Rett Syndrome: Treatment with IGF-I, Melatonin, Blackcurrant Extracts, and Rehabilitation
Abstract
1. Introduction
2. Case Presentation Section
2.1. Medical History
2.2. Blood Analysis
2.3. Medical Treatments
2.4. Rehabilitation and Results
2.4.1. Speech Therapy
2.4.2. Neurostimulation and Occupational Therapy
2.4.3. EINA
2.4.4. Physiotherapy
3. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rett, A. On an unusual brain atropic syndrome with hyperammonemia in childhood. Wien Med. Wochenschr. 1966, 116, 723–726. [Google Scholar] [PubMed]
- Hagberg, B.; Aicardi, J.; Dias, K.; Ramos, O. A progressive syndrome of autism, dementia, ataxia, and loss of purposeful hand use in girls: Rett’s syndrome: Report of 35 cases. Ann. Neurol. 1983, 14, 471–479. [Google Scholar] [CrossRef] [PubMed]
- Amir, R.E.; Van den Veyver, I.B.; Wan, M.; Tran, C.Q.; Francke, U.; Zoghbi, H.Y. Rett syndrome is caused by mutations in X-linked MECP2, encoding methyl-CpG-binding protein 2. Nat. Genet. 1999, 23, 185–188. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.D.; Meehan, R.R.; Henzel, W.J.; Maurer-Fogy, I.; Jeppesen, P.; Klein, F.; et al. Purification, sequence, and cellular localization of a novel chromosomal protein that binds to methylated DNA. Cell 1992, 69, 905–914. [Google Scholar] [CrossRef]
- D’Esposito, M.; Quaderi, N.A.; Ciccodicola, A.; Bruni, P.; Esposito, T.; D’Urso, M.; Brown, S.D. Isolation, physical mapping, and northern analysis of the X-linked human gene encoding methyl CpG-binding protein, MECP2. Mamm. Genome 1996, 7, 533–535. [Google Scholar] [CrossRef] [PubMed]
- Galvão, T.C.; Thomas, J.O. Structure-specific binding of MeCP2 to four-way junction DNA through its methyl CpG-binding domain. Nucleic Acids Res. 2005, 33, 6603–6609. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.U.; Su, Y.; Shin, J.H.; Shin, J.; Li, H.; Xie, B.; Zhong, C.; Hu, S.; Le, T.; Fan, G. Distribution, recognition and regulation of non-CpG methylation in the adult mammalian brain. Nat. Neurosci. 2014, 17, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Shahbazian, M.D.; Antalffy, B.; Armstrong, D.L.; Zoghbi, H.Y. Insight into Rett syndrome: MeCP2 levels display tissue- and cell-specific differences and correlate with neuronal maturation. Hum. Mol. Genet. 2002, 11, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Meehan, R.R.; Lewis, J.D.; Bird, A.P. Characterization of MeCP2, a vertebrate DNA binding protein with affinity for methylated DNA. Nucleic Acids Res. 1992, 20, 5085–5092. [Google Scholar] [CrossRef] [PubMed]
- Chechnik, G.; Meilijson, I.; Ruppin, E. Synaptic pruning in development: A computational account. Neural Comput. 1998, 10, 1759–1777. [Google Scholar] [CrossRef]
- Neul, J.L.; Kaufmann, W.E.; Glaze, D.G.; Christodolou, J.; Clarke, A.J.; Bahi-Buisson, N.; Leonard, H.; Bailey, M.E.; Schanen, N.C.; Zapella, M.; et al. Rett Syndrome: Revised Diagnostic Criteria and Nomenclature. Ann. Neurol. 2010, 68, 944–950. [Google Scholar] [CrossRef] [PubMed]
- Olson, H.E.; Tambunan, D.; LaCoursiere, C.; Godenberg, M.; Pinsky, R.; Martin, E.; Ho, E.; Khwaja, O.; Kaufmann, W.E.; Poduri, A. Mutations in Epilepsy and Intellectual Disability in Patients with Features of Rett Syndrome. Am. J. Med. Genet. A 2015, 167, 2017–2025. [Google Scholar] [CrossRef] [PubMed]
- Guerrini, R.; Parrini, E. Epilepsy in Rett syndrome, and CDL5- and FOXG1-gene-related encephalopathies. Epilepsia 2012, 53, 2067–2078. [Google Scholar] [CrossRef] [PubMed]
- Hagebeuk, E.E.; van den Bossche, R.A.; de Weerd, A.W. Respiratory and sleep disorders in female children with atypical Rett syndrome caused by mutations in the CDKL5 gene. Dev. Med. Child Neurol. 2013, 55, 480–484. [Google Scholar] [CrossRef] [PubMed]
- Das, D.K.; Mehta, B.; Menon, S.R.; Raha, S.; Udani, V. Novel mutations in cyclin-dependent kinase-like 5 (CDKL5) gene in Indian cases of Rett syndrome. Neuromol. Med. 2013, 15, 218–225. [Google Scholar] [CrossRef] [PubMed]
- Yuge, K.; Iwama, K.; Yonee, C.; Matsufuji, M.; Sano, N.; Saikusa, T.; Yae, Y.; Yamashita, Y.; Mizuguchi, T.; Matsumoto, N.; et al. A novel STXBP1 mutation causes typical Rett syndrome in a Japanese girl. Brain Dev. 2018, 40, 493–497. [Google Scholar] [CrossRef] [PubMed]
- Percy, A.K.; Lane, J.; Annese, F.; Warren, H.; Skinner, S.A.; Neul, J.L. When Rett syndrome is due to genes other than MECP2. Transl. Sci. Rare Dis. 2018, 13, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Ariani, F.; Hayek, G.; Rondinella, D.; Artuso, R.; Mencarelli, M.A.; Spanhol-Rosetto, A.; Pollazon, M.; Buoni, S.; Spiga, O.; Ricciardi, S.; et al. FOXG1 is responsible for the congenital variant of Rett syndrome. Am. J. Hum. Genet. 2008, 83, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Naidu, S.; Johnston, M.V. Neurodevelopmental disorders: Clinical criteria for Rett syndrome. Nat. Rev. Neurol. 2011, 7, 312–314. [Google Scholar] [CrossRef] [PubMed]
- Ehrhart, F.; Sangani, N.B.; Curfs, L.M.G. Current developments in the genetics of Rett and Rett-like syndrome. Curr. Opin. Psychiatry 2018, 3, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Neul, J.L.; Lane, J.B.; Lee, H.S.; Geerts, S.; Barrish, J.O.; Annese, F.; Baggett, L.M.; Barnes, K.; Skinner, S.M.; Motil, K.J.; et al. Developmental delay in Rett syndrome: Data from the natural history study. J. Neurodev. Disord. 2014, 6, 20. [Google Scholar] [CrossRef] [PubMed]
- Tokaji, N.; Ito, H.; Kohmoto, T.; Naruto, T.; Takahashi, R.; Goji, A.; Mori, T.; Toda, Y.; Saito, M.; Tange, S.; et al. A rare male patient with classic Rett syndrome cause by MeCP2_e1 mutation. Am. J. Med. Genet. 2018, 176, 699–702. [Google Scholar] [CrossRef] [PubMed]
- Tarquinio, D.C.; Hou, W.; Neul, J.L.; Kaufmann, W.E.; Glaze, D.G.; Motil, K.J.; Skinner, S.A.; Lee, H.S. The Changing Face of Survival in Rett Syndrome and MECP2-Related Disorders. Pediatr. Neurol. 2015, 53, 402–411. [Google Scholar] [CrossRef] [PubMed]
- Shioda, T.; Takahashi, S.; Kaname, T.; Yamauchi, T.; Fukuoka, T. MECP2 mutation in a boy with severe apnea and sick sinus syndrome. Brain Dev. 2018, 40, 714–718. [Google Scholar] [CrossRef] [PubMed]
- Leonard, H.; Bower, C. Is the girl with Rett syndrome normal at birth? Dev. Med. Child Neurol. 1988, 40, 115–121. [Google Scholar]
- Kerr, A.M. Early clinical signs in the Rett disorder. Neuropediatrics 1995, 26, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Katz, D.M.; Bird, A.; Coenraads, M.; Gray, S.J.; Menon, D.U.; Philpot, B.D.; Tarquinio, D.C. Rett Syndrome: Crossing the Treshold to Clinical Translation. Trends Neurosci. 2016, 39, 100–113. [Google Scholar] [CrossRef] [PubMed]
- Lonard, H.; Cobb, S.; Downs, J. Clinical and biological progress over 50 years in Rett syndrome. Nat. Rev. Neurol. 2017, 13, 37–51. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, H.M.; Kauffmann, W.E.; Barnes, K.V.; Rakesh, K.; Kapur, K.; Tarquinio, D.C.; Cantwell, N.G.; Roche, K.J.; Rose, S.A.; Walco, A.C.; et al. Placebo-controlled crossover assessment of mecasermin for the treatment of Rett syndrome. Ann. Clin. Transl. Neurol. 2018, 5, 323–332. [Google Scholar] [CrossRef] [PubMed]
- Pini, G.; Scusa, M.F.; Congiu, L.; Benincassa, A.; Morescalchi, P.; Bottiglioni, I.; Di Marco, P.; Borelli, P.; Bonuccelli, U.; Della-Chiesa, A.; et al. IGF1 as a Potential Treatment for Rett Syndrome: Safety Assessment in Six Rett Patients. Autism Res. Treat. 2012, 2012, 679801. [Google Scholar] [CrossRef] [PubMed]
- Pini, G.; Scusa, M.F.; Benincassa, A.; Bottiglioni, I.; Congiu, L.; Vadhatpour, C.; Romanelli, A.M.; Gemo, I.; Puccetti, C.; McNamara, R.; et al. Repeated insulin-like growth factor 1 treatment in a patient with Rett syndrome: A single case study. Front. Pediatr. 2014, 2, 52. [Google Scholar] [CrossRef] [PubMed]
- Belichenko, P.V.; Wright, E.E.; Belichenko, N.P.; Masliah, E.; Li, H.H.; Mobley, W.C.; Francke, U. Widespread changes in dendritic and axonal morphology in Mecpp2-mutant mouse models of Rett syndrome: Evidence for disruption of neuronal networks. J. Comp. Neurol. 2009, 514, 240–258. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Cerdeño, V. Dendrite and spine modifications in autism and related neurodevelopmental disorders in patients and animal models. Dev. Neurobiol. 2017, 77, 419–437. [Google Scholar] [CrossRef] [PubMed]
- Glaze, D.G.; Neul, J.L.; Percy, A.; Feyma, T.; Beisang, A.; Yaroshinsky, A.; Zuchero, D.; Horrigan, J.; Glass, L.; Jones, N.E. A Double-Blind, Randomized, Placebo-Controlled Clinical Study of Trofinetide in the Treatment of Rett Syndrome. Pediatr. Neurol. 2017, 76, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Eeg-Olofsson, O.; Al-Zuhair, A.G.; Teebi, A.S.; Al-Essa, M.M. Abnormal mitochondria in the Rett syndrome. Brain Dev. 1988, 10, 260–262. [Google Scholar] [CrossRef]
- Shulyakova, N.; Andreazza, A.C.; Mills, L.R.; Eubanks, J.H. Mitochondrial Dysfunction in the Pathogenesis of Rett syndrome: Implications for Mitochondria-Targeted Therapies. Front. Cell Neurosci. 2017, 11, 58. [Google Scholar] [CrossRef] [PubMed]
- Hoffbuhr, K.; Devaney, J.M.; LaFleur, B.; Sirianni, N.; Scacheri, C.; Giron, J.; Schuette, J.; Innis, J.; Marino, M.; Philippart, M.; et al. MeCP2 mutations in children with and without the phenotype of Rett syndrome. Neurology 2001, 56, 1486–1495. [Google Scholar] [CrossRef] [PubMed]
- Matijevic, T.; Knezevic, J.; Slavica, M.; Pavelic, J. Rett syndrome: From the gene to the disease. Eur. Neurol. 2009, 61, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Zhao, P.W.; He, X.L.; Lin, J.; Wu, G.F.; Yue, X.; Bi, B.; Hu, J.S.; Liu, Z.S. Clinical features and MECP2 mutations in children with Rett syndrome. Zhongguo Dang Dai Er Ke Za Zhi 2014, 16, 393–396. [Google Scholar] [PubMed]
- Killian, J.T.; Lane, J.B.; Cutter, G.R.; Skinner, S.A.; Kaufmann, W.E.; Tarquinio, D.C.; Glaze, D.G.; Motil, K.J.; Neul, J.L.; Percy, A.K. Pubertal Development in Rett Syndrome Deviates from Typical Females. Pediatr. Neurol. 2014, 51, 769–775. [Google Scholar] [CrossRef] [PubMed]
- Quintana, A.; Agra, C.; Outeiral, L.; Devesa, A.; Llorente, D.; Devesa, J. Cognitive evolution of a patient who suffered a Subarachnoid Haemorrhage eight years ago, after being treated with Growth Hormone, Melatonin and Neurorehabilitation. Reports 2018, 1, 2. [Google Scholar] [CrossRef]
- Hadzsiev, K.; Polgar, N.; Bene, J.; Komlosi, K.; Karteszi, J.; Hollody, K.; Kosztolanyi, G.; Renieri, A.; Melegh, B. Analysis of Hungarian patients with Rett syndrome phenotype for MECP2, CDKL5 and FOXG1 gene mutations. J. Hum. Genet. 2011, 56, 183–187. [Google Scholar] [CrossRef] [PubMed]
- Kaufmann, W.E.; Johnston, M.V.; Blue, M.E. MeCP2 expression and function during brain development: Implications for Rett syndrome’s pathogenesis and clinical evolution. Brain Dev. 2005, 27 (Suppl. 1), 77–87. [Google Scholar] [CrossRef] [PubMed]
- Nelson, S.B.; Valakh, V. Excitatory/Inbitory Balance and Circuit Homeostasis in Autism Spectrum Disorders. Neuron 2015, 87, 684–698. [Google Scholar] [CrossRef] [PubMed]
- Ballestar, E.; Yusufzai, T.M.; Wolffe, A.P. Effects of Rett syndrome mutations of the methyl-CpG binding domain of the transcriptional repressor MeCP2 on selectivity for association with methylated DNA. Biochemistry 2000, 39, 7100–7106. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Miller, E.C.; Pozzo-Miller, L. Dendritic spine dysgenesis in Rett syndrome. Front. Neuroanat. 2014, 8, 97. [Google Scholar] [CrossRef] [PubMed]
- Chang, Q.; Khare, G.; Dani, V.; Nelson, S.; Jaenisch, R. The disease progression of Mecp2 mutant mice is affected by the level of BDNF expression. Neuron 2006, 49, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Abuhatzira, L.; Makendoski, K.; Kaufman, Y.; Razin, A.; Shemer, R. MeCP2 deficiency in the brain decreases BDNF levels by REST/CoREST-mediated repression and increases TRKB production. Epigenetics 2007, 2, 214–222. [Google Scholar] [CrossRef] [PubMed]
- Giacometti, E.; Luikenhuis, S.; Beard, C.; Jaenisch, R. Partial rescue of MeCP2 deficiency by postnatal activation of MeCP2. Proc. Natl. Acad. Sci. USA 2007, 104, 1931–1936. [Google Scholar] [CrossRef] [PubMed]
- Guy, J.; Gan, J.; Selfridge, J.; Cobb, S.; Bird, A. Reversal of neurological defects in a mouse model of Rett syndrome. Science 2007, 315, 1143–1147. [Google Scholar] [CrossRef] [PubMed]
- Landi, S.; Putignano, E.; Boggio, E.M.; Giustetto, M.; Pizzorusso, T.; Ratto, G.M. The short-time structural plasticity of dendritic spines is altered in a model of Rett syndrome. Sci. Rep. 2011, 1, 45. [Google Scholar] [CrossRef] [PubMed]
- Tropea, D.; Giacometti, E.; Wilson, N.R.; Beard, C.; McCurry, C.; Fu, D.D.; Flannery, R.; Jaenisch, R.; Sur, M. Partial reversal of Rett Syndrome-like symptoms in MeCP2 mutant mice. Proc. Natl. Acad. Sci. USA 2009, 106, 2029–2034. [Google Scholar] [CrossRef] [PubMed]
- Castro, J.; García, R.I.; Kwok, S.; Banerjee, A.; Petravicz, J.; Woodson, J.; Mellios, N.; Tropea, D.; Sur, M. Functional recovery with recombinant human IGF1 treatment in a mouse model of Rett Syndrome. Proc. Natl. Acad. Sci. USA 2014, 111, 9941–9946. [Google Scholar] [CrossRef] [PubMed]
- Khwaja, O.S.; Ho, E.; Barnes, K.V.; O’Leary, H.M.; Pereira, L.M.; Finkelstein, Y.; Nelson, C.A., 3rd; Vogel-Farley, V.; DeGregorio, G.; Holm., I.A.; et al. Safety, pharmacokinetics, and preliminary assessment of efficacy of mecasermin (recombinant human IGF-1) for the treatment of Rett syndrome. Proc. Natl. Acad. Sci. USA 2014, 111, 4596–4601. [Google Scholar] [CrossRef] [PubMed]
- Pini, G.; Congiu, L.; Benincasa, A.; DiMarco, P.; Bigoni, S.; Dyer, A.H.; Mortimer, N.; Della-Chiesa, A.; O'Leary, S.; McNamara, R.; et al. Illness Severity, Social and Cognitive Analysis, and EEG Analysis of Ten Patients with Rett Syndrome Treated with Mecasermin (Recombinant Human IGF-1). Autism Res. Treat. 2016, 2016, 5073078. [Google Scholar] [CrossRef] [PubMed]
- Riikonen, R. Treatment of autistic spectrum disorder with insulin-like growth factors. Eur. J. Paediatr. Neurol. 2016, 20, 816–823. [Google Scholar] [CrossRef] [PubMed]
- Pathipati, P.; Gorba, T.; Scheepens, A.; Goffin, V.; Sun, Y.; Fraser, M. Growth hormone and prolactin regulate human neural stem cell regenerative activity. Neuroscience 2011, 190, 409–427. [Google Scholar] [CrossRef] [PubMed]
- McGinley, L.M.; Sims, E.; Lunn, J.S.; Kashlan, O.N.; Chen, K.S.; Bruno, E.S.; Pacut, C.M.; Hazel, T.; Johe, K.; Sakowski, S.A.; et al. Human cortical neural stem cells expressing Insulin-Like Growth Factor-I: A Novel Cellular Therapy for Alzheimer’s Disease. Stem Cells Transl. Med. 2016, 5, 379–391. [Google Scholar] [CrossRef] [PubMed]
- Bondy, C.; Werner, H.; Roberts, C.T., Jr.; LeRoith, D. Cellular pattern of type-I insulin-like growth factor receptor gene expression during maturation of the rat brain: Comparison with insulin-like growth factors I and II. Neuroscience 1992, 46, 909–923. [Google Scholar] [CrossRef]
- Fernández, A.M.; Torres-Alemán, I. The many faces of insulin-like peptide signaling in the brain. Nat. Rev. Neurosci. 2012, 13, 225–239. [Google Scholar] [CrossRef] [PubMed]
- Costales, J.; Kolevzon, A. The Therapeutic Potential of Insulin-Like Growth Factor-1 in Central Nervous System Disorders. Neurosci. Biobehav. Rev. 2016, 63, 207–222. [Google Scholar] [CrossRef] [PubMed]
- O’Kusky, J.; Ye, P. Neurodevelopmental effects of insulin-like growth factor signaling. Front. Neuroendocrinol. 2012, 33, 230–251. [Google Scholar] [CrossRef] [PubMed]
- Huppke, P.; Roth, C.; Christen, H.J.; Brockmann, K.; Hanefeld, F. Endocrinological study on growth retardation in Rett syndrome. Acta Paediatr. 2001, 90, 1257–1261. [Google Scholar] [CrossRef] [PubMed]
- Hara, M.; Nishi, Y.; Yamashita, Y.; Hirata, R.; Takahashi, S.; Nagamitsu, S.; Hosoda, H.; Kangawa, K.; Kojima, M.; Matsuishi, T. Relation between circulating levels of GH, IGF-1, ghrelin and somatic growth in Rett syndrome. Brain Dev. 2014, 36, 794–800. [Google Scholar] [CrossRef] [PubMed]
- Devesa, J.; Almengló, C.; Devesa, P. Multiple Effects of Growth hormone in the Body: Is it Really the Hormone for Growth? Clin. Med. Insights Endocrinol. Diabetes 2016, 9, 47–71. [Google Scholar] [CrossRef] [PubMed]
- Tarquinio, D.C.; Motil, K.J.; Hou, W.; Lee, H.S.; Glaze, D.G.; Skinner, S.A.; Neul, J.L.; Annese, F.; McNair, L.; Barrish, J.O.; et al. Growth failure and outcome in Rett syndrome: Specific growth references. Neurology 2012, 76, 1653–1661. [Google Scholar] [CrossRef] [PubMed]
- Ranke, M.B. Insulin-like growth factor binding-protein-3 (IGFBP-3). Best Pract. Res. Clin. Endocrinol. Metab. 2015, 29, 701–711. [Google Scholar] [CrossRef] [PubMed]
- Conti, V.; Gandaglia, A.; Galli, F.; Tirone, M.; Bellini, E.; Campana, L.; Kilstrup-Nielsen, C.; Rovere-Querini, P.; Brunelli, S.; Landsberger, N. MeCP2 Affects Skeletal Muscle Growth and Morphology through Non Cell-Autonomous Mechanisms. PLoS ONE 2015, 10, e0130183. [Google Scholar] [CrossRef] [PubMed]
- Poling, J.S.; Frye, R.E.; Shoffner, J.; Zimmerman, A.W. Developmental regression and Mitochondrial Dysfunction in a Child with Autism. J. Child Neurol. 2006, 21, 170–172. [Google Scholar] [CrossRef] [PubMed]
- De Felice, C.; Signorini, C.; Leoncini, S.; Pecorelli, A.; Durand, T.; Valacchi, G.; Ciccoli, L.; Hayek, J. Oxidative stress and Rett syndrome. Minerva Pediatr. 2014, 66, 41–62. [Google Scholar] [PubMed]
- Filosa, S.; Pecorelli, A.; D’Esposito, M.; Valacchi, G.; Hajek, J. Exploring the possible link between MeCP2 and oxidative stress in Rett syndrome. Free Radic. Biol. Med. 2015, 88, 81–90. [Google Scholar] [CrossRef] [PubMed]
- De Filippis, B.; Valenti, D.; de Bari, L.; De Rasmo, D.; Musto, M.; Fabbri, A.; Ricceri, L.; Fiorentini, C.; Laviola, G.; Vacca, R.A. Mitochondrial free radical overproduction due to respiratory chain impairment in the brain of a mouse model of Rett syndrome: Protective effect of CNF1. Free Radic. Biol. Med. 2015, 83, 167–177. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J. Functional aspects of the pineal hormone melatonin in combating cell and tissue damage induced by free radicals. Eur. J. Endocrinol. 1996, 134, 412–430. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J. Antioxidant actions of melatonin. Adv. Pharmacol. 1997, 38, 103–117. [Google Scholar] [PubMed]
- Sánchez, A.; Calpena, A.C.; Clares, B. Evaluating the Oxidative Stress in Inflammation. Int. J. Mol. Sci. 2015, 16, 16981–17004. [Google Scholar] [CrossRef] [PubMed]
- Hardeland, R.; Cardinali, D.P.; Brown, G.M.; Pandi-Perumal, S.R. Melatonin and brain inflammaging. Prog. Neurobiol. 2015, 127–128, 46–63. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Fan, C.; Hu, W.; Jiang, S.; Ma, Z.; Yan, X.; Deng, C.; Di, S.; Xin, Z.; Wu, G.; et al. Melatonin attenuated early brain injury induced by subarachnoid hemorrhage via regulating NLRP3 inflammasome and apoptosis signaling. J. Pineal Res. 2016, 60, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Cardinali, D.P.; Vigo, D.E. Melatonin, mitochondria, and the metabolic syndrome. Cell Mol. Sci. 2017, 74, 3941–3954. [Google Scholar] [CrossRef] [PubMed]
- Favero, G.; Franceschetti, L.; Bonomini, F.; Rodella, L.F.; Rezzani, R. Melatonin as an Anti-Inflammatory Agent Modulating Inflammasome Activation. Int. J. Endocrinol. 2017, 2017, 1835195. [Google Scholar] [CrossRef] [PubMed]
- Carrascal, L.; Nunez-Abades, P.; Ayala, A.; Cano, M. Role of melatonin in the inflammatory process and its therapeutic potential. Curr. Pharm. Des. 2018, in press. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, M.I.; Carretero, M.; Escames, G.; López, L.C.; Maldonado, M.D.; Tan, D.X.; Reiter, R.J.; Acuña-Castroviejo, D. Chronic melatonin treatment prevents age-dependent cardiac mitochondrial dysfunction in senescence-accelerated mice. Free Radic. Res. 2007, 41, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Sara, V.R.; Carlsson-Skwirut, C.; Bergman, T.; Jörnvall, H.; Roberts, P.J.; Crawford, M.; Hakansson, L.N.; Civalero, I.; Nordberg, A. Identification of Gly-Pro-Glu (GPE), the aminoterminal tripeptide of insulin-like growth factor 1 which is truncated in brain, as a novel neuroactive peptide. Biochem. Biophys. Res. Commun. 1989, 165, 766–771. [Google Scholar] [CrossRef]
- Yamamoto, H.; Murphy, L.J. Enzymatic conversion of IGF-I to des (1–3) IGF-I in rat serum and tissues: A further potential site of growth hormone regulation of IGF-I action. J. Endocrinol. 1995, 146, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, H.; Murphy, L.J. N-terminal truncated insulin-like growth factor-I in human urine. J. Clin. Endocrinol. Metab. 1995, 80, 1179–1183. [Google Scholar] [PubMed]
- Almengló, C.; Devesa, P.; Devesa, J.; Arce, V. GPE promotes the proliferation and migration of mouse embryonic neural stem cells and their progeny in vitro. Int. J. Mol. Sci. 2017, 18, 1280. [Google Scholar] [CrossRef] [PubMed]
- Guan, J.; Gluckman, P.; Yang, P.; Krissansen, G.; Sun, X.; Zhou, Y.; Wen, J.; Phillips, G.; Shorten, P.R.; McMahon, C.D.; et al. Cyclic glycine-proline regulates IGF-1 homeostasis by altering the binding of IGFBP-3 to IGF-1. Sci. Rep. 2014, 4, 4388. [Google Scholar] [CrossRef] [PubMed]
- Sing-Mallah, G.; McMahon, C.D.; Guan, J.; Singh, K. Cyclic-glycine-proline accelerates mammary involution by promoting apoptosis and inhibiting IGF-1 function. J. Cell Physiol. 2017, 232, 3369–3383. [Google Scholar] [CrossRef] [PubMed]
- Singh-Mallah, G.; Sing, K.; McMahon, C.D.; Harris, P.; Brimble, M.A.; Thorstensen, E.; Guan, J. Maternally administered Cyclic Glycine-Proline increases Insulin-Like Growth Factor-1 Bioavailability and Novelty Recognition in Developing Offspring. Endocrinology 2016, 157, 3130–3139. [Google Scholar] [CrossRef] [PubMed]
- Tapiero, H.; Tew, K.D.; Ba, G.N.; Mathe, G. Polyphenols: Do they play a role in the prevention of human pathologies? Biomed. Pharmacother. 2002, 56, 200–207. [Google Scholar] [CrossRef]
- Ghosh, D.K. Anthocyanins and anthocyanin-rich extracts in biology and medicine: Biochemical, cellular and medicinal properties. Curr. Top. Nutraceut. Res. 2005, 3, 113–124. [Google Scholar]
- Kim, M.J.; Rehman, S.U.; Amin, F.U.; Kim, M.O. Enhanced neuroprotection of anthocyanin-loaded PEG-gold nanoparticles against Aß1-42-induced neuroinflammation and neurodegeneration via the NF-KB/JNK/GSK3ß signaling pathway. Nanomedicine 2017, 13, 2533–2544. [Google Scholar] [CrossRef] [PubMed]
- Winter, A.N.; Ross, E.K.; Wilkins, H.M.; Stankiewicz, T.R.; Wallace, T.; Miller, K.; Linseman, D.A. An anthocyanin-enriched extract from strawberries delays disease onset and extends survival in the hSOD1G93A mouse model of amyotrophic lateral sclerosis. Nutr. Neurosci. 2018, 21, 414–426. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.S.; Ali, T.; Kim, M.W.; Jo, M.H.; Chung, J.I.; Kim, M.O. Anthocyanins Improve Hippocampus-Dependent Memory Function and Prevent Neurodegeneration via JNK/Akt/GSK3ß Signaling in LPS-Treated Adult Mice. Mol. Neurobiol. 2018, in press. [Google Scholar] [CrossRef] [PubMed]
- Fan, D.; Alamri, Y.; Liu, K.; MacAskill, M.; Harris, P.; Brimble, M.; Dalrympe-Alford, J.; Prickett, T.; Menzies, O.; Laurenson, A.; et al. Supplementation of Blackcurrant Anthocyanins Increased Cyclic Glicine-Proline in the Cerebrospinal Fluid of Parkinson Patients: Potential Treatment to Improve Insulin-Like Growth Factor-1 Function. Nutrients 2018, 10, 714. [Google Scholar] [CrossRef] [PubMed]
- Bas, V.N.; Çetinkaya, S.; Ağladıoğlu, S.Y.; Aksoy, A.; Gülpınar, B.; Aycan, Z. Report of the first case of precocious puberty in Rett syndrome. J. Pediatr. Endocrinol. Metab. 2013, 26, 937–939. [Google Scholar] [CrossRef] [PubMed]
- Ben-Schachar, S.; Chahrour, M.; Thaller, C.; Shaw, C.A.; Zoghbi, H.Y. Mouse models of MeCP2 disorders share gene expression changes in the cerebellum and hypothalamus. Hum. Mol. Genet. 2009, 18, 2431–2442. [Google Scholar] [CrossRef] [PubMed]
- Westberry, J.M.; Trout, A.L.; Wilson, M.E. Epigenetic regulation of estrogen receptor alpha gene expression in the mouse cortex during early postnatal development. Endocrinology 2010, 151, 731–740. [Google Scholar] [CrossRef] [PubMed]
- Devesa, J.; Lima, L.; Tresguerres, J.A. Neuroendocrine control of growth hormone secretion in humans. Trends Endocrinol. Metab. 1992, 3, 175–183. [Google Scholar] [CrossRef]
- Keshin, M.; Erdeve, S.S.; Aycan, Z. Rett syndrome and precocius puberty association. J. Pediatr. Endocrinol. Metab. 2015, 28, 1197. [Google Scholar]
- McGill, B.E.; Bundle, S.F.; Yaylaoglu, M.B.; Carson, J.P.; Thaller, C.; Zoghbi, H.Y. Enhanced anxiety and stress-induced corticosterone release are associated with increased Crh expression in a mouse model of Rett syndrome. Proc. Natl. Acad. Sci. USA 2006, 103, 18267–18272. [Google Scholar] [CrossRef] [PubMed]
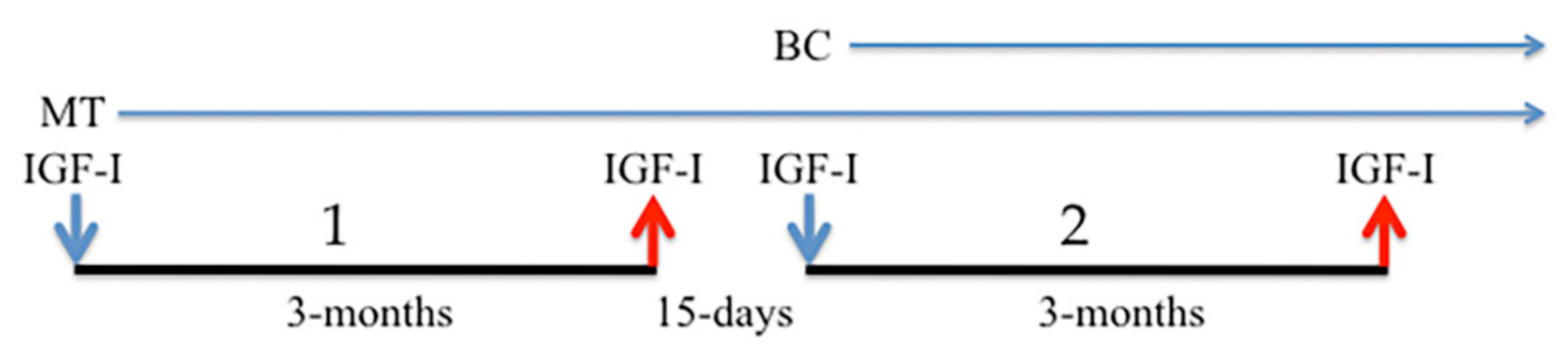
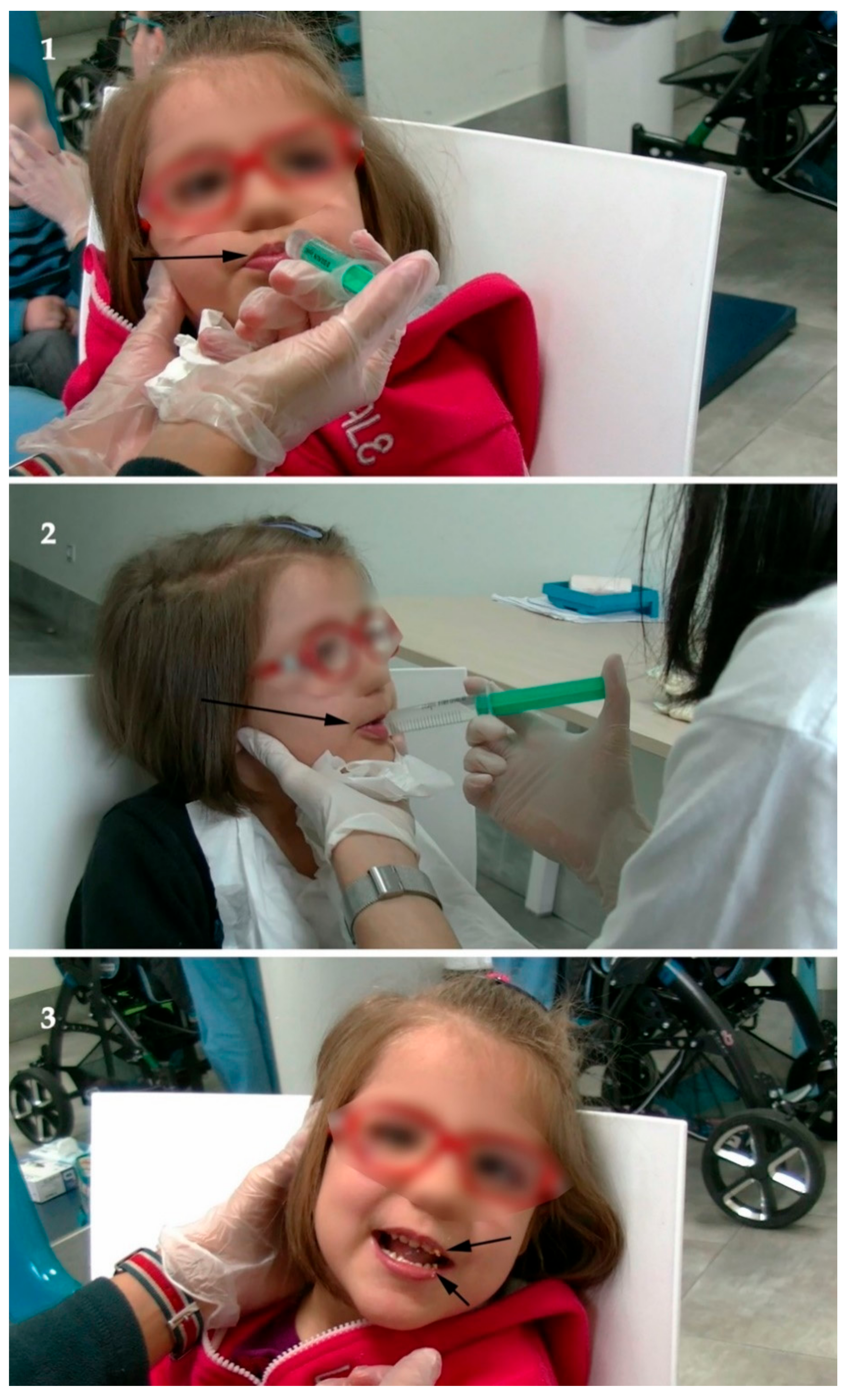


| Parameter | Admission | 3-Months | 6-Months | Normal Values |
|---|---|---|---|---|
| Erythrocytes | 5.37 × 106 | 4.12 × 106 | 5.1 × 106 | 3.9–4.6/µL (106) |
| Hematocrit | 42.2 | 39.7 | 41.8 | 35–41.5% |
| Hemoglobin | 14.40 | 12.3 | 14.2 | 11–14.2 g/dL |
| Leukocytes | 6.700 | 5.850 | 6.280 | 5.3–12.2/µL (103) |
| Platelets | 278 × 103 | 315 × 103 | 301 × 103 | 150–370/µL (103) |
| Glucose | 76 | 79 | 81 | 70–105 mg/dL |
| Cholesterol (total) | 166 | 172 | 180 | <200 mg/dL |
| Triglycerides | 68 | 83 | 92 | <150 mg/dL |
| Creatinine | 0.8 | 0.6 | 0.7 | 0.55–1 mg/dL |
| GOT | 19 | 17 | 22 | <31 U/L |
| GPT | 15 | 21 | 18 | <34 U/L |
| GGT | 29 | 25 | 23 | <42 U/L |
| CPK | 315.2 | 271 | 195 | 20–195 U/L |
| TSH | 2.1 | 2.8 | 1.9 | 0.4–3.7 µIU/mL |
| fT4 | 0.99 | 1.1 | 1.3 | 0.8–1.85 ng/dL |
| Cortisol | 16.5 | 19 | 18.4 | 5–25 µg/dL |
| IGF-I | 68 | 166 | 185 | 55–248 ng/mL |
| IGFBP3 | 4.8 | 3.4 | 2.7 | 2.6–5.8 ng/mL |
| FSH | 0.80 | 3.30 | 4.1 | 0.31–4.84 mU/mL |
| LH | <0.1 | 0.1 | 1.1 | 0.1–0.53 mU/mL |
| Estradiol | <5 | 8 | 12 | 5–12.3 pg/mL |
| Testosterone | 3.98 | <0.025 | 3.2 | 0.04–0.14 ng/mL |
| DHEA-S | 196 | 212 | 357 | 78–1100 ng/mL |
| Androstenedione | 52 | 67 | 92 | 43–176 ng/dL |
| Tanner stage | II | I | III | |
| Treatment | None | IGF-I + MT | IGF-I + MT + BC |
| Admission | Discharge |
|---|---|
| 3 | 1 |
| AREA | PRE- | POST- |
|---|---|---|
| Social/Personal | 0 | 2 |
| Adaptive | 1 | 3 |
| Gross motor | 6 | 8 |
| Fine motor | 0 | 2 |
| TOTAL MOTOR | 3 | 6 |
| Receptive communication | 1 | 5 |
| Expressive communication | 1 | 2 |
| TOTAL COMMUNICATION | 1 | 3 |
| Cognition | 1 | 4 |
| TOTAL | 2 | 3 |
| Disorders | Pre- | 1 | 2 |
|---|---|---|---|
| Hand skills | +++ | ++ | + |
| Acquired spoken language | +++ | +++ | −/+ |
| Gait abnormalities | + | − | − |
| Stereotypic hand movements | +++ | ++ | − |
| Breathing disturbances | +++ | ++ | + |
| Bruxism | +++ | − | − |
| Impaired sleep pattern | ++ | − | − |
| Hypotonia | ++ | + | − |
| Cognition | +++ | ++ | + |
| Scoliosis/Kyphosis | + | + | + |
| Growth retardation | + | − | − |
| Social interaction and communication | +++ | ++ | − |
| Chew/Crush | +++ | ++ | − |
| Drooling | ++ | + | − |
| Rejection of warm liquids | +++ | ++ | − |
| Rejection of textures and flavors | +++ | ++ | − |
| Memory | +++ | ++ | + |
| Perceptive discrimination | +++ | ++ | + |
| Tracking objects | +++ | ++ | − |
| Seizures | − | − | − |
| ECG | − | − | − |
| Tanner stage | 2 | 1 | 3 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Devesa, J.; Devesa, O.; Carrillo, M.; Casteleiro, N.; Devesa, A.; Llorente, D.; González, C. Rett Syndrome: Treatment with IGF-I, Melatonin, Blackcurrant Extracts, and Rehabilitation. Reports 2018, 1, 14. https://doi.org/10.3390/reports1020014
Devesa J, Devesa O, Carrillo M, Casteleiro N, Devesa A, Llorente D, González C. Rett Syndrome: Treatment with IGF-I, Melatonin, Blackcurrant Extracts, and Rehabilitation. Reports. 2018; 1(2):14. https://doi.org/10.3390/reports1020014
Chicago/Turabian StyleDevesa, Jesús, Olga Devesa, María Carrillo, Nerea Casteleiro, Ana Devesa, David Llorente, and Cristina González. 2018. "Rett Syndrome: Treatment with IGF-I, Melatonin, Blackcurrant Extracts, and Rehabilitation" Reports 1, no. 2: 14. https://doi.org/10.3390/reports1020014
APA StyleDevesa, J., Devesa, O., Carrillo, M., Casteleiro, N., Devesa, A., Llorente, D., & González, C. (2018). Rett Syndrome: Treatment with IGF-I, Melatonin, Blackcurrant Extracts, and Rehabilitation. Reports, 1(2), 14. https://doi.org/10.3390/reports1020014






