Influence of Tillage Practices, Organic Manures and Extrinsic Factors on β-Glucosidase Activity: The Final Step of Cellulose Hydrolysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Field Site and Experiments
2.2. Measured Variables
2.3. Data Analysis
3. Results
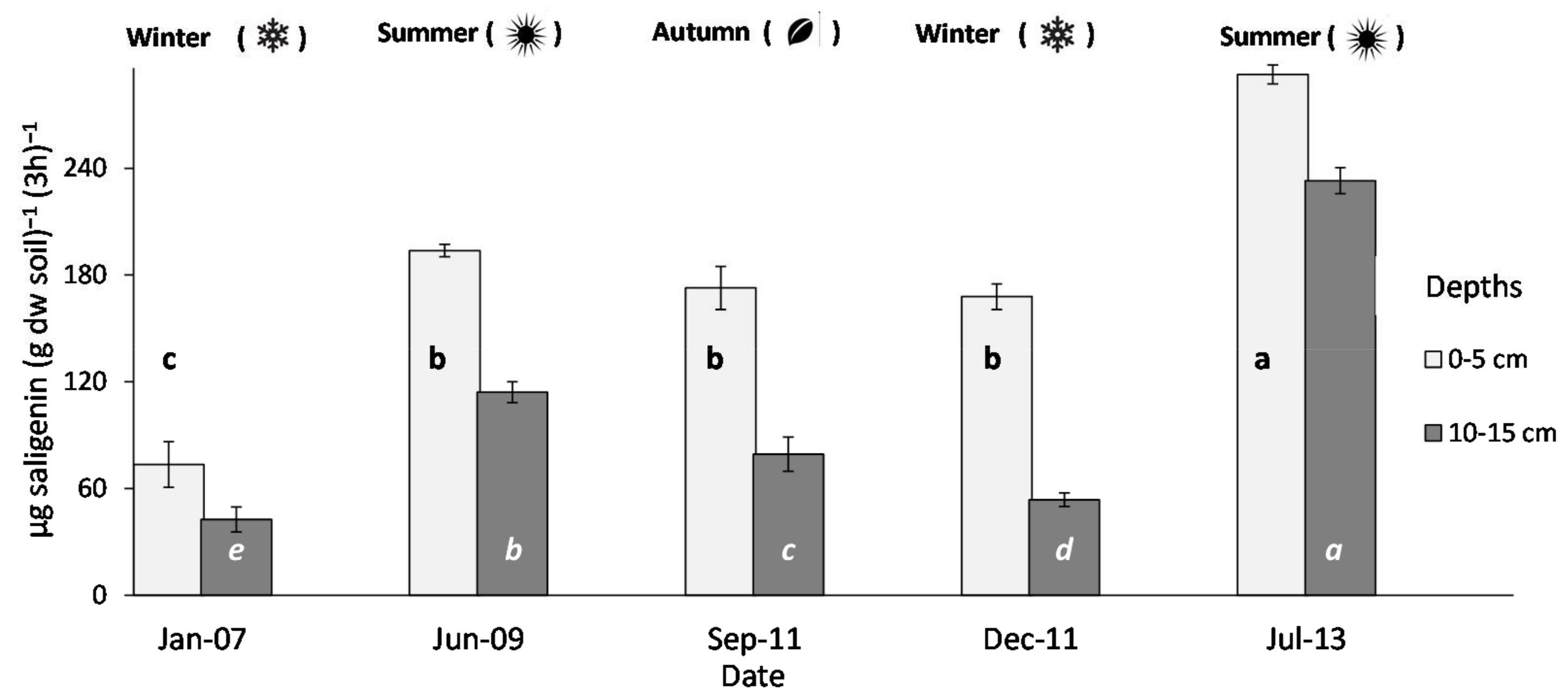
3.1. β-Glucosidase Activity under Grass-Reference Soil and Relationships with Meteorological Data
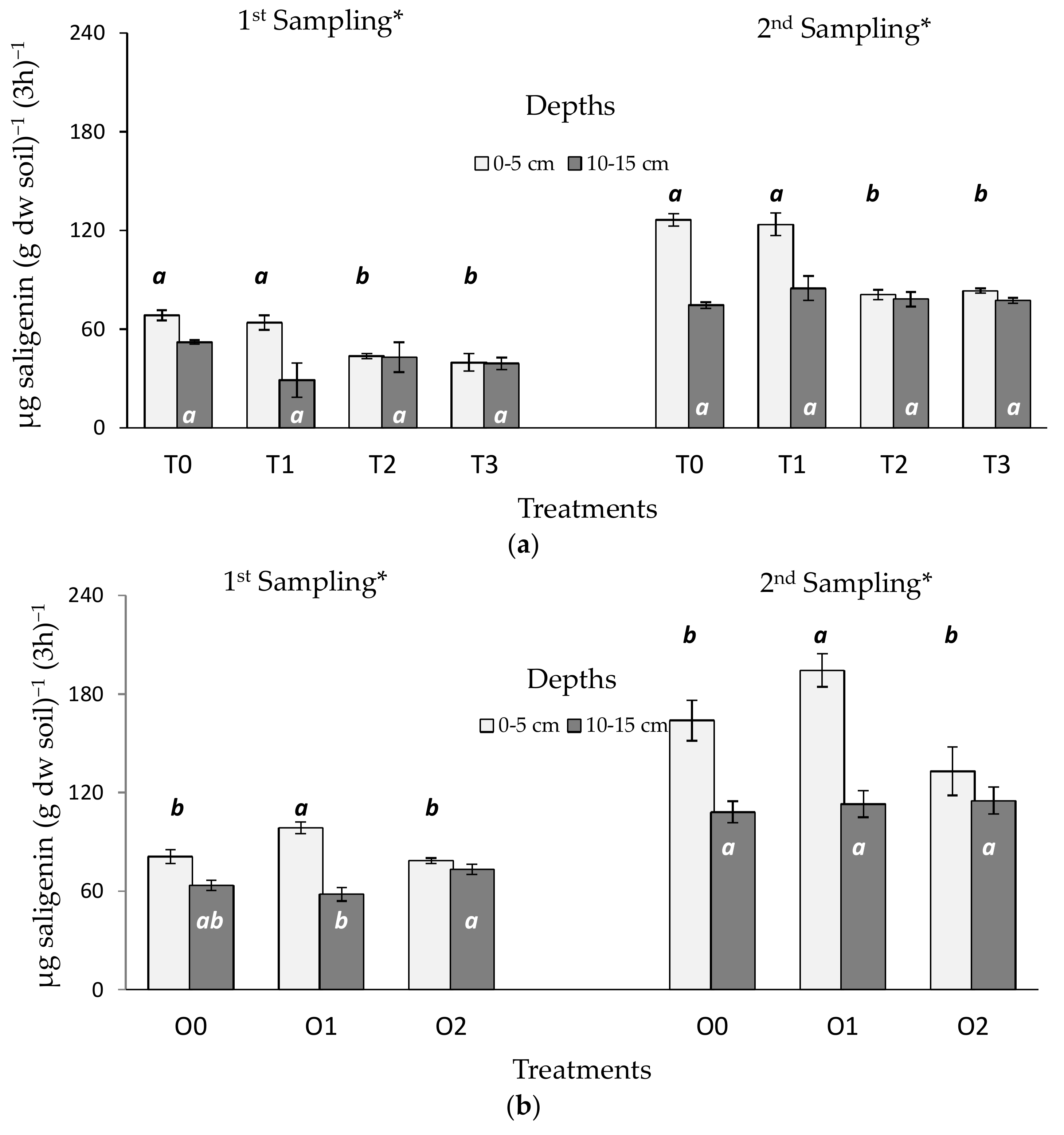
3.2. Tillage and Organic Residue Experiments
4. Discussion
4.1. Temporal Variability of β-Glucosidase Activity and Rainfall
4.2. Tillage and Organic Treatments
4.3. Response Ratio for Comparing Samples Taken in Different Seasons
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Eriksson, K.; Blanchette, R.A.; Ander, P. Biodegradation of cellulose. In Microbial and Enzymatic Degradation of Wood and Wood Components; Eriksson, K., Blanchette, R.A., Ander, P., Eds.; Springer: Berlin, Germany, 1990; pp. 89–180. [Google Scholar]
- Lammirato, C.; Miltner, A.; Wick, L.Y.; Kästner, M. Hydrolysis of cellobiose by β-glucosidase in the presence of soil minerals–Interactions at solid–liquid interfaces and effects on enzyme activity levels. Soil Biol. Biochem. 2010, 42, 2203–2210. [Google Scholar] [CrossRef]
- Stege, P.W.; Messina, G.A.; Bianchi, G.; Olsina, R.A.; Raba, J. Determination of β-glucosidase activity in soils with a bioanalytical sensor modified with multiwalled carbon nanotubes. Anal. Bioanal. Chem. 2010, 397, 1347–1353. [Google Scholar] [CrossRef] [PubMed]
- Maurya, B.; Singh, V.; Dhyani, P.; Kumar, A. Influence of altitudes on activity of soil health bioindicators β-glucosidase and urease in agricultural soils of Almora district of central Himalaya. Res. J. Soil Biol. 2012, 4, 1–9. [Google Scholar] [CrossRef]
- Błońska, E.; Lasota, J.; Zwydak, M.; Klamerus-Iwan, A.; Gołąb, J. Restoration of forest soil and vegetation 15 years after landslides in a lower zone of mountains in temperate climates. Ecol. Eng. 2016, 97, 503–515. [Google Scholar] [CrossRef]
- Turner, B.L.; Hopkins, D.W.; Haygarth, P.M.; Ostle, N. β-Glucosidase activity in pasture soils. Appl. Soil Ecol. 2002, 20, 157–162. [Google Scholar] [CrossRef]
- Gil-Sotres, F.; Trasar-Cepeda, C.; Leirós, M.; Seoane, S. Different approaches to evaluating soil quality using biochemical properties. Soil Biol. Biochem. 2005, 37, 877–887. [Google Scholar] [CrossRef]
- Błońska, E.; Lasota, J.; Gruba, P. Enzymatic activity and stabilization of organic matter in soil with different detritus inputs. Soil Sci. Plant Nutr. 2017, 63, 242–247. [Google Scholar] [CrossRef]
- León, P.; Espejo, R.; Gómez-Paccard, C.; Hontoria, C.; Mariscal, I.; Renella, G.; Benito, M. No tillage and sugar beet foam amendment enhanced microbial activity of degraded acidic soils in South West Spain. Appl. Soil Ecol. 2017, 109, 69–74. [Google Scholar] [CrossRef]
- Dilly, O.; Munch, J.C.; Pfeiffer, E. Enzyme activities and litter decomposition in agricultural soils in northern, central, and southern Germany. J. Plant Nutr. Soil Sci. 2007, 170, 197–204. [Google Scholar] [CrossRef]
- Soil Survey Staff. Keys to Soil Taxonomy; USDA-Natural Resources Conservation Service: Washington, DC, USA, 2014. [Google Scholar]
- Newton, A.; Guy, D.; Bengough, A.; Gordon, D.; McKenzie, B.; Sun, B.; Valentine, T.; Hallett, P. Soil tillage effects on the efficacy of cultivars and their mixtures in winter barley. Field Crop. Res. 2012, 128, 91–100. [Google Scholar] [CrossRef]
- British Standards Institution PAS 100: Specification for Composted Materials; British Standards Institution (BSI): London, UK, 2005. [CrossRef]
- Paterson, E.; Neilson, R.; Midwood, A.J.; Osborne, S.M.; Sim, A.; Thornton, B.; Millard, P. Altered food web structure and C-flux pathways associated with mineralisation of organic amendments to agricultural soil. Appl. Soil Ecol. 2011, 48, 107–116. [Google Scholar] [CrossRef]
- Ball, B.; Watson, C.; Baddeley, J. Soil physical fertility, soil structure and rooting conditions after ploughing organically managed grass/clover swards. Soil Use Manag. 2007, 23, 20–27. [Google Scholar] [CrossRef]
- Hoffmann, G.; Dedeken, M. Eine Methode zur colorimetrischen Bestimmung der β-Glucosidase-Aktivität in Böden. J. Plant Nutr. Soil Sci. 1965, 108, 193–198. [Google Scholar] [CrossRef]
- Mariscal-Sancho, I.; Santano, J.; Mendiola, M.Á.; Peregrina, F.; Espejo, R. Carbon dioxide emission rates and β-glucosidase activity in Mediterranean Ultisols under different soil management. Soil Sci. 2010, 175, 453–460. [Google Scholar] [CrossRef]
- Strobl, W.; Traunmüller, M.; Schinner, F.; Öhlinger, R.; Kandeler, E.; Margesin, R. β-Glucosidase Activity. In Methods in Soil Biology; Springer: Berlin, Germany, 1996; pp. 198–200. [Google Scholar]
- Böhme, L.; Böhme, F. Soil microbiological and biochemical properties affected by plant growth and different long-term fertilisation. Eur. J. Soil Biol. 2006, 42, 1–12. [Google Scholar] [CrossRef]
- Ball, B.; Campbell, D.; Hunter, E. Soil compactibility in relation to physical and organic properties at 156 sites in UK. Soil Tillage Res. 2000, 57, 83–91. [Google Scholar] [CrossRef]
- Mariscal-Sancho, I.; Ball, B.; Peregrina, F. Soil quality dynamics following long-term application of poultry manure and sewage sludge on grassland. Commun. Soil Sci. Plant Anal. 2011, 42, 656–668. [Google Scholar] [CrossRef]
- Kemper, W.D.; Rosenau, R.C. Aggregate Stability and Size Distribution. In Methods of Soil Analysis. Part 1: Physical and Mineralogical Methods; Klute, A., Ed.; American Society of Agronomy: Madison, WI, USA, 1986; pp. 425–442. [Google Scholar]
- Dermott, W.; De Boodt, M. Dispersion ratio determination according to Dermott. West Eur. Methods Soil Struct. Determ. 1967, 71–72. [Google Scholar] [CrossRef]
- Niemi, R.; Vepsäläinen, M.; Wallenius, K.; Simpanen, S.; Alakukku, L.; Pietola, L. Temporal and soil depth-related variation in soil enzyme activities and in root growth of red clover (Trifolium pratense) and timothy (Phleum pratense) in the field. Appl. Soil Ecol. 2005, 30, 113–125. [Google Scholar] [CrossRef]
- Vepsäläinen, M.; Erkomaa, K.; Kukkonen, S.; Vestberg, M.; Wallenius, K.; Niemi, R.M. The impact of crop plant cultivation and peat amendment on soil microbial activity and structure. Plant Soil 2004, 264, 273–286. [Google Scholar] [CrossRef]
- Dick, R.P.; Kandeler, E. Enzymes in soils. In Encyclopedia of Soils in the Environment; Hillel, D., Hatfield, J.L., Eds.; Elsevier Academic Press: Amsterdam, The Netherlands, 2005; Volume 1, pp. 448–456. [Google Scholar]
- Gutknecht, J.L.M.; Henry, H.A.L.; Balser, T.C. Inter-annual variation in soil extra-cellular enzyme activity in response to simulated global change and fire disturbance. Pedobiologia 2010, 53, 283–293. [Google Scholar] [CrossRef]
- Bell, T.H.; Henry, H.A.L. Fine scale variability in soil extracellular enzyme activity is insensitive to rain events and temperature in a mesic system. Pedobiologia 2011, 54, 141–146. [Google Scholar] [CrossRef]
- Yan, J.; Pan, G.; Li, L.; Quan, G.; Ding, C.; Luo, A. Adsorption, immobilization, and activity of β-glucosidase on different soil colloids. J. Colloid Interface Sci. 2010, 348, 565–570. [Google Scholar] [CrossRef] [PubMed]
- Landgraf, D.; Klose, S. Mobile and readily available C and N fractions and their relationship to microbial biomass and selected enzyme activities in a sandy soil under different management systems. J. Plant Nutr. Soil Sci. 2002, 165, 9–16. [Google Scholar] [CrossRef]
- Sardans, J.; Peñuelas, J.; Estiarte, M. Changes in soil enzymes related to C and N cycle and in soil C and N content under prolonged warming and drought in a Mediterranean shrubland. Appl. Soil Ecol. 2008, 39, 223–235. [Google Scholar] [CrossRef]
- Dick, W. Organic carbon, nitrogen, and phosphorus concentrations and pH in soil profiles as affected by tillage intensity. Soil Sci. Soc. Am. J. 1983, 47, 102–107. [Google Scholar] [CrossRef]
- Soane, B.D.; Ball, B.C.; Arvidsson, J.; Basch, G.; Moreno, F.; Roger-Estrade, J. No-till in northern, western and south-western Europe: A review of problems and opportunities for crop production and the environment. Soil Tillage Res. 2012, 118, 66–87. [Google Scholar] [CrossRef]
- Zaman, M.; Di, H.; Cameron, K.; Frampton, C. Gross nitrogen mineralization and nitrification rates and their relationships to enzyme activities and the soil microbial biomass in soils treated with dairy shed effluent and ammonium fertilizer at different water potentials. Biol. Fertil. Soils 1999, 29, 178–186. [Google Scholar] [CrossRef]
- Paterson, E.; Osler, G.; Dawson, L.A.; Gebbing, T.; Sim, A.; Ord, B. Labile and recalcitrant plant fractions are utilised by distinct microbial communities in soil: Independent of the presence of roots and mycorrhizal fungi. Soil Biol. Biochem. 2008, 40, 1103–1113. [Google Scholar] [CrossRef]
- Griffiths, B.; Ball, B.; Daniell, T.; Hallett, P.; Neilson, R.; Wheatley, R.; Osler, G.; Bohanec, M. Integrating soil quality changes to arable agricultural systems following organic matter addition, or adoption of a ley-arable rotation. Appl. Soil Ecol. 2010, 46, 43–53. [Google Scholar] [CrossRef]
- Newton, A.C.; Bengough, A.G.; Guy, D.C.; McKenzie, B.M.; Hallett, P.D. Interactions between Barley Cultivars and Soil Cultivation—Effects on Yield and Disease. In Proceedings of the Crop Protection in Northern Britain, Dundee, UK, 23–24 February 2010; pp. 137–142. [Google Scholar]
- White, P.J.; George, T.S.; Gregory, P.J.; Bengough, A.G.; Hallett, P.D.; McKenzie, B.M. Matching roots to their environment. Ann. Bot. 2013, 112, 207–222. [Google Scholar] [CrossRef] [PubMed]
- James, B.R.; Riha, S.J. pH buffering in forest soil organic horizons: Relevance to acid precipitation. J. Environ. Qual. 1986, 15, 229–234. [Google Scholar] [CrossRef]
- Wick, B.; Kühne, R.F.; Vielhauer, K.; Vlek, P.L. Temporal variability of selected soil microbiological and biochemical indicators under different soil quality conditions in south-western Nigeria. Biol. Fertil. Soils 2002, 35, 155–167. [Google Scholar] [CrossRef]
- Tukey, J.W. One degree of freedom for non-additivity. Biometrics 1949, 5, 232–242. [Google Scholar] [CrossRef]
| Study Factors in Each Experiment | ||||
|---|---|---|---|---|
| Tillage Experiment | Organic Experiment | Grass-Reference | ||
| Zero (control) | (T0) | Control | (O0) | Grass-reference |
| Minimum | (T1) | Slurry | (O1) | |
| Conventional | (T2) | Compost | (O2) | |
| Deep | (T3) | |||
| Study variables in all experiments | ||||
| Soil Organic Matter content (SOM) | ||||
| Electric Conductivity (EC) | ||||
| pH | ||||
| Dispersion ratio | ||||
| Percentage of water stable aggregates | ||||
| β-glucosidase activity (BGa) | ||||
| Response ratio of β-glucosidase activity (BGa-ratio) | ||||
| Weather variables (air and soil temperature *, solar radiation * and precipitation) | ||||
| Treatment | Depth | SOM | EC | pH1:2.5 | Dispersion Ratio | Water Stable | 1st BGa-Ratio * | 2nd BGa-Ratio * | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (cm) | (%) | (µS cm−1) | Aggregates (%) | (%) | (%) | ||||||||||||||||||||||||
| T0 (zero) | 0–5 | 5.14 | ± | 0.29 | a† | 173 | ± | 16 | a | 5.42 | ± | 0.07 | c | 6.90 | ± | 0.91 | a | 98.8 | ± | 0.5 | a | 100.0 | ± | 5.9 | a A § | 100.0 | ± | 3.5 | a A |
| T1 (minimum) | 0–5 | 4.58 | ± | 0.29 | ab | 143 | ± | 16 | ab | 5.79 | ± | 0.07 | b | 7.47 | ± | 0.91 | a | 99.4 | ± | 0.5 | a | 93.8 | ± | 5.9 | a A | 105.0 | ± | 3.5 | a A |
| T2 (conventional) | 0–5 | 3.83 | ± | 0.29 | c | 103 | ± | 16 | b | 6.11 | ± | 0.07 | a | 4.60 | ± | 0.91 | ab | 94.1 | ± | 0.5 | b | 64.1 | ± | 5.9 | b A | 63.5 | ± | 3.5 | b A |
| T3 (deep) | 0–5 | 3.94 | ± | 0.29 | bc | 98 | ± | 16 | b | 6.18 | ± | 0.07 | a | 2.55 | ± | 0.91 | c | 95.4 | ± | 0.5 | b | 58.3 | ± | 5.9 | b A | 70.8 | ± | 3.5 | b A |
| O0 (control) | 0–5 | 4.52 | ± | 0.50 | b | 222 | ± | 59 | b | 5.84 | ± | 0.25 | b | 7.51 | ± | 0.47 | a | 94.2 | ± | 0.5 | b | 100.0 | ± | 4.7 | b A | 100.0 | ± | 4.8 | ab A |
| O1 (slurry) | 0–5 | 5.24 | ± | 0.50 | b | 276 | ± | 59 | b | 5.85 | ± | 0.25 | b | 9.41 | ± | 0.47 | a | 94.8 | ± | 0.5 | b | 122.0 | ± | 4.7 | a A | 113.8 | ± | 4.8 | a A |
| O2 (compost) | 0–5 | 15.18 | ± | 0.50 | a | 896 | ± | 59 | a | 6.74 | ± | 0.25 | a | 4.78 | ± | 0.47 | b | 96.9 | ± | 0.5 | a | 97.7 | ± | 4.7 | b A | 90.0 | ± | 4.8 | b A |
| Grass-reference | 0–5 | 4.56 | ± | 0.20 | 243 | ± | 23 | 6.00 | ± | 0.01 | 7.83 | ± | 5.12 | 99.4 | ± | 0.5 | N/A | N/A | |||||||||||
| T0 (zero) | 10–15 | 4.08 | ± | 0.10 | a | 98 | ± | 6 | a | 6.13 | ± | 0.02 | b | 7.09 | ± | 1.30 | a | 96.1 | ± | 1.1 | ab | 100.0 | ± | 13.9 | a A | 100.0 | ± | 4.6 | a A |
| T1 (minimum) | 10–15 | 4.03 | ± | 0.10 | a | 92 | ± | 6 | a | 6.07 | ± | 0.02 | b | 10.40 | ± | 1.30 | a | 96.2 | ± | 1.1 | a | 55.5 | ± | 13.9 | a A | 113.4 | ± | 4.6 | a A |
| T2 (conventional) | 10–15 | 3.90 | ± | 0.10 | a | 84 | ± | 6 | a | 6.23 | ± | 0.02 | a | 8.53 | ± | 1.30 | a | 92.7 | ± | 1.1 | b | 83.3 | ± | 13.9 | a A | 104.7 | ± | 4.6 | a A |
| T3 (deep) | 10–15 | 3.87 | ± | 0.10 | a | 78 | ± | 6 | a | 6.26 | ± | 0.02 | a | 9.33 | ± | 1.30 | a | 93.0 | ± | 1.1 | ab | 75.0 | ± | 13.9 | a B | 104.0 | ± | 4.6 | a A |
| O0 (control) | 10–15 | 4.42 | ± | 0.77 | b | 133 | ± | 38 | b | 5.85 | ± | 0.07 | b | 6.96 | ± | 0.62 | b | 94.8 | ± | 1.4 | b | 100.0 | ± | 2.5 | b A | 100.0 | ± | 9.7 | a A |
| O1 (slurry) | 10–15 | 4.62 | ± | 0.77 | b | 139 | ± | 38 | b | 5.98 | ± | 0.07 | b | 12.77 | ± | 0.62 | a | 93.3 | ± | 1.4 | b | 91.3 | ± | 2.5 | c A | 106.1 | ± | 9.7 | a A |
| O2 (compost) | 10–15 | 10.86 | ± | 0.77 | a | 470 | ± | 38 | a | 6.99 | ± | 0.07 | a | 8.48 | ± | 0.62 | b | 97.8 | ± | 1.4 | a | 115.4 | ± | 2.5 | a A | 107.4 | ± | 9.7 | a A |
| Grass-reference | 10–15 | 4.26 | ± | 0.11 | 439 | ± | 66 | 6.25 | ± | 0.04 | 9.81 | ± | 3.21 | 94.8 | ± | 0.8 | N/A | N/A | |||||||||||
| (a) | ||||||
|---|---|---|---|---|---|---|
| EC | pH | Dispersion Ratio | WSA % | BGa | BGa-Ratio | |
| SOM % | 0.90 ** | −0.80 ** | 0.66 * | 0.69 * | 0.82 ** | 0.68 * |
| EC | −0.90 ** | 0.64 * | 0.60 * | 0.85 ** | 0.75 * | |
| pH | −0.64 * | −0.60 * | −0.81 ** | −0.68 * | ||
| Dispersion Ratio | 0.67 * | 0.56 | 0.59 | |||
| WSA % | 0.77 * | 0.79 ** | ||||
| BGa | 0.91 ** | |||||
| (b) | ||||||
| SOM % | 0.80 * | 0.57 | −0.60 | 0.94 ** | −0.43 | −0.76 * |
| EC | 0.47 | −0.60 | 0.90 * | −0.22 | −0.61− | |
| pH | −0.37 | 0.56 | −0.32 | −0.56− | ||
| Dispersion Ratio | −0.64 | 0.67 | 0.92 * | |||
| WSA % | −0.24 | −0.71 * | ||||
| BGa | 0.76 * | |||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mariscal-Sancho, I.; Ball, B.; McKenzie, B. Influence of Tillage Practices, Organic Manures and Extrinsic Factors on β-Glucosidase Activity: The Final Step of Cellulose Hydrolysis. Soil Syst. 2018, 2, 21. https://doi.org/10.3390/soilsystems2020021
Mariscal-Sancho I, Ball B, McKenzie B. Influence of Tillage Practices, Organic Manures and Extrinsic Factors on β-Glucosidase Activity: The Final Step of Cellulose Hydrolysis. Soil Systems. 2018; 2(2):21. https://doi.org/10.3390/soilsystems2020021
Chicago/Turabian StyleMariscal-Sancho, Ignacio, Bruce Ball, and Blair McKenzie. 2018. "Influence of Tillage Practices, Organic Manures and Extrinsic Factors on β-Glucosidase Activity: The Final Step of Cellulose Hydrolysis" Soil Systems 2, no. 2: 21. https://doi.org/10.3390/soilsystems2020021
APA StyleMariscal-Sancho, I., Ball, B., & McKenzie, B. (2018). Influence of Tillage Practices, Organic Manures and Extrinsic Factors on β-Glucosidase Activity: The Final Step of Cellulose Hydrolysis. Soil Systems, 2(2), 21. https://doi.org/10.3390/soilsystems2020021





