Bemotrizinol-Loaded Lipid Nanoparticles for the Development of Sunscreen Emulsions
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Nanostructured Lipid Carriers (NLC)
2.3. Characterization of Unloaded and Bemotrizinol-Loaded NLC
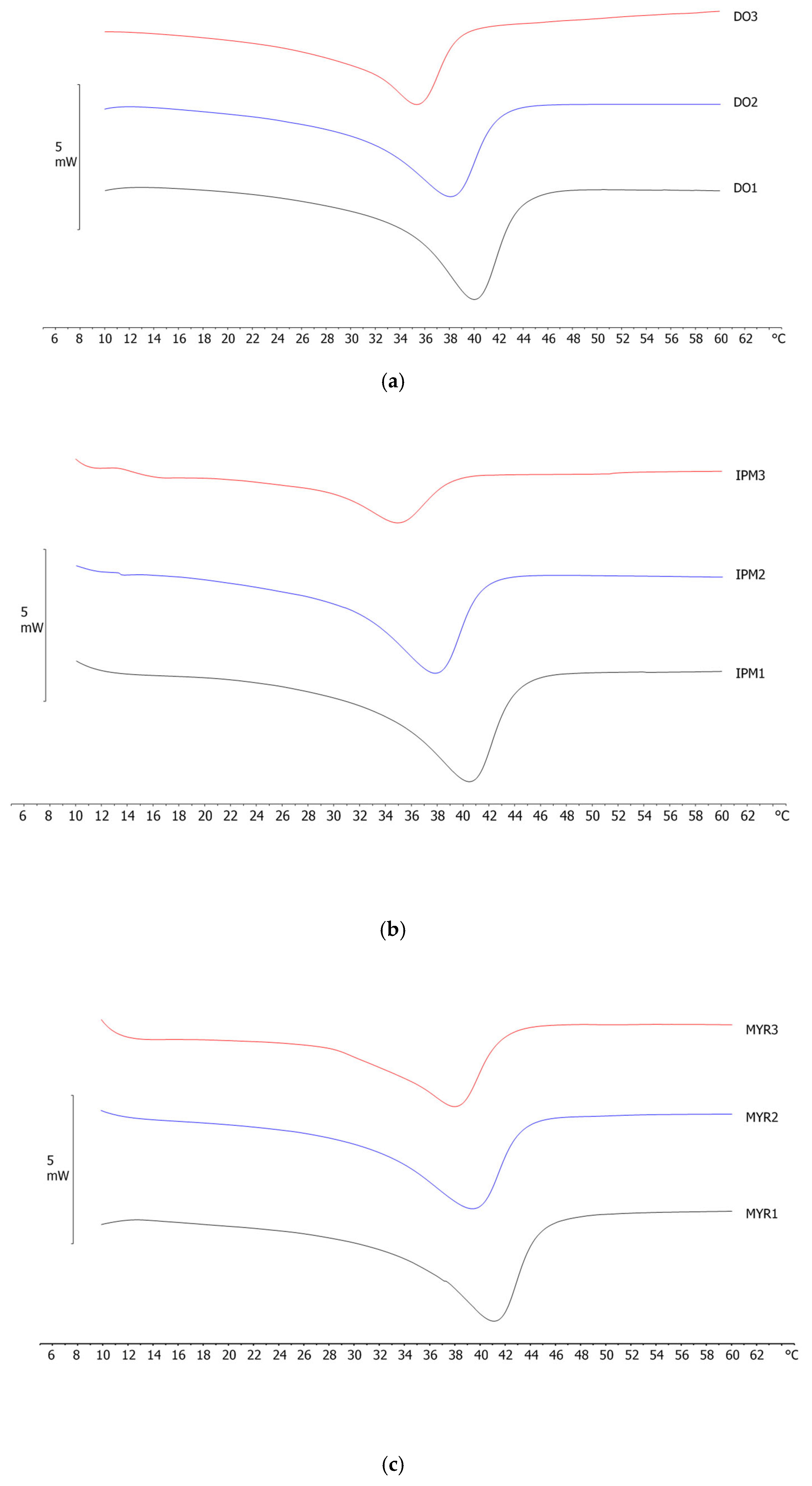
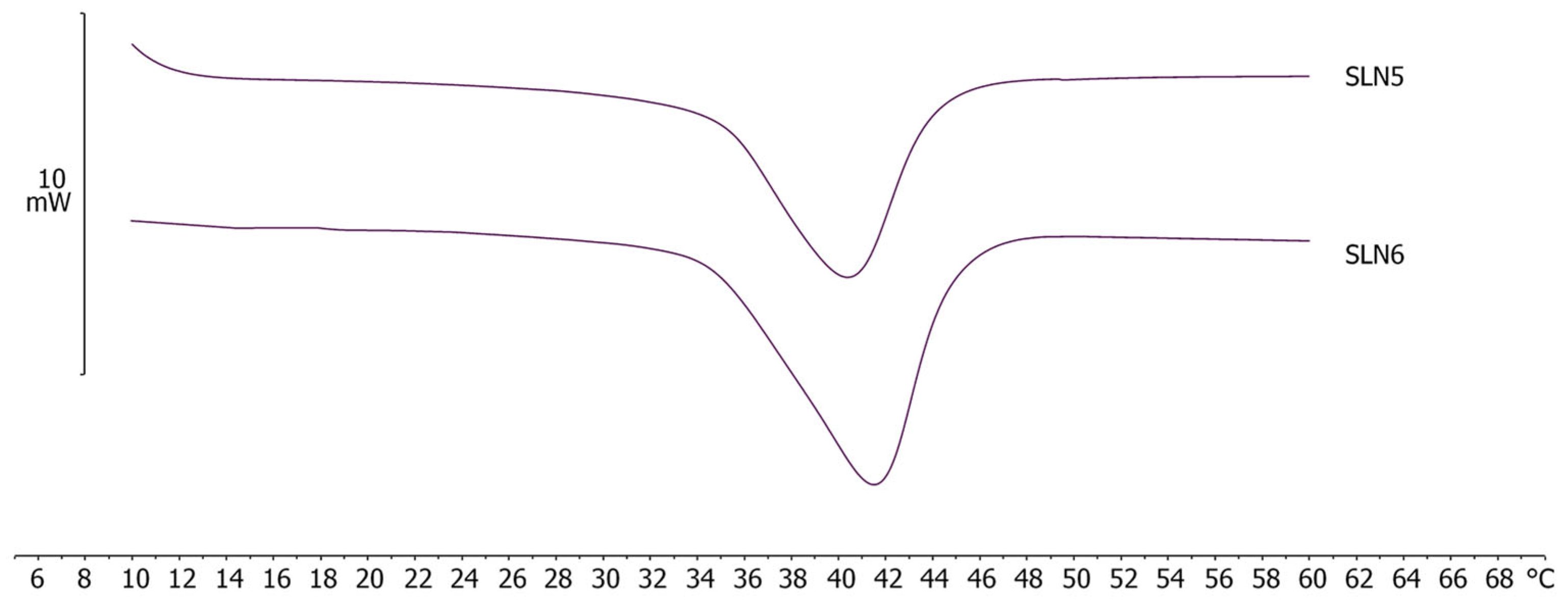
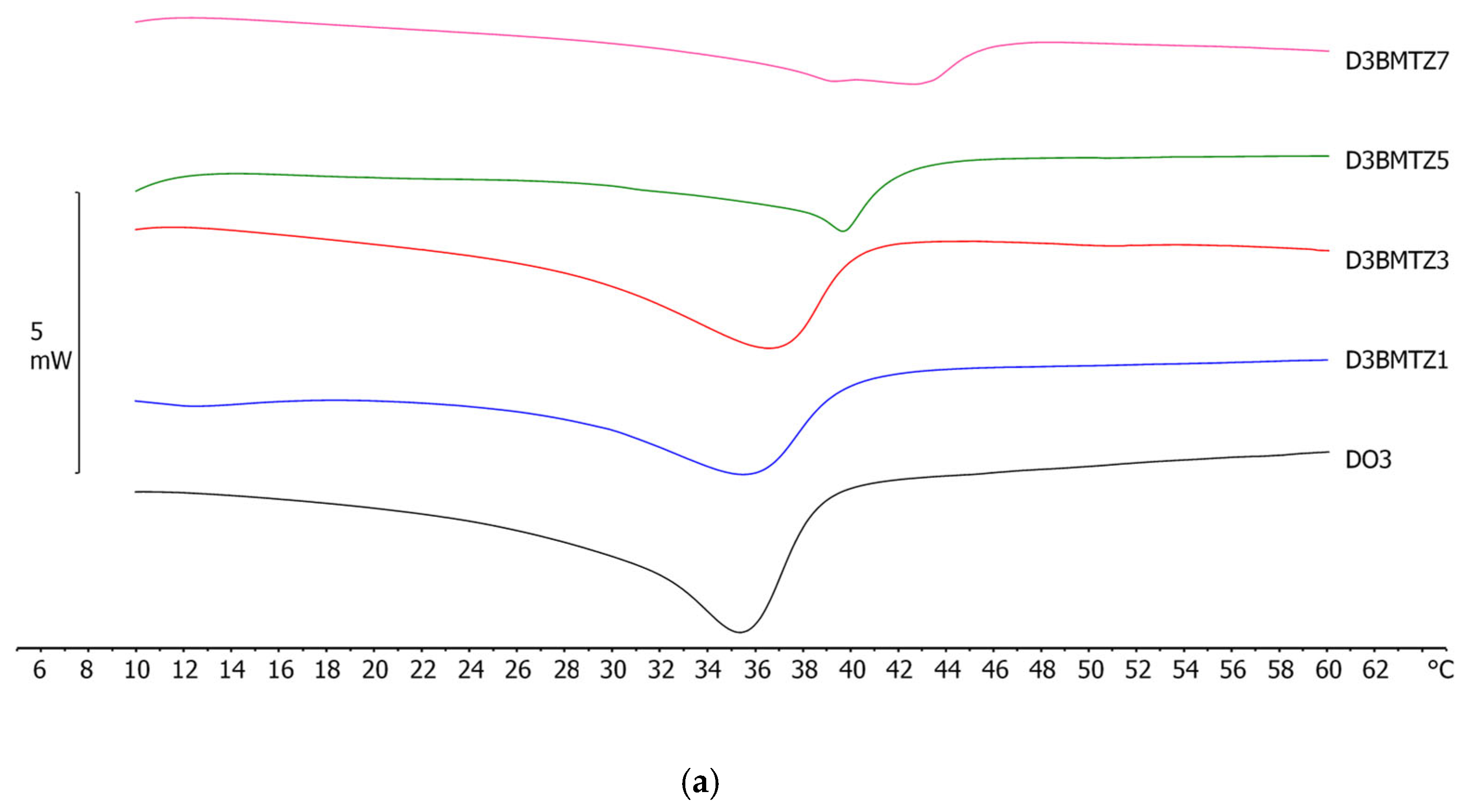
2.4. Differential Scanning Calorimetry (DSC) Analyses
2.5. Stability Studies on Nanostructured Lipid Carriers (NLC)
2.6. Preparation of O/W Emulsions
2.7. Stability Studies on O/W Emulsions
2.8. Spreadability
2.9. Occlusive Properties
2.10. Viscosity
2.11. In Vitro Release of Bemotrizinol
2.12. In Vitro Skin Permeation Experiments
2.13. In Vitro Sun Protection Factor (SPF)
2.14. Statistical Analysis
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Lionetti, N.; Rigano, L. The New Sunscreens among Formulation Strategy, Stability Issues, Changing Norms, Safety and Efficacy Evaluations. Cosmetics 2017, 4, 15. [Google Scholar] [CrossRef]
- Sander, M.; Sander, M.; Burbidge, T.; Beecker, J. The Efficacy and Safety of Sunscreen Use for the Prevention of Skin Cancer. Can. Med. Assoc. J. 2020, 192, E1802–E1808. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Colantonio, S.; Dawson, A.; Lin, X.; Beecker, J. Sunscreen Application, Safety, and Sun Protection: The Evidence. J. Cutan. Med. Surg. 2019, 23, 357–369. [Google Scholar] [CrossRef]
- Montenegro, L.; Turnaturi, R.; Parenti, C.; Pasquinucci, L. In Vitro Evaluation of Sunscreen Safety: Effects of the Vehicle and Repeated Applications on Skin Permeation from Topical Formulations. Pharmaceutics 2018, 10, 27. [Google Scholar] [CrossRef] [PubMed]
- Ouchene, L.; Litvinov, I.V.; Netchiporouk, E. Systemic Absorption of Common Organic Sunscreen Ingredients Raises Possible Safety Concerns for Patients. J. Cutan. Med. Surg. 2019, 23, 449–450. [Google Scholar] [CrossRef]
- Oral, D.; Yirun, A.; Erkekoglu, P. Safety Concerns of Organic Ultraviolet Filters: Special Focus on Endocrine-Disrupting Properties. J. Environ. Pathol. Toxicol. Oncol. 2020, 39, 201–212. [Google Scholar] [CrossRef]
- Nesseem, D. Formulation of Sunscreens with Enhancement Sun Protection Factor Response Based on Solid Lipid Nanoparticles. Int. J. Cosmet. Sci. 2011, 33, 70–79. [Google Scholar] [CrossRef]
- Nikolić, S.; Keck, C.M.; Anselmi, C.; Müller, R.H. Skin Photoprotection Improvement: Synergistic Interaction between Lipid Nanoparticles and Organic UV Filters. Int. J. Pharm. 2011, 414, 276–284. [Google Scholar] [CrossRef]
- Khater, D.; Nsairat, H.; Odeh, F.; Saleh, M.; Jaber, A.; Alshaer, W.; Al Bawab, A.; Mubarak, M.S. Design, Preparation, and Characterization of Effective Dermal and Transdermal Lipid Nanoparticles: A Review. Cosmetics 2021, 8, 39. [Google Scholar] [CrossRef]
- Chavda, V.P.; Acharya, D.; Hala, V.; Daware, S.; Vora, L.K. Sunscreens: A Comprehensive Review with the Application of Nanotechnology. J. Drug Deliv. Sci. Technol. 2023, 86, 104720. [Google Scholar] [CrossRef]
- de Araújo, M.M.; Schneid, A.C.; Oliveira, M.S.; Mussi, S.V.; de Freitas, M.N.; Carvalho, F.C.; Bernes Junior, E.A.; Faro, R.; Azevedo, H. NLC-Based Sunscreen Formulations with Optimized Proportion of Encapsulated and Free Filters Exhibit Enhanced UVA and UVB Photoprotection. Pharmaceutics 2024, 16, 427. [Google Scholar] [CrossRef]
- Sharma, B.; Chauhan, I.; Tiwari, R.K. Development of NLC-Based Sunscreen Gel of Green Tea Extract and Its In Vitro Characterization. Curr. Bioact. Compd. 2024, 20, 31–42. [Google Scholar] [CrossRef]
- Paliwal, R.; Paliwal, S.R.; Kenwat, R.; Das Kurmi, B.; Sahu, M.K. Solid Lipid Nanoparticles: A Review on Recent Perspectives and Patents. Expert. Opin. Ther. Pat. 2020, 30, 179–194. [Google Scholar] [CrossRef] [PubMed]
- Eroğlu, C.; Sinani, G.; Ulker, Z. Current State of Lipid Nanoparticles (SLN and NLC) for Skin Applications. Curr. Pharm. Des. 2023, 29, 1632–1644. [Google Scholar] [CrossRef]
- Queiroz, M.d.C.V.; Muehlmann, L.A. Characteristics and Preparation of Solid Lipid Nanoparticles and Nanostructured Lipid Carriers. J. Nanotheranostics 2024, 5, 188–211. [Google Scholar] [CrossRef]
- Pardeike, J.; Hommoss, A.; Müller, R.H. Lipid Nanoparticles (SLN, NLC) in Cosmetic and Pharmaceutical Dermal Products. Int. J. Pharm. 2009, 366, 170–184. [Google Scholar] [CrossRef]
- Kakadia, P.; Conway, B. Lipid Nanoparticles for Dermal Drug Delivery. Curr. Pharm. Des. 2015, 21, 2823–2829. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.-H.; Jeon, Y.-E.; Kang, S.; Lee, J.-Y.; Lee, K.W.; Kim, K.-T.; Kim, D.-D. Lipid Nanoparticles for Enhancing the Physicochemical Stability and Topical Skin Delivery of Orobol. Pharmaceutics 2020, 12, 845. [Google Scholar] [CrossRef]
- Tran, P.; Lee, S.-E.; Kim, D.-H.; Pyo, Y.-C.; Park, J.-S. Recent Advances of Nanotechnology for the Delivery of Anticancer Drugs for Breast Cancer Treatment. J. Pharm. Investig. 2020, 50, 261–270. [Google Scholar] [CrossRef]
- Shirodkar, R.K.; Kumar, L.; Mutalik, S.; Lewis, S. Solid Lipid Nanoparticles and Nanostructured Lipid Carriers: Emerging Lipid Based Drug Delivery Systems. Pharm. Chem. J. 2019, 53, 440–453. [Google Scholar] [CrossRef]
- Chatelain, E.; Gabard, B. Photostabilization of Butyl Methoxydibenzoylmethane (Avobenzone) and Ethylhexyl Methoxycinnamate by Bis-Ethylhexyloxyphenol Methoxyphenyl Triazine (Tinosorb S), a New UV Broadband Filter. Photochem. Photobiol. 2001, 74, 401. [Google Scholar] [CrossRef]
- Teixeira Gomes, J.V.; Cherem Peixoto da Silva, A.; Lamim Bello, M.; Rangel Rodrigues, C.; Aloise Maneira Corrêa Santos, B. Molecular Modeling as a Design Tool for Sunscreen Candidates: A Case Study of Bemotrizinol. J. Mol. Model. 2019, 25, 362. [Google Scholar] [CrossRef]
- Medeiros, T.S.; Moreira, L.M.C.C.; Oliveira, T.M.T.; Melo, D.F.; Azevedo, E.P.; Gadelha, A.E.G.; Fook, M.V.L.; Oshiro-Júnior, J.A.; Damasceno, B.P.G.L. Bemotrizinol-Loaded Carnauba Wax-Based Nanostructured Lipid Carriers for Sunscreen: Optimization, Characterization, and In Vitro Evaluation. AAPS PharmSciTech 2020, 21, 288. [Google Scholar] [CrossRef]
- Wissing, S.A.; Müller, R.H. The Influence of the Crystallinity of Lipid Nanoparticles on Their Occlusive Properties. Int. J. Pharm. 2002, 242, 377–379. [Google Scholar] [CrossRef] [PubMed]
- Souto, E.B.; Almeida, A.J.; Müller, R.H. Lipid Nanoparticles (SLN®, NLC®) for Cutaneous Drug Delivery:Structure, Protection and Skin Effects. J. Biomed. Nanotechnol. 2007, 3, 317–331. [Google Scholar] [CrossRef]
- Subramaniam, B.; Siddik, Z.H.; Nagoor, N.H. Optimization of Nanostructured Lipid Carriers: Understanding the Types, Designs, and Parameters in the Process of Formulations. J. Nanoparticle Res. 2020, 22, 141. [Google Scholar] [CrossRef]
- Anton, N.; Benoit, J.-P.; Saulnier, P. Design and Production of Nanoparticles Formulated from Nano-Emulsion Templates—A Review. J. Control. Release 2008, 128, 185–199. [Google Scholar] [CrossRef]
- Safta, D.A.; Bogdan, C.; Moldovan, M.-L. SLNs and NLCs for Skin Applications: Enhancing the Bioavailability of Natural Bioactives. Pharmaceutics 2024, 16, 1270. [Google Scholar] [CrossRef]
- Montenegro, L.; Santagati, L.M.; Sarpietro, M.G.; Castelli, F.; Panico, A.; Siciliano, E.A.; Lai, F.; Valenti, D.; Sinico, C. In Vitro Skin Permeation of Idebenone from Lipid Nanoparticles Containing Chemical Penetration Enhancers. Pharmaceutics 2021, 13, 1027. [Google Scholar] [CrossRef]
- Sarpietro, M.G.; Torrisi, C.; Pignatello, R.; Castelli, F.; Montenegro, L. Assessment of the Technological Properties of Idebenone and Tocopheryl Acetate Co-Loaded Lipid Nanoparticles. Appl. Sci. 2021, 11, 3553. [Google Scholar] [CrossRef]
- Ruktanonchai, U.; Limpakdee, S.; Meejoo, S.; Sakulkhu, U.; Bunyapraphatsara, N.; Junyaprasert, V.; Puttipipatkhachorn, S. The Effect of Cetyl Palmitate Crystallinity on Physical Properties of Gamma-Oryzanol Encapsulated in Solid Lipid Nanoparticles. Nanotechnology 2008, 19, 095701. [Google Scholar] [CrossRef]
- Montenegro, L.; Rapisarda, L.; Ministeri, C.; Puglisi, G. Effects of Lipids and Emulsifiers on the Physicochemical and Sensory Properties of Cosmetic Emulsions Containing Vitamin E. Cosmetics 2015, 2, 35–47. [Google Scholar] [CrossRef]
- Chaudhary, B.; Verma, S. Preparation and Evaluation of Novel in Situ Gels Containing Acyclovir for the Treatment of Oral Herpes Simplex Virus Infections. Sci. World J. 2014, 2014, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Elena, O.B.; Maria, N.A.; Michael, S.Z.; Natalia, B.D.; Alexander, I.B.; Ivan, I.K. Dermatologic Gels Spreadability Measuring Methods Comparative Study. Int. J. Appl. Pharm. 2022, 14, 164–168. [Google Scholar] [CrossRef]
- Wissing, S.; Lippacher, A.; Müller, R. Investigations on the Occlusive Properties of Solid Lipid Nanoparticles (SLN). J. Cosmet. Sci. 2001, 52, 313–324. [Google Scholar]
- Montenegro, L.; Parenti, C.; Turnaturi, R.; Pasquinucci, L. Resveratrol-Loaded Lipid Nanocarriers: Correlation between In Vitro Occlusion Factor and In Vivo Skin Hydrating Effect. Pharmaceutics 2017, 9, 58. [Google Scholar] [CrossRef] [PubMed]
- Fallica, F.; Leonardi, C.; Toscano, V.; Santonocito, D.; Leonardi, P.; Puglia, C. Assessment of Alcohol-Based Hand Sanitizers for Long-Term Use, Formulated with Addition of Natural Ingredients in Comparison to WHO Formulation 1. Pharmaceutics 2021, 13, 571. [Google Scholar] [CrossRef]
- Puglia, C.; Santonocito, D.; Bonaccorso, A.; Musumeci, T.; Ruozi, B.; Pignatello, R.; Carbone, C.; Parenti, C.; Chiechio, S. Lipid Nanoparticle Inclusion Prevents Capsaicin-Induced TRPV1 Defunctionalization. Pharmaceutics 2020, 12, 339. [Google Scholar] [CrossRef]
- Mansur, J.S.; Breder, M.N.R.; Mansur, M.C.A.; Azulay, R.D. Determinação Do Fator de Proteção Solar Por Espectrofotometria. An. Bras. Dermatol. 1986, 61, 121–1245. [Google Scholar]
- Ricci-Junior, E.; Dellamora Ortiz, G.M.; Pereira dos Santos, E.; de Carvalho Varjão Mota, A.; Antonio Ozzetti, R.; Luiz Vergnanini, A.; Santos-Oliveira, R.; Santos Silva, R.; Lira Ribeiro, V.; Maria Faria de Freitas, Z. In Vivo and in Vitro Evaluation of Octyl Methoxycinnamate Liposomes. Int. J. Nanomed. 2013, 8, 4689–4701. [Google Scholar] [CrossRef]
- Fiume, M.M.; Bergfeld, W.F.; Belsito, D.V.; Hill, R.A.; Klaassen, C.D.; Liebler, D.C.; Marks, J.G.; Shank, R.C.; Slaga, T.J.; Snyder, P.W.; et al. Amended Safety Assessment of Triglycerides as Used in Cosmetics. Int. J. Toxicol. 2022, 41, 22–68. [Google Scholar] [CrossRef]
- Fiume, M.M.; Heldreth, B.A.; Bergfeld, W.F.; Belsito, D.V.; Hill, R.A.; Klaassen, C.D.; Liebler, D.C.; Marks, J.G.; Shank, R.C.; Slaga, T.J.; et al. Safety Assessment of Alkyl Esters as Used in Cosmetics. Int. J. Toxicol. 2015, 34, 5S–69S. [Google Scholar] [CrossRef]
- Gonzalez Solveyra, E.; Szleifer, I. What Is the Role of Curvature on the Properties of Nanomaterials for Biomedical Applications? WIREs Nanomed. Nanobiotechnol. 2016, 8, 334–354. [Google Scholar] [CrossRef]
- Alajami, H.N.; Fouad, E.A.; Ashour, A.E.; Kumar, A.; Yassin, A.E.B. Celecoxib-Loaded Solid Lipid Nanoparticles for Colon Delivery: Formulation Optimization and In Vitro Assessment of Anti-Cancer Activity. Pharmaceutics 2022, 14, 131. [Google Scholar] [CrossRef] [PubMed]
- Müller, R.H.; Mäder, K.; Gohla, S. Solid Lipid Nanoparticles (SLN) for Controlled Drug Delivery—A Review of the State of the Art. Eur. J. Pharm. Biopharm. 2000, 50, 161–177. [Google Scholar] [CrossRef] [PubMed]
- Pardeshi, C.; Rajput, P.; Belgamwar, V.; Tekade, A.; Patil, G.; Chaudhary, K.; Sonje, A. Solid Lipid Based Nanocarriers: An Overview / Nanonosači Na Bazi Čvrstih Lipida: Pregled. Acta Pharm. 2012, 62, 433–472. [Google Scholar] [CrossRef]
- Jenning, V.; Thünemann, A.F.; Gohla, S.H. Characterisation of a Novel Solid Lipid Nanoparticle Carrier System Based on Binary Mixtures of Liquid and Solid Lipids. Int. J. Pharm. 2000, 199, 167–177. [Google Scholar] [CrossRef]
- Bunjes, H.; Westesen, K.; Koch, M.H.J. Crystallization Tendency and Polymorphic Transitions in Triglyceride Nanoparticles. Int. J. Pharm. 1996, 129, 159–173. [Google Scholar] [CrossRef]
- Samimi, S.; Maghsoudnia, N.; Eftekhari, R.B.; Dorkoosh, F. Characterization and Biology of Nanomaterials for Drug Delivery; Elsevier: Amsterdam, The Netherlands, 2019; ISBN 9780128140314. [Google Scholar]
- Lardy, F.; Vennat, B.; Pouget, M.P.; Pourrat, A. Functionalization of Hydrocolloids: Principal Component Analysis Applied to the Study of Correlations Between Parameters Describing the Consistency of Hydrogels. Drug Dev. Ind. Pharm. 2000, 26, 715–721. [Google Scholar] [CrossRef] [PubMed]
- Garg, A.; Aggarwal, D.; Garg, S.; Singla, A.K. Spreading of Semisolid Formulations: An Update. Pharm. Technol. 2002, 26, 84–105. [Google Scholar]
- Santonocito, D.; Puglia, C.; Montenegro, L. Effects of Lipid Phase Content on the Technological and Sensory Properties of O/W Emulsions Containing Bemotrizinol-Loaded Nanostructured Lipid Carriers. Cosmetics 2024, 11, 123. [Google Scholar] [CrossRef]
- Ahmad, J. Lipid Nanoparticles Based Cosmetics with Potential Application in Alleviating Skin Disorders. Cosmetics 2021, 8, 84. [Google Scholar] [CrossRef]
- Chu, C.C.; Hasan, Z.A.A.; Tan, C.P.; Nyam, K.L. In Vitro Safety Evaluation of Sunscreen Formulation from Nanostructured Lipid Carriers Using Human Cells and Skin Model. Toxicol. Vitr. 2022, 84, 105431. [Google Scholar] [CrossRef]
- Assali, M.; Zaid, A.-N. Features, Applications, and Sustainability of Lipid Nanoparticles in Cosmeceuticals. Saudi Pharm. J. 2022, 30, 53–65. [Google Scholar] [CrossRef]
- Souza, C.; Maia Campos, P.M.B.G. Development of a HPLC Method for Determination of Four UV Filters in Sunscreen and Its Application to Skin Penetration Studies. Biomed. Chromatogr. 2017, 31, e4029. [Google Scholar] [CrossRef] [PubMed]
- Sheu, M.-T.; Lin, C.-W.; Huang, M.-C.; Shen, C.-H.; Ho, H.-O. Correlation of in Vivo and in Vitro Measurements of Sun Protection Factor. J. Food Drug Anal. 2003, 11, 12. [Google Scholar] [CrossRef]
- Saraf, S.; Kaur, C. In Vitro Sun Protection Factor Determination of Herbal Oils Used in Cosmetics. Pharmacogn. Res. 2010, 2, 22. [Google Scholar] [CrossRef]
- Pissavini, M.; Tricaud, C.; Wiener, G.; Lauer, A.; Contier, M.; Kolbe, L.; Trullás Cabanas, C.; Boyer, F.; Meredith, E.; de Lapuente, J.; et al. Validation of a New in Vitro Sun Protection Factor Method to Include a Wide Range of Sunscreen Product Emulsion Types. Int. J. Cosmet. Sci. 2020, 42, 421–428. [Google Scholar] [CrossRef] [PubMed]
- Dimitrovska Cvetkovska, A.; Manfredini, S.; Ziosi, P.; Molesini, S.; Dissette, V.; Magri, I.; Scapoli, C.; Carrieri, A.; Durini, E.; Vertuani, S. Factors Affecting SPF in Vitro Measurement and Correlation with in Vivo Results. Int. J. Cosmet. Sci. 2017, 39, 310–319. [Google Scholar] [CrossRef]
- Jose, J.; Netto, G. Role of Solid Lipid Nanoparticles as Photoprotective Agents in Cosmetics. J. Cosmet. Dermatol. 2019, 18, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Ácsová, A.; Hojerová, J.; Janotková, L.; Bendová, H.; Jedličková, L.; Hamranová, V.; Martiniaková, S. The Real UVB Photoprotective Efficacy of Vegetable Oils: In Vitro and in Vivo Studies. Photochem. Photobiol. Sci. 2021, 20, 139–151. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.I.; Liu, S.; Brooks, G.J.; Lanctot, Y.; Gruber, J. V Reliable and Simple Spectrophotometric Determination of Sun Protection Factor: A Case Study Using Organic UV Filter-Based Sunscreen Products. J. Cosmet. Dermatol. 2018, 17, 518–522. [Google Scholar] [CrossRef] [PubMed]
- Hermund, D.B.; Torsteinsen, H.; Vega, J.; Figueroa, F.L.; Jacobsen, C. Screening for New Cosmeceuticals from Brown Algae Fucus Vesiculosus with Antioxidant and Photo-Protecting Properties. Mar. Drugs 2022, 20, 687. [Google Scholar] [CrossRef] [PubMed]

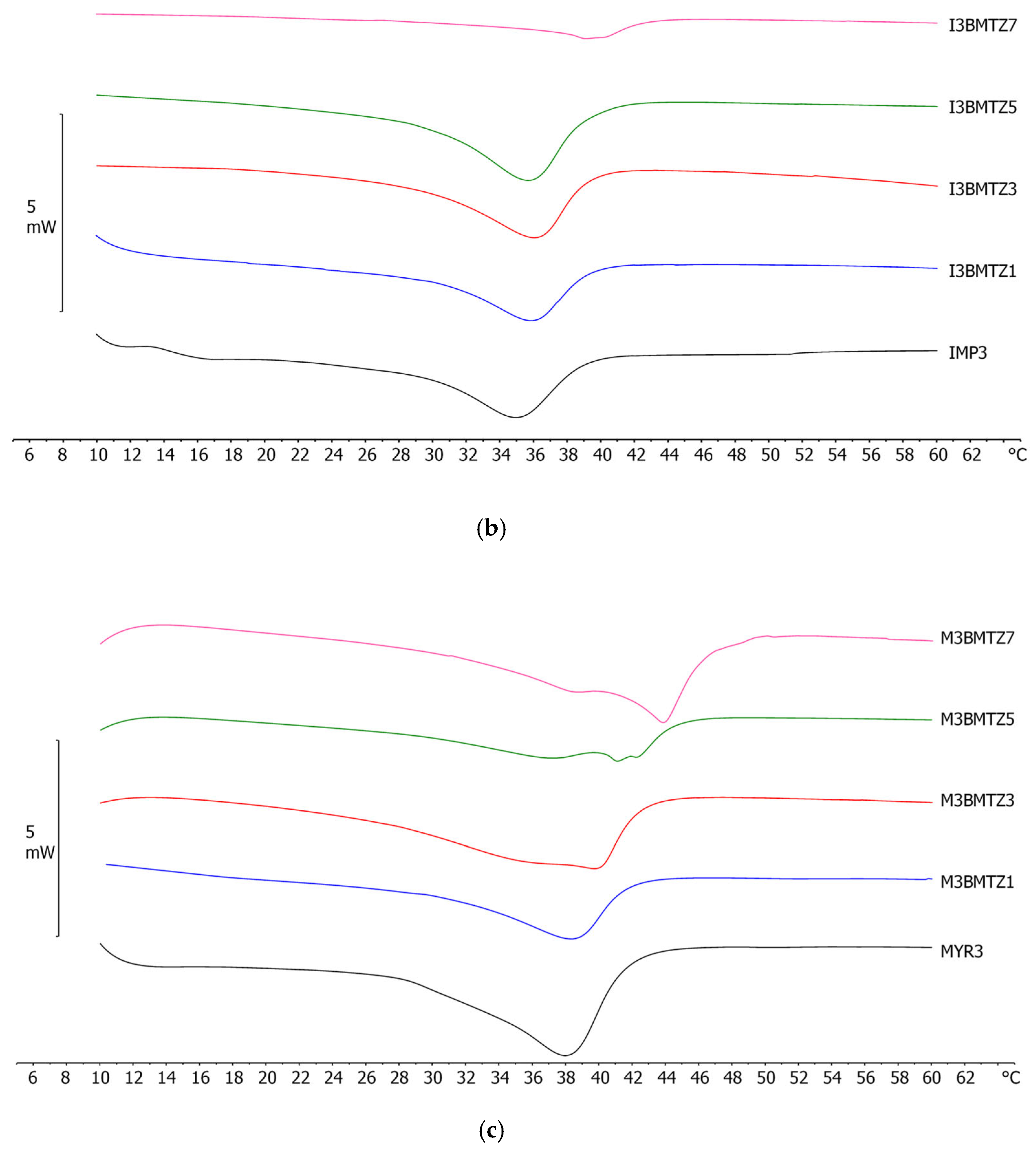
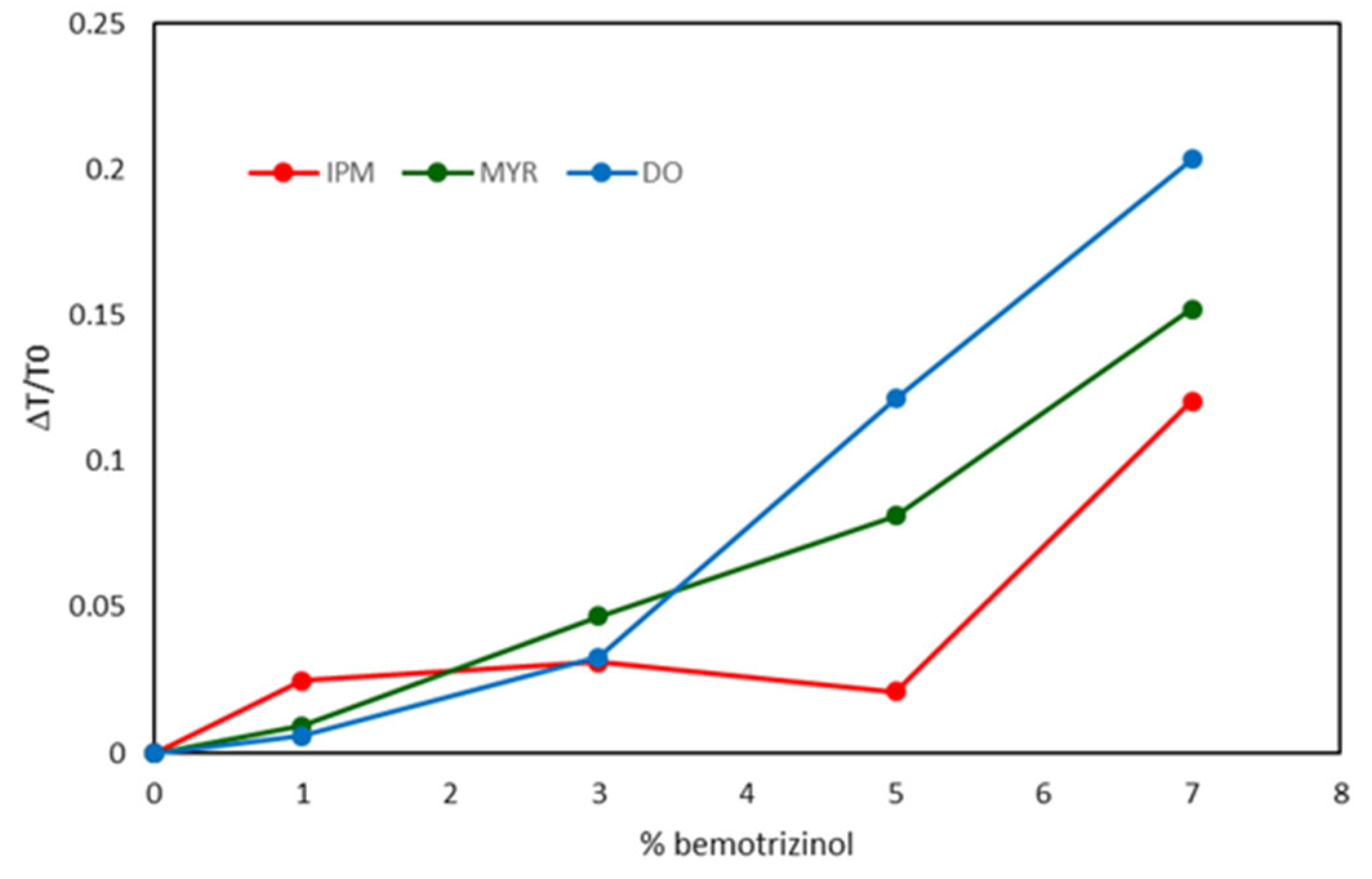
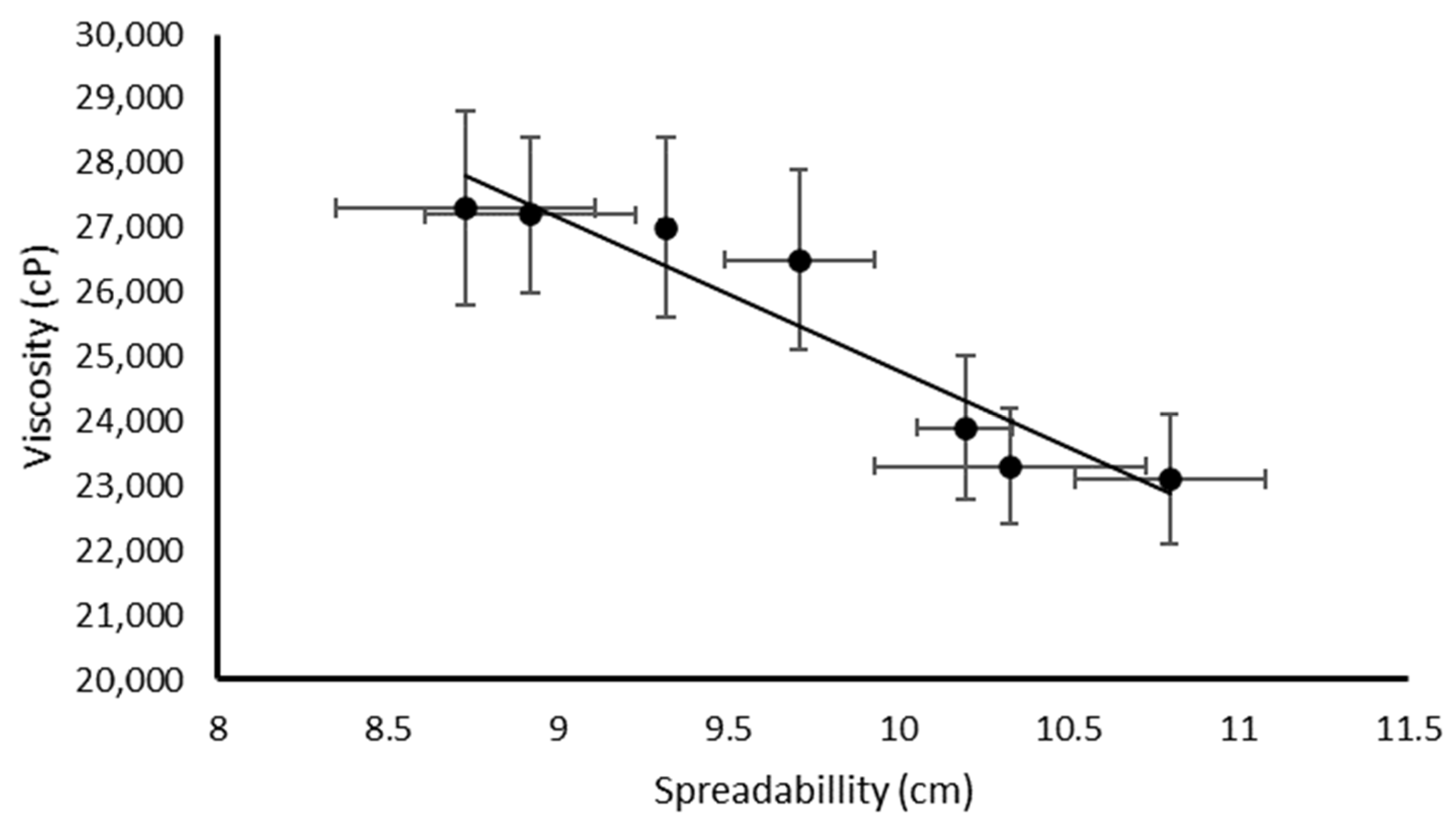
| NLC Code | Oleth-20 | GO a | CP b | IPM c | MYR d | DO e |
|---|---|---|---|---|---|---|
| IPM1 | 8.7 | 4.4 | 6.0 | 1.0 | --- | --- |
| IPM2 | 8.7 | 4.4 | 5.0 | 2.0 | --- | --- |
| IPM3 | 8.7 | 4.4 | 4.0 | 3.0 | --- | --- |
| MYR1 | 8.7 | 4.4 | 6.0 | --- | 1.0 | --- |
| MYR2 | 8.7 | 4.4 | 5.0 | --- | 2.0 | --- |
| MYR3 | 8.7 | 4.4 | 4.0 | --- | 3.0 | --- |
| DO1 | 8.7 | 4.4 | 6.0 | --- | --- | 1.0 |
| DO2 | 8.7 | 4.4 | 5.0 | --- | --- | 2.0 |
| DO3 | 8.7 | 4.4 | 4.0 | --- | --- | 3.0 |
| NLC Code | Oleth-20 | GO a | CP b | IPM c | MYR d | DO e | BMTZ f |
|---|---|---|---|---|---|---|---|
| IBMTZ1 | 8.7 | 4.4 | 4.0 | 3.0 | --- | --- | 1.0 |
| IBMTZ3 | 8.7 | 4.4 | 4.0 | 3.0 | --- | --- | 3.0 |
| IBMTZ5 | 8.7 | 4.4 | 4.0 | 3.0 | --- | --- | 5.0 |
| IBMTZ7 | 8.7 | 4.4 | 4.0 | 3.0 | --- | --- | 7.0 |
| MBMTZ1 | 8.7 | 4.4 | 4.0 | --- | 3.0 | --- | 1.0 |
| MBMTZ3 | 8.7 | 4.4 | 4.0 | --- | 3.0 | --- | 3.0 |
| MBMTZ5 | 8.7 | 4.4 | 4.0 | --- | 3.0 | --- | 5.0 |
| MBMTZ7 | 8.7 | 4.4 | 4.0 | --- | 3.0 | --- | 7.0 |
| DBMTZ1 | 8.7 | 4.4 | 4.0 | --- | --- | 3.0 | 1.0 |
| DBMTZ3 | 8.7 | 4.4 | 4.0 | --- | --- | 3.0 | 3.0 |
| DBMTZ5 | 8.7 | 4.4 | 4.0 | --- | --- | 3.0 | 5.0 |
| DBMTZ7 | 8.7 | 4.4 | 4.0 | --- | --- | 3.0 | 7.0 |
| Ingredient | Formulation Code | ||||||
|---|---|---|---|---|---|---|---|
| A | B | BNLC | C | CNLC | D | DNLC | |
| Phase A | |||||||
| Cetiol S a | 3.0 | 3.0 | 3.0 | 3.0 | 3.0 | 3.0 | 3.0 |
| MYR b | 2.0 | 2.0 | 2.0 | 2.0 | 2.0 | 2.0 | 2.0 |
| Greensyl c | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Crodamol CAP d | 2.0 | 2.0 | 2.0 | 2.0 | 2.0 | 2.0 | 2.0 |
| Argan oil | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Wheat oil | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Shea oil | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| TA e | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 |
| Brij 72 f | 3.0 | 3.0 | 3.0 | 3.0 | 3.0 | 3.0 | 3.0 |
| Brij 721 g | 2.0 | 2.0 | 2.0 | 2.0 | 2.0 | 2.0 | 2.0 |
| Cetyl palmitate | 4.0 | 4.0 | 4.0 | 4.0 | 4.0 | 4.0 | 4.0 |
| Cetyl alcohol | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| BMTZ h | --- | 0.4 | --- | 0.8 | --- | 1.6 | --- |
| Phase B | |||||||
| EDTA i | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 |
| Water | q.s. l | q.s. l | q.s. l | q.s. l | q.s. l | q.s. l | q.s. l |
| Phase C | |||||||
| Kemipur 100 m | 0.35 | 0.35 | 0.35 | 0.35 | 0.35 | 0.35 | 0.35 |
| Benzyl alcohol | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 | 0.25 |
| Phase D | |||||||
| Parfum | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 |
| Phase E | |||||||
| BMTZ-NLC n | --- | --- | 5.0 | --- | 10.0 | --- | 20.0 |
| NLC Code | Z-Average ± S.D. (nm) | PDI | Zeta ± S.D. (mV) |
|---|---|---|---|
| IPM1 | 38.5 ± 1.5 | 0.102 ± 0.008 | −10.3 ± 1.1 |
| IPM2 | 37.4 ± 1.8 | 0.111 ± 0.007 | −9.2 ± 1.2 |
| IPM3 | 37.9 ± 2.0 | 0.107 ± 0.011 | −11.3 ± 0.9 |
| MYR1 | 47.9 ± 2.8 | 0.115 ± 0.008 | −8.9 ± 1.0 |
| MYR2 | 45.6 ± 1.6 | 0.105 ± 0.009 | −10.4 ± 1.7 |
| MYR3 | 48.1 ± 1.9 | 0.109 ± 0.007 | −9.4 ± 0.8 |
| DO1 | 45.3 ± 1.4 | 0.153 ± 0.009 | −11.5 ± 1.6 |
| DO2 | 41.1 ± 2.0 | 0.171 ± 0.012 | −9.9 ± 1.2 |
| DO3 | 41.7 ± 2.3 | 0.166 ± 0.008 | −10.6 ± 1.8 |
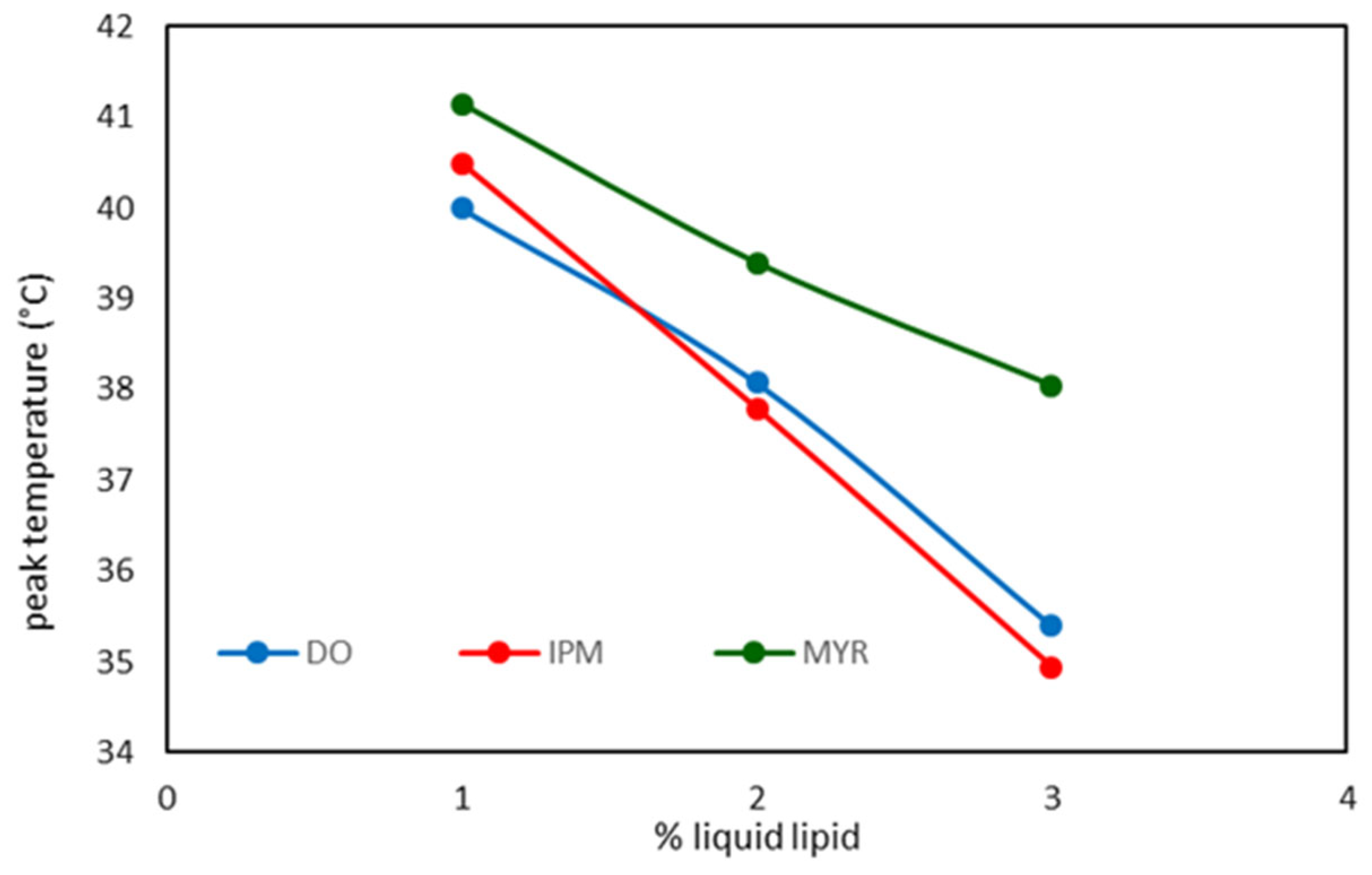
| NLC Code | ΔH ± S.D. (J/g) | RI % |
|---|---|---|
| DO 1 | 9.46 ± 0.38 | 63.9 |
| DO 2 | 7.22 ± 0.14 | 58.5 |
| DO 3 | 5.01 ± 0.05 | 50.8 |
| IPM 1 | 8.96 ± 0.24 | 60.5 |
| IPM 2 | 6.59 ± 0.20 | 53.4 |
| IPM 3 | 2.23 ± 0.08 | 22.6 |
| MYR 1 | 10.37 ± 0.45 | 70.0 |
| MYR 2 | 7.80 ± 0.15 | 63.0 |
| MYR 3 | 4.21 ± 0.17 | 42.6 |
| NLC Code | Z-Average ± S.D. (nm) | PDI | Zeta ± S.D. (mV) |
|---|---|---|---|
| IBMTZ1 | 33.2 ± 2.1 | 0.217 ± 0.015 | −10.4 ± 1.3 |
| IBMTZ3 | 35.5 ± 1.9 | 0.090 ± 0.004 | −9.9 ± 1.7 |
| IBMTZ5 | 45.3 ± 2.3 | 0.115 ± 0.009 | −11.8 ± 1.4 |
| IBMTZ7 | 72.5 ± 6.3 | 0.149 ± 0.014 | −9.9 ± 1.3 |
| MBMTZ1 | 49.8 ± 2.9 | 0.127 ± 0.011 | −11.2 ± 1.4 |
| MBMTZ3 | 58.4 ± 2.1 | 0.136 ± 0.009 | −10.2 ± 1.9 |
| MBMTZ5 | 78.1 ± 3.9 | 0.191 ± 0.018 | −11.5 ± 0.9 |
| MBMTZ7 | 99.3 ± 2.5 | 0.289 ± 0.019 | −9.9 ± 1.2 |
| DBMTZ1 | 45.0 ± 4.1 | 0.129 ± 0.013 | −10.7 ± 1.5 |
| DBMTZ3 | 50.8 ± 4.3 | 0.167 ± 0.015 | −11.2 ± 1.4 |
| DBMTZ5 | 66.9 ± 3.3 | 0.181 ± 0.018 | −11.3 ± 1.8 |
| DBMTZ7 | 81.5 ± 2.8 | 0.264 ± 0.019 | −10.8 ± 0.7 |
| NLC Code | ΔH ± S.D. (J/g) | RI % |
|---|---|---|
| DBMTZ1 | 2.72 ± 0.12 | 27.5 |
| DBMTZ3 | 3.67 ± 0.13 | 37.1 |
| DBMTZ5 | 2.03 ± 0.04 | 20.6 |
| DBMTZ7 | 1.94 ± 0.09 | 19.6 |
| IBMTZ1 | 2.00 ± 0.04 | 20.2 |
| IBMTZ3 | 2.46 ± 0.11 | 24.8 |
| IBMTZ5 | 2.93 ± 0.06 | 29.6 |
| IBMTZ7 | 0.83 ± 0.03 | 8.4 |
| MBMTZ1 | 2.10 ± 0.10 | 21.2 |
| MBMTZ3 | 4.04 ± 0.12 | 40.9 |
| MBMTZ5 | 2.58 ± 0.05 | 26.1 |
| MBMTZ7 | 4.37 ± 0.21 | 44.2 |
| Emulsion Code | pH | F | S (cm) | V (cP) | Q (μg) | SPF |
|---|---|---|---|---|---|---|
| A | 6.6 ± 0.1 | 50.41 ± 1.52 | 8.92 ± 0.31 | 27,200 ± 1200 | --- | 1.22 ± 0.04 |
| B | 7.5 ± 0.2 | 53.31 ± 1.43 | 9.71 ± 0.22 | 26,500 ± 1400 | 0.60 ± 0.12 | 3.03 ± 0.11 |
| BNLC | 7.0 ± 0.1 | 51.45 ± 0.99 | 10.33 ± 0.40 | 23,300 ± 900 | 0.98 ± 0.23 | 3.54 ± 0.12 |
| C | 7.4 ± 0.1 | 51.70 ± 1.32 | 8.73 ± 0.38 | 27,300 ± 1500 | 0.88 ± 0.17 | 4.14 ± 0.18 |
| CNLC | 7.4 ± 0.2 | 49.83 ± 1.44 | 10.2 ± 0.14 | 23,900 ± 1100 | 0.87 ± 0.19 | 4.78 ± 0.21 |
| D | 7.5 ± 0.1 | 50.66 ± 0.98 | 9.32 ± 0.02 | 27,000 ±1400 | 0.71 ± 0.11 | 5.26 ± 0.28 |
| DNLC | 7.4 ± 0.1 | 51.07 ± 1.02 | 10.8 ± 0.28 | 23,100 ± 1000 | 0.66 ± 0.13 | 6.34 ± 0.29 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sarpietro, M.G.; Santonocito, D.; Greco, G.; Russo, S.; Puglia, C.; Montenegro, L. Bemotrizinol-Loaded Lipid Nanoparticles for the Development of Sunscreen Emulsions. Colloids Interfaces 2025, 9, 54. https://doi.org/10.3390/colloids9050054
Sarpietro MG, Santonocito D, Greco G, Russo S, Puglia C, Montenegro L. Bemotrizinol-Loaded Lipid Nanoparticles for the Development of Sunscreen Emulsions. Colloids and Interfaces. 2025; 9(5):54. https://doi.org/10.3390/colloids9050054
Chicago/Turabian StyleSarpietro, Maria Grazia, Debora Santonocito, Giuliana Greco, Stefano Russo, Carmelo Puglia, and Lucia Montenegro. 2025. "Bemotrizinol-Loaded Lipid Nanoparticles for the Development of Sunscreen Emulsions" Colloids and Interfaces 9, no. 5: 54. https://doi.org/10.3390/colloids9050054
APA StyleSarpietro, M. G., Santonocito, D., Greco, G., Russo, S., Puglia, C., & Montenegro, L. (2025). Bemotrizinol-Loaded Lipid Nanoparticles for the Development of Sunscreen Emulsions. Colloids and Interfaces, 9(5), 54. https://doi.org/10.3390/colloids9050054












