Reversal of Forest Soil Acidification in the Northeastern United States and Eastern Canada: Site and Soil Factors Contributing to Recovery
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Sites, Experimental Design and Field Sampling
2.2. Laboratory Analysis
2.3. Calculations and Statistical Analysis
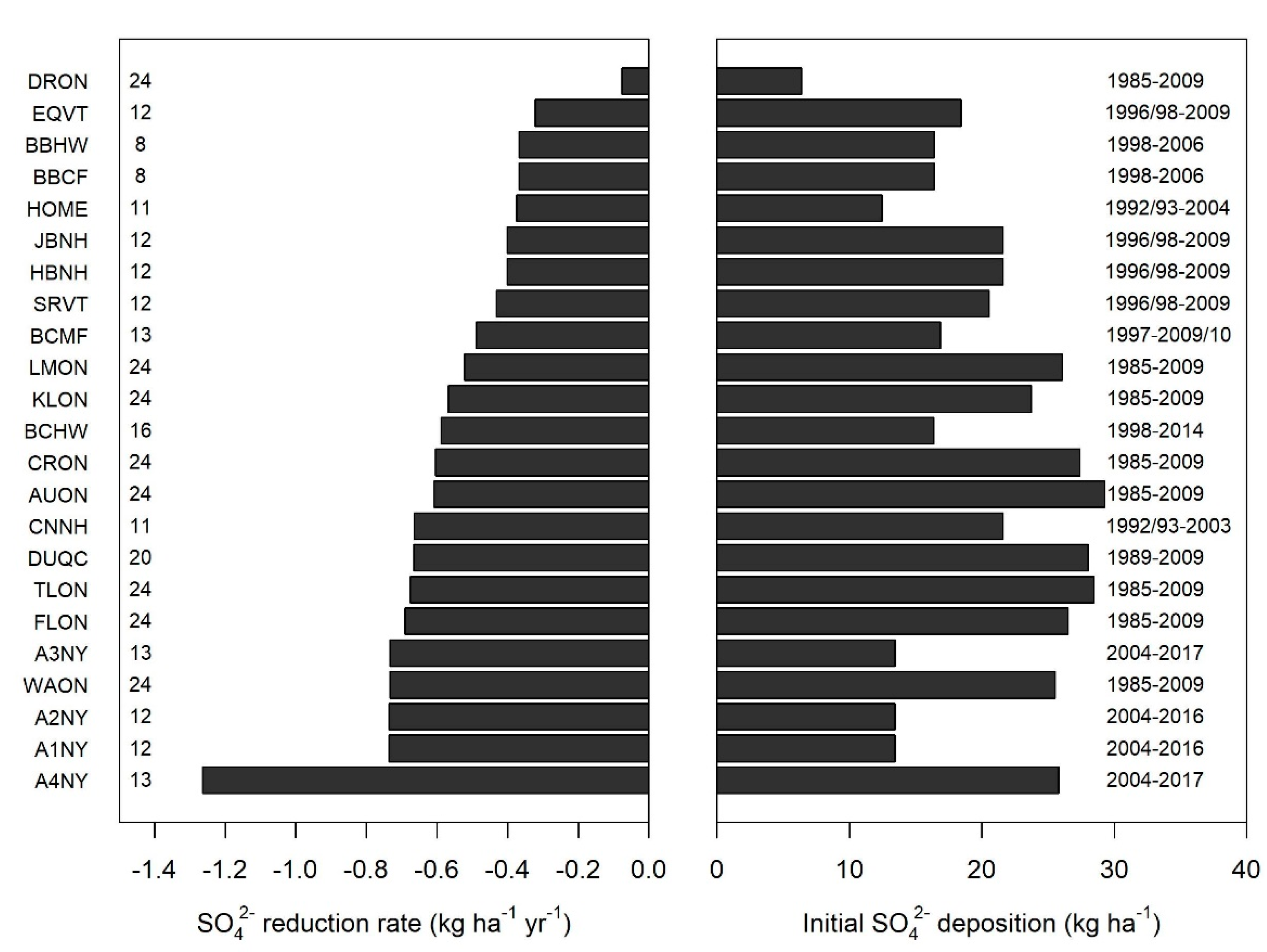
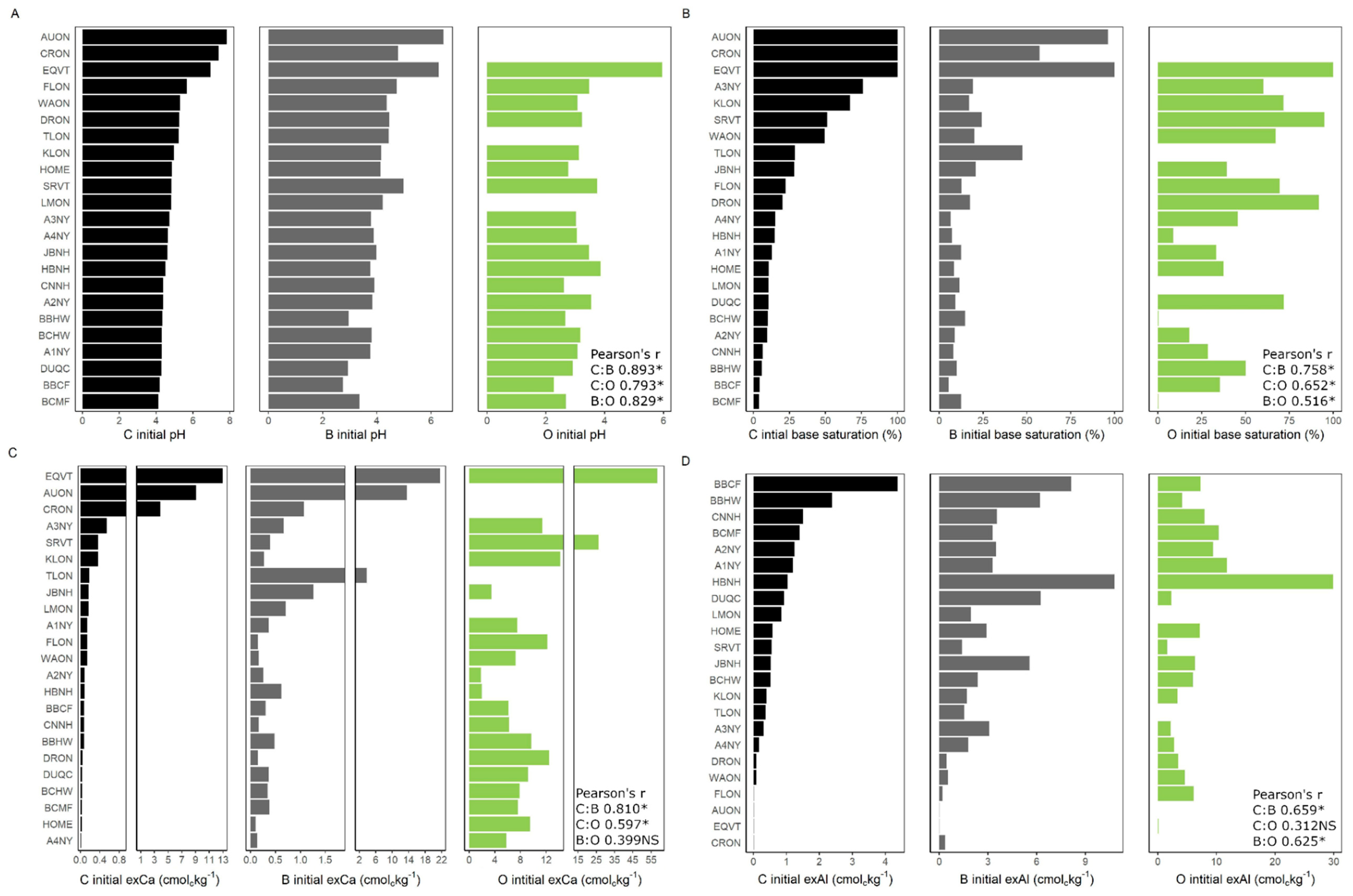
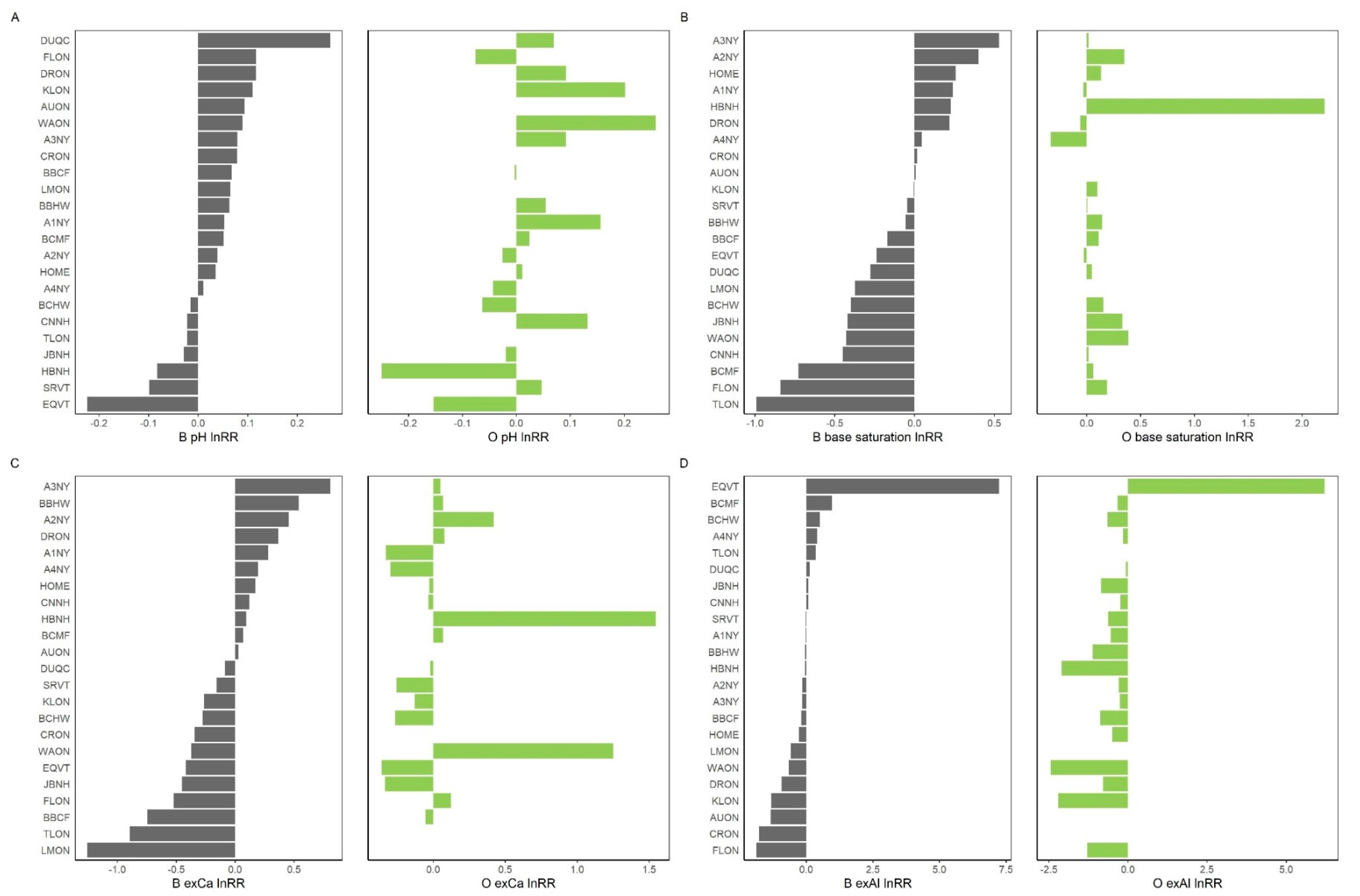
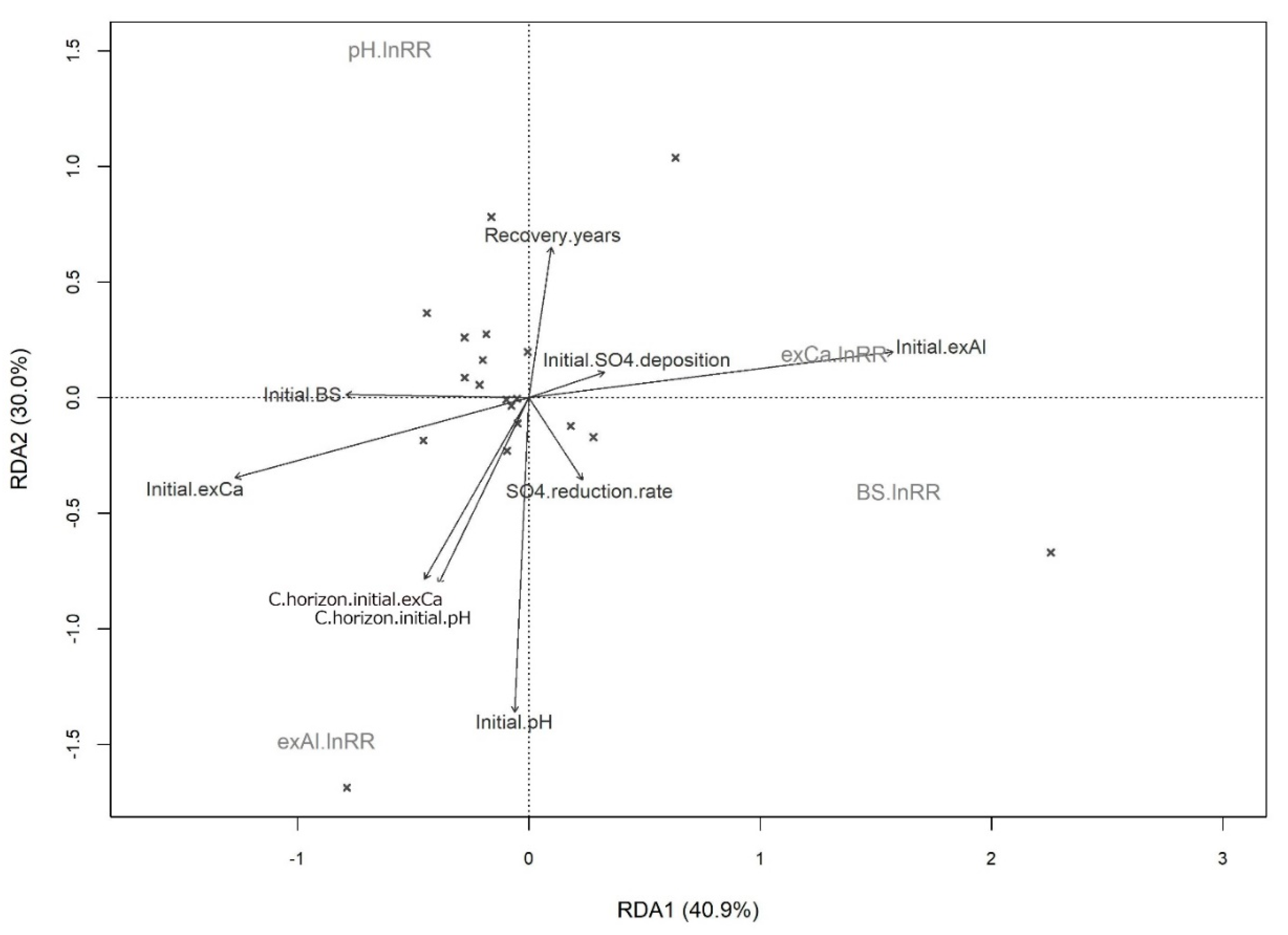
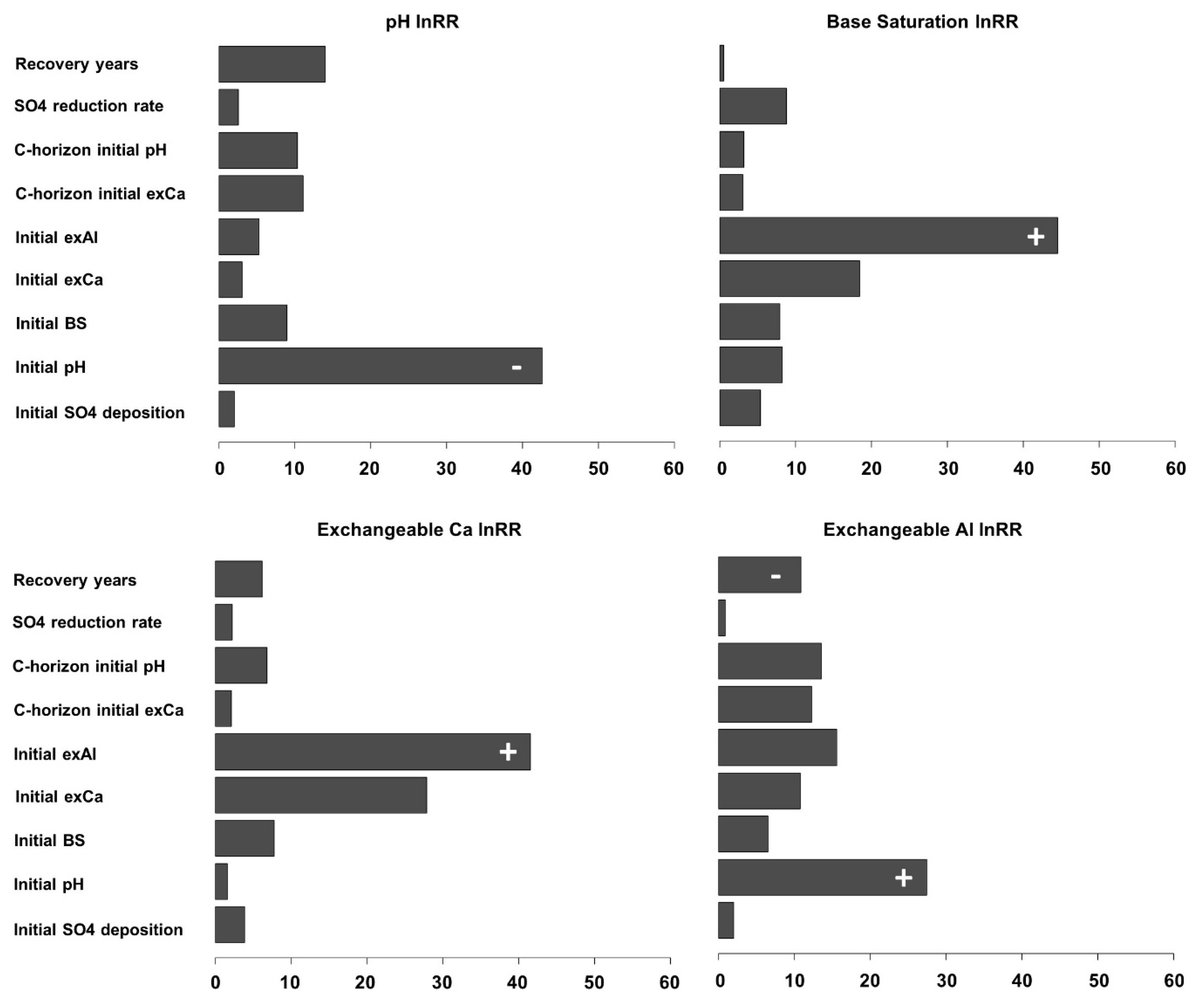
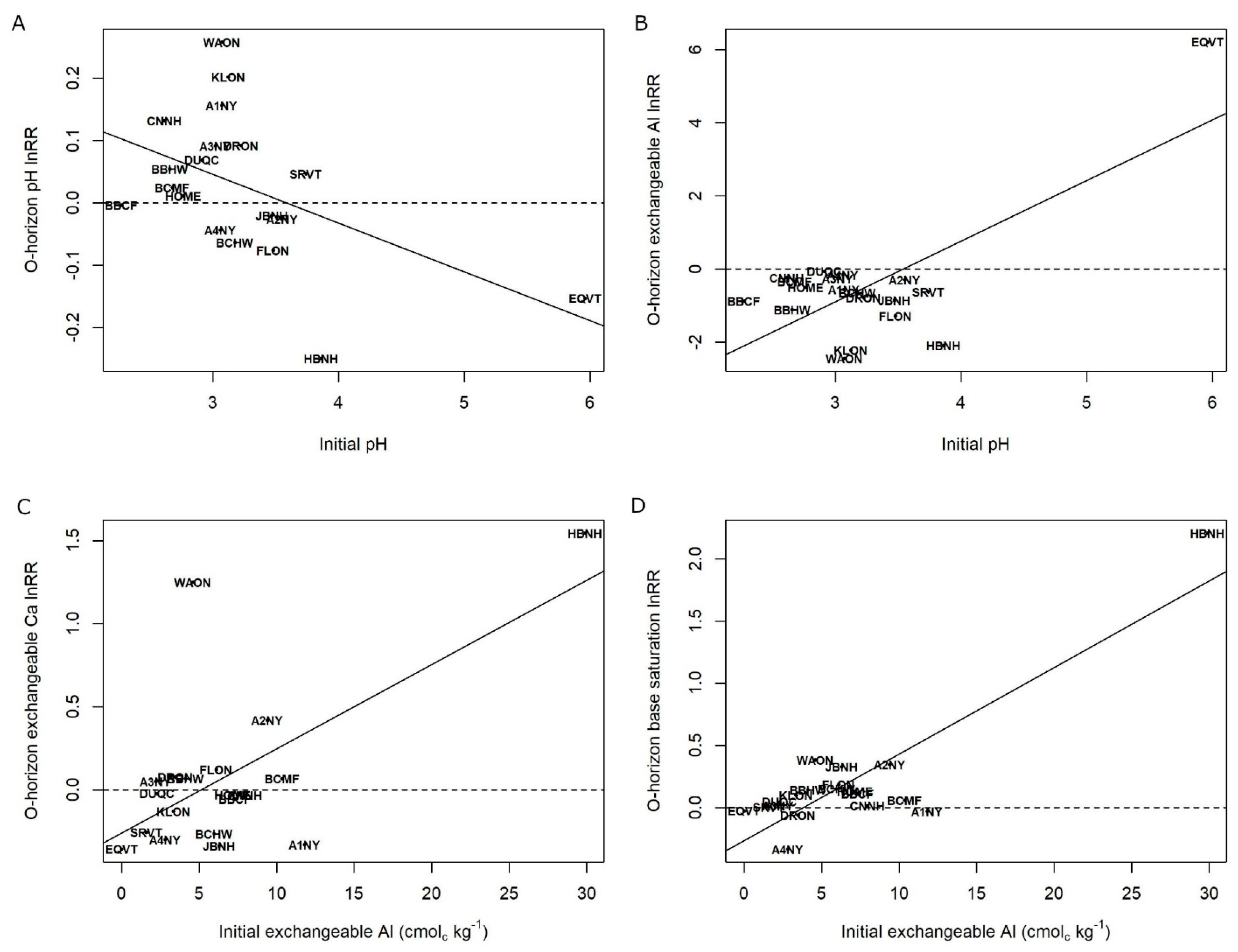
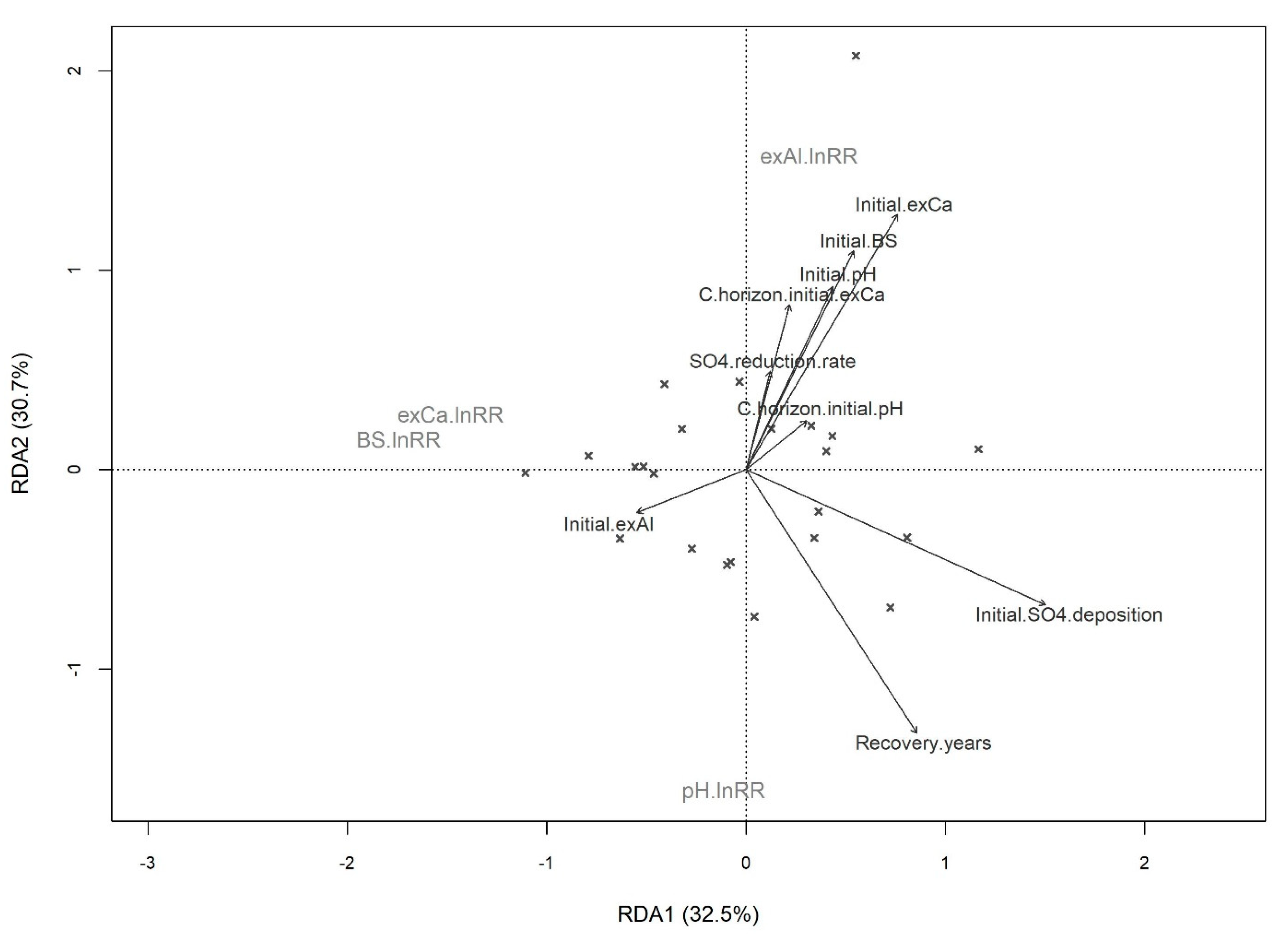
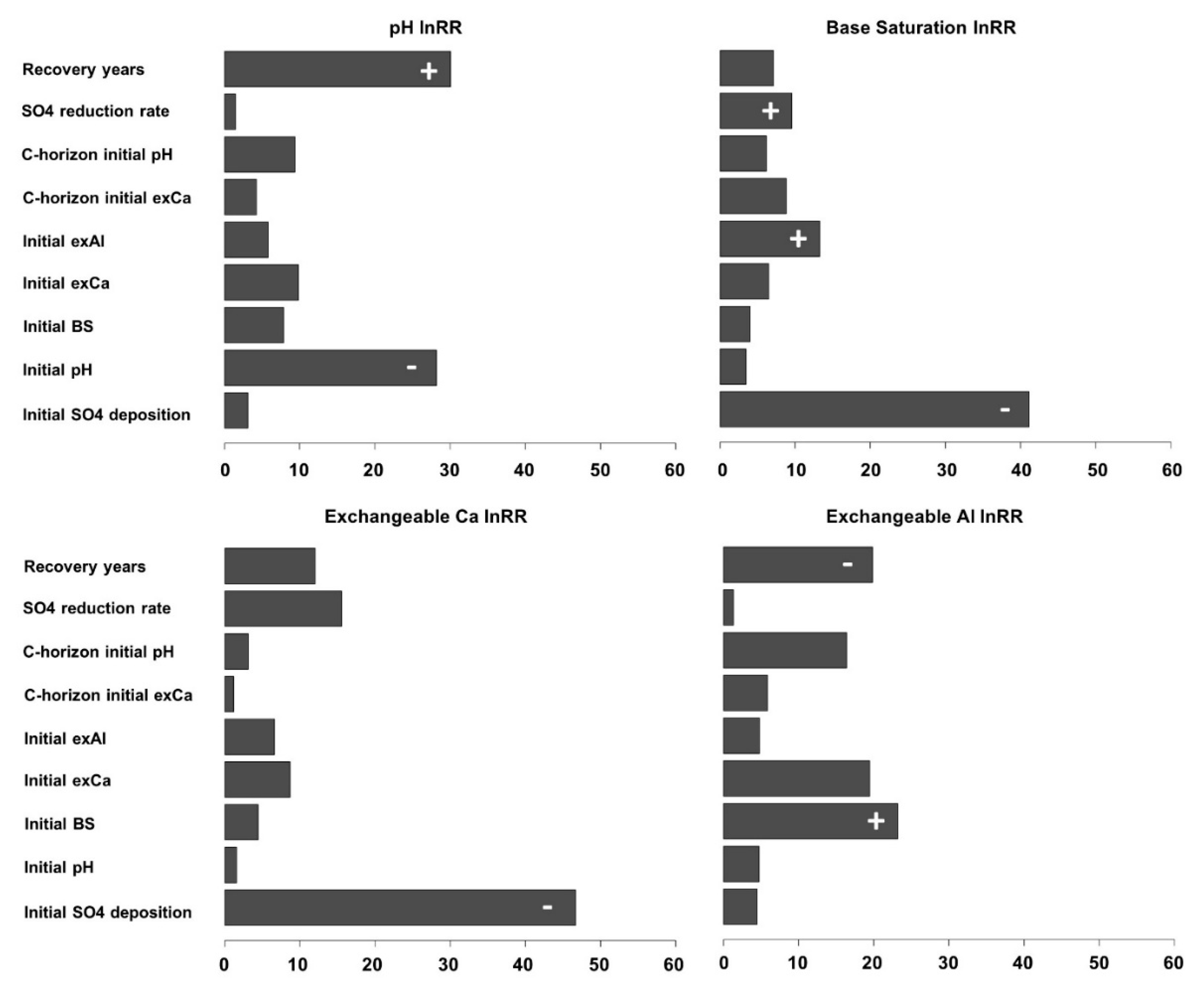
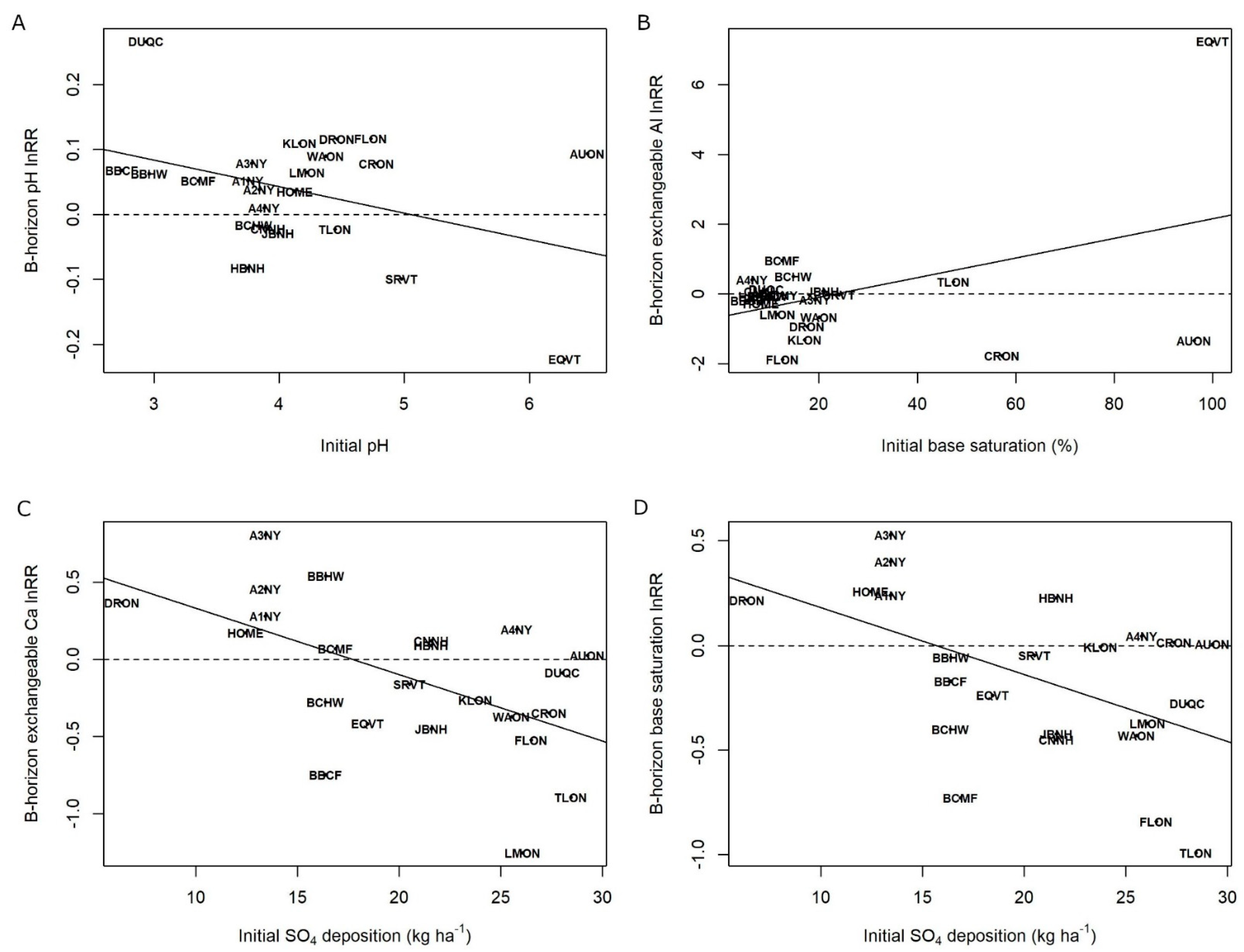
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Engstrom, A.; Backstrand, G.; Stenram, H. Air Pollution across National Boundaries: The Impact on the Environment of Sulfur in Air and Precipitation, Report Editor; Royal Ministry for Foreign Affairs: Stockholm, Sweden, 1971.
- Odén, S. The acidity problem-an outline of concepts. Water Air Soil Pollut. 1976, 6, 137–166. [Google Scholar] [CrossRef]
- Abrahamsen, G. Effects of acidic deposition on forest soil and vegetation. Phil. Trans. R. Soc. Lond. 1984, 305, 369–392. [Google Scholar]
- Johnson, D.W.; Turner, J.; Kelly, J.M. The effects of acid rain on forest nutrient status. Water Resour. Res. 1982, 18, 449–461. [Google Scholar] [CrossRef]
- Harrison, R.B.; Johnson, D.W.; Todd, D.E. Sulfate adsorption and desorption reversibility in a variety of forest soils. J. Environ. Qual. 1989, 18, 419–426. [Google Scholar] [CrossRef]
- Berden, M.; Nilsson, S.I.; Rosen, K.; Tyler, G. Soil Acidification Extent, Causes and Consequences. An Evaluation of Literature Information and Current Research [Acid Neutralizing Capacity, Proton Sources, Proton Budgets, Biological and Chemical Consequences, Incl. Natural Soil Acidification Processes; Rapport-Naturvaardsverket: Solna, Sweden, 1987. [Google Scholar]
- Ulrich, B.; Mayer, R.; Khanna, P.K. Chemical changes due to acid precipitation in a loess-derived soil in central Europe. Soil Sci. 1980, 130, 193–199. [Google Scholar] [CrossRef]
- Grimm, R.; Rehfuess, K.E. Kurzfristige Veranderungen von Bodenreaktien und Kationenaustauscheigenschaften in einen Meliorationsversuch zu Kiefer (Pinus sylv. L.) auf Podsol-Pseudogley in der Oberpfalz. Allg. Forst Jagdztg. 1986, 157, 205–213. [Google Scholar]
- Linzon, S.N.; Temple, P.J. Soil Resampling and PH Measurements after an 18-Year Period in Ontario. Proc. Int. Conf. on Ecological Impact of Acid Precipitation; SNSF Project: Sandefjord, Norway, 1980; pp. 176–177. [Google Scholar]
- Johnson, A.H.; Andersen, S.B.; Siccama, T.G. Acid rain and soils of the Adirondacks. 1. Changes in pH and available calcium, 1930–1984. Can. J. Res. 1994, 24, 39–45. [Google Scholar] [CrossRef]
- Johnson, A.H.; Moyer, A.J.; Bedison, J.E.; Richter, S.L.; Willig, S.A. Seven decades of calcium depletion in organic horizons of Adirondack forest soils. Soil Sci. Soc. Am. J. 2008, 72, 1824–1830. [Google Scholar] [CrossRef]
- Bedison, J.E.; Johnson, A.H. Seventy-four years of calcium loss from forest soils of the Adirondack Mountains, New York. Soil Sci. Soc. Am. J. 2010, 74, 2187–2195. [Google Scholar] [CrossRef]
- Bailey, S.W.; Horsley, S.B.; Long, R.P. Thirty years of change in forest soils of the Allegheny Plateau, Pennsylvania. Soil Sci. Soc. Am. J. 2005, 69, 681–690. [Google Scholar] [CrossRef]
- Johnson, D.W.; Henderson, G.S.; Todd, D.E. Changes in nutrient distribution in forests and soils of the Walker Branch Watershed, Tennessee, over an eleven-year period. Biogeochemistry 1988, 5, 275–293. [Google Scholar] [CrossRef]
- Trettin, C.F.; Johnson, D.W.; Todd, D.E. Forest nutrient and carbon pools at the Walker Branch Watershed: Changes during a 21-year period. Soil Sci. Soc. Am. J. 1999, 63, 1436–1448. [Google Scholar] [CrossRef]
- Johnson, D.W.; Todd, D.E.; Trettin, C.F.; Mulholland, P.J. Decadal changes in potassium, calcium, and magnesium in a deciduous forest soil. Soil Sci. Soc. Am. J. 2008, 72, 1795–1805. [Google Scholar] [CrossRef]
- Watmough, S.A.; Dillon, P.J. Major element fluxes from a coniferous catchment in central Ontario, 1983–1999. Biogeochemistry 2004, 67, 369–398. [Google Scholar] [CrossRef]
- Warby, R.A.F.; Johnson, C.E.; Driscoll, C.T. Continuing acidification of organic soils across the northeastern USA: 1984–2001. Soil Sci. Soc. Am. J. 2009, 73, 274–284. [Google Scholar] [CrossRef]
- Miller, D.E.; Watmough, S.A. Soil acidification and foliar nutrient status of Ontario’s deciduous forest in 1986 and 2005. Environ. Pollut. 2009, 157, 664–672. [Google Scholar] [CrossRef] [PubMed]
- Sanderson, T.M.; Barton, C.; Cotton, C.; Karathanasis, T. Long-term evaluation of acidic atmospheric deposition on soils and soil solution chemistry in the Daniel Boone National Forest, USA. Water Air Soil Pollut. 2017, 228, 403. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency (USEPA). Clean Air Act (CAA) Amendments; USEPA: Washington, DC, USA, 1990; pp. 7401–7671.
- Government of Canada. Canadian Environmental Protection Act (CEPA); Minister of Justice Canada: Ottawa, ON, Canada, 1999. [Google Scholar]
- Vet, R.; Artz, R.S.; Carou, S.; Shaw, M.; Ro, C.U.; Aas, W.; Baker, A.; Bowersox, V.C.; Dentener, F.; Galy-Lacaux, C.; et al. A global assessment of precipitation chemistry and deposition of sulfur, nitrogen, sea salt, base cations, organic acids, acidity and pH, and phosphorus. Atmos. Environ. 2014, 93, 3–100. [Google Scholar] [CrossRef]
- International Joint Commission (IJC); Canada-US Air Quality Committee. Canada, United States Air Quality Agreement: Progress Report 2016; Minister of Environment and Climate Change Canada: Ottawa, ON, Canada, 2017. [Google Scholar]
- Lawrence, G.B.; Shortle, W.C.; David, M.B.; Smith, K.T.; Warby, R.A.F.; Lapenis, A.G. Early indications of soil recovery from acidic deposition in US red spruce forests. Soil Sci. Soc. Am. J. 2012, 76, 1407–1417. [Google Scholar] [CrossRef]
- Lawrence, G.B.; Hazlett, P.W.; Fernandez, I.J.; Ouimet, R.; Bailey, S.W.; Shortle, W.C.; Smith, W.T.; Antidormi, M.R. Declining acidic deposition begins reversal of forest-soil acidification in the northeastern U.S. and eastern Canada. Environ. Sci. Technol. 2015, 49, 13103–13111. [Google Scholar] [CrossRef]
- Berger, T.W.; Türtscher, S.; Berger, P.; Lindebner, L. A slight recovery of soils from Acid Rain over the last three decades is not reflected in the macro nutrition of beech (Fagus sylvatica) at 97 forest stands of the Vienna Woods. Environ. Pollut. 2016, 216, 624–635. [Google Scholar] [CrossRef] [PubMed]
- Siemion, J.; McHale, M.R.; Lawrence, G.B.; Burns, D.A.; Antidormi, M. Long-term changes in soil and stream chemistry across an acid deposition gradient in the northeastern United States. J. Environ. Qual. 2018, 47, 410–418. [Google Scholar] [CrossRef] [PubMed]
- Fraser, O.L.; Bailey, S.W.; Ducey, M.J. Decadal change in soil chemistry of northern hardwood forests on the White Mountain National Forest, New Hampshire, USA. Soil Sci. Soc. Am. J. 2019, 83, S96–S104. [Google Scholar] [CrossRef]
- Norton, S.A. Geologic factors controlling the sensitivity of aquatic ecosystems to acidic precipitation. In Atmospheric Sulfur Deposition: Environmental Input and Health Effects; Shriner, D.S., Richmond, C.R., Lindberg, S.E., Eds.; Ann Arbor Science Pub. Inc.: Ann Arbor, MI, USA, 1979; pp. 521–531. [Google Scholar]
- Hendry, R.; Galloway, J.N.; Norton, S.A.; Schofield, C.L.; Burns, D.A.; Schaffer, P.W. Sensitivity of the eastern United States to acid precipitation impacts on surface waters. In Ecological Impact of Acid Precipitation, Proceedings of the International Conference, Sandefjord, Norway, 11–14 March 1980; Drablos, D., Tollan, A., Eds.; US Department of Energy and Environment, Brookhaven National Laboratory: Upton, NY, USA, 1980; pp. 216–217. [Google Scholar]
- Johnson, D.W.; Hornbeck, J.W.; Kelly, J.M.; Swank, W.T.; Todd, D.E. Regional patterns of soil sulfate accumulation: Relevance to ecosystem sulfur budgets. In Atmospheric Sulfur Deposition: Environmental Impact and Health Effects; Shriner, D.S., Richmond, C.R., Lindberg, S.E., Eds.; Ann Arbor Science: Ann Arbor, MI, USA, 1980; pp. 507–520. [Google Scholar]
- Lawrence, G.B.; Antidormi, M.R.; McDonnell, T.C.; Sullivan, T.J.; Bailey, S.W. Adirondack New York soil chemistry data, 1992–2017: USA. Geol. Surv. Data Release 2020. [Google Scholar] [CrossRef]
- Lawrence, G.B.; Fernandez, I.J.; Hazlett, P.W.; Bailey, S.W.; Ross, D.S.; Villars, T.R.; Quintana, A.; Ouimet, R.; McHale, M.R.; Johnson, C.E.; et al. Methods of soil resampling to monitor changes in the chemical concentrations of forest soils. J. Vis. Exp. 2016. [Google Scholar] [CrossRef]
- McKenney, D.W.; Hutchinson, M.F.; Papadopol, P.; Lawrence, K.; Pedlar, J.; Campbell, K.; Milewska, E.; Hopkinson, R.F.; Price, D.; Owen, T. Customized spatial climate models for North America. Bull. Am. Meteorol. Soc. 2011, 92, 1611–1622. [Google Scholar] [CrossRef]
- CAPMoN Canadian Air and Precipitation Monitoring Network. Available online: https://www.canada.ca/en/environment-climate-change/services/air-pollution/monitoring-networks-data/canadian-air-precipitation.html (accessed on 1 June 2020).
- NADP National Atmospheric Deposition Program. Available online: http://nadp.slh.wisc.edu/ (accessed on 1 June 2020).
- Ross, D.; Bailey, S.; Briggs, R.; Curry, J.; Fernandez, I.; Fredriksen, G.; Goodale, C.; Hazlett, P.; Heine, P.; Johnson, C.; et al. Inter-laboratory variation in the chemical analysis of acidic forest soil reference samples from Eastern North America. Ecosphere 2015, 6, 73. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020; Available online: https://www.R-project.org (accessed on 28 July 2020).
- Hedges, L.V.; Gurevitch, J.; Curtis, P.S. The meta-analysis of response ratios in experimental ecology. Ecology 1999, 80, 1150–1156. [Google Scholar] [CrossRef]
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, R.B.; Simpson, J.L.; Solymos, P.; et al. Vegan: Community Ecology Package, R package version 2.5–6. 2019. Available online: https://CRAN.R-project.org/package=vegan (accessed on 28 July 2020).
- Bertini, S.C.B.; Azevedo, L.C.B.; de Mendes, I.C.; Cardoso, E.J.B.N. Hierarchical partitioning for selection of microbial and chemical indicators of soil quality. Pedobiologia 2014, 57, 293–301. [Google Scholar] [CrossRef]
- MacNally, R.; Walsh, C.J. Hierarchical partitioning public-domain software. Biodivers. Conserv. 2004, 13, 659–660. Available online: https://search.proquest.com/openview/6f19c4d612b6cf75b106c479e7084a58/1 (accessed on 28 July 2020).
- Chevan, A.; Sutherland, M. Hierarchical partitioning. Am. Stat. 1991, 45, 90–96. [Google Scholar]
- Fox, J.; Weisberg, S. An {R} Companion to Applied Regression, 3rd ed.; Sage: Thousand Oaks, CA, USA, 2019; Available online: https://socialsciences.mcmaster.ca/jfox/Books/Companion/ (accessed on 28 July 2020).
- Olea, P.P.; Mateo-Tomás, P.; de Frutos, Á. Estimating and modelling bias of the hierarchical partitioning public-domain software: Implications in environmental management and conservation. PLoS ONE 2010, 5, e11698. [Google Scholar] [CrossRef] [PubMed]
- Jandl, R.; Smidt, S.; Mutsch, F.; Fürst, A.; Zechmeister, H.; Bauer, H.; Dirnböck, T. Acidification and nitrogen eutrophication of Austrian forest soils. Appl. Environ. Soil Sci. 2012, 2012. [Google Scholar] [CrossRef]
- Kirk, G.J.D.; Bellamy, P.H.; Lark, R.M. Changes in soil pH across England and Wales in response to decreased acid deposition. Glob. Chang. Biol. 2010, 16, 3111–3119. [Google Scholar] [CrossRef]
- Cincotta, M.M.; Perdrial, J.M.; ALibenson, A.S.; Landsman-Gerjoi, M.; Perdrial, N.; Armfield, J.; Adle, T.; Shanley, J. Soil aggregates as a source of dissolved organic carbon to streams: An experimental study on the effect of solution chemistry on water extractable carbon. Front. Environ. Sci. 2019, 7, 172. [Google Scholar] [CrossRef]
- Reuss, J.O. Implications of the calcium-aluminum exchange system for the effect of acid precipitation on soils. J. Environ. Qual. 1983, 12, 591–595. [Google Scholar] [CrossRef]
- McHale, M.R.; Burns, D.A.; Siemion, J.; Antidormi, M.R. The response of soil and stream chemistry to decreases in acid deposition in the Catskill Mountains, New York, USA. Environ. Pollut. 2017, 229, 607–620. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, G.B.; Scanga, S.E.; Sabo, R.D. Recovery of soils from acidic deposition may exacerbate nitrogen export from forested watersheds. J. Geophys. Res. Biogeosci. 2020, 125, e2019JG005036. [Google Scholar] [CrossRef]
- Hazlett, P.W.; Curry, J.M.; Weldon, T.P. Assessing decadal change in mineral soil cation chemistry at the Turkey Lakes Watershed. Soil Sci. Soc. Am. J. 2011, 75, 287–305. [Google Scholar] [CrossRef]
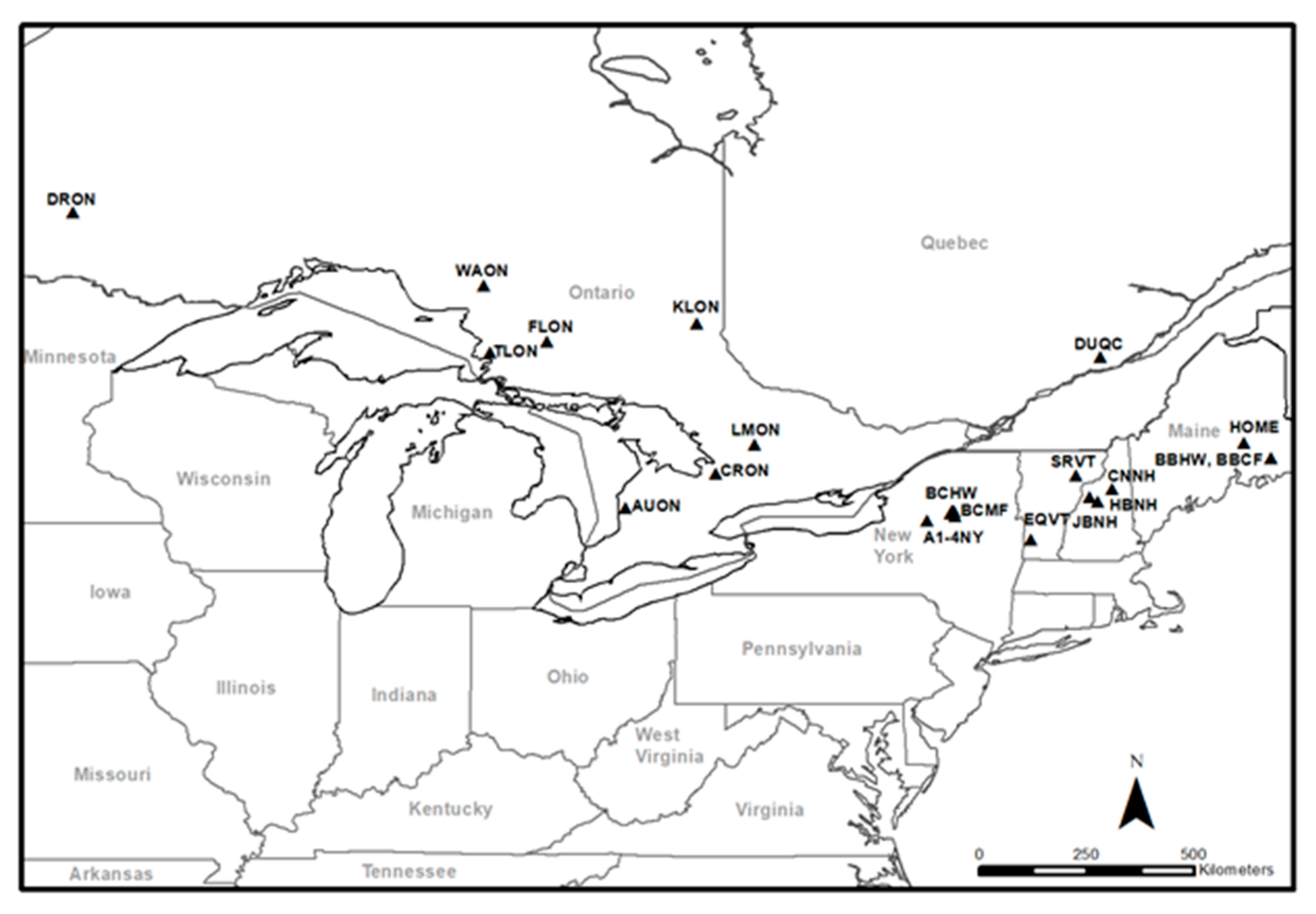

| State/Province, Location | Site Code | Forest Type | Soil Type (US Great Group) | Soil Type (Cdn Great Group) | Pits Sampled | Soil Texture a B Horizon | Annual Precipitation b (mm) | Annual Mean Temperature b (°C) |
|---|---|---|---|---|---|---|---|---|
| MaineBear Brook | BBCF | spruce | Haplorthod | Humo-ferric podzol | 10 | SL | 1256 | 6.9 |
| MaineBear Brook | BBHW | northern hardwood | Haplorthod | Humo-ferric podzol | 10 | SL | 1256 | 6.9 |
| MaineHowland | HOME | mixed softwood | Haplorthod | Humo-ferric podzol | 12 | SL | 1180 | 6.4 |
| New Hampshire, Crawford Notch | CNNH | spruce | Haplorthod | Humo-ferric podzol | 12 | SL | 1703 | 1.8 |
| QuebecDuchesnay | DUQC | northern hardwood | Haplorthod | Humo-ferric podzol | 4 | SL | 1341 | 3.8 |
| New HampshireHubbard Brook | HBNH | northern hardwood | Haplorthod | Humo-ferric podzol | 4 | SL | 1281 | 5.6 |
| New HampshireJeffers Brook | JBNH | northern hardwood | Haplorthod | Humo-ferric podzol | 4 | SL | 1434 | 4.0 |
| VermontSleepers River | SRVT | northern hardwood | Haplorthod | Humo-ferric podzol | 4 | SL | 1295 | 4.5 |
| Vermont Mount Equinox | EQVT | northern hardwood | Dystrudept | Dystric brunisol | 4 | SL | 1383 | 5.9 |
| New York Adirondack 28014 | A2NY | northern hardwood | Haplorthod | Humo-ferric podzol | 15 | SL | 1342 | 4.4 |
| New YorkBuck Creek | BCMF | spruce, northern hardwood | Haplorthod | Humo-ferric podzol | 28 | SL | 1258 | 4.8 |
| New YorkBuck Creek | BCHW | northern hardwood | Haplorthod | Humo-ferric podzol | 28 | SL | 1258 | 4.8 |
| New YorkAdirondack 28030 | A3NY | northern hardwood | Haplorthod | Humo-ferric podzol | 15 | SL | 1196 | 5.2 |
| New YorkAdirondack 27020 | A1NY | northern hardwood | Haplorthod | Humo-ferric podzol | 15 | SL | 1305 | 4.6 |
| New YorkAdirondack 29012 | A4NY | softwood, hardwood | Haplorthod | Humo-ferric podzol | 15 | SL | 1259 | 6.4 |
| OntarioLittle Margaret Lake | LMON | northern hardwood | Hapludalf | Gray brown luvisol | 25 | SiL | 1080 | 5.3 |
| Ontario Craighurst | CRON | northern hardwood | Eutrustept | Melanic brunisol | 25 | SL | 968 | 6.6 |
| OntarioKirkland Lake | KLON | boreal jack pine | Haplorthod | Humo-ferric podzol | 25 | SiS | 743 | 3.1 |
| OntarioAuburn | AUON | northern hardwood | Hapludalf | Gray brown luvisol | 25 | SiC | 991 | 7.8 |
| OntarioFlame Lake | FLON | boreal jack pine | Haplorthod | Humo-ferric podzol | 25 | SCL | 896 | 3.1 |
| OntarioTurkey Lakes | TLON | northern hardwood | Haplohumod | Ferro-humic podzol | 25 | SiL | 1069 | 3.3 |
| OntarioWawa | WAON | boreal jack pine | Haplorthod | Humo-ferric podzol | 25 | LS | 838 | 2.0 |
| OntarioDryden | DRON | boreal jack pine | Haplorthod | Humo-ferric podzol | 25 | SiS | 706 | 2.6 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hazlett, P.; Emilson, C.; Lawrence, G.; Fernandez, I.; Ouimet, R.; Bailey, S. Reversal of Forest Soil Acidification in the Northeastern United States and Eastern Canada: Site and Soil Factors Contributing to Recovery. Soil Syst. 2020, 4, 54. https://doi.org/10.3390/soilsystems4030054
Hazlett P, Emilson C, Lawrence G, Fernandez I, Ouimet R, Bailey S. Reversal of Forest Soil Acidification in the Northeastern United States and Eastern Canada: Site and Soil Factors Contributing to Recovery. Soil Systems. 2020; 4(3):54. https://doi.org/10.3390/soilsystems4030054
Chicago/Turabian StyleHazlett, Paul, Caroline Emilson, Greg Lawrence, Ivan Fernandez, Rock Ouimet, and Scott Bailey. 2020. "Reversal of Forest Soil Acidification in the Northeastern United States and Eastern Canada: Site and Soil Factors Contributing to Recovery" Soil Systems 4, no. 3: 54. https://doi.org/10.3390/soilsystems4030054
APA StyleHazlett, P., Emilson, C., Lawrence, G., Fernandez, I., Ouimet, R., & Bailey, S. (2020). Reversal of Forest Soil Acidification in the Northeastern United States and Eastern Canada: Site and Soil Factors Contributing to Recovery. Soil Systems, 4(3), 54. https://doi.org/10.3390/soilsystems4030054





