Adsorption of Uranium (VI) from Aqueous Solutions by Amino-functionalized Clay Minerals
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Materials
2.2. Silylation with APTES
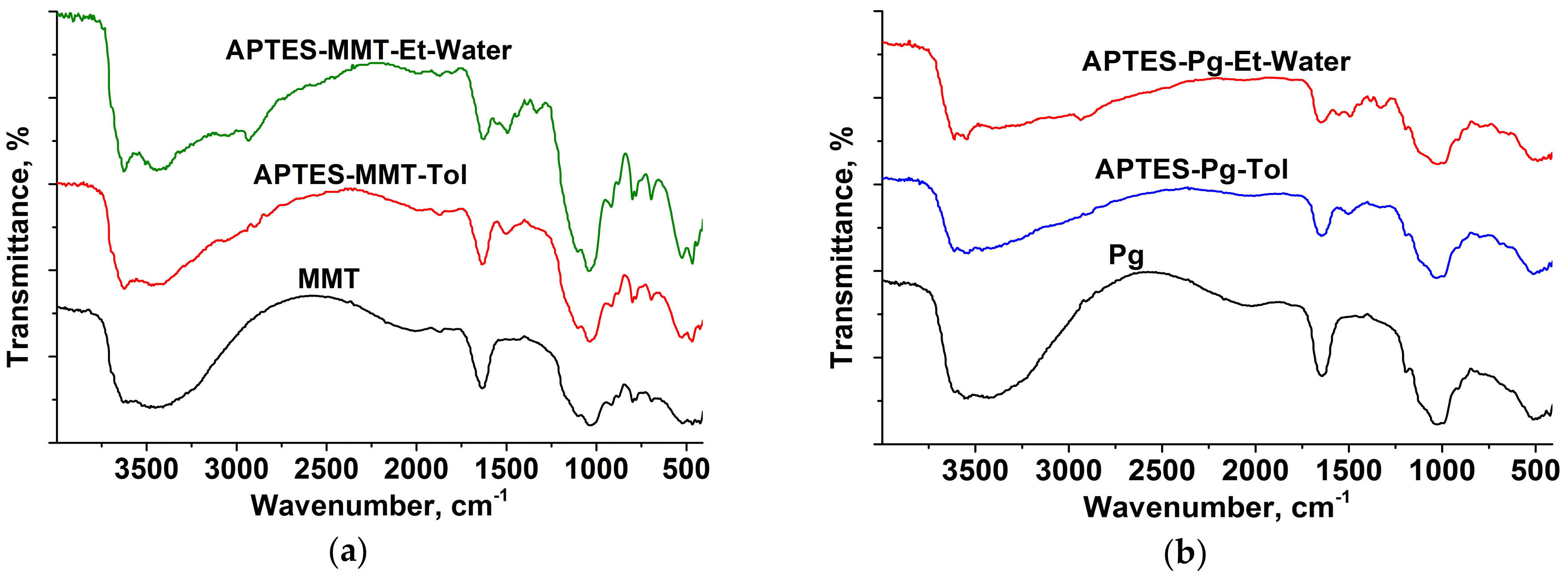
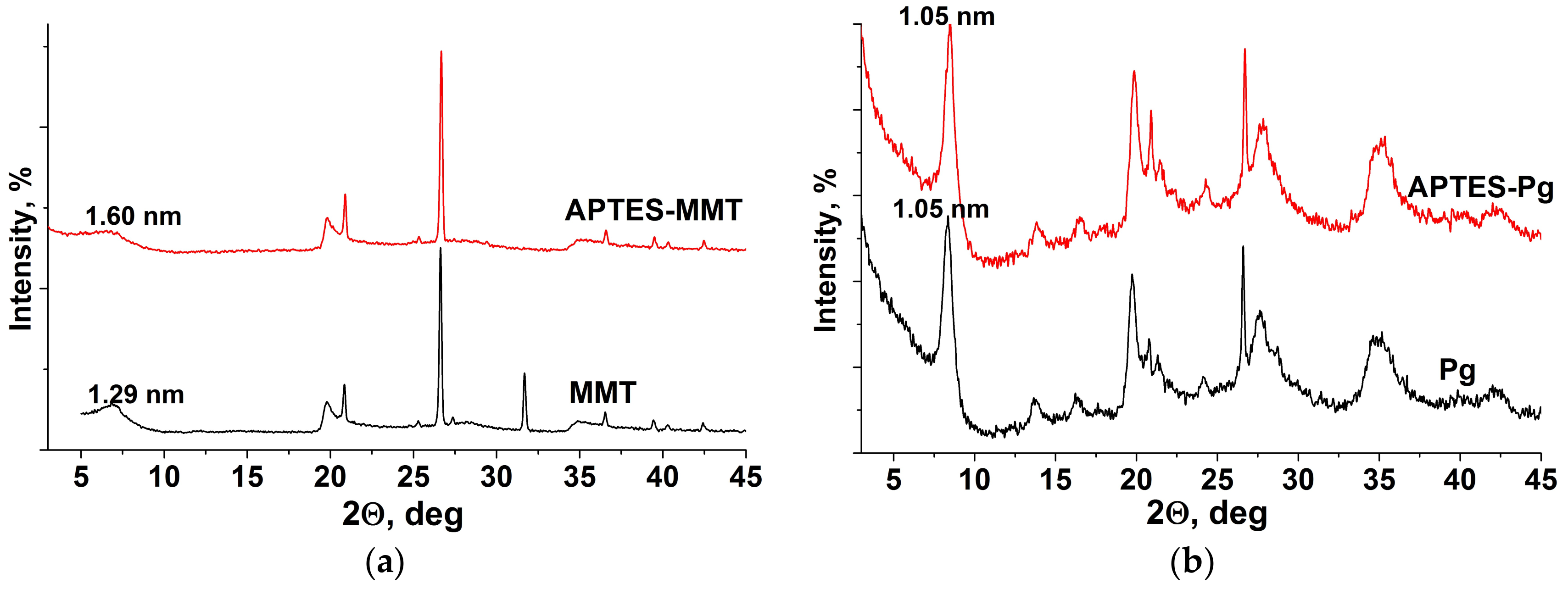
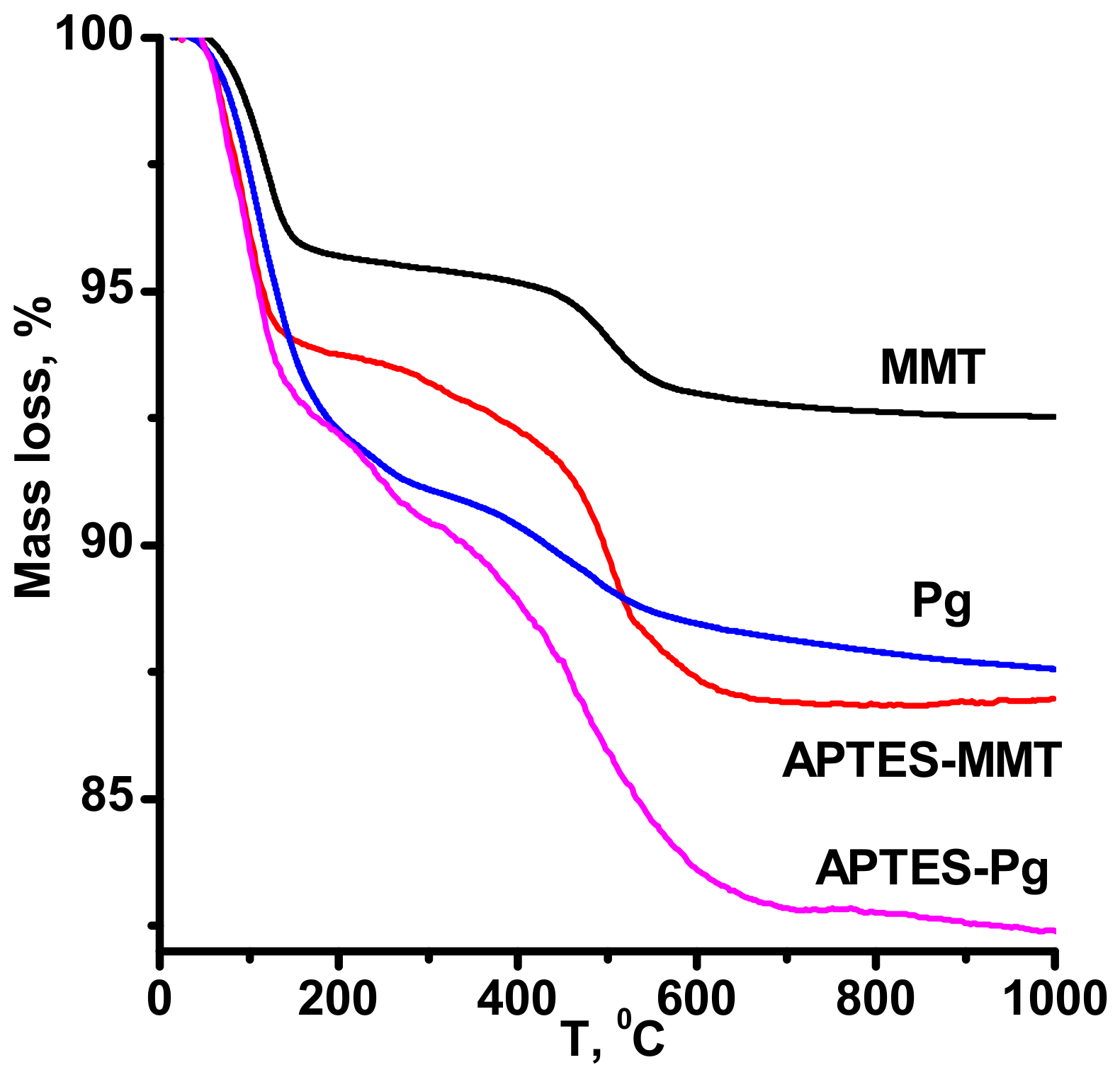
2.3. Characterization Methods
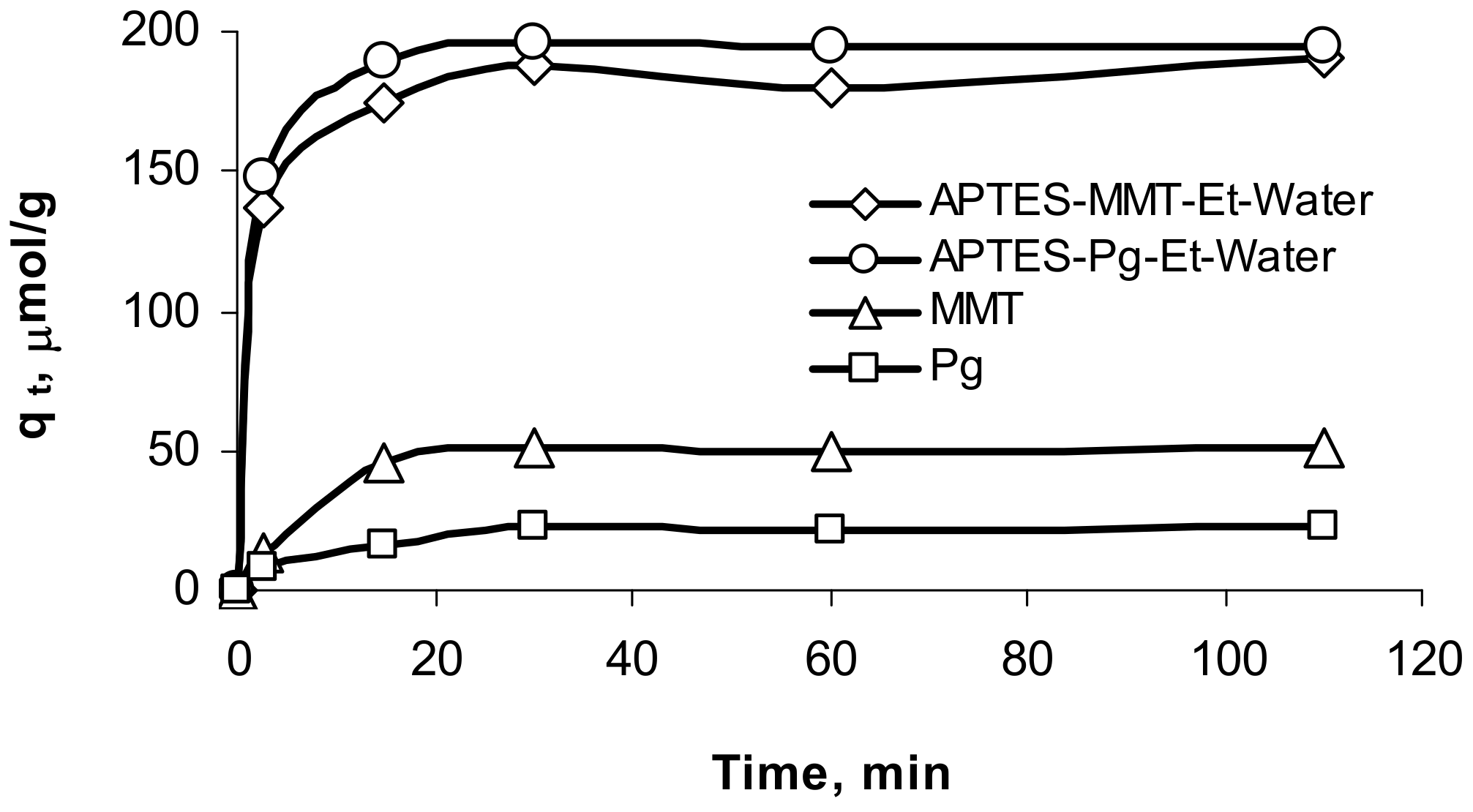
2.4. Adsorption Experiments
3. Results
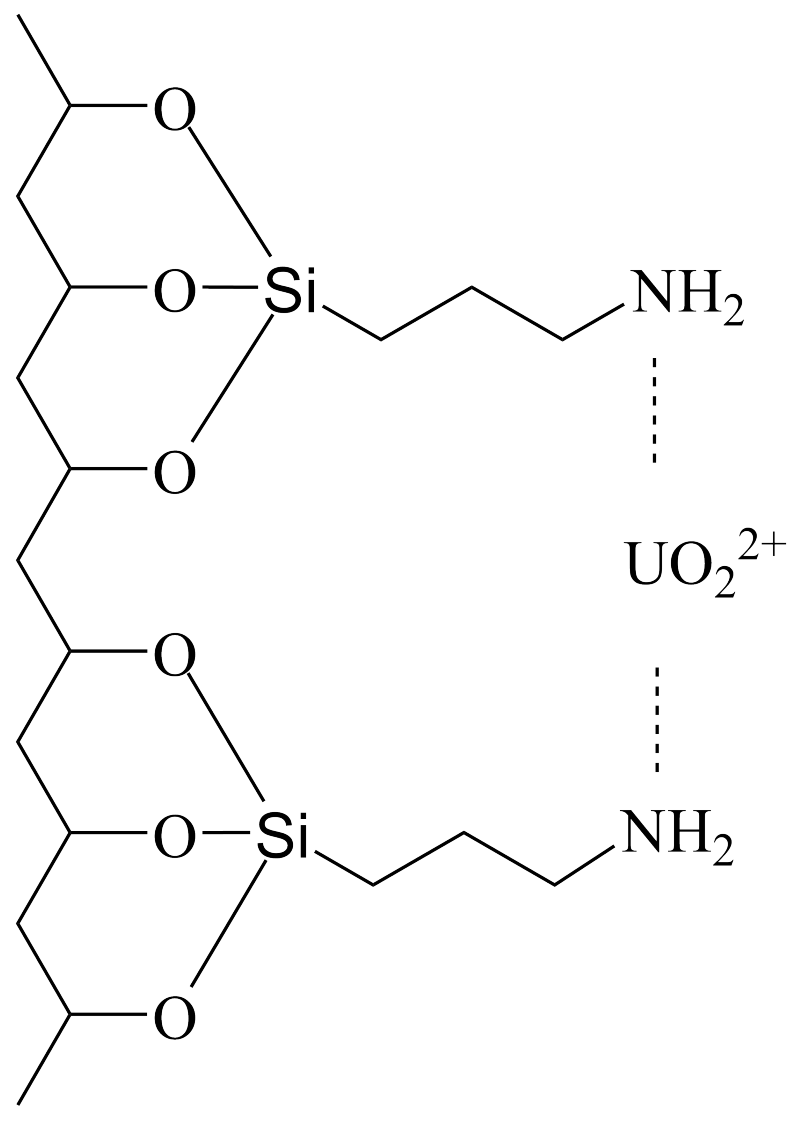
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bergaya, F.; Theng, B.K.; Lagaly, G. (Eds.) Handbook of Clay Science; Developments in Clay Science; Elsevier Ltd.: Amsterdam, The Netherlands, 2006; Volume 1. [Google Scholar]
- Ngulube, T.; Gumbo, J.R.; Masindi, V.; Maity, A. An update on synthetic dyes adsorption onto clay based minerals: A state-of-art review. J. Environ. Manag. 2017, 191, 35–57. [Google Scholar] [CrossRef]
- Cecilia, J.A.; García-Sancho, C.; Vilarrasa-García, E.; Jiménez-Jiménez, J.; Rodriguez-Castellón, E. Synthesis, Characterization, Uses and Applications of Porous Clays Heterostructures: A Review. Chem. Rec. 2018, 18, 1–21. [Google Scholar] [CrossRef]
- Kami´nska, G. Removal of Organic Micropollutants by Grainy Bentonite-Activated Carbon Adsorbent in a Fixed Bed Column. Water 2018, 10, 1791. [Google Scholar] [CrossRef]
- Bogolepov, A.A.; Pshinko, G.N.; Kornilovich, B.Yu. The impact of complexing agents on the processes of sorption treatment of waters containing uranium. J. Water Chem. Technol. 2007, 29, 9–14. [Google Scholar] [CrossRef]
- Najafi, M.; Yousefi, Y.; Rafati, A.A. Synthesis, characterization and adsorption studies of several heavy metal ions on amino-functionalized silica nano hollow sphere and silica gel. Sep. Purif. Technol. 2012, 85, 193–205. [Google Scholar] [CrossRef]
- Nakanishi, K.; Tomita, M.; Kato, K. Synthesis of amino-functionalized mesoporous silica sheets and their application for metal ion capture. J. Asian Ceram. Soc. 2015, 3, 70–76. [Google Scholar] [CrossRef]
- Dána, E. Adsorption of heavy metals on functionalized-mesoporous silica: A review. Microporous Mesoporous Mater. 2017, 247, 145–157. [Google Scholar] [CrossRef]
- Ramasamy, D.L.; Repo, E. Synthesis of mesoporous and microporous amine and non-amine functionalized silica gels for the application of rare earth elements (REE) recovery from the waste water-understanding the role of pH, temperature, calcination and mechanism in Light REE and Heavy REE separation. Chem. Eng. J. 2017, 322, 56–65. [Google Scholar] [CrossRef]
- Diagboya, P.N.E.; Dikio, E.D. Silica-based mesoporous materials; emerging designer adsorbents for aqueous pollutants removal and water treatment. Microporous Mesoporous Mater. 2018, 266, 252–267. [Google Scholar] [CrossRef]
- Yang, S.; Yuan, P.; He, H.-P.; Qin, Z. Effect of reaction temperature on grafting of γ-aminopropyl triethoxysilane (APTES) onto kaolinite. Appl. Clay Sci. 2012, 62–63, 8–14. [Google Scholar] [CrossRef]
- Fatimah, I. Preparation, characterization and physicochemical study of 3-amino propyl trimethoxy silane-modified kaolinite for Pb(II) adsorption. J. King Saud Univ. 2017, 30, 250–257. [Google Scholar] [CrossRef]
- Balomenou, G.; Stathi, P.; Enotiadis, A.; Gournis, D.; Deligiannakis, Y. Physicochemical study of amino-functionalized organosilicon cubes intercalated in montmorillonite clay: H-binding and metal uptake. J. Colloid Interface Sci. 2008, 325, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Addy, M.; Losey, B.; Mohseni, R.; Zlotnikov, E.; Vasiliev, A. Adsorption of heavy metal ions on mesoporous silica-modified montmorillonite containing a grafted chelate ligand. Appl. Clay Sci. 2012, 59–60, 115–120. [Google Scholar] [CrossRef]
- Su, L.; Tao, Q.; He, H.; Zhu, J.; Yuan, P.; Zhu, R. Silylation of montmorillonite surfaces: Dependence on solvent nature. J. Colloid Interface Sci. 2013, 391, 16–20. [Google Scholar] [CrossRef]
- Fang, L.; Wang, L.; Zhou, T.; Liu, L.; Li, M. Preparation and characterization of Fe,Co,Si-pillared montmorillonites with aminosilanes as silicon pillars precursor. Appl. Clay Sci. 2017, 141, 88–94. [Google Scholar] [CrossRef]
- Xue, A.; Zhou, Sh.; Zhao, Yi.; Lu, X. Adsorption of reactive dyes from aqueous solution by silylated palygorskite. Appl. Clay Sci. 2010, 48, 638–640. [Google Scholar] [CrossRef]
- Guerra, D.L.; Silva, E.M.; Airoldi, C. Application of modified attapulgites as adsorbents for uranyl uptake from aqueous solution—Thermodynamic approach. Process Saf. Environ. Prot. 2010, 88, 53–61. [Google Scholar] [CrossRef]
- Liang, X.; Xu, Y.; Tan, X.; Wang, L.; Sun, Y.; Lin, D.; Sun, Y.; Qin, X.; Wang, Q. Heavy metal adsorbents mercapto and amino functionalized palygorskite: Preparation and characterization. Colloids Surf. A 2013, 426, 98–105. [Google Scholar] [CrossRef]
- Borrego, T.; Andrade, M.; Pinto, M.L.; Silva, A.R.; Carvalho, A.P.; Rocha, J.; Freire, C.; Pires, J. Physicochemical characterization of silylated functionalized materials. J. Colloid Interface Sci. 2010, 344, 603–610. [Google Scholar] [CrossRef]
- Sousa, C.; Pereira, C.; Rodriguez-Borges, J.E.; Freire, C. L-Serine functionalized clays: Preparation and characterization. Polyhedronics 2015, 102, 121–129. [Google Scholar] [CrossRef]
- He, H.; Tao, Q.; Zhu, J.; Yuan, P.; Shen, W.; Yang, S. Silylation of clay mineral surfaces. Appl. Clay Sci. 2013, 71, 15–20. [Google Scholar] [CrossRef]
- Silva, A.A.; Dahmouche, K.; Soares, B.G. Nanostructure and dynamic mechanical properties of silane-functionalized montmorillonite/epoxy nanocomposites. Appl. Clay Sci. 2011, 54, 151–158. [Google Scholar] [CrossRef]
- Asgari, M.; Abouelmagd, A.; Sundararaj, U. Silane functionalization of sodium montmorillonite nanoclay and its effect on rheological and mechanical properties of HDPE/clay nanocomposites. Appl. Clay Sci. 2017, 146, 439–448. [Google Scholar] [CrossRef]
- Zhang, S.; Liu, Q.; Cheng, H.; Zhang, Yu.; Li, X.; Frost, R.L. Intercalation of γ-aminopropyl triethoxysilane (APTES) into kaolinite interlayer with methanol-grafted kaolinite as intermediate. Appl. Clay Sci. 2015, 114, 484–490. [Google Scholar] [CrossRef]
- Murray, H.H. Applied Clay Mineralogy; Elsevier: Amsterdam, The Netherlands, 2007; ISBN 9780444517012. [Google Scholar]
- Galan, E.; Singer, A. (Eds.) Developments in Palygorskite-Sepiolite Research; Elsevier: Amsterdam, The Netherlands, 2011; ISBN 9780444536075. [Google Scholar]
- Nguyen-Trung, C.; Begun, G.M.; Palmer, D.A. Aqueous uranium complexes. 2. Raman spectroscopic study of the complex formation of the dioxouranium (VI) ion with a variety of inorganic and organic ligands. Inorg. Chem. 1992, 31, 5280–5287. [Google Scholar] [CrossRef]
- Kornilovych, B.; Wireman, M.; Ubaldini, S.; Guglietta, D.; Koshik, Yu.; Caruso, B.; Kovalchuk, I. Uranium Removal from Groundwater by Permeable Reactive Barrier with Zero-Valent Iron and Organic Carbon Mixtures: Laboratory and Field Studies. Metals 2018, 8, 408. [Google Scholar] [CrossRef]
- Langmuir, D. Aqueous Environmental Geochemistry; Prentice-Hall, Inc.: Upper Saddle River, NJ, USA, 1997; ISBN 978-0023674129. [Google Scholar]
- Tarasevich, Y.I. Poverhnostnyie yavleniya na dispesnyih materialah [The Superficial Phenomena on Disperse Materials]; Naukova dumka: Kyiv, Ukraine, 2011; p. 390. ISBN 978-966-00-1137-3. [Google Scholar]
- Zhou, L.; Chen, H.; Jiang, X.; Lu, F.; Zhou, Y.; Yin, W.; Ji, X. Modification of montmorillonite surfaces using a novel class of cationic gemini surfactants. J. Colloid Interface Sci. 2009, 332, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Moreira, M.A.; Ciuffi, K.; Rives, V.; Vicente, M.A.; Trujillano, R.; Gil, A.; Korili, S.A.; Faria, E.H. Effect of chemical modification of palygorskite and sepiolite by 3-aminopropyltriethoxisilane on adsorption of cationic and anionic dyes. Appl. Clay Sci. 2017, 135, 394–404. [Google Scholar] [CrossRef]
- Sing, K.S.W.; Everett, D.H.; Haul, R.A.W.; Moscou, L.; Pierotti, R.A.; Rouquerol, J.; Siemieniewska, T. Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity. Pure Appl. Chem. Res. 1985, 57, 603–619. [Google Scholar] [CrossRef]
- Rouquerol, F.; Rouquerol, J.; Sing, K.S.W.; Llewellyn, P.; Maurin, G. Adsorption by Powders and Porous Solids; Elsevier: Amsterdam, The Netherlands, 2014; ISBN 9780080970356. [Google Scholar]
- Kornilovich, B.Y.; Pshinko, G.N.; Bogolepov, A.A. Effects of EDTA and NTA on sorption of U(VI) on the clay fraction of soil. Radiochemistry 2006, 48, 584–588. [Google Scholar] [CrossRef]
- Abd El-Magied, M.O.; Mohammaden, T.F.; El-Aassy, I.K.; Gad, H.M.H.; Hassan, A.M.; Mahmoud, M.A. Decontamination of Uranium-Polluted Groundwater by Chemically-Enhanced, Sawdust-Activated Carbon. Colloids Interfaces 2017, 1, 2. [Google Scholar] [CrossRef]
- Sheng, G.; Shao, X.; Li, Y.; Li, J.; Dong, H.; Cheng, W.; Gao, X.; Huang, Y. Enhanced Removal of Uranium(VI) by Nanoscale Zerovalent Iron Supported on Na-Bentonite and an Investigation of Mechanism. J. Phys. Chem. A 2014, 118, 2952–2958. [Google Scholar] [CrossRef] [PubMed]
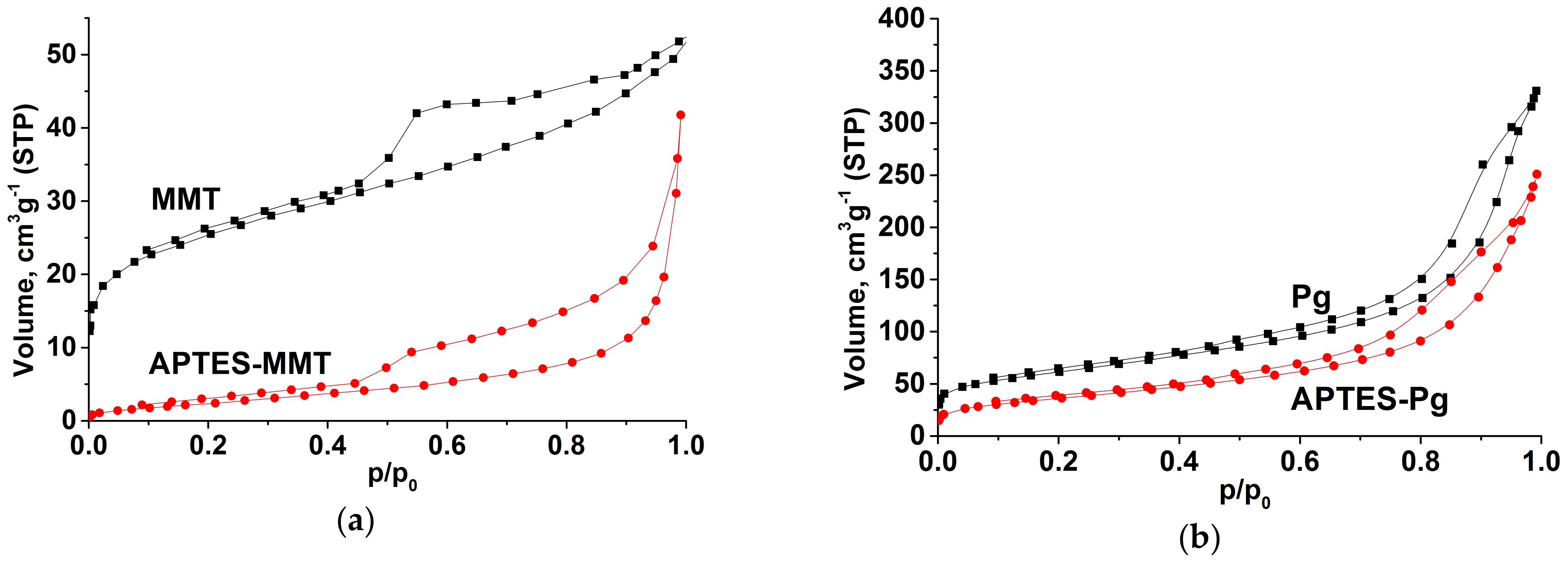
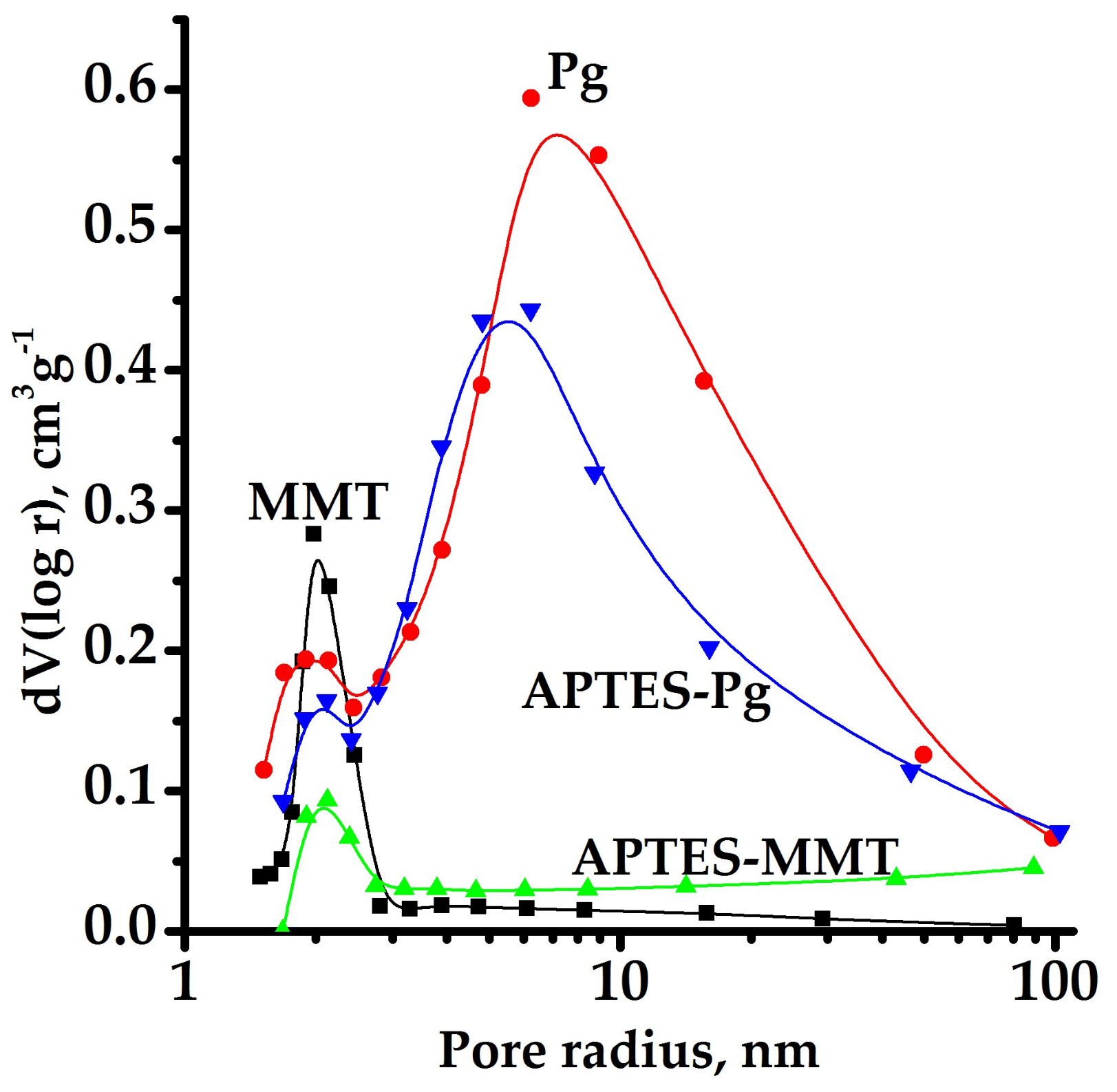
| Sample | S, m2/g | V∑, cm3/g | Vμ, cm3/g | Vμ%, % | Distribution of Pore Sizes, nm | |
|---|---|---|---|---|---|---|
| BJH dV (r) | ||||||
| r1 | r2 | |||||
| Montmorillonite | 89 | 0.081 | 0.016 | 19.80 | 1.41 | – |
| APTES- Montmorillonite | 10 | 0.064 | 0.004 | 5.87 | 2.13 | – |
| Palygorskite | 213 | 0.512 | 0.084 | 16.56 | 1.90 | 6.26 |
| APTES- Palygorskite | 129 | 0.389 | 0.048 | 12.30 | 2.12 | 6.23 |
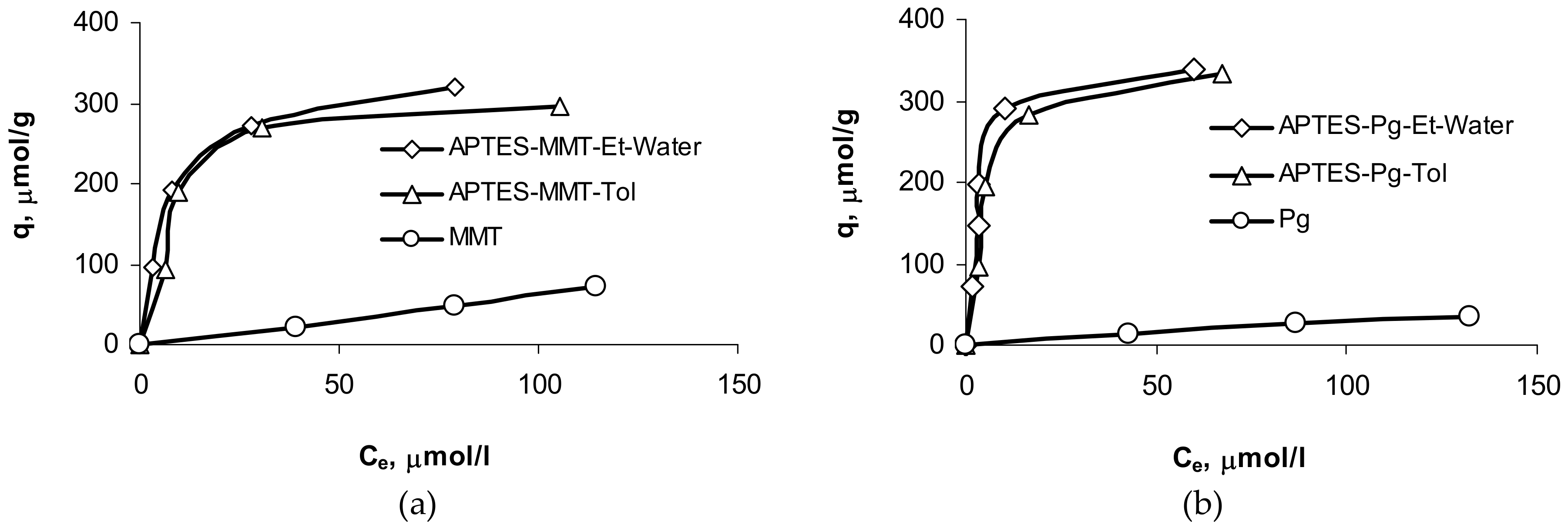
| Sample | Langmuir | Freundlich | ||||
|---|---|---|---|---|---|---|
| qm, μmol/g | KL, L/μmol | R2 | KF, L/g | n | R2 | |
| MMT | 125 | 0.0084 | 0.956 | 5.3 | 1.97 | 0.887 |
| APTES-MMT-Tol | 337 | 0.0977 | 0.974 | 89.0 | 3.69 | 0.765 |
| APTES-MMT-Et-Water | 350 | 0.1328 | 0.998 | 92.1 | 3.37 | 0.923 |
| Pg | 59 | 0.0096 | 0.975 | 3.3 | 2.14 | 0.851 |
| APTES-Pg-Tol | 372 | 0.1624 | 0.963 | 109.1 | 3.61 | 0.882 |
| APTES-Pg-Et-Water | 379 | 0.2317 | 0.960 | 119.6 | 3.70 | 0.770 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tobilko, V.; Spasonova, L.; Kovalchuk, I.; Kornilovych, B.; Kholodko, Y. Adsorption of Uranium (VI) from Aqueous Solutions by Amino-functionalized Clay Minerals. Colloids Interfaces 2019, 3, 41. https://doi.org/10.3390/colloids3010041
Tobilko V, Spasonova L, Kovalchuk I, Kornilovych B, Kholodko Y. Adsorption of Uranium (VI) from Aqueous Solutions by Amino-functionalized Clay Minerals. Colloids and Interfaces. 2019; 3(1):41. https://doi.org/10.3390/colloids3010041
Chicago/Turabian StyleTobilko, Victoria, Larysa Spasonova, Iryna Kovalchuk, Borys Kornilovych, and Yurij Kholodko. 2019. "Adsorption of Uranium (VI) from Aqueous Solutions by Amino-functionalized Clay Minerals" Colloids and Interfaces 3, no. 1: 41. https://doi.org/10.3390/colloids3010041
APA StyleTobilko, V., Spasonova, L., Kovalchuk, I., Kornilovych, B., & Kholodko, Y. (2019). Adsorption of Uranium (VI) from Aqueous Solutions by Amino-functionalized Clay Minerals. Colloids and Interfaces, 3(1), 41. https://doi.org/10.3390/colloids3010041





