Clinical Use of Surface Electromyography to Track Acute Upper Extremity Muscle Recovery after Stroke: A Descriptive Case Study of a Single Patient
Abstract
1. Introduction
2. Materials and Methods
2.1. Participant
2.2. Intervention
3. Results
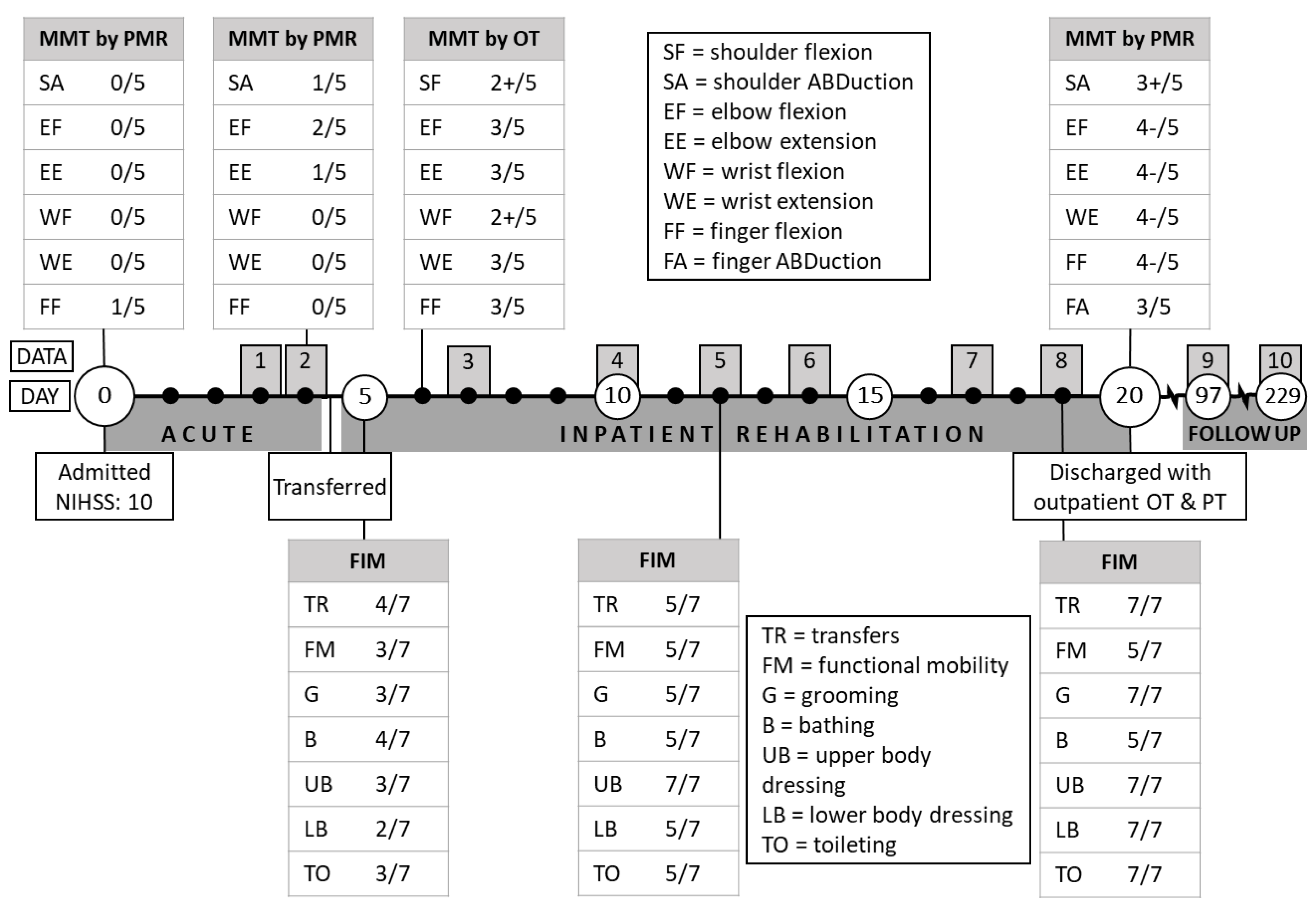
3.1. Acute Recovery
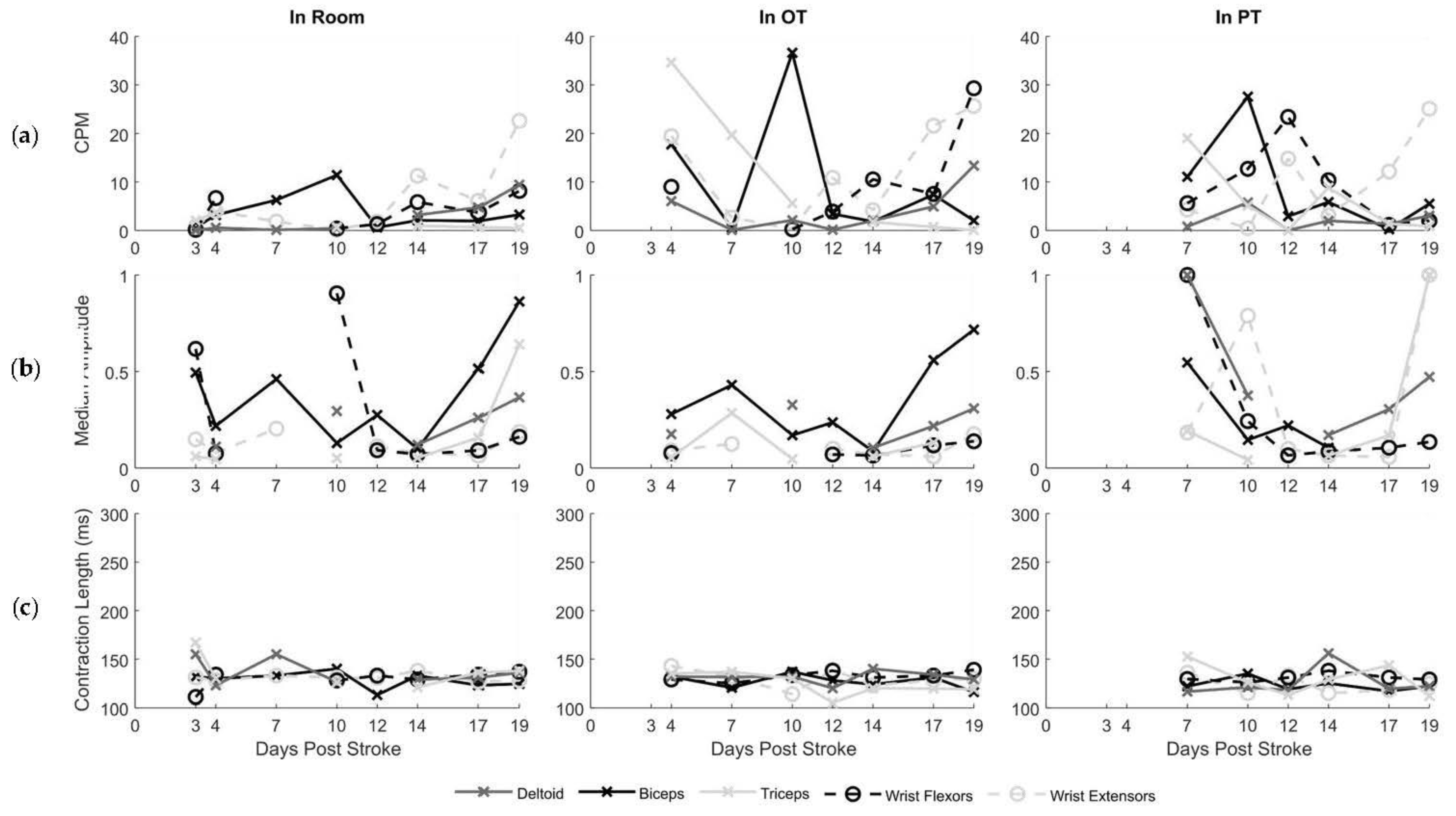
3.2. Inpatient Rehabilitation
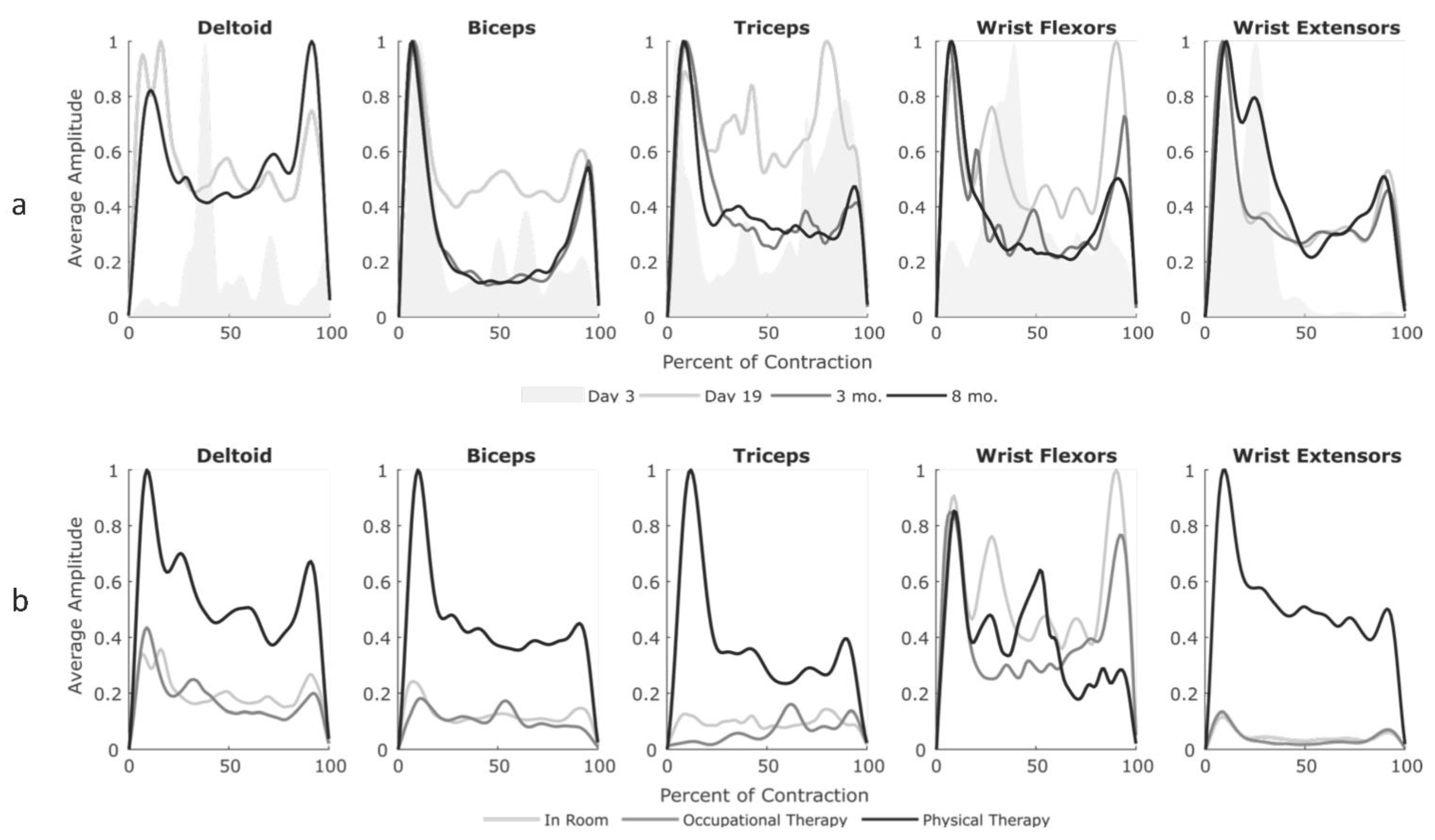
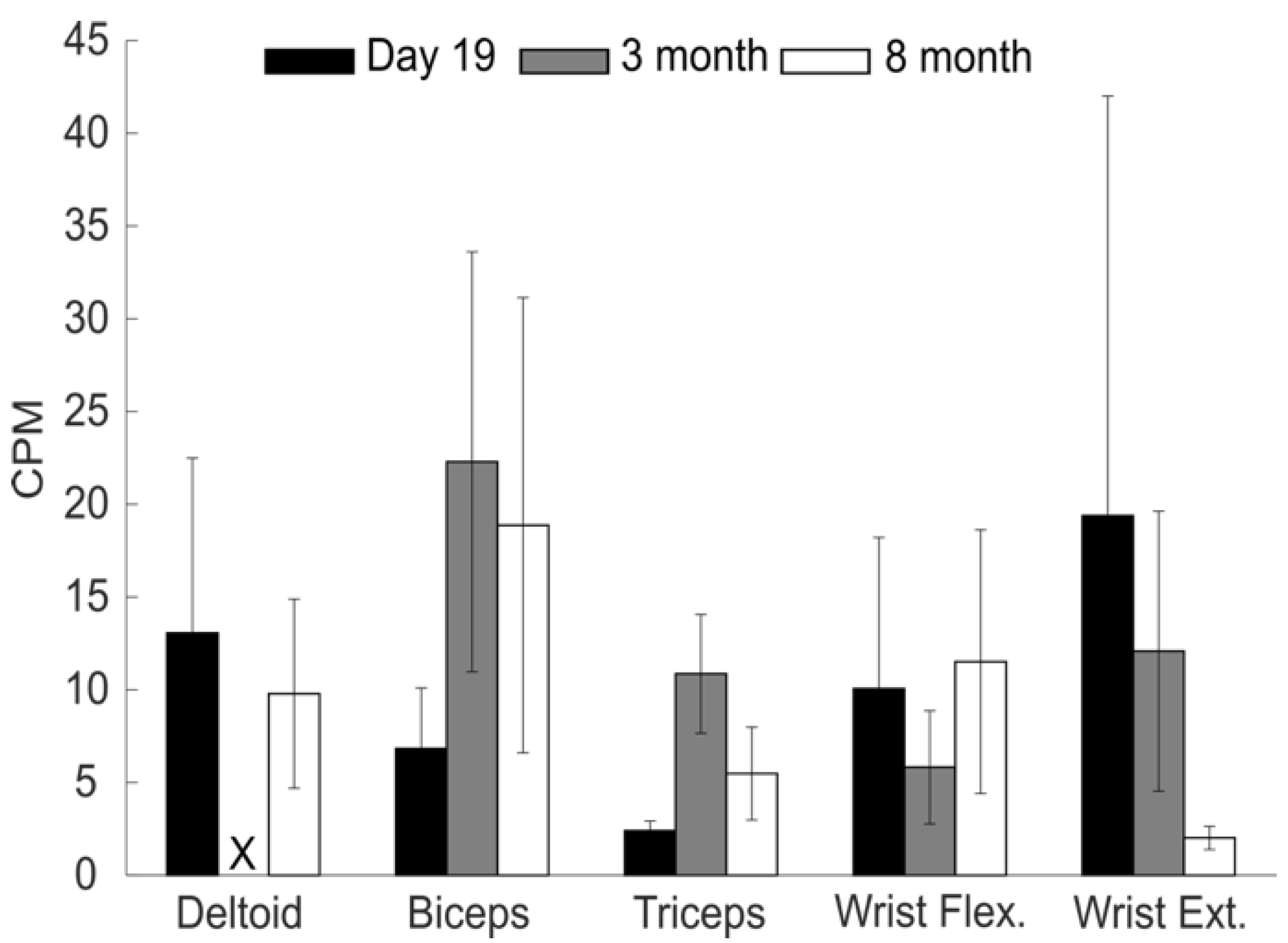
3.3. Recovery at Home
3.4. Perceptions of sEMG Use
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Olesen, J.; Gustavsson, A.; Svensson, M.Y.; Wittchen, H.; Jonsson, B.H.; on behalf of the CDBE2010 study group; The European Brain Council. The economic cost of brain disorders in Europe. Eur. J. Neurol. 2011, 19, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Jolkkonen, J.; Kwakkel, G. Translational Hurdles in Stroke Recovery Studies. Transl. Stroke Res. 2016, 7, 331–342. [Google Scholar] [CrossRef]
- Roger, V.L.; Go, A.S.; Lloyd-Jones, D.M.; Adams, R.J.; Berry, J.D.; Brown, T.M.; Carnethon, M.R.; Dai, S.; De Simone, G.; Ford, E.S. Heart disease and stroke statistics—2011 update: A report from the American Heart Association. Circulation 2011, 123, e18–e209. [Google Scholar] [CrossRef]
- Lenker, J.A.; Harris, F.; Taugher, M.; Smith, R.O. Consumer perspectives on assistive technology outcomes. Disabil. Rehabil. Assist. Technol. 2013, 8, 373–380. [Google Scholar] [CrossRef]
- Franceschini, M.; La Porta, F.; Agosti, M.; Massucci, M. Is health-related-quality of life of stroke patients influenced by neurological impairments at one year after stroke? Eur. J. Phys. Rehabil. Med. 2010, 46, 389–399. [Google Scholar]
- Kong, K.; Chua, K.; Lee, J. Symptomatic upper limb spasticity in patients with chronic stroke attending a rehabilitation clinic: Frequency, clinical correlates and predictors. J. Rehabil. Med. 2010, 42, 453–457. [Google Scholar] [CrossRef]
- Murphy, T.H.; Corbett, D. Plasticity during stroke recovery: From synapse to behaviour. Nat. Rev. Neurosci. 2009, 10, 861–872. [Google Scholar] [CrossRef] [PubMed]
- Murphy, M.A.; Bergquist, F.; Hagström, B.; Hernández, N.; Johansson, D.; Ohlsson, F.; Sandsjö, L.; Wipenmyr, J.; Malmgren, K. An upper body garment with integrated sensors for people with neurological disorders—Early development and evaluation. BMC Biomed. Eng. 2019, 1, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Merletti, R.; Farina, D. Surface Electromyography: Physiology, Engineering, and Applications; John Wiley & Sons: Hoboken, NJ, USA, 2016. [Google Scholar]
- Logan, L.R. Rehabilitation Techniques to Maximize Spasticity Management. Top. Stroke Rehabil. 2011, 18, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Dobkin, B.H.; Martinez, C. Wearable Sensors to Monitor, Enable Feedback, and Measure Outcomes of Activity and Practice. Curr. Neurol. Neurosci. Rep. 2018, 18, 87. [Google Scholar] [CrossRef]
- Adans-Dester, C.; Hankov, N.; O’Brien, A.; Vergara-Diaz, G.; Black-Schaffer, R.; Zafonte, R.; Dy, J.; Lee, S.I.; Bonato, P. Enabling precision rehabilitation interventions using wearable sensors and machine learning to track motor recovery. NPJ Digit. Med. 2020, 3, 1–10. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, H.; Zhao, W.; Zhang, M.; Qin, H.; Xie, Y. Flexible, Stretchable Sensors for Wearable Health Monitoring: Sensing Mechanisms, Materials, Fabrication Strategies and Features. Sensors 2018, 18, 645. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.-C.; Hsu, Y.-L. A review of accelerometry-based wearable motion detectors for physical activity monitoring. Sensors 2010, 10, 7772–7788. [Google Scholar] [CrossRef] [PubMed]
- Maceira-Elvira, P.; Popa, T.; Schmid, A.-C.; Hummel, F.C. Wearable technology in stroke rehabilitation: Towards improved diagnosis and treatment of upper-limb motor impairment. J. Neuroeng. Rehabil. 2019, 16, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Woodford, H.J.; Price, C.I. EMG biofeedback for the recovery of motor function after stroke. Cochrane Database Syst. Rev. 2007, 38, CD004585. [Google Scholar] [CrossRef]
- Dewald, J.P.A.; Pope, P.S.; Given, J.D.; Buchanan, T.S.; Rymer, W.Z. Abnormal muscle coactivation patterns during isometric torque generation at the elbow and shoulder in hemiparetic subjects. Brain 1995, 118, 495–510. [Google Scholar] [CrossRef] [PubMed]
- Brown, E.V.D.; McCoy, S.W.; Fechko, A.S.; Price, R.; Gilbertson, T.; Moritz, C.T. Preliminary Investigation of an Electromyography-Controlled Video Game as a Home Program for Persons in the Chronic Phase of Stroke Recovery. Arch. Phys. Med. Rehabil. 2014, 95, 1461–1469. [Google Scholar] [CrossRef] [PubMed]
- Kasman, G.S.; Wolf, S.L. Surface EMG Made Easy: A Beginner’s Guide for Rehabilitation Clinicians; Noraxon USA, Inc.: Scottsdale, AZ, USA, 2002. [Google Scholar]
- Winstein, C.; Varghese, R. Been there, done that, so what’s next for arm and hand rehabilitation in stroke? NeuroRehabilitation 2018, 43, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Hesam-Shariati, N.; Trinh, T.; Thompson-Butel, A.G.; Shiner, C.T.; McNulty, P.A. A Longitudinal Electromyography Study of Complex Movements in Poststroke Therapy. 2: Changes in Coordinated Muscle Activation. Front. Neurol. 2017, 8, 277. [Google Scholar] [CrossRef]
- Dobkin, B.H. A Rehabilitation-Internet-of-Things in the Home to Augment Motor Skills and Exercise Training. Neurorehabil. Neural Repair 2017, 31, 217–227. [Google Scholar] [CrossRef]
- Lane, K.; Chandler, E.; Payne, D.; Pomeroy, V.M. Stroke survivors’ recommendations for the visual representation of movement analysis measures: A technical report. Physiotherapy 2020, 107, 36–42. [Google Scholar] [CrossRef]
- Schwartz, S.; Cohen, M.E.; Herbison, G.J.; Shah, A. Relationship between two measures of upper extremity strength: Manual muscle test compared to hand-held myometry. Arch. Phys. Med. Rehabil. 1992, 73, 1063–1068. [Google Scholar]
- Sethi, A.; Ting, J.; Allen, M.; Clark, W.; Weber, D. Advances in motion and electromyography based wearable technology for upper extremity function rehabilitation: A review. J. Hand Ther. 2020, 33, 180–187. [Google Scholar] [CrossRef]
- Merletti, R. Surface electromyography: The SENIAM project. Eur. J. Phys. Rehabil. Med. 2000, 36, 167. [Google Scholar]
- Zeiler, S.R. Should We Care About Early Post-Stroke Rehabilitation? Not Yet, but Soon. Curr. Neurol. Neurosci. Rep. 2019, 19, 13. [Google Scholar] [CrossRef] [PubMed]
- Feldner, H.A.; Howell, D.; Kelly, V.E.; McCoy, S.W.; Steele, K.M. “Look, Your Muscles Are Firing!”: A Qualitative Study of Clinician Perspectives on the Use of Surface Electromyography in Neurorehabilitation. Arch. Phys. Med. Rehabil. 2019, 100, 663–675. [Google Scholar] [CrossRef] [PubMed]
- Feldner, H.A.; Papazian, C.; Peters, K.; Steele, K.M. “It’s All Sort of Cool and Interesting… but What Do I Do With It?” A Qualitative Study of Stroke Survivors’ Perceptions of Surface Electromyography. Front. Neurol. 2020, 11, 1037. [Google Scholar] [CrossRef]
- Sensor Location Recommendations. Available online: www.SENIAM.org (accessed on 16 November 2020).
- Li, X.; Zhou, P.; Aruin, A.S. Teager–Kaiser Energy Operation of Surface EMG Improves Muscle Activity Onset Detection. Ann. Biomed. Eng. 2007, 35, 1532–1538. [Google Scholar] [CrossRef] [PubMed]
- Solnik, S.; Rider, P.; Steinweg, K.; DeVita, P.; Hortobágyi, T. Teager–Kaiser energy operator signal conditioning improves EMG onset detection. Graefe Arch. Clin. Exp. Ophthalmol. 2010, 110, 489–498. [Google Scholar] [CrossRef]
- Yang, D.; Zhang, H.; Gu, Y.; Liu, H. Accurate EMG onset detection in pathological, weak and noisy myoelectric signals. Biomed. Signal Process. Control. 2017, 33, 306–315. [Google Scholar] [CrossRef]
- Wilson, R.D.; Page, S.J.; Delahanty, M.; Knutson, J.S.; Gunzler, D.D.; Sheffler, L.R.; Chae, J. Upper-limb recovery after stroke: A randomized controlled trial comparing EMG-triggered, cyclic, and sensory electrical stimulation. Neurorehabilit. Neural Repair 2016, 30, 978–987. [Google Scholar] [CrossRef]
- Kwakkel, G.; Winters, C.; Van Wegen, E.E.; Nijland, R.H.; Van Kuijk, A.A.; Visser-Meily, A.; De Groot, J.; De Vlugt, E.; Arendzen, J.H.; Geurts, A.C. Effects of unilateral upper limb training in two distinct prognostic groups early after stroke: The EXPLICIT-stroke randomized clinical trial. Neurorehabilit. Neural Repair 2016, 30, 804–816. [Google Scholar] [CrossRef] [PubMed]
- Shahar, N.; Schwartz, I.; Portnoy, S. Differences in muscle activity and fatigue of the upper limb between Task-Specific training and robot assisted training among individuals post stroke. J. Biomech. 2019, 89, 28–33. [Google Scholar] [CrossRef] [PubMed]
- Hogrel, J.-Y. Clinical applications of surface electromyography in neuromuscular disorders. Neurophysiol. Clin. Neurophysiol. 2005, 35, 59–71. [Google Scholar] [CrossRef] [PubMed]
- Winters, C.; Kwakkel, G.; Van Wegen, E.E.; Nijland, R.H.; Veerbeek, J.M.; Meskers, C.G. Moving stroke rehabilitation forward: The need to change research. NeuroRehabilitation 2018, 43, 19–30. [Google Scholar] [CrossRef]
- Kwakkel, G.; Lannin, N.A.; Borschmann, K.; English, C.; Coralie, E.; Churilov, L.; Saposnik, G.; Winstein, C.; Van Wegen, E.E.H.; Wolf, S.L.; et al. Standardized Measurement of Sensorimotor Recovery in Stroke Trials: Consensus-Based Core Recommendations from the Stroke Recovery and Rehabilitation Roundtable. Neurorehabilit. Neural Repair 2017, 31, 784–792. [Google Scholar] [CrossRef]
- Ahmad, S.A.; Chappell, P.H. Surface EMG pattern analysis of the wrist muscles at different speeds of contraction. J. Med. Eng. Technol. 2009, 33, 376–385. [Google Scholar] [CrossRef]
- Fisher, J.M.; Hammerla, N.Y.; Rochester, L.; Andras, P.; Walker, R.W. Body-worn sensors in Parkinson’s disease: Evaluating their acceptability to patients. Telemed. e-Health 2016, 22, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Luker, J.; Lynch, E.; Bernhardsson, S.; Bennett, L.; Bernhardt, J. Stroke Survivors’ Experiences of Physical Rehabilitation: A Systematic Review of Qualitative Studies. Arch. Phys. Med. Rehabil. 2015, 96, 1698–1708.e10. [Google Scholar] [CrossRef]
- Winstein, C.J.; Stein, J.; Arena, R.; Bates, B.; Cherney, L.R.; Cramer, S.C.; DeRuyter, F.; Eng, J.J.; Fisher, B.; Harvey, R.L.; et al. Guidelines for Adult Stroke Rehabilitation and Recovery. Stroke 2016, 47, e98–e169. [Google Scholar] [CrossRef]
- Stegeman, D.; Hermens, H. Standards for Surface Electromyography: The European Project Surface EMG for Non-Invasive Assessment of Muscles (SENIAM). 2007. Available online: https://citeseerx.ist.psu.edu/viewdoc/download?doi=10.1.1.623.2040&rep=rep1&type=pdf (accessed on 6 May 2021).
- Merriam, S.B.; Tisdell, E.J. Qualitative Research: A Guide to Design and Implementation, 4th ed.; John Wiley & Sons: San Francisco, CA, USA, 2015. [Google Scholar]
- Dideriksen, J.L.; Negro, F.; Falla, D.; Kristensen, S.R.; Mrachacz-Kersting, N.; Farina, D. Coherence of the Surface EMG and Common Synaptic Input to Motor Neurons. Front. Hum. Neurosci. 2018, 12, 207. [Google Scholar] [CrossRef] [PubMed]
- Farina, D.; Merletti, R.; Enoka, R.M. The extraction of neural strategies from the surface EMG: An update. J. Appl. Physiol. 2014, 117, 1215–1230. [Google Scholar] [CrossRef] [PubMed]
- McManus, L.; De Vito, G.; Lowery, M.M. Analysis and Biophysics of Surface EMG for Physiotherapists and Kinesiologists: Toward a Common Language with Rehabilitation Engineers. Front. Neurol. 2020, 11, 576729. [Google Scholar] [CrossRef] [PubMed]
- Jung, C.Y.; Park, J.-S.; Lim, Y.; Kim, Y.-B.; Park, K.K.; Moon, J.H.; Song, J.-H.; Lee, S. Estimating fatigue level of femoral and gastrocemius muscles based on surface electromyography in time and frequency domain. J. Mech. Med. Biol. 2018, 18, 1850042. [Google Scholar] [CrossRef]

| Days Post Stroke | Qualitative Functional Assessment |
|---|---|
| 7 | Patient demonstrated progress but also having difficulty with engaging left upper extremity fingers…Patient presented with emerging 2 point pinch and full palm grasp and release today. |
| 10 | Patient showing increased function in his left upper extremity. Fine/gross motor activity with great improvement and effort compared to previous session. |
| 12 | Patient participated in fine and gross motor strengthening and coordination with great effort. Demonstrating great key pinch and emerging pincer pinch. |
| 14 | Continues to demonstrate good progress with left upper extremity function performing exercises with great effort and improved control. Continues to need more work on wrist extension, thumb abduction, and middle/ring finger control. Difficulty with coordinating movements. |
| 17 | Patient presented with improved left upper extremity function. Increased coordination compared to previous session. |
| 19 | Patient demonstrated great progress again. Participated in fine motor activity with great effort and minimum cues for coordination. |
| Data | Total Recording Time | In Room | In OT | In PT | |||
|---|---|---|---|---|---|---|---|
| Time | Activity | Time | Activity | Time | Activity | ||
| Day 3 | 192 min | 192 min | Resting in bed | ||||
| Day 4 | 184 min | 161 min | Sitting in wheelchair | 23 min | Range of motion and strength testing | ||
| Day 7 | 186 min | 66 min | Back in room | 60 min | Fine motor coordination in pinch and grasp, passive and assisted range of motion, mirror box, and e-stim | 60 min | Therapeutic functional activity/bed mobility |
| Day 10 | 250 min | 135 min | Eating lunch and napping | 60 min | Therasponge and theraputty for L hand | 55 min | Therapeutic functional activity/bed mobility, gait/stair training |
| Day 12 | 240 min | 120 min | Eating lunch and napping | 60 min | Self-care/ADL management- cued to use L upper extremity as much as possible | 60 min | Therapeutic exercise/procedure, gait/stair training |
| Day 14 | 232 min | 112 min | Eating lunch and napping | 60 min | Left upper extremity exercises of thumb, finger, and wrist | 60 min | Shoulder flexion with towel, balance/vestibular training |
| Day 17 | 222 min | 102 min | Eating lunch and resting | 60 min | Self-care/ADL management, left upper extremity finger exercises | 60 min | Core strengthening and balance, upper extremity mirror therapy |
| Day 19 | 206 min | 86 min | Small group conference | 30 min | Self-care/ADL management, fine motor activity to increase coordination and endurance | 90 min | Balance/vestibular training |
| Sample Interview Questions | Tell Me about What You Remember about Having your Stroke. |
| What were your initial goals for recovery? | |
| Did your goals for recovery change over time? | |
| Describe what it was like for you to wear the sEMG sensors in the hospital. | |
| What kinds of information do you wish you could have received while wearing the sensors? | |
| In what way would you have preferred receiving such information? (i.e., visual or auditory signals, via cell phone or tablet, written report, etc.) | |
| Why might it be beneficial/detrimental to track muscle signals with sEMG during recovery from stroke? | |
| In which settings might using sEMG to track muscle activity be most useful? (i.e., home/community vs. hospital or rehab) |
| Quote | Theme/Topic | Participant Quote |
|---|---|---|
| 1 | Acute Recovery | “So when I had my stroke, um, it was pretty shocking. I knew what it was, I’ve had enough first aid training to know the signs. I knew I was having one, though I was still surprised…I was in the (first) hospital for five days, and had absolutely no motion in my arm, my hand, or my shoulder, on the left side.” |
| 2 | Inpatient Rehab | “The thing about rehab is you, you start to learn that it can become pretty routine. And they can actually set you up for doing a lot of stuff on your own. So you have to be very motivated to do that. Motivation was not a problem for me. Early on, because (I) had the time, and I had the drive to want to use, particularly, my arm and hand much more than I could…I told (the second) hospital that I wanted two plus weeks of rehab. By the time I left, I could walk on my own with a cane…I didn’t have, I had very, very limited motion in my arm, and my shoulder, and my hand. But their goal was to make me self-sufficient.” |
| 3 | Recovery at Home | “The arm took a long time, a frustratingly long time. When I went back to work, I still had to get help via software to type. I couldn’t use my hand…I could lift my arm and shrug my shoulders but the fingers itself wouldn’t work. And I was given a lot of home exercises, I would start my days doing all that. At least an hour or two of home exercises, um, pretty religiously too. And for the most part I tried to do normal things. I tried to do dishes, fold clothes, mow my lawn, clean the house…we played a lot of board games, and I would totally use my left hand for everything, which wasn’t normal for me, but was good for that…recovery. And it got better and better, you know, to the point where for the first time I could cut a piece of meat with a fork and a knife. It was pretty exhilarating, that was a big celebration, even though (laughing) my hand would still dip, like, into my horseradish sauce eating prime rib!” |
| 4 | Recovery is Ongoing | “(When people ask me how far I’ve come) I usually answer that in three ways. Totally, about 70%. My leg, about 90%, but there’s still differences and weakness in my knee. Arm…probably 60%. There are days it feels like 90%, and there are days, or times in a day, where it feels less. I know I’m not 100%, and I may not be either, and I’m okay with that. You know, cause I can walk. I can run, I can talk. But the things that I notice now, they’re subtle... subtle to most people, but they’re very noticeable to me.” |
| 5 | Perceptions of sEMG Use | “I was intrigued…however, I don’t know what all the readings tell you, I mean, so that is of interest, what you all were seeing… I didn’t dislike anything though, but what I thought was compelling about it was everybody told me that this (hand function return) would be slow. Well, guess what? Eight months later, this is still recovering. So I was, I was hopeful that it would show signs of things that are occurring when I couldn’t physically feel it…if you had other scientific evidence that things were happening, even beyond their notion that it would, it gives you a lot of hope. You just have to be patient, and it’s harder to take when someone tells you, but easier to understand if someone actually shows you.” |
| 6 | Limitations of sEMG Sensors | “When you and I got together, it was a lot to take on and off. That’s kind of a pain, right? I’m wondering if there is a way to do kind of both. That… that has multiple individual muscle sensors where you pull a sleeve on, for example. As long as you align it correctly, it’s getting a, a number of muscles.” |
| 7 | sEMG as a Motivator for Improvement | “For the most part when I was at my, my worst, I couldn’t tell if things were really going differently, but maybe it was cause it was so subtle. Cause I want big changes or I want big improvements. But again seeing some improvement, whatever scale, scientifically with your data, could be a big boost. Because there were times where I can tell no difference at all, but I’m sure there was something there. And at home, you’re doing this on your own, that’s the longer-term harder stuff. If you have a way of telling that at home, it’s kind of nice to get that affirmation through any means you can.” |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feldner, H.A.; Papazian, C.; Peters, K.M.; Creutzfeldt, C.J.; Steele, K.M. Clinical Use of Surface Electromyography to Track Acute Upper Extremity Muscle Recovery after Stroke: A Descriptive Case Study of a Single Patient. Appl. Syst. Innov. 2021, 4, 32. https://doi.org/10.3390/asi4020032
Feldner HA, Papazian C, Peters KM, Creutzfeldt CJ, Steele KM. Clinical Use of Surface Electromyography to Track Acute Upper Extremity Muscle Recovery after Stroke: A Descriptive Case Study of a Single Patient. Applied System Innovation. 2021; 4(2):32. https://doi.org/10.3390/asi4020032
Chicago/Turabian StyleFeldner, Heather A., Christina Papazian, Keshia M. Peters, Claire J. Creutzfeldt, and Katherine M. Steele. 2021. "Clinical Use of Surface Electromyography to Track Acute Upper Extremity Muscle Recovery after Stroke: A Descriptive Case Study of a Single Patient" Applied System Innovation 4, no. 2: 32. https://doi.org/10.3390/asi4020032
APA StyleFeldner, H. A., Papazian, C., Peters, K. M., Creutzfeldt, C. J., & Steele, K. M. (2021). Clinical Use of Surface Electromyography to Track Acute Upper Extremity Muscle Recovery after Stroke: A Descriptive Case Study of a Single Patient. Applied System Innovation, 4(2), 32. https://doi.org/10.3390/asi4020032






