Microbe Biomass in Relation to Organic Carbon and Clay in Soil
Abstract
1. Introduction
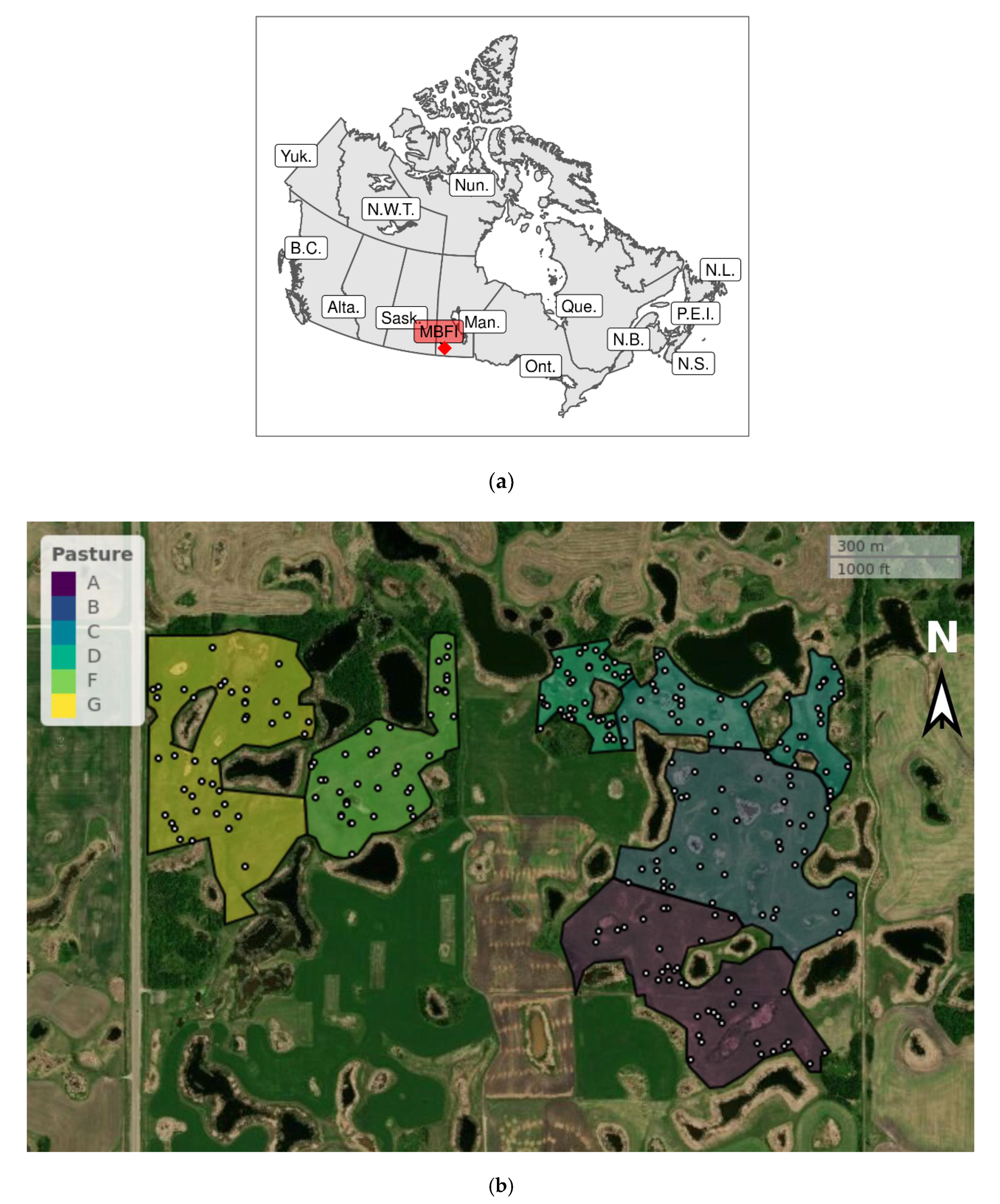
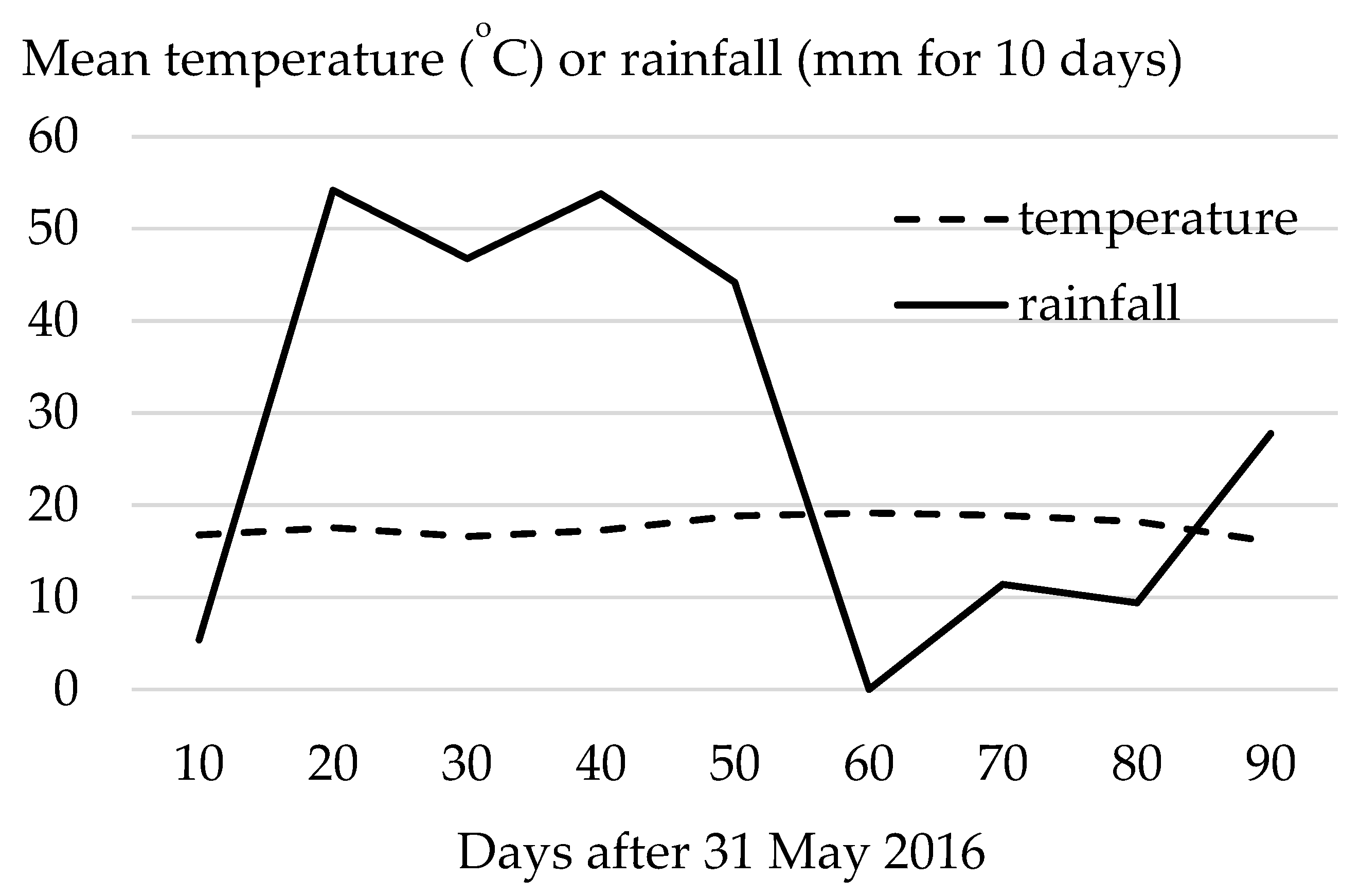
2. Materials and Methods
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bach, E.M.; Ramirez, K.S.; Fraser, T.D.; Wall, D.H. Soil biodiversity integrates solutions for a sustainable future. Sustainability 2020, 12, 2662. [Google Scholar] [CrossRef]
- Jenkinson, D.S.; Brookes, P.C.; Powlson, D.S. Measuring soil microbial biomass. Soil Biol. Biochem. 2004, 36, 5–7. [Google Scholar] [CrossRef]
- Hassink, J. Density fractions of soil macroorganic matter and microbial biomass as predictors of C and N mineralization. Soil Biol. Biochem. 1995, 27, 1099–1108. [Google Scholar] [CrossRef]
- Murphy, D.V.; Sparling, G.P.; Fillery, I.R.P. Stratification of microbial biomass C and N and gross N mineralization with soil depth in two contrasting Western Australian agricultural soils. Aust. J. Soil Res. 1998, 36, 45–55. [Google Scholar] [CrossRef]
- McGonigle, T.P.; Turner, W.G. Grasslands and croplands have different microbial biomass carbon levels per unit soil organic carbon. Agriculture 2017, 7, 57. [Google Scholar] [CrossRef]
- Gupta, V.V.S.R.; Germida, J.J. Soil aggregation: Influence on microbial biomass and implications for biological processes. Soil Biol. Biochem. 2015, 80, A3–A9. [Google Scholar] [CrossRef]
- Totsche, K.U.; Amelung, W.; Gerzabek, M.H.; Guggenberger, G.; Klumpp, E.; Knief, C.; Lehndorff, E.; Mikutta, R.; Peth, S.; Prechtel, A.; et al. Microaggregates in soils. J. Plant Nutr. Soil Sci. 2018, 181, 104–136. [Google Scholar] [CrossRef]
- Diochon, A.C.; Kellman, L. Physical fractionation of soil organic matter: Destabilization of deep soil carbon following harvesting of a temperate coniferous forest. J. Geophys. Res. Biogeosci. 2009, 114, 1–9. [Google Scholar] [CrossRef]
- Alvarez, C.R.; Alvarez, R. Are active organic matter fractions suitable indices of management effects on soil carbon? A meta-analysis from the Pampas. Arch. Agron. Soil Sci. 2016, 62, 1592–1601. [Google Scholar] [CrossRef]
- Carter, M.R. The influence of tillage on the proportion of organic carbon and nitrogen in the microbial biomass of medium-textured soils in a humid climate. Biol. Fert. Soils 1991, 11, 135–139. [Google Scholar] [CrossRef]
- Moore, J.M.; Klose, S.; Tabatabai, M.A. Soil microbial biomass carbon and nitrogen as affected by cropping systems. Biol. Fert. Soils 2000, 31, 200–210. [Google Scholar] [CrossRef]
- Sawada, K.; Funakawa, S.; Kosaki, T. Different effects of pH on microbial biomass carbon and metabolic quotients by fumigation–Extraction and substrate-induced respiration methods in soils under different climatic conditions. Soil Sci. Plant. Nutr. 2009, 55, 363–374. [Google Scholar] [CrossRef]
- Franzluebbers, A.J.; Haney, R.L.; Hons, F.M.; Zuberer, D.A. Active fractions of organic matter in soils with different texture. Soil Biol. Biochem. 1996, 28, 1367–1372. [Google Scholar] [CrossRef]
- Seita, R.; Verma, S.L.; Marschner, P. Measuring microbial biomass carbon by direct extraction—Comparison with chloroform fumigation-extraction. Eur. J. Soil Biol. 2012, 53, 103–106. [Google Scholar]
- Hassink, J. Effects of soil texture and structure on carbon and nitrogen mineralization in grassland soils. Biol. Fert. Soil 1992, 14, 126–134. [Google Scholar] [CrossRef]
- Sparling, G.P. Ratio of microbial biomass carbon to soil organic carbon as a sensitive indicator of changes in soil organic matter. Aust. J. Soil Res. 1992, 30, 195–207. [Google Scholar] [CrossRef]
- Environment Canada. Canadian Climate Normals. Available online: https://climate.weather.gc.ca/climate_normals (accessed on 28 May 2020).
- Sloan, C.E. Ground-Water Hydrology of Prairie Potholes in North Dakota; U.S. Geological Survey: Washington, DC, USA, 1972; pp. 1–28. [CrossRef]
- Vance, E.D.; Brookes, P.C.; Jenkinson, D.S. An extraction method for measuring soil microbial biomass carbon. Soil Biol. Biochem. 1987, 19, 703–707. [Google Scholar] [CrossRef]
- Nelson, D.W.; Sommers, L.E. Total carbon, organic carbon, and organic matter. In Methods of Soil Analysis. Part 3. Chemical Methods; Sparks, D.L., Ed.; Soil Science Society of America: Madison, WI, USA, 1996; pp. 961–1010. [Google Scholar]
- Gee, G.W.; Or, D. Particle-size analysis. In Methods of Soil Analysis. Part 4. Physical Methods; Dane, G.H., Topp, G.C., Eds.; Soil Science Society of America: Madison, WI, USA, 2002; pp. 255–293. [Google Scholar]
- Maynard, D.G.; Kalra, Y.P. Nitrate and exchangeable ammonium nitrogen. In Soil Sampling and Methods of Analysis; Carter, M.R., Ed.; Lewis: Boca Raton, FL, USA, 1993; pp. 25–38. [Google Scholar]
- Schoenau, J.J.; Karamanos, R.E. Sodium bicarbonate-extractable P, K, and N. In Soil Sampling and Methods of Analysis; Carter, M.R., Ed.; Lewis: Boca Raton, FL, USA, 1993; pp. 51–58. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing. Available online: http://www.R-project.org (accessed on 28 May 2020).
- RStudio. RStudio: Integrated Development Environment for R. Available online: http://www.rstudio.org/ (accessed on 28 May 2020).
- Bates, D.; Maechler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Software 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Kuznetsova, A.; Brockhoff, P.B.; Christensen, R.H.B. lmerTest package: Tests in linear mixed effects models. J. Stat. Softw. 2017, 82, 1–26. [Google Scholar] [CrossRef]
- Quinn, G.P.; Keough, M.J. Experimental Design and Data Analysis for Biologists; Cambridge University Press: Cambridge, UK, 2002. [Google Scholar]
- Nakagawa, S.; Johnson, P.C.D.; Schielzeth, H. The coefficient of determination R² and intra-class correlation coefficient from generalized linear mixed-effects models revisited and expanded. J. R. Soc. Interface 2017, 14, 20170213. [Google Scholar] [CrossRef] [PubMed]
- Bartoń, K. MuMIn: Multi-Model Inference. R Package Version 1.43.17. Available online: https://CRAN.R-project.org/package=MuMIn (accessed on 28 May 2020).
- Roberts, T.L.; Stewart, J.W.B.; Bettany, J.R. The influence of topography on the distribution of organic and inorganic soil phosphorus across a narrow environmental gradient. Can. J. Soil Sci. 1985, 65, 651–665. [Google Scholar] [CrossRef]
- Fissore, C.; Dazell, B.J.; Berhe, A.A.; Voegtle, M.; Evans, M.; Wu, A. Influence of topography on soil organic carbon dynamics in a Southern California grassland. Catena 2017, 149, 140–149. [Google Scholar] [CrossRef]
- Garcia-Tomillo, A.; Miras-Avalos, J.M.; Dafonte-Dafonte, J.; Paz-Gonzalez, A. Mapping soil texture using geostatistical interpolation combined with electromagnetic induction measurements. Soil Sci. 2017, 182, 278–284. [Google Scholar] [CrossRef]
- Teller, J.T. The ice age and its legacy. In Natural Heritage of Manitoba: Legacy of the Ice Age; Teller, J.T., Ed.; Manitoba Museum of Man and Nature: Winnipeg, MB, Canada, 1984; pp. 23–47. [Google Scholar]
- Li, X.; McCarty, G.W.; Du, L.; Lee, S. Use of topographic models for mapping soil properties and processes. Soil Syst. 2020, 4, 32. [Google Scholar] [CrossRef]
- Garcia-Tomillo, A.; Miras-Avalos, J.M.; Dafonte-Dafonte, J.; Paz-Gonzalez, A. Estimating soil organic matter using interpolation methods with an electromagnetic induction sensor and topographic parameters: A case study in a humid region. Precis. Agric. 2017, 18, 882–897. [Google Scholar] [CrossRef]
- Ladd, J.N.; Foster, R.C.; Nannipieri, P.; Oades, J.M. Soil structure and biological activity. Soil Biochem. 1996, 9, 23–78. [Google Scholar]
- Catroux, G.; Chaussod, R.; Nicolardot, B. Assessment of nitrogen supply form the soil. C.R. Acad. Agric. Fr. 1987, 3, 71–79. [Google Scholar]
- Verberne, E.L.J.; Hassink, J.; de Willigen, P.; Groot, J.J.R.; van Veen, J.A. 1990. Modelling organic matter dynamics in different soils. Neth. J. Agric. Sci. 1990, 38, 221–238. [Google Scholar]
- Tisdall, J.M.; Oades, J.M. Organic matter and water-stable aggregates in soils. Eur. J. Soil Sci. 1982, 33, 141–163. [Google Scholar] [CrossRef]
- Elliott, E.T.; Coleman, D.C. Let the soil work for us. Ecol. Bull. 1988, 39, 2–32. [Google Scholar]
- Hassink, J.; Bouwman, L.A.; Zwart, K.B.; Bloem, J.; Brussaard, L. Relationships between soil texture, physical protection of organic matter, soil biota, and C and N mineralization in grassland soils. Geoderma 1993, 57, 105–128. [Google Scholar] [CrossRef]
- Bae, H.C.; Cota-Robles, E.H.; Casida, L.E. Microflora of soil as viewed by transmission electron microscopy. Appl. Microbiol. 1972, 23, 637–648. [Google Scholar] [CrossRef] [PubMed]
- Blankinship, J.C.; Schimel, J.P. Biotic versus abiotic controls on bioavailable soil organic carbon. Soil Syst. 2018, 2, 10. [Google Scholar] [CrossRef]
- Aitkenhead, J.A.; McDowell, W.H. Soil C:N as a predictor of annual riverine DOC flux at local and global scales. Glob. Biogeo. Cycles 2000, 14, 127–138. [Google Scholar] [CrossRef]
- Gales, K.; Ayling, S.M.; Cannell, R.Q. Effects of waterlogging and drought on winter wheat and winter barley grown on a clay and sandy loam soil. II. Soil and plant water relationships. Plant. Soil 1984, 80, 67–78. [Google Scholar] [CrossRef]
- Wu, J. Change in soil microbial biomass and regulating factors in an alpine meadow site on the Qinghai-Tibetan Plateau. Soil Sci. Plant. Nutr. 2020, 66, 177–194. [Google Scholar] [CrossRef]
- Liu, N.; Zhang, Y.; Chang, S.; Kan, H.; Lin, L. Impact of grazing on soil carbon and microbial biomass in typical steppe and desert steppe of Inner Mongolia. PLoS ONE 2012, 7, e36434. [Google Scholar] [CrossRef]


| Parameter | Clay (%) | SOC (g kg−1) | ED (m) |
|---|---|---|---|
| Clay (%) | 1.00 | −0.27 | 0.07 |
| SOC (g kg−1) | −0.27 | 1.00 | −0.28 |
| ED (m) | 0.07 | −0.28 | 1.00 |
| Analysis | Parameter | Estimate | Std. Error | df | t Value | p-Value |
|---|---|---|---|---|---|---|
| MBC | Intercept | 3.045 | 0.043 | 4.9 | 68.779 | <0.001 |
| log10 | Clay (%) | −0.007 | 0.002 | 230.4 | −4.273 | <0.001 |
| (mg kg−1) | SOC (g kg−1) | 0.008 | 0.001 | 230.0 | 9.174 | <0.001 |
| ED (m) | −0.003 | 0.007 | 228.2 | −0.370 | 0.712 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liddle, K.; McGonigle, T.; Koiter, A. Microbe Biomass in Relation to Organic Carbon and Clay in Soil. Soil Syst. 2020, 4, 41. https://doi.org/10.3390/soilsystems4030041
Liddle K, McGonigle T, Koiter A. Microbe Biomass in Relation to Organic Carbon and Clay in Soil. Soil Systems. 2020; 4(3):41. https://doi.org/10.3390/soilsystems4030041
Chicago/Turabian StyleLiddle, Kaylin, Terence McGonigle, and Alexander Koiter. 2020. "Microbe Biomass in Relation to Organic Carbon and Clay in Soil" Soil Systems 4, no. 3: 41. https://doi.org/10.3390/soilsystems4030041
APA StyleLiddle, K., McGonigle, T., & Koiter, A. (2020). Microbe Biomass in Relation to Organic Carbon and Clay in Soil. Soil Systems, 4(3), 41. https://doi.org/10.3390/soilsystems4030041





