On the Role of Bonding Time on Microstructure and Mechanical Properties of TLP Bonded Al/Mg2Si Composite
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Preparation
2.2. Joining Procedure
2.3. Microstructural Analyses
3. Results
3.1. Bonding Temperature
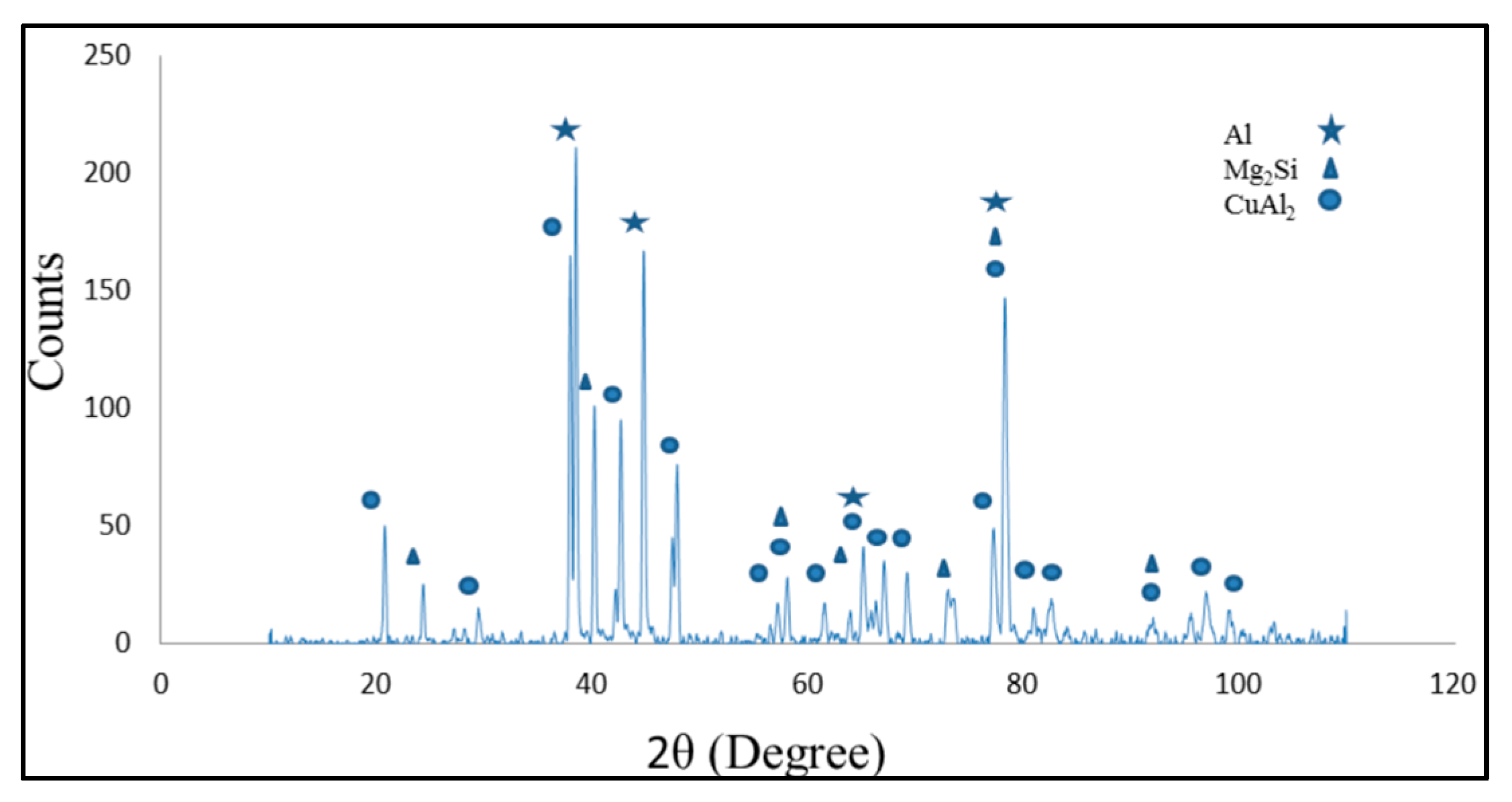
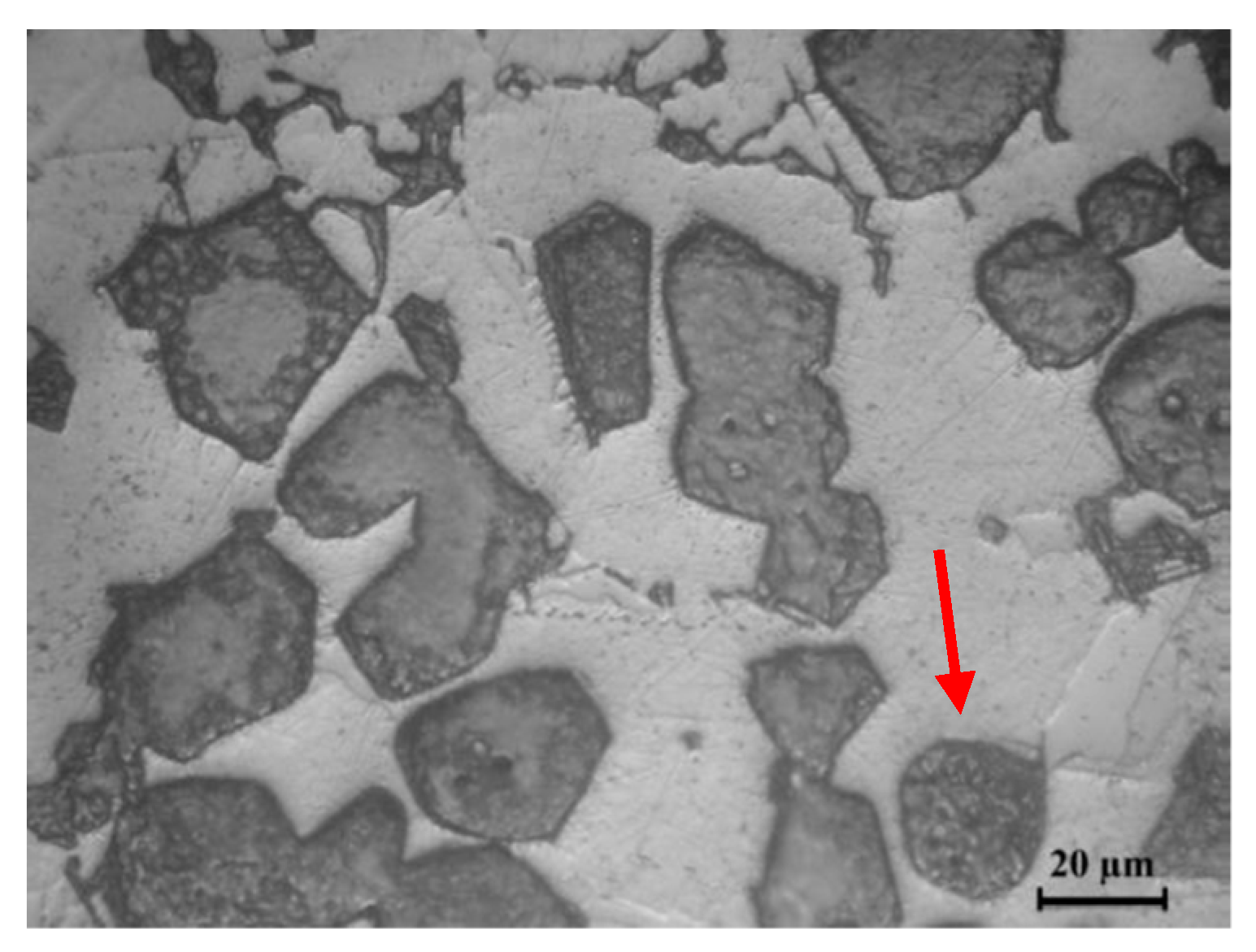
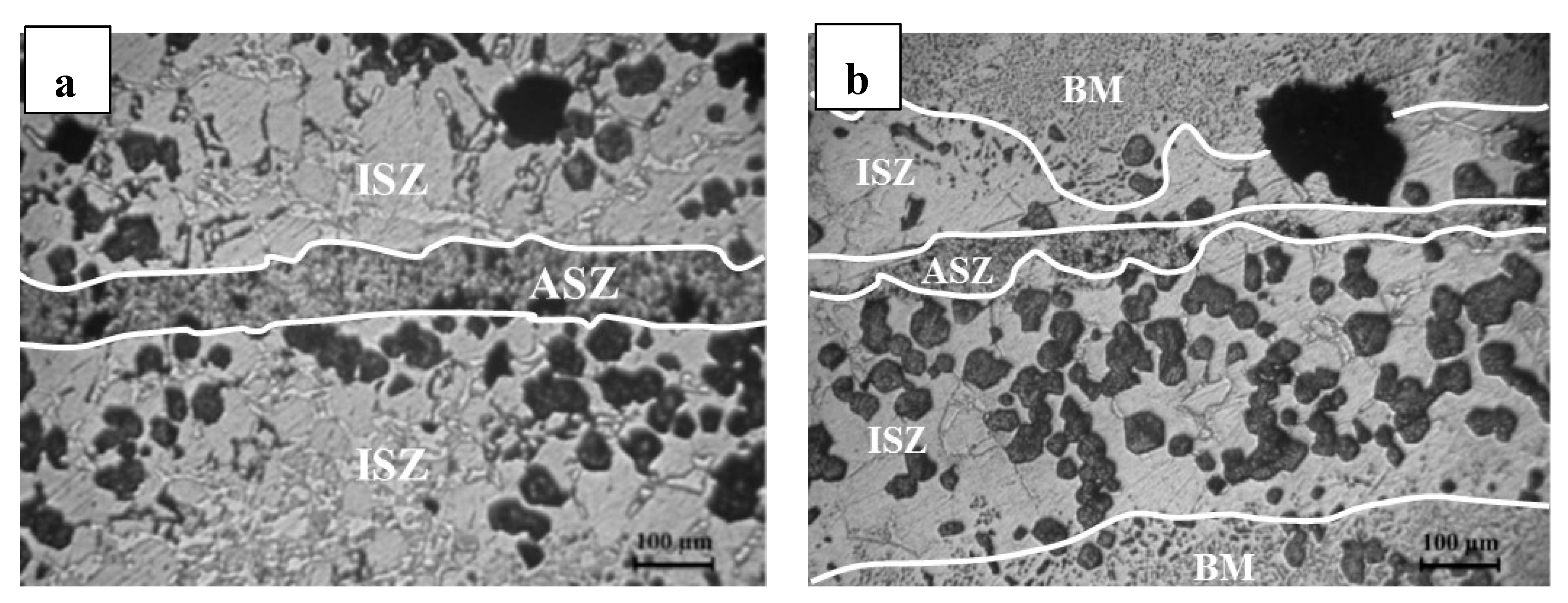
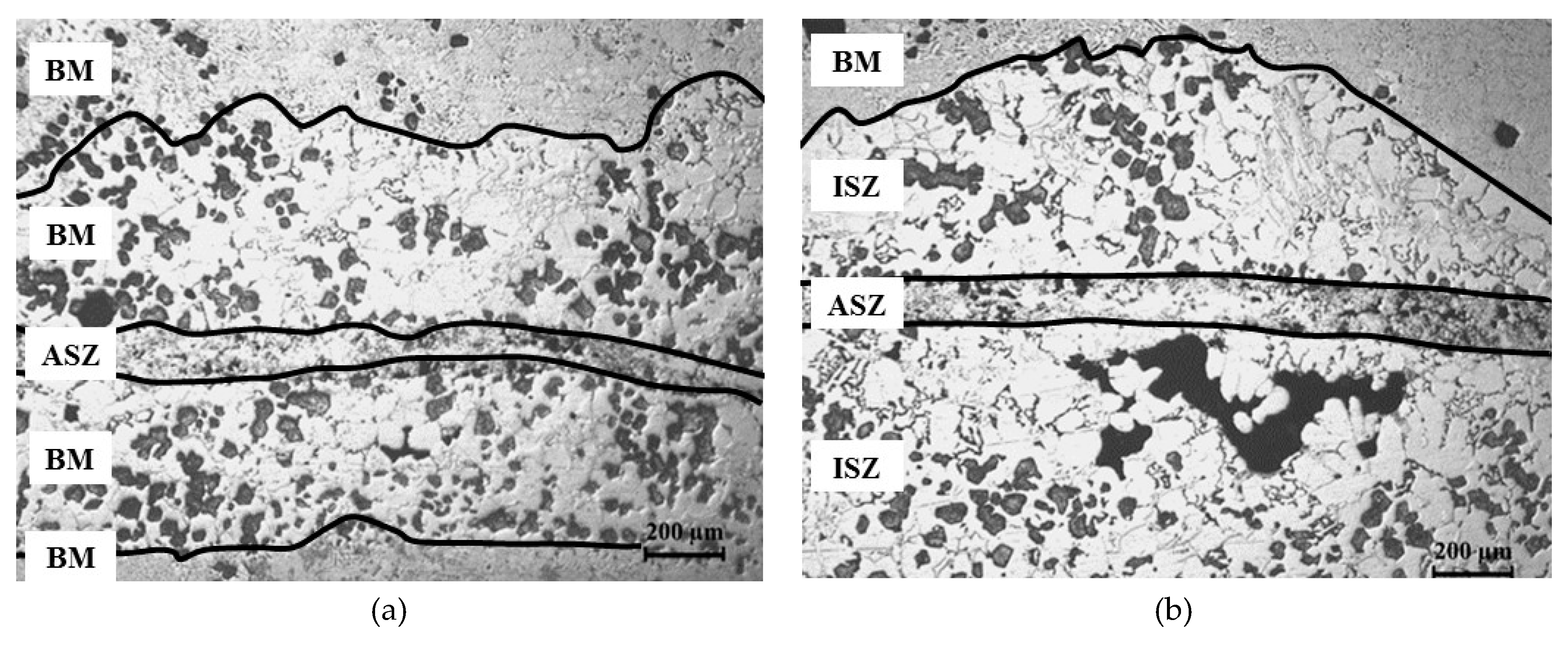
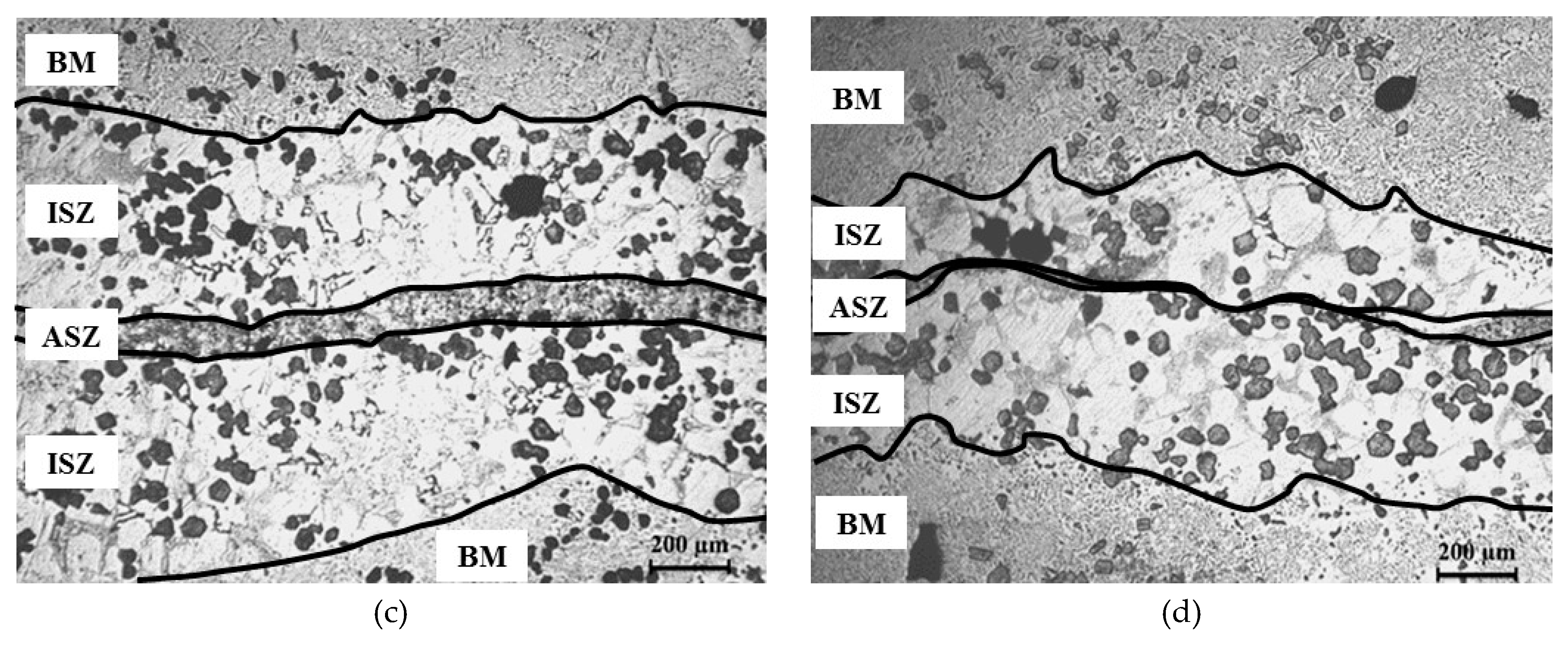
3.2. Microstructure of Base Material and the Bonding Zone
- (1)
- Athermally Solidified Zone (ASZ), which solidified by decreasing temperature from bonding temperature to room temperature.
- (2)
- Isothermally Solidified Zone (ISZ), which solidified at the constant temperature (bonding temperature) and segregation of reinforcement particles was obvious in this zone.
- (3)
- Base Material (BM), which did not have an effect on this zone with increasing temperature.
4. Discussion
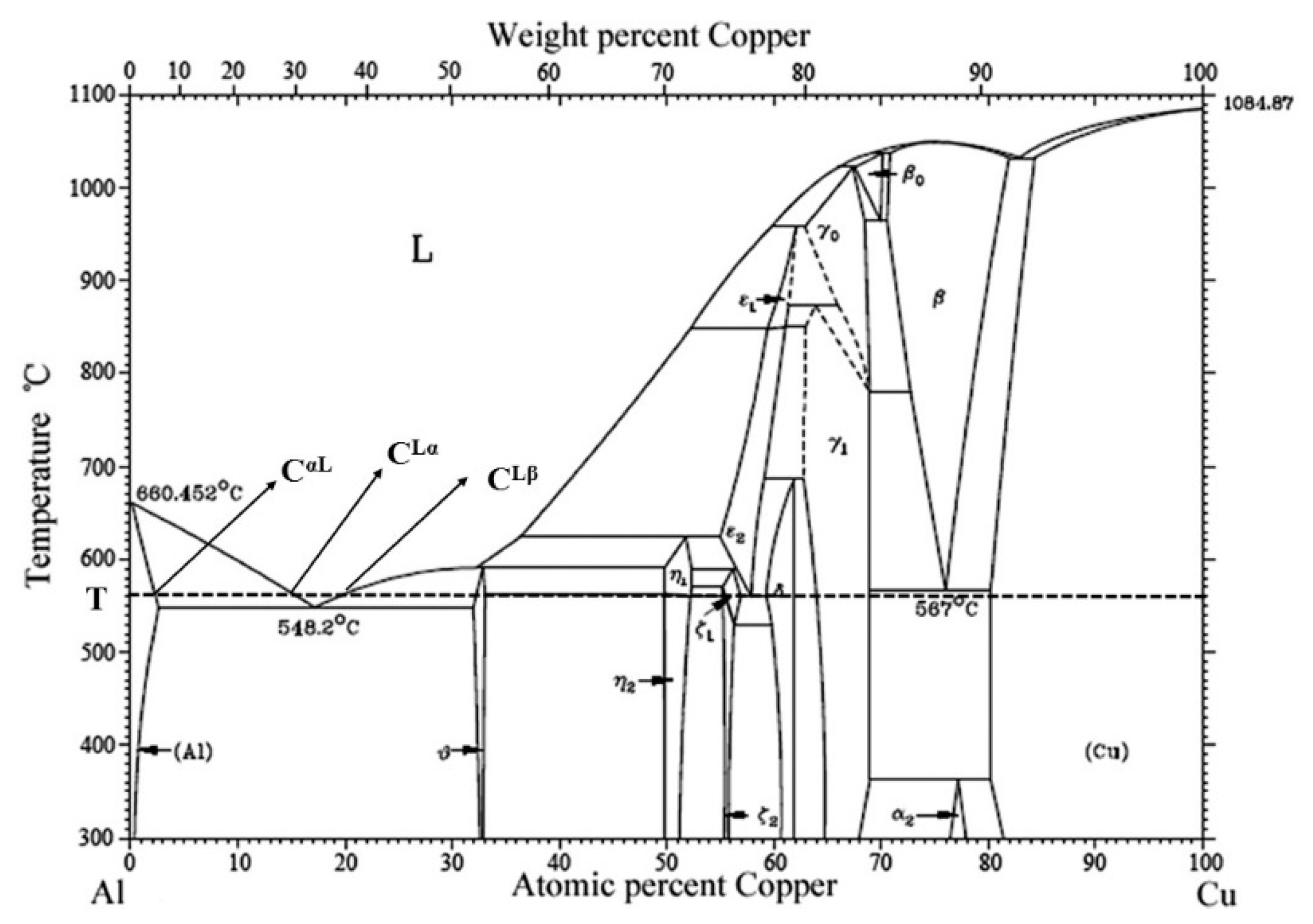
4.1. Athermally Solidification Zone (ASZ)
4.2. Isothermal Solidification Zone (ISZ)
- (1)
- Isothermal solidified α-Al
- (2)
- Primary α-Al precipitated from the liquid (Cαl), solidifying from the bonding temperature to eutectic temperature
- (3)
- Eutectic (CuAl2+α) phase, which solidified from the liquid under the eutectic temperature
4.3. Width of ASZ
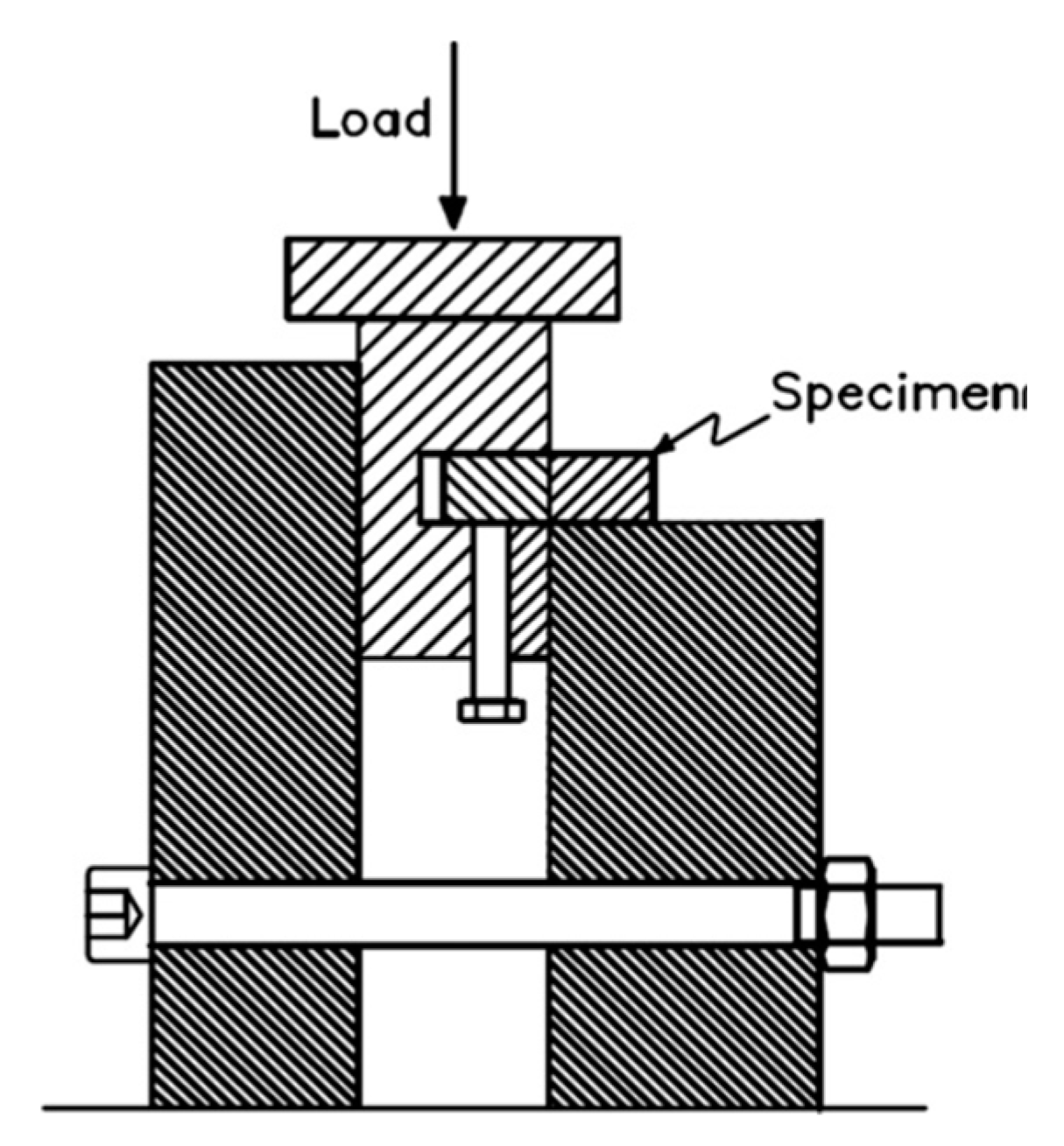
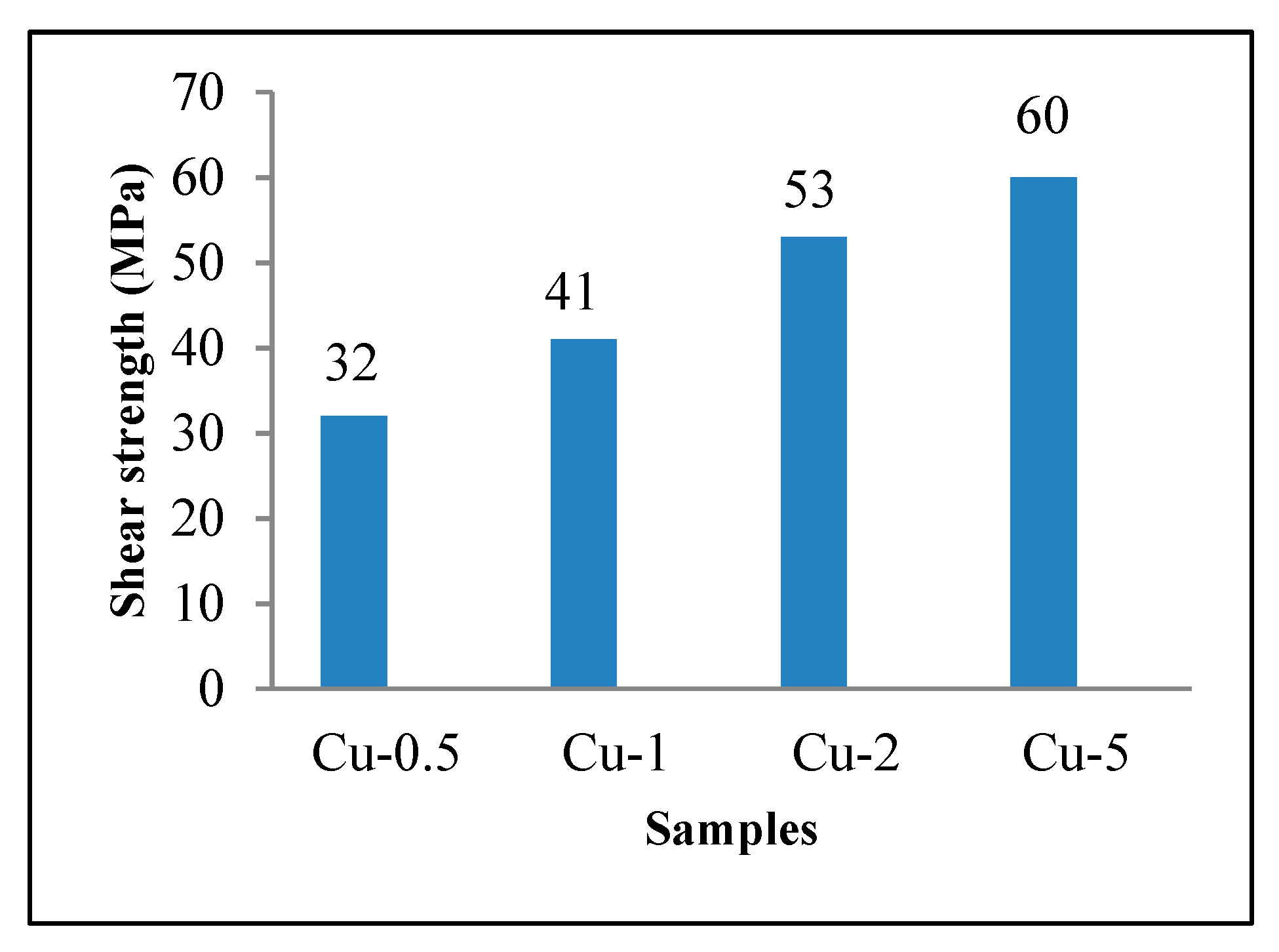
4.4. Shear Strength of Samples
5. Conclusions
- (1)
- Three zones were identified in the bonding interface:
- (i)
- An athermally solidified zone, which was in the center of the bonding line; it had porosities due to the use of Cu power
- (ii)
- An isothermally solidified zone, which can be characterized by the segregation of reinforcement particles
- (iii)
- Base metal, which did not have an effect on the microstructure even with increasing temperature
- (2)
- By increasing bonding time due to the diffusion of copper from the interlayer to the base metal, the width of ASZ decreased. In addition, by decreasing the amount of liquid formed at the bonding temperature, the reinforcement particles engulfed in the liquid decreased. Therefore, the width of the segregated zone (ISZ) decreased.
- (3)
- By increasing bonding time and diffusion of Cu from the interlayer, the amount of intermetallic phase (CuAl2) decreased in ISZ.
- (4)
- The maximum shear strength, 60 MPa (75% shear strength of as received composite), was obtained from the Cu-5 sample. Decrease in the amount of CuAl2, the width of ASZ, and homogenizing of the bonding zone due to the diffusion of copper were considered as the reasons for increasing shear strength by increasing bonding time.
Author Contributions
Funding
Conflicts of Interest
References
- Olakanmi, E.O.; Cochrane, R.; Dalgarno, K. A review on selective laser sintering/melting (SLS/SLM) of aluminium alloy powders: Processing, microstructure, and properties. Prog. Mater. Sci. 2015, 74, 401–477. [Google Scholar] [CrossRef]
- Polmear, I.; StJohn, D.; Nie, J.-F.; Qian, M. Light Alloys: Metallurgy of the Light Metals; Butterworth-Heinemann: Oxford, UK, 2017. [Google Scholar]
- Ellis, M. Joining of Al-based metal matrix composites-a review. Mater. Manuf. Proc. 1996, 11, 45–66. [Google Scholar] [CrossRef]
- Hadian, R.; Emamy, M.; Varahram, N.; Nemati, N. The effect of Li on the tensile properties of cast Al–Mg2Si metal matrix composite. Mater. Sci. Eng. A 2008, 490, 250–257. [Google Scholar] [CrossRef]
- Shirzadi, A.; Wallach, E. New approaches for transient liquid phase diffusion bonding of aluminium based metal matrix composites. Mater. Sci. Technol. 1997, 13, 135–142. [Google Scholar] [CrossRef]
- Pasebani, S.; Ghayoor, M.; Badwe, S.; Irrinki, H.; Atre, S.V. Effects of atomizing media and post processing on mechanical properties of 17-4 PH stainless steel manufactured via selective laser melting. Add. Manuf. 2018, 22, 127–137. [Google Scholar] [CrossRef]
- Ghayoor, M.; Lee, K.; He, Y.; Chang, C.-h.; Paul, B.K.; Pasebani, S. Microstructural Analysis of Additively Manufactured 304L Stainless Steel Oxide Dispersion Strengthened Alloy. Microsc. Microanal. 2019. accepted. [Google Scholar]
- Dehghanghadikolaei, A.; Ibrahim, H.; Amerinatanzi, A.; Hashemi, M.; Moghaddam, N.S.; Elahinia, M. Improving corrosion resistance of additively manufactured nickel–titanium biomedical devices by micro-arc oxidation process. J. Mater. Sci. 2019, 54, 7333–7355. [Google Scholar] [CrossRef]
- Ghayoor, M.; Badwe, S.B.; Irrinki, H.; Atre, S.V.; Pasebani, S. Water Atomized 17-4 PH Stainless Steel Powder as a Cheaper Alternative Powder Feedstock for Selective Laser Melting. In Proceedings of Materials Science Forum; Trans Tech Publications: Zurich, Switzerland, 2018; pp. 698–703. [Google Scholar]
- Brandl, E.; Heckenberger, U.; Holzinger, V.; Buchbinder, D. Additive manufactured AlSi10Mg samples using Selective Laser Melting (SLM): Microstructure, high cycle fatigue, and fracture behavior. Mater. Design 2012, 34, 159–169. [Google Scholar] [CrossRef]
- Ghayoor, M.; Lee, K.; He, Y.; Chang, C.-h.; Paul, B.K.; Pasebani, S. The Role of Volumetric Energy Density on the Microstructure and Texture of 304L Stainless Steel Produced via Selective Laser Melting. Add. Manuf. 2019. accepted. [Google Scholar]
- Mirzababaei, S.; Filip, P. Impact of humidity on wear of automotive friction materials. Wear 2017, 376, 717–726. [Google Scholar] [CrossRef]
- Zhang, G.; Zhang, J.; Pei, Y.; Li, S.; Chai, D. Joining of Al2O3p/Al composites by transient liquid phase (TLP) bonding and a novel process of active-transient liquid phase (A-TLP) bonding. Mater. Sci. Eng. A 2008, 488, 146–156. [Google Scholar] [CrossRef]
- MacDonald, W.; Eagar, T. Transient liquid phase bonding. Ann. Rev. Mater. Sci. 1992, 22, 23–46. [Google Scholar] [CrossRef]
- Maity, J.; Pal, T.K.; Maiti, R. Transient liquid phase diffusion bonding of 6061-15 wt% SiCp in argon environment. J. Mater. Proc. Technol. 2009, 209, 3568–3580. [Google Scholar] [CrossRef]
- Natsume, Y.; Ohsasa, K.; Tayu, Y.; Momono, T.; Narita, T. Numerical modeling of the transient liquid-phase diffusion bonding process of Al using Cu filler metal. ISIJ Int. 2003, 43, 1976–1982. [Google Scholar] [CrossRef]
- Li, Z.; Fearis, W.; North, T. Particulate segregation and mechanical properties in transient liquid phase bonded metal matrix composite material. Mater. Sci. Technol. 1995, 11, 363–369. [Google Scholar] [CrossRef]
- Askew, J.; Wilde, J.; Khan, T. Transient liquid phase bonding of 2124 aluminium metal matrix composite. Mater. Sci. Technol. 1998, 14, 920–924. [Google Scholar] [CrossRef]
- Baghbani, M.G.; Hadian, A.M. Transient Liquid Phase Bonding of Al/Mg2Si Composite Using a Cu-Ni Mixed Powder. In Proceedings of Advanced Materials Research; Trans Tech Publications: Zurich, Switzerland, 2014; pp. 632–637. [Google Scholar]
- Okamoto, H.; Schlesinger, M.; Mueller, E. ASM Handbook; Volume 3: Alloy Phase Diagram; ASM International: Materials Park, OH, USA, 2016; pp. 292–294. ISBN 978-1-62708-163-4. [Google Scholar]
- Li, Z.; Zhou, Y.; North, T. Counteraction of particulate segregation during transient liquid-phase bonding of aluminium-based MMC material. J. Mater. Sci. 1995, 30, 1075–1082. [Google Scholar] [CrossRef]
- Zhang, J.; Fan, Z.; Wang, Y.; Zhou, B. Microstructural development of Al–15wt.% Mg2Si in situ composite with mischmetal addition. Mater. Sci. Eng. A 2000, 281, 104–112. [Google Scholar] [CrossRef]
- Stefanescu, D.; Dhindaw, B.; Kacar, S.; Moitra, A. Behavior of ceramic particles at the solid-liquid metal interface in metal matrix composites. Metall. Trans. A 1988, 19, 2847–2855. [Google Scholar] [CrossRef]
- Maity, J.; Pal, T.K.; Maiti, R. Transient liquid phase diffusion bonding of 6061-13 vol.% SiCp composite using Cu powder interlayer: mechanism and interface characterization. J. Mater. Sci. 2010, 45, 3575–3587. [Google Scholar] [CrossRef]




| Element | Al | Si | Mg | Fe | Cu | Mn | Cr | Ni | Zn | Ti |
|---|---|---|---|---|---|---|---|---|---|---|
| wt.% | Bal | 5.5 | 9.7 | 0.09 | 0.001 | 0.02 | 0.003 | 0.007 | 0.007 | 0.009 |
| Element | Cu | P | Ag | As | Fe | Mn | Pb | Sb | Sn |
|---|---|---|---|---|---|---|---|---|---|
| wt.% | Bal | ≤0.001% | ≤0.002% | ≤0.0005% | ≤0.005% | ≤0.001% | ≤0.01% | ≤0.001% | ≤0.01% |
| Bonding Time | 30 min | 1 h | 2 h | 5 h |
|---|---|---|---|---|
| Width of ASZ (μm) | 128 ± 15 | 119 ± 9 | 98 ± 11 | 56 ± 8 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghayoor, M.; Hadian, A.M. On the Role of Bonding Time on Microstructure and Mechanical Properties of TLP Bonded Al/Mg2Si Composite. J. Compos. Sci. 2019, 3, 66. https://doi.org/10.3390/jcs3030066
Ghayoor M, Hadian AM. On the Role of Bonding Time on Microstructure and Mechanical Properties of TLP Bonded Al/Mg2Si Composite. Journal of Composites Science. 2019; 3(3):66. https://doi.org/10.3390/jcs3030066
Chicago/Turabian StyleGhayoor, Milad, and Ali M. Hadian. 2019. "On the Role of Bonding Time on Microstructure and Mechanical Properties of TLP Bonded Al/Mg2Si Composite" Journal of Composites Science 3, no. 3: 66. https://doi.org/10.3390/jcs3030066
APA StyleGhayoor, M., & Hadian, A. M. (2019). On the Role of Bonding Time on Microstructure and Mechanical Properties of TLP Bonded Al/Mg2Si Composite. Journal of Composites Science, 3(3), 66. https://doi.org/10.3390/jcs3030066




