Thermal Transformation of Caffeic Acid on the Nanoceria Surface Studied by Temperature Programmed Desorption Mass-Spectrometry, Thermogravimetric Analysis and FT–IR Spectroscopy
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Fourier Transform–Infrared (FT–IR) Spectroscopy of Caffeic Acid (CA) Surface Complexes
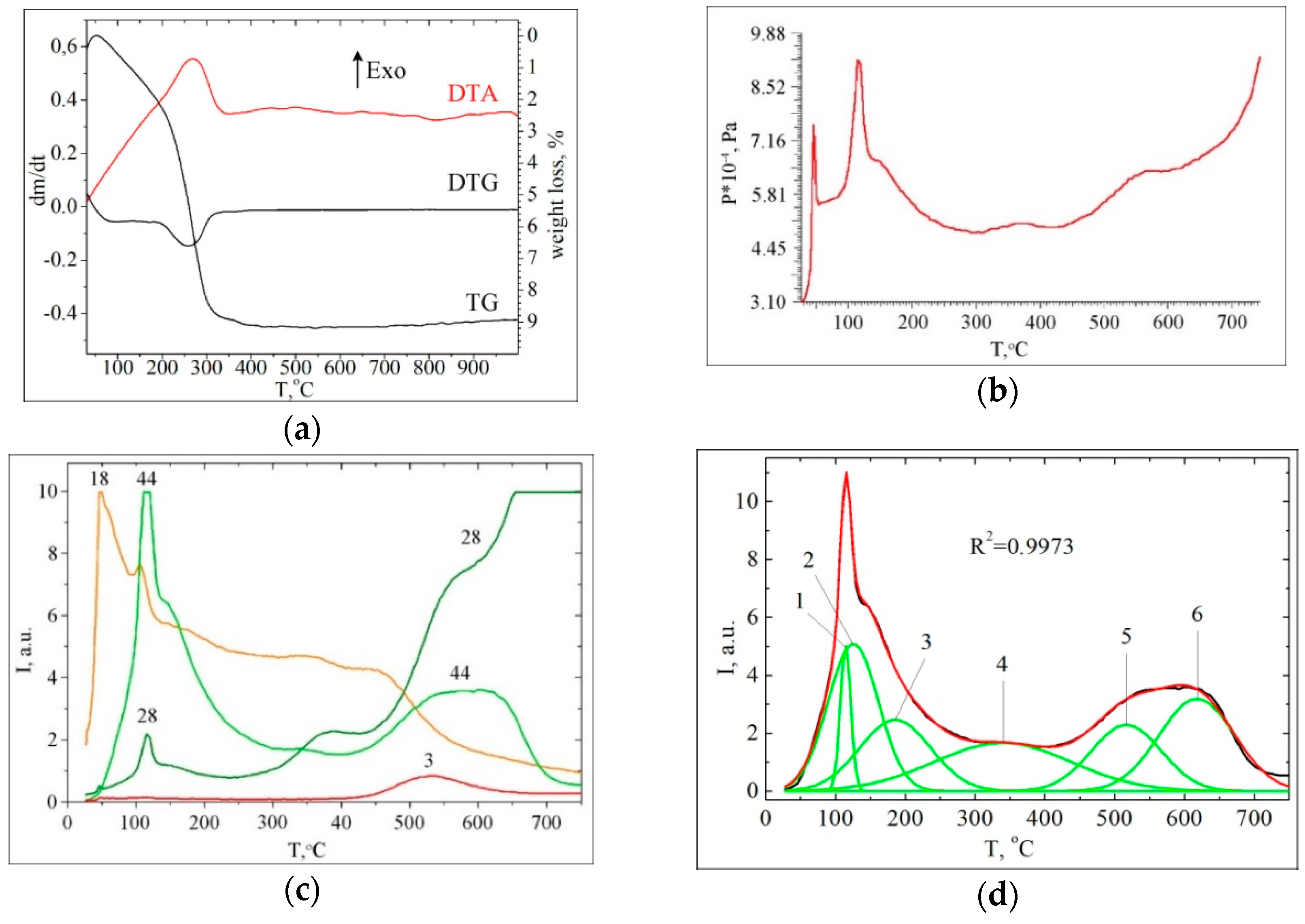
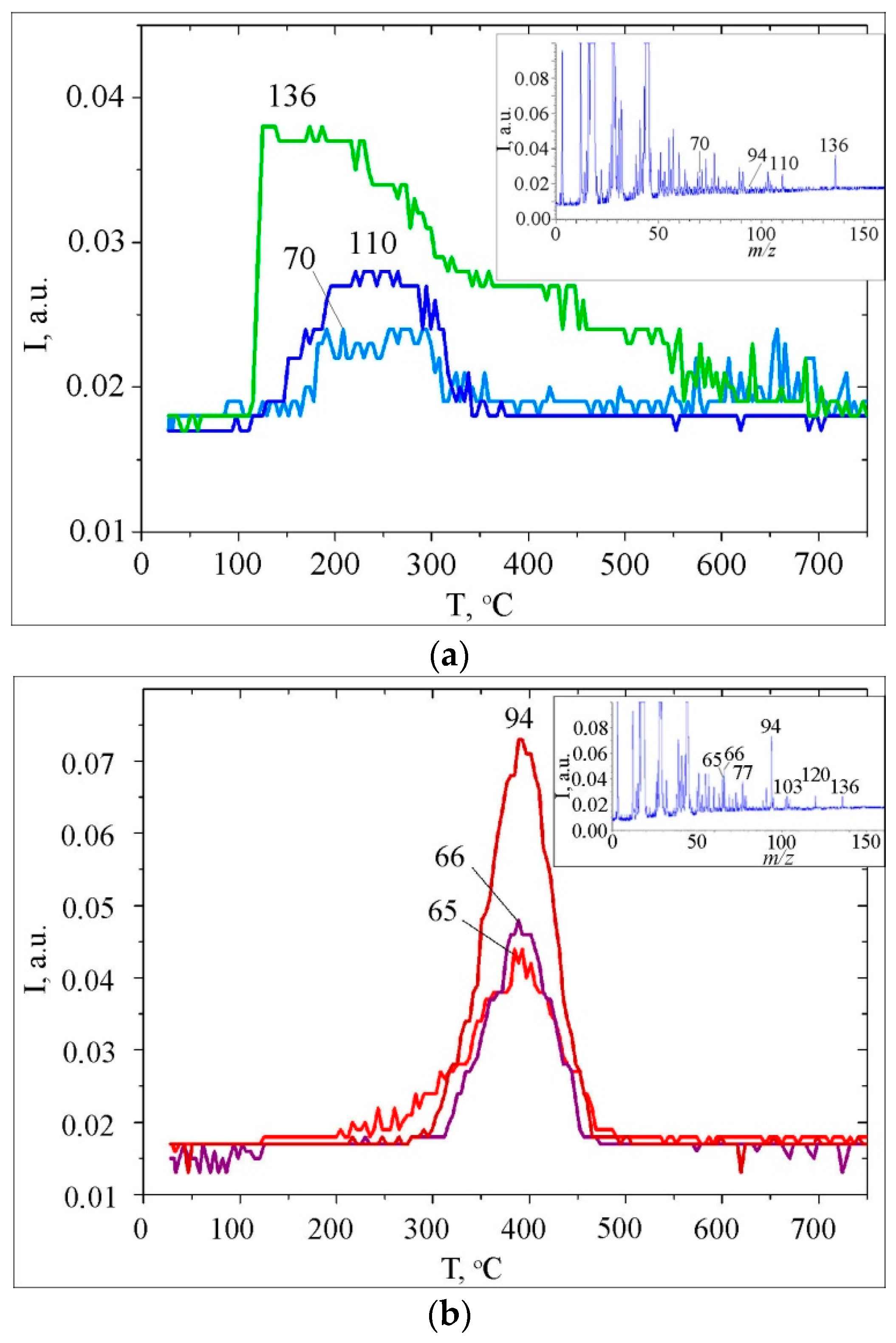
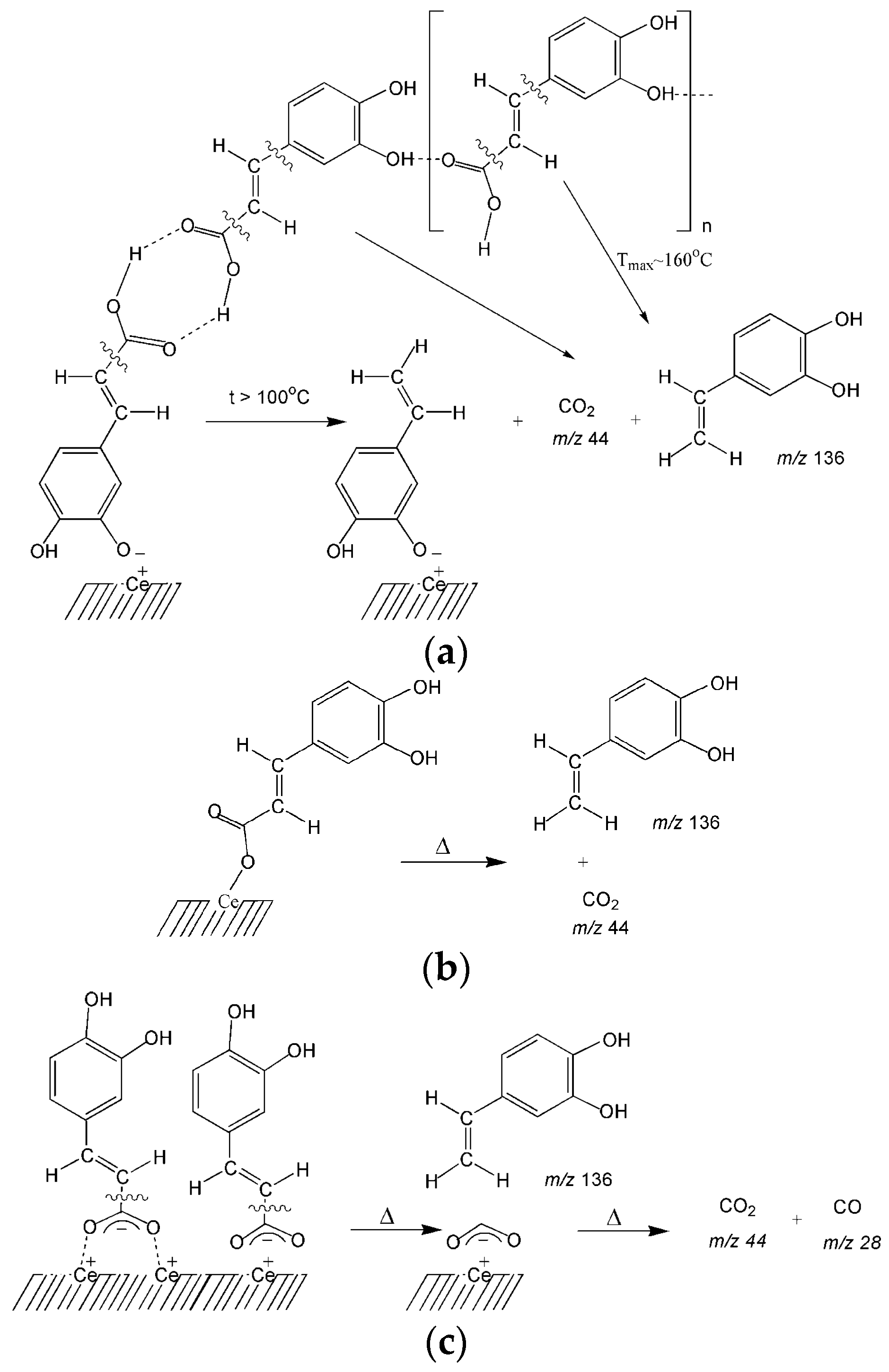
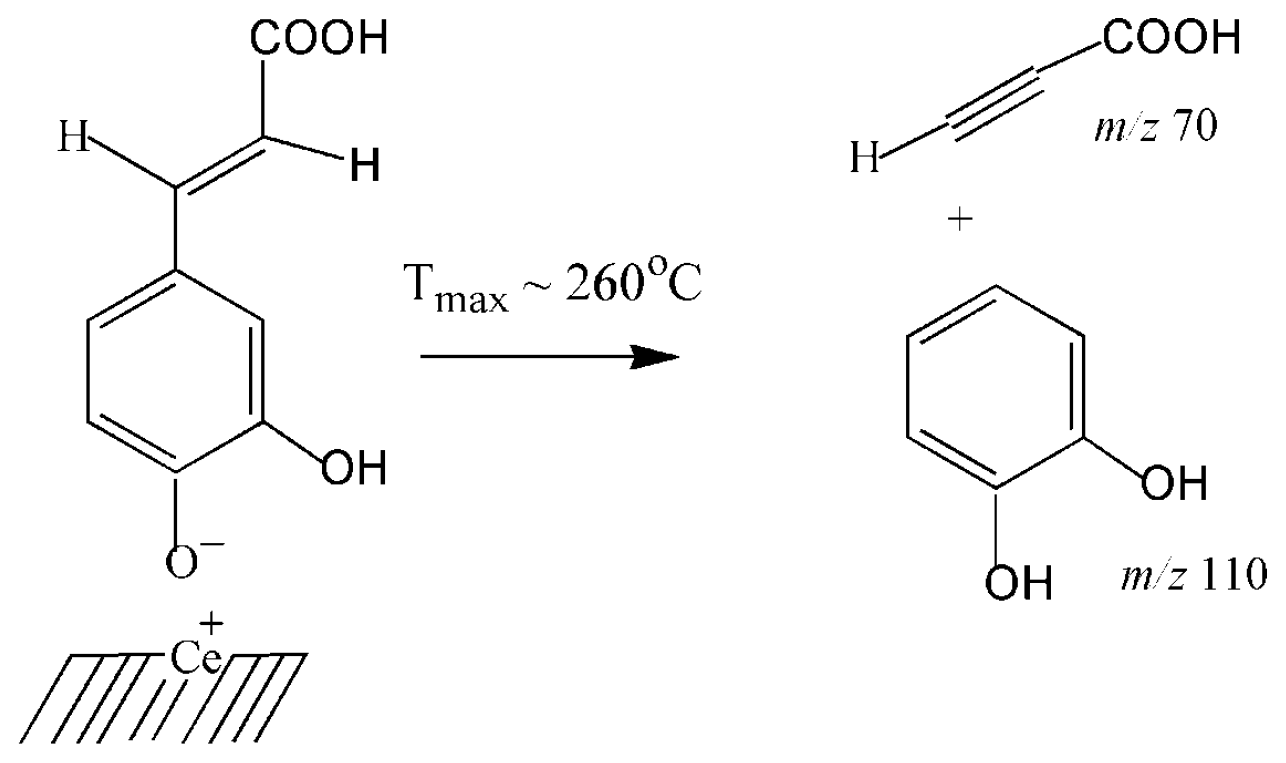
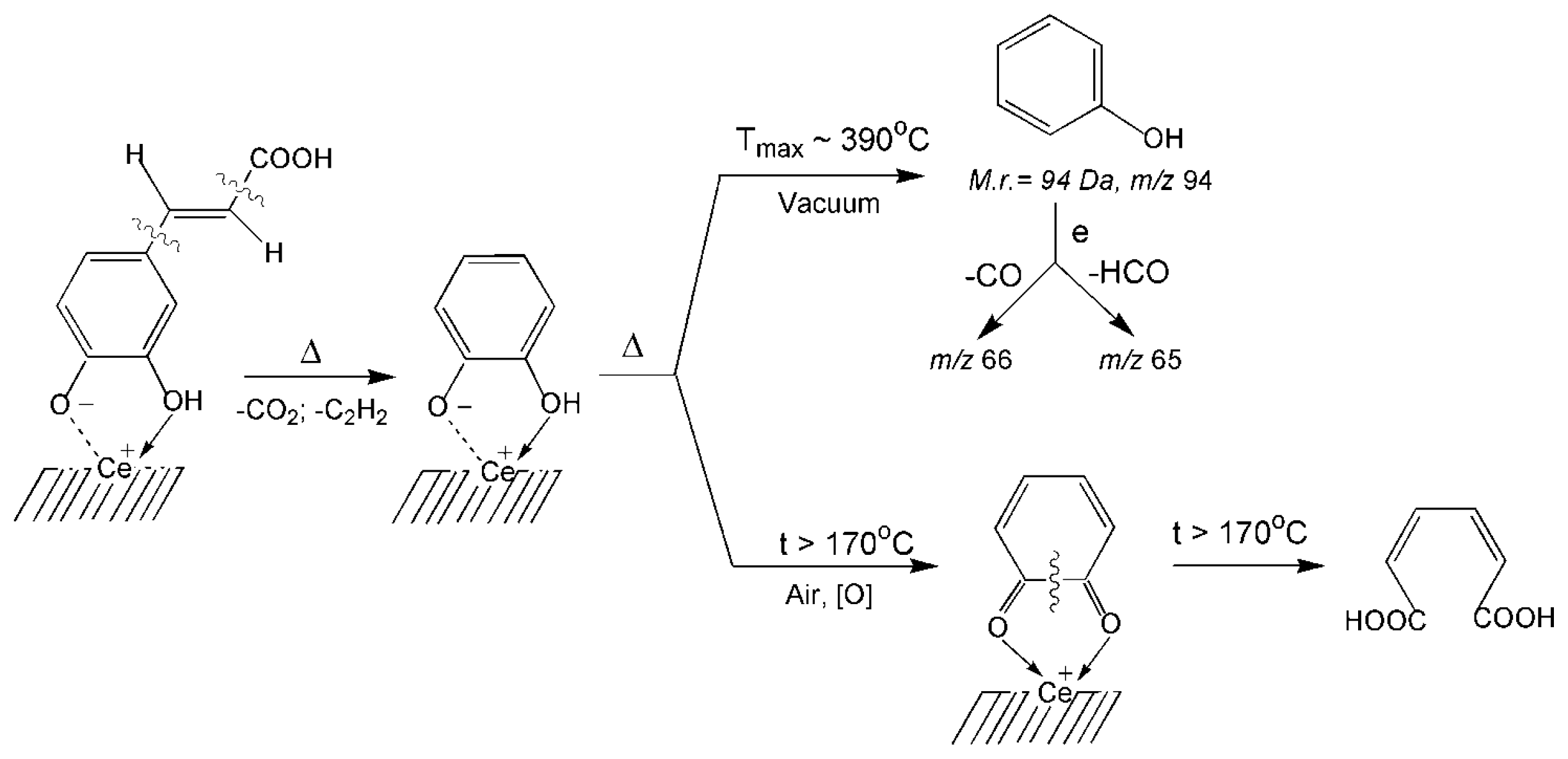
3.2. Thermal Transformations of CA on the Nanoceria Surface
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Campbell, C.T.; Peden, C.H.F. Oxygen Vacancies and Catalysis on Ceria Surfaces. Science 2005, 309, 713–714. [Google Scholar] [CrossRef] [PubMed]
- Orozco, L.M.; Renz, M.; Corma, A. Cerium oxide as a catalyst for the ketonization of aldehydes: Mechanistic insights and a convenient way to alkanes without the consumption of external hydrogen. Green Chem. 2017, 619, 1555–1569. [Google Scholar] [CrossRef]
- Montini, T.; Melchionna, M.; Monai, M.; Fornasiero, P. Fundamentals and Catalytic Applications of CeO2-Based Materials. Chem. Rev. 2016, 116, 5987–6041. [Google Scholar] [CrossRef] [PubMed]
- Trovarelli, A. Catalytic Properties of Ceria and CeO2-Containing Materials. Catal. Rev. 1996, 38, 439–520. [Google Scholar] [CrossRef]
- Vivier, L.; Duprez, D. Ceria-Based Solid Catalysts for Organic Chemistry. ChemSusChem 2010, 3, 654–678. [Google Scholar] [CrossRef] [PubMed]
- Nelson, N. Catalytic Upgrading of Phenolic Compounds Using Ceria-Based Materials. Ph.D. Thesis, Iowa State University, Ames, IA, USA, 2016. [Google Scholar]
- Das, S.; Dowding, J.M.; Klump, K.E.; McGinnis, J.F.; Self, W.; Seal, S. Cerium oxide nanoparticles: Applications and prospects in nanomedicine. Nanomedicine 2013, 8, 1483–1508. [Google Scholar] [CrossRef] [PubMed]
- Caputo, F.; Mameli, M.; Sienkiewicz, A.; Licoccia, S.; Stellacci, F.; Ghibelli, L.; Traversa, E. A novel synthetic approach of cerium oxide nanoparticles with improved biomedical activity. Sci. Rep. 2017, 7, 4636–4649. [Google Scholar] [CrossRef] [PubMed]
- Grulke, E.A.; Reed, K.; Beck, M.J.; Huang, X.; Cormack, A.N.; Seal, S. Nanoceria: Factors affecting its pro- and antioxidant Properties. Environ. Sci. Nano 2014, 1, 429–444. [Google Scholar] [CrossRef]
- Asati, M.; Santimukul Santra, S.; Kaittanis, C.; Nath, S.; Perez, J.M. Oxidase Activity of Polymer-Coated Cerium Oxide Nanoparticles. Angew. Chem. Int. Ed. Engl. 2009, 48, 2308–2312. [Google Scholar] [CrossRef] [PubMed]
- Hancock, M.L.; Grulke, E.A.; Yokel, R. Ceria Nanoparticle Dissolution and Stability in Acidic Aqueous Environments. In Proceedings of the AIChE Annual Meeting, Pittsburgh, PA, USA, 28 October–2 November 2018; p. 405a. [Google Scholar]
- Yokel, R.A.; Hancock, M.L.; Grulke, E.A.; Unrine, J.M.; Graham, U.M. Nanoceria Dissolution and Carboxylic Acid Stabilization in Aqueous Dispersions. FASEB J. 2017, 31, lb624. Available online: https://www.fasebj.org/doi/abs/10.1096/fasebj.31.1_supplement.lb6 24 (accessed on 6 December 2018).
- Ralph, J. Hydroxycinnamates in lignification. Phytochem. Rev. 2010, 9, 65–83. [Google Scholar] [CrossRef]
- Clifford, M.N. Chlorogenic acids and other cinnamatesnature, occurrence, dietary burden, absorption and metabolism. J. Sci. Food Agric. 2000, 80, 1033–1043. [Google Scholar] [CrossRef]
- Higdon, J.V.; Frei, B. Coffe and health: a rewiew of recent human research. Crit. Rev. Food Sci. 2006, 103, 101–123. [Google Scholar] [CrossRef] [PubMed]
- Clifford, N. Chlorogenic acid and other cinnamates: Nature, occurrence and dietary burden. J. Sci. Food Agric. 1999, 79, 362–372. [Google Scholar] [CrossRef]
- Lustosa, S.R.; Galindo, A.B.; Nunes, L.C.C.; Randau, K.P.; Rolim Neto, P.J. Prόpolis: Atualizações sobre a química e a farmacologia. Rev. Bras. Farmacogn. 2008, 18, 447–454. [Google Scholar] [CrossRef]
- Pandino, G.; Lombardo, S.; Williamson, G.; Mauromicale, G. Polyphenol profile and content in wild and cultivated. Cynara cardunculus L. Ital. J. Agron. 2012, 7, 254–261. [Google Scholar] [CrossRef]
- Chen, J.H.; Ho, C.T. Antioxidant activities of caffeic acid and its related hydroxycinamic acid compounds. J. Agric. Food Chem. 1997, 45, 2374–2378. [Google Scholar] [CrossRef]
- Lu, Y.; Foo, L. Antioxidant and radical scavenging activities of polyphenols from apple pomace. Food Chem. 2000, 68, 81–85. [Google Scholar] [CrossRef]
- Gulcin, I. Antioxidant activity of caffeic acid. Toxicology 2006, 217, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Magnani, C.; Isaac, V.L.B.; Correa, M.A.; Salgado, H.R.N. Caffeic acid: A review of its potential use in medications and cosmetics. Anal. Methods 2014, 6, 3203–3210. [Google Scholar] [CrossRef]
- Graf, E. Antioxidant potential of ferulic acid. Free Radic. Biol. Med. 1992, 13, 435–448. [Google Scholar] [CrossRef]
- Cuvelier, M.E.; Richard, H.; Berset, C. Antioxidative activity and phenolic composition of pilot-plant and commercial extracts of sage and rosemary. J. Am. Oil Chem. Soc. 1996, 73, 645–652. [Google Scholar] [CrossRef]
- Brand-Willams, W.; Cuvelier, M.E.; Berset, C. Use of free radical method to evaluate antioxidant activity. Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Almajano, M.P.; Carbo, R.; Delgado, M.E.; Gordon, M.H. Effect of pH on the Antimicrobial activity and oxidative stability of oil-in-water emulsions containing caffeic acid. J. Food Sci. 2007, 72, 258–263. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, V.; Verma, P. An overview of key pretreatment processes employed for bioconversion of lignocellulosic biomass into biofuels and value added products. 3 Biotech 2013, 3, 415–431. [Google Scholar] [CrossRef] [PubMed]
- Hodasova, L.; Jablonský, M.; Škulcová, A.; Ház, A. Lignin, potential products and their market value. Wood Res. 2015, 60, 973–986. [Google Scholar]
- Luo, Z.; Wang, Y.; He, M.; Zhao, C. Precise oxygen scission of lignin derived aryl ethers to quantitatively produce aromatic hydrocarbons in water. Green Chem. 2016, 18, 433–441. [Google Scholar] [CrossRef]
- Azizova, L.R.; Kulik, T.V.; Palianytsia, B.B.; Lipkovska, N.A. Thermal and hydrolytic stability of grafted ester groups of carboxylic acids on the silica surface. J. Therm. Anal. Calorim. 2015, 122, 517–523. [Google Scholar] [CrossRef]
- Kulik, T.V. Use of TPD–MS and Linear Free Energy Relationships for assessing the reactivity of aliphatic carboxylic acids on a silica surface. J. Phys. Chem. C 2011, 116, 570–580. [Google Scholar] [CrossRef]
- Kwart, H.; King, K. The Chemistry of Carboxylic Acids and Esters; Patai, S., Ed.; Interscience Publishers: London, UK, 1969. [Google Scholar]
- Nagashima, O.; Sato, S.; Takahashi, R.; Sodesawa, T. Ketonization of carboxylic acids over CeO2–based composite oxides. J. Mol. Catal. A Chem. 2005, 227, 231–239. [Google Scholar] [CrossRef]
- Pacchioni, G. Ketonization of Carboxylic Acids in Biomass Conversion over TiO2 and ZrO2 Surfaces: A DFT Perspective. ACS Catal. 2014, 4, 2874–2888. [Google Scholar] [CrossRef]
- Kulik, T.V.; Barvinchenko, V.N.; Palyanitsa, B.B.; Smirnova, O.V.; Pogorelyi, V.K.; Chuiko, A.A. A desorption mass spectrometry study of the interaction of cinnamic acid with a silica surface. Russ. J. Phys. Chem. 2007, 8, 83–90. [Google Scholar] [CrossRef]
- Kulik, T.V.; Lipkovska, N.A.; Barvinchenko, V.N.; Palyanytsya, B.B.; Kazakova, O.A.; Dovbiy, O.A.; Pogorelyi, V.K. Interactions between bioactive ferulic acid and fumed silica by UV–vis spectroscopy, FT–IR, TPD MS investigation and quantum chemical methods. J. Colloid Interface Sci. 2009, 339, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Kulik, T.V.; Lipkovska, N.O.; Barvinchenko, V.M.; Palyanytsya, B.B.; Kazakova, O.A.; Dudik, O.O.; Menyhárd, A.; László, K. Thermal transformation of bioactive caffeic acid on fumed silica seen by UV–Vis spectroscopy, thermogravimetric analysis, temperature programmed desorption mass spectrometry and quantum chemical methods. J. Colloid Interface Sci. 2016, 470, 132–141. [Google Scholar] [CrossRef] [PubMed]
- Kulik, T.V.; Barvinchenko, V.N.; Palyanytsya, B.B.; Lipkovska, N.A.; Dudik, O.O. Thermal transformations of biologically active derivatives of cinnamic acid by TPD MS investigation. J. Anal. Appl. Pyrol. 2011, 90, 219–223. [Google Scholar] [CrossRef]
- Kumar, N.; Pruthi, V.; Goel, N. Structural, thermal and quantum chemical studies of p-coumaric and caffeic acids. J. Mol. Struct. 2015, 1085, 242–248. [Google Scholar] [CrossRef]
- Davalos, J.Z.; Herrero, R.; Chana, A.; Guerrero, A.; Jiménez, P.; Santiuste, J.M. Energetics and structural properties, in the gas phase, of trans-hydroxycinnamic acids. J. Phys. Chem. A 2012, 116, 2261–2267. [Google Scholar] [CrossRef] [PubMed]
- Xing, Y.; Peng, H.; Zhang, M.; Li, X.; Zeng, W.; Yang, X. Caffeic acid product from the highly copper-tolerant plant Elsholtzia splendens post-phytoremediation: Its extraction, purification, and identification. J. Zhejiang Univ. Sci. B 2012, 13, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Sẃisłocka, R. Spectroscopic (FT-IR, FT-Raman, UV absorption, 1H and 13C NMR) and theoretical (in B3LYP/6-311++G** level) studies on alkali metal salts of caffeic acid. Spectrochim. Acta Part A 2013, 100, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Williams, P.A.M.; Baro, A.G.; Ferrer, E.G. Study of the interaction of oxovanadium(IV) with a plant component (caffeic acid). Synthesis and characterization of a solid compound. Polyhedron 2002, 21, 1979–1984. [Google Scholar] [CrossRef]
- Tošović, J. Spectroscopic feature s of caffeic acid: Theoretical study. Kragujev. J. Sci. 2017, 39, 99–108. [Google Scholar] [CrossRef]
- Tarasevich, B.N. IR Spectra of the Main Classes of Organic Molecule. Reference Materials; Moskow Lomonosov University: Moskow, Russia, 2012. (In Russian) [Google Scholar]
- Bellamy, L. Infra-Red Spectra of Complex Molecule; Methuen & Co LTD: London, UK, 1963. [Google Scholar]
- Świsłocka, R.; Kowczyk-Sadowy, M.; Kalinowska, M.; Lewandowski, W. Spectroscopic (FT-IR, FT-Raman, 1H and 13C NMR) and theoretical studies of p-coumaric acid and alkali metal p-coumarates. Spectroscopy 2012, 27, 35–48. [Google Scholar] [CrossRef]
- Brand, J.; Eglinton, G. Applications of Spectroscopy to Organic Chemistry; Oldbourne Press: London, UK, 1965. [Google Scholar]
- Ferrer, E.G.; Salinas, M.V.; Correa, M.G.; Vrdoljak, F.; Williams, P.A.M. ALP Inhibitors: Vanadyl(IV) Complexes of Ferulic and Cinnamic Acid. Z. Naturforsch. 2005, 60b, 305–311. [Google Scholar] [CrossRef]
- Kulyk, K.; Palianytsia, B.; Alexander, J.; Azizova, L.; Borysenko, M.; Larsson, M.; Kartel, M.; Kulik, T. Kinetics of Valeric Acid Ketonization and Ketenization in Catalytic Pyrolisis on Nanosized SiO2, γ-Al2O3 CeO2/SiO2, Al2O3/TiO2 and TiO2/Al2O3. ChemPhysChem 2017, 18, 1943–1955. [Google Scholar] [CrossRef] [PubMed]
- Palacios, E.G.; Juares-Lopes, G.; Monhemius, A.J. Infrared spectroscopy of metal carboxylates: II. Analysis of Fe(III), Ni and Zn carboxylate solutions. Hydrometallurgy 2004, 72, 139–148. [Google Scholar] [CrossRef]
- Stehfest, K.; Boese, M.; Kerns, G.; Piry, A.; Wilhelm, C. Fourier transform infrared spectroscopy as a new tool to determine rosmarinic acid in situ. J. Plant Physiol. 2004, 161, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Kharlampovich, G.D.; Churkin, Y.V. Phenolz; Khimiya Publ.: Moskow, Russia, 1974. (In Russian) [Google Scholar]
- Nakanishi, K. Infrared Adsorption Spectroscopy (Practical); Holden Day. Inc.: San Francisco, CA, USA, 1962. [Google Scholar]
- Kotorlenko, A.; Alexandrova, V.S. Spectral manifestations of change in electronic structure in phenol-phenolate anion-phenoxi radical series. Theor. Exp. Chem. 1982, 18, 115–118. [Google Scholar] [CrossRef]
- Śanchez-Cortés1, S.; Garćıa-Ramos, J.V. Adsorption and Chemical Modification of Phenols on a Silver Surface. J. Colloid. Interface Sci. 2000, 231, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Barton, D.; Ollis, W. Comprehensive Organic Chemistry: The Synthesis and Reactions of Organic Compounds, 3rd ed.; Oxygen, Compounds, Stoddart, J.F., Eds.; Pergamon Press: Oxford, UK, 1979; Volume 1. [Google Scholar]
- Pillar, E.A.; Zhou, R.; Guzman, M.I. Heterogeneous Oxidation of Catechol. J. Phys. Chem. A 2015, 119, 10349–10359. [Google Scholar] [CrossRef] [PubMed]
- Dai, G.; Zou, Q.; Wang, S.; Zhao, Y.; Zhu, L.; Huang, Q. Effect of Torrefaction on the Structure and Pyrolysis Behavior of Lignin. Energy Fuels 2018, 32, 4160–4166. [Google Scholar] [CrossRef]
- Evtuguin, D.V.; Rocha, G.; Goodfellow, B.J. Detection of muconic acid type structures in oxidised lignins using 2D NMR. Holzforschung 2009, 63, 675–680. [Google Scholar] [CrossRef]
- Fuchs, G.; Boll, M.; Heider, J. Microbial degradation of aromatic compounds—From one strategy to four. Nat. Rev. Microbiol. 2011, 9, 803–816. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Wang, J.; Yang, Y.; Zhang, Y.; Zhao, C.; Yu, Y.; Wang, S. Comparison of the thermal degradation behaviors and kinetics of palm oil waste under nitrogen and air atmosphere in TGA-FTIR with a complementary use of model-free and model-fitting approaches. J. Anal. Appl. Pyrolysis 2018, 134, 12–24. [Google Scholar] [CrossRef]
- Ma, Z.; Wanga, J.; Zhoua, H.; Zhanga, Y.; Yanga, Y.; Liua, X.; Yea, J.; Chenc, D.; Wangb, S. Relationship of thermal degradation behavior and chemical structure of lignin isolated from palm kernel shell under different process severities. Fuel Process. Technol. 2018, 181, 141–156. [Google Scholar] [CrossRef]
- Woodruff, D.; Delchar, T. Modern Techniques of Surface Science; Cambridge Solid State Science Series; Cambridge University Press: London, UK, 1986. [Google Scholar]
- Cvetanovic, R.J.; Amenomiya, Y. A temperature programmed desorption technique for investigation of practical catalysts. Catal. Rev. 1972, 6, 21–85. [Google Scholar] [CrossRef]
- Nicholl, S.I.; Talley, J.W. Development of thermal programmed desorption mass spectrometry methods for environmental applications. Chemosphere 2006, 63, 132–141. [Google Scholar] [CrossRef] [PubMed]
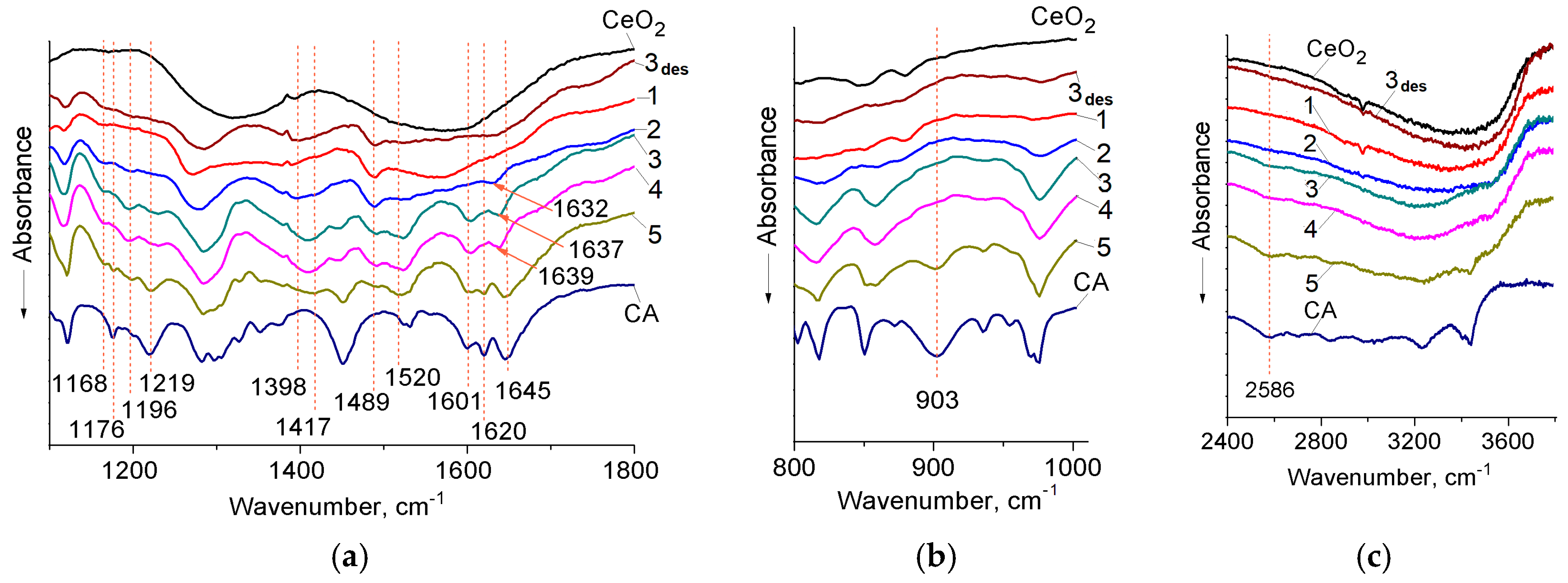
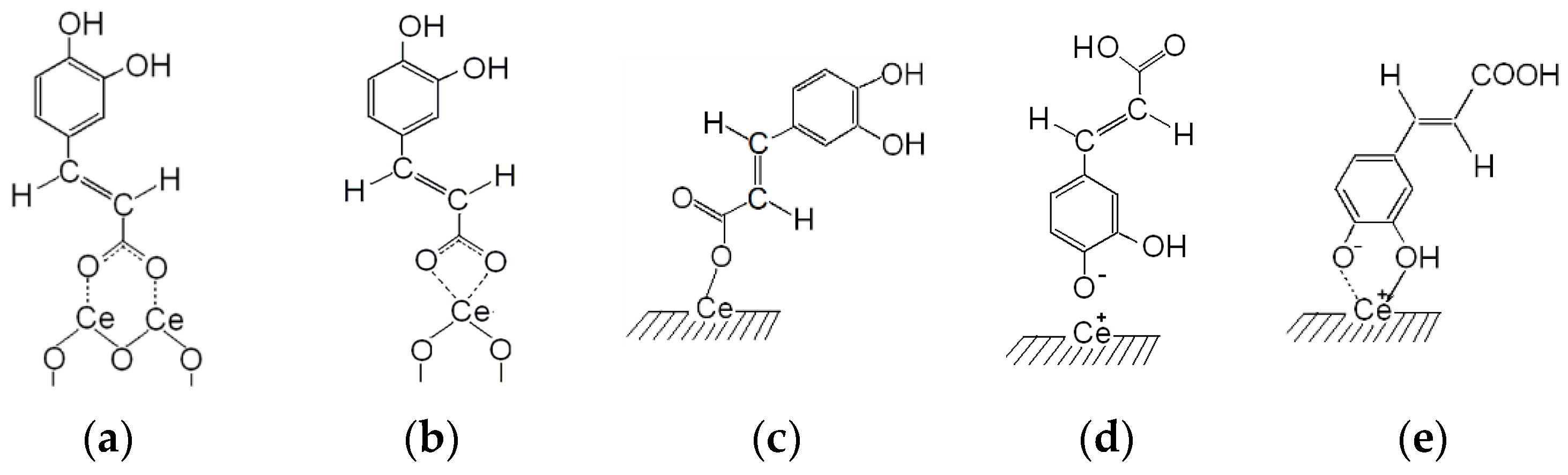
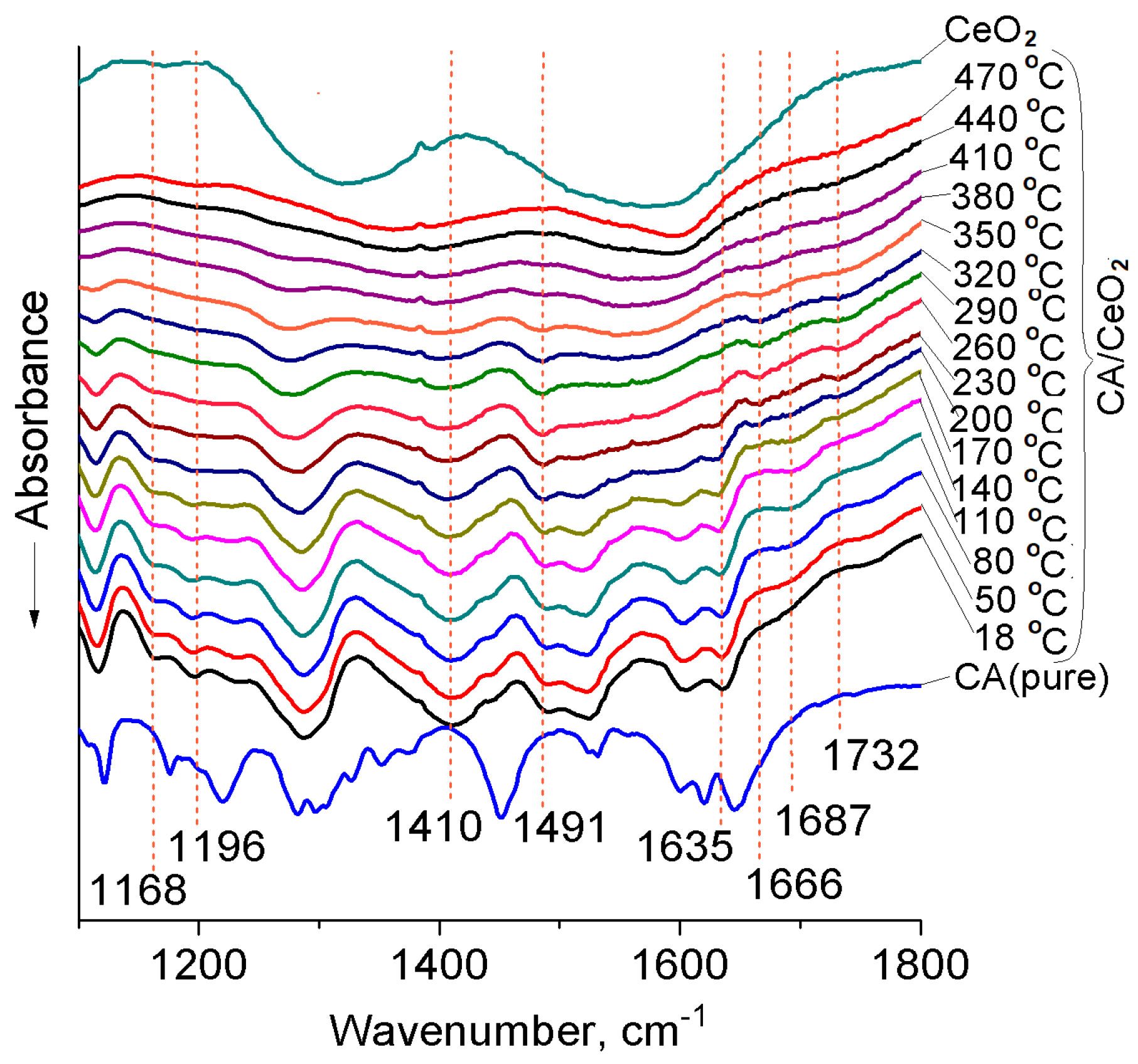
| CA | CA/CeO2 | Assignments 1 | Literary Data |
|---|---|---|---|
| Phenolic group | |||
| 903 | – | δCOH | 955–890 [45] |
| 1176 | – | βCOHar | 1180 [52], 1178 [53], |
| – | 1196 1166 | νCO– | 1195 [55] |
| 1219 | 1219 1230 2 | νCOHar | 1140–1230 [45], 1230 [53], 1200 [54] |
| 1282 | 1281 | νCOH | 1210–1320 [45,46,54] |
| Carboxylic group | |||
| 903 | – | δCOH | 955–890 [45] |
| – | |||
| – | 1396 | νC-O | |
| – | 1410 | νsCOO− | 1300–1400 [45], 1420–1470 [46] |
| – | 1417 | ||
| – | 1489 | νasCOO− | 1550–1610 [46], 1550–1650 [45,54] |
| – | 1520 | ||
| 1645 | 1632 | νC=O | 1645 [41,42,52] |
| 1620 | 1620 | νC=C | 1635 [48,49], 1626 [46] |
| 2500–2700 3 | 2500–2700 3 | νOH | 2500–2700 [46] |
| Stage | Temperature Range/Tmax (°C) | Volatiles (%) | Char (%) |
|---|---|---|---|
| I | 20–200 | 22.6 | - |
| II | 260 | 68.7 | - |
| Σ(I+II) | 91.3 | 8.7 |
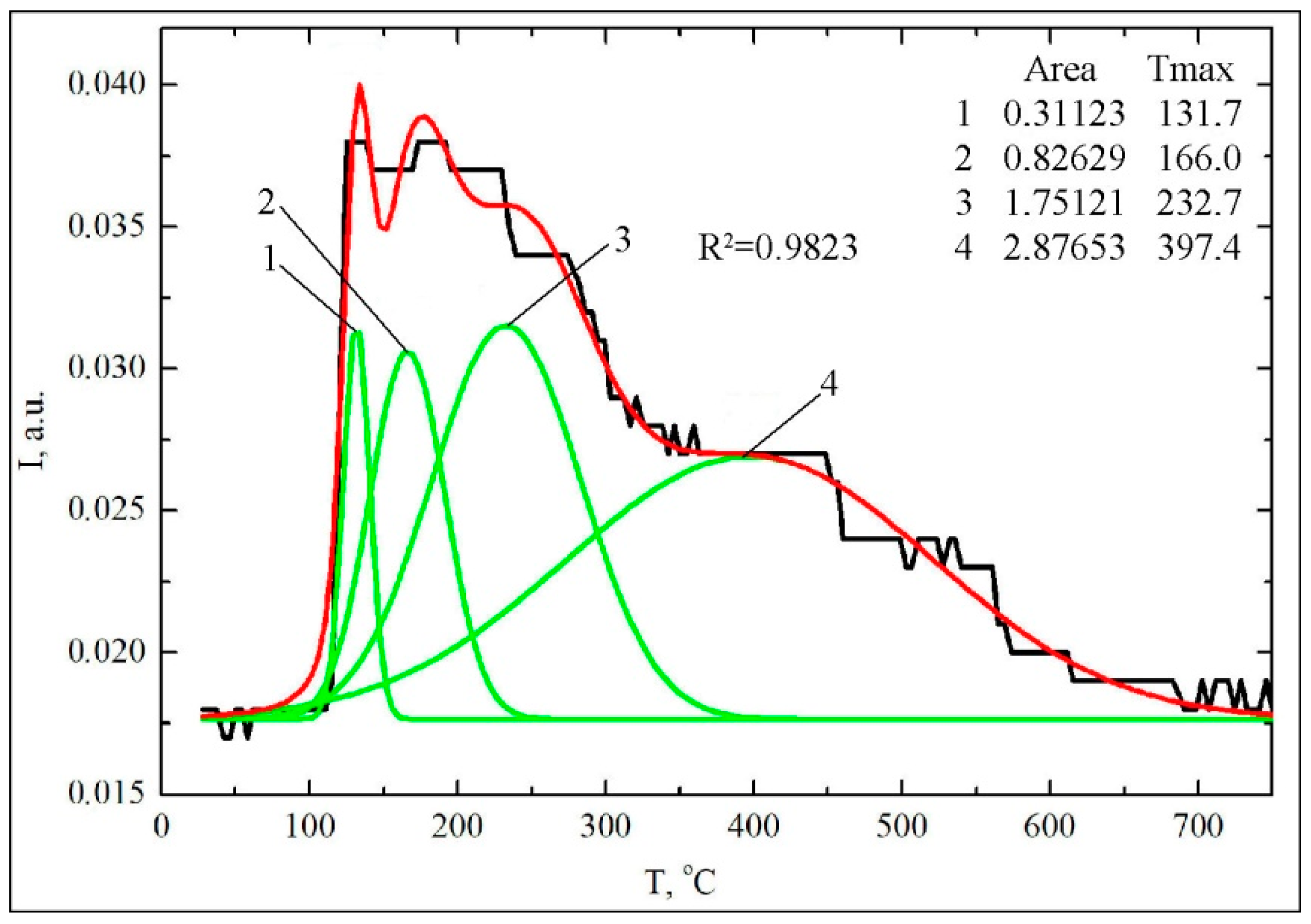
| No | Surface Complexes (SC) | m/z | Tmax (°C) | Scheme | Peak Area (a.u.) | % |
|---|---|---|---|---|---|---|
| -COOH | ||||||
| 1 | Hydrogen-bonded association | 136 | 132 | 2a | 0.311 | 2.6 |
| 2 | Dimers | 136 | 166 | 2a | 0.826 | 6.9 |
| 3 | Monodentate bonded complexes | 136 | 232 | 2b | 1.751 | 14.6 |
| 4 | Bidentate carboxylates | 136 | 397 | 2c | 2.877 | 24.0 |
| ΣSCCOOH = 5.8 | 39.0 | |||||
| -Ph(OH)2 | ||||||
| 5 | Phenolate complexes | 110 | 241 | 3 | 1.507 | 12.6 |
| 6 | Chelate phenolate complexes | 94 | 390 | 4 | 4.706 | 39.2 |
| ΣSCPhOH = 6.2 | 52.2 | |||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nastasiienko, N.; Palianytsia, B.; Kartel, M.; Larsson, M.; Kulik, T. Thermal Transformation of Caffeic Acid on the Nanoceria Surface Studied by Temperature Programmed Desorption Mass-Spectrometry, Thermogravimetric Analysis and FT–IR Spectroscopy. Colloids Interfaces 2019, 3, 34. https://doi.org/10.3390/colloids3010034
Nastasiienko N, Palianytsia B, Kartel M, Larsson M, Kulik T. Thermal Transformation of Caffeic Acid on the Nanoceria Surface Studied by Temperature Programmed Desorption Mass-Spectrometry, Thermogravimetric Analysis and FT–IR Spectroscopy. Colloids and Interfaces. 2019; 3(1):34. https://doi.org/10.3390/colloids3010034
Chicago/Turabian StyleNastasiienko, Nataliia, Borys Palianytsia, Mykola Kartel, Mats Larsson, and Tetiana Kulik. 2019. "Thermal Transformation of Caffeic Acid on the Nanoceria Surface Studied by Temperature Programmed Desorption Mass-Spectrometry, Thermogravimetric Analysis and FT–IR Spectroscopy" Colloids and Interfaces 3, no. 1: 34. https://doi.org/10.3390/colloids3010034
APA StyleNastasiienko, N., Palianytsia, B., Kartel, M., Larsson, M., & Kulik, T. (2019). Thermal Transformation of Caffeic Acid on the Nanoceria Surface Studied by Temperature Programmed Desorption Mass-Spectrometry, Thermogravimetric Analysis and FT–IR Spectroscopy. Colloids and Interfaces, 3(1), 34. https://doi.org/10.3390/colloids3010034




