Prey Size Decline as a Unifying Ecological Selecting Agent in Pleistocene Human Evolution
Abstract
1. Introduction
2. Pleistocene Decline in Prey Size
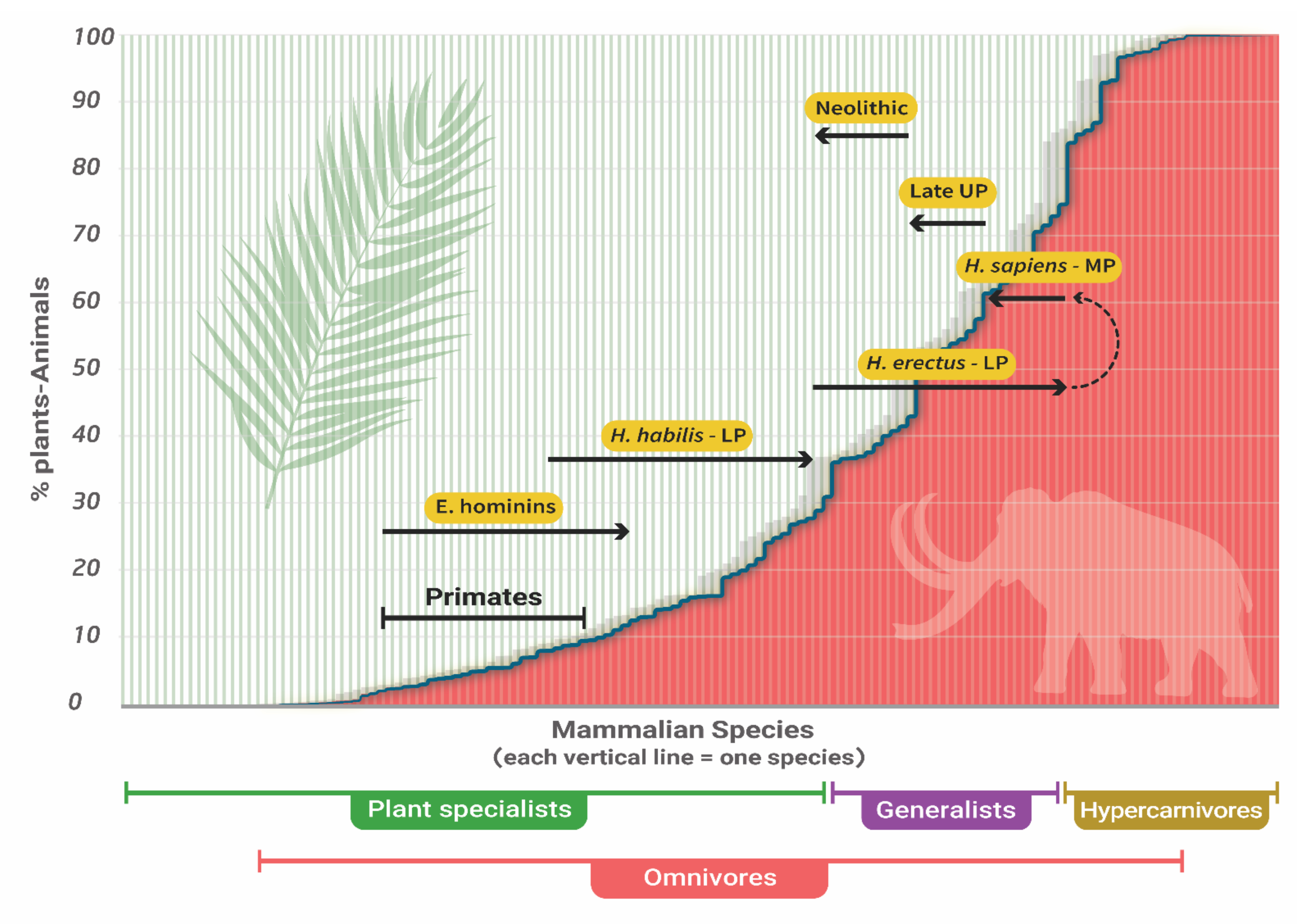
3. The Trophic Position of Humans
4. Specialization in Large Prey
4.1. High Relative Biomass
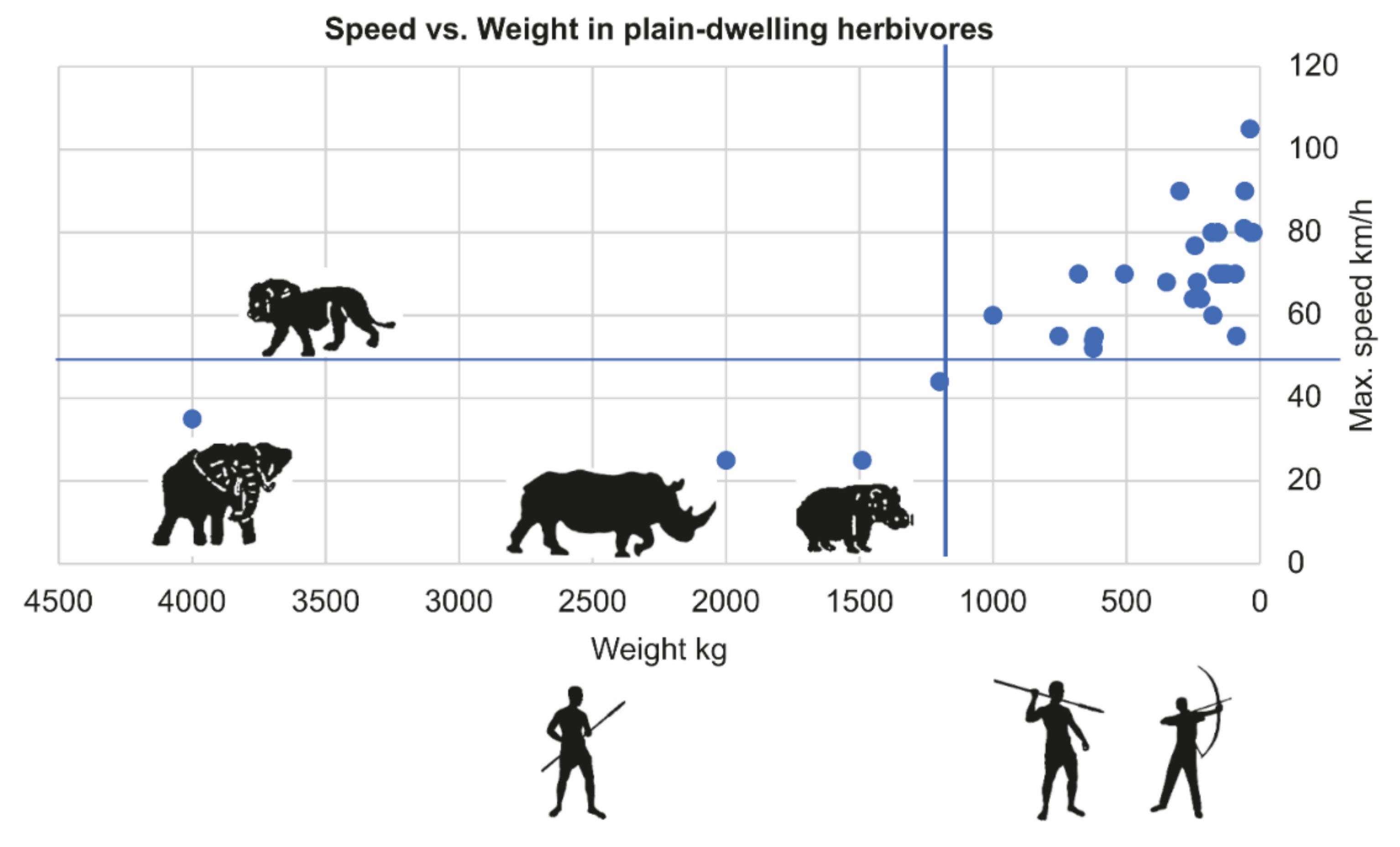
4.2. Not Escaping–Easier Tracking and Less Complex Hunting Tools
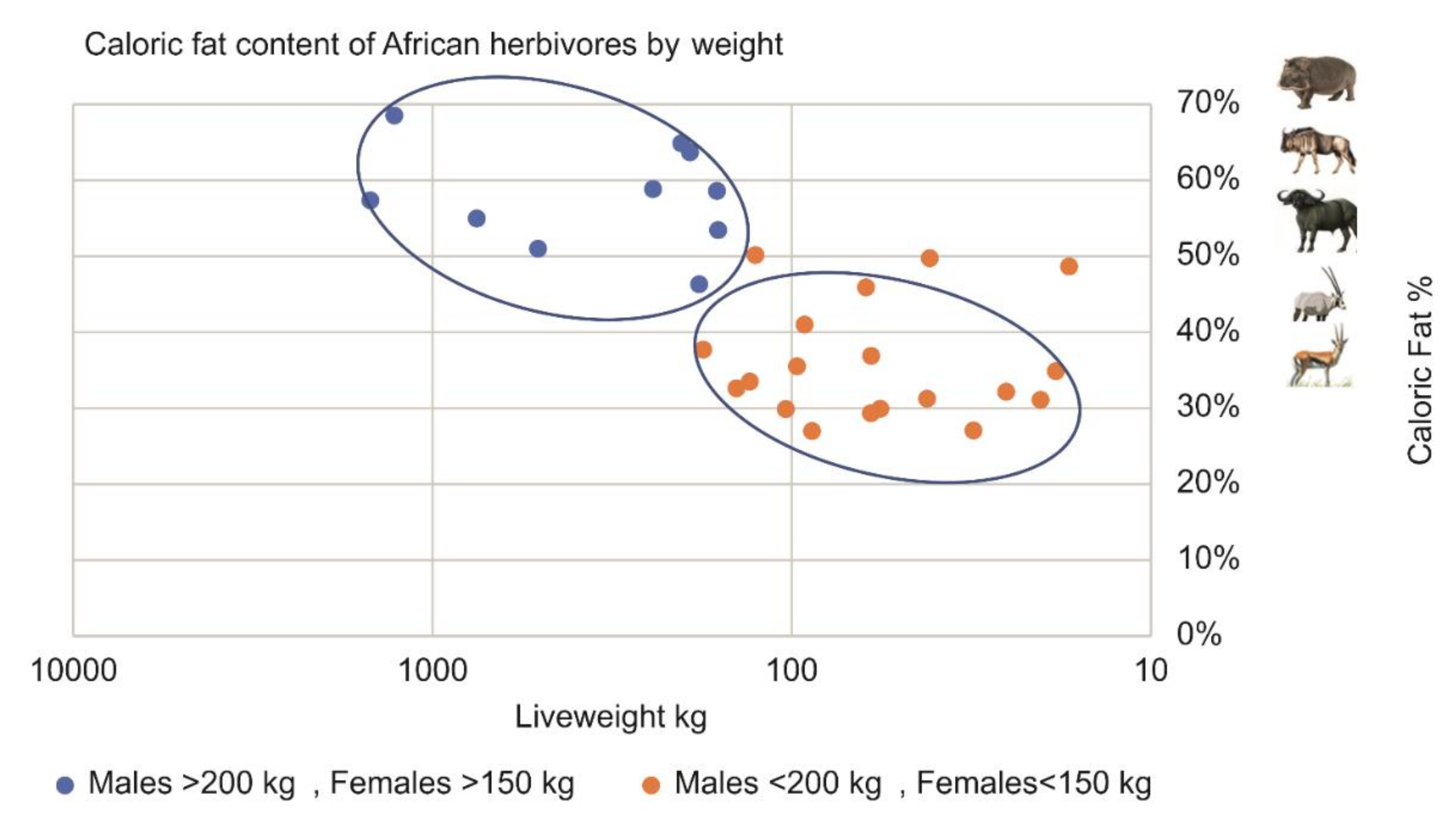
4.3. Larger Prey Contains Higher Body Fat Levels
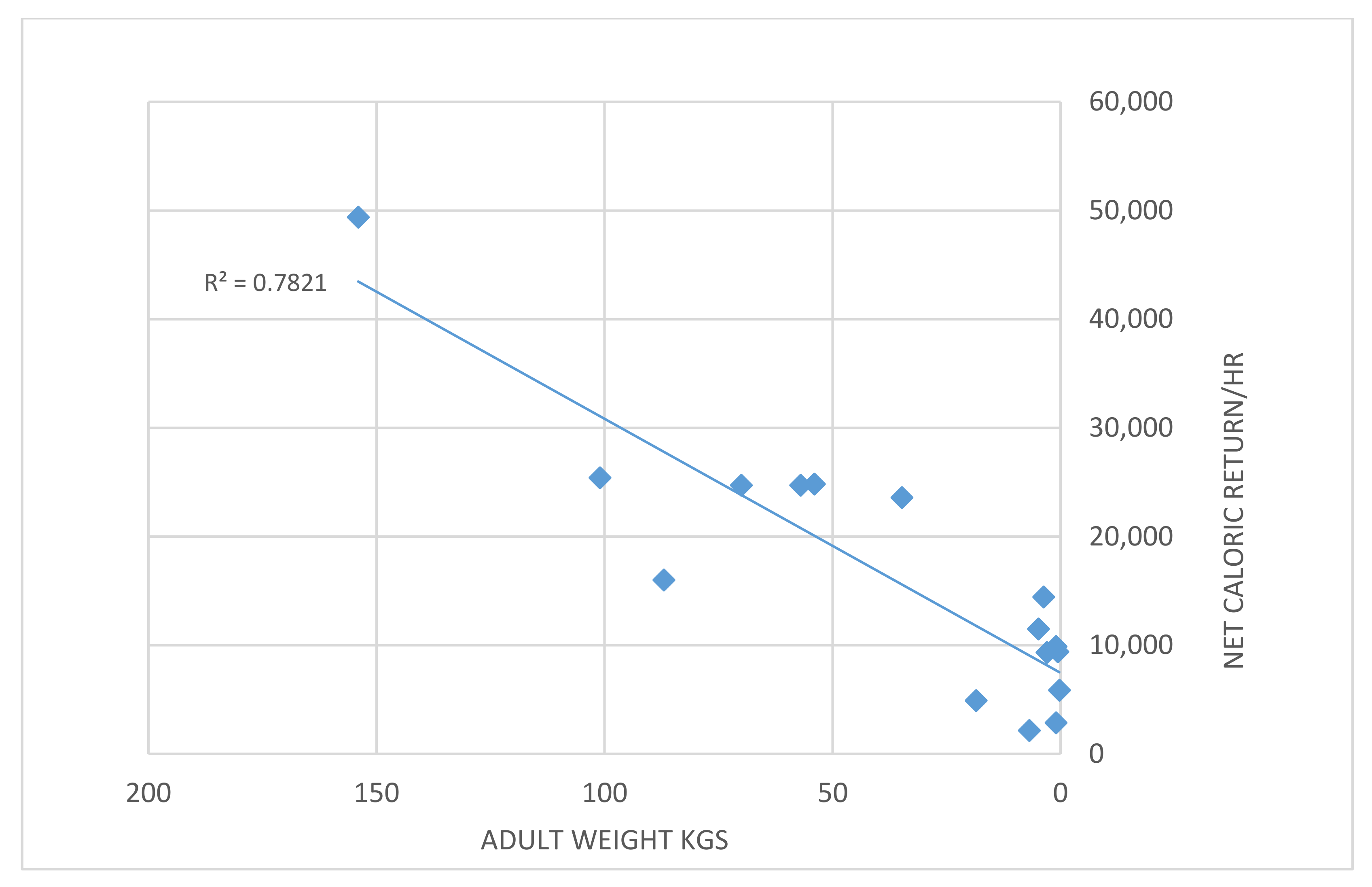
4.4. Larger Animals Provide a Higher Energetic Return
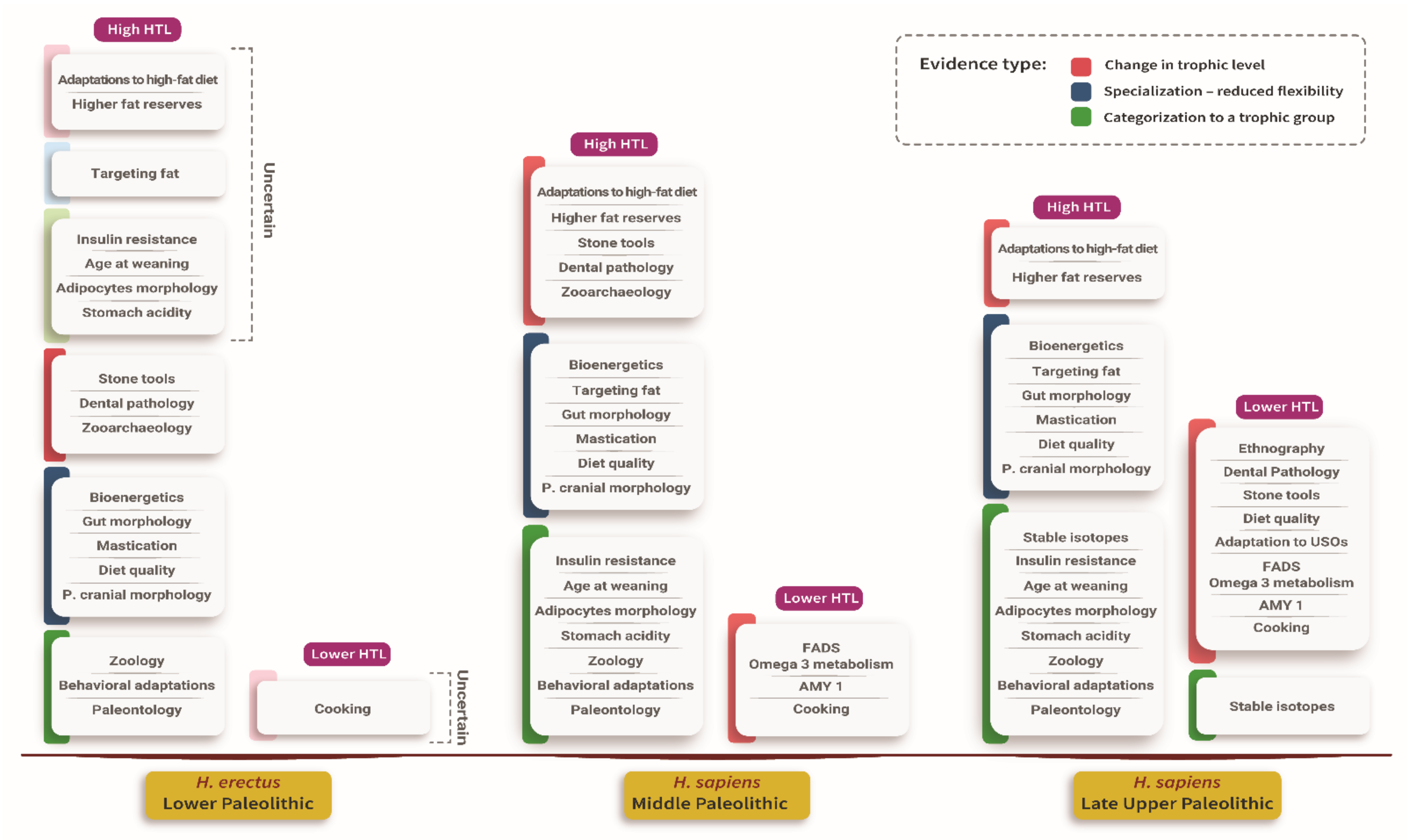
4.5. Evidence for Specialization in Large Prey
5. Anthropogenic Contribution to Prey Size Decline
6. The Decline in Prey Size as an Agent of Selection: Preliminary Case Studies
6.1. Brain Size, Language, Stone-Tools, and Fire
6.2. Hunting of Large Animals by H. erectus (sensu lato)
6.3. The Evolution of H. sapiens
6.4. The Extinction of the Neandertal
6.5. Increased Plant Food Consumption from the Upper Paleolithic Onward
6.6. Dog Domestication
6.7. Plant and Animal Domestication at Different Times and Places
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- MacColl, A.D. The ecological causes of evolution. Trends Ecol. Evol. 2011, 26, 514–522. [Google Scholar] [CrossRef] [PubMed]
- Ben-Dor, M.; Barkai, R. The importance of large prey animals during the Pleistocene and the implications of their extinction on the use of dietary ethnographic analogies. J. Anthropol. Archaeol. 2020, 59, 101192. [Google Scholar] [CrossRef]
- Ben-Dor, M.; Barkai, R. Supersize does matter: The importance of large prey in Paleolithic subsistence and a method for measurement of its significance in zooarchaeological assemblages. In Human-Elephant Interactions: From Past to Present; Konidaris, G., Barkai, R., Tourloukis, V., Harvati, K., Eds.; Tübingen University Press: Tübingen, Germany, in press.
- Ben-Dor, M.; Sirtoli, R.; Barkai, R. The evolution of the human trophic level during the Pleistocene. Yb. Phys. Anthropol. 2021. [Google Scholar]
- Faith, J.T.; Rowan, J.; Du, A. Early hominins evolved within non-analog ecosystems. Proc. Natl. Acad. Sci. USA 2019, 116, 21478–21483. [Google Scholar] [CrossRef] [PubMed]
- Smith, F.A.; Smith, R.E.E.; Lyons, S.K.; Payne, J.L. Body size downgrading of mammals over the late Quaternary. Science 2018, 360, 310–313. [Google Scholar] [CrossRef]
- Faith, J.T.; Rowan, J.; Du, A.; Barr, W.A. The uncertain case for human-driven extinctions prior to Homo sapiens. Quat. Res. 2020, 96, 88–104. [Google Scholar] [CrossRef]
- Faith, J.T.; Potts, R.; Plummer, T.W.; Bishop, L.C.; Marean, C.W.; Tryon, C.A. New perspectives on middle Pleistocene change in the large mammal faunas of East Africa: Damaliscus hypsodon sp. nov.(Mammalia, Artiodactyla) from Lainyamok, Kenya. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2012, 361, 84–93. [Google Scholar] [CrossRef]
- Potts, R.; Behrensmeyer, A.K.; Faith, J.T.; Tryon, C.A.; Brooks, A.S.; Yellen, J.E.; Deino, A.L.; Kinyanjui, R.; Clark, J.B.; Haradon, C.M. Environmental dynamics during the onset of the Middle Stone Age in eastern Africa. Science 2018, 360, 86–90. [Google Scholar] [CrossRef]
- Ben-Dor, M.; Gopher, A.; Hershkovitz, I.; Barkai, R. Man the fat hunter: The demise of Homo erectus and the emergence of a new hominin lineage in the Middle Pleistocene (ca. 400 kyr) Levant. PLoS ONE 2011, 6, e28689. [Google Scholar] [CrossRef]
- Dembitzer, J.; Barkai, R.; Ben-Dor, M.; Meiri, S. 1.5 Million Years of Hunting Down the Body Size Distribution in the Paleolithic Southern Levant. Unpublished work. 2021. [Google Scholar]
- Koch, P.L.; Barnosky, A.D. Late Quaternary Extinctions: State of the Debate. Annu. Rev. Ecol. Evol. Syst. 2006, 37, 215–252. [Google Scholar] [CrossRef]
- Boivin, N.L.; Zeder, M.A.; Fuller, D.Q.; Crowther, A.; Larson, G.; Erlandson, J.M.; Denham, T.; Petraglia, M.D. Ecological consequences of human niche construction: Examining long-term anthropogenic shaping of global species distributions. Proc. Natl. Acad. Sci. USA 2016, 113, 6388–6396. [Google Scholar] [CrossRef] [PubMed]
- Sandom, C.; Faurby, S.; Sandel, B.; Svenning, J.-C. Global late Quaternary megafauna extinctions linked to humans, not climate change. Proc. R. Soc. B Biol. Sci. 2014, 281, 20133254. [Google Scholar] [CrossRef] [PubMed]
- Smith, F.A.; Smith, R.E.E.; Lyons, S.K.; Payne, J.L.; Villaseñor, A. The accelerating influence of humans on mammalian macroecological patterns over the late Quaternary. Quat. Sci. Rev. 2019, 211, 1–16. [Google Scholar] [CrossRef]
- Dirzo, R.; Young, H.S.; Galetti, M.; Ceballos, G.; Isaac, N.J.; Collen, B. Defaunation in the Anthropocene. Science 2014, 345, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Bodmer, R.E. Managing Amazonian wildlife: Biological correlates of game choice by detribalized hunters. Ecol. Appl. 1995, 5, 872–877. [Google Scholar] [CrossRef]
- Jerozolimski, A.; Peres, C.A. Bringing home the biggest bacon: A cross-site analysis of the structure of hunter-kill profiles in Neotropical forests. Biol. Conserv. 2003, 111, 415–425. [Google Scholar] [CrossRef]
- Andermann, T.; Faurby, S.; Turvey, S.T.; Antonelli, A.; Silvestro, D. The past and future human impact on mammalian diversity. Sci. Adv. 2020, 6, eabb2313. [Google Scholar] [CrossRef]
- Meltzer, D.J. Overkill, glacial history, and the extinction of North America’s Ice Age megafauna. Proc. Natl. Acad. Sci. USA 2020, 117, 28555–28563. [Google Scholar] [CrossRef]
- Rendu, W.; Renou, S.; Soulier, M.-C.; Rigaud, S.; Roussel, M.; Soressi, M. Subsistence strategy changes during the Middle to Upper Paleolithic transition reveals specific adaptations of Human Populations to their environment. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Wilkinson, P. Ecosystem models and demographic hypotheses: Predation and prehistory in North America. In Models in Archaeology; Clark, D., Ed.; Routledge: London, UK, 2014; pp. 543–576. [Google Scholar]
- Pineda-Munoz, S.; Alroy, J. Dietary characterization of terrestrial mammals. Proc. R. Soc. B Biol. Sci. 2014, 281, 20141173. [Google Scholar] [CrossRef]
- Hempson, G.P.; Archibald, S.; Bond, W.J. A continent-wide assessment of the form and intensity of large mammal herbivory in Africa. Science 2015, 350, 1056–1061. [Google Scholar] [CrossRef]
- Agam, A.; Barkai, R. Elephant and mammoth hunting during the Paleolithic: A Review of the relevant archaeological, ethnographic and ethno-historical records. Quaternary 2018, 1, 3. [Google Scholar] [CrossRef]
- Churchill, S.E. Weapon technology, prey size selection, and hunting methods in modern hunter-gatherers: Implications for hunting in the Palaeolithic and Mesolithic. Archeol. Pap. Am. Anthropol. Assoc. 1993, 4, 11–24. [Google Scholar] [CrossRef]
- Ben Dor, M. Data file—Supersize does matter. The importance of large prey in Paleolithic subsistence and a method for measuring its significance in zooarchaeological assemblages. Mendeley Data 2020, 2. [Google Scholar] [CrossRef]
- Speth, J.D.; Spielmann, K.A. Energy source, protein metabolism, and hunter-gatherer subsistence strategies. J. Anthropol. Archaeol. 1983, 2, 1–31. [Google Scholar] [CrossRef]
- Kelly, R.L. The Lifeways of Hunter-Gatherers: The Foraging Spectrum; Cambridge University Press: Cambridge, UK, 2013. [Google Scholar]
- Pitts, G.C.; Bullard, T.R. Some interspecific aspects of body composition in mammals. In Body Composition in Animals and Man; National Academy of Sciences: Washington, DC, USA, 1967; pp. 45–70. [Google Scholar]
- Ledger, H.P. Body composition as a basis for a comparative study of some East African mammals. Symp. Zool. Soc. Lond. 1968, 21, 289–310. [Google Scholar]
- Futuyma, D.J.; Moreno, G. The evolution of ecological specialization. Annu. Rev. Ecol. Syst. 1988, 19, 207–233. [Google Scholar] [CrossRef]
- Speth, J.D. Big-Game Hunting: Protein, Fat, or Politics. In The Paleoanthropology and Archaeology of Big-Game Hunting; Springer: Berlin/Heidelberg, Germany, 2010; pp. 149–161. [Google Scholar]
- Hawkes, K.; O’Connell, J.F.; Blurton Jones, N.G.; Bell, D.; Bird, R.; Bird, D.; Hames, R.; Ivey, P.; Judge, D.; Kazankov, A. Hunting and nuclear families: Some lessons from the Hadza about mens work. Curr. Anthropol. 2001, 42, 681–709. [Google Scholar] [CrossRef]
- Wood, B.M.; Marlowe, F.W. Household and kin provisioning by Hadza men. Hum. Nat. 2013, 24, 280–317. [Google Scholar] [CrossRef]
- Gurven, M.; Hill, K. Hunting as subsistence and mating effort? A re-evaluation of “Man the Hunter”, the sexual division of labor and the evolution of the nuclear family. In Proceedings of IUSSP Seminar on Male Life History; University of California: Santa Barbara, CA, USA, 2006. [Google Scholar]
- Dominguez-Rodrigo, M.; Bunn, H.T.; Mabulla, A.Z.P.; Baquedano, E.; Uribelarrea, D.; Perez-Gonzalez, A.; Gidna, A.; Yravedra, J.; Diez-Martin, F.; Egeland, C.P.; et al. On meat eating and human evolution: A taphonomic analysis of BK4b (Upper Bed II, Olduvai Gorge, Tanzania), and its bearing on hominin megafaunal consumption. Quat. Int. 2014, 322, 129–152. [Google Scholar] [CrossRef]
- Stiner, M.C.; Gopher, A.; Barkai, R. Hearth-side socioeconomics, hunting and paleoecology during the late Lower Paleolithic at Qesem Cave, Israel. J. Hum. Evol. 2011, 60, 213–233. [Google Scholar] [CrossRef]
- Saladié, P.; Huguet, R.; Díez, C.; Rodríguez-Hidalgo, A.; Cáceres, I.; Vallverdú, J.; Rosell, J.; de Castro, J.M.B.; Carbonell, E. Carcass transport decisions in Homo antecessor subsistence strategies. J. Hum. Evol. 2011, 61, 425–446. [Google Scholar] [CrossRef]
- Bunn, H.T.; Pickering, T.R. Bovid mortality profiles in paleoecological context falsify hypotheses of endurance running–hunting and passive scavenging by early Pleistocene hominins. Quat. Res. 2010, 74, 395–404. [Google Scholar] [CrossRef]
- Stiner, M.C. The use of mortality patterns in archaeological studies of hominid predatory adaptations. J. Anthropol. Archaeol. 1990, 9, 305–351. [Google Scholar] [CrossRef]
- Castel, J.-C.; Discamps, E.; Soulier, M.-C.; Sandgathe, D.; Dibble, H.L.; McPherron, S.J.; Goldberg, P.; Turq, A. Neandertal subsistence strategies during the Quina Mousterian at Roc de Marsal (France). Quat. Int. 2016, 433, 140–156. [Google Scholar] [CrossRef]
- Adler, D. Ahead of the game. Curr. Anthropol. 2006, 47, 89–118. [Google Scholar] [CrossRef]
- Gaudzinski, S.; Roebroeks, W. Adults only. Reindeer hunting at the Middle Palaeolithic site Salzgitter Lebenstedt, Northern Germany. J. Hum. Evol. 2000, 38, 497–521. [Google Scholar] [CrossRef]
- Owen-Smith, R.N. Adaptive Herbivore Ecology: From Resources to Populations in Variable Environments; Cambridge University Press: Cambridge, UK, 2002. [Google Scholar]
- Cordain, L.; Eaton, S.B.; Sebastian, A.; Mann, N.; Lindeberg, S.; Watkins, B.A.; O’Keefe, J.H.; Brand-Miller, J. Origins and evolution of the Western diet: Health implications for the 21st century. Am. J. Clin. Nutr. 2005, 81, 341–354. [Google Scholar] [CrossRef] [PubMed]
- Speth, J.D. Bison Kills and Bone Counts: Decision Making by Ancient Hunters; University of Chicago Press: Chicago, IL, USA, 1983. [Google Scholar]
- Binford, L.R. Nunamiut Ethnoarchaeology; Academic Press: New York, NY, USA, 1978. [Google Scholar]
- Morin, E. Fat composition and Nunamiut decision-making: A new look at the marrow and bone grease indices. JAS 2007, 34, 69–82. [Google Scholar] [CrossRef]
- Bar-Oz, G.; Munro, N.D. Gazelle bone marrow yields and Epipalaeolithic carcass exploitation strategies in the southern Levant. JAS 2007, 34, 946–956. [Google Scholar] [CrossRef]
- Stiner, M.C. An Unshakable Middle Paleolithic? Trends versus Conservatism in the Predatory Niche and Their Social Ramifications. Curr. Anthropol. 2013, 54, S288–S304. [Google Scholar] [CrossRef]
- McKinney, M.L. Extinction vulnerability and selectivity: Combining ecological and paleontological views. Annu. Rev. Ecol. Syst. 1997, 28, 495–516. [Google Scholar] [CrossRef]
- Gaillard, J.-M.; Festa-Bianchet, M.; Yoccoz, N.; Loison, A.; Toigo, C. Temporal variation in fitness components and population dynamics of large herbivores. Annu. Rev. Ecol. Syst. 2000, 31, 367–393. [Google Scholar] [CrossRef]
- Lincoln, A.E.; Quinn, T.P. Optimal foraging or surplus killing: Selective consumption and discarding of salmon by brown bears. Behav. Ecol. 2019, 30, 202–212. [Google Scholar] [CrossRef]
- Speth, J.D.; Clark, J.L. Hunting and overhunting in the Levantine Late Middle Palaeolithic. Before Farm. 2006, 2006, 1–42. [Google Scholar] [CrossRef]
- Lee, S.-H.; Wolpoff, M.H. The pattern of evolution in Pleistocene human brain size. Paleobiology 2003, 29, 186–196. [Google Scholar] [CrossRef]
- Hawks, J. Selection for smaller brains in Holocene human evolution. arXiv preprint 2011, arXiv:1102.5604. [Google Scholar]
- Henneberg, M. Decrease of human skull size in the Holocene. Hum. Biol. 1988, 60, 395–405. [Google Scholar]
- Reardon, P.; Seidlitz, J.; Vandekar, S.; Liu, S.; Patel, R.; Park, M.T.M.; Alexander-Bloch, A.; Clasen, L.S.; Blumenthal, J.D.; Lalonde, F.M. Normative brain size variation and brain shape diversity in humans. Science 2018, 360, 1222–1227. [Google Scholar] [CrossRef]
- Benson-Amram, S.; Dantzer, B.; Stricker, G.; Swanson, E.M.; Holekamp, K.E. Brain size predicts problem-solving ability in mammalian carnivores. Proc. Natl. Acad. Sci. USA 2016, 113, 2532–2537. [Google Scholar] [CrossRef]
- Dunbar, R. The social brain hypothesis. Evol. Anthropol. Issues News Rev. 1998, 6, 178–190. [Google Scholar] [CrossRef]
- Burini, R.C.; Leonard, W.R. The evolutionary roles of nutrition selection and dietary quality in the human brain size and encephalization. Nutrire 2018, 43, 19. [Google Scholar] [CrossRef]
- DeCasien, A.R.; Williams, S.A.; Higham, J.P. Primate brain size is predicted by diet but not sociality. Nat. Ecol. Evol. 2017, 1, 0112. [Google Scholar] [CrossRef] [PubMed]
- González-Forero, M.; Gardner, A. Inference of ecological and social drivers of human brain-size evolution. Nature 2018, 557, 554. [Google Scholar] [CrossRef]
- Leonard, W.R.; Snodgrass, J.J.; Robertson, M.L. Effects of brain evolution on human nutrition and metabolism. Annu. Rev. Nutr. 2007, 27, 311–327. [Google Scholar] [CrossRef]
- Powell, L.E.; Isler, K.; Barton, R.A. Re-evaluating the link between brain size and behavioural ecology in primates. Proc. R. Soc. B 2017, 284, 20171765. [Google Scholar] [CrossRef] [PubMed]
- Rosati, A.G. Foraging cognition: Reviving the ecological intelligence hypothesis. Trends Cogn. Sci. 2017, 21, 691–702. [Google Scholar] [CrossRef]
- Aiello, L.C.; Wheeler, P. The expensive-tissue hypothesis: The brain and the digestive system in human and primate evolution. Curr. Anthropol. 1995, 36, 199–221. [Google Scholar] [CrossRef]
- Navarrete, A.; Schaik, C.P.V.; Isler, K. Energetics and the evolution of human brain size. Nature 2011, 480, 91–93. [Google Scholar] [CrossRef]
- Stanford, C.B.; Bunn, H.T. Meat-Eating & Human Evolution; Oxford University Press: Oxford, UK, 2001. [Google Scholar]
- Brain, C.K. Do we owe our intelligence to a predatory past? James Arthur Lect. Evol. Hum. Brain 2000, 70, 1–32. [Google Scholar]
- Churchill, S.E.; Walker, C.S.; Schwartz, A.M. Home-range size in large-bodied carnivores as a model for predicting neandertal territory size. Evol. Anthropol. Issues News Rev. 2016, 25, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Pontzer, H. Constrained total energy expenditure and the evolutionary biology of energy balance. Exerc. Sport Sci. Rev. 2015, 43, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Flinn, M.V.; Geary, D.C.; Ward, C.V. Ecological dominance, social competition, and coalitionary arms races: Why humans evolved extraordinary intelligence. Evol. Hum. Behav. 2005, 26, 10–46. [Google Scholar] [CrossRef]
- Geary, D.C. Evolution and cognitive development. In Evolutionary Perspectives on Human Development; Burgess, R.L., MacDonald, K., Eds.; Sage: Thousand Oaks, CA, USA, 2004; pp. 99–103. [Google Scholar]
- Blurton Jones, N.; Konner, M.J. Kung knowledge of animal behavior. In Kalahari Hunter-Gatherers; Lee, B.R., DeVore, I., Eds.; Harvard University Press: Cambridge, MA, USA, 1976; pp. 325–348. [Google Scholar]
- Liebenberg, L. Persistence hunting by modern hunter-gatherers. Curr. Anthropol. 2006, 47, 1017–1026. [Google Scholar] [CrossRef]
- Liebenberg, L. The Origin of Science—The Evolutionary Roots of Scientific Reasoning and Its Implications for Citizen Science; CyberTracker: Cape Town, South Africa, 2013. [Google Scholar]
- Brink, J. Imagining Head-Smashed-in Aboriginal Buffalo Hunting on the Northern Plains; Athabasca University Press: Edmonton, AB, Canada, 2008. [Google Scholar]
- Davidson, I. Cognitive evolution and origins of language and speech. In Encyclopedia of Global Archaeology; Smith, C., Ed.; Springer: New York, NY, USA, 2013; pp. 1530–1543. [Google Scholar]
- Corballis, M.C. The gradual evolution of language. HUMANA MENTE J. Philos. Stud. 2014, 7, 39–60. [Google Scholar]
- Dornhaus, A.; Chittka, L. Why do honey bees dance? Behav. Ecol. Sociobiol. 2004, 55, 395–401. [Google Scholar] [CrossRef]
- Herzlinger, G.; Wynn, T.; Goren-Inbar, N. Expert cognition in the production sequence of Acheulian cleavers at Gesher Benot Ya’aqov, Israel: A lithic and cognitive analysis. PLoS ONE 2017, 12, e0188337. [Google Scholar] [CrossRef]
- Putt, S.S. Human Brain Activity During Stone Tool Production: Tracing the Evolution of Cognition and Language; The University of Iowa: Iowa City, IA, USA, 2016. [Google Scholar]
- Vaesen, K. The cognitive bases of human tool use. Behav. Brain Sci. 2012, 35, 203–218. [Google Scholar] [CrossRef]
- Smith, H.L.; Goebel, T. Origins and spread of fluted-point technology in the Canadian Ice-Free Corridor and eastern Beringia. Proc. Natl. Acad. Sci. USA 2018, 115, 4116–4121. [Google Scholar] [CrossRef]
- Gärdenfors, P.; Lombard, M. Causal cognition, force dynamics and early hunting technologies. Front. Psychol. 2018, 9, 87. [Google Scholar] [CrossRef]
- Stiner, M.C.; Kuhn, S.L. Changes in the “Connectedness” and resilience of paleolithic societies in Mediterranean ecosystems. Hum. Ecol. 2006, 34, 693–712. [Google Scholar] [CrossRef]
- Wrangham, R. Control of Fire in the Paleolithic Evaluating the Cooking Hypothesis. Curr. Anthropol. 2017, 58, S303–S313. [Google Scholar] [CrossRef]
- Gowlett, J. The discovery of fire by humans: A long and convoluted process. Philos. Trans. R. Soc. B Biol. Sci. 2016, 371, 20150164. [Google Scholar] [CrossRef] [PubMed]
- Shimelmitz, R.; Kuhn, S.L.; Jelinek, A.J.; Ronen, A.; Clark, A.E.; Weinstein-Evron, M. ‘Fire at will’: The emergence of habitual fire use 350,000 years ago. J. Hum. Evol. 2014, 77, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Roebroeks, W.; Villa, P. On the earliest evidence for habitual use of fire in Europe. Proc. Natl. Acad. Sci. USA 2011, 108, 5209–5214. [Google Scholar] [CrossRef] [PubMed]
- Barkai, R.; Blasco, R.; Rosell, J.; Gopher, A. A land of flint and fallow deer: Human persistence at Middle Pleistocene Qesem Cave. In Crossing the Human Threshold; Pope, M., McNabb, J., Gamble, C., Eds.; Routledge: London, UK, 2017; pp. 82–104. [Google Scholar]
- Venditti, F.; Cristiani, E.; Nunziante-Cesaro, S.; Agam, A.; Lemorini, C.; Barkai, R. Animal residues found on tiny Lower Paleolithic tools reveal their use in butchery. Sci. Rep. 2019, 9, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Barkai, R.; Rosell, J.; Blasco, R.; Gopher, A. Fire for a reason: Barbecue at Middle Pleistocene Qesem Cave, Israel. Curr. Anthropol. 2017, 58, S314–S328. [Google Scholar] [CrossRef]
- Pearce, E.; Stringer, C.; Dunbar, R.I. New insights into differences in brain organization between Neanderthals and anatomically modern humans. Proc. R. Soc. B Biol. Sci. 2013, 280, 20130168. [Google Scholar] [CrossRef]
- Wynn, T.; Overmann, K.A.; Coolidge, F.L. The false dichotomy: A refutation of the Neandertal indistinguishability claim. J. Anthropol. Sci. 2016, 94, 1–22. [Google Scholar]
- Lombard, M.; Högberg, A. Four-field co-evolutionary model for human cognition: Variation in the Middle Stone Age/Middle Palaeolithic. J. Archaeol. Method Theory 2021, 1–36. [Google Scholar]
- Antón, S.C.; Potts, R.; Aiello, L.C. Evolution of early Homo: An integrated biological perspective. Science 2014, 345, 1236828. [Google Scholar] [CrossRef]
- Owen, R.B.; Muiruri, V.M.; Lowenstein, T.K.; Renaut, R.W.; Rabideaux, N.; Luo, S.; Deino, A.L.; Sier, M.J.; Dupont-Nivet, G.; McNulty, E.P. Progressive aridification in East Africa over the last half million years and implications for human evolution. Proc. Natl. Acad. Sci. USA 2018, 115, 11174–11179. [Google Scholar] [CrossRef] [PubMed]
- Domínguez-Rodrigo, M.; Pickering, T.R. The meat of the matter: An evolutionary perspective on human carnivory. Azania Archaeol. Res. Afr. 2017, 52, 4–32. [Google Scholar] [CrossRef]
- Roach, N.T.; Du, A.; Hatala, K.G.; Ostrofsky, K.R.; Reeves, J.S.; Braun, D.R.; Harris, J.W.; Behrensmeyer, A.K.; Richmond, B.G. Pleistocene animal communities of a 1.5 million-year-old lake margin grassland and their relationship to Homo erectus paleoecology. J. Hum. Evol. 2018, 122, 70–83. [Google Scholar] [CrossRef]
- Bunn, H.T.; Ezzo, J.A. Hunting and scavenging by Plio-Pleistocene hominids: Nutritional constraints, archaeological patterns, and behavioural implications. JAS 1993, 20, 365–398. [Google Scholar] [CrossRef]
- Isaac, G.L. The archaeology of human origins: Studies of the Lower Pleistocene in East Africa, 1971–1981. Adv. World Archaeol. 1984, 3, 1–86. [Google Scholar]
- Klein, R.G. The archaeological significance of animal bones from Acheulean sites in southern Africa. Afr. Archaeol. Rev. 1988, 6, 3–25. [Google Scholar] [CrossRef]
- Bunn, H.T.; Gurtov, A.N. Prey mortality profiles indicate that Early Pleistocene Homo at Olduvai was an ambush predator. Quat. Int. 2014, 322, 44–53. [Google Scholar] [CrossRef]
- Bunn, H.T. Large ungulate mortality profiles and ambush hunting by Acheulean-age hominins at Elandsfontein, Western Cape Province, South Africa. JAS 2019, 107, 40–49. [Google Scholar] [CrossRef]
- Hublin, J.-J.; Ben-Ncer, A.; Bailey, S.E.; Freidline, S.E.; Neubauer, S.; Skinner, M.M.; Bergmann, I.; Le Cabec, A.; Benazzi, S.; Harvati, K. New fossils from Jebel Irhoud, Morocco and the pan-African origin of Homo sapiens. Nature 2017, 546, 289. [Google Scholar] [CrossRef] [PubMed]
- Potts, R. Variability selection in hominid evolution. Evol. Anthropol. Issues News Rev. 1998, 7, 81–96. [Google Scholar] [CrossRef]
- Snell-Rood, E.C.; Steck, M.K. Behaviour shapes environmental variation and selection on learning and plasticity: Review of mechanisms and implications. Anim. Behav. 2019, 147, 147–156. [Google Scholar] [CrossRef]
- Fox, R.J.; Donelson, J.M.; Schunter, C.; Ravasi, T.; Gaitán-Espitia, J.D. Beyond buying time: The role of plasticity in phenotypic adaptation to rapid environmental change. Philos. Trans. R. Soc. B 2019. [Google Scholar] [CrossRef]
- Román-Palacios, C.; Scholl, J.P.; Wiens, J.J. Evolution of diet across the animal tree of life. Evol. Lett. 2019, 3, 339–347. [Google Scholar] [CrossRef]
- McHenry, H.M. Body size and proportions in early hominids. Am. J. Phys. Anthropol. 1992, 87, 407–431. [Google Scholar] [CrossRef]
- Steudel-Numbers, K.L.; Tilkens, M.J. The effect of lower limb length on the energetic cost of locomotion: Implications for fossil hominins. J. Hum. Evol. 2004, 47, 95–109. [Google Scholar] [CrossRef] [PubMed]
- Pontzer, H.; Brown, M.H.; Raichlen, D.A.; Dunsworth, H.; Hare, B.; Walker, K.; Luke, A.; Dugas, L.R.; Durazo-Arvizu, R.; Schoeller, D. Metabolic acceleration and the evolution of human brain size and life history. Nature 2016, 533, 390. [Google Scholar] [CrossRef] [PubMed]
- Lacruz, R.; Stringer, C.; Kimble, W.; Wood, B.; Harvati, K.; O’Higgins, P.; Arsuaga, J.-L. The evolutionary history of the human face. Nat. Ecol. Evol. 2019, 3, 726–736. [Google Scholar] [CrossRef]
- Smith, F.H. The fate of the neandertals. J. Anthropol. Res. 2013, 69, 167–200. [Google Scholar] [CrossRef]
- Bocherens, H. Neanderthal dietary habits: Review of the isotopic evidence. In Evolution of Hominin Diets: Integrating Approaches to the Study Paleolithic Subsistence; Hublin, J.J., Richards, M.P., Eds.; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2009; pp. 241–250. [Google Scholar] [CrossRef]
- Churchill, S.E. Thin on the Ground: Neandertal Biology, Archeology and Ecology; John Wiley & Sons: Hoboken, NJ, USA, 2014. [Google Scholar]
- Gaudzinski, S. Monospecific or species-dominated faunal assemblages during the Middle Paleolithic in Europe. In Transitions before the Transition: Evolution and Stability in the Middle Paleolithic and Middle Stone Age; Hovers, E., Kuhn, S., Eds.; Springer: New York, NY, USA, 2006; pp. 137–147. [Google Scholar]
- Hockett, B.; Haws, J.A. Nutritional ecology and the human demography of Neandertal extinction. Quat. Int. 2005, 137, 21–34. [Google Scholar] [CrossRef]
- O’Connell, J.F. How did modern humans displace Neanderthals? Insights from hunter-gatherer ethnography and archaeology. In When Neanderthals and Modern Humans Met; Conard, N.J., Ed.; Kerns Verlag: Tübingen, Germany, 2006; pp. 43–64. [Google Scholar]
- Richards, M.P.; Trinkaus, E. Isotopic evidence for the diets of European Neanderthals and early modern humans. Proc. Natl. Acad. Sci. USA 2009, 106, 16034–16039. [Google Scholar] [CrossRef]
- Salazar-Garcia, D.C.; Power, R.C.; Serra, A.S.; Villaverde, V.; Walker, M.J.; Henry, A.G. Neanderthal diets in central and southeastern Mediterranean Iberia. Quat. Int. 2013, 318, 3–18. [Google Scholar] [CrossRef]
- Villa, P.; Roebroeks, W. Neandertal Demise: An Archaeological Analysis of the Modern Human Superiority Complex. PLoS ONE 2014, 9. [Google Scholar] [CrossRef]
- Wissing, C.; Rougier, H.; Crevecoeur, I.; Germonpré, M.; Naito, Y.I.; Semal, P.; Bocherens, H. Isotopic evidence for dietary ecology of Late Neandertals in North-Western Europe. Quat. Int. 2015, 10, 327–345. [Google Scholar]
- Lev, E.; Kislev, M.E.; Bar-Yosef, O. Mousterian vegetal food in Kebara Cave, Mt. Carmel. JAS 2005, 32, 475–484. [Google Scholar] [CrossRef]
- Madella, M.; Jones, M.K.; Goldberg, P.; Goren, Y.; Hovers, E. The exploitation of plant resources by Neanderthals in Amud Cave (Israel): The evidence from Phytolith studies. JAS 2002, 29, 703–719. [Google Scholar] [CrossRef]
- Fiorenza, L.; Benazzi, S.; Henry, A.G.; Salazar-Garcia, D.C.; Blasco, R.; Picin, A.; Wroe, S.; Kullmer, O. To Meat or Not to Meat? New Perspectives on Neanderthal Ecology. Am. J. Phys. Anthropol. 2015, 156, 43–71. [Google Scholar] [CrossRef]
- Gaudzinski-Windheuser, S.; Niven, L. Hominin Subsistence Patterns During the Middle and Late Paleolithic in Northwestern Europe. In Evolution of Hominin Diets: Integrating Approaches to the Study of Palaeolithic Subsistence; Hublin, J.J., Richards, M.P., Eds.; Springer: Berlin/Heidelberg, Germany, 2009; pp. 99–111. [Google Scholar] [CrossRef]
- Panera, J.; Rubio-Jara, S.; Yravedra, J.; Blain, H.-A.; Sese, C.; Perez-Gonzalez, A. Manzanares Valley (Madrid, Spain): A good country for Proboscideans and Neanderthals. Quat. Int. 2014, 326, 329–343. [Google Scholar] [CrossRef]
- Bocherens, H. Diet and Ecology of Neanderthals: Implications from C and N Isotopes. In Neanderthal Lifeways, Subsistence and Technology: One Hundred Fifty Years of Neanderthal Study; Springer: Dordrecht, The Netherlands, 2011; pp. 73–85. [Google Scholar] [CrossRef]
- Bocherens, H.; Drucker, D.G.; Billiou, D.; Patou-Mathis, M.; Vandermeersch, B. Isotopic evidence for diet and subsistence pattern of the Saint-Césaire I Neanderthal: Review and use of a multi-source mixing model. J. Hum. Evol. 2005, 49, 71–87. [Google Scholar] [CrossRef]
- Bocherens, H.; Drucker, D.G. Dietary competition between Neanderthals and modern humans: Insights from stable isotopes. In When Neanderthals and Modern Humans Met; Conard, N.J., Ed.; Kerns Verlag: Tübingen, Germany, 2006; pp. 129–143. [Google Scholar]
- Naito, Y.I.; Chikaraishi, Y.; Drucker, D.G.; Ohkouchi, N.; Semal, P.; Wißing, C.; Bocherens, H. Ecological niche of Neanderthals from Spy Cave revealed by nitrogen isotopes of individual amino acids in collagen. J. Hum. Evol. 2016, 93, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Blasco, R.; Fernández Peris, J. A uniquely broad spectrum diet during the Middle Pleistocene at Bolomor Cave (Valencia, Spain). Quat. Int. 2012, 252, 16–31. [Google Scholar] [CrossRef]
- Hardy, K.; Buckley, S.; Collins, M.J.; Estalrrich, A.; Brothwell, D.; Copeland, L.; García-Tabernero, A.; García-Vargas, S.; de la Rasilla, M.; Lalueza-Fox, C.; et al. Neanderthal medics? Evidence for food, cooking, and medicinal plants entrapped in dental calculus. Naturwissenschaften 2012, 99, 617–626. [Google Scholar] [CrossRef]
- Henry, A.G.; Brooks, A.S.; Piperno, D.R. Plant foods and the dietary ecology of Neanderthals and early modern humans. J. Hum. Evol. 2014, 69, 44–54. [Google Scholar] [CrossRef] [PubMed]
- Weyrich, L.S.; Duchene, S.; Soubrier, J.; Arriola, L.; Llamas, B.; Breen, J.; Morris, A.G.; Alt, K.W.; Caramelli, D.; Dresely, V. Neanderthal behaviour, diet, and disease inferred from ancient DNA in dental calculus. Nature 2017, 544, 357–361. [Google Scholar] [CrossRef]
- Sistiaga, A.; Mallol, C.; Galvan, B.; Everett Summons, R. The Neanderthal Meal: A New Perspective Using Faecal Biomarkers. PLoS ONE 2014, 9, e101045. [Google Scholar] [CrossRef]
- Higham, T.; Douka, K.; Wood, R.; Ramsey, C.B.; Brock, F.; Basell, L.; Camps, M.; Arrizabalaga, A.; Baena, J.; Barroso-Ruiz, C.; et al. The timing and spatiotemporal patterning of Neanderthal disappearance. Nature 2014, 512, 306–309. [Google Scholar] [CrossRef]
- Froehle, A.W.; Churchill, S.E. Energetic Competition Between Neandertals and Anatomically Modern Humans. Paleo Anthropol. 2009, 96, 116. [Google Scholar]
- Ben-Dor, M.; Gopher, A.; Barkai, R. Neandertals’ large lower thorax may represent adaptation to high protein diet. Am. J. Phys. Anthropol. 2016, 160, 367–378. [Google Scholar] [CrossRef]
- Lindstedt, S.L.; Boyce, M.S. Seasonality, fasting, endurance, and body size in mammals. Am. Nat. 1985, 125, 873–878. [Google Scholar] [CrossRef]
- Geist, V. Neanderthal the hunter. Nat. Hist. 1981, 90, 26. [Google Scholar]
- Stewart, J.R. Neanderthal extinction as part of the faunal change in Europe during Oxygen Isotope Stage 3. Acta Zool. Cracoviensia Ser. A Vertebr. 2007, 50A, 93–120. [Google Scholar] [CrossRef]
- Larbey, C.; Mentzer, S.M.; Ligouis, B.; Wurz, S.; Jones, M.K. Cooked starchy food in hearths ca. 120 kya and 65 kya (MIS 5e and MIS 4) from Klasies River Cave, South Africa. J. Hum. Evol. 2019, 131, 210–227. [Google Scholar] [CrossRef] [PubMed]
- Aranguren, B.; Becattini, R.; Lippi, M.M.; Revedin, A. Grinding flour in Upper Palaeolithic Europe (25000 years bp). Antiquity 2007, 81, 845–855. [Google Scholar] [CrossRef]
- Bar-Yosef, O. Upper Paleolithic hunter-gatherers in Western Asia. In The Oxford Handbook of the Archaeology and Anthropology of Hunter-Gatherers; Cummings, V., Jordan, P., Zvelebil, M., Eds.; Oxford University Press: Oxford, UK, 2014; pp. 252–278. [Google Scholar]
- Kuhn, S.L.; Stiner, M.C. The antiquity of hunter-gatherers. In Hunter–Gatherers: Interdisciplinary Perspectives; Panter-Brick, C., Layton, R., Rowley-Conwy, P., Eds.; Cambridge University Press: Cambridge, UK, 2001; pp. 99–142. [Google Scholar]
- Revedin, A.; Aranguren, B.; Becattini, R.; Longo, L.; Marconi, E.; Lippi, M.M.; Skakun, N.; Sinitsyn, A.; Spiridonova, E.; Svoboda, J. Thirty thousand-year-old evidence of plant food processing. Proc. Natl. Acad. Sci. USA 2010, 107, 18815–18819. [Google Scholar] [CrossRef]
- Soffer, O. Storage, sedentism and the Eurasian Palaeolithic record. Antiquity 1989, 63, 719. [Google Scholar] [CrossRef]
- Germonpré, M.; Sablin, M.V.; Lázničková-Galetová, M.; Després, V.; Stevens, R.E.; Stiller, M.; Hofreiter, M. Palaeolithic dogs and Pleistocene wolves revisited: A reply to Morey (2014). JAS 2015, 54, 210–216. [Google Scholar] [CrossRef]
- Macdonald, M.L.; Rogers, Q.R.; Morris, J.G. Nutrition of the domestic cat, a mammalian carnivore. Annu. Rev. Nutr. 1984, 4, 521–562. [Google Scholar] [CrossRef]
- Lahtinen, M.; Clinnick, D.; Mannermaa, K.; Salonen, J.S.; Viranta, S. Excess protein enabled dog domestication during severe Ice Age winters. Sci. Rep. 2021, 11, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Lupo, K.D. When and where do dogs improve hunting productivity? The empirical record and some implications for early Upper Paleolithic prey acquisition. J. Anthropol. Archaeol. 2017, 47, 139–151. [Google Scholar] [CrossRef]
- Yeomans, L.; Martin, L.; Richter, T. Close companions: Early evidence for dogs in northeast Jordan and the potential impact of new hunting methods. J. Anthropol. Archaeol. 2019, 53, 161–173. [Google Scholar] [CrossRef]
- Munro, N.; BarOz, G.; Dayan, T.; Broughton, J.; Ugan, A.; Davis, S.; Hayden, B.; Jones, E.; Lyman, R.L.; Valla, F. Zooarchaeological measures of hunting pressure and occupation intensity in the Natufian: Implications for agricultural origins. Curr. Anthropol. 2004, 45, S5–S34. [Google Scholar] [CrossRef]
- Munro, N.D.; Bar-Oz, G.; Meier, J.S.; Sapir-Hen, L.; Stiner, M.C.; Yeshurun, R. The emergence of animal management in the Southern Levant. Sci. Rep. 2018, 8, 1–11. [Google Scholar] [CrossRef]
- Davis, S.J. Why domesticate food animals? Some zoo-archaeological evidence from the Levant. JAS 2005, 32, 1408–1416. [Google Scholar] [CrossRef]
- Munro, N.D.; Bar-Oz, G. Gazelle bone fat processing in the Levantine Epipalaeolithic. JAS 2005, 32, 223–239. [Google Scholar] [CrossRef]
- Outram, A.K. Identifying dietary stress in marginal environments: Bone fats, optimal foraging theory and the seasonal round. In Colonisation, Migration and Marginal Areas: A Zooarchaeological Approach; Miondini, M., Munoz, S., Wickler, S., Eds.; Oxford Books: Oxford, UK, 2004; pp. 74–85. [Google Scholar]
- Bar-Yosef, O. Multiple origins of agriculture in Eurasia and Africa. In On Human Nature: Biology, Psychology, Ethics, Politics, and Religion; Tibayrenc, M., Francisco, J.A., Eds.; Academic Press: Cambridge, MA, USA, 2017; pp. 297–331. [Google Scholar]
- Price, T.D.; Bar-Yosef, O. The origins of agriculture: New data, new ideas: An introduction to supplement 4. Curr. Anthropol. 2011, 52, S163–S174. [Google Scholar] [CrossRef]
- Vigne, J.-D. The origins of animal domestication and husbandry: A major change in the history of humanity and the biosphere. C. R. Biol. 2011, 334, 171–181. [Google Scholar] [CrossRef] [PubMed]
| Species | Weight Kg | Ledger–Raw Data (E%) | African Adjusted (E%) | Non-African Adjusted (E%) |
|---|---|---|---|---|
| Females | ||||
| Hippo | 1277 | 67% | 71% | 76% |
| Wildebeest K | 192 | 62% | 67% | 71% |
| Waterbuck | 181 | 45% | 50% | 55% |
| Oryx | 161.5 | 57% | 62% | 67% |
| Wilderbeest S | 160.3 | 52% | 57% | 62% |
| Kongoni | 126.2 | 49% | 54% | 59% |
| Topi | 103.9 | 29% | 33% | 38% |
| Kobe | 62.1 | 45% | 49% | 55% |
| Warthog | 60.2 | 28% | 32% | 37% |
| Impala | 42 | 30% | 34% | 39% |
| Grant’s Gazelle | 41.3 | 48% | 53% | 59% |
| Thomson’s Gazelle | 18.4 | 34% | 38% | 43% |
| Thomson’s Gazelle (S) | 16.9 | 47% | 52% | 57% |
| Males | ||||
| Hippo | 1489 | 56% | 61% | 66% |
| Buffalo | 753 | 54% | 58% | 64% |
| Eland | 508.1 | 50% | 54% | 60% |
| Wildebeest K | 243.3 | 58% | 62% | 67% |
| Waterbuck | 237.7 | 20% | 23% | 27% |
| Wildebeest S | 203 | 64% | 68% | 72% |
| Oryx | 176.4 | 36% | 41% | 46% |
| Kongoni | 142.5 | 31% | 36% | 41% |
| Topi | 130.8 | 32% | 37% | 42% |
| Kobe | 96.7 | 34% | 39% | 44% |
| Lesser Kudu | 92.1 | 40% | 44% | 50% |
| Warthog | 87.8 | 26% | 30% | 35% |
| Grant’s Gazelle | 60.1 | 36% | 40% | 45% |
| Impala | 56.7 | 29% | 33% | 38% |
| Gerenuk | 31.2 | 26% | 30% | 35% |
| Thomson’s Gazelle | 25.3 | 31% | 35% | 40% |
| Thomson’s Gazelle (S) | 20.3 | 30% | 34% | 39% |
| Average | 227 | 41% | 46% | 52% |
| Evidence Name | Evidence Description |
|---|---|
| Bioenergetics | Large prey provides higher energetic returns per hour than smaller prey. The need to replace large prey with smaller prey is energetically costly. |
| Higher fat reserves | Humans have relatively high-fat reserves. Large prey is less abundant than smaller prey. Fat reserves may have evolved to allow extended fasting of several weeks, thereby bridging a variable encountering rate with large prey. Humans have adapted to efficiently synthesize ketones to replace glucose as an energy source for the brain during fasting. |
| Stomach acidity | Stomach acidity evolved, among other things, to guard against pathogens. Similar acidity level to scavengers in humans, higher than in carnivores, can be interpreted as an adaptation to a large prey’s protracted consumption over days and weeks, whereby humans are acting as scavengers of their prey. |
| Targeting fat | Humans targeted fat by hunting large and prime-adult animals, both of which have a higher fat level, by bringing fatty parts to central places and exploiting bone fats at a great energetic expense. The recognition of targeting fat as a driver of human behavior supports the importance of large, higher fat bearing animals to humans’ survival. |
| Stable isotopes | Researchers interpreted higher levels of nitrogen isotope 15 in humans than in carnivores as testifying to the higher consumption of large prey than other carnivores. |
| Paleontology | A decline in the guild of large prey carnivores 1.5 Mya was interpreted as resulting from humans’ entrance to the guild. Moreover, the extinction of large prey throughout the Pleistocene is interpreted by some researchers as anthropogenic, testifying to humans’ preference for large prey. |
| Zoological analogy | Large social carnivores get most of their energy from large prey. |
| Ethnography | Interpreting ethnographic and Upper Paleolithic technologies as an adaptation to smaller prey acquisition means humans were less adapted to smaller prey acquisition in earlier periods. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ben-Dor, M.; Barkai, R. Prey Size Decline as a Unifying Ecological Selecting Agent in Pleistocene Human Evolution. Quaternary 2021, 4, 7. https://doi.org/10.3390/quat4010007
Ben-Dor M, Barkai R. Prey Size Decline as a Unifying Ecological Selecting Agent in Pleistocene Human Evolution. Quaternary. 2021; 4(1):7. https://doi.org/10.3390/quat4010007
Chicago/Turabian StyleBen-Dor, Miki, and Ran Barkai. 2021. "Prey Size Decline as a Unifying Ecological Selecting Agent in Pleistocene Human Evolution" Quaternary 4, no. 1: 7. https://doi.org/10.3390/quat4010007
APA StyleBen-Dor, M., & Barkai, R. (2021). Prey Size Decline as a Unifying Ecological Selecting Agent in Pleistocene Human Evolution. Quaternary, 4(1), 7. https://doi.org/10.3390/quat4010007







