Transformation or Progression from Adenocarcinoma to Small Cell Lung Cancer Detected by Serially Tracking Mutations in the Blood
Abstract
1. Introduction
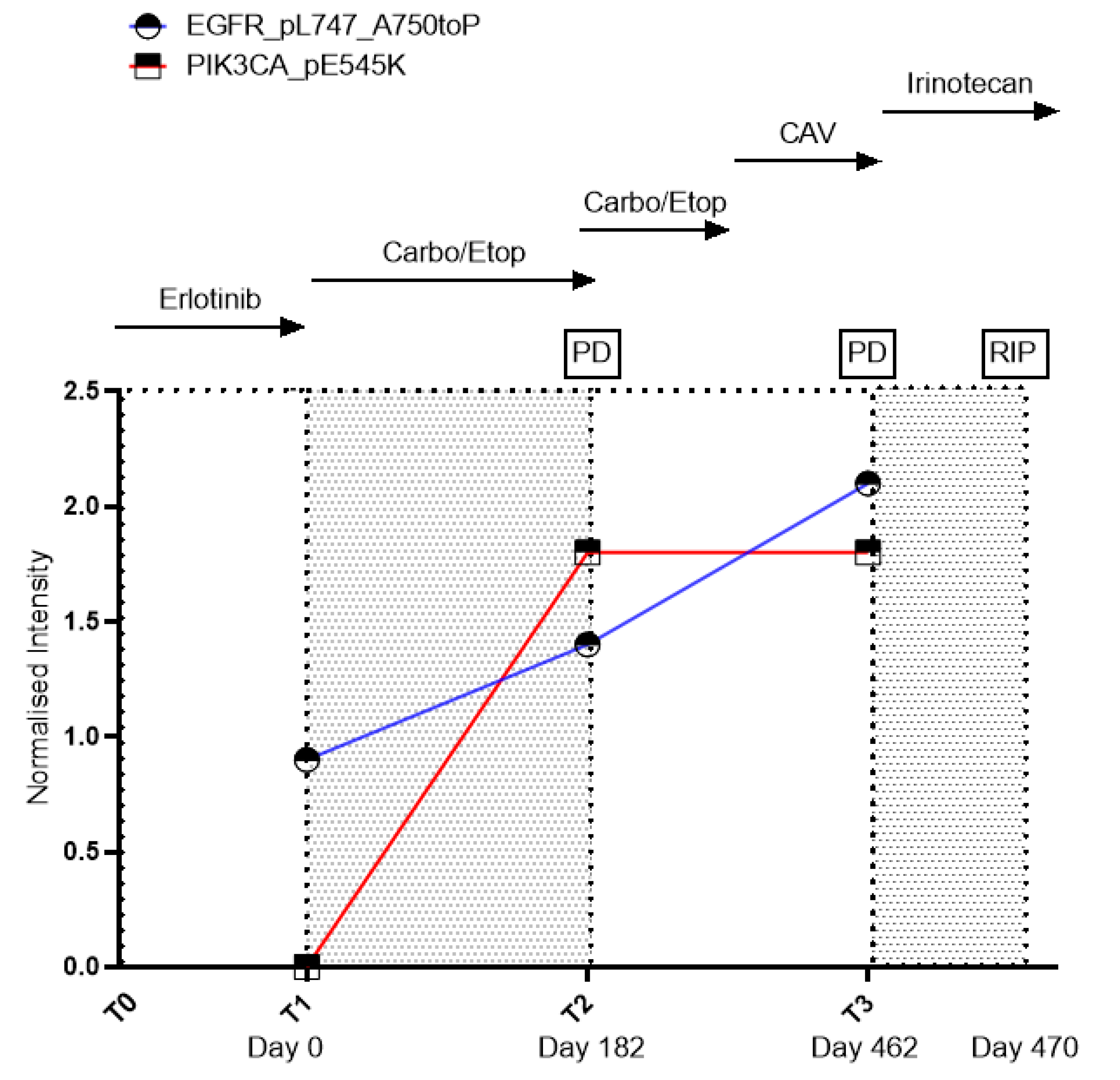
2. Case Report
3. Ethics Approvals
Funding
Conflicts of Interest
References
- Yu, H.A.; Arcila, M.E.; Rekhtman, N.; Sima, C.S.; Zakowski, M.F.; Pao, W.; Kris, M.G.; Miller, V.A.; Ladanyi, M.; Riely, G.J. Analysis of tumor specimens at the time of acquired resistance to EGFR-TKI therapy in 155 patients with EGFR-mutant lung cancers. Clin. Cancer Res. 2013, 19, 2240–2247. [Google Scholar] [CrossRef]
- Smyth, R.J.; Toomey, S.M.; Sartori, A.; O’Hanrahan, E.; Cuffe, S.D.; Breathnach, O.S.; Morgan, R.K.; Hennessy, B.T. Brief Report on the Detection of the EGFR T790M Mutation in Exhaled Breath Condensate from Lung Cancer Patients. J. Thorac. Oncol. 2018, 13, 1213–1216. [Google Scholar] [CrossRef]
- Truini, A.; Starrett, J.H.; Stewart, T.; Ashtekar, K.; Walther, Z.; Wurtz, A.; Lu, D.; Park, J.H.; DeVeaux, M.; Song, X.; et al. The EGFR Exon 19 Mutant L747–A750 > P Exhibits Distinct Sensitivity to Tyrosine Kinase Inhibitors in Lung Adenocarcinoma. Clin. Cancer Res. 2019, 25, 6382–6391. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kulasinghe, A.; Monkman, J.; Nalder, M.; O’Leary, C.; Ladwa, R.; O’Byrne, K. Transformation or Progression from Adenocarcinoma to Small Cell Lung Cancer Detected by Serially Tracking Mutations in the Blood. Reports 2020, 3, 33. https://doi.org/10.3390/reports3040033
Kulasinghe A, Monkman J, Nalder M, O’Leary C, Ladwa R, O’Byrne K. Transformation or Progression from Adenocarcinoma to Small Cell Lung Cancer Detected by Serially Tracking Mutations in the Blood. Reports. 2020; 3(4):33. https://doi.org/10.3390/reports3040033
Chicago/Turabian StyleKulasinghe, Arutha, James Monkman, Mark Nalder, Connor O’Leary, Rahul Ladwa, and Ken O’Byrne. 2020. "Transformation or Progression from Adenocarcinoma to Small Cell Lung Cancer Detected by Serially Tracking Mutations in the Blood" Reports 3, no. 4: 33. https://doi.org/10.3390/reports3040033
APA StyleKulasinghe, A., Monkman, J., Nalder, M., O’Leary, C., Ladwa, R., & O’Byrne, K. (2020). Transformation or Progression from Adenocarcinoma to Small Cell Lung Cancer Detected by Serially Tracking Mutations in the Blood. Reports, 3(4), 33. https://doi.org/10.3390/reports3040033





