Abstract
Most of our terrestrial carbon (C) storage occurs in soils as organic C derived from living organisms. Therefore, the fate of soil organic C (SOC) in response to changes in climate, land use, and management is of great concern. Here we provide a unified conceptual model for SOC cycling by gathering the available information on SOC sources, dissolved organic C (DOC) dynamics, and soil biogeochemical processes. The evidence suggests that belowground C inputs (from roots and microorganisms) are the dominant source of both SOC and DOC in most ecosystems. Considering our emerging understanding of SOC protection mechanisms and long-term storage, we highlight the present need to sample (often ignored) deeper soil layers. Contrary to long-held biases, deep SOC—which contains most of the global amount and is often hundreds to thousands of years old—is susceptible to decomposition on decadal timescales when the environmental conditions under which it accumulated change. Finally, we discuss the vulnerability of SOC in different soil types and ecosystems globally, as well as identify the need for methodological standardization of SOC quality and quantity analyses. Further study of SOC protection mechanisms and the deep soil biogeochemical environment will provide valuable information about controls on SOC cycling, which in turn may help prioritize C sequestration initiatives and provide key insights into climate-carbon feedbacks.
1. Introduction
Soils have the unique ability to sequester and store large amounts of carbon (C). They are estimated to contain about two to three times the amount of C stored in the atmosphere and vegetation combined [1,2]. Most soil C is in the form of organic C derived from living organisms and has been stored for hundreds to thousands of years in deeper soil layers (below ~20 cm) [3,4]. Therefore, the loss of even relatively small amounts of this soil organic C (SOC) could exacerbate global climate change by releasing substantial amounts of greenhouse gases, such as carbon dioxide (CO2) and methane (CH4), to the atmosphere. Various components of the soil environment interact to protect SOC from loss or, alternatively, enhance the microbial decomposition of organic C compounds. Changes in environmental constraints affect SOC cycling by altering soil properties and C inputs, which in turn alter soil microbial communities and processes, as well as their access to organic matter and other nutrients.
Our shifting understanding of SOC “stability” and the interactions of organic C with the soil environment have resulted in the proposal of numerous conceptual and mechanistic SOC models [2,5,6]. Here we provide a unified conceptual model for SOC cycling by gathering the available information on SOC sources, dissolved organic C (DOC) dynamics, and deep soil biogeochemical processes. Emphasis is placed on how our emerging understanding of SOC protection mechanisms translates into a pressing need to sample deeper soil layers. We also discuss SOC vulnerability on a global scale in different soil types and ecosystems. Finally, we identify additional common practices that may affect SOC quality and quantity analyses and their comparability, such as the use of different soil sampling methods [7], the exclusion of the coarse soil (>2 mm) fraction [8], and the use of varying SOC fractionation techniques [9,10,11].
2. Vertical Distribution and Storage: Our Shifting Understanding of Soil Organic Carbon “Stability”
Soil depth plays an important role in SOC distribution. Although SOC concentrations decrease rather linearly with increasing depth, total SOC storage is greater in deeper soil layers than at the surface. In fact, due to increased mass in deeper soil layers, well over 50% of the global SOC stock is stored in deeper soil layers (below ~20 cm) [12,13]. Radiocarbon ages of SOC also increase with depth, with deep SOC commonly being as old as 1000 to more than 10,000 years [3,4,14,15]. The sharp increase in SOC age from surface to deeper soil suggests that SOC is stored longer in deeper soil layers.
2.1. “Humus” Versus a Decomposition Continuum
Historically, long-term storage (or preservation) of SOC was thought to occur via the formation of complex and “stable” SOC compounds, generally referred to as “humus”. While this theory of “humification” is based on alkaline extraction of SOC in the laboratory (which Lehmann and Kleber [2] describe as “incomplete, selective, and prone to creating artifacts”), contemporary, in situ spectromicroscopic techniques have revealed the presence of much smaller, more chemically simple organic C compounds in the soil environment [3]. Moreover, no modern analytic techniques have observed “humic substances” in the soil [2], and alkaline extraction is unable to confirm the existence of these substances [16]. The emerging concept of SOC storage as an ecosystem property, rather than simply the result of chemical “recalcitrance” or “stability,” is well-supported in the literature over the past few decades [2,3,5,17]. This concept is based on the premise that organic matter in the soil decomposes along a continuum. Microbial processing increases the solubility and potential for protection of organic C compounds [2,18,19]. Protection of these compounds is provided by the interaction of the physical, chemical, and biological properties of the soil environment, which reduce the probability (and thus rate) of decomposition, creating the capacity of soil to sequester C [2,3].
2.2. Physical Protection and Mineral Sorption
In most mineral soils, SOC protection occurs primarily through abiotic mechanisms involving physical protection and mineral sorption [2]. Physical protection takes place in the fine pores of soil microaggregates (0.053 mm < diameter (Ø) < 0.25 mm) in which anoxic conditions prevail or which are inaccessible to, or spatially separated from, microbial decomposers [20,21]. Microaggregates are often formed and stabilized within macroaggregates (Ø > 0.25 mm), the formation of which is fostered primarily by roots [21,22]. Microorganisms, particularly root-associated microorganisms, and organic matter also play key roles in fostering aggregate formation [21,22,23]. The sorption of dissolved organic C (DOC) compounds to minerals occurs in large part with the clay fraction [24,25]. The iron and aluminum hydrous oxides (secondary minerals containing iron and aluminum, respectively, hydroxides [OH−], and weakly bound H2O) of some clay separates are especially effective sorbents of SOC [5,24,26,27,28,29]. Mineral sorption is likely the primary means of SOC protection [2,30,31,32,33,34,35,36] and is particularly important in deeper soil layers [2,5,26,37,38]. The fraction of SOC sorbed to minerals has been shown to increase with depth and comprise the majority of organic C stored in soils [25,26,39,40,41]. While the age of SOC in the surface soil is primarily dependent on climate and land use variables, deep SOC age is more strongly related to soil type, highlighting the importance of soil texture and mineralogy—specifically the clay fraction—in promoting long-term SOC storage [42,43].
2.3. Vertical Movement and Cycling
Major mechanisms contributing to deep SOC include C inputs from roots and root-associated-microorganisms [25,44,45,46,47,48,49,50], as well as the translocation of DOC to deeper soil layers [5,6,26,27,30,51]. Rain events and new inputs of organic matter are important drivers of DOC flux [44,52]. Waterflow or the saturation of mineral sorption sites in surface soil layers by organic matter inputs preferentially releases less-strongly-sorbed DOC from sorption sites [52,53]. Dissolved organic C released into the soil solution is susceptible to further microbial processing and transport to deeper soil layers, where minerals often have greater surface area (higher clay content and greater abundance of iron and aluminum hydrous oxides) and mineral surfaces are not yet saturated with C [21,53,54]. As DOC is transported vertically down the soil profile, certain organic C compounds are preferentially sorbed to mineral surfaces. Preferential sorption appears to occur with smaller, microbial-derived, highly-oxidized, hydrophobic, and nitrogenous organic C compounds [2,5,18,24,27,55,56,57,58,59], likely due to the stronger affinity for mineral surfaces of proteinaceous microbial residues versus less proteinaceous plant residues [29,60,61,62]. Therefore, the quality (i.e., composition) of organic C compounds plays a role in long-term SOC storage [63] in relation to soil mineralogy and mineral associations, particularly at depth [64].
Dissolved organic C fluxes contribute indirectly and directly to SOC storage by supporting microorganisms and sorbing to the mineral fraction, respectively. In fact, nearly one-third of deep soil microbial activity may be supported by DOC fluxes [44]. Similar to SOC concentrations, DOC concentrations decrease with increasing soil depth [27,52,54,65]. However, this decrease appears to be primarily due to sorption rather than respiration losses [54,66]. Dissolved organic C age has been found to increase with depth [52] and to approximate the age of SOC at the same depth [67,68], supporting the framework of a continuum of organic matter decomposition and protection and release processes.
3. Inputs and Sources: A Deeper Look at Shoot Litter, Roots, and Microorganisms
Quantifying organic matter inputs to soil from various sources and their relative and total contributions to SOC can provide key insights into processes related to SOC protection and cycling [18]. Organic matter inputs to soil include shoot litter, roots (including root death and a wide variety of rhizodeposition processes, such as passive and active secretion of exudates and the sloughing of root tissues), and microbial biomass [69,70,71]. Relative contributions to SOC from these three sources, and the complex dynamics of their interactions with the soil environment, are largely debated.
Shoot-derived C is incorporated into the bulk SOC through the transport of DOC from the litter layer [5,6,30,65], as well as through the mixing of particulate organic matter into superficial soil layers via soil fauna (i.e., bioturbation). Bioturbation can play an important role in SOC cycling in some ecosystems [26]. However, the abundance and effect (particularly long-term) of bioturbation agents usually declines sharply with depth [72], and their contributions to deep SOC may be negligible compared to DOC transport [73]. Because root- and microbial-derived C are input belowground, incorporation into the DOC pool and bulk SOC may be more direct. Nonetheless, most belowground C inputs undergo stages of decomposition via repeated microbial processing, protection, and release into the DOC pool.
Studies that reviewed or modeled DOC dynamics in forest ecosystems estimated that DOC-derived SOC represented ~20%–89% of total SOC [44,74,75,76]. Unfortunately, only shoot-derived C contributions to the DOC pool were considered in these reviews and models, leaving gaps in our knowledge of DOC dynamics [70,77] and the relative contributions of different sources to SOC [78]. Here we review the evidence regarding SOC and DOC sources and provide a unified conceptual model for SOC cycling (Figure 1).
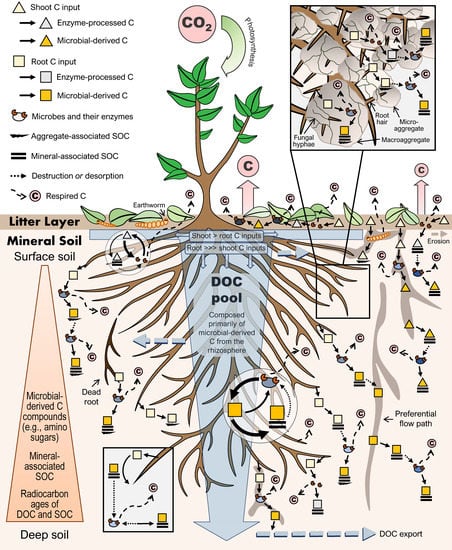
Figure 1.
Proposed model for soil organic carbon (SOC) cycling showing root carbon (C) inputs as the primary source of both SOC and dissolved organic C (DOC) in most ecosystems. Root-derived C is shown undergoing multiple stages of microbial processing, protection, and release into the DOC pool as it is transported vertically down the soil profile. Unlike shoot C inputs—which are often mineralized in the litter layer or undergo partial oxidation via microbial extracellular enzymes—most root C inputs will undergo microbial assimilation, biosynthesis, and turnover prior to SOC incorporation. Microbial processing increases the solubility and potential for protection of organic C compounds, which are protected primarily through abiotic mechanisms involving physical protection within soil micro- and macro-aggregates (upper right inset) and mineral sorption of DOC compounds. Microbial activities, and thus SOC decomposition, can be stimulated by multiple mechanisms, and the protection of SOC can be counteracted by physical or biochemical mechanisms, such as by certain root exudates fostering the release of organic C compounds from protective mineral associations (lower left inset). Soil fauna (e.g., detritivores) are represented by earthworms, which contribute to bioturbation. Respired C is shown for the whole soil (in relative amounts), as well as for individual processes. Differences in microbial communities between the litter layer, rhizosphere (i.e., the portion of soil in the immediate vicinity of roots), bulk soil, and with depth are indicated by color. Within the circles illustrating DOC cycling, thicker lines indicate more rapid rates. The size of the ends of wedges represents a relative increase or decrease. Controls on the processes shown include temperature, moisture, the flora and fauna present, and other ecosystem and soil properties (e.g., parent materials, texture, mineralogy, and pH).
3.1. Root Versus Shoot Contributions
About 40–50% of all C fixed by plants via photosynthesis is allocated belowground, and about one-fourth of this C is released into the soil environment by growing roots via rhizodeposition [69,71,79,80]. Compared to aboveground contributions to mineral SOC, root-derived C contributions are approximately 1.5–10 times greater, and they may make up as much as 75% of total SOC [25,44,45,46,47,48,49,50]. In a recent review, Jackson et al. [37] found that average root-derived C contributions to SOC in agricultural systems were 45%, compared to just 8% for aboveground C inputs. Several direct and indirect mechanisms are responsible for the incorporation of root C into the bulk SOC. Direct mechanisms include exudate sorption to mineral surfaces and root litter protection in aggregate interiors, while indirect mechanisms include processing or assimilation by root-associated microorganisms or mycorrhizae [19,21,81]. For example, ectomycorrhizal fungi appear to obtain all their C from tree roots, thus acting primarily as a transfer of root C to the surrounding soil environment [82]. In a chronosequence of boreal forested islands, 14C bomb-carbon modeling revealed that 50–70% of the SOC was root- or root-associated-microbial-derived [83]. Similarly, in a poplar plantation, mycorrhizal fungal external mycelium were found to contribute ~60% of new SOC over three growing seasons, a larger contribution than fine root turnover inputs and over twice that of shoot litter inputs [84].
Shoot litter and root manipulation experiments have provided mixed results as to whether aboveground or belowground C inputs are more important for maintaining SOC stocks [85,86,87]. Recolonization of roots in “root exclusion” plots [86], potential changes in bulk density between plots over time [86,88], and confounding variables, such as microbial biomass [89], can complicate the interpretation of results. For example, shoot litter manipulations affected SOC concentrations (at 0–10 cm in depth) after only two years in a wet tropical forest; however, changes in fine root biomass, surface DOC fluxes, microbial biomass, nutrient fluxes, and soil moisture accompanied the SOC changes [89]. Therefore, the effects of shoot litter manipulation on SOC concentrations may have been indirect and more related to modifications in fine root biomass and root C inputs, which in turn may have driven changes in microbial activity and microbial biomass turnover [89]. Although some studies have shown the importance of shoot C inputs to maintaining SOC stocks [85,87,90], changes in SOC quantity from shoot litter manipulations have not resulted in similar changes in SOC quality [86,89]. In a temperate deciduous forest, SOC composition and microbial biomass (at ~0–10 cm in depth) were strongly influenced by root manipulations over 20 years, while shoot litter manipulations had no effect on either [91]. This indicates that SOC stocks are primarily derived from underground sources. However, modifications in aboveground C inputs may rapidly affect SOC quantity to varying degrees in different ecosystems, likely due to changes in both particulate organic C [87] and complex biogeochemical interactions in the soil environment [86,90].
3.2. Microbial Processing and Turnover
Incubation and in situ root growth experiments have shown that the mean transit time of root-derived C in soils is over twice that of shoot-derived C [21], which is often mineralized in the litter layer [3,52]. The creation of micrometer-scale pores and aggregates by mycorrhiza and root-hair activities enhance the physical protection of root-derived C from microorganisms [18,21,92], as does the fact that C input belowground has more opportunities for interactions with the mineral soil and sorption to the clay fraction than does shoot-derived C. Nonetheless, due to organic matter inputs by roots, the portion of soil in the immediate vicinity of roots (i.e., the rhizosphere) has higher concentrations of SOC and other nutrients, and thus enhanced microbial activity compared to the bulk soil [21,93]. Therefore, a substantial portion of rhizodeposits undergoes microbial processing and bio-transformation into other microbial-derived C compounds prior to SOC incorporation [6,25,79,81]. For example, root exudates—which are the primary form of rhizodeposits [69]—have been found to rapidly approach the signature of microbial sugars, while rhizospheric sugars as a whole were found to rapidly approach the chemical composition of the bulk SOC [79]. This suggests that a substantial portion of root exudates, following microbial processing, is ultimately incorporated into the bulk SOC.
Microbial necromass and products may make up as much as 80% of SOC [94]. Isotopic and biomarker analyses confirm that root-derived C is the primary source of C for soil microorganisms [95,96]. Most root C inputs undergo microbial assimilation, biosynthesis, and turnover [6]. In contrast, aboveground C inputs may evade passing through a living microbial body, rather undergoing partial oxidation via microbial extracellular enzymes [6]. Repeated microbial processing of root C inputs and interrelated SOC protection mechanisms (i.e., mineral sorption) are particularly important in contributing to SOC storage in deeper soil layers [51,97]. In fact, microbial-derived C compounds (amino sugars) associated with root C inputs have been found to increase in abundance (amino sugars per g C) in the rhizosphere and with increasing soil depth [25,98]. Additionally, soils with higher initial microbial biomass and clay fractions result in greater incorporation of C inputs into microbial biomass and SOC [99,100], illustrating the key role soil texture plays in SOC storage. By modelling root mass, amino sugars, the clay fraction, and the particulate organic C fraction, Angst et al. [25] were able to account for >90% of variability in SOC stocks [35,101].
3.3. Dissolved Organic Carbon Pool
A larger clay fraction leads to increased opportunities for SOC protection via mineral sorption of DOC [25,27,32], the primary protection mechanism promoting long-term SOC storage [2,30,31,32,33,34,35,36]. At the same time, because preferential sorption to mineral surfaces occurs with proteinaceous microbial residues [29,60,61,62], SOC stored long-term is enriched in microbial-derived C compounds [63,98]. The strong relationships between root C inputs, microbial-derived C, the clay fraction, and SOC storage indicate that the DOC pool is likely comprised primarily of root-associated-microbial-derived C. Multiple studies reveal a sharp contrast between litter layer and mineral soil DOC sources and fluxes. For example, in a meta-analysis of shoot litter manipulation experiments in temperate forests, DOC differed only in the litter layer in response to aboveground C input changes, while there was no response in the mineral soil [90]. Similarly, although isotopically-labelled shoot-derived C contributed to ~70% of DOC leached from the litter layer in two deciduous forests, this fraction decreased to 8% and 5% at 5 and 10 cm in depth, respectively [65], indicating that the majority of DOC in the soil profile was derived from other sources. This is consistent with the lack of relationship found between mineral soil DOC fluxes and environmental conditions in temperate forests, despite DOC fluxes from the litter layer generally increasing with annual precipitation [102]. The evidence suggests that the sources and drivers of DOC flux in mineral soil may be largely unrelated to the litter layer.
Root biomass and the DOC pool follow similar distribution patterns, with the majority of root biomass across all terrestrial biomes in the upper 30 cm of soil [103] and the majority of DOC emanating from the surface soil (above ~20 cm) in most ecosystems and decreasing with increasing soil depth [44]. In a temperate forest, mean microbial biomass and DOC concentration were two times higher in rhizosphere compared to bulk soils, and the size of these organic C pools was positively related to exudation rates [104]. Similarly, DOC concentrations in a temperate coastal prairie were highest directly below the main rooting zone and the DOC was similar in magnitude and chemical characteristics to DOC collected from a nearby forest [52]. Although there was some seasonal variation in surface DOC concentration and composition in these contrasting grassland and forest ecosystems, the fluctuations declined steeply with depth [52]. This indicates that fresh shoot C inputs are only a minor component of DOC pools, limited primarily to the surface soil and fluxes following rain events after long dry periods [52].
Isotopic and spectroscopic evidence have revealed that the DOC pool is comprised primarily of smaller, more soluble, highly-oxidized organic C compounds released from the bulk SOC following microbial processing [52]. In the deeper soil layers (below ~20 cm) of a temperate forest, Ahrens et al. [51] determined that the DOC pool was comprised of 50–60% microbial-derived C. Microbial diversity and DOC chemistry are both strongly associated with soil depth. In fact, microbial population composition and the chemical character of DOC are interdependent and vary more with soil depth than with landscape position or local vegetation [105]. Compared to shoot-derived C, root-derived C has a much greater influence on soil microbial community composition [106] and biomass [91]. Similarly, SOC composition is strongly associated with soil depth, root C inputs, and microbial biomass [86,89,91]. On the other hand, manipulations in aboveground C inputs have resulted in only minor or no changes in SOC composition in the mineral soil, even after up to 20 years of shoot litter manipulations [86,89,91].
3.4. Synthesis
The evidence suggests that, in most ecosystems, both SOC and DOC are primarily derived from belowground sources (i.e., roots and microorganisms). Because shoot litter leachate may contribute more substantially to bulk SOC as opposed to rhizosphere SOC [6], the ratio of rhizosphere to bulk soil is an important factor in determining the relative contribution to SOC of aboveground versus belowground sources [81]. Additionally, the litter layer likely plays an important indirect role in maintaining SOC stocks in some forest ecosystems [89]. Our review indicates that microbial-derived C, primarily from microbial-processed and bio-transformed root C inputs [25,95,96], comprises the majority of SOC in most ecosystems [6,25,51,79,83,94] and is protected via the sorption of DOC to minerals [27,52,79,104], particularly to clay [24,25,35]. As shown in Figure 1, root-derived C undergoes multiple stages of microbial processing, protection, and release into the DOC pool as it is transported vertically down the soil profile—helping resolve the argument that the phenomenon of SOC radiocarbon ages increasing with depth is not well explained if roots are considered the primary source of SOC [5,21,107].
4. Deep and Dynamic: Old Soil Organic Carbon is Susceptible to Abrupt Decomposition
Despite organic C in deeper soil layers having longer transit times than in more superficial layers [108], SOC traditionally considered as “stabilized” is vulnerable to abrupt decomposition [3,109]. Numerous studies have indicated that deep and old SOC may be more susceptible than superficial SOC to substantial and potentially rapid decomposition resulting from changes in environmental conditions [33,109,110,111,112,113,114,115,116,117]. Important knowledge gaps exist regarding deep soil biogeochemical processes, as well as the global amount of SOC and its vertical and spatial distribution [118,119,120,121]. Common bias against sampling deeper soil layers not only contributes to uncertainty in global SOC estimates and climate models, but it also limits our understanding of SOC cycling and protection mechanisms. Here we highlight the need to sample deeper soil layers considering our emerging understanding of SOC cycling and the growing body of evidence revealing changes in deep SOC over relatively modest periods of time in response to changes in climate, land use, and management.
4.1. Global Stocks and Sampling Bias
Global SOC stocks from 0–100 cm in depth are estimated to range from 1220–1576 Pg C (1 Pg = 1015 g) [12,13,122,123,124,125,126]. In a comparison of 27 studies over the past several decades, Scharlemann et al. [118] found the median across all estimates for the global SOC stock was roughly 1500 Pg C in the upper 100 cm. Estimates for global SOC stocks from 0–200 cm in depth range from ~2000–2500 Pg C [12,13], while the 200–300 cm stock was estimated at 351 Pg C [13]. Far fewer observations exist of the size of SOC stocks in soil below ~20 cm, causing substantial uncertainty in global SOC estimates [12,13,18,25,109,119]. Using a compilation of 86 published soil profiles, Jandl et al. [119] found that about half were sampled to 20 cm or less and ~90% were sampled to 30 cm or less.
Ignoring the component of an ecosystem that often contains the most organic C (i.e., deep soil) can lead to drastic misinterpretations of C cycling and changes over time [109]. For example, following 20 years of shoot litter manipulations in a temperate deciduous forest, Bowden et al. [86] found that most of the SOC loss (compared to the control) in plots with no aboveground C inputs occurred between 10 and 50 cm in depth. However, in shoot litter manipulation experiments across five ecosystems and totaling 70 publications used for a meta-analysis, the majority of sampling was conducted in the top 10 cm of mineral soil [90], which may have led to substantial underestimations of SOC changes. Land use conversion and climate change studies have also illustrated the need to account for deeper soil layers when assessing SOC changes over time, wherein incorrect conclusions about the direction or magnitude of SOC change can be drawn by shallow sampling [43,114,127]. Future progress toward understanding the effects of climate, land use, and management change on global C cycling will come from recognizing that deep SOC can (and does) change on decadal timescales.
4.2. Climate Change
Global surface temperature change by the end of the 21st century is projected to be in the range of +2–4 °C, with greater mean warming over land masses than over the ocean and with the most warming occurring in the Arctic region [128]. Although considerable variation in temperature can occur in the surface soil and even in deeper soil layers, the soil profile as a whole approximates the mean annual air temperature [129,130]. Therefore, soils will warm at roughly the same rate as surface temperatures over the next century. Recent studies underscore the importance of the deep SOC response to global warming. For example, in permafrost soils experiencing thaw, old deeper SOC contributions to ecosystem respiration flux increased with soil temperature and ecosystem respiration flux [113]. While in a temperate forest ecosystem, subjecting the mineral soil profile to 100 cm in depth to 4 °C warming increased CO2 production at all depths and increased total soil respiration by 34–37% [114]. About 50% and 40% of soil respiration and the warming response, respectively, occurred below 15 cm [114]. Unfortunately, most studies do not report the warming response below 20 cm [114], leaving additional uncertainty in climate models.
Climate change also may have drastic implications for DOC flux. By the end of this century, extreme precipitation events are expected to become more intense and more frequent over mid-latitude and wet tropical terrestrial ecosystems [128]. These extreme precipitation events could transport to deeper soil layers large amounts of DOC comprised primarily of recently-fixed C, especially after prolonged periods without rain events where fresh C compounds have accrued in the surface soil [44,52]. While this could lead to enhanced potential for mineral associations at depth, and thus longer-term SOC storage, rapid fluxes of DOC can also result in increased DOC export [67,131]. Moreover, increased and continuing fresh C inputs to deeper soil layers can enhance bulk SOC decomposition for years to decades [115,117,132,133] by providing deep microbial communities with the energy to synthesize extracellular enzymes for SOC decomposition [4,133,134,135]. That is, the introduction of fresh organic C compounds to deeper soil layers can stimulate, or prime, microbial activities, alleviating potential energetic barriers to SOC decomposition that may have existed.
Similarly, fresh root C inputs can enhance microbial activity and SOC decomposition, a phenomenon referred to as (positive) rhizosphere priming [136,137]. Under changing environmental conditions, wherein deep soil microbial communities are suddenly exposed to additional root C inputs, increased bulk SOC decomposition can occur. For example, elevated atmospheric CO2 concentrations and increased aboveground and belowground plant growth have resulted in losses of old SOC that exceeded the formation of new SOC, potentially due to increases in rooting depth and root C inputs [138,139,140,141]. In some cases, root exudates may drive increases in bulk SOC decomposition without associated increases in microbial biomass, indicating that the microorganisms are allocating the energy provided by fresh root C exudates to mineralization activities rather than to growth [133].
Interactions between root exudates and the soil mineral matrix can enhance the availability of SOC. For example, the common root exudate, oxalic acid, fosters the release of organic C compounds from protective mineral associations, thereby promoting SOC loss via microbial decomposition or export [33]. This may be why soils with finer texture experience greater rhizosphere priming effects than soils with coarser texture [132]. In other words, rhizosphere priming appears to impact mineral-associated SOC—that potentially has been stored long-term—more than particulate SOC [132]. According to a recent meta-analysis, rhizosphere priming increases SOC decomposition rates by an average of 59% [132], a phenomenon likely driven by root exudates rather than root litter [133]. Compared to surface soil, deeper soil layers appear to be as sensitive or more sensitive to rhizosphere priming [116,135]. In addition to possible rhizosphere priming effects, the introduction of roots to deeper soil layers increases soil moisture variability and aeration through transpiration, exposing soil microorganisms to potentially more favorable metabolic conditions [115].
Projected increases in hot temperature extremes over most land areas during the next century, especially on seasonal timescales [128], could promote the loss of deep SOC by modifying fine root distribution and corresponding rhizodeposits in the soil. In a study that modeled root allocation in temperate forest ecosystems, increased soil temperatures, reductions in soil moisture, and subsequent increases in soil strength led to fine root colonization of deeper soil layers [49]. Compared to coarse roots, fine roots likely release more exudates that travel further from the root surface [142], and they have faster turnover rates [143], both of which can induce rhizosphere priming effects in the surrounding bulk soil. On the other hand, root litter appears to decompose slower in deeper soil layers, likely due to shifts in microbial communities and decreased microbial biomass and available nitrogen [130,144]. In the absence of rhizosphere priming effects, this slowly decomposing root litter could increase SOC stocks and long-term storage under climate-change-induced increases in primary production and rooting depth [130].
Despite multiple soil biogeochemical models projecting reductions in SOC stocks and increases in CO2 emissions from soils under warming, a substantial fraction of experiments diverge with these modeled results [145]. Additionally, some studies have shown varying priming effects with fresh C inputs depending on soil properties or nutrition, plant nutrient contents, C sources, or relative amounts of C inputs to soil [146,147,148,149,150,151,152]. Although additional shoot litter may be unlikely to increase SOC stocks [86,87,90], roots and root-associated microorganisms contribute more substantially to SOC storage, particularly in deeper soil layers. Therefore, climate-change-induced increases in root biomass and rooting depth would likely increase SOC stocks eventually [153]. However, such increases in SOC stocks could take decades to manifest and may be preceded by priming-induced deep and old SOC losses.
4.3. Land Use Change and Management
Land use change and management have been observed to impact deep SOC on decadal timescales [154]. In a meta-analysis, conversion from forest to pasture was found to increase SOC stocks by 7–13% above 100 cm, with similar increases for all depths sampled (<30, 30–50, and 50–100 cm) [155], whereas conversion from forest to crop decreased SOC stocks by ~50% above 60 cm, with similar decreases for the depths sampled (<30 and 30–60 cm) [155]. Conversion from crop to secondary forest increased SOC stocks by 53% [155]; however, sampling depth was not noted in the analysis. When deeper soil layers were sampled following conversion from crop to forest, SOC losses from >15 cm in depth were found to offset surface SOC gains for at least 40 years after conversion [50,115]. Forest harvest also has been shown to affect deep soil, reducing SOC from 60–100+ cm in depth by an average of ~18%, according to a recent meta-analysis [156]. Compared to harvest, thinning resulted in even greater (+9%) SOC reductions in the mineral soil [156]. Deep SOC seems to be especially sensitive to thinning practices [115]. For example, in a temperate forest, thinning reduced SOC stocks by 25% compared to the control over ~11 years of treatments, with most of the loss occurring below ~20 cm [157].
Reductions in deep SOC following land use change and management practices likely result from a combination of effects and complex interactions in the soil environment that enhance microbial activity, SOC decomposition, and DOC export. Land use change and management can affect soil temperature, moisture, and DOC flux, among other soil properties and processes [158]. For example, mean soil temperature and mean daily soil temperature flux at 100 cm in depth have been observed to increase by ~2–4 °C and ~4–6 °C, respectively, in the first two years following harvest compared to an unharvested reference in a temperate forest ecosystem [159]. Soils compacted by harvest treatments experienced greater temperature increases than soils not compacted by the same treatments [159], probably due to the higher thermal conductivity of compacted soils [129]. These increases in soil temperature can substantially hasten rates of SOC decomposition by enhancing microbial metabolism [129]. Similarly, DOC flux is an important driver of soil biogeochemical processes. Increases in DOC flux from surface soil layers due to land use change or management practices can introduce substantial quantities of fresh C compounds to deeper soil layers, potentially enhancing old SOC decomposition and DOC export [67,68,131,160].
Disturbance or changes in the litter layer in response to land use or management can also affect deeper soil layers. For example, the exclusion of aboveground C inputs over 20 years in a temperate deciduous forest resulted in reductions (−24%) in SOC stocks to at least 50 cm in depth, with much of this loss occurring below 10 cm [86]. On the other hand, increased aboveground C inputs, which could result from enhanced forest productivity, have shown mixed promise toward increasing SOC stocks [86,87,90]. Although few shoot litter manipulation experiments have sampled deeper soil layers [90], a recent incubation experiment revealed that deep SOC as opposed to surface SOC would be more susceptible to priming induced by increased aboveground C inputs [117]. Fresh C from shoot litter could potentially reach deeper soil layers via preferential pathways of interconnected large soil pores. Preferential pathways, which can persist for decades in forest ecosystems [161], represent means for recently-fixed superficial C to enter deeper soil layers without substantial abiotic and biotic interactions with the soil environment along the way [54]. Because decomposition rates in preferential flow paths are likely enhanced [161] due to increased SOC concentrations (+10–70%) and microbial biomass (+9–92%) compared to nearby bulk soil [27,162], these pathways can enhance SOC decomposition in the surrounding soil.
Deep SOC losses observed following reforestation of formerly cultivated land may be primarily due to rhizosphere priming effects induced by changes in root biomass and surface area, rooting depth, and rhizodeposits [50,115]. That is, when relatively shallow-rooted agricultural ecosystems are converted to deep-rooted forests, root C inputs are introduced to deeper soil layers and can enhance or promote the decomposition of older SOC that was formerly protected under the agricultural ecosystem [115,138]. Forest management practices such as fertilization also have been observed to increase soil rooting depth [163], and the added nitrogen could enhance the potential rhizosphere priming response in deeper soil layers [116,144]. On the other hand, increased live fine root length, and thus increased activity and exudation, is considered the likely explanation for gains in SOC storage following conversion from forest to pasture [153]. As noted previously, practices or environmental conditions that increase root C inputs to soil are likely to also increase microbial biomass, which in turn could eventually increase SOC stocks and long-term storage. Nonetheless, in the case of conversion from crop to forest, SOC stocks took nearly 50 years to stabilize, resulting in deep and old SOC losses before potential gains could manifest [50,115].
5. Vulnerability on a Global Scale: Permafrost, Peatland, Wetland, and Forest Soils
Our ability to increase SOC storage and decrease greenhouse gas emissions from soils on a large scale is partially dependent on our gaining a deeper understanding of the mechanisms and controls responsible for the protection of SOC [145,164]. Assessing the vulnerability of both shallow and deep SOC in various soil types and ecosystems is essential to prioritize C sequestration initiatives and constrain climate models. Not only is SOC distributed unequally in the vertical profile, with the majority of SOC stored below ~20 cm [13], but it is also distributed unequally on global and spatial scales, varying substantially across biomes and landscapes [35]. As the magnitude of SOC loss due to both warming and priming increases with the size of the initial stock, soils with larger standing SOC stocks are especially at risk in our changing world [163,165].
5.1. Permafrost Soils
Organic C is stored in soil wherever primary production rates and organic matter inputs are greater than decomposition rates. This relationship of production versus decomposition is highly dependent on microbial controls, which in turn are highly dependent on climate. In general, maximum decomposition rates occur in warm, humid environments. Therefore, the northern hemisphere stores more SOC than does the southern hemisphere [126]. Soils in the northern permafrost (i.e., any subsurface materials that remain below 0 °C for two or more consecutive years) region are estimated to contain ~1700 Pg C to 300+ cm in depth [166], which is more than the global SOC stock estimated for the upper 100 cm [118]. Nearly 90% of this ~1700 Pg C occurs in perennially frozen soils and deposits [166].
Permafrost SOC is especially vulnerable to abrupt and potentially rapid decomposition due to global warming [164]. While SOC in most mineral soils is protected in aggregate interiors or through adsorption to mineral surfaces [2], protection from microbial decomposition in permafrost soils is primarily dependent on the frozen conditions of the soil [164,167]. The thawing of permafrost thus enables the decomposition of ancient SOC. Unfortunately, the Arctic region is likely to warm more rapidly than the global mean [128], which is expected to increase seasonal permafrost thaw depths by at least 30–50% by 2050 [168]. Substantial reductions, ranging from 37–81% by 2100, in the upper 3.5 m of permafrost in the northern hemisphere have been projected by numerous climate models [128]. During and after permafrost thaw, protection and long-term storage of unfrozen SOC resulting from soil processes, such as the development of soil structure or mineral sorption, is possible [3]. However, thawed permafrost is also susceptible to erosion and mass movement by mudslides [129].
5.2. Peatland and Wetland Soils
Similar to permafrost soils, peatland and wetland soils are more vulnerable than most mineral soils to rapid change because the protection of SOC in these systems is primarily dependent on the anaerobic conditions under which the soils formed [164,167]. Peatlands and wetlands are estimated to contain ~500 Pg C [169,170], or about one-third of the global SOC stock estimated for the upper 100 cm [118]. While peatlands occupy only 3% of the terrestrial global surface [171], they contain over 80% of this estimated 500 Pg C [169]. Expected decreases in soil moisture during the summertime in continental areas [128,172] could potentially dry out surface layers of peat, thus making substantial amounts of SOC available for aerobic decomposition [167]. While additional C sequestration in vegetation and reduced CH4 emissions may offset increases in CO2 emissions from peatlands under aerobic conditions, increases in peat fire frequency and severity expected due to higher temperatures and peat drying would rapidly release large amounts of C to the atmosphere [167].
5.3. Forest Soils
Forest ecosystems are estimated to contain over half the global SOC stock [12,13,18], and soils not affected by land use change have been acting as a global C sink for centuries [1,173]. Between 1990 and 2007, the world’s forests sequestered as much as 30% of annual global anthropogenic CO2 emissions [174,175]. However, differences between the two large fluxes of forest soil C inputs and soil respiratory C losses are small [176,177]. Therefore, even minor changes in C inputs or losses from forest soils can potentially affect the global C cycle [176,178]. In our warming world, it is unclear if forest soils will continue to act as a global C sink. Hastened microbial metabolic processes due to warming are projected to result in net global reductions in SOC storage [179]. Moreover, in some forest ecosystems, warming-induced increases in primary production and rhizodeposition could result in priming effects, further decreasing SOC stocks for decades, particularly in deeper soil layers [33,110,111,112,132,138,139,140,180]. On the other hand, given the pivotal role that microorganisms play in SOC cycling—that is, increasing the solubility and potential for protection of organic C compounds [2,18,19]—warming and increased belowground C inputs could potentially lead toward greater opportunities for SOC protection and long-term storage [164].
5.4. Models
Current soil models used for climate change projections do not reflect our emerging understanding of SOC cycling, protection, and long-term storage mechanisms [164]. A recently developed SOC model—Carbon, Organisms, Rhizosphere, and Protection in the Soil Environment (CORPSE)—revealed that priming effects, when accounted for, resulted in net SOC storage losses in most terrestrial ecosystems under elevated CO2, counteracting SOC gains from increased primary production and C inputs [181]. However, priming effects are generally unaccounted for in global models [138]. In fact, standard models used to predict SOC dynamics do not incorporate direct microbial controls on the vertical and spatial distribution of SOC stocks [126]. By modelling microbial processes, Wieder et al. [126] were able to explain 50% of the variation in global SOC observations in the upper 100 cm of soil compared to traditional models that explained only 28–30% of the variation. These results underscore the importance of including microbial controls on SOC dynamics in global models, as well as the need for further study of deep soil microbial processes, SOC protection mechanisms, and their interactions [78,141,145,164]. Much uncertainty remains in controls on SOC cycling, particularly in the deep soil environment. Long-term studies focused on changes in C inputs to soil—and resulting shifts in superficial and deeper soil microbial communities and activity, DOC fluxes and chemistry, SOC fractions, and greenhouse gas fluxes—are needed to help constrain soil biogeochemical models and, by extension, climate models [145].
6. Sampling and Processing: The Need for Methodological Standardization
To understand the effects of climate, land use, and management change on C cycling in soils, our ability to accurately quantify and compare SOC stocks over time is critical. Unfortunately, all soil sampling and processing methods are not equally accurate, precise, or comparable. The inaccuracy or incompatibility of SOC quality and quantity results obtained with different techniques presents additional challenges toward understanding soil biogeochemical processes and SOC protection mechanisms. Identifying these actual or potential issues and developing standards will move us closer to creating more reliable global SOC databases and models.
6.1. Sampling Methods
Numerous soil sampling tools and methods exist. Of the three most common methods—which include core, clod, and excavation methods—the core method is by far the most commonly used [7]. Despite being the most used method, the core method frequently has been found to underestimate soil mass and bulk density compared with other soil sampling methods, which can lead to substantial underestimations of SOC stocks [7,182,183,184,185,186]. Therefore, it is recommended that the accuracy of the core method for measuring these parameters be assessed for a given soil and adjusted appropriately using a more reliable method, such as the excavation method [7]. Additional incompatibility issues arise due to whether soil is sampled by depth or horizon and how (or if) bulk density is used as the parameter to calculate SOC stocks [7,187,188]. In general, sampling by horizon is recommended for studying pedogenic controls on soil properties, while sampling by depth is recommended for nutrition-based studies because it requires a smaller sampling size for a given error limit [189,190]. Calculating SOC stocks using the mass-based approach is commonly recommended to avoid confounding effects caused by land-use- or management-related changes in bulk density over time [88,191,192,193,194,195].
6.2. Coarse Fraction
Many studies exclude the coarse soil (>2 mm) fraction of SOC, which may contain a substantial portion of organic C content [8,109,196]. The coarse soil fraction has been found to contain as much as 75% of total SOC in rocky forest soils [184]. Zabowski et al. [8] concluded that soils with a coarse fraction greater than 20% (by weight) were likely to have substantial (>10% of total SOC) organic C content in the coarse fraction. Within the 48 conterminous United States alone, skeletal (≥35% by volume rock content) soils comprise ~33% of the area [188]. Therefore, the common practice of discarding the coarse fraction of soils could result in substantial underestimations of the SOC stocks of rocky soils if the volume of soil occupied by coarse fragments is considered void (i.e., coarse fragment mass but not volume is subtracted from SOC estimates, as suggested by Throop et al. [188]) [8,109,196]. Although the coarse fraction of SOC has been historically ignored and considered chemically inert [196], Koele and Hildebrand [197] concluded that nutrient exchange mechanisms were essentially the same for both free and stone-protected fine soil (<2 mm) and that the coarse fraction contribution to short-term nutritional dynamics was substantial. This evidence suggests that we cannot assume coarse fraction SOC is inactive on decadal timescales. While it would be more labor-intensive, determining the contribution of the coarse soil fraction to SOC in rocky soils would improve the accuracy of regional and global SOC stock estimates [8,109,196].
6.3. Processing and Analysis
Soil can be processed field moist, wet, air-dried, oven-dried, or rewetted after drying. These different methods, understandably, can yield very different results. For example, air-drying soil increases the quantity or strength of organic C interactions with minerals, aggregate stability, and DOC, while decreasing microbial biomass C and altering microbial community structure [9,198,199]. Similarly, the common practice of oven-drying soil prior to analysis was shown to increase DOC concentrations [200]. Such effects should be considered and air- or oven-drying should be strictly avoided when microorganisms [201], DOC, or SOC fractions are measured or analyzed. Sieving with mesh sizes smaller than 5–6 mm can also alter soil microbial biomass and activity, as well as DOC concentrations [201,202,203]. Dissolved organic C quality and quantity analyses are further complicated by the different techniques available, including in situ and laboratory collection techniques [44,200,204] and the use of various extraction methods (e.g., cold or hot with water or a CaCl2, K2SO4, or KCl solution) [11,198,205,206], filter types [11], and pore sizes [207], all of which can lead to different results or potential artefacts.
In general, DOC extracted with different salts is more similar than DOC extracted with water, and the amount of DOC released from mineral soil is greatest for K2SO4, similar for KCl and water, and slightly lower for CaCl2 [205]. Chantigny et al. [206] and Gabor et al. [205] recommend using CaCl2 extraction in the laboratory for mineral soils to potentially limit organic matter desorption from mineral surfaces or to reduce possible artifacts and interference for fluorescence analysis. Although DOC extracted in the laboratory is not equivalent to DOC in soil solution extracted in the field by lysimeters [205], soil pore water DOC also is not perfectly represented by lysimeters [208,209]. Concerning filter type, Chantigny et al. [206] and Zsolnay [11] recommend polycarbonate filters. Unlike cellulose and acetate filters, polycarbonate filters do not appear to adsorb or release DOC [11]. Dissolved organic C is commonly extracted with a filter pore size of 0.45 µm. However, most DOC compounds are smaller than 0.025 µm, and thus pore sizes between 0.4 and 1.2 µm have resulted in DOC filtrates with similar properties [11,207]. To provide DOC properties that are more homogeneous and to reduce possible interference for fluorescence analysis, Chow et al. [207] recommend a filter pore size of 0.1 µm or smaller.
6.4. Fractionation Methods
Finally, varying methods used for physical fractionation of SOC [10]—to isolate organic C contained in differently sized aggregates, density fractions, and mineral-associations—add ambiguity to meta-analyses and limit our ability to draw overall conclusions about organic C protection mechanisms in the soil environment and in different soil types, ecosystems, and biomes. Not only can SOC fractionation provide insight into mechanisms of C cycling in soils and controls on long-term storage, but it can also reveal changes in SOC fractions over time that may not be evident in the SOC stock as a whole [100]. Although there are many viable and useful SOC fractionation techniques [10], given the essential roles that both mineral sorption [2,25,26,30,31,32,33,34,35,36,40,41] and aggregate formation [36,41,151,152,210,211] play in SOC cycling and protection, methods that include aggregate, density, and particle-size fractionation [210] will provide the most robust data, moving us toward a better understanding of SOC dynamics [145].
7. Synthesis and Future Directions
Any changes in microbial controls, root biomass and surface area, organic matter, or soil properties (e.g., temperature and moisture) can affect SOC accumulation, decomposition, and transit time—even in deeper soil layers. Unfortunately, available data on SOC in the literature is often confined to surface soil layers (above ~20 cm), which store less than half of the global SOC stock [12,13,18,25,109,119]. Deep SOC is not intrinsically “stable” and is vulnerable to potentially rapid decomposition when the environmental conditions under which it accumulated change, as the studies reviewed herein have revealed. With biological activity (and associated SOC cycling processes) often extending to at least 100 cm in depth [212,213], and with half the variation in global SOC observations to 100 cm in depth explained by modeling microbial processes [126], sampling soil to 100 cm in depth should be the standard rather than the exception. However, sampling even deeper may be necessary in certain cases, with some soils containing substantial SOC and microbial activity well below 100 cm [214,215,216,217].
The increased accuracy and precision of data gained by sampling deeper soil layers and by following a standardized methodology for SOC quality and quantity analyses would enhance our knowledge of SOC cycling and assist in reducing uncertainty in climate change projections [119]. Due to increasing SOC radiocarbon ages with depth, losses of deeper SOC result in additions of relatively long-sequestered C to the atmosphere and to a comparably short-term C cycle [3,14,15]. Further study of SOC sources and biogeochemical processes in the deep soil environment will contribute substantially to our emerging understanding of SOC protection mechanisms and vulnerability, which can provide us with key insights into climate-carbon feedbacks, as well as help us to prioritize C sequestration initiatives. Importantly, the evidence suggests that belowground C inputs (from roots and microorganisms) are the dominant source of both SOC and DOC in most ecosystems. However, the litter layer likely plays an important indirect role in maintaining SOC stocks in some forest ecosystems. Our shifting understanding of SOC “stability” has important implications for future research and for our understanding of the effects of climate, land use, and management change on SOC cycling.
Author Contributions
Conceptualization, C.D.G. and R.B.H.; investigation, C.D.G.; data curation, C.D.G.; writing—original draft preparation, C.D.G.; writing—review and editing, C.D.G.; supervision and review, R.B.H.
Funding
This research received no external funding.
Acknowledgments
Thank you to Darlene Zabowski, Eric Turnblom, Patrick Tobin, and David Butman for their feedback. We also thank the anonymous reviewers and the editor for their constructive comments that helped improve the quality of this paper.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Ciais, P.; Sabine, C.; Bala, G.; Bopp, L.; Brovkin, V.; Canadell, J.; Chhabra, A.; DeFries, R.; Galloway, J.; Heimann, M.; et al. Carbon and other biogeochemical cycles. In Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Stocker, T.F., Qin, D., Plattner, G.-K., Tignor, M., Allen, S.K., Boschung, J., Nauels, A., Xia, Y., Bex, V., Midgley, P.M., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013; pp. 465–570. ISBN 978-1-107-66182-0. [Google Scholar]
- Lehmann, J.; Kleber, M. The contentious nature of soil organic matter. Nature 2015, 528, 60–68. [Google Scholar] [CrossRef]
- Schmidt, M.W.I.; Torn, M.S.; Abiven, S.; Dittmar, T.; Guggenberger, G.; Janssens, I.A.; Kleber, M.; Kögel-Knabner, I.; Lehmann, J.; Manning, D.A.C.; et al. Persistence of soil organic matter as an ecosystem property. Nature 2011, 478, 49–56. [Google Scholar] [CrossRef]
- Fontaine, S.; Barot, S.; Barré, P.; Bdioui, N.; Mary, B.; Rumpel, C. Stability of organic carbon in deep soil layers controlled by fresh carbon supply. Nature 2007, 450, 277–280. [Google Scholar] [CrossRef]
- Kaiser, K.; Kalbitz, K. Cycling downwards—Dissolved organic matter in soils. Soil Biol. Biochem. 2012, 52, 29–32. [Google Scholar] [CrossRef]
- Sokol, N.W.; Sanderman, J.; Bradford, M.A. Pathways of mineral-associated soil organic matter formation: Integrating the role of plant carbon source, chemistry, and point of entry. Glob. Chang. Biol. 2018, 12–24. [Google Scholar] [CrossRef]
- Gross, C.D.; Harrison, R.B. Quantifying and comparing soil carbon stocks: Underestimation with the core sampling method. Soil Sci. Soc. Am. J. 2018, 82, 949. [Google Scholar] [CrossRef]
- Zabowski, D.; Whitney, N.; Gurung, J.; Hatten, J. Total soil carbon in the coarse fraction and at depth. For. Sci. 2011, 57, 11–18. [Google Scholar] [CrossRef]
- Kaiser, M.; Kleber, M.; Berhe, A.A. How air-drying and rewetting modify soil organic matter characteristics: An assessment to improve data interpretation and inference. Soil Biol. Biochem. 2015, 80, 324–340. [Google Scholar] [CrossRef]
- Moni, C.; Derrien, D.; Hatton, P.J.; Zeller, B.; Kleber, M. Density fractions versus size separates: Does physical fractionation isolate functional soil compartments? Biogeosciences 2012, 9, 5181–5197. [Google Scholar] [CrossRef]
- Zsolnay, Á. Dissolved organic matter: Artefacts, definitions, and functions. Geoderma 2003, 113, 187–209. [Google Scholar] [CrossRef]
- Batjes, N.H. Total carbon and nitrogen in the soils of the world. Eur. J. Soil Sci. 1996, 47, 151–163. [Google Scholar] [CrossRef]
- Jobbágy, E.G.; Jackson, R.B. The vertical distribution of soil organic carbon and its relation to climate and vegetation. Ecol. Appl. 2000, 10, 423–436. [Google Scholar] [CrossRef]
- Rumpel, C.; Kogel-Knabner, I.; Bruhn, F. Vertical distribution, age, and chemical composition of organic carbon in two forest soils of different pedogenesis. Org. Geochem. 2002, 33, 1131–1142. [Google Scholar] [CrossRef]
- Paul, E.A.; Follett, R.F.; Leavitt, S.W.; Halvorson, A.; Peterson, G.A.; Lyon, D.J. Radiocarbon dating for determination of soil organic matter pool sizes and dynamics. Soil Sci. Soc. Am. J. 1997, 61, 1058–1067. [Google Scholar] [CrossRef]
- Kleber, M.; Lehmann, J. Humic substances extracted by alkali are invalid proxies for the dynamics and functions of organic matter in terrestrial and aquatic ecosystems. J. Environ. Qual. 2019, 48, 207–216. [Google Scholar] [CrossRef]
- Dwivedi, D.; Riley, W.J.; Torn, M.S.; Spycher, N.; Maggi, F.; Tang, J.Y. Mineral properties, microbes, transport, and plant-input profiles control vertical distribution and age of soil carbon stocks. Soil Biol. Biochem. 2017, 107, 244–259. [Google Scholar] [CrossRef]
- Stockmann, U.; Adams, M.A.; Crawford, J.W.; Field, D.J.; Henakaarchchi, N.; Jenkins, M.; Minasny, B.; McBratney, A.B.; de Remy de Courcelles, V.; Singh, K.; et al. The knowns, known unknowns and unknowns of sequestration of soil organic carbon. Agric. Ecosyst. Environ. 2013, 164, 80–99. [Google Scholar] [CrossRef]
- Pett-Ridge, J.; Firestone, M.K. Using stable isotopes to explore root-microbe-mineral interactions in soil. Rhizosphere 2017, 3, 244–253. [Google Scholar] [CrossRef]
- Six, J.; Conant, R.T.; Paul, E.A.; Paustian, K. Stabilization mechanisms of soil organic matter: Implications for C-saturatin of soils. Plant Soil 2002, 241, 155–176. [Google Scholar] [CrossRef]
- Rasse, D.P.; Rumpel, C.; Dignac, M.F. Is soil carbon mostly root carbon? Mechanisms for a specific stabilisation. Plant Soil 2005, 269, 341–356. [Google Scholar] [CrossRef]
- Ge, Z.; Fang, S.; Chen, H.; Zhu, R.; Peng, S.; Ruan, H.; Ge, Z.; Fang, S.; Chen, H.Y.H.; Zhu, R.; et al. Soil aggregation and organic carbon dynamics in poplar plantations. Forests 2018, 9, 508. [Google Scholar] [CrossRef]
- Tisdall, J.M.; Oades, J.M. Organic matter and water stable aggregates in soil. J. Soil Sci. 1982, 33, 141–163. [Google Scholar] [CrossRef]
- Kaiser, K.; Zech, W. Dissolved organic matter sorption by mineral constituents of subsoil clay fractions. J. Plant Nutr. Soil Sci. 2000, 163, 531–535. [Google Scholar] [CrossRef]
- Angst, G.; Messinger, J.; Greiner, M.; Häusler, W.; Hertel, D.; Kirfel, K.; Kögel-Knabner, I.; Leuschner, C.; Rethemeyer, J.; Mueller, C.W. Soil organic carbon stocks in topsoil and subsoil controlled by parent material, carbon input in the rhizosphere, and microbial-derived compounds. Soil Biol. Biochem. 2018, 122, 19–30. [Google Scholar] [CrossRef]
- Lützow, M.V.; Kögel-Knabner, I.; Ekschmitt, K.; Matzner, E.; Guggenberger, G.; Marschner, B.; Flessa, H. Stabilization of organic matter in temperate soils: Mechanisms and their relevance under different soil conditions—A review. Eur. J. Soil Sci. 2006, 57, 426–445. [Google Scholar] [CrossRef]
- Guggenberger, G.; Kaiser, K. Dissolved organic matter in soil: Challenging the paradigm of sorptive preservation. Geoderma 2003, 113, 293–310. [Google Scholar] [CrossRef]
- Kaiser, K.; Eusterhues, K.; Rumpel, C.; Guggenberger, G.; Kogel-Knabner, I. Stabilization of organic matter by soil minerals—Investigation of density and particle-size fractions from two acid forest soils. J. Plant Nutr. Soil Sci. 2002, 165, 451–459. [Google Scholar] [CrossRef]
- Kleber, M.; Sollins, P.; Sutton, R. A conceptual model of organo-mineral interactions in soils: Self-assembly of organic molecular fragments into zonal structures on mineral surfaces. Biogeochemistry 2007, 85, 9–24. [Google Scholar] [CrossRef]
- Schrumpf, M.; Kaiser, K.; Guggenberger, G.; Persson, T.; Kögel-Knabner, I.; Schulze, E.D. Storage and stability of organic carbon in soils as related to depth, occlusion within aggregates, and attachment to minerals. Biogeosciences 2013, 10, 1675–1691. [Google Scholar] [CrossRef]
- Kalbitz, K.; Schwesig, D.; Rethemeyer, J.; Matzner, E. Stabilization of dissolved organic matter by sorption to the mineral soil. Soil Biol. Biochem. 2005, 37, 1319–1331. [Google Scholar] [CrossRef]
- Kaiser, K.; Guggenberger, G. The role of DOM sorption to mineral surfaces in the preservation of organic matter in soils. Org. Geochem. 2000, 31, 711–725. [Google Scholar] [CrossRef]
- Keiluweit, M.; Bougoure, J.J.; Nico, P.S.; Pett-Ridge, J.; Weber, P.K.; Kleber, M. Mineral protection of soil carbon counteracted by root exudates. Nat. Clim. Chang. 2015, 5, 588–595. [Google Scholar] [CrossRef]
- Rumpel, C.; Eusterhues, K.; Kögel-Knabner, I. Location and chemical composition of stabilized organic carbon in topsoil and subsoil horizons of two acid forest soils. Soil Biol. Biochem. 2004, 36, 177–190. [Google Scholar] [CrossRef]
- Wiesmeier, M.; Urbanski, L.; Hobley, E.; Lang, B.; von Lützow, M.; Marin-Spiotta, E.; van Wesemael, B.; Rabot, E.; Ließ, M.; Garcia-Franco, N.; et al. Soil organic carbon storage as a key function of soils—A review of drivers and indicators at various scales. Geoderma 2019, 333, 149–162. [Google Scholar] [CrossRef]
- Balabane, M.; Plante, A.F. Aggregation and carbon storage in silty soil using physical fractionation techniques. Eur. J. Soil Sci. 2004, 55, 415–427. [Google Scholar] [CrossRef]
- Jackson, R.B.; Lajtha, K.; Crow, S.E.; Hugelius, G.; Kramer, M.G. The Ecology of Soil Carbon: Pools, Vulnerabilities, and Biotic and Abiotic Controls. Annu. Rev. Ecol. Evol. Syst. 2017, 48, 419–445. [Google Scholar] [CrossRef]
- Lorenz, K.; Lal, R.; Shipitalo, M.J. Stabilized soil organic carbon pools in subsoils under forest are potential sinks for atmospheric CO2. For. Sci. 2011, 57, 19–25. [Google Scholar]
- Diochon, A.C.; Kellman, L. Physical fractionation of soil organic matter: Destabilization of deep soil carbon following harvesting of a temperate coniferous forest. J. Geophys. Res. Biogeosciences 2009, 114, 1–9. [Google Scholar] [CrossRef]
- Lopez-Sangil, L.; Rovira, P. Sequential chemical extractions of the mineral-associated soil organic matter: An integrated approach for the fractionation of organo-mineral complexes. Soil Biol. Biochem. 2013, 62, 57–67. [Google Scholar] [CrossRef]
- John, B.; Yamashita, T.; Ludwig, B.; Flessa, H. Storage of organic carbon in aggregate and density fractions of silty soils under different types of land use. Geoderma 2005, 128, 63–79. [Google Scholar] [CrossRef]
- Mathieu, J.A.; Hatte, C.; Balesdent, J.; Parent, E. Deep soil carbon dynamics are driven more by soil type than by climate: A worldwide meta-analysis of radiocarbon profiles. Glob. Chang. Biol. 2015, 21, 4278–4292. [Google Scholar] [CrossRef]
- Don, A.; Scholten, T.; Schulze, E.D. Conversion of cropland into grassland: Implications for soil organic-carbon stocks in two soils with different texture. J. Plant Nutr. Soil Sci. 2009, 172, 53–62. [Google Scholar] [CrossRef]
- Neff, J.C.; Asner, G.P. Dissolved organic carbon in terrestrial ecosystems: Synthesis and a model. Ecosystems 2001, 4, 29–48. [Google Scholar] [CrossRef]
- Bird, J.A.; Torn, M.S. Fine roots vs. needles: A comparison of 13C and 15N dynamics in a ponderosa pine forest soil. Biogeochemistry 2006, 79, 361–382. [Google Scholar] [CrossRef]
- Bird, J.A.; Kleber, M.; Torn, M.S. 13C and 15N stabilization dynamics in soil organic matter fractions during needle and fine root decomposition. Org. Geochem. 2008, 39, 465–477. [Google Scholar] [CrossRef]
- Balesdent, J.; Balabane, M. Major contribution of roots to soil carbon storage inferred from maize cultivated soils. Soil Biol. Biochem. 1996, 28, 1261–1263. [Google Scholar] [CrossRef]
- Kong, A.Y.Y.; Six, J. Tracing root vs. residue carbon into soils from conventional and alternative cropping systems. Soil Sci. Soc. Am. J. 2010, 74, 1201–1210. [Google Scholar] [CrossRef]
- Rasse, D.P.; Longdoz, B.; Ceulemans, R. TRAP: A modelling approach to below-ground carbon allocation in temperate forests. Plant Soil 2001, 229, 281–293. [Google Scholar] [CrossRef]
- Richter, D.D.; Markewitz, D.; Trumbore, S.E.; Wells, C.G. Rapid accumulation and turnover of soil carbon in a re-establishing forest. Nature 1999, 400, 56–58. [Google Scholar] [CrossRef]
- Ahrens, B.; Braakhekke, M.C.; Guggenberger, G.; Schrumpf, M.; Reichstein, M. Contribution of sorption, DOC transport and microbial interactions to the 14C age of a soil organic carbon profile: Insights from a calibrated process model. Soil Biol. Biochem. 2015, 88, 390–402. [Google Scholar] [CrossRef]
- Sanderman, J.; Baldock, J.A.; Amundson, R.; Baldock, J.A. Dissolved organic carbon chemistry and dynamics in contrasting forest and grassland soils. Biogeochemistry 2008, 89, 181–198. [Google Scholar] [CrossRef]
- McCarthy, J.F. Carbon fluxes in soil: Long-term sequestration in deeper soil horizons. J. Geogr. Sci. 2005, 15, 149–154. [Google Scholar] [CrossRef]
- Deb, S.K.; Shukla, M.K. A review of dissolved organic matter transport processes affecting soil and environmental quality. J. Environ. Anal. Toxicol. 2011, 1, 1–11. [Google Scholar] [CrossRef]
- Jardine, P.M.; Weber, N.L.; Mccarthy, J.F. Mechanisms of dissolved organic carbon adsorption on soil. Soil Sci. Soc. Am. 1989, 53, 1378–1385. [Google Scholar] [CrossRef]
- Guo, M.; Chorover, J. Transport and fractionation of dissolved organic matter in soil columns. Soil Sci. 2003, 168, 108–118. [Google Scholar] [CrossRef]
- Hedges, J.I.; Oades, J.M. Comparative organic geochemistries of soils and marine sediments. Org. Geochem. 1997, 27, 319–361. [Google Scholar] [CrossRef]
- Omoike, A.; Chorover, J. Adsorption to goethite of extracellular polymeric substances from Bacillus subtilis. Geochim. Cosmochim. Acta 2006, 70, 827–838. [Google Scholar] [CrossRef]
- Kaiser, K.; Zech, W. Competitive sorption of dissolved organic matter fractions to soils and related mineral phases. Soil Sci. Soc. Am. J. 1997, 61, 64–69. [Google Scholar] [CrossRef]
- Gleixner, G.; Poirier, N.; Bol, R.; Balesdent, J. Molecular dynamics of organic matter in a cultivated soil. Org. Geochem. 2002, 33, 357–366. [Google Scholar] [CrossRef]
- Kiem, R.; Kögel-Knabner, I. Contribution of lignin and polysaccharides to the refractory carbon pool in C-depleted arable soils. Soil Biol. Biochem. 2003, 35, 101–118. [Google Scholar] [CrossRef]
- Knicker, H. Stabilization of N-compounds in soil and organic-matter-rich sediments - What is the difference? Mar. Chem. 2004, 92, 167–195. [Google Scholar] [CrossRef]
- Barré, P.; Quénéa, K.; Vidal, A.; Cécillon, L.; Christensen, B.T.; Kätterer, T.; Macdonald, A.; Petit, L.; Plante, A.F.; van Oort, F.; et al. Microbial and plant-derived compounds both contribute to persistent soil organic carbon in temperate soils. Biogeochemistry 2018, 140, 81–92. [Google Scholar] [CrossRef]
- Kramer, M.G.; Lajtha, K.; Audfenkampe, A. Depth trends of soil organic matter C:N and 15N natural abundance controlled by association with minerals. Biogeochemistry 2017, 136, 1–12. [Google Scholar] [CrossRef]
- Hagedorn, F.; Kammer, A.; Schmidt, M.W.I.; Goodale, C.L. Nitrogen addition alters mineralization dynamics of 13C-depleted leaf and twig litter and reduces leaching of older DOC from mineral soil. Glob. Chang. Biol. 2012, 18, 1412–1427. [Google Scholar] [CrossRef]
- Qualls, R.G.; Haines, B. Biodegradability of dissolved organic matter in forest throughfall, soil solution, and stream water. Am. J. Soil Sci. 1992, 56, 578–586. [Google Scholar] [CrossRef]
- Evans, C.D.; Freeman, C.; Cork, L.G.; Thomas, D.N.; Reynolds, B.; Billett, M.F.; Garnett, M.H.; Norris, D. Evidence against recent climate-induced destabilisation of soil carbon from 14C analysis of riverine dissolved organic matter. Geophys. Res. Lett. 2007, 34, 1–5. [Google Scholar] [CrossRef]
- Schiff, S.L.; Aravena, R.; Trumbore, S.E.; Dillon, P.J. Dissolved organic carbon cycling in forested wetlands: A carbon isotope approach. Water Resour. Res. 1990, 26, 2949–2957. [Google Scholar] [CrossRef]
- Nguyen, C. Rhizodeposition of organic C by plants: Mechanisms and controls. Agronomie 2003, 23, 375–396. [Google Scholar] [CrossRef]
- Kalbitz, K.; Solinger, S.; Park, J.-H.; Michalzik, B.; Matzner, E. Controls on the dynamics of dissolved organic matter in soils: A review. Soil Sci. 2000, 165, 277–304. [Google Scholar] [CrossRef]
- Jones, D.L.; Nguyen, C.; Finlay, R.D. Carbon flow in the rhizosphere: Carbon trading at the soil-root interface. Plant Soil 2009, 321, 5–33. [Google Scholar] [CrossRef]
- Jiménez, J.J.; Decaëns, T. Vertical distribution of earthworms in grassland soils of the Colombian Llanos. Biol. Fertil. Soils 2000, 32, 463–473. [Google Scholar] [CrossRef]
- Braakhekke, M.C.; Wutzler, T.; Beer, C.; Kattge, J.; Schrumpf, M.; Ahrens, B.; Schoning, I.; Hoosbeek, M.R.; Kruijt, B.; Kabat, P.; et al. Modeling the vertical soil organic matter profile using Bayesian parameter estimation. Biogeosciences 2013, 10, 399–420. [Google Scholar] [CrossRef]
- Kalbitz, K.; Kaiser, K. Contribution of dissolved organic matter to carbon storage in forest mineral soils. J. Plant Nutr. Soil Sci. 2008, 171, 52–60. [Google Scholar] [CrossRef]
- Michalzik, B.; Tipping, E.; Mulder, J.; Gallardo-Lancho, J.F.; Matzner, E.; Bryant, C.L.; Clarke, N.; Lofts, S.; Vicente-Esteban, M.A. Modelling the production and transport of dissolved organic carbon in forest soils. Biogeochemistry 2003, 66, 241–264. [Google Scholar] [CrossRef]
- Sanderman, J.; Amundson, R. A comparative study of dissolved organic carbon transport and stabilization in California forest and grassland soils. Biogeochemistry 2009, 92, 41–59. [Google Scholar] [CrossRef]
- McDowell, W.H. Dissolved organic matter in soils—future directions and unanswered questions. Geoderma 2003, 113, 179–186. [Google Scholar] [CrossRef]
- Rumpel, C.; Kögel-Knabner, I. Deep soil organic matter-a key but poorly understood component of terrestrial C cycle. Plant Soil 2011, 338, 143–158. [Google Scholar] [CrossRef]
- Derrien, D.; Marol, C.; Balesdent, J. The dynamics of neutral sugars in the rhizosphere of wheat. An approach by 13C pulse-labelling and GC/C/IRMS. Plant Soil 2004, 267, 243–253. [Google Scholar] [CrossRef]
- Lambers, H. Growth, respiration, exudation and symbiotic associations: The fate of carbon translocated to the root. In Root Develpment and Function. Soc. Exp. Biol. Seminar Ser. 30; Gregory, P.J., Lake, J.V., Rose, D.A., Eds.; Cambridge University Press: Cambridge, UK, 1987; pp. 124–145. [Google Scholar]
- Sokol, N.W.; Bradford, M.A. Microbial formation of stable soil carbon is more efficient from belowground than aboveground input. Nat. Geosci. 2019, 12, 46–53. [Google Scholar] [CrossRef]
- Wallander, H.; Göransson, H.; Rosengren, U. Production, standing biomass and natural abundance of 15N and 13C in ectomycorrhizal mycelia collected at different soil depths in two forest types. Oecologia 2004, 139, 89–97. [Google Scholar] [CrossRef]
- Clemmensen, K.E.; Bahr, A.; Ovaskainen, O.; Dahlberg, A.; Ekblad, A.; Wallander, H.; Stenlid, J.; Finlay, R.D.; Wardle, D.A.; Lindahl, B.D. Roots and associated fungi drive long-term carbon sequestration in boreal forest. Science 2013, 339, 1615–1618. [Google Scholar] [CrossRef]
- Godbold, D.L.; Hoosbeek, M.R.; Lukac, M.; Cotrufo, M.F.; Janssens, I.A.; Ceulemans, R.; Polle, A.; Velthorst, E.J.; Scarascia-Mugnozza, G.; De Angelis, P.; et al. Mycorrhizal hyphal turnover as a dominant process for carbon input into soil organic matter. Plant Soil 2006, 281, 15–24. [Google Scholar] [CrossRef]
- Fekete, I.; Kotroczó, Z.; Varga, C.; Nagy, P.T.; Várbíró, G.; Bowden, R.D.; Tóth, J.A.; Lajtha, K. Alterations in forest detritus inputs influence soil carbon concentration and soil respiration in a Central-European deciduous forest. Soil Biol. Biochem. 2014, 74, 106–114. [Google Scholar] [CrossRef]
- Bowden, R.D.; Deem, L.; Plante, A.F.; Peltre, C.; Nadelhoffer, K.; Lajtha, K. Litter input controls on soil carbon in a temperate deciduous forest. Soil Sci. Soc. Am. J. 2014, 78, S66. [Google Scholar] [CrossRef]
- Lajtha, K.; Townsend, K.L.; Kramer, M.G.; Swanston, C.; Bowden, R.D.; Nadelhoffer, K. Changes to particulate versus mineral-associated soil carbon after 50 years of litter manipulation in forest and prairie experimental ecosystems. Biogeochemistry 2014, 119, 341–360. [Google Scholar] [CrossRef]
- Wendt, J.W.; Hauser, S. An equivalent soil mass procedure for monitoring soil organic carbon in multiple soil layers. Eur. J. Soil Sci. 2013, 64, 58–65. [Google Scholar] [CrossRef]
- Leff, J.W.; Wieder, W.R.; Taylor, P.G.; Townsend, A.R.; Nemergut, D.R.; Grandy, A.S.; Cleveland, C.C. Experimental litterfall manipulation drives large and rapid changes in soil carbon cycling in a wet tropical forest. Glob. Chang. Biol. 2012, 18, 2969–2979. [Google Scholar] [CrossRef]
- Xu, S.; Liu, L.L.; Sayer, E.J. Variability of above-ground litter inputs alters soil physicochemical and biological processes: A meta-analysis of litterfall-manipulation experiments. Biogeosciences 2013, 10, 7423–7433. [Google Scholar] [CrossRef]
- Wang, J.J.; Pisani, O.; Lin, L.H.; Lun, O.O.Y.; Bowden, R.D.; Lajtha, K.; Simpson, A.J.; Simpson, M.J. Long-term litter manipulation alters soil organic matter turnover in a temperate deciduous forest. Sci. Total Environ. 2017, 607–608, 865–875. [Google Scholar] [CrossRef]
- Mendez-Millan, M.; Dignac, M.F.; Rumpel, C.; Rasse, D.P.; Derenne, S. Molecular dynamics of shoot vs. root biomarkers in an agricultural soil estimated by natural abundance 13C labelling. Soil Biol. Biochem. 2010, 42, 169–177. [Google Scholar] [CrossRef]
- Grayston, S.J.; Vaughan, D.; Jones, D. Rhizosphere carbon flow in trees, in comparison with annual plants: The importance of root exudation and its impact on microbial activity and nutrient availability. Appl. Soil Ecol. 1997, 5, 29–56. [Google Scholar] [CrossRef]
- Liang, C.; Balser, T.C. Microbial production of recalcitrant organic matter in global soils: Implications for productivity and climate policy. Nat. Rev. Microbiol. 2010, 9, 75. [Google Scholar] [CrossRef]
- Kramer, C.; Trumbore, S.; Fröberg, M.; Cisneros Dozal, L.M.; Zhang, D.; Xu, X.; Santos, G.M.; Hanson, P.J. Recent (<4 year old) leaf litter is not a major source of microbial carbon in a temperate forest mineral soil. Soil Biol. Biochem. 2010, 42, 1028–1037. [Google Scholar] [CrossRef]
- Liang, B.C.; Wang, X.L.; Ma, B.L. Maize root-induced change in soil organic carbon pools. Soil Sci. Soc. Am. J. 2002, 66, 845–847. [Google Scholar] [CrossRef]
- Riley, W.J.; Maggi, F.; Kleber, M.; Torn, M.S.; Tang, J.Y.; Dwivedi, D.; Guerry, N. Long residence times of rapidly decomposable soil organic matter: Application of a multi-phase, multi-component, and vertically resolved model (BAMS1) to soil carbon dynamics. Geosci. Model Dev. 2014, 7, 1335–1355. [Google Scholar] [CrossRef]
- Liang, C.; Balser, T.C. Preferential sequestration of microbial carbon in subsoils of a glacial-landscape toposequence, Dane County, WI, USA. Geoderma 2008, 148, 113–119. [Google Scholar] [CrossRef]
- Creamer, C.A.; Jones, D.L.; Baldock, J.A.; Rui, Y.; Murphy, D.V.; Hoyle, F.C.; Farrell, M. Is the fate of glucose-derived carbon more strongly driven by nutrient availability, soil texture, or microbial biomass size? Soil Biol. Biochem. 2016, 103, 201–212. [Google Scholar] [CrossRef]
- Vogel, C.; Heister, K.; Buegger, F.; Tanuwidjaja, I.; Haug, S.; Schloter, M.; Kögel-Knabner, I. Clay mineral composition modifies decomposition and sequestration of organic carbon and nitrogen in fine soil fractions. Biol. Fertil. Soils 2015, 51, 427–442. [Google Scholar] [CrossRef]
- Liang, C.; Schimel, J.P.; Jastrow, J.D. The importance of anabolism in microbial control over soil carbon storage. Nat. Microbiol. 2017, 2, 1–6. [Google Scholar] [CrossRef]
- Michalzik, B.; Kalbitz, K.; Park, J.; Solinger, S.; Matzner, E. Fluxes and concentrations of dissolved organic carbon and nitrogen–a synthesis for temperate forests. Biogeochemistry 2001, 52, 173–205. [Google Scholar] [CrossRef]
- Jackson, R.B.; Canadell, J.; Ehleringer, J.R.; Mooney, H.A.; Sala, O.E.; Schulze, E.D. A global analysis of root distributions for terrestrial biomes. Oecologia 1996, 108, 389–411. [Google Scholar] [CrossRef]
- Sun, L.; Ataka, M.; Kominami, Y.; Yoshimura, K. Relationship between fine-root exudation and respiration of two Quercus species in a Japanese temperate forest. Tree Physiol. 2017, 37, 1011–1020. [Google Scholar] [CrossRef]
- Gabor, R.S.; Eilers, K.; McKnight, D.M.; Fierer, N.; Anderson, S.P. From the litter layer to the saprolite: Chemical changes in water-soluble soil organic matter and their correlation to microbial community composition. Soil Biol. Biochem. 2014, 68, 166–176. [Google Scholar] [CrossRef]
- Wang, Q.; He, T.; Wang, S.; Liu, L. Carbon input manipulation affects soil respiration and microbial community composition in a subtropical coniferous forest. Agric. For. Meteorol. 2013, 178–179, 152–160. [Google Scholar] [CrossRef]
- Gaudinski, J.B.; Trumbore, S.E.; Davidson, E.A.; Cook, A.C.; Markewitz, D.; Richter, D.D. The age of fine-root carbon in three forests of the eastern United States measured by radiocarbon. Oecologia 2001, 129, 420–429. [Google Scholar] [CrossRef]
- Sierra, C.A.; Müller, M.; Metzler, H.; Manzoni, S.; Trumbore, S.E. The muddle of ages, turnover, transit, and residence times in the carbon cycle. Glob. Chang. Biol. 2017, 23, 1763–1773. [Google Scholar] [CrossRef]
- Harrison, R.B.; Footen, P.W.; Strahm, B.D. Deep soil horizons: Contribution and importance to soil carbon pools and in assessing whole-ecosystem response to management and global change. For. Sci. 2011, 57, 67–76. [Google Scholar]
- Bernal, B.; McKinley, D.C.; Hungate, B.A.; White, P.M.; Mozdzer, T.J.; Megonigal, J.P. Limits to soil carbon stability; Deep, ancient soil carbon decomposition stimulated by new labile organic inputs. Soil Biol. Biochem. 2016, 98, 85–94. [Google Scholar] [CrossRef]
- Karhu, K.; Hilasvuori, E.; Fritze, H.; Biasi, C.; Nykänen, H.; Liski, J.; Vanhala, P.; Heinonsalo, J.; Pumpanen, J. Priming effect increases with depth in a boreal forest soil. Soil Biol. Biochem. 2016, 99, 104–107. [Google Scholar] [CrossRef]
- Bernal, B.; Megonigal, J.P.; Mozdzer, T.J. An invasive wetland grass primes deep soil carbon pools. Glob. Chang. Biol. 2017, 23, 2104–2116. [Google Scholar] [CrossRef]
- Hicks Pries, C.E.; Schuur, E.A.G.; Natali, S.M.; Crummer, K.G. Old soil carbon losses increase with ecosystem respiration in experimentally thawed tundra. Nat. Clim. Chang. 2016, 6, 214–218. [Google Scholar] [CrossRef]
- Hicks Pries, C.E.; Castanha, C.; Porras, R.; Torn, M.S. The whole-soil carbon flux in response to warming. Science 2017, 355, 1420–1423. [Google Scholar] [CrossRef]
- Mobley, M.L.; Lajtha, K.; Kramer, M.G.; Bacon, A.R.; Heine, P.R.; Richter, D.D. Surficial gains and subsoil losses of soil carbon and nitrogen during secondary forest development. Glob. Chang. Biol. 2015, 21, 986–996. [Google Scholar] [CrossRef]
- Tian, Q.; Yang, X.; Wang, X.; Liao, C.; Li, Q.; Wang, M.; Wu, Y.; Liu, F. Microbial community mediated response of organic carbon mineralization to labile carbon and nitrogen addition in topsoil and subsoil. Biogeochemistry 2016, 128, 125–139. [Google Scholar] [CrossRef]
- Shahzad, T.; Anwar, F.; Hussain, S.; Mahmood, F.; Arif, M.S.; Sahar, A.; Nawaz, M.F.; Perveen, N.; Sanaullah, M.; Rehman, K.; et al. Carbon dynamics in surface and deep soil in response to increasing litter addition rates in an agro-ecosystem. Geoderma 2018, 333, 1–9. [Google Scholar] [CrossRef]
- Scharlemann, J.P.; Tanner, E.V.; Hiederer, R.; Kapos, V. Global soil carbon: Understanding and managing the largest terrestrial carbon pool. Carbon Manag. 2014, 5, 81–91. [Google Scholar] [CrossRef]
- Jandl, R.; Rodeghiero, M.; Martinez, C.; Cotrufo, M.F.; Bampa, F.; van Wesemael, B.; Harrison, R.B.; Guerrini, I.A.; deB Richter, D.; Rustad, L.; et al. Current status, uncertainty and future needs in soil organic carbon monitoring. Sci. Total Environ. 2014, 468–469, 376–383. [Google Scholar] [CrossRef]
- Balesdent, J.; Basile-Doelsch, I.; Chadoeuf, J.; Cornu, S.; Derrien, D.; Fekiacova, Z.; Hatté, C. Atmosphere–soil carbon transfer as a function of soil depth. Nature 2018, 559, 599–602. [Google Scholar] [CrossRef]
- Trumbore, S.E.; Czimczik, C.I. An uncertain future for soil carbon. Science 2008, 321, 1455–1456. [Google Scholar] [CrossRef]
- Sombroek, W.G.; Nachtergaele, F.O.; Hebel, A. Amounts, dynamics and sequestering of carbon in tropical and subtropical soils. Ambio 1993, 22, 417–426. [Google Scholar]
- Post, W.M.; Emanuel, W.R.; Zinke, P.J.; Stangenberger, A.G. Soil carbon pools and world life zones. Nature 1982, 298, 156–159. [Google Scholar] [CrossRef]
- Eswaran, H.; Van Den Berg, E.; Reich, P. Organic carbon in soils of the world. Soil Sci. Soc. Am. J. 1993, 57, 192–194. [Google Scholar] [CrossRef]
- Köchy, M.; Hiederer, R.; Freibauer, A. Global distribution of soil organic carbon – Part 1: Masses and frequency distributions of SOC stocks for the tropics, permafrost regions, wetlands, and the world. Soil 2015, 1, 351–365. [Google Scholar] [CrossRef]
- Wieder, W.R.; Bonan, G.B.; Allison, S.D. Global soil carbon projections are improved by modelling microbial processes. Nat. Clim. Chang. 2013, 3, 909–912. [Google Scholar] [CrossRef]
- Osher, L.J.; Matson, P.A.; Amundson, R. Effect of land use change on soil organic carbon. Biogeochemistry 2003, 65, 213–232. [Google Scholar] [CrossRef]
- Collins, M.; Knutti, R.; Arblaster, J.; Dufresne, J.-L.; Fichefet, T.; Friedlingstein, P.; Gao, X.; Gutowski, W.J.; Johns, T.; Krinner, G.; et al. Long-term climate change: Projections, commitments and irreversibility. In Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Stocker, T.F., Qin, D., Plattner, G.-K., Tignor, M., Allen, S.K., Boschung, J., Nauels, A., Xia, Y., Bex, V., Midgley, P.M., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013; pp. 1029–1136. ISBN 9781107415324. [Google Scholar]
- Brady, N.C.; Weil, R.R. The Nature and Properties of Soils, 14th ed.; Pearson: Upper Saddle River, NJ, USA, 2008. [Google Scholar]
- Hicks Pries, C.E.; Sulman, B.N.; West, C.; O’Neill, C.; Poppleton, E.; Porras, R.C.; Castanha, C.; Zhu, B.; Wiedemeier, D.B.; Torn, M.S. Root litter decomposition slows with soil depth. Soil Biol. Biochem. 2018, 125, 103–114. [Google Scholar] [CrossRef]
- Nieminen, M. Export of dissolved organic carbon, nitrogen and phosphorus following clear-cutting of three Norway spruce forests growing on drained peatlands in southern Finland. Silva Fenn. 2004, 38, 123–132. [Google Scholar] [CrossRef]
- Huo, C.; Luo, Y.; Cheng, W. Rhizosphere priming effect: A meta-analysis. Soil Biol. Biochem. 2017, 111, 78–84. [Google Scholar] [CrossRef]
- Shahzad, T.; Chenu, C.; Genet, P.; Barot, S.; Perveen, N.; Mougin, C.; Fontaine, S. Contribution of exudates, arbuscular mycorrhizal fungi and litter depositions to the rhizosphere priming effect induced by grassland species. Soil Biol. Biochem. 2015, 80, 146–155. [Google Scholar] [CrossRef]
- Mason-Jones, K.; Schmücker, N.; Kuzyakov, Y. Contrasting effects of organic and mineral nitrogen challenge the N-Mining Hypothesis for soil organic matter priming. Soil Biol. Biochem. 2018, 124, 38–46. [Google Scholar] [CrossRef]
- Shahzad, T.; Rashid, M.I.; Maire, V.; Barot, S.; Perveen, N.; Alvarez, G.; Mougin, C.; Fontaine, S. Root penetration in deep soil layers stimulates mineralization of millennia-old organic carbon. Soil Biol. Biochem. 2018, 124, 150–160. [Google Scholar] [CrossRef]
- Pausch, J.; Zhu, B.; Kuzyakov, Y.; Cheng, W. Plant inter-species effects on rhizosphere priming of soil organic matter decomposition. Soil Biol. Biochem. 2013, 57, 91–99. [Google Scholar] [CrossRef]
- Zhu, B.; Gutknecht, J.L.M.; Herman, D.J.; Keck, D.C.; Firestone, M.K.; Cheng, W. Rhizosphere priming effects on soil carbon and nitrogen mineralization. Soil Biol. Biochem. 2014, 76, 183–192. [Google Scholar] [CrossRef]
- Dijkstra, F.A.; Cheng, W. Interactions between soil and tree roots accelerate long-term soil carbon decomposition. Ecol. Lett. 2007, 10, 1046–1053. [Google Scholar] [CrossRef]
- Kuzyakov, Y. Priming effects: Interactions between living and dead organic matter. Soil Biol. Biochem. 2010, 42, 1363–1371. [Google Scholar] [CrossRef]
- Phillips, R.P.; Meier, I.C.; Bernhardt, E.S.; Grandy, A.S.; Wickings, K.; Finzi, A.C. Roots and fungi accelerate carbon and nitrogen cycling in forests exposed to elevated CO2. Ecol. Lett. 2012, 15, 1042–1049. [Google Scholar] [CrossRef]
- Burns, R.G.; DeForest, J.L.; Marxsen, J.; Sinsabaugh, R.L.; Stromberger, M.E.; Wallenstein, M.D.; Weintraub, M.N.; Zoppini, A. Soil enzymes in a changing environment: Current knowledge and future directions. Soil Biol. Biochem. 2013, 58, 216–234. [Google Scholar] [CrossRef]
- Finzi, A.C.; Abramoff, R.Z.; Spiller, K.S.; Brzostek, E.R.; Darby, B.A.; Kramer, M.A.; Phillips, R.P. Rhizosphere processes are quantitatively important components of terrestrial carbon and nutrient cycles. Glob. Chang. Biol. 2015, 21, 2082–2094. [Google Scholar] [CrossRef]
- Poirier, V.; Roumet, C.; Munson, A.D. The root of the matter: Linking root traits and soil organic matter stabilization processes. Soil Biol. Biochem. 2018, 120, 246–259. [Google Scholar] [CrossRef]
- Liang, Z.; Elsgaard, L.; Nicolaisen, M.H.; Lyhne-Kjærbye, A.; Olesen, J.E. Carbon mineralization and microbial activity in agricultural topsoil and subsoil as regulated by root nitrogen and recalcitrant carbon concentrations. Plant Soil 2018, 433, 65–82. [Google Scholar] [CrossRef]
- Sulman, B.N.; Moore, J.A.M.; Abramoff, R.; Averill, C.; Kivlin, S.; Georgiou, K.; Sridhar, B.; Hartman, M.D.; Wang, G.; Wieder, W.R.; et al. Multiple models and experiments underscore large uncertainty in soil carbon dynamics. Biogeochemistry 2018, 141, 109–123. [Google Scholar] [CrossRef]
- Carrillo, Y.; Bell, C.; Koyama, A.; Canarini, A.; Boot, C.M.; Wallenstein, M.; Pendall, E. Plant traits, stoichiometry and microbes as drivers of decomposition in the rhizosphere in a temperate grassland. J. Ecol. 2017. [Google Scholar] [CrossRef]
- Koranda, M.; Schnecker, J.; Kaiser, C.; Fuchslueger, L.; Kitzler, B.; Stange, C.F.; Sessitsch, A.; Zechmeister-Boltenstern, S.; Richter, A. Microbial processes and community composition in the rhizosphere of European beech—The influence of plant C exudates. Soil Biol. Biochem. 2011, 43, 551–558. [Google Scholar] [CrossRef] [PubMed]
- Blagodatskaya, E.V.; Blagodatsky, S.A.; Anderson, T.H.; Kuzyakov, Y. Priming effects in Chernozem induced by glucose and N in relation to microbial growth strategies. Appl. Soil Ecol. 2007, 37, 95–105. [Google Scholar] [CrossRef]
- De Graaff, M.-A.; Classen, A.T.; Castro, H.F.; Schadt, C.W. Labile soil carbon inputs mediate the soil microbial community composition and plant residue decomposition rates. New Phytol. 2010, 188, 1055–1064. [Google Scholar] [CrossRef]
- Li, L.J.; Zhu-Barker, X.; Ye, R.; Doane, T.A.; Horwath, W.R. Soil microbial biomass size and soil carbon influence the priming effect from carbon inputs depending on nitrogen availability. Soil Biol. Biochem. 2018, 119, 41–49. [Google Scholar] [CrossRef]
- Tian, J.; Pausch, J.; Yu, G.; Blagodatskaya, E.; Gao, Y.; Kuzyakov, Y. Aggregate size and their disruption affect 14C-labeled glucose mineralization and priming effect. Appl. Soil Ecol. 2015, 90, 1–10. [Google Scholar] [CrossRef]
- Tian, J.; Pausch, J.; Yu, G.; Blagodatskaya, E.; Kuzyakov, Y. Aggregate size and glucose level affect priming sources: A three-source-partitioning study. Soil Biol. Biochem. 2016, 97, 199–210. [Google Scholar] [CrossRef]
- Guo, L.B.; Halliday, M.J.; Siakimotu, S.J.M.; Gifford, R.M. Fine root production and litter input: Its effects on soil carbon. Plant Soil 2005, 272, 1–10. [Google Scholar] [CrossRef]
- Hobley, E.; Baldock, J.; Hua, Q.; Wilson, B. Land-use contrasts reveal instability of subsoil organic carbon. Glob. Chang. Biol. 2017, 23, 955–965. [Google Scholar] [CrossRef]
- Guo, L.B.; Gifford, R.M. Soil carbon stocks and land use change: A meta analysis. Glob. Chang. Biol. 2002, 8, 345–360. [Google Scholar] [CrossRef]
- James, J.; Harrison, R. The effect of harvest on forest soil carbon: A meta-analysis. Forests 2016, 7, 308. [Google Scholar] [CrossRef]
- Gross, C.D.; James, J.N.; Turnblom, E.C.; Harrison, R.B. Thinning treatments reduce deep soil carbon and nitrogen stocks in a coastal Pacific Northwest forest. Forests 2018, 9, 238. [Google Scholar] [CrossRef]
- Clarke, N.; Gundersen, P.; Jönsson-Belyazid, U.; Kjønaas, O.J.; Persson, T.; Sigurdsson, B.D.; Stupak, I.; Vesterdal, L. Influence of different tree-harvesting intensities on forest soil carbon stocks in boreal and northern temperate forest ecosystems. For. Ecol. Manag. 2015, 351, 9–19. [Google Scholar] [CrossRef]
- Gallo, A.C. Response of Soil Temperature, Moisture, and Respiration Two Years Following Intensive Organic Matter and Compaction Manipulations in Oregon Cascade Forests. Master’s Thesis, Oregon State University, Corvallis, OR, USA, 2016. [Google Scholar]
- Butman, D.E.; Wilson, H.F.; Barnes, R.T.; Xenopoulos, M.A.; Raymond, P.A. Increased mobilization of aged carbon to rivers by human disturbance. Nat. Geosci. 2015, 8, 112–116. [Google Scholar] [CrossRef]
- Hagedorn, F.; Bundt, M. The age of preferential flow paths. Geoderma 2002, 108, 119–132. [Google Scholar] [CrossRef]
- Bundt, M.; Widmer, F.; Pesaro, M.; Zeyer, J.; Blaser, P. Preferential flow paths: Biological “hot spots” in soils. Soil Biol. Biochem. 2001, 33, 729–738. [Google Scholar] [CrossRef]
- Kuzyakov, Y.; Friedelb, J.K.; Stahra, K. Review of mechanisms and quantification of priming effects. Soil Biol. Biochem. 2000, 32, 1485–1498. [Google Scholar] [CrossRef]
- Bradford, M.A.; Wieder, W.R.; Bonan, G.B.; Fierer, N.; Raymond, P.A.; Crowther, T.W. Managing uncertainty in soil carbon feedbacks to climate change. Nat. Clim. Chang. 2016, 6, 751–758. [Google Scholar] [CrossRef]
- Crowther, T.; Todd-Brown, K.; Rowe, C.; Wieder, W.; Carey, J.; Machmuller, M.; Snoek, L.; Fang, S.; Zhou, G.; Allison, S.; et al. Quantifying global soil C losses in response to warming. Nature 2016, 104, 104–108. [Google Scholar] [CrossRef]
- Tarnocai, C.; Canadell, J.G.; Schuur, E.A.G.; Kuhry, P.; Mazhitova, G.; Zimov, S. Soil organic carbon pools in the northern circumpolar permafrost region. Glob. Biogeochem. Cycles 2009, 23, 1–11. [Google Scholar] [CrossRef]
- Davidson, E.A.; Janssens, I.A. Temperature sensitivity of soil carbon decomposition and feedbacks to climate change. Nature 2006, 440, 165–173. [Google Scholar] [CrossRef]
- Anisimov, O.; Reneva, S. Permafrost and changing climate: The Russian perspective. Ambio 2006, 35, 169–175. [Google Scholar] [CrossRef]
- Bridgham, S.D.; Megonigal, J.P.; Keller, J.K.; Bliss, N.B.; Trettin, C. The carbon balance of North American wetlands. Wetlands 2006, 26, 889–916. [Google Scholar] [CrossRef]
- Gorham, E. Northern peatlands: Role in the carbon cycle and probable responses to climatic warming. Ecol. Appl. 1991, 1, 182–195. [Google Scholar] [CrossRef]
- Maltby, E.; Immirzi, P. Carbon dynamics in peatlands and other wetland soils regional and global perspectives. Chemosphere 1993, 27, 999–1023. [Google Scholar] [CrossRef]
- Wetherald, R.T.; Manabe, S. Detectability of summer dryness caused by greenhouse warming. Clim. Chang. 1999, 43, 495–511. [Google Scholar] [CrossRef]
- Le Mer, J.; Roger, P. Production, oxidation, emission and consumption of methane by soils: A review. Eur. J. Soil Biol. 2001, 37, 25–50. [Google Scholar] [CrossRef]
- Bellassen, V.; Luyssaert, S. Carbon sequestration: Managing forests in uncertain times. Nature 2014, 506, 153–155. [Google Scholar] [CrossRef]
- Pan, Y.; Birdsey, R.A.; Fang, J.; Houghton, R.; Kauppi, P.E.; Kurz, W.A.; Phillips, O.L.; Shvidenko, A.; Lewis, S.L.; Canadell, J.G.; et al. A large and persistent carbon sink in the world’s forests. Science 2011, 333, 988–993. [Google Scholar] [CrossRef]
- Nave, L.E.; Vance, E.D.; Swanston, C.W.; Curtis, P.S. Harvest impacts on soil carbon storage in temperate forests. For. Ecol. Manag. 2010, 259, 857–866. [Google Scholar] [CrossRef]
- Schimel, D. Terrestrial ecosystems and the carbon cycle. Glob. Chang. Biol. 1995, 1, 77–91. [Google Scholar] [CrossRef]
- Lal, R. Forest soils and carbon sequestration. For. Ecol. Manag. 2005, 220, 242–258. [Google Scholar] [CrossRef]
- Jones, C.; McConnell, C.; Coleman, K.; Cox, P.; Falloon, P.; Jenkinson, D.; Powlson, D. Global climate change and soil carbon stocks; predictions from two contrasting models for the turnover of organic carbon in soil. Glob. Chang. Biol. 2005, 11, 154–166. [Google Scholar] [CrossRef]
- Song, W.; Tong, X.; Zhang, J.; Meng, P. Three-source partitioning of soil respiration by 13C natural abundance and its variation with soil depth in a plantation. J. For. Res. 2016, 27, 533–540. [Google Scholar] [CrossRef]
- Sulman, B.N.; Phillips, R.P.; Oishi, A.C.; Shevliakova, E.; Pacala, S.W. Microbe-driven turnover offsets mineral-mediated storage of soil carbon under elevated CO2. Nat. Clim. Chang. 2014, 4, 1099–1102. [Google Scholar] [CrossRef]
- Andraski, B.J. Balloon and core sampling for determining bulk density of alluvial desert soil. Soil Sci. Soc. Am. J. 1991, 55, 1188–1190. [Google Scholar] [CrossRef]
- Muller, R.N.; Hamilton, M.E. A simple, effective method for determining the bulk density of stony soils. Commun. Soil Sci. Plant Anal. 1992, 23, 313–319. [Google Scholar] [CrossRef]
- Harrison, R.B.; Adams, A.B.; Licata, C.; Flaming, B.; Wagoner, G.L.; Carpenter, P.; Vance, E.D. Quantifying deep-soil and coarse-soil fractions: Avoiding sampling bias. Soil Sci. Soc. Am. J. 2003, 67, 1602–1606. [Google Scholar] [CrossRef]
- Kulmatiski, A.; Vogt, D.J.; Siccama, T.G.; Beard, K.H. Detecting nutrient pool changes in rocky forest soils. Soil Sci. Soc. Am. J 2003, 67, 1282–1286. [Google Scholar] [CrossRef]
- Beem-Miller, J.P.; Kong, A.Y.Y.; Ogle, S.; Wolfe, D. Sampling for soil carbon stock assessment in rocky agricultural soils. Soil Sci. Soc. Am. J. 2016, 80, 1411. [Google Scholar] [CrossRef]
- Hobley, E.U.; Murphy, B.; Simmons, A. Comment on “Soil organic stocks are systematically overestimated by misuse of the parameters bulk density and rock fragment content” by Poeplau et al. (2017). Soil 2018, 4, 169–171. [Google Scholar] [CrossRef]
- Throop, H.L.; Archer, S.R.; Monger, H.C.; Waltman, S. When bulk density methods matter: Implications for estimating soil organic carbon pools in rocky soils. J. Arid Environ. 2012, 77, 66–71. [Google Scholar] [CrossRef]
- Grüneberg, E.; Schöning, I.; Kalko, E.K.V.; Weisser, W.W. Regional organic carbon stock variability: A comparison between depth increments and soil horizons. Geoderma 2010, 155, 426–433. [Google Scholar] [CrossRef]
- Wang, H.; Wang, W.; Chang, S.X. Sampling method and tree-age affect soil organic C and N contents in larch plantations. Forests 2017, 8, 28. [Google Scholar] [CrossRef]
- Ellert, B.H.; Bettany, J.R. Calculation of organic matter and nutrients stored in soils under contrasting management regimes. Can. J. Soil Sci. 1995, 75, 529–538. [Google Scholar] [CrossRef]
- Lee, J.; Hopmans, J.W.; Rolston, D.E.; Baer, S.G.; Six, J. Determining soil carbon stock changes: Simple bulk density corrections fail. Agric. Ecosyst. Environ. 2009, 134, 251–256. [Google Scholar] [CrossRef]
- Wuest, S.B. Correction of bulk density and sampling method biases using soil mass per unit area. Soil Sci. Soc. Am. J. 2009, 73, 312. [Google Scholar] [CrossRef]
- Schrumpf, M.; Schulze, E.D.; Kaiser, K.; Schumacher, J. How accurately can soil organic carbon stocks and stock changes be quantified by soil inventories? Biogeosciences 2011, 8, 1193–1212. [Google Scholar] [CrossRef]
- Mikha, M.M.; Benjamin, J.G.; Halvorson, A.D.; Nielsen, D.C. Soil carbon changes influenced by soil management and calculation method. Open J. Soil Sci. 2013, 3, 123–131. [Google Scholar] [CrossRef]
- Corti, G.; Ugolini, F.C.; Agnelli, A.; Certini, G.; Cuniglio, R.; Berna, F.; Fernández Sanjurjo, M.J. The soil skeleton, a forgotten pool of carbon and nitrogen in soil. Eur. J. Soil Sci. 2002, 53, 283–298. [Google Scholar] [CrossRef]
- Koele, N.; Hildebrand, E.E. The ecological significance of the coarse soil fraction for Picea abies (L.) Karst. seedling nutrition. Plant Soil 2008, 312, 163–174. [Google Scholar] [CrossRef]
- Sun, S.Q.; Cai, H.Y.; Chang, S.X.; Bhatti, J.S. Sample storage-induced changes in the quantity and quality of soil labile organic carbon. Sci. Rep. 2015, 5, 1–10. [Google Scholar] [CrossRef]
- Blaud, A.; Menon, M.; van der Zaan, B.; Lair, G.J.; Banwart, S.A. Effects of Dry and Wet Sieving of Soil on Identification and Interpretation of Microbial Community Composition, 1st ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2017; Volume 142, ISBN 9780128122228. [Google Scholar]
- Koopmans, G.F.; Groenenberg, J.E. Effects of soil oven-drying on concentrations and speciation of trace metals and dissolved organic matter in soil solution extracts of sandy soils. Geoderma 2011, 161, 147–158. [Google Scholar] [CrossRef]
- Horwath, W.R.; Paul, E.A. Microbial Biomass. In Methods of Soil Analysis: Part 2—Microbiological and Biochemical Properties, SSSA Book Series no. 5; Bottomley, P.S., Angle, J.S., Weaver, R.W., Eds.; Soil Science Society of America: Madison, WI, USA, 1994; pp. 753–774. [Google Scholar]
- Datta, R.; Vranová, V.; Pavelka, M.; Rejšek, K.; Formánek, P. Effect of soil sieving on respiration induced by low-molecular-weight substrates. Int. Agrophysics 2014, 28, 119–124. [Google Scholar] [CrossRef]
- Ross, D.J. Influence of Sieve Mesh Size on Estimates of Microbial Carbon and Nitrogen by Fumigation Extraction Procedures in Soils under Pasture. Soil Biol. Biochem. 1992, 24, 343–350. [Google Scholar] [CrossRef]
- De Feudis, M.; Cardelli, V.; Massaccesi, L.; Hofmann, D.; Berns, A.E.; Bol, R.; Cocco, S.; Corti, G.; Agnelli, A. Altitude affects the quality of the water-extractable organic matter (WEOM) from rhizosphere and bulk soil in European beech forests. Geoderma 2017, 302, 6–13. [Google Scholar] [CrossRef]
- Gabor, R.S.; Burns, M.A.; Lee, R.H.; Elg, J.B.; Kemper, C.J.; Barnard, H.R.; McKnight, D.M. Influence of leaching solution and catchment location on the fluorescence of water-soluble organic matter. Environ. Sci. Technol. 2015, 49, 4425–4432. [Google Scholar] [CrossRef]
- Chantigny, M.H.; Angers, D.A.; Kaiser, K.; Kalbitz, K. Extraction and characterization of dissolved organic matter. In Soil Sampling and Methods of Analysis; Carter, M.R., Gregorich, E.G., Eds.; CRC Press: Boca Raton, FL, USA, 2007; pp. 617–635. ISBN 9781420005271. [Google Scholar]
- Chow, A.T.; Guo, F.; Gao, S.; Breuer, R.; Dahlgren, R.A. Filter pore size selection for characterizing dissolved organic carbon and trihalomethane precursors from soils. Water Res. 2005, 39, 1255–1264. [Google Scholar] [CrossRef]
- Perdrial, J.N.; Perdrial, N.; Harpold, A.; Gao, X.; Gabor, R.; LaSharr, K.; Chorover, J. Impacts of sampling dissolved organic matter with passive capillary wicks versus aqueous soil extraction. Soil Sci. Soc. Am. J. 2012, 76, 2019. [Google Scholar] [CrossRef]
- Weihermüller, L.; Siemens, J.; Deurer, M.; Knoblauch, S.; Rupp, H.; Göttlein, A.; Pütz, T. In situ soil water extraction: A review. J. Environ. Qual. 2007, 36, 1735–1748. [Google Scholar] [CrossRef]
- Yamashita, T.; Flessa, H.; John, B.; Helfrich, M.; Ludwig, B. Organic matter in density fractions of water-stable aggregates in silty soils: Effect of land use. Soil Biol. Biochem. 2006, 38, 3222–3234. [Google Scholar] [CrossRef]
- Rillig, M.C.; Muller, L.A.H.; Lehmann, A. Soil aggregates as massively concurrent evolutionary incubators. ISME J. 2017, 11, 1943–1948. [Google Scholar] [CrossRef]
- Stone, E.L.; Kalisz, P.J. On the maximum extent of tree roots. For. Ecol. Manag. 1991, 46, 59–102. [Google Scholar] [CrossRef]
- Kramer, C.; Gleixner, G. Soil organic matter in soil depth profiles: Distinct carbon preferences of microbial groups during carbon transformation. Soil Biol. Biochem. 2008, 40, 425–433. [Google Scholar] [CrossRef]
- Harper, R.J.; Tibbett, M. The hidden organic carbon in deep mineral soils. Plant Soil 2013, 368, 641–648. [Google Scholar] [CrossRef]
- Ajwa, H.A.; Rice, C.W.; Sotomayor, D. Carbon and nitrogen mineralization in tallgrass prairie and agricultural soil profiles. Soil Sci. Soc. Am. J. 1998, 62, 942–951. [Google Scholar] [CrossRef]
- Charnay, M.P.; Tuis, S.; Coquet, Y.; Barriuso, E. Spatial variability in 14C-herbicide degradation in surface and subsurface soils. Pest Manag. Sci. 2005, 61, 845–855. [Google Scholar] [CrossRef]
- James, J.N.; Dietzen, C.; Furches, J.C.; Harrison, R.B. Lessons on buried horizons and pedogenesis from deep forest soils. Soil Horizons 2015, 56. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).