Ultrastructural Remodeling of the Neurovascular Unit in the Female Diabetic db/db Model—Part I: Astrocyte
Abstract
:1. Introduction
2. Methods
2.1. Animal Studies
2.2. Tissue Collection and Preparation for Transmission Electron Microscopy
3. Results
3.1. Endothelial Cell Remodeling of the Neurovascular Unit
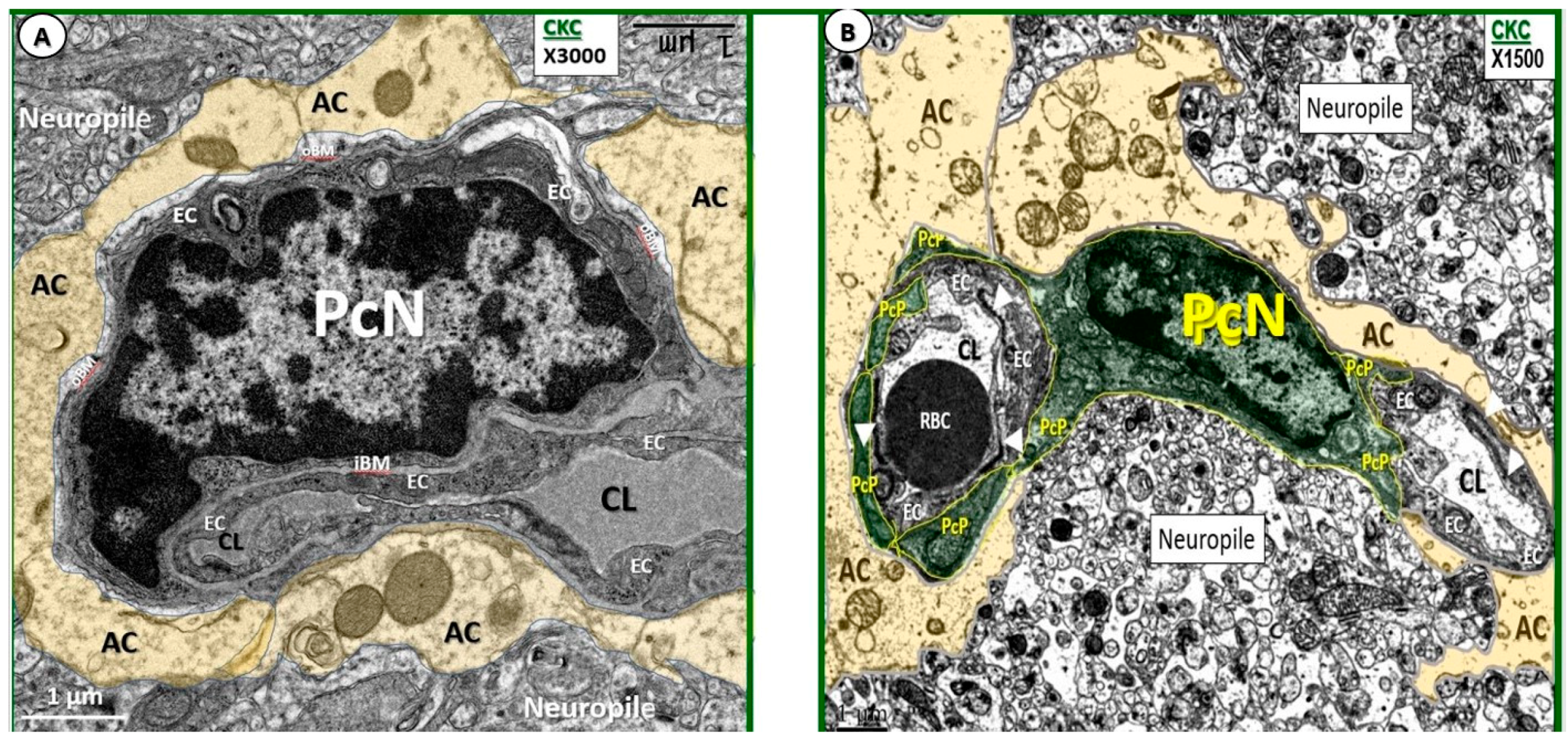
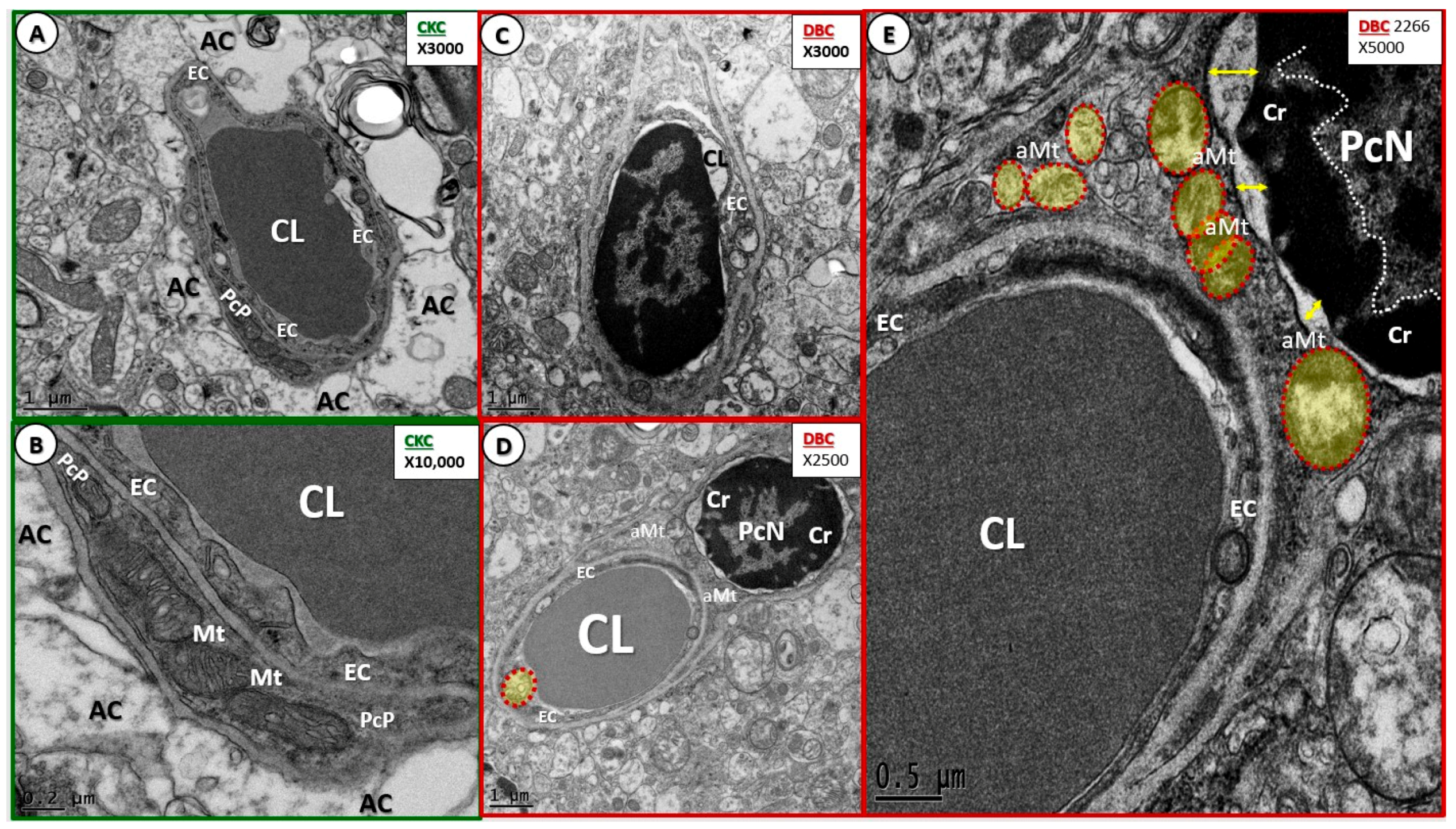
3.2. Remodeling of Pericytes and Pericyte Foot Processes
3.3. Protoplasmic Astrocyte Remodeling in Cortical Gray Matter Diabetic DBC Models
3.4. Sticky, Adhesive Red Blood Cells in the Neurovascular Unit of Diabetic DBC Mice
3.5. Neurovascular Unit Microbleeds in the Diabetic DBC Mice
3.6. Nanometer Channels as Possible Origins of the Glymphatic Pathway
4. Discussion
- i.
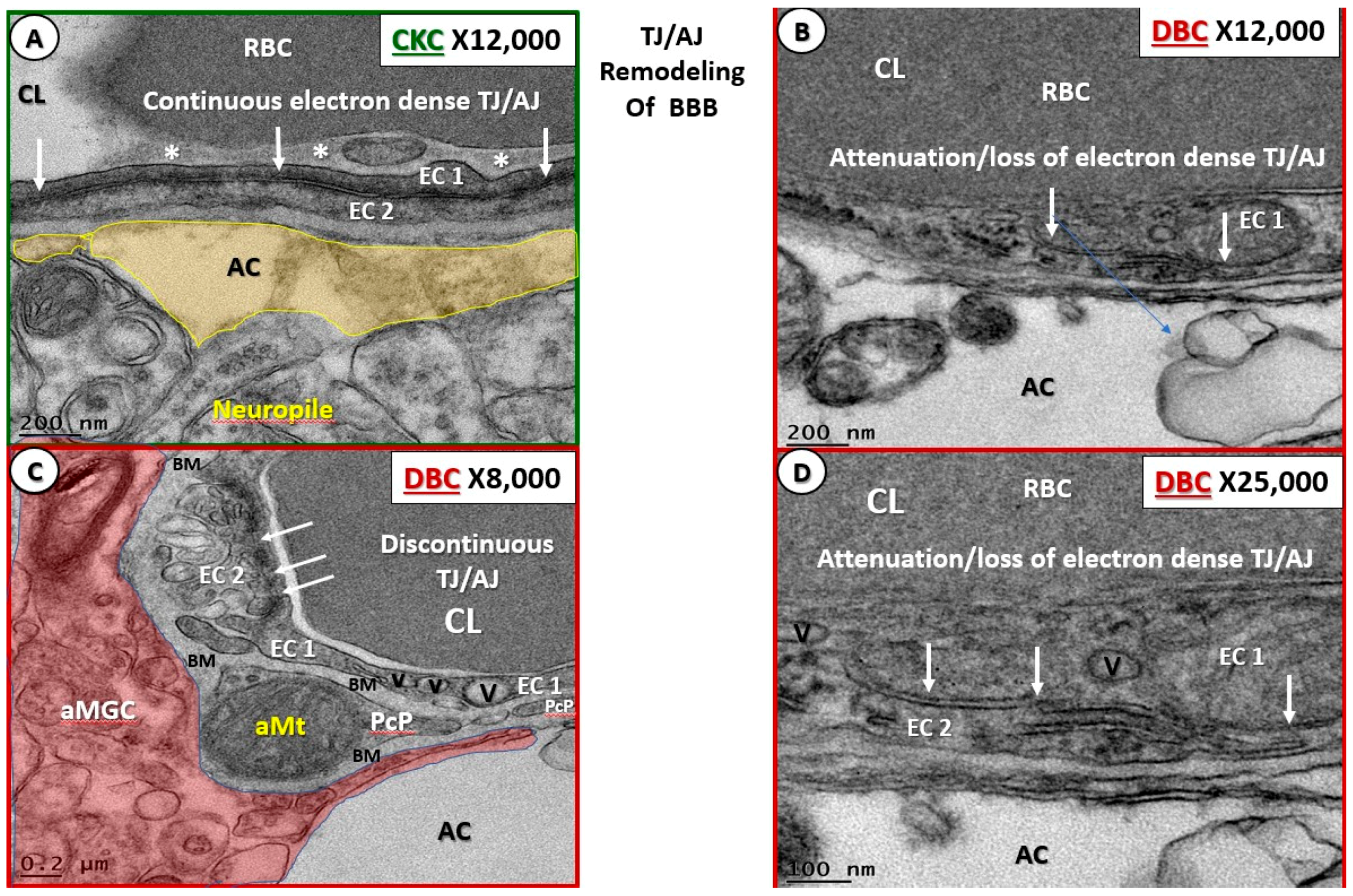
- Endothelial cell BBB TJ/AJ attenuation and/or loss would allow increased permeability (due to dysfunction, attenuation, and/or loss of its permeability barrier), which would allow the accumulation of multiple vasculotoxic and neurotoxic moieties within the NVU parenchyma (Figure 3B–D, Figure 4C,D, Figure 5, Figure 7C,D, Figure 8, and Figure 9) [6]. This compromise of the EC BBB TJ/AJ may aid in the understanding as to why there is concurrent remodeling in the surrounding cellular and extracellular regional constituents of the NVU. Importantly, these ultrastructural maladaptive remodeling changes could also interfere with neurovascular signaling/coupling functions of astrocytes to both pericytes and endothelial cells, which could lead to dysfunction and/or loss of function. As a result, there could also be a reduction in regional cerebral blood flow with regional hypoperfusion and ischemia [4,5,6,7,8].
- ii.
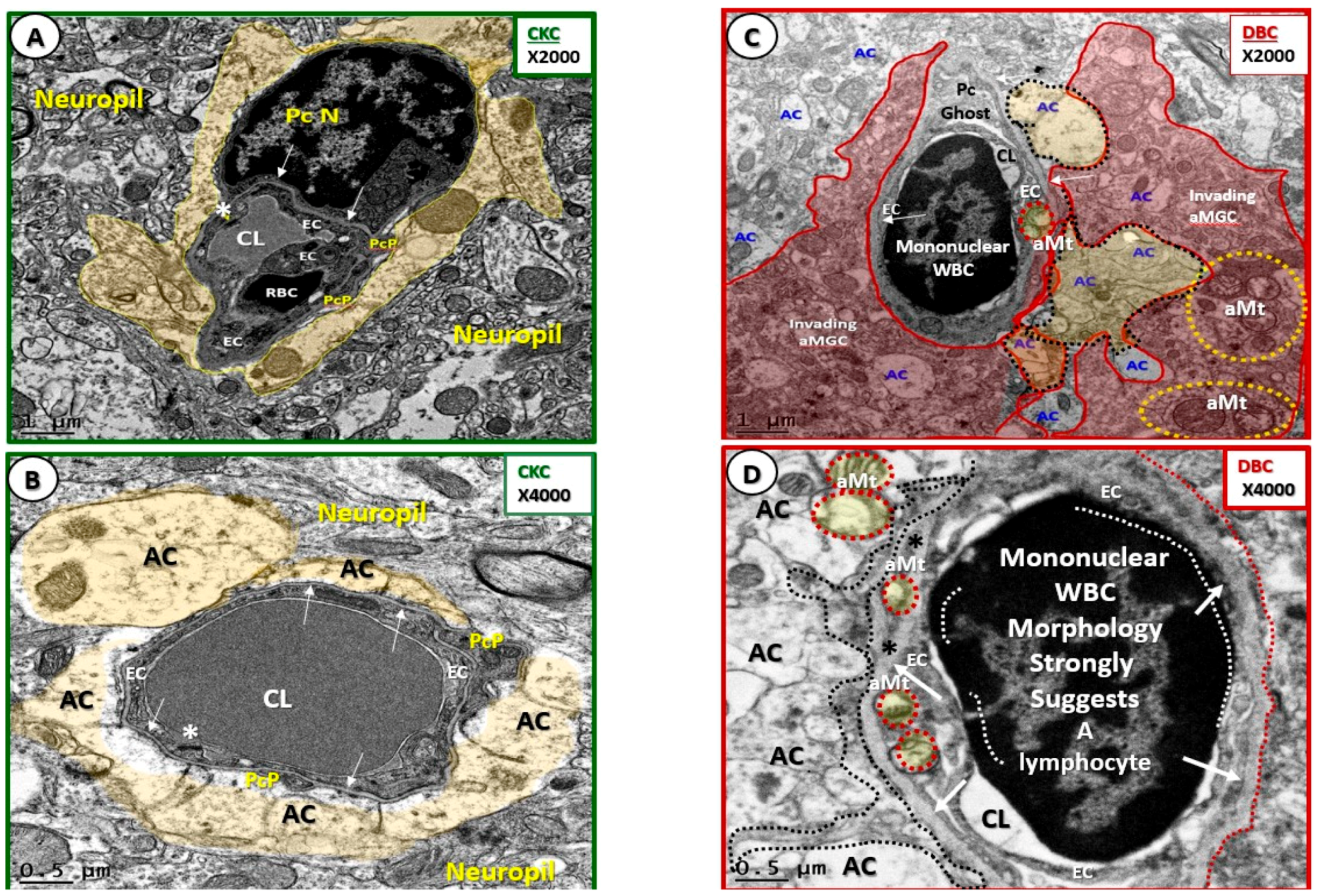
- Luminal RBCs were observed to become adherent (via adherence plaques) in DBC models, which were not observed in the CKC models (Figure 10). Red blood cells become excessively glycated and form advanced glycation end products (AGE) with hemoglobin in human and DBC models of T2DM. Previously, our group showed elevations of hemoglobin A1c (HbA1c) [21], which is known to increase RBC stiffness that is associated with the loss of RBC deformability [40]. The accumulation of AGE in the RBC outer plasma membrane regions will serve as a ligand to the EC AGE receptor (RAGE). Additionally, the inner plasma membrane may translocate phosphatidylserine (PS) to the outer leaflet in the hyperglycemic microenvironment of DBC models. The translocated or “flipped” outer leaflet PS will contribute to the adherence of RBCs to the EC PS receptor, as well as to the EC matrix of thrombospondin, αvβ1, and CD36, which may add to the increased electron density of the proteinaceous electron-dense adhesion plaques of the RBC and EC [41,42]. Importantly, there were also adherent mononuclear white blood cells within the capillary lumen of NVUs observed in the cortical gray matter in DBC models, as depicted in previous figures (Figure 4C,D, Figure 7C and Figure 8B), which will be discussed in greater detail as they relate to microglia remodeling. While the elusive endothelial glycocalyx was not specifically studied in this experiment, it is known that hyperglycemia results in the loss or shedding of the protective endothelial glycocalyx, and thus, may result in a more vulnerable and activated endothelium with increased inflammation and injury, decreased endothelial bioavailability of nitric oxide, and impaired vasodilation, in addition to becoming a more pro-coagulant surface in the DBC models (Figure 10C,D) [43].
- ii.
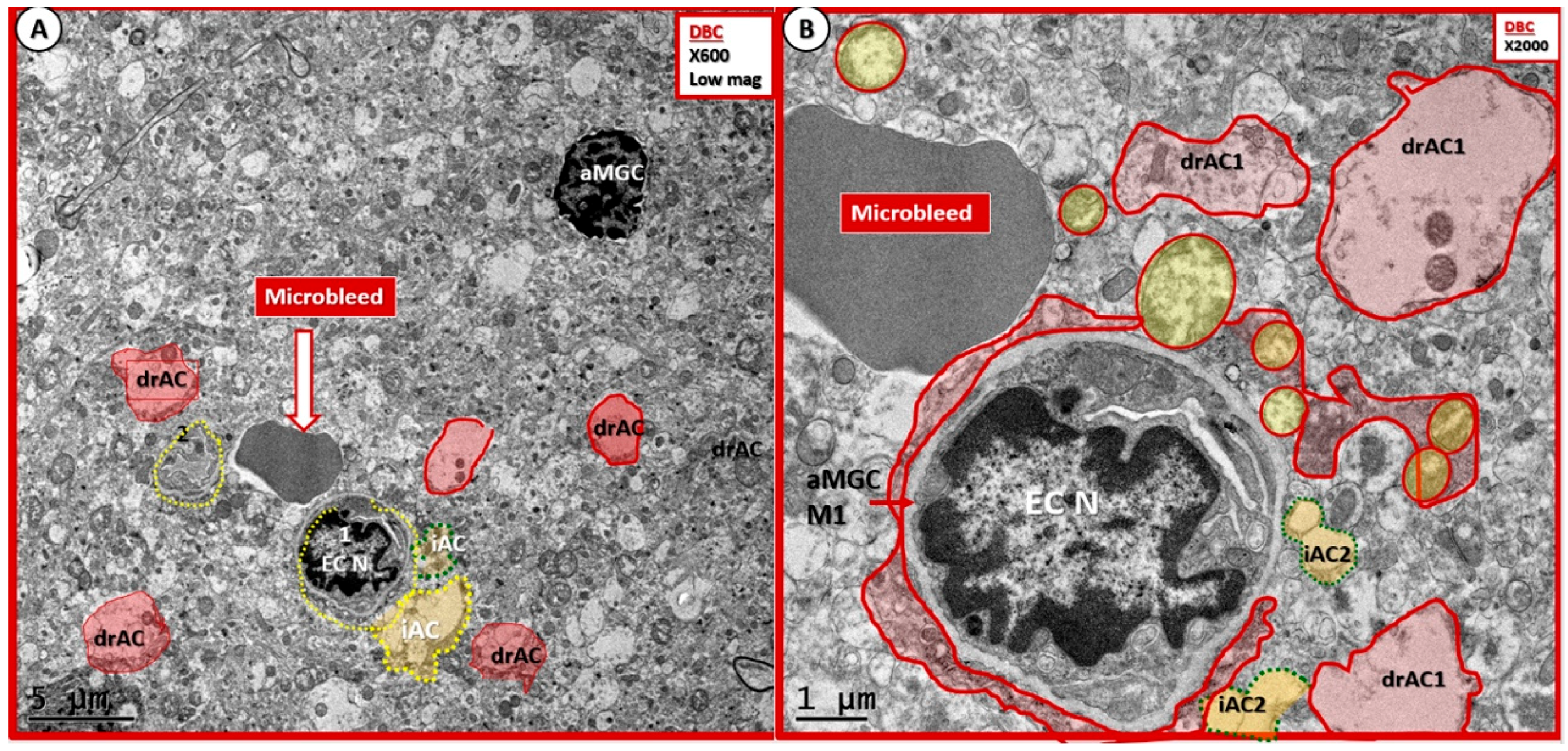
- Microbleeds/microhemorrhages within the gray matter of the cortical layers were observed in DBCs, which certainly could be related to incompetent EC TJ/AJ BBB proteins. These hemoglobin-containing extrusions/microbleeds would contain iron that could promote additional oxidative stress to the NVU and the immediate surrounding tissues (Figure 11 and Figure 12). Importantly, RBC remodeling can result in increasing dysfunction and/or damage to the NVU as a result of adherence, escaping, or loss of deformability within NVU capillary lumen in DBC models. Of note, cerebral microbleeds are being increasingly found on magnetic resonance imaging (MRI) [44]. Currently, the significance of microbleeds in diabetic preclinical models is yet to be evaluated extensively at the transmission electron microscopic ultrastructural level; however, there may be some similarities to retinal microbleeds and hemorrhages [45]. Notwithstanding, these observed microbleeds may be related to remodeled and dysfunctional TJ/AJ. The combination of adherent RBCs and microbleeds in the DBC may have detrimental consequences in local regional blood flow with resultant regional ischemia and loss of neurovascular coupling in the DBC models and may have a predisposition to accelerated neurodegeneration.
- iii.
- Maladaptive Pc remodeling (Figure 7) in diabetes may contribute to increased BBB TJ/AJ permeability, neuronal dysfunction, injury, and eventual neurodegeneration [13,15,18,25]. Recently, in streptozotozin-induced type 1 diabetes, it was demonstrated that pericytes are also attenuated and/or lost, and that the mitochondrial-specific carbonic anhydrase inhibitor (toprimate) was able to rescue pericyte loss and normalise BBB permeability [46]. Our findings of aberrant mitochondria in DBC pericytes may be playing a detrimental role in our observed pericyte attenuation and/or loss (Figure 7E).
- iv.
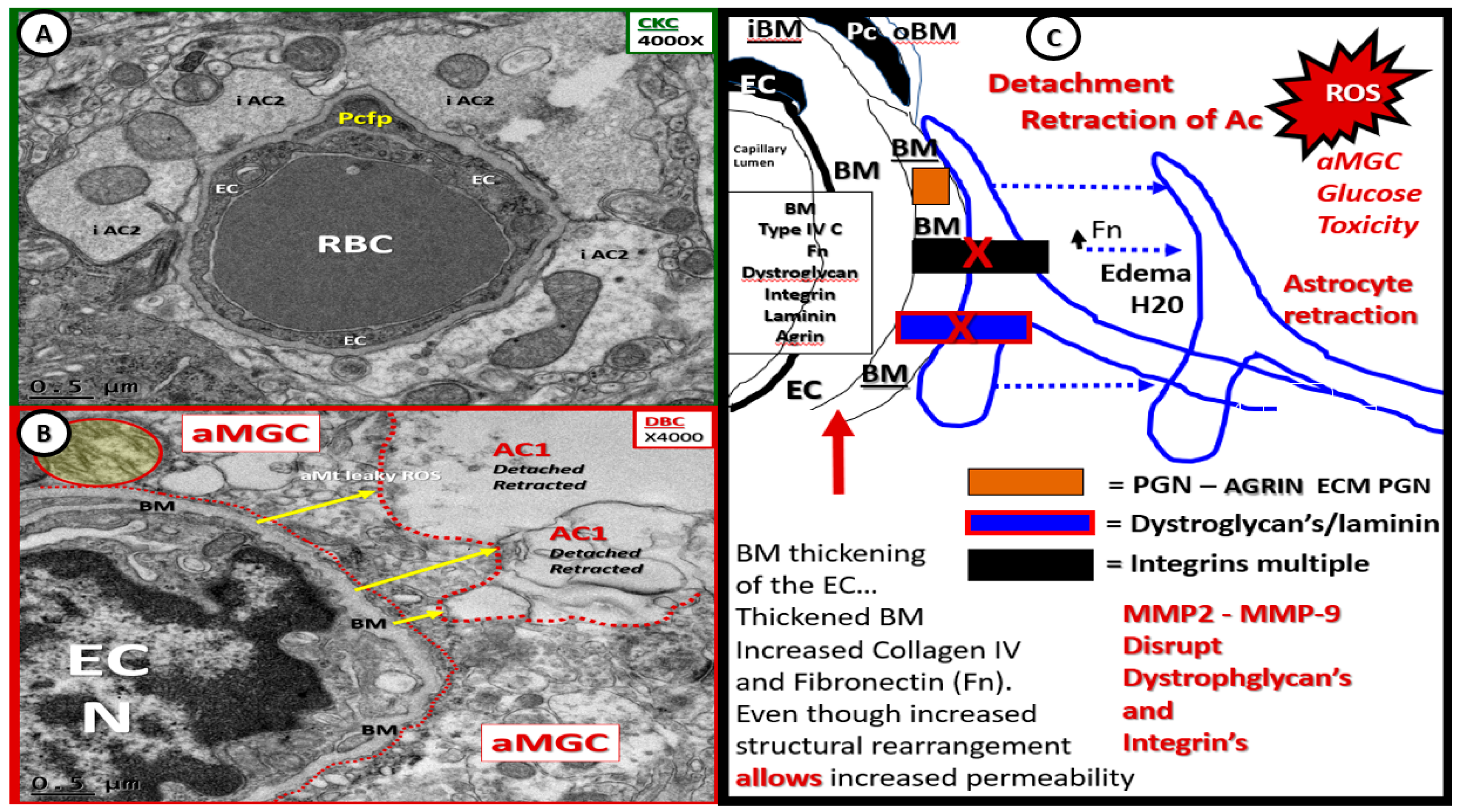
- Capillary NVU BM thickening was observed in the cortical gray matter of DBC (~≥200 nm) as compared to the CKC (~≤80 nm) models (Figure 4 and Figure 5). Capillary BM thickening is a fundamental ultrastructural central finding in most diabetic affected end-organs and nearly a pathognomonic ultrastructural finding in human and rodent models of diabetes. Capillary NVU BM thickening was reported in human diabetics in the cortical regions of the brain [47]. Basement membrane thickening was also reported in the retinas of type 1 diabetic rats (streptozotocin-induced) at six months of age [48], whereas we did not observe BM thickening in our type 1 diabetic mice mid-brain models studied at four months in a previous study [18]. Mechanisms of BM thickening may be related to glucotoxicity, increased protein kinase C, increased vascular endothelial cell growth factor, increased AGE, type IV collagen AGE crosslinking, and oxidative stress [23]. The observed increase in BM thickness may increase NVU permeability and could also play a role in the detachment of ACs from the capillary EC and Pc NVU BM due to an interference in cell–matrix interactions and dysfunctional alterations of BM integrins/dystroglycans. The current finding of BM thickening in DBC models filled in some gaps in our knowledge regarding the lack of BM thickening in diet-induced obesity Western mice models (cortical gray matter) [13] and the type 1 diabetic mice models previously studied (cortical mid-brain) [23]. To the best of our knowledge, we are the first to identify BM thickening in capillary EC/Pc NVU microvessels in the cortical gray matter of the female obese, insulin-resistant, type 2 diabetic db/db mouse model.
- v.
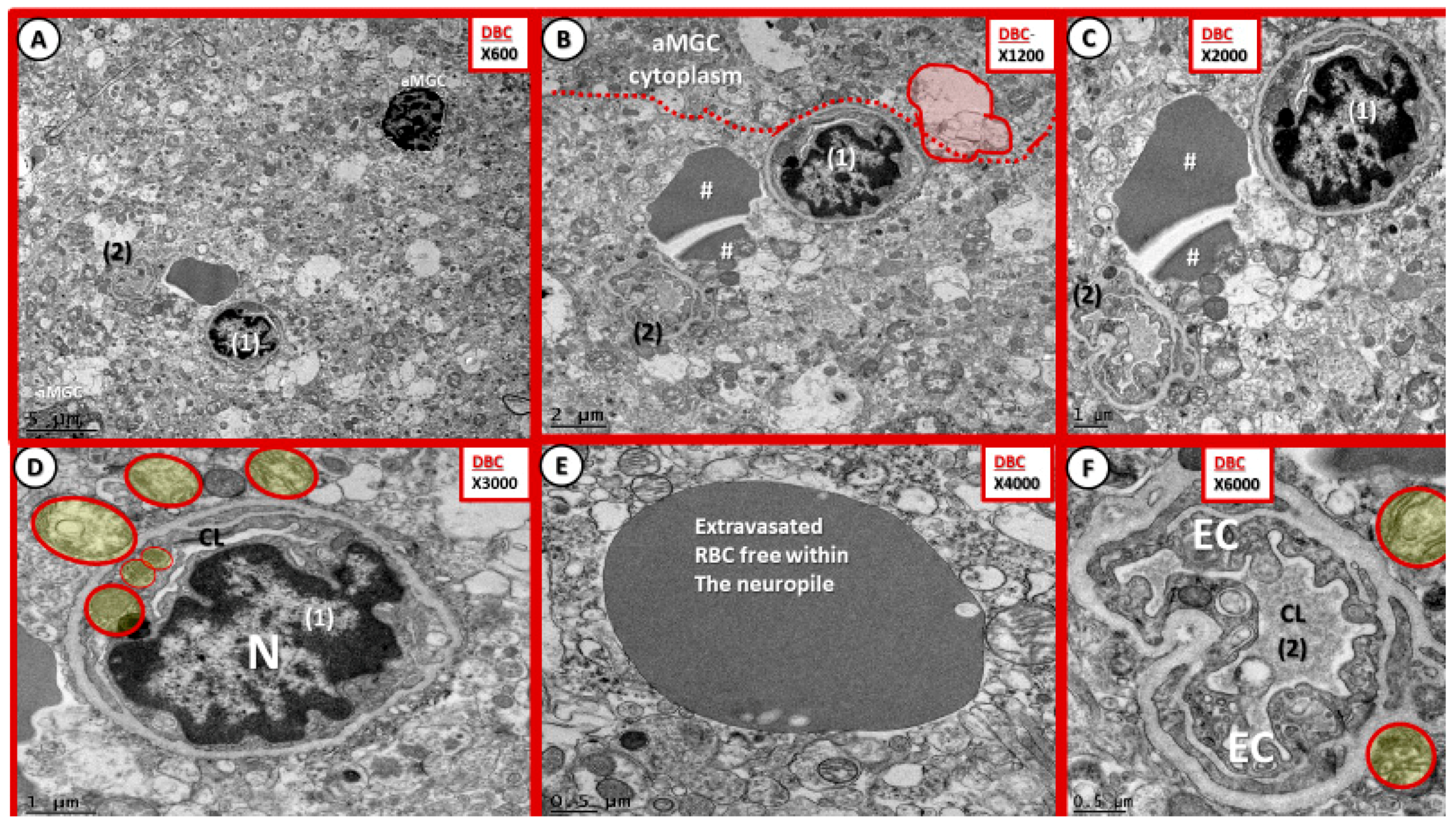
- Abnormal remodeling changes of the supportive and connecting protoplasmic astrocytes were observed in the DBC models. Astrocytes foot processes were depicted in CKC images as being colored “golden” due to their important function and location as connecting cells between the capillary NVU and their regional neurons when they were observed to have intact connections with the EC and Pc basement membranes. We observed how these connecting intact astrocytes became detached and retracted from the capillary EC and Pc BMs in the DBC, and how they lost their connective essential role of neurovascular coupling. This loss of neurovascular coupling and loss of vasodilation when being actively signaled by regional neurons could result in a loss of function that could result in localized decreased cerebral blood flow, resulting in regional hypoxia with the potential for increased neurodegeneration. In the detached astrocyte, it is possible that the AC soma may remodel its F-actin cytoskeleton such that the AC protoplasmic processes retract toward the soma of the AC in a phenotypic response to injury (brain wounding mechanism), which may result from a combination of excessive reactive oxygen/nitrogen species, toxic cytokines, and ischemia in the DBC models. Some suggested that the activated-reactive detached AC are induced by activated microglia cells [49,50,51], and furthermore, that these activated microglia may be responsible for the actual physical detachment and subsequent retraction of the capillary NVU astrocytes in the DBC (Figure 4C,D and Figure 5).
5. Type 2 Diabetes Mellitus Increases the Risk of the Neurodegenerative Diseases, Alzheimer’s and Parkinson’s Disease
6. Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wild, S.; Roglic, G.; Green, A.; Sicree, R.; King, H. Global prevalence of diabetes: Estimates for the year 2000 and projections for 2030. Diabetes Care 2004, 27, 1047–1053. [Google Scholar] [CrossRef] [PubMed]
- Ott, A.; Breteler, M.M.; van Harskamp, F.; Claus, J.J.; van der Cammen, T.J.; Grobbee, D.E.; Hofman, A. Prevalence of Alzheimer’s disease and vascular dementia: Association with education. The Rotterdam study. BMJ 1995, 310, 970–973. [Google Scholar] [CrossRef] [PubMed]
- Reeve, A.; Simcox, E.; Turnbull, D. Ageing and Parkinson’s disease: Why is advancing age the biggest risk factor? Ageing Res. Rev. 2014, 14, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Nelson, A.R.; Sweeney, M.D.; Sagare, A.P.; Zlokovic, B.J. Neurovascular dysfunction and neurodegeneration in dementia and Alzheimer’s disease. Biochim. Biophys. Acta 2016, 1862, 887–900. [Google Scholar] [CrossRef] [PubMed]
- Snyder, H.M.; Corriveau, R.A.; Craft, S.; Faber, J.E.; Greenberg, S.M.; Knopman, D.; Lamb, B.T.; Montine, T.J.; Nedergaard, M.; Schaffer, C.B.; et al. Vascular contributions to cognitive impairment and dementia including Alzheimer’s disease. Alzheimer’s Dement. 2015, 11, 710–717. [Google Scholar] [CrossRef] [PubMed]
- Zlokovic, B.V. Neurovascular pathways to neurodegeneration in Alzheimer’s disease and other disorders. Nat. Rev. Neurosci. 2011, 12, 723–738. [Google Scholar] [CrossRef] [PubMed]
- Iadecola, C. The pathobiology of vascular dementia. Neuron 2013, 80, 844–866. [Google Scholar] [CrossRef] [PubMed]
- Kisler, K.; Nelson, A.R.; Montagne, A.; Zlokovic, B.V. Cerebral blood flow regulation and neurovascular dysfunction in Alzheimer disease. Nat. Rev. Neurosci. 2017, 18, 419–434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rocca, W.A.; Petersen, R.C.; Knopman, D.S.; Herbert, L.E.; Evans, D.A.; Hall, K.S.; Gao, S.; Unverzawt, F.W.; Langa, K.M.; Larson, E.B.; et al. Trends in the incidence and prevalence of Alzheimer’s disease, dementia, and cognitive impairment in the United States. Alzheimer’s Dement. 2011, 7, 80–93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leibson, C.L.; Rocca, W.A.; Hanson, V.A.; Cha, R.; Kokmen, E.; O’Brien, P.C.; Palumbo, P.J. Risk of dementia among persons with diabetes mellitus: A population-based cohort study. Am. J. Epidemiol. 1996, 145, 301–308. [Google Scholar] [CrossRef]
- Ott, A.; Stolk, R.P.; van Harskamp, F.; Pols, H.A.; Hofman, A.; Breteler, M.M. Diabetes mellitus and the risk of dementia: The Rotterdam Study. Neurology 1999, 53, 1937–1942. [Google Scholar] [CrossRef] [PubMed]
- Peila, R.; Rodriguez, B.L.; Launer, L.J. Type 2 diabetes, APOE gene, and the risk for dementia and related pathologies: The Honolulu-Asia Aging Study. Diabetes 2002, 51, 1256–1262. [Google Scholar] [CrossRef] [PubMed]
- Hayden, M.R.; Banks, W.A.; Shah, G.N.; Gu, Z.; Sowers, J.R. Cardiorenal metabolic syndrome and diabetic cognopathy. Cardiorenal Med. 2013, 3, 265–282. [Google Scholar] [CrossRef] [PubMed]
- McConnell, H.L.; Kersch, C.N.; Woltjer, R.L.; Neuwelt, E.A. The Translational Significance of the Neurovascular Unit. J. Biol. Chem. 2017, 292, 762–770. [Google Scholar] [CrossRef] [PubMed]
- Hall, C.N.; Reynell, C.; Gesslein, B.; Hamilton, N.B.; Mishra, A.; Sutherland, B.A.; O’Farrell, F.M.; Buchan, A.M.; Lauritzen, M.; Attwell, D. Capillary pericytes regulate cerebral blood flow in health and disease. Nature 2014, 508, 55–60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mishra, A.; Reynolds, J.P.; Chen, Y.; Gourine, A.V.; Rusakov, D.A.; Attwell, D. Astrocytes mediate neurovascular signaling to capillary pericytes but not to arterioles. Nat. Neurosci. 2016, 19, 1619–1627. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Petzold, G.C.; Murthy, V.N. Role of astrocytes in neurovascular coupling. Neuron 2011, 71, 782–797. [Google Scholar] [CrossRef] [PubMed]
- Salameh, T.S.; Shah, G.N.; Price, T.O.; Hayden, M.R.; Banks, W.A. Blood-brain barrier disruption and neurovascular unit dysfunction in diabetic mice: Protection with the mitochondrial carbonic anhydrase inhibitor topiramate. J. Pharmacol. Exp. Ther. 2016, 359, 452–459. [Google Scholar] [CrossRef] [PubMed]
- Hu, G.; Jousilahti, P.; Bidel, S.; Antikainen, R.; Tuomilehto, J. Type 2 diabetes and the risk of Parkinsons’s disease. Diabetes Care 2007, 30, 842–847. [Google Scholar] [CrossRef] [PubMed]
- Butt, A.; Mihaila, D.; Verkhratsky, A. Neuroglia: A New Open-Access Journal Publishing All Aspects of Glial Research. Neuroglia 2018, 1, 1. [Google Scholar] [CrossRef]
- Habibi, J.; Aroor, A.R.; Sowers, J.R.; Jia, G.; Hayden, M.R.; Garro, M.; Barron, B.; Mayoux, E.; Rector, R.S.; Whaley-Connell, A.; et al. Sodium glucose transporter-2 (SGLT-2) inhibition with empagliflozin improves cardiac diastolic function in a female rodent model of diabetes. Cardiovasc. Diabetol. 2017, 16, 9. [Google Scholar] [CrossRef] [PubMed]
- Laws, K.R.; Irvine, K.; Gale, T.M. Sex differences in cognitive impairment in Alzheimer’s disease. World J. Psychiatry 2016, 6, 54–65. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hayden, M.R.; Sowers, J.R.; Tyagi, S.C. The central role of vascular extracellular matrix and basement membrane remodeling in metabolic syndrome and type 2 diabetes: The matrix preloaded. Cardiovasc. Diabetol. 2005, 4, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thomsen, M.S.; Routhe, L.J.; Moos, T. The vascular basement membrane in the healthy and pathological brain. J. Cereb. Blood Flow Metab. 2017, 37, 3300–3317. [Google Scholar] [CrossRef] [PubMed]
- Bell, R.D.; Winkler, E.A.; Sagare, A.P.; Singh, I.; LaRue, B.; Deane, R.; Zlokovic, B.V. Pericytes control key neurovascular functions and neuronal phenotype in the adult brain and during brain aging. Neuron 2010, 68, 409–427. [Google Scholar] [CrossRef] [PubMed]
- Verkhratsky, A.; Nedergaard, M. Astroglial cradle in the life of the synapse. Phil. Trans. R. Soc. Lond. B Biol. Sci. 2014, 369, 20130595. [Google Scholar] [CrossRef] [PubMed]
- Verkhratsky, A.; Nedergaard, M. Physiology of Astroglia. Physiol. Rev. 2018, 98, 239–389. [Google Scholar] [CrossRef] [PubMed]
- Verkhratsky, A.; Bush, N.A.; Nedergaard, M.; Butt, A. The Special Case of Human Astrocytes. Neuroglia 2018, 1, 4. [Google Scholar] [CrossRef]
- Scemes, E.; Spray, D.C. Chapter: The astrocytic syncytium. In Non-Neural Cells in the Nervous System: Function and Dysfunction; Hertz, L., Ed.; Elsevier: New York, NY, USA, 2004; Volume 31, pp. 165–179. [Google Scholar]
- Abbott, N.J.; Rönnbäck, L.; Hansson, E. Astrocyte-endothelial interactions at the blood-brain barrier. Nat. Rev. Neurosci. 2006, 7, 41–53. [Google Scholar] [CrossRef] [PubMed]
- Del Zoppo, G.J.; Milner, R. Integrin–Matrix Interactions in the Cerebral Microvasculature. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 1966–1975. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reske-Nielsen, E.; Lundbaek, K. Diabetic Encephalopathy. In Pathophysiologie und Klinik/Pathophysiology and Clinical Considerations; Pfeiffer, E.F., Ed.; Springer: Berlin/Heidelberg, Germany, 1971. [Google Scholar]
- De la Monte, S.M.; Wands, J.R. Alzheimer’s Disease Is Type 3 Diabetes—Evidence Reviewed. J. Diabetes Sci. Technol. 2008, 2, 1101–1113. [Google Scholar] [CrossRef] [PubMed]
- Li, X.L.; Aou, S.; Oomura, Y.; Hori, N.; Fukunaga, K.; Hori, T. Impairment of long-term potentiation and spatial memory in leptin receptor-deficient rodents. Neuroscience 2002, 113, 607–615. [Google Scholar] [CrossRef]
- Zheng, H.; Zheng, Y.; Zhao, L.; Chen, M.; Bai, G.; Hu, Y.; Hu, W.; Yan, Z.; Gao, H. Cognitive decline in type 2 diabetic db/db mice may be associated with brain region-specific metabolic disorders. Biochim. Biophys. Acta 2017, 1863, 266–273. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Rodriguez, J.J.; Ortiz, O.; Jimenez-Palomares, M.; Kay, K.R.; Berrocoso, E.; Murillo-Carretero, M.I.; Perdomo, G.; Spires-Jones, T.; Cozar-Castellano, I.; Lechuga-Sancho, A.M.; et al. Differential central pathology and cognitive impairment in pre-diabetic and diabetic mice. Psychoneuroendocrinology 2013, 38, 2462–2475. [Google Scholar] [CrossRef] [PubMed]
- Andersen, J.V.; Nissen, J.D.; Christensen, S.K.; Markussen, K.H.; Waagepetersen, H.S. Impaired Hippocampal Glutamate and Glutamine Metabolism in the db/db Mouse Model of Type 2 Diabetes Mellitus. Neural Plast. 2017, 2017, 2107084. [Google Scholar] [CrossRef] [PubMed]
- Ernst, A.; Sharma, A.N.; Elased, K.M.; Guest, P.C.; Rahmoune, H.; Bahn, S. Diabetic db/db mice exhibit central nervous system and peripheral molecular alterations as seen in neurological disorders. Transl. Psychiatry 2013, 3, e263. [Google Scholar] [CrossRef] [PubMed]
- Kalani, A.; Chaturvedi, P.; Maldonado, C.; Bauer, P.; Joshua, I.G.; Tyagi, S.C.; Tyagi, N. Dementia-like pathology in type-2 diabetes: A novel microRNA mechanism. Mol. Cell. Neurosci. 2017, 80, 58–65. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tomaiuolo, G. Biomechanical properties of red blood cells in health and disease towards microfluidics. Biomicrofluidics 2014, 8, 051501. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carelli-Alinovi, C.; Misiti, F. Erythrocytes as Potential Link between Diabetes and Alzheimer’s Disease. Front. Aging Neurosci. 2017, 9, 276. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Koo, S.; Lin, C.S.; Neu, B. Macromolecular depletion modulates the binding of red blood cells to activated endothelial cells. Biointerphases 2010, 5, FA19–FA23. [Google Scholar] [CrossRef] [PubMed]
- Nieuwdorp, M.; van Haeften, T.W.; Gouverneur, M.C.; Mooij, H.L.; van Lieshout, M.H.; Levi, M.; Meijers, J.C.; Holleman, F.; Hoekstra, J.B.; Vink, H.; et al. Loss of endothelial glycocalyx during acute hyperglycemia coincides with endothelial dysfunction and coagulation activation in vivo. Diabetes 2006, 55, 480–486. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Ramirez, S.; Greenberg, S.M.; Viswanathan, A. Cerebral microbleeds: Overview and implications in cognitive impairment. Alzheimers Res. Ther. 2014, 6, 33. [Google Scholar] [CrossRef] [PubMed]
- Qiu, C.; Cotch, M.F.; Sigurdsson, S.; Jonsson, P.V.; Jonsdottir, M.K.; Sveinbjrnsdottir, S.; Eiriksdottir, G.; Klein, R.; Harris, T.B.; van Buchem, M.A.; et al. Cerebral microbleeds, retinopathy and dementia: The AGES-Reykjavik study. Neurology 2010, 75, 2221–2228. [Google Scholar] [CrossRef] [PubMed]
- Price, T.O.; Eranki, V.; Banks, W.A.; Ercal, N.; Shah, G.N. Topiramate treatment protects blood-brain barrier pericytes from hyperglycemia-induced oxidative damage in diabetic mice. Endocrinology 2012, 153, 362–372. [Google Scholar] [CrossRef] [PubMed]
- Johnson, P.C.; Brendel, K.; Meezan, E. Thickened cerebral cortical capillary basement membranes in diabetics. Arch. Pathol. Lab. Med. 1982, 106, 214–217. [Google Scholar] [PubMed]
- Cherian, S.; Roy, S.; Pinheiro, A.; Roy, S. Tight Glycemic Control Regulates Fibronectin Expression and Basement Membrane Thickening in Retinal and Glomerular Capillaries of Diabetic Rats. Investig. Ophthalmol. Vis. Sci. 2009, 50, 943–949. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liddelow, S.A.; Guttenplan, K.A.; Clarke, L.E.; Bennett, F.C.; Bohlen, C.J.; Schirmer, L.; Bennett, M.L.; Münch, A.E.; Chung, W.S.; Peterson, T.C.; et al. Neurotoxic reactive astrocytes are induced by activated microglia. Nature 2017, 541, 481–487. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iliff, J.J.; Wang, M.; Liao, Y.; Plogg, B.A.; Peng, W.; Gundersen, G.A.; Benveniste, H.; Vates, G.E.; Deane, R.; Goldman, S.A.; et al. A Paravascular Pathway Facilitates CSF Flow Through the Brain Parenchyma and the Clearance of Interstitial Solutes, Including Amyloid β. Sci. Transl. Med. 2012, 4, 147ra111. [Google Scholar] [CrossRef] [PubMed]
- Iliff, J.J.; Nedergaard, M. Is there a cerebral lymphatic system? Stroke 2013, 44, S93–S95. [Google Scholar] [CrossRef] [PubMed]
- Jessen, N.A.; Munk, A.S.; Lundgarrd, I.; Nedergaard, M. The glymphatic system—A beginner’s guide. Neurochem. Res. 2015, 40, 2583–2599. [Google Scholar] [CrossRef] [PubMed]
- Abbott, N.J.; Pizzo, M.E.; Preston, J.E.; Janigro, D.; Thorne, R.G. The role of brain barriers in fluid movement in the CNS: Is there a ‘glymphatic’ system? Acta Neuropathol. 2018, 135, 387–407. [Google Scholar] [CrossRef] [PubMed]
- Weller, R.O.; Sharp, M.M.; Christodoulides, M.; Carare, R.O.; Mollgard, K. The meninges as barriers and facilitators for the movement of fluid, cells and pathogens related to the rodent and human CNS. Acta Neuropathol. 2018, 135, 363–385. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bacyinski, A.; Maosheng, X.U.; Wang, W.; Hu, J. The paravascular pathway for brain current understanding, significance and controversy. Front. Neroanat. 2017, 11, 101. [Google Scholar] [CrossRef] [PubMed]
- Albargothy, N.J.; Johnston, D.A.; MacGregor-Sharp, M.; Weller, R.O.; Verma, A.; Hawkes, C.A.; Carare, R.O. Convective influx/glymphatic system: Tracers injected into the CSF enter and leave the brain along separate periarterial basement membrane pathways. Acta Neuropathol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, P.M. Early vascular aging (EVA): Consequences and Prevention. Vasc. Health Risk Manag. 2008, 4, 547–552. [Google Scholar] [CrossRef] [PubMed]
- Wrighten, S.A.; Piroli, G.G.; Grillo, C.A.; Reagan, L.P. A look inside the diabetic brain: Contributors to diabetes-induced brain aging. Biochim. Biophys. Acta 2009, 1792, 444–453. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ryan, C.M.; Geckle, M. Why is learning and memory dysfunction in Type 2 diabetes limited to older adults? Diabetes Metab. Res. Rev. 2000, 16, 308–315. [Google Scholar] [CrossRef]
- De Pablo-Fernandez, E.; Goldacre, R.; Pakpoor, J.; Noyce, A.J.; Warner, T.T. Association between diabetes and subsequent Parkinson disease: A record-linkage cohort study. Neurology 2018. [Google Scholar] [CrossRef] [PubMed]
- Knott, G.; Marchman, H.; Wall, D.; Lich, B. Serial section scanning electron microscopy of adult brain tissue using focused ion beam milling. J. Neurosci. 2008, 28, 2959–2964. [Google Scholar] [CrossRef] [PubMed]
- Pizzo, M.E.; Wolak, D.J.; Kumar, N.N.; Brunette, E.; Brunnquell, C.L.; Hannocks, M.J.; Abbott, N.J.; Meyerand, M.E.; Sorokin, L.; Stanimirovic, D.B.; et al. Intrathecal antibody distribution in the rat brain: Surface diffusion, perivascular transport and osmotic enhancement of delivery. J. Physiol. 2018, 596, 445–475. [Google Scholar] [CrossRef] [PubMed]






| Mural Cells | |
| Endothelial cell(s) (EC) | Line the entire vascular system (macrovascular and microvascular) in a mononuclear layer. |
| The ECs are the first cell one encounters from the vascular lumen as one proceeds from the luminal surface to the outermost abluminal regions of the neurovascular unit (NVU). | |
| Endothelial cells have an elusive glycocalyx on their luminal cytoplasm; however, this structure is usually eliminated by dehydration in the preparation for microscopy and staining. | |
| The ECs have an intermediate electron-dense cytoplasm and are thin except where one encounters a larger and greater electron-dense nucleus. | |
| Neurovascular ECs have very few to no pinocytotic vesicles as compared to peripheral capillary ECs with an increase in EC mitochondria. Next one encounters the ECs hyaline basement membrane (BM) with a less electron density. | |
| The ECs cytoplasm and nuclei are elongated and their terminating cytoplasm ends most commonly with overlapping junctions creating a paracellular–inter-endothelial space that is lined by very election-dense protein staining of tight and adherens junctions that form the brain’s specific blood-brain barrier (Figure 1). | |
| Pericyte(s) (Pc) | Are the next abluminal encountered cell in the NVU capillary. Pericytes are embedded within the shared BM synthesized by both the ECs and Pcs. The Pcs wrap around (peri-) the ECs of the NVU capillary and those transitioning to very small arterioles with an internal elastic lamina. |
| Similar to the ECs, Pcs have an electron-dense cytoplasm with elongated cytoplasmic processes and nuclei and contain prominent electron-dense lysosomes and mitochondria. Importantly, Pcs are known to be contractile cells that allow for NVU capillary contraction/relaxation to permit relaxation in regions of highly active neurons, which allow for increased regional cerebral blood flow (CBF) and neurovascular coupling (NVC) with intact astrocytes. | |
| In cross section one sometimes can only identify Pc foot processes (Pcfp) with their encasing inner and outer BMs, whereas in longitudinal sections one can better identify their elongated character (Figure 1) | |
| Glial Cells | |
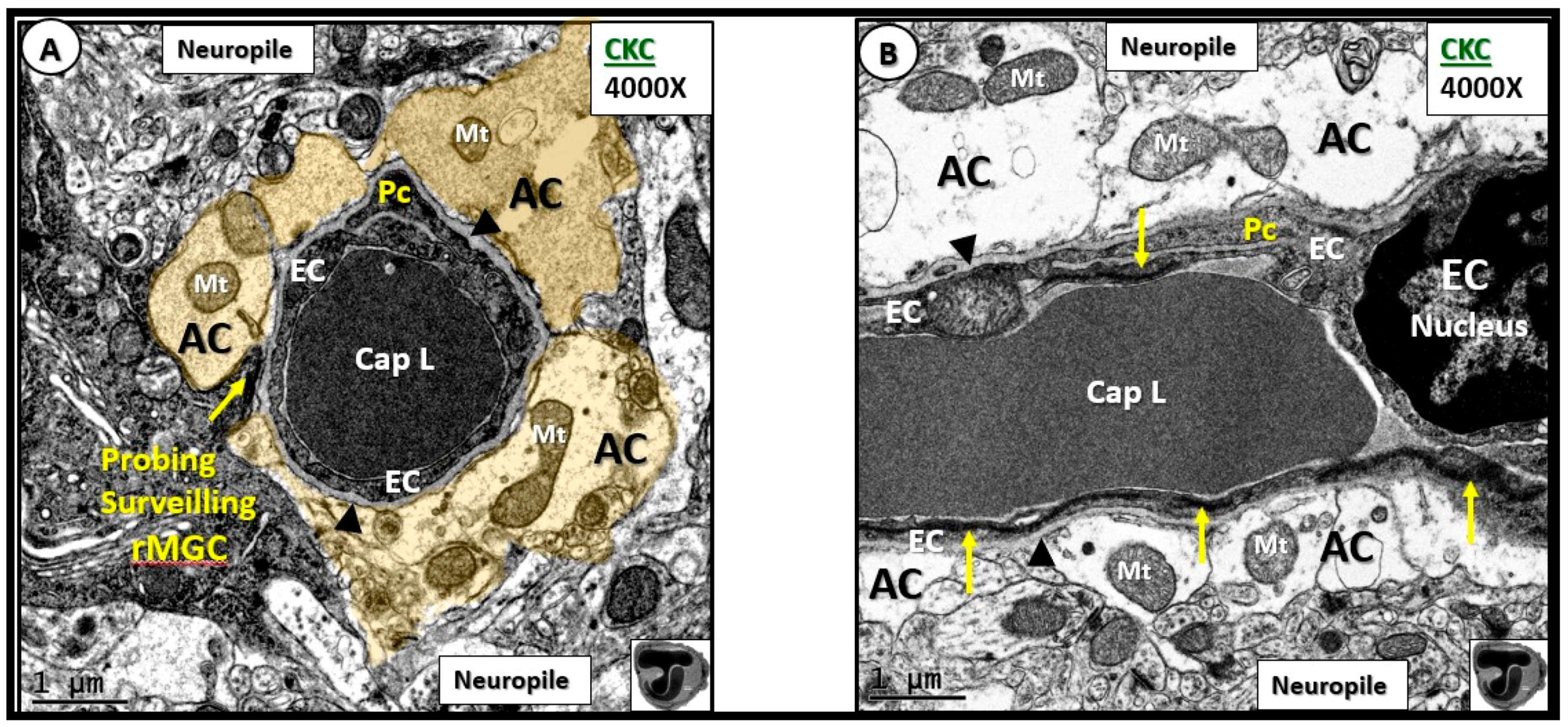
| Astrocyte cell(s) (AC) | Are the largest cell of the NVU, which assume a more cuboidal morphology in contrast to ECs and Pcs. The AC are also considered to be the brains connecting cell to regional neurons and form a clear zone, halo or corona around the Pcs and ECs. |
| Characteristically, they are the most electron-lucent cell of the NVU and brain cells and one often observes scattered electron-dense line’s, which represent their endoplasmic reticulum proteins. | |
| The ACs electron-dense thinned plasma membranes tightly adhere or abut the outer BMs of the ECs and Pcs. | |
| The AC completes the third key cell of the neurovascular unit; however, the microglia are also an important part and in both the grey matter and especially the white matter oligodendrocytes become a highly important part of the NVU as well (Figure 1). | |
| Microglia cells (MGC) | Are the smallest of the glia cells and their cytoplasm is the most electron-dense of the NVU and the brain. |
| In their non-activated phenotypic state, they have elongated cytoplasmic process in ramified form. They have an extensive endoplasmic reticulum, Golgi body system and contain multiple mitochondria. Their cytoplasmic processes are known to be capable of extending and contracting. They have a unique morphology of their nuclei with an outer stippled chromatin at its neurolemma and a more stippled diffuse chromatin electron dense appearance of the central nuclei (Figure 1). | |
| Oligodendrocyte cell(s) (OL) | As suggested by their name Oligo-, these cells are intermediate in size and their thinned cytoplasm also have an intermediate electron density that is helpful when comparing to AC and MGCs. They also may occur in groups or nests and are more often found in the deeper white matter regions of the brain |
| Neurons, Interneurons and the Neuropil | |
| Neurons (N)-interneurons | May be myelinated or unmyelinated. |
| Neurons have electron-lucent cytoplasmic axons with a greater electron dense and orderly layered network of neurofilaments and contain the usual cytoplasmic organelles. Neurons are also known to have and axon hillock and very long cytoplasmic axon extensions, which connect to other neurons via their dendritic synapses. | |
| Sometimes, neurons and especially interneurons can be noted to be closely interacting and in close proximity to the EC, Pc and astrocytes of the NVU. | |
| Neuronal cytoplasmic axons are also electron-lucent; however, their more electron-dense neurofilaments are layered in an orderly fashion in contrast to the electron densities of the AC, which are randomly scattered throughout the cytoplasm | |
| Neuropil–neuropile | Is an all-inclusive term and appears to form the background tissue along with a very thinned extracellular matrix–interstitium within the cortical grey matter, which includes the vast number of dendritic synapses, neurons (myelinated/unmyelinated axons) passing through the neuropil along with other glial cells and processes (Figure 1). |
| The EC, Pc, AC, MGC, oligodendrocytes and neurons–interneurons are the six major cell types that are present within a vast neuropile responsible for forming the NVU within the cortical grey matter | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hayden, M.R.; Grant, D.G.; Aroor, A.R.; DeMarco, V.G. Ultrastructural Remodeling of the Neurovascular Unit in the Female Diabetic db/db Model—Part I: Astrocyte. Neuroglia 2018, 1, 220-244. https://doi.org/10.3390/neuroglia1010015
Hayden MR, Grant DG, Aroor AR, DeMarco VG. Ultrastructural Remodeling of the Neurovascular Unit in the Female Diabetic db/db Model—Part I: Astrocyte. Neuroglia. 2018; 1(1):220-244. https://doi.org/10.3390/neuroglia1010015
Chicago/Turabian StyleHayden, Melvin R., DeAna G. Grant, Annayya R. Aroor, and Vincent G. DeMarco. 2018. "Ultrastructural Remodeling of the Neurovascular Unit in the Female Diabetic db/db Model—Part I: Astrocyte" Neuroglia 1, no. 1: 220-244. https://doi.org/10.3390/neuroglia1010015
APA StyleHayden, M. R., Grant, D. G., Aroor, A. R., & DeMarco, V. G. (2018). Ultrastructural Remodeling of the Neurovascular Unit in the Female Diabetic db/db Model—Part I: Astrocyte. Neuroglia, 1(1), 220-244. https://doi.org/10.3390/neuroglia1010015







