Supplementary Cementitious Material from Epsom Salt Production Waste
Abstract
1. Introduction
2. Materials and Methods
3. Results
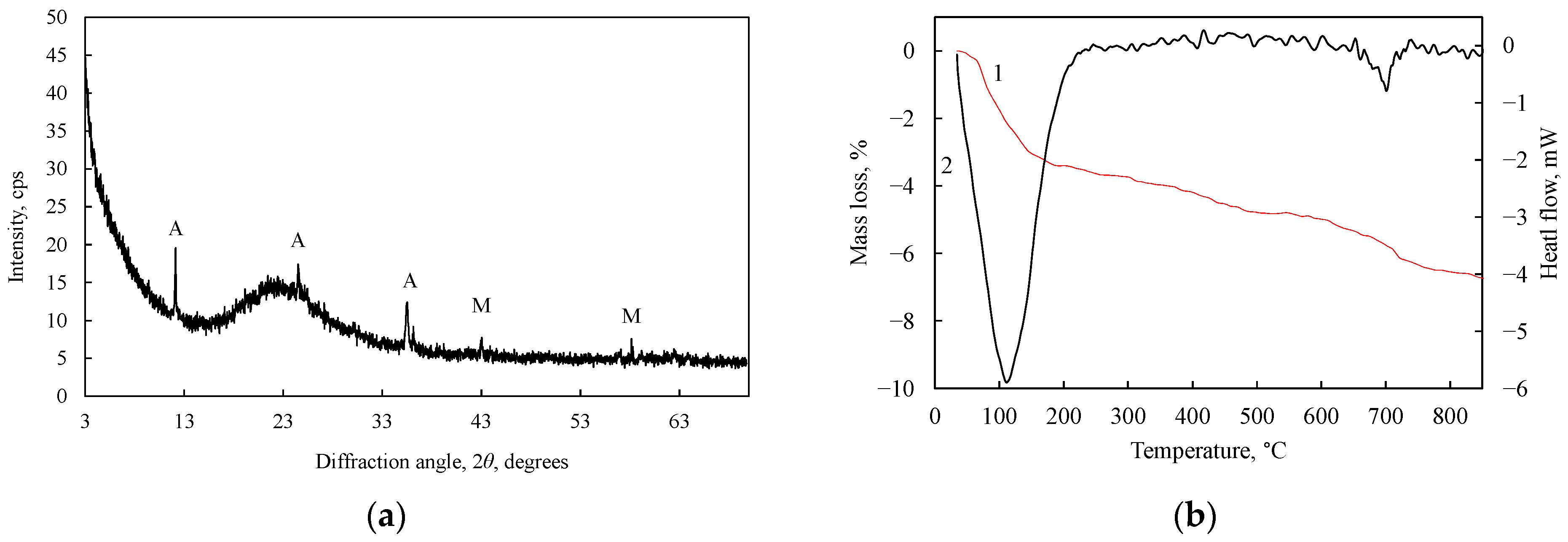
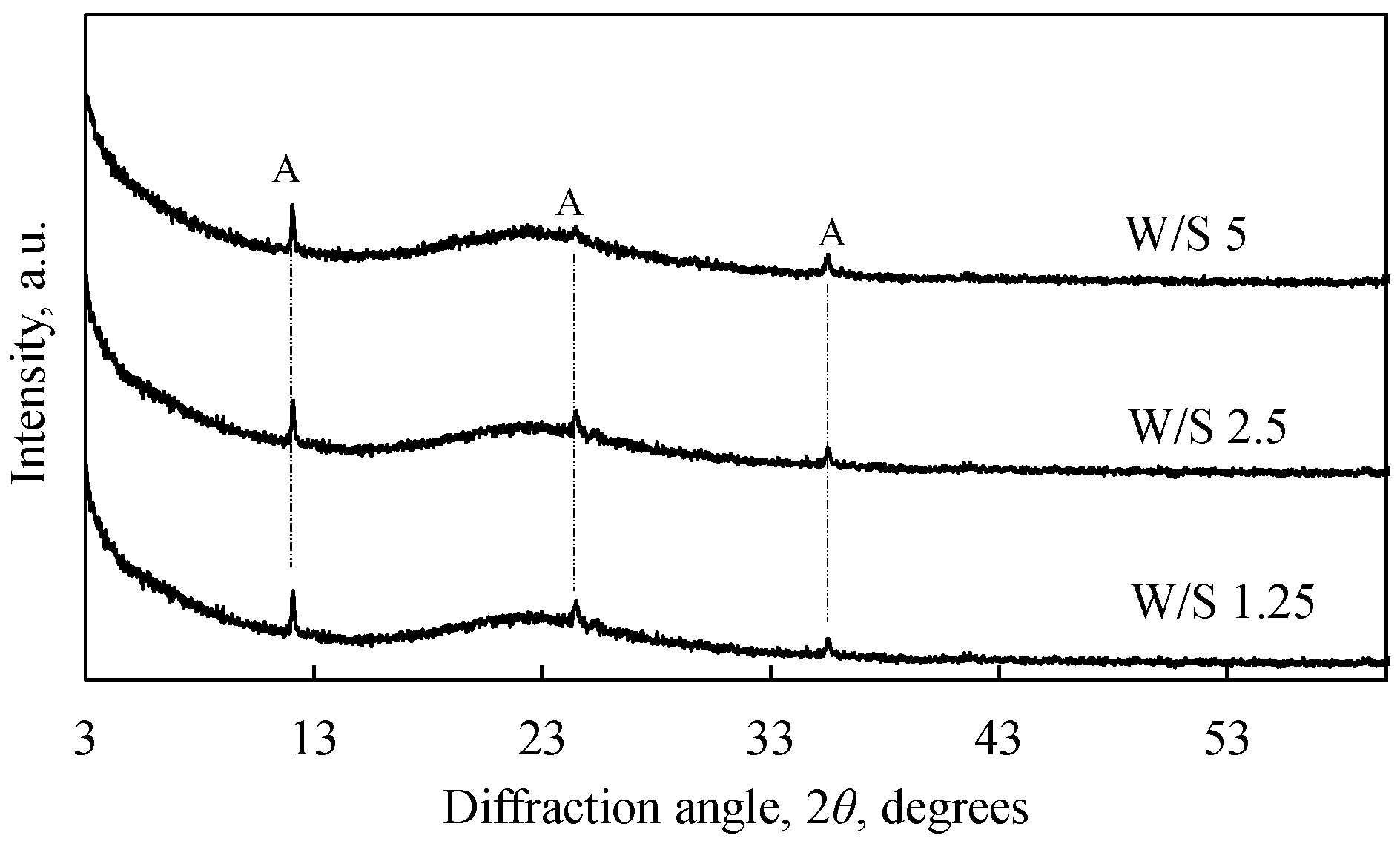
3.1. Neutralization of ESW
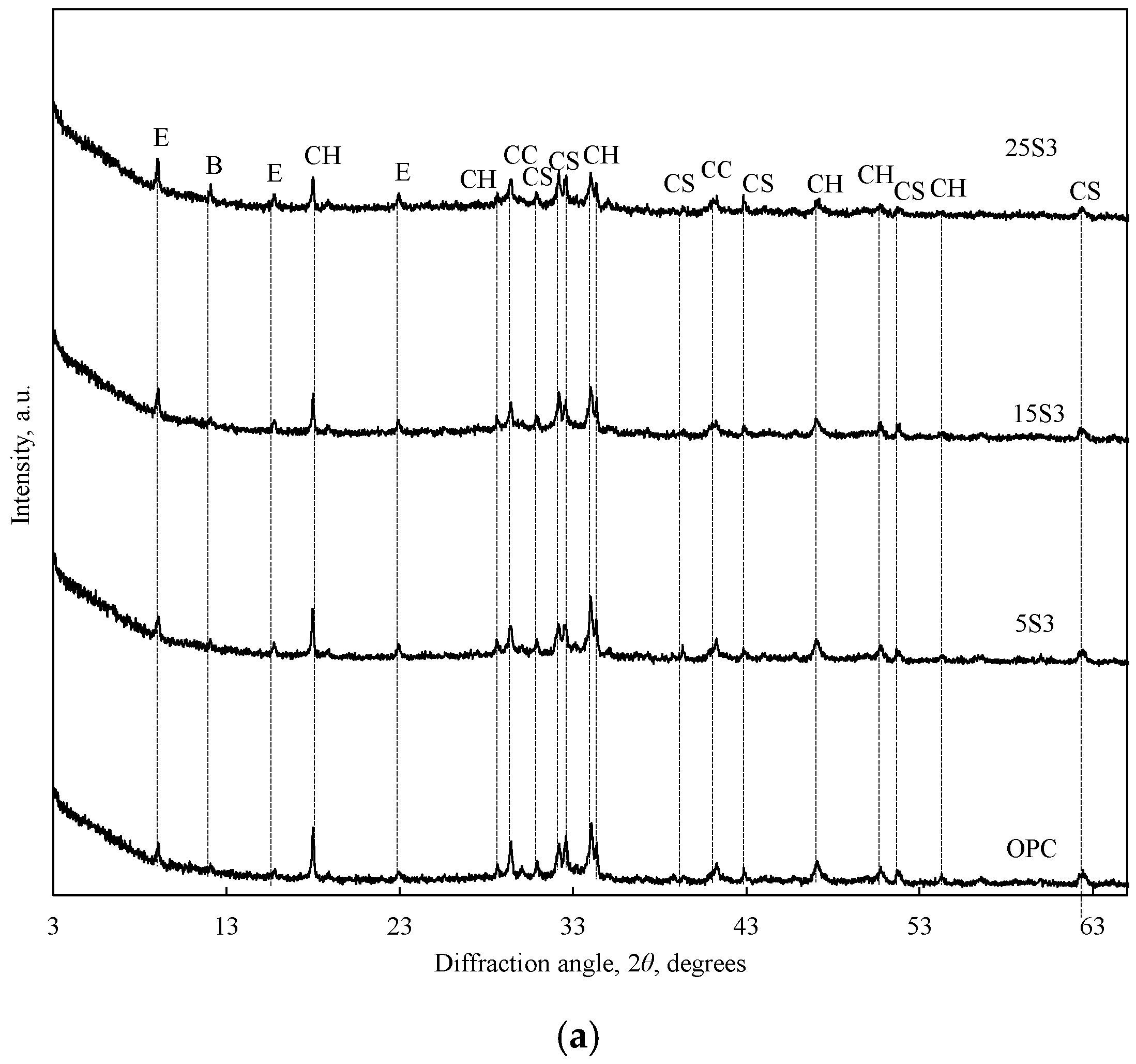
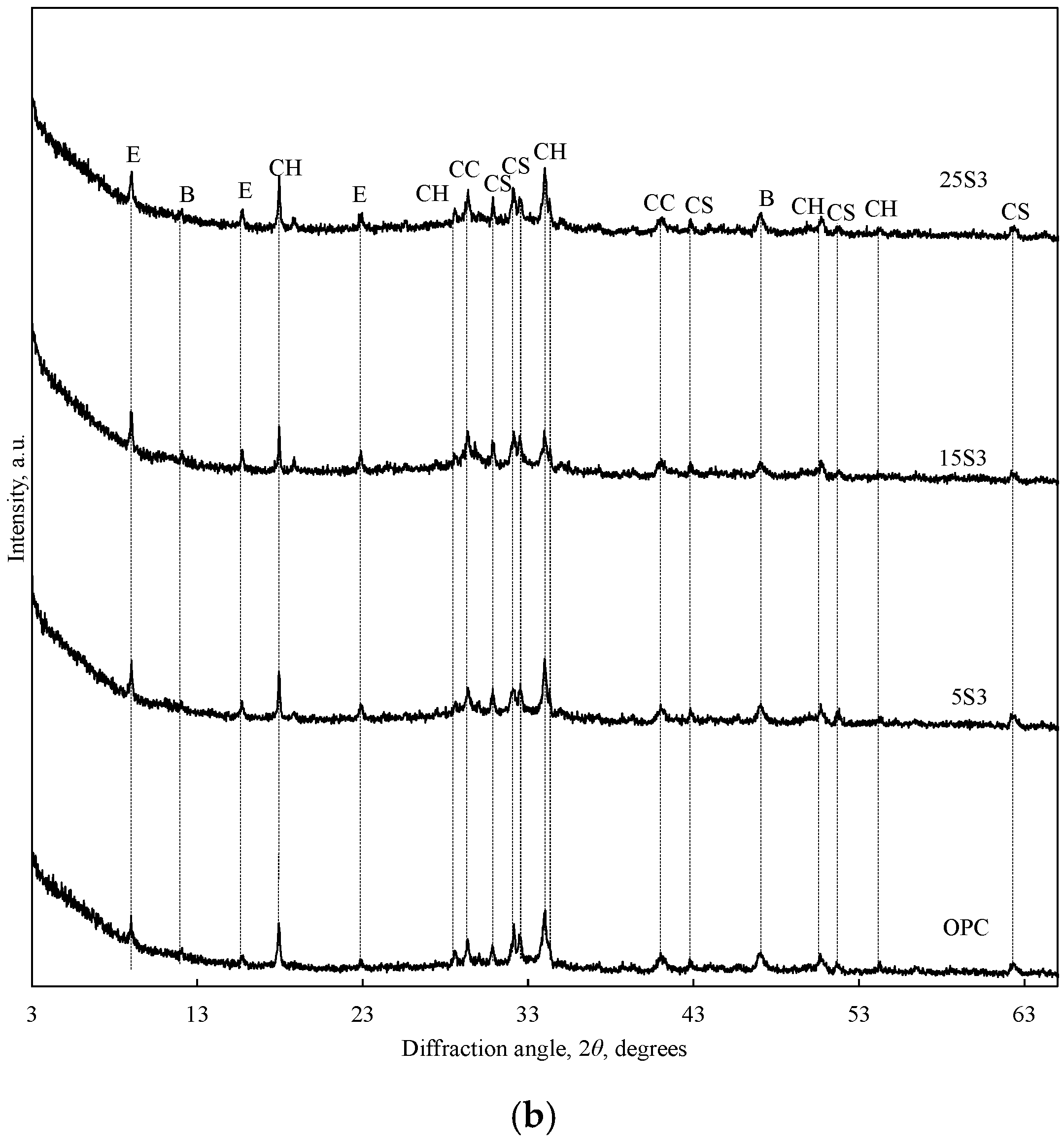
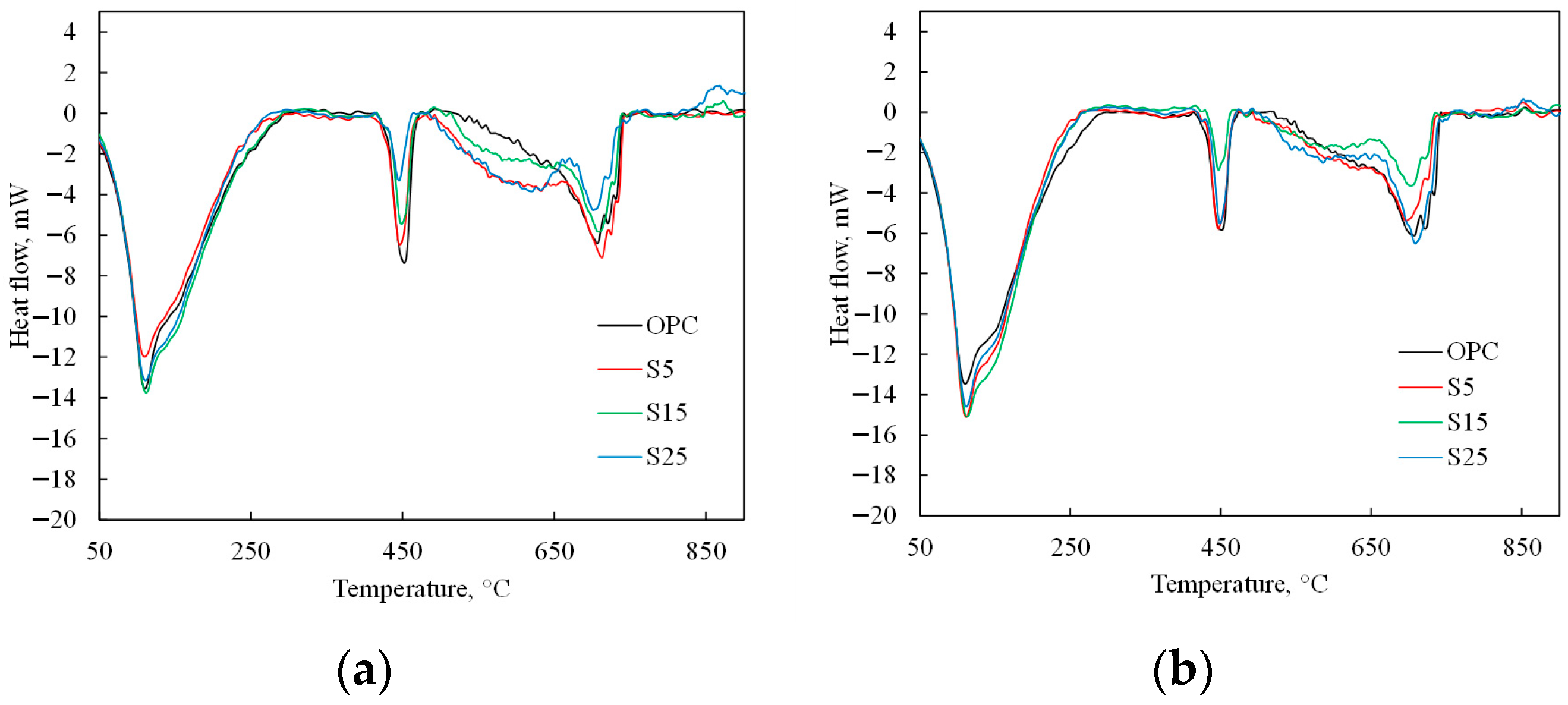
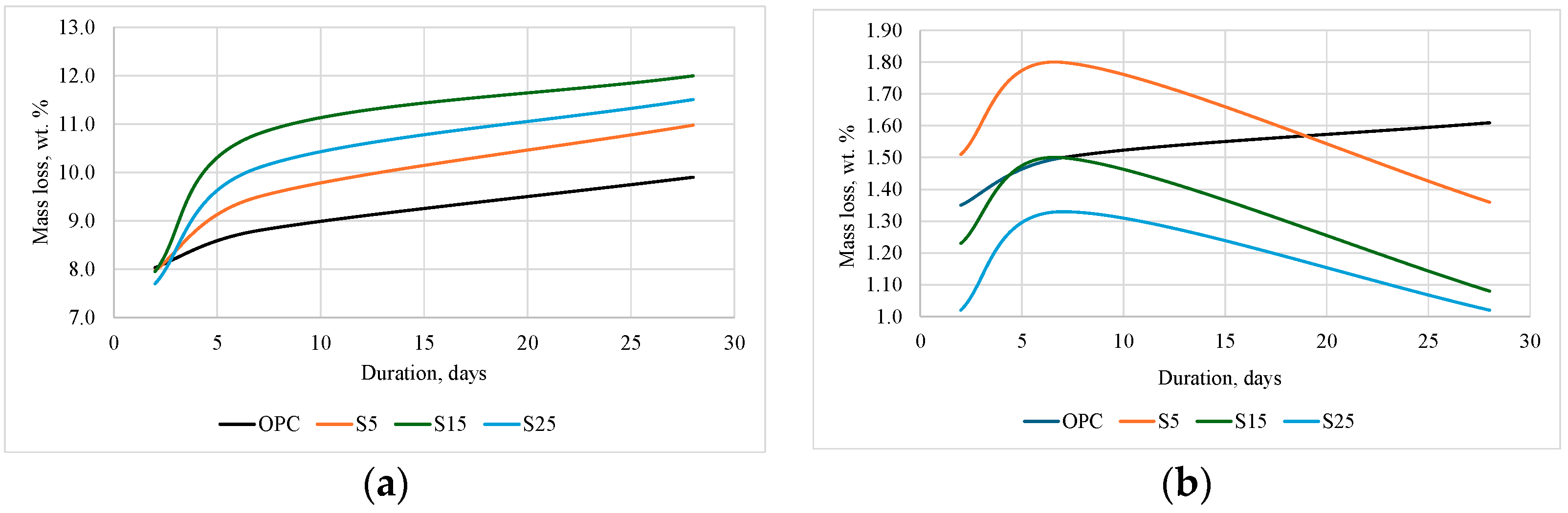
3.2. Influence of ESW on OPC Characteristics
4. Conclusions
- Epsom salt production waste consists of irregularly shaped sharp-edged amorphous materials with crystalline antigorite and magnetite impurities. It is an acidic material with a pH of 3.47.
- ESW can be neutralized with lime milk, and the water/solid material (W/S) ratio does not have a decisive influence on the neutralization process. After neutralization, the ESW becomes a non-acidic material with a pH value of 7.5, because during neutralization, the acidic components are bound to neutral compounds that do not settle on the surface of the ESW material, but pass into the liquid medium.
- Neutralized Epsom salt production waste (NESW) is characterized by very high pozzolanic activity (1085 mg CaO/g). NESW results in a slight increase in water consumption to achieve a normal consistency in cement pastes and a modest extension of the setting time of Portland cement paste.
- The addition of NESW accelerates the initial hydration of Portland cement and induces a strong pozzolanic reaction, clearly observable after 28 days of hydration.
- NESW is a very effective supplementary cementitious material, and can replace as much as 25 wt.% of Portland cement without reducing the strength class of Portland cement.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| OPC | Ordinary Portland cement |
| SCM | Supplementary cementitious materials |
| C–S–H | Calcium silicate hydrates |
| ESW | Epsom salt production waste |
| DSC | Differential scanning calorimetry |
| TG | Thermogravimetric analysis |
| STA | Simultaneous thermal analysis |
| XRD | X-ray diffraction analysis |
| XRF | X-ray fluorescence analysis |
| SEM | Scanning electron microscope |
| IC | Isothermal calorimetry analysis |
| NESW | Neutralized Epsom salt production waste |
| W/S | water/solid material ratio |
| C3S | Calcium silicates |
| C4AF | Brownmillerite |
References
- Ige, O.E.; Kabeya, M. Decarbonizing the Cement Industry: Technological, Economic, and Policy Barriers to CO2 Mitigation Adoption. Clean Technol. 2025, 7, 85. [Google Scholar] [CrossRef]
- Li, G.; Zhou, C.; Ahmad, W.; Usanova, K.I.; Karelina, M.; Mohamed, A.M.; Khallaf, R. Fly Ash Application as Supplementary Cementitious Material: A Review. Materials 2022, 15, 2664. [Google Scholar] [CrossRef]
- Chen, B.; Perumal, P.; Illikainen, M.; Ye, G. A Review on the Utilization of Municipal Solid Waste Incineration (MSWI) Bottom Ash as a Mineral Resource for Construction Materials. J. Build. Eng. 2023, 71, 106386. [Google Scholar] [CrossRef]
- Hansted, F.A.S.; Mantegazini, D.Z.; Ribeiro, T.M.; Gonçalves, C.E.C.; Balestieri, J.A.P. A mini-review on the use of waste in the production of sustainable Portland cement composites. Waste Manag. Res. 2022, 41, 828–838. [Google Scholar] [CrossRef] [PubMed]
- Ndahirwa, D.; Zmamou, H.; Lenormand, H.; Leblanc, N. The role of supplementary cementitious materials in hydration, durability and shrinkage of cement-based materials, their environmental and economic benefits: A review. Clean. Mater. 2022, 5, 100123. [Google Scholar] [CrossRef]
- Ma, M.; Tam, V.W.Y.; Le, K.N.; Osei-Kyei, R. Factors affecting the price of recycled concrete: A critical review. J. Build. Eng. 2022, 46, 103743. [Google Scholar] [CrossRef]
- Al-Hellali, N.; Bengtsson, M.; Nagy, A.; Sadagopan, M. Glass waste as a supplementary cementitious material in climate reduced concrete—A review. Nord. Concr. Res. 2025, 72, 167–181. [Google Scholar] [CrossRef]
- Xiong, Y.; Zhang, A.; Zhao, Y.; Xu, Q.; Ding, Y. A mini review on sewage sludge and red mud recycling for thermal energy storage. Energies 2024, 17, 2079. [Google Scholar] [CrossRef]
- Fode, T.A.; Jande, Y.A.C.; Kivevele, T. Effects of different supplementary cementitious materials on durability and mechanical properties of cement composite–comprehensive review. Heliyon 2023, 9, e17924. [Google Scholar] [CrossRef]
- Ahmed, A. Assessing the effects of supplementary cementitious materials on concrete properties: A review. Discov. Civ. Eng. 2024, 1, 145. [Google Scholar] [CrossRef]
- Gao, S.; Chu, H.; Shi, W.; Wang, F.; Jiang, J. Utilization of municipal solid waste incineration fly ash in ecological concrete and pavement bricks: Mechanical properties and environmental impact. Case Stud. Constr. Mater. 2024, 21, e03999. [Google Scholar] [CrossRef]
- Kaminskas, R.; Barauskas, I. Influence of pozzolana on sulfate attack of cement stone affected by chloride ions. Mater. Struct. 2014, 47, 1901–1910. [Google Scholar] [CrossRef]
- Hu, L.; He, Z.; Zhang, S. Sustainable use of rice husk ash in cement-based materials: Environmental evaluation and performance improvement. J. Clean. Prod. 2020, 264, 121744. [Google Scholar] [CrossRef]
- Castellanos, N.T.; Agredo, J.T. Using spent fluid catalytic cracking (FCC) catalyst as pozzolanic addition—A review. Ing. Investig. 2010, 30, 35–42. [Google Scholar] [CrossRef]
- Zito, S.V.; Irassar, E.F.; Rahhal, V.F. Recycled construction and demolition waste as supplementary cementing materials in eco-friendly concrete. Recycling 2023, 8, 54. [Google Scholar] [CrossRef]
- Doneliene, J.; Eisinas, A.; Baltakys, K.; Bankauskaite, A. The effect of synthetic hydrated calcium aluminate additive on the hydration properties of OPC. Adv. Mater. Sci. Eng. 2016, 2016, 3605845. [Google Scholar] [CrossRef]
- Kaminskas, R.; Savickaite, B.; Eisinas, A. Hydrothermally treated cement bypass dust as a supplementary cementitious material. Sustainability 2025, 17, 6757. [Google Scholar] [CrossRef]
- Auyeshov, A.; Arynov, K.; Yeskibayeva, C.; Ibrayeva, A.; Zhumadildayeva, A. Efficient production of high-purity magnesium hydroxide from serpentinite. Molecules 2025, 30, 3484. [Google Scholar] [CrossRef]
- Zhao, Q.; Liu, C.-J.; Jiang, M.-F.; Saxén, H.; Zevenhoven, R. Preparation of magnesium hydroxide from serpentinite by sulfuric acid leaching for CO2 mineral carbonation. Miner. Eng. 2015, 79, 116–124. [Google Scholar] [CrossRef]
- Romão, I.; Gando-Ferreira, L.M.; Zevenhoven, R. Separation and recovery of valuable metals extracted from serpentinite during the production of Mg(OH)2 for CO2 Sequestration. Miner. Eng. 2015, 77, 25–33. [Google Scholar] [CrossRef]
- Hamada, H.M.; Abed, F.; Beddu, S.; Humada, A.M.; Majdi, A. Effect of volcanic ash and natural pozzolana on mechanical properties of sustainable cement concrete: A comprehensive review. Case Stud. Constr. Mater. 2023, 19, e02425. [Google Scholar] [CrossRef]
- Jeong, Y.; Kang, S.-H.; Kim, M.O.; Moon, J. Acceleration of cement hydration from supplementary cementitious materials: Performance comparison between silica fume and hydrophobic silica. Cem. Concr. Compos. 2020, 112, 103688. [Google Scholar] [CrossRef]
- Shanks, B.; Howe, C.; Draper, S.; Wong, H.; Cheeseman, C. Production of low-carbon amorphous SiO2 for use as a supplementary cementitious material and nesquehonite from olivine. Mater. Lett. 2024, 361, 136133. [Google Scholar] [CrossRef]
- Lin, Y.; Alengaram, U.J.; Ibrahim, Z. Effect of treated and untreated rice husk ash, palm oil fuel ash, and sugarcane bagasse ash on the mechanical, durability, and microstructure characteristics of blended concrete–a comprehensive review. J. Build. Eng. 2023, 78, 107500. [Google Scholar] [CrossRef]
- Zulumyan, N.; Isahakyan, A.; Beglaryan, H.; Melikyan, S. A study of thermal decomposition of antigorite from dunite and lizardite from peridotite. J. Therm. Anal. Calorim. 2018, 131, 1201–1211. [Google Scholar] [CrossRef]
- EN 196-3:2016; Methods of Testing Cement. Determination of Setting Times and Soundness. European Committee for Standardization (CEN): Brussels, Belgium, 2016.
- EN 196-1:2016; Methods of Testing Cement. Determination of Strength. European Committee for Standardization (CEN): Brussels, Belgium, 2016.
- NF P18-513:2012; Metakaolin. Pozzolanic Addition for Concrete. Association Française de Normalisation: La Plaine Saint-Denis, France, 2012.
- Quarcioni, Y.; Chotoli, V.A.F.F.; Coelho, A.C.V.; Cincotto, M.A. Indirect and direct chapelle’s methods for the determination of lime consumption in pozzolanic materials. Rev. IBR. Estrut. Mat. 2015, 8, 1–7. [Google Scholar] [CrossRef]
- Hesse, C.; Goetz-Neunhoeffer, F.; Neubauer, J.A. New Approach in Quantitative In-Situ XRD of Cement Pastes: Correlation of Heat Flow Curves with Early Hydration Reactions. Cem. Concr. Res. 2011, 41, 123–128. [Google Scholar] [CrossRef]
- Pichler, C.; Perfler, L.; Lackner, R. Deconvolution of main hydration kinetic peaks in properly sulfated Portland cements with boundary nucleation and growth models and relation to early-age concrete strength development. Constr. Build. Mater. 2022, 348, 128602. [Google Scholar] [CrossRef]
- Scrivener, K.L.; Juilland, P.; Monteiro, P.J.M. Advances in understanding hydration of Portland cement. Cem. Concr. Res. 2015, 78, 38–56. [Google Scholar] [CrossRef]
- EN 197-1:2011; Cement–Part 1: Composition, Specifications and Conformity Criteria for Common Cements. European Committee for Standardization (CEN): Brussels, Belgium, 2011.
- El-Diadamony, H.; Amer, A.A.; Sokkary, T.M.; El-Hoseny, S. Hydration and Characteristics of Metakaolin Pozzolanic Cement Pastes. HBRC J. 2018, 14, 150–158. [Google Scholar] [CrossRef]
- Gineika, A.; Siauciunas, R.; Baltakys, K. Synthesis of wollastonite from AlF3-rich silica gel and its hardening in the CO2 atmosphere. Sci. Rep. 2019, 9, 18063. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Walder, B.J.; Kunhi Mohamed, A.; Hofstetter, A.; Srinivasan, B.; Rossini, A.J.; Scrivener, K.; Emsley, L.; Bowen, P. The atomic-level structure of cementitious calcium silicate hydrate. J. Phys. Chem. C 2017, 121, 17188–17196. [Google Scholar] [CrossRef]
- Miah, M.J.; Huaping, R.; Paul, S.C.; Babafemi, A.J.; Li, Y. Long-term strength and durability performance of eco-friendly concrete with supplementary cementitious materials. Innov. Infrastruct. Solut. 2023, 8, 255. [Google Scholar] [CrossRef]






| Component (wt.%) | ESW | OPC |
|---|---|---|
| SiO2 | 88.35 | 19.72 |
| Al2O3 | 0.10 | 4.93 |
| Fe2O3 | 2.47 | 3.25 |
| CaO | 0.48 | 61.19 |
| MgO | 2.64 | 3.93 |
| K2O | - | 1.04 |
| Na2O | - | 0.14 |
| Cr2O3 | 0.07 | - |
| NiO | 0.12 | - |
| SO3 | 2.73 | 2.6 |
| P2O5 | 0.83 | - |
| MnO | 0.05 | - |
| Other | 2.16 | 3.2 |
| Specific surface area, m2/kg | 320 | 320 |
| Component (wt.%) | ESW | NESW; W/S = 1.25 | NESW; W/S = 2.5 | NESW; W/S = 5 |
|---|---|---|---|---|
| SiO2 | 88.35 | 92.25 | 92.15 | 92.47 |
| Al2O3 | 0.10 | 0.12 | 0.13 | 0.13 |
| Fe2O3 | 2.47 | 0.86 | 0.93 | 0.90 |
| CaO | 0.48 | 0.56 | 0.55 | 0.58 |
| MgO | 2.64 | 1.52 | 1.51 | 1.51 |
| Cr2O3 | 0.07 | 0.07 | 0.06 | 0.06 |
| NiO | 0.12 | 0.06 | - | 0.08 |
| SO3 | 2.73 | 0.84 | 0.76 | 0.76 |
| P2O5 | 0.83 | 0.56 | 0.55 | 0.52 |
| Other | 2.21 | 3.16 | 3.36 | 2.99 |
| Component (wt.%) | Abbreviation | Normal Consistency W/C (%) | Setting Time (min) | ||
|---|---|---|---|---|---|
| OPC | NESW | Initial | Final | ||
| 100 | - | OPC | 0.27 | 84 | 126 |
| 95 | 5 | S5 | 0.28 | 82 | 115 |
| 85 | 15 | S15 | 0.30 | 80 | 105 |
| 75 | 25 | S25 | 0.32 | 75 | 100 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaminskas, R.; Zuzevicius, A.; Barauskas, I. Supplementary Cementitious Material from Epsom Salt Production Waste. J. Compos. Sci. 2025, 9, 708. https://doi.org/10.3390/jcs9120708
Kaminskas R, Zuzevicius A, Barauskas I. Supplementary Cementitious Material from Epsom Salt Production Waste. Journal of Composites Science. 2025; 9(12):708. https://doi.org/10.3390/jcs9120708
Chicago/Turabian StyleKaminskas, Rimvydas, Arijus Zuzevicius, and Irmantas Barauskas. 2025. "Supplementary Cementitious Material from Epsom Salt Production Waste" Journal of Composites Science 9, no. 12: 708. https://doi.org/10.3390/jcs9120708
APA StyleKaminskas, R., Zuzevicius, A., & Barauskas, I. (2025). Supplementary Cementitious Material from Epsom Salt Production Waste. Journal of Composites Science, 9(12), 708. https://doi.org/10.3390/jcs9120708






