Effect of Biochar on Soil Greenhouse Gas Emissions at the Laboratory and Field Scales
Abstract
1. Introduction
2. Materials and Methods
2.1. Biochar
2.2. Field Site
2.3. Incubation Study
2.4. Field Study
2.5. Gas Sample Analysis
2.6. Calculations and Statistical Analyses
3. Results
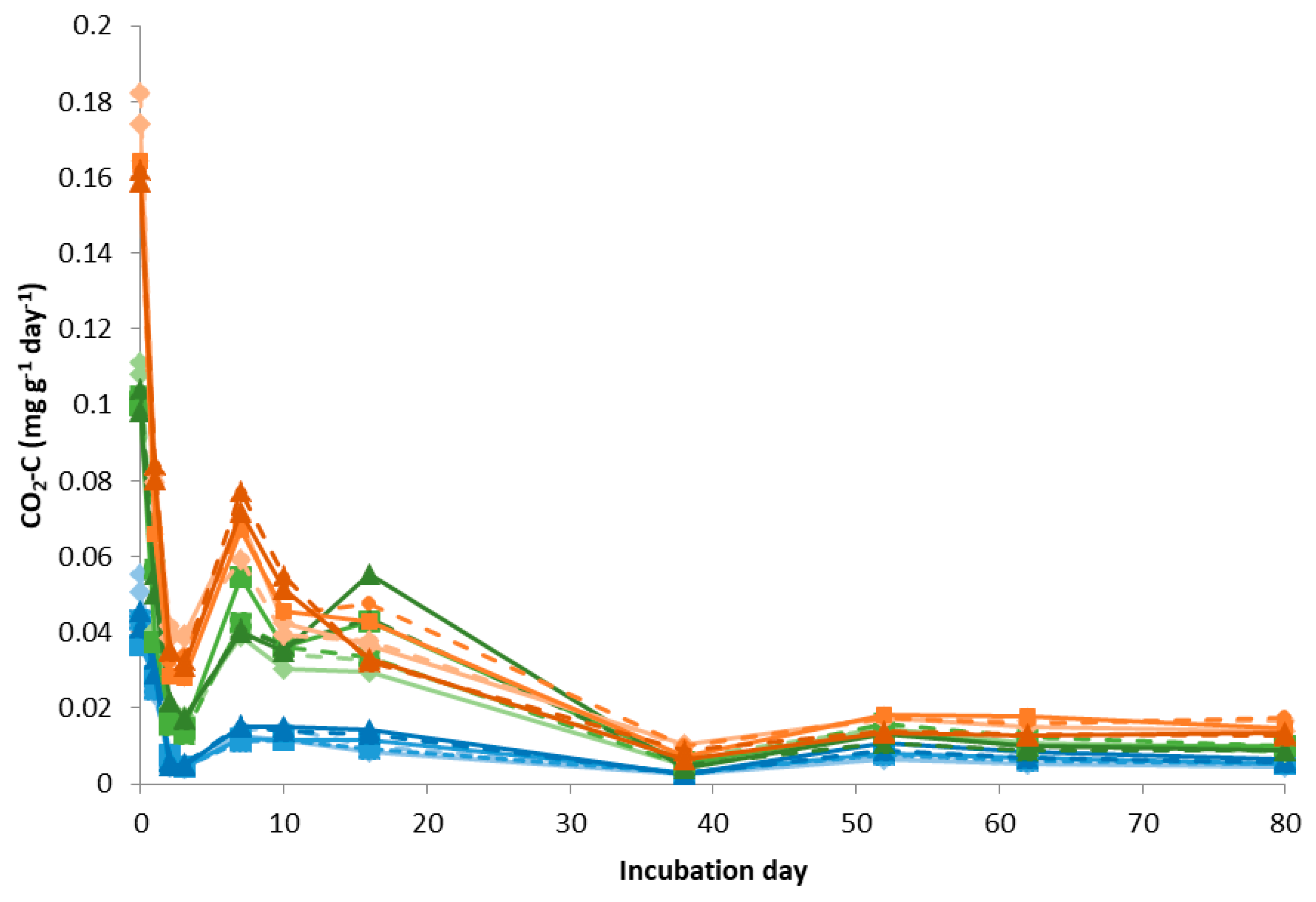
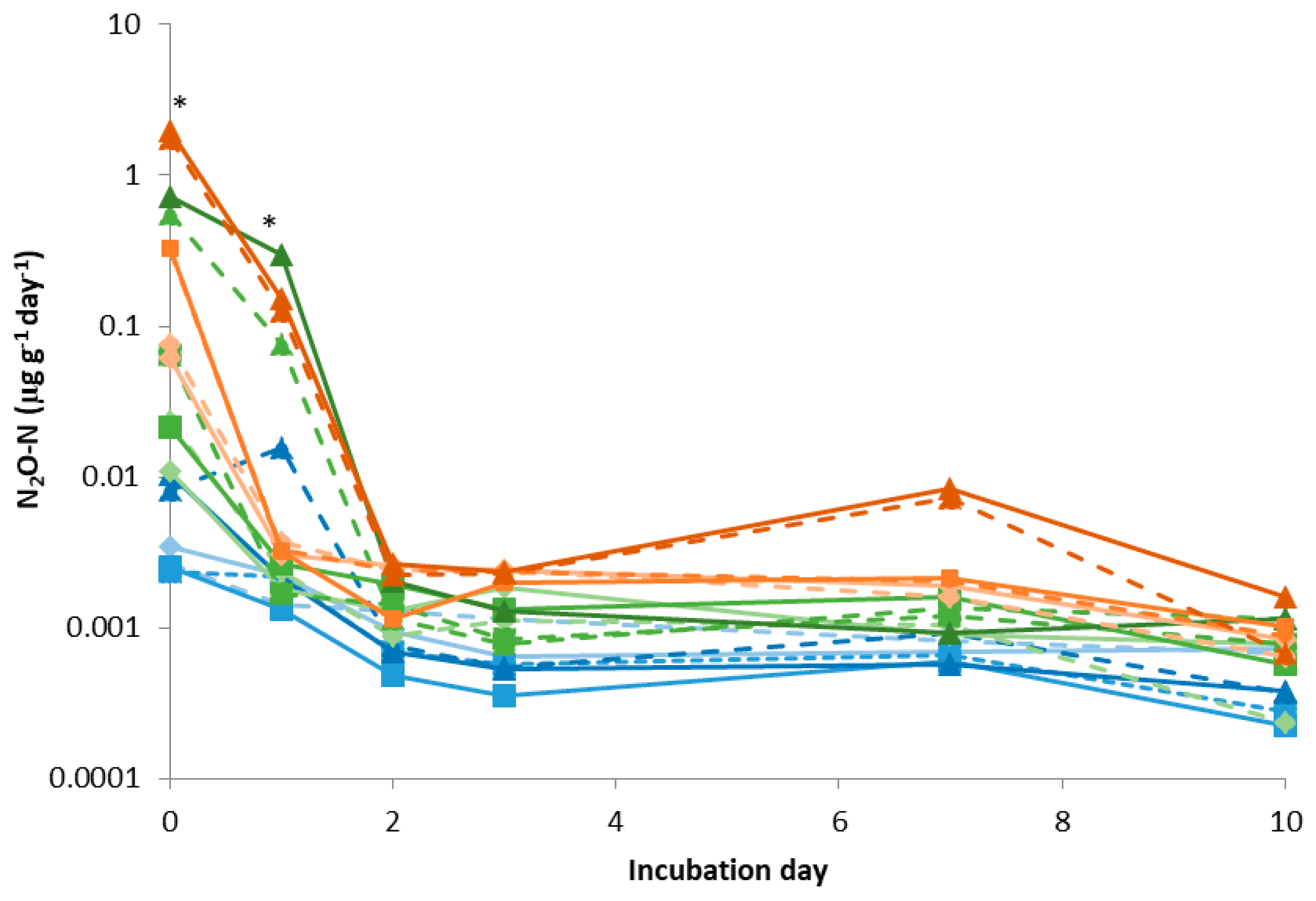
3.1. Soil Laboratory Incubation
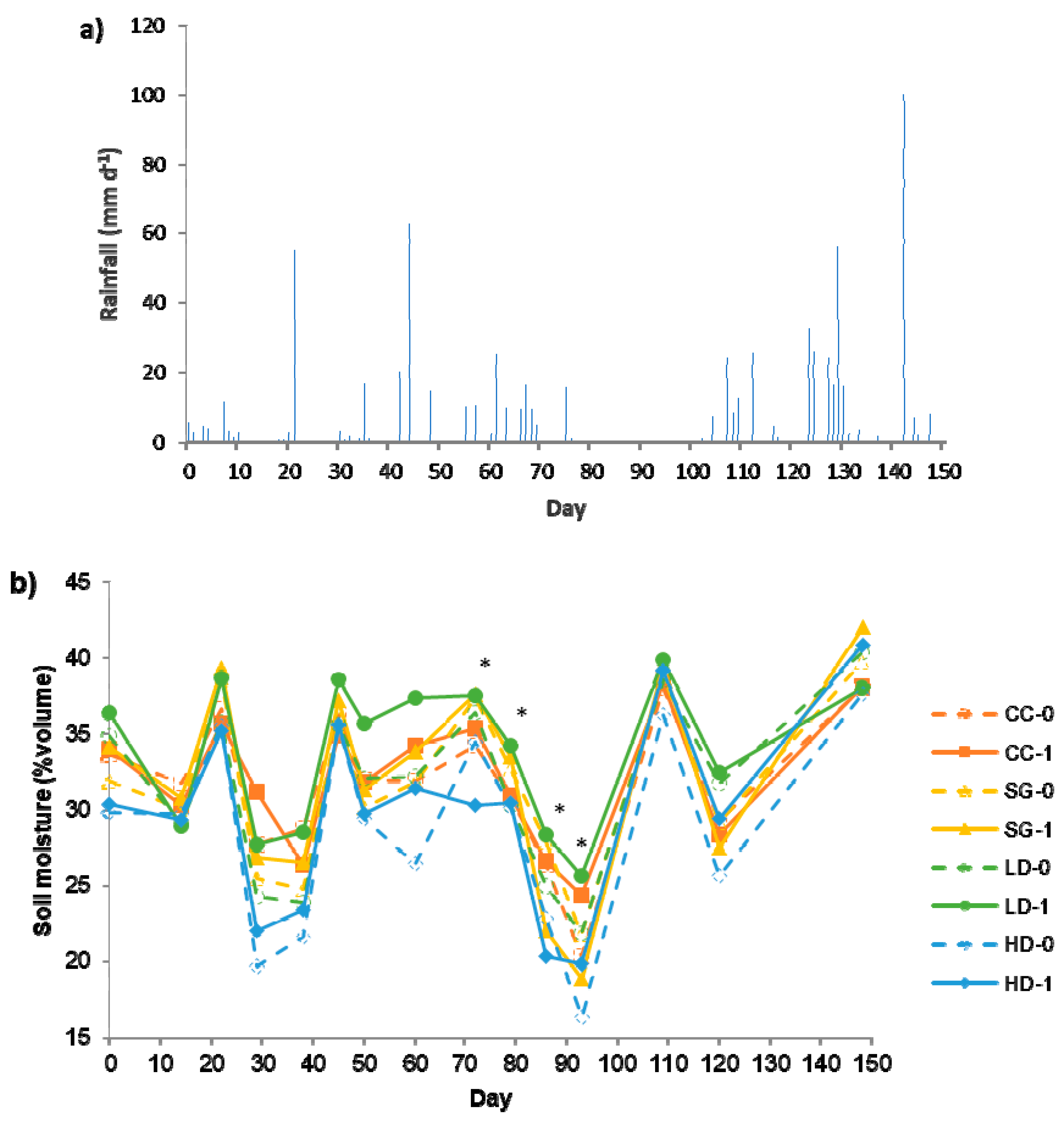
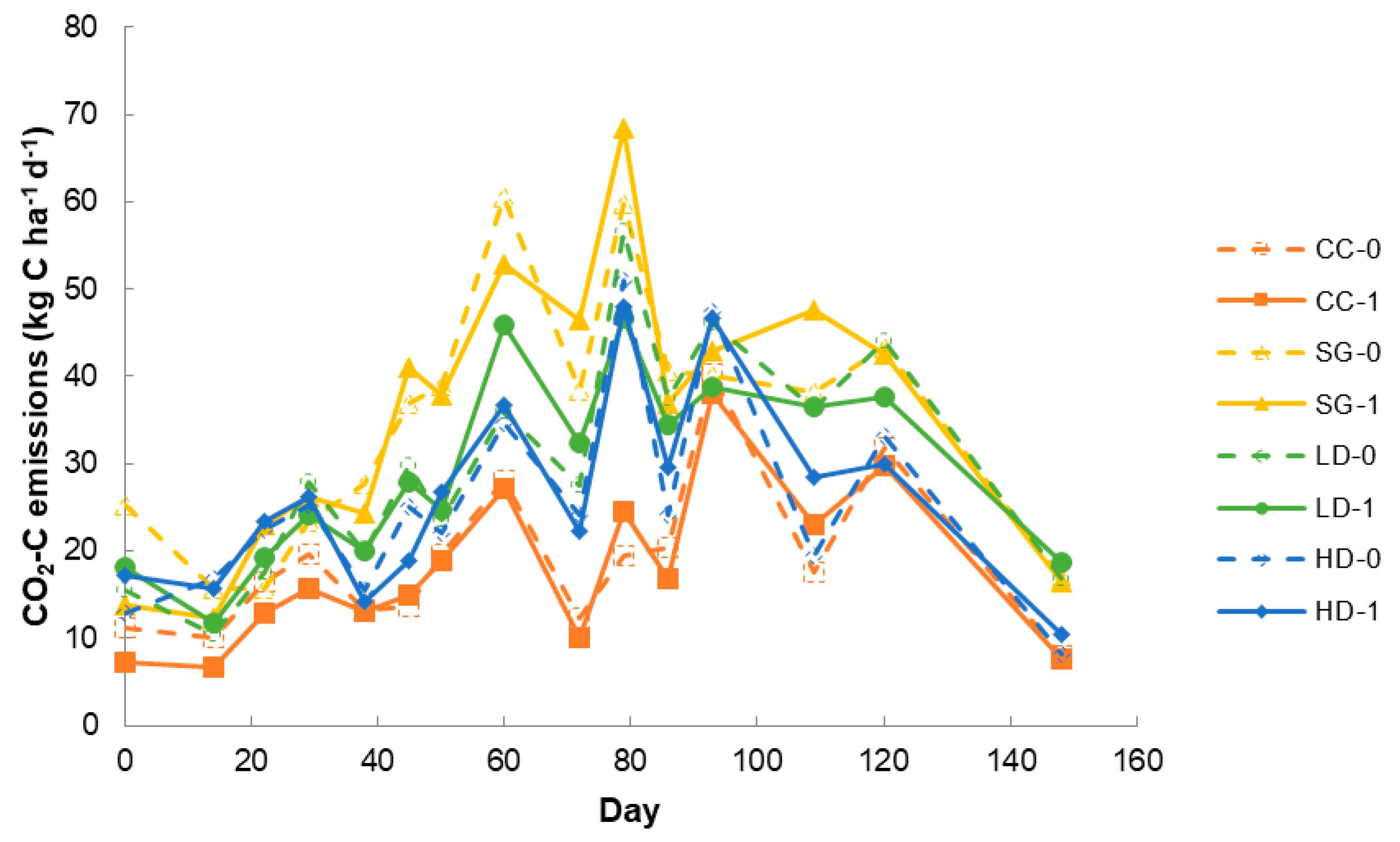
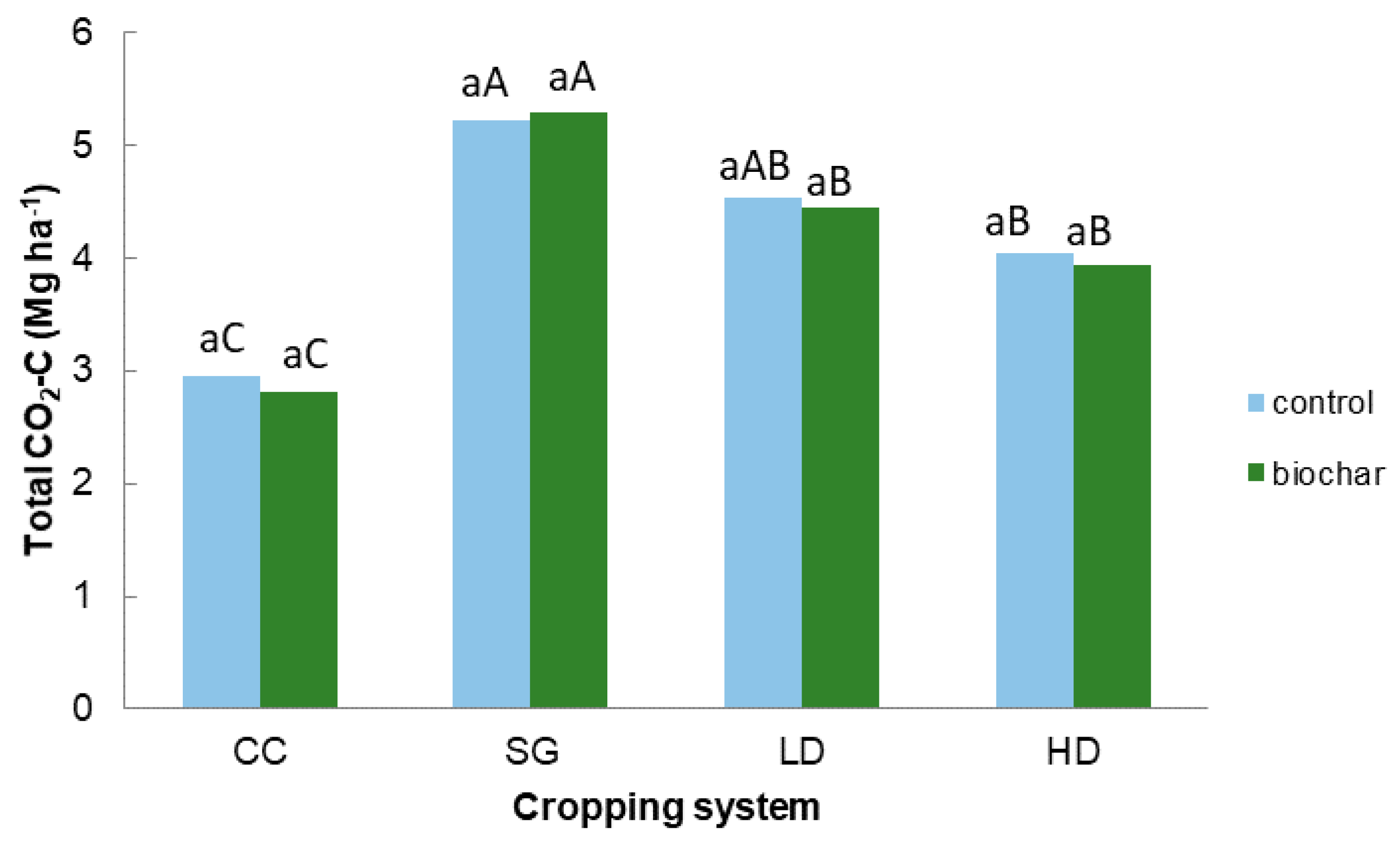
3.2. Field Study: CO2 Emissions
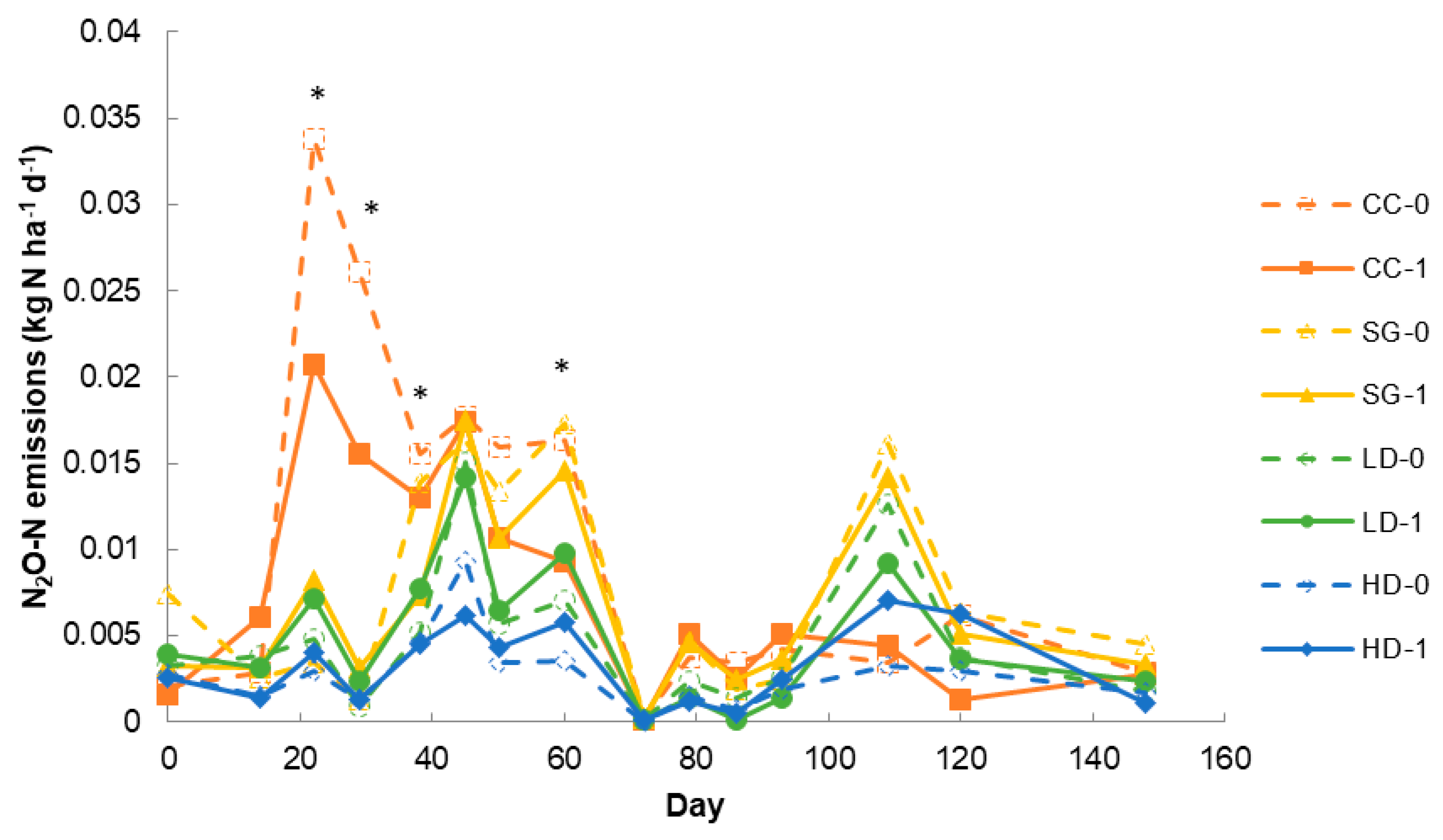
3.3. Field Study: N2O Emissions
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lehmann, J.; Gaunt, J.; Rondon, M. Bio-Char Sequestration in Terrestrial Ecosystems—A Review. Mitig. Adapt. Strateg. Glob. Chang. 2006, 11, 395–419. [Google Scholar] [CrossRef]
- Lehmann, J. A Handful of Carbon. Nature 2007, 447, 143–144. [Google Scholar] [CrossRef] [PubMed]
- Laird, D.A. The Charcoal Vision: A Win–win–win Scenario for Simultaneously Producing Bioenergy, Permanently Sequestering Carbon, while Improving Soil and Water Quality. Agron. J. 2008, 100, 178. [Google Scholar] [CrossRef]
- Roberts, K.G.; Gloy, B.A.; Joseph, S.; Scott, N.R.; Lehmann, J. Life Cycle Assessment of Biochar Systems: Estimating the Energetic, Economic, and Climate Change Potential. Environ. Sci. Technol. 2010, 44, 827–833. [Google Scholar] [CrossRef] [PubMed]
- Kauffman, N.; Dumortier, J.; Hayes, D.J.; Brown, R.C.; Laird, D.A. Producing Energy While Sequestering Carbon? The Relationship between Biochar and Agricultural Productivity. Biomass Bioenergy 2014, 63, 167–176. [Google Scholar] [CrossRef]
- Jeffery, S.; Abalos, D.; Spokas, K.A.; Verheijen, F.G.A. Biochar Effects on Crop Yield. In Biochar for Environmental Management: Science, Technology and Implementation; Routledge: Abingdon, UK, 2015; pp. 301–326. [Google Scholar]
- Crane-Droesch, A.; Abiven, S.; Jeffery, S.; Torn, M.S. Heterogeneous Global Crop Yield Response to Biochar: A Meta-Regression Analysis. Environ. Res. Lett. 2013, 8, 044049. [Google Scholar] [CrossRef]
- Biederman, L.A.; Harpole, W.S. Biochar and Its Effects on Plant Productivity and Nutrient Cycling: A Meta-Analysis. GCB Bioenergy 2013, 5, 202–214. [Google Scholar] [CrossRef]
- Woolf, D.; Amonette, J.E.; Street-Perrott, F.A.; Lehmann, J.; Joseph, S. Sustainable Biochar to Mitigate Global Climate Change. Nat. Commun. 2010, 1, 1–9. [Google Scholar] [CrossRef]
- Spokas, K.A.; Reicosky, D.C. Impacts of Sixteen Different Biochars on Soil Greenhouse Gas Production. Ann. Environ. Sci. 2009, 3, 179–193. [Google Scholar]
- Cayuela, M.L.; van Zwieten, L.; Singh, B.P.; Jeffery, S.; Roig, A.; Sánchez-Monedero, M.A. Biochar’s Role in Mitigating Soil Nitrous Oxide Emissions: A Review and Meta-Analysis. Agric. Ecosyst. Environ. 2014, 191, 5–16. [Google Scholar] [CrossRef]
- Cayuela, M.L.; Sánchez-Monedero, M.A.; Roig, A.; Hanley, K.; Enders, A.; Lehmann, J. Biochar and Denitrification in Soils: When, How Much and Why Does Biochar Reduce N2O Emissions? Sci. Rep. 2013, 3, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Archontoulis, S.V.; Huber, I.; Miguez, F.E.; Thorburn, P.J.; Rogovska, N.; Laird, D.A. A Model for Mechanistic and System Assessments of Biochar Effects on Soils and Crops and Trade-Offs. GCB Bioenergy 2016, 8, 1–18. [Google Scholar] [CrossRef]
- Saarnio, S. Impacts of Biochar Amendment on Greenhouse Gas Emissions from Agricultural Soils. In Agricultural and Environmental Applications of Biochar: Advances and Barriers; Soil Science Society of America, Inc.: Madison, WI, USA, 2015; pp. 1–36. [Google Scholar]
- Song, X.; Pan, G.; Zhang, C.; Zhang, L.; Wang, H. Effects of Biochar Application on Fluxes of Three Biogenic Greenhouse Gases: A Meta-Analysis. Ecosyst. Heal. Sustain. 2016, 2, e01202. [Google Scholar] [CrossRef]
- Liu, S.; Zhang, Y.; Zong, Y.; Hu, Z.; Wu, S.; Zhou, J.; Jin, Y.; Zou, J. Response of Soil Carbon Dioxide Fluxes, Soil Organic Carbon and Microbial Biomass Carbon to Biochar Amendment: A Meta-Analysis. GCB Bioenergy 2016, 8, 392–406. [Google Scholar] [CrossRef]
- He, Y.; Zhou, X.; Jiang, L.; Li, M.; Du, Z.; Zhou, G.; Shao, J.; Wang, X.; Xu, Z.; Hosseini Bai, S.; et al. Effects of Biochar Application on Soil Greenhouse Gas Fluxes: A Meta-Analysis. GCB Bioenergy 2016, 1–13. [Google Scholar] [CrossRef]
- Jones, D.L.; Murphy, D.V.; Khalid, M.; Ahmad, W.; Edwards-Jones, G.; DeLuca, T.H. Short-Term Biochar-Induced Increase in Soil CO2 Release Is Both Biotically and Abiotically Mediated. Soil Biol. Biochem. 2011, 43, 1723–1731. [Google Scholar] [CrossRef]
- Cross, A.; Sohi, S.P. The Priming Potential of Biochar Products in Relation to Labile Carbon Contents and Soil Organic Matter Status. Soil Biol. Biochem. 2011, 43, 2127–2134. [Google Scholar] [CrossRef]
- Bruun, S.; Clauson-Kaas, S.; Bobuľská, L.; Thomsen, I.K. Carbon Dioxide Emissions from Biochar in Soil: Role of Clay, Microorganisms and Carbonates. Eur. J. Soil Sci. 2014, 65, 52–59. [Google Scholar] [CrossRef]
- Farrell, M.; Kuhn, T.K.; Macdonald, L.M.; Maddern, T.M.; Murphy, D.V.; Hall, P.A.; Singh, B.P.; Baumann, K.; Krull, E.S.; Baldock, J.A. Microbial Utilisation of Biochar-Derived Carbon. Sci. Total Environ. 2013, 465, 288–297. [Google Scholar] [CrossRef]
- Fang, Y.; Singh, B.; Singh, B.P.; Krull, E. Biochar Carbon Stability in Four Contrasting Soils. Eur. J. Soil Sci. 2014, 65, 60–71. [Google Scholar] [CrossRef]
- Lin, X.W.; Xie, Z.B.; Zheng, J.Y.; Liu, Q.; Bei, Q.C.; Zhu, J.G. Effects of Biochar Application on Greenhouse Gas Emissions, Carbon Sequestration and Crop Growth in Coastal Saline Soil. Eur. J. Soil Sci. 2015, 66, 329–338. [Google Scholar] [CrossRef]
- Lu, W.; Ding, W.; Zhang, J.; Li, Y.; Luo, J.; Bolan, N.; Xie, Z. Biochar Suppressed the Decomposition of Organic Carbon in a Cultivated Sandy Loam Soil: A Negative Priming Effect. Soil Biol. Biochem. 2014, 76, 12–21. [Google Scholar] [CrossRef]
- Smith, J.L.; Collins, H.P.; Bailey, V.L. The Effect of Young Biochar on Soil Respiration. Soil Biol. Biochem. 2010, 42, 2345–2347. [Google Scholar] [CrossRef]
- Zimmerman, A.R.; Gao, B.; Ahn, M.-Y. Positive and Negative Carbon Mineralization Priming Effects among a Variety of Biochar-Amended Soils. Soil Biol. Biochem. 2011, 43, 1169–1179. [Google Scholar] [CrossRef]
- Whitman, T.; Enders, A.; Lehmann, J. Pyrogenic Carbon Additions to Soil Counteract Positive Priming of Soil Carbon Mineralization by Plants. Soil Biol. Biochem. 2014, 73, 33–41. [Google Scholar] [CrossRef]
- Keith, A.; Singh, B.; Singh, B.P. Interactive Priming of Biochar and Labile Organic Matter Mineralization in a Smectite-Rich Soil. Environ. Sci. Technol. 2011, 45, 9611–9618. [Google Scholar] [CrossRef] [PubMed]
- Watzinger, A.; Feichtmair, S.; Kitzler, B.; Zehetner, F.; Kloss, S.; Wimmer, B.; Zechmeister-Boltenstern, S.; Soja, G. Soil Microbial Communities Responded to Biochar Application in Temperate Soils and Slowly Metabolized 13C-Labelled Biochar as Revealed by 13C PLFA Analyses: Results from a Short-Term Incubation and Pot Experiment. Eur. J. Soil Sci. 2014, 65, 40–51. [Google Scholar] [CrossRef]
- Zhang, Y.; Hu, X.; Zou, J.; Zhang, D.; Chen, W.; Liu, Y.; Chen, Y.; Wang, X. Response of surface albedo and soil carbon dioxide fluxes to biochar amendment in farmland. J. Soils Sediments 2018, 18, 1590–1601. [Google Scholar] [CrossRef]
- Cayuela, M.L.; Jeffery, S.; van Zwieten, L. The Molar H:Corg Ratio of Biochar Is a Key Factor in Mitigating N2O Emissions from Soil. Agric. Ecosyst. Environ. 2015, 202, 135–138. [Google Scholar] [CrossRef]
- Zhang, A.; Cui, L.; Pan, G.; Li, L.; Hussain, Q.; Zhang, X.; Zheng, J.; Crowley, D. Effect of Biochar Amendment on Yield and Methane and Nitrous Oxide Emissions from a Rice Paddy from Tai Lake Plain, China. Agric. Ecosyst. Environ. 2010, 139, 469–475. [Google Scholar] [CrossRef]
- Zhang, A.; Liu, Y.; Pan, G.; Hussain, Q.; Li, L.; Zheng, J.; Zhang, X. Effect of Biochar Amendment on Maize Yield and Greenhouse Gas Emissions from a Soil Organic Carbon Poor Calcareous Loamy Soil from Central China Plain. Plant Soil 2012, 351, 263–275. [Google Scholar] [CrossRef]
- Case, S.D.C.; McNamara, N.P.; Reay, D.S.; Whitaker, J. Can Biochar Reduce Soil Greenhouse Gas Emissions from a Miscanthus Bioenergy Crop? GCB Bioenergy 2014, 6, 76–89. [Google Scholar] [CrossRef]
- Zhang, A.; Bian, R.; Hussain, Q.; Li, L.; Pan, G.; Zheng, J.; Zhang, X.; Zheng, J. Change in Net Global Warming Potential of a Rice–wheat Cropping System with Biochar Soil Amendment in a Rice Paddy from China. Agric. Ecosyst. Environ. 2013, 173, 37–45. [Google Scholar] [CrossRef]
- Scheer, C.; Grace, P.R.; Rowlings, D.W.; Kimber, S.; Van Zwieten, L. Effect of Biochar Amendment on the Soil-Atmosphere Exchange of Greenhouse Gases from an Intensive Subtropical Pasture in Northern New South Wales, Australia. Plant Soil 2011, 345, 47–58. [Google Scholar] [CrossRef]
- Liu, X.; Qu, J.; Li, L.; Zhang, A.; Jufeng, Z.; Zheng, J.; Pan, G. Can Biochar Amendment Be an Ecological Engineering Technology to Depress N2O Emission in Rice Paddies?—A Cross Site Field Experiment from South China. Ecol. Eng. 2012, 42, 168–173. [Google Scholar] [CrossRef]
- Bian, R.; Zhang, A.; Li, L.; Pan, G.; Zheng, J.; Zhang, X.; Zheng, J.; Joseph, S.; Chang, A. Effect of Municipal Biowaste Biochar on Greenhouse Gas Emissions and Metal Bioaccumulation in a Slightly Acidic Clay Rice Paddy. BioResources 2014, 9, 685–703. [Google Scholar] [CrossRef]
- Felber, R.; Leifeld, J.; Horák, J.; Neftel, A. Nitrous Oxide Emission Reduction with Greenwaste Biochar: Comparison of Laboratory and Field Experiments. Eur. J. Soil Sci. 2014, 65, 128–138. [Google Scholar] [CrossRef]
- Pandey, A.; Mai, V.T.; Vu, D.Q.; Bui, T.P.L.; Mai, T.L.A.; Jensen, L.S.; de Neergaard, A. Organic Matter and Water Management Strategies to Reduce Methane and Nitrous Oxide Emissions from Rice Paddies in Vietnam. Agric. Ecosyst. Environ. 2014, 196, 137–146. [Google Scholar] [CrossRef]
- Shen, J.; Tang, H.; Liu, J.; Wang, C.; Li, Y.; Ge, T.; Jones, D.L.; Wu, J. Contrasting Effects of Straw and Straw-Derived Biochar Amendments on Greenhouse Gas Emissions within Double Rice Cropping Systems. Agric. Ecosyst. Environ. 2014, 188, 264–274. [Google Scholar] [CrossRef]
- Van Zwieten, L.; Singh, B.P.; Kimber, S.W.L.; Murphy, D.V.; Macdonald, L.M.; Rust, J.; Morris, S. An Incubation Study Investigating the Mechanisms That Impact N2O Flux from Soil Following Biochar Application. Agric. Ecosyst. Environ. 2014, 191, 53–62. [Google Scholar] [CrossRef]
- Spokas, K.A. Impact of Biochar Field Aging on Laboratory Greenhouse Gas Production Potentials. GCB Bioenergy 2013, 5, 165–176. [Google Scholar] [CrossRef]
- Keith, A.; Singh, B.; Dijkstra, F.A.; van Ogtrop, F. Biochar Field Study: Greenhouse Gas Emissions, Productivity, and Nutrients in Two Soils. Agron. J. 2016, 0, 0. [Google Scholar] [CrossRef]
- Luo, G.J.; Kiese, R.; Wolf, B.; Butterbach-Bahl, K. Effects of Soil Temperature and Moisture on Methane Uptake and Nitrous Oxide Emissions across Three Different Ecosystem Types. Biogeosciences 2013, 10, 3205–3219. [Google Scholar] [CrossRef]
- Smith, K.A.; Thomson, P.E.; Clayton, H.; McTaggart, I.P.; Conen, F. Effects of Temperature, Water Content and Nitrogen Fertilisation on Emissions of Nitrous Oxide by Soils. Atmos. Environ. 1998, 32, 3301–3309. [Google Scholar] [CrossRef]
- Castellano, M.J.; Schmidt, J.P.; Kaye, J.P.; Walker, C.; Graham, C.B.; Lin, H.; Dell, C. Hydrological Controls on Heterotrophic Soil Respiration across an Agricultural Landscape. Geoderma 2011, 162, 273–280. [Google Scholar] [CrossRef]
- Castellano, M.J.; Schmidt, J.P.; Kaye, J.P.; Walker, C.; Graham, C.B.; Lin, H.; Dell, C.J. Hydrological and Biogeochemical Controls on the Timing and Magnitude of Nitrous Oxide Flux across an Agricultural Landscape. Glob. Chang. Biol. 2010, 16, 2711–2720. [Google Scholar] [CrossRef]
- Watanabe, A.; Ikeya, K.; Kanazaki, N.; Makabe, S.; Sugiura, Y.; Shibata, A. Five Crop Seasons’ Records of Greenhouse Gas Fluxes from Upland Fields with Repetitive Applications of Biochar and Cattle Manure. J. Environ. Manag. 2014, 144, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Bass, A.M.; Bird, M.I.; Kay, G.; Muirhead, B. Soil Properties, Greenhouse Gas Emissions and Crop Yield under Compost, Biochar and Co-Composted Biochar in Two Tropical Agronomic Systems. Sci. Total Environ. 2016, 550, 459–470. [Google Scholar] [CrossRef]
- Fidel, R.B.; Laird, D.A.; Parkin, T.B. Impact of Six Lignocellulosic Biochars on C and N Dynamics of Two Contrasting Soils. GCB Bioenergy 2017, 9, 1279–1291. [Google Scholar] [CrossRef]
- Bonin, C.L.; Fidel, R.B.; Banik, C.; Laird, D.A.; Mitchell, R.; Heaton, E.A. Perennial Biomass Crop Establishment, Community Characteristics, and Productivity in the Upper US Midwest: Effects of Cropping Systems Seed Mixtures and Biochar Applications. Eur. J. Agron. 2018, 101, 121–128. [Google Scholar] [CrossRef]
- Parkin, T.B.; Venterea, R.T. Chamber-Based Trace Gas Flux Measurements. In Sampling Protocols; Follett, R.F., Ed.; USDA-ARS: Washington, DC, USA, 2010. [Google Scholar]
- Rochette, P.; Eriksen-Hamel, N.S. Chamber measurements of soil nitrous oxide flux: Are absolute values reliable? Soil Sci. Soc. Am. J. 2008, 72, 331–342. [Google Scholar] [CrossRef]
- Iqbal, J.; Castellano, M.J.; Parkin, T.B. Evaluation of Photoacoustic Infrared Spectroscopy for Simultaneous Measurement of N2O and CO2 Gas Concentrations and Fluxes at the Soil Surface. Glob. Chang. Biol. 2013, 19, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Danevčič, T.; Mandic-Mulec, I.; Stres, B.; Stopar, D.; Hacin, J. Emissions of CO2, CH4 and N2O from Southern European peatlands. Soil Biol. Biochem. 2010, 42, 1437–1446. [Google Scholar] [CrossRef]
- Parkin, T.B.; Venterea, R.T.; Hargreaves, S.K. Calculating the Detection Limits of Chamber-based Soil Greenhouse Gas Flux Measurements. J. Environ. Qual. 2012, 41, 705–715. [Google Scholar] [CrossRef] [PubMed]
- Thomazini, A.; Spokas, K.; Hall, K.; Ippolito, J.; Lentz, R.; Novak, J. GHG Impacts of Biochar: Predictability for the Same Biochar. Agric. Ecosyst. Environ. 2015, 207, 183–191. [Google Scholar] [CrossRef]
- Fidel, R.B.; Laird, D.A.; Parkin, T.B. Impact of Biochar Organic and Inorganic C on Soil CO2 and N2O Emissions. J. Environ. Qual. 2017, 46, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Velthof, G.L.; Kuikman, P.J.; Oenema, O. Nitrous Oxide Emission from Soils Amended with Crop Residues. Nutr. Cycl. Agroecosyst. 2002, 62, 249–261. [Google Scholar] [CrossRef]
- Novak, J.M.; Busscher, W.J.; Watts, D.W.; Amonette, J.E.; Ippolito, J.A.; Lima, I.M.; Gaskin, J.; Das, K.C.; Steiner, C.; Ahmedna, M.; et al. Biochars Impact on Soil-Moisture Storage in an Ultisol and Two Aridisols. Soil Sci. 2012, 177, 310–320. [Google Scholar] [CrossRef]
- Ulyett, J.; Sakrabani, R.; Kibblewhite, M.; Hann, M. Impact of Biochar Addition on Water Retention, Nitrification and Carbon Dioxide Evolution from Two Sandy Loam Soils. Eur. J. Soil Sci. 2014, 65, 96–104. [Google Scholar] [CrossRef]
- Karhu, K.; Mattila, T.; Bergström, I.; Regina, K. Biochar Addition to Agricultural Soil Increased CH4 Uptake and Water Holding Capacity—Results from a Short-Term Pilot Field Study. Agric. Ecosyst. Environ. 2011, 140, 309–313. [Google Scholar] [CrossRef]
- Bateman, E.J.; Baggs, E.M. Contributions of Nitrification and Denitrification to N2O Emissions from Soils at Different Water-Filled Pore Space. Biol. Fertil. Soils 2005, 41, 379–388. [Google Scholar] [CrossRef]
- Deng, Q.; Hui, D.; Wang, J.; Iwuozo, S.; Yu, C.-L.; Jima, T.; Smart, D.; Reddy, C.; Dennis, S. Corn Yield and Soil Nitrous Oxide Emission under Different Fertilizer and Soil Management: A Three-Year Field Experiment in Middle Tennessee. PLoS ONE 2015, 10, e0125406. [Google Scholar] [CrossRef] [PubMed]

| Temperature | Moisture (%) | CO2 (mg CO2 g−1) | N2O (µg N2O g−1) | ||
|---|---|---|---|---|---|
| control | biochar | control | biochar | ||
| 10°C | 27 | 2.33 ±0.05 | 2.1 ±0.1 | 0.043±0.003 | 0.047 ±0.002 |
| 31 | 2.21 ±0.09 | 2.4 ±0.1 | 0.041±0.006 | 0.029 ±0.006 | |
| 35 | 2.6 ±0.1 | 2.9 ±0.1 | 0.1 ±0.05 | 0.10 ±0.07 | |
| 20°C | 27 | 5.7 ±0.4 | 5.4 ±0.4 | 0.15 ±0.02 | 0.09 ±0.01 |
| 31 | 5.8 ±0.5 | 6.2 ±0.5 | 0.34 ±0.1 | 0.15 ±0.02 | |
| 35 | 5.9 ±0.2 | 6.6 ±0.6 | 3.2 ±0.4 | 4.3 ±0.9 | |
| 30°C | 27 | 8.0 ±0.5 | 7.9 ±0.4 | 0.4 ±0.05 | 0.4 ±0.07 |
| 31 | 8.4 ±0.5 | 8.2 ±0.6 | 1.6 ±0.5 | 1.6 ±0.5 | |
| 35 | 7.5 ±0.1 | 7.2 ±0.3 | 7 ±2 | 10 ±2 | |
| %C | %N | |||
|---|---|---|---|---|
| Crop | Control | Biochar | Control | Biochar |
| CC | 2.5 ± 0.3 | 2.9 ± 0.2 | 0.26 ± 0.01 | 0.26 ± 0.02 |
| SG | 2.5 ± 0.1 | 3.2 ± 0.3 | 0.26 ± 0.01 | 0.26 ± 0.01 |
| LD | 2.2 ± 0.1 | 3.4 ± 0.2 | 0.24 ± 0.01 | 0.27 ± 0.01 |
| HD | 2.9 ± 0.3 | 3.2 ± 0.3 | 0.27 ± 0.01 | 0.27 ± 0.02 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fidel, R.B.; Laird, D.A.; Parkin, T.B. Effect of Biochar on Soil Greenhouse Gas Emissions at the Laboratory and Field Scales. Soil Syst. 2019, 3, 8. https://doi.org/10.3390/soilsystems3010008
Fidel RB, Laird DA, Parkin TB. Effect of Biochar on Soil Greenhouse Gas Emissions at the Laboratory and Field Scales. Soil Systems. 2019; 3(1):8. https://doi.org/10.3390/soilsystems3010008
Chicago/Turabian StyleFidel, Rivka B., David A. Laird, and Timothy B. Parkin. 2019. "Effect of Biochar on Soil Greenhouse Gas Emissions at the Laboratory and Field Scales" Soil Systems 3, no. 1: 8. https://doi.org/10.3390/soilsystems3010008
APA StyleFidel, R. B., Laird, D. A., & Parkin, T. B. (2019). Effect of Biochar on Soil Greenhouse Gas Emissions at the Laboratory and Field Scales. Soil Systems, 3(1), 8. https://doi.org/10.3390/soilsystems3010008





