Water Vapor Adsorption by Some Manganese Oxide Forms
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Phase Composition
3.2. Morphology
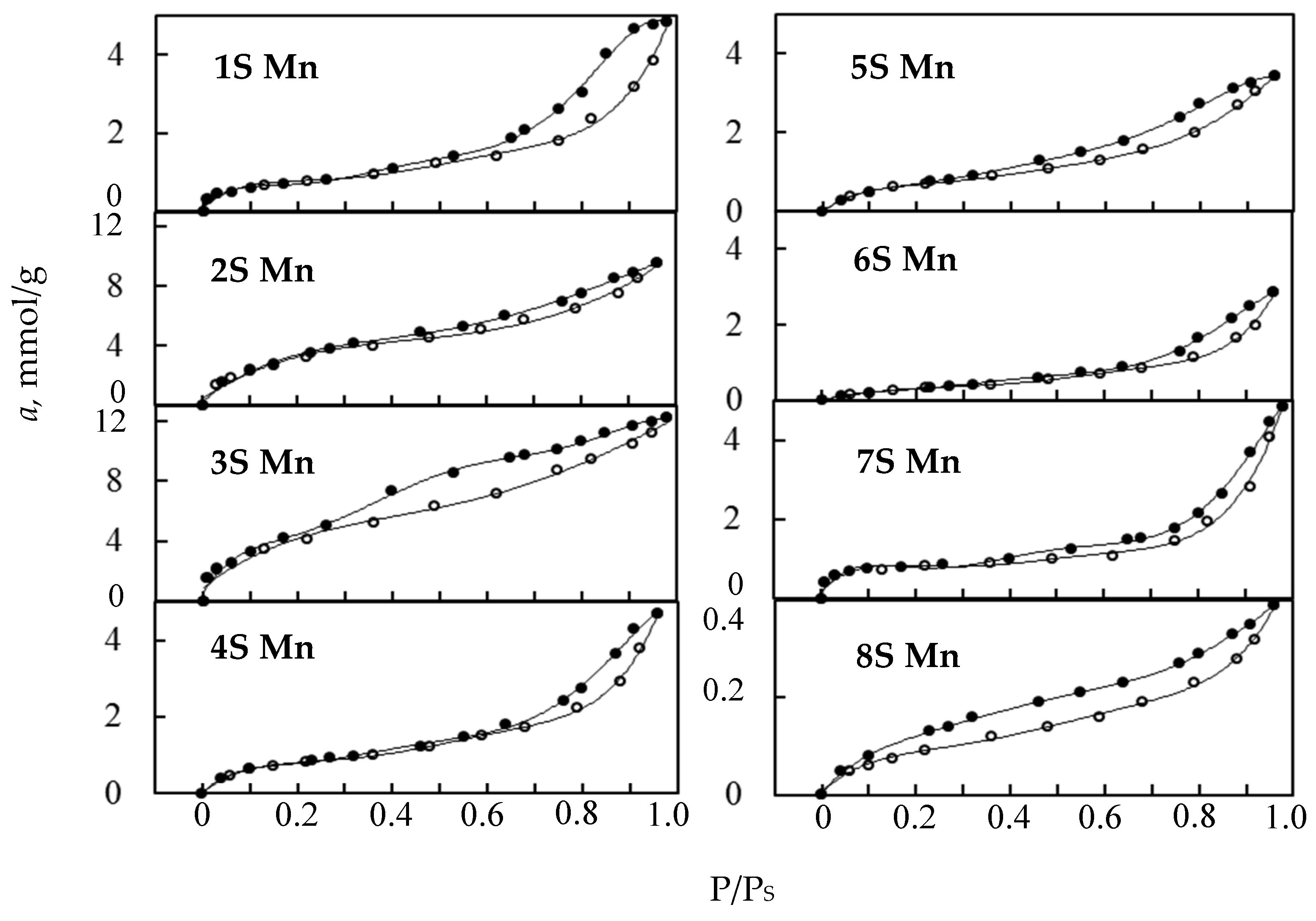
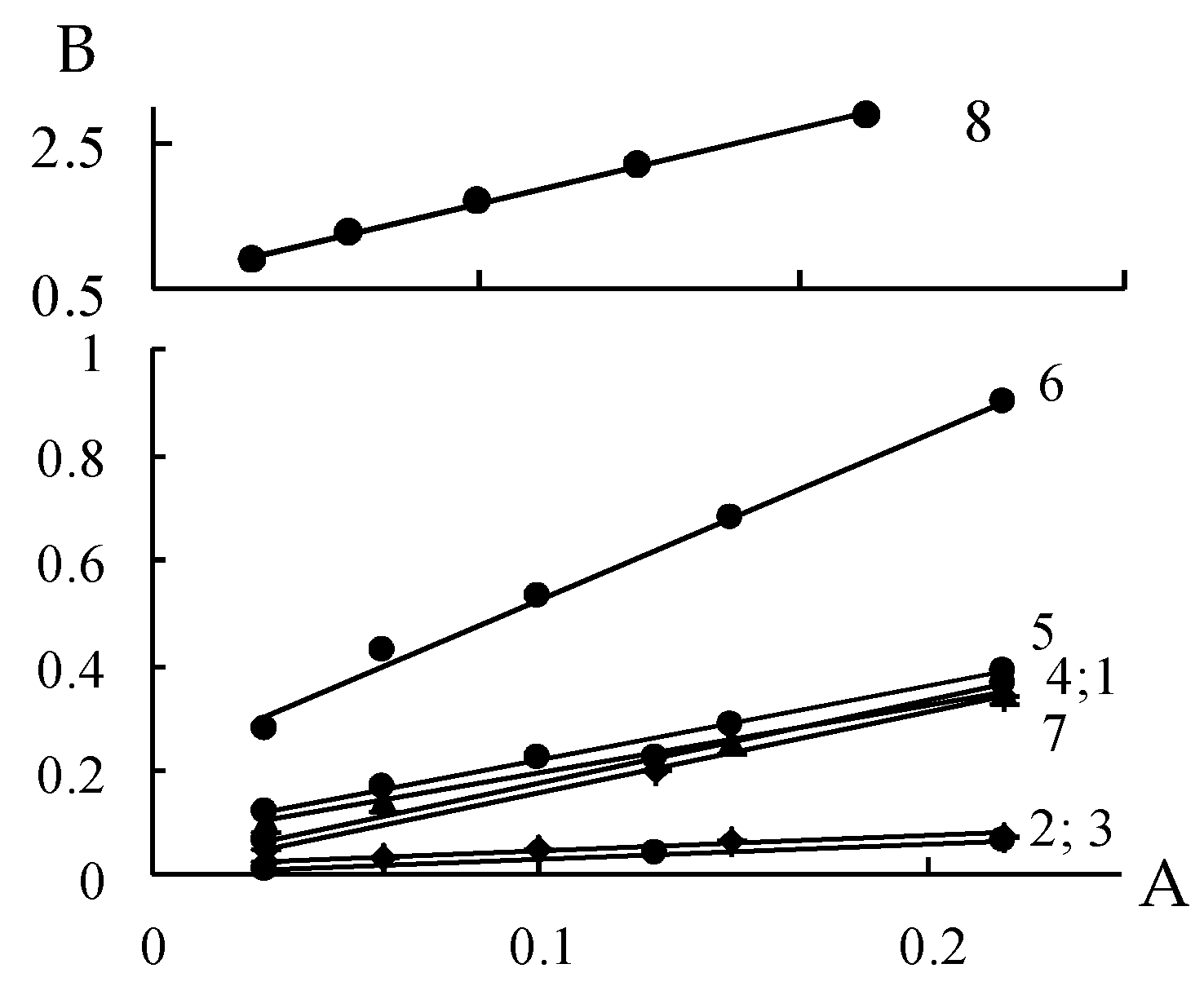
3.3. Water Vapor Sorption
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Jia, J.; Zhang, P.; Chen, L. Catalytic decomposition of gaseous ozone over manganese dioxides with different crystal structures. Appl. Catal. B 2016, 189, 210–218. [Google Scholar] [CrossRef]
- Wang, C.; Ma, J.; Liu, F.; He, H.; Zhang, R. The Effects of Mn2+ Precursors on the Structure and Ozone Decomposition Activity of Cryptomelane-Type Manganese Oxide (OMS-2) Catalysts. J. Phys. Chem. C 2015, 119, 23119–23126. [Google Scholar] [CrossRef]
- Ma, J.; Wang, C.; He, H. Transition metal doped cryptomelane-type manganese oxide catalysts for ozone decomposition. Appl. Catal. B 2017, 201, 503–510. [Google Scholar] [CrossRef]
- Yang, J.; Zhou, H.; Wang, L.; Zhang, Y.; Chen, C.; Hu, H.; Zhang, J. Cobalt-Doped K-OMS-2 Nanofibers: A Novel and Efficient Water-Tolerant Catalyst for the Oxidation of Carbon Monoxide. ChemCatChem 2017, 9, 1163–1167. [Google Scholar] [CrossRef]
- Carabineiro, S.A.; Santos, V.P.; Pereira, M.F.R.; Órfão, J.J.; Figueiredo, J.L. CO oxidation over gold supported on Cs, Li and Ti-doped cryptomelane materials. J. Colloid Interface Sci. 2016, 480, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Gao, C.; Shi, J.W.; Fan, Z.; Gao, G.; Niu, C. Sulfur and Water Resistance of Mn-Based Catalysts for Low-Temperature Selective Catalytic Reduction of NOx: A Review. Catalysts 2018, 8, 11. [Google Scholar] [CrossRef]
- Huang, W.; Shi, J. Water-promoted low-concentration NO removal at room temperature by Mg-doped manganese oxides OMS-2. Appl. Catal. A 2015, 507, 65–74. [Google Scholar] [CrossRef]
- Selvakumar, S.; Nuns, N.; Trentesaux, M.; Batra, V.S.; Giraudon, J.-M.; Lamonier, J.-F. Reaction of formaldehyde over birnessite catalyst: A combined XPS and ToF-SIMS study. Appl. Catal. B Environ. 2018, 223, 192–200. [Google Scholar] [CrossRef]
- Luo, J.; Zhang, Q.; Huang, A.; Suib, S.L. Total oxidation of volatile organic compounds with hydrophobic cryptomelane-type octahedral molecular sieves. Micropor. Mesopor. Mater. 2000, 35, 209–217. [Google Scholar] [CrossRef]
- Burtch, N.C.; Jasuja, H.; Walton, K.S. Water stability and adsorption in metal-organic frameworks. Chem. Rev. 2014, 114, 10575–10612. [Google Scholar] [CrossRef] [PubMed]
- Ng, E.P.; Mintova, S. Nanoporous materials with enhanced hydrophilicity and high water sorption capacity. Micropor. Mesopor. Mater. 2008, 114, 1–26. [Google Scholar] [CrossRef]
- Naono, H.; Hakuman, M. Analysis of adsorption isotherms of water vapor for nonporous and porous adsorbents. J. Colloid Interface Sci. 1991, 145, 405–412. [Google Scholar] [CrossRef]
- Muttakin, M.; Mitra, S.; Thu, K.; Ito, K.; Saha, B.B. Theoretical framework to evaluate minimum desorption temperature for IUPAC classified adsorption isotherms. Int. J. Heat Mass Transfer 2018, 122, 795–805. [Google Scholar] [CrossRef]
- Luo, J.; Zhang, Q.; Garcia-Martinez, J.; Suib, S.L. Adsorptive and acidic properties, reversible lattice oxygen evolution, and catalytic mechanism of cryptomelane-type manganese oxides as oxidation catalysts. J. Am. Chem. Soc. 2008, 130, 3198–3207. [Google Scholar] [CrossRef] [PubMed]
- Gregg, S.J.; Sing, K.S.W. Adsorption, Surface Area and Porosity; Academic Press: New York, NY, USA, 1982; ISBN 0-12-300956-1. [Google Scholar]
- Suib, S.L. Structure, porosity, and redox in porous manganese oxide octahedral layer and molecular sieve materials. J. Mater. Chem. 2008, 18, 1623–1631. [Google Scholar] [CrossRef]
- Gac, W. The influence of silver on the structural, redox and catalytic properties of the cryptomelane-type manganese oxides in the low-temperature CO oxidation reaction. Appl. Catal. B 2007, 75, 107–117. [Google Scholar] [CrossRef]
- Wang, Z.M.; Tezuka, S.; Kanoh, H. Gaseous molecular sieving property of a microporous hollandite-type hydrous manganese oxide. Chem. Lett. 2000, 29, 560–561. [Google Scholar] [CrossRef]
- Lee, D.G.; Yang, C.M.; Kim, B.H. Enhanced electrochemical properties of boron functional groups on porous carbon nanofiber/MnO2 materials. J. Electroanal. Chem. 2017, 788, 192–197. [Google Scholar] [CrossRef]
- Tatlier, M.; Munz, G.; Henninger, S.K. Relation of water adsorption capacities of zeolites with their structural properties. Micropor. Mesopor. Mater. 2018, 788, 192–197. [Google Scholar] [CrossRef]
- Zhu, B.; Xu, Z.; Wang, C.; Gao, Y. Shape evolution of metal nanoparticles in water vapor environment. Nano Lett. 2016, 16, 2628–2632. [Google Scholar] [CrossRef] [PubMed]
- Rakitskaya, T.L.; Khitrich, V.F.; Raskola, L.A.; Makordey, F.V.; Sirovetnik, O.V. Decomposition of ozone microconcentrations by fine-dispersed MnO2 catalyst. Herald ONU(Ukr). Chemistry 2004, 9, 117–124. [Google Scholar]
- Karyakin, Y.V.; Angelov, I.I. Pure Chemical Substances. M. Chem. 1974, 407. [Google Scholar]
- DeGuzman, R.N.; Shen, Y.F.; Neth, E.J.; Suib, S.L.; O’Young, C.L.; Levine, S.; Newsam, J.M. Synthesis and characterization of octahedral molecular sieves (OMS-2) having the hollandite structure. Chem. Mater. 1994, 6, 815–821. [Google Scholar] [CrossRef]
- Zheng, H.; Feng, C.; Kim, S.-J.; Yin, S.; Wu, H.; Wang, S.; Li, S. Synthesis and electrochemical properties of KMn8O16 nanorods for Lithium ion batteries. Electrochim. Acta 2013, 88, 225–230. [Google Scholar] [CrossRef]
- Tian, H.; He, J.; Liu, L.; Wang, D. Effects of textural parameters and noble metal loading on the catalytic activity of cryptomelane-type manganese oxides for formaldehyde oxidation. Ceram. Int. 2013, 39, 315–321. [Google Scholar] [CrossRef]
- Sul, N.; Duan, Y.; Jiao, X.; Chen, D. Large-scale preparation and catalytic properties of one-dimensional α/β-MnO2 nanostructures. J. Phys. Chem. 2009, 113, 8560–8565. [Google Scholar] [CrossRef]
- Rakitskaya, T.L.; Truba, A.S.; Ennan, A.A.; Dlubovskii, R.M.; Volkova, V.Y. Water vapour adsorption by nanostructured polyphase compositions based on the solid component of welding aerosol. Adsorpt. Sci. Technol. 2017, 35, 389–395. [Google Scholar] [CrossRef]
- Musil, M.; Choi, B.; Tsutsumi, A. Morphology and Electrochemical Properties of α-, β-, γ-, and δ-MnO2 Synthesized by Redox Method. J. Electrochem. Soc. 2015, 162, A2058–A2065. [Google Scholar] [CrossRef]
- Prélot, B.; Villiéras, F.; Pelletier, M.; Razafitianamaharavo, A.; Thomas, F.; Poinsignon, C. Structural–chemical disorder of manganese dioxides: II. Influence on textural properties. J. Colloid Interface Sci. 2003, 264, 343–353. [Google Scholar] [CrossRef]
- Tombácz, E.; Hajdú, A.; Illés, E.; László, K.; Garberoglio, G.; Jedlovszky, P. Water in contact with magnetite nanoparticles, as seen from experiments and computer simulations. Langmuir 2009, 25, 13007–13014. [Google Scholar] [CrossRef] [PubMed]
- Foote, H.W.; Dixon, J.K. The adsorption of water and benzene vapors by manganese dioxide. J. Am. Chem. Soc. 1930, 52, 2170–2179. [Google Scholar] [CrossRef]
- Genuino, H.C.; Dharmarathna, S.; Njagi, E.C.; Mei, M.C.; Suib, S.L. Gas-phase total oxidation of benzene, toluene, ethylbenzene, and xylenes using shape-selective manganese oxide and copper manganese oxide catalysts. J. Phys. Chem. 2012, 116, 12066–12078. [Google Scholar] [CrossRef]
Sample Availability: Not Available. |
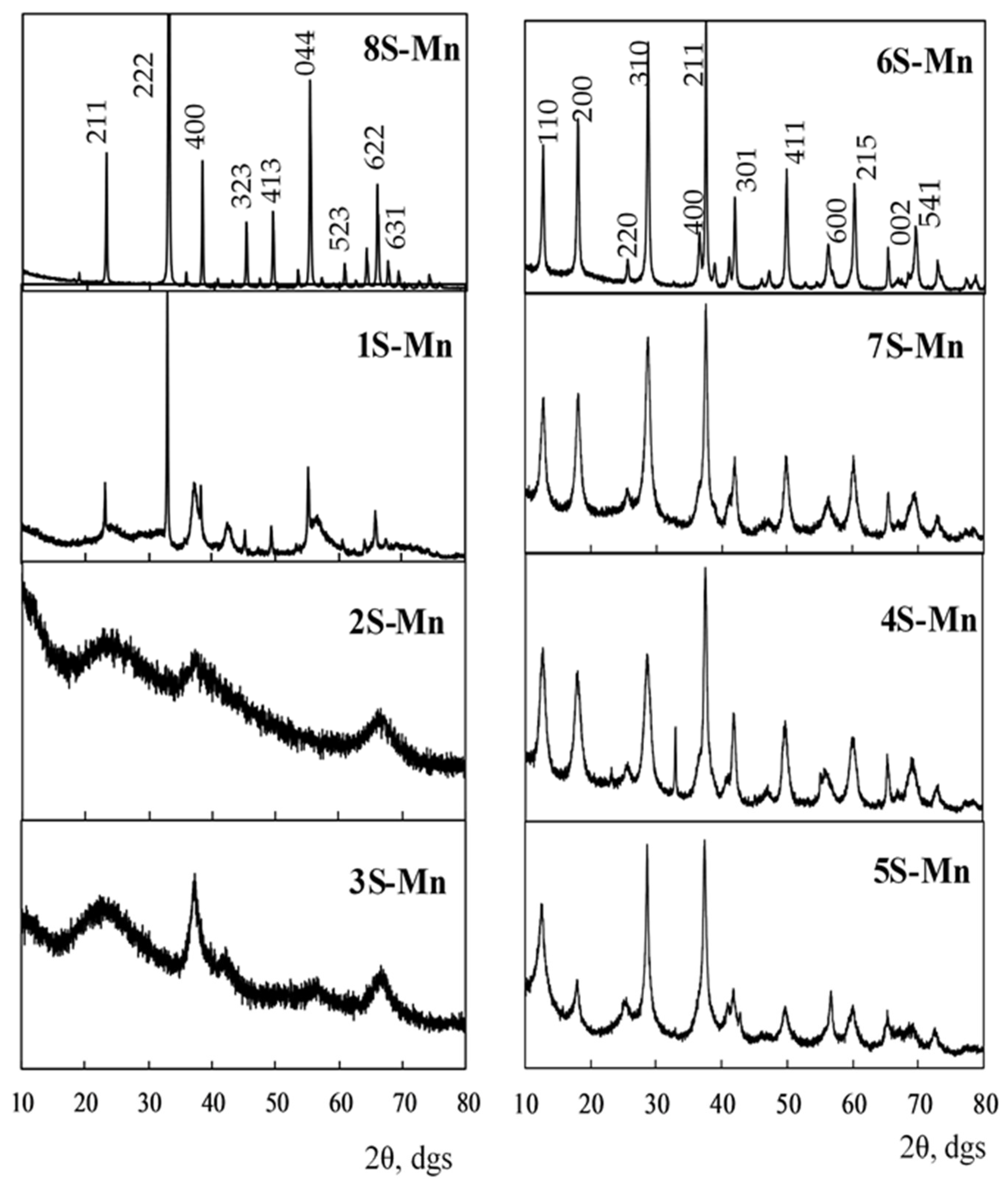

| Sample | Phase | Phase Content (wt.%) | Crystallite Size (nm) |
|---|---|---|---|
| 1S-Mn | β-Mn2O3 (bixbyite) | 30.0 (3) | 87 |
| β-MnO2 (pyrolusite) | 48.7 (6) | 2 | |
| ε-MnO2 (akhtenskite) | 19.6 (3) | 7 | |
| 2S-Mn | ε-MnO2 (akhtenskite) | 97.0 (1) | 3 |
| 3S-Mn | ε-MnO2 (akhtenskite) | 97.0 (1) | 3 |
| 4S-Mn | α-MnO2 (cryptomelane) | 90.5 (3) | 15 |
| β-Mn2O3 (bixbyite) | 9.49 (2) | 66 | |
| 5S-Mn | α-MnO2 (cryptomelane) | 85.3 (4) | 16 |
| β-MnO2 (pyrolusite) | 14.72 (4) | 28 | |
| 6S-Mn | α-MnO2 (cryptomelane) | 100.0 (4) | 36 |
| 7S-Mn | α-MnO2 (cryptomelane) | 100.0 (1) | 14 |
| 8S-Mn | β-Mn2O3 (bixbyite) | 100.0 (5) | 66 |
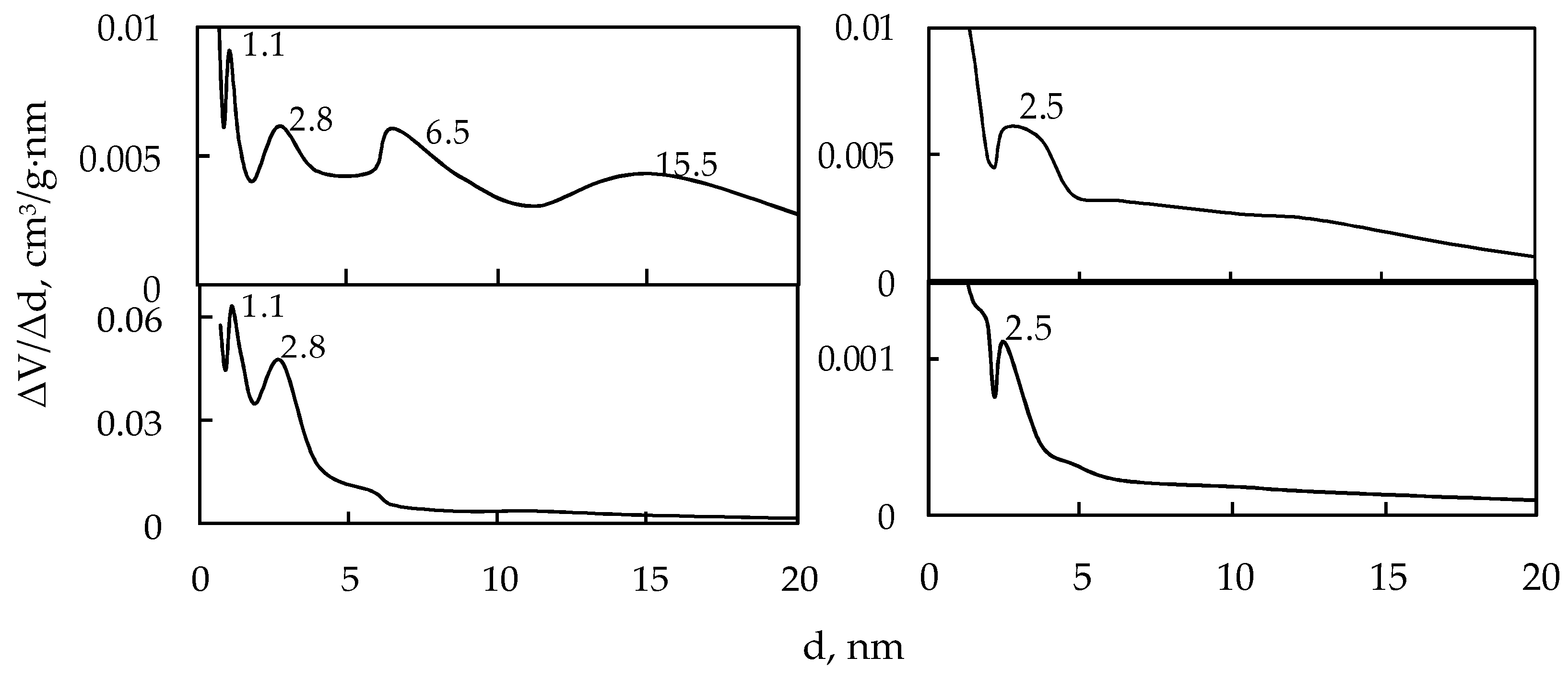
| Sample | Phase (Content in wt.%) | аm (mmol/g) | С | Ssp (m2/g) | Maxima of Pore Diameter Distribution Curves (nm) | |
|---|---|---|---|---|---|---|
| 10.6 Å2 | 14.8 Å2 | |||||
| 1S-Mn | β-Mn2O3 (30) ε- | 0.62 | 110.4 | 40.3 | 56.3 | 1.1; 2.8; 6.5; 15.5 |
| β-MnO2 (48.7) | ||||||
| MnO2 (19.6) | ||||||
| 2S-Mn | ε-MnO2 (97.0) | 2.43 | 15.1 | 158.0 | 216.5 | 2.0; 2.5; 6.4 |
| semicrystalline | ||||||
| 3S-Mn | ε-MnO2 (97.0) | 3.43 | 62.0 | 223.0 | 311.4 | 1.1; 2.8 |
| semicrystalline | ||||||
| 4S-Mn | α-MnO2 (90.5) | 0.78 | 19.6 | 44.5 | 69.9 | 2.0; 4.8 |
| Mn2O3 (9.5) | ||||||
| 5S-Mn | α-MnO2 (85.3) | 0.70 | 16.6 | 46.0 | 63.0 | 3.7 |
| β-MnO2 (14.7) | ||||||
| 6S-Mn | α-MnO2 (100.0) | 0.29 | 16.5 | 19.0 | 26.0 | 2.5 |
| 7S-Mn | α-MnO2 (100.0) | 0.65 | 243.4 | 42.4 | 59.2 | 4.0; 11.3 |
| 8S-Mn | β-Mn2O3 (100.0) | 0.07 | 30.3 | 5.0 | 6.9 | 2.5 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rakitskaya, T.; Truba, A.; Dzhyga, G.; Nagaevs’ka, A.; Volkova, V. Water Vapor Adsorption by Some Manganese Oxide Forms. Colloids Interfaces 2018, 2, 61. https://doi.org/10.3390/colloids2040061
Rakitskaya T, Truba A, Dzhyga G, Nagaevs’ka A, Volkova V. Water Vapor Adsorption by Some Manganese Oxide Forms. Colloids and Interfaces. 2018; 2(4):61. https://doi.org/10.3390/colloids2040061
Chicago/Turabian StyleRakitskaya, Tatyana, Alla Truba, Ganna Dzhyga, Anna Nagaevs’ka, and Vitaliya Volkova. 2018. "Water Vapor Adsorption by Some Manganese Oxide Forms" Colloids and Interfaces 2, no. 4: 61. https://doi.org/10.3390/colloids2040061
APA StyleRakitskaya, T., Truba, A., Dzhyga, G., Nagaevs’ka, A., & Volkova, V. (2018). Water Vapor Adsorption by Some Manganese Oxide Forms. Colloids and Interfaces, 2(4), 61. https://doi.org/10.3390/colloids2040061





