Tensile Properties, Fracture Mechanics Properties and Toughening Mechanisms of Epoxy Systems Modified with Soft Block Copolymers, Rigid TiO2 Nanoparticles and Their Hybrids
Abstract
1. Introduction
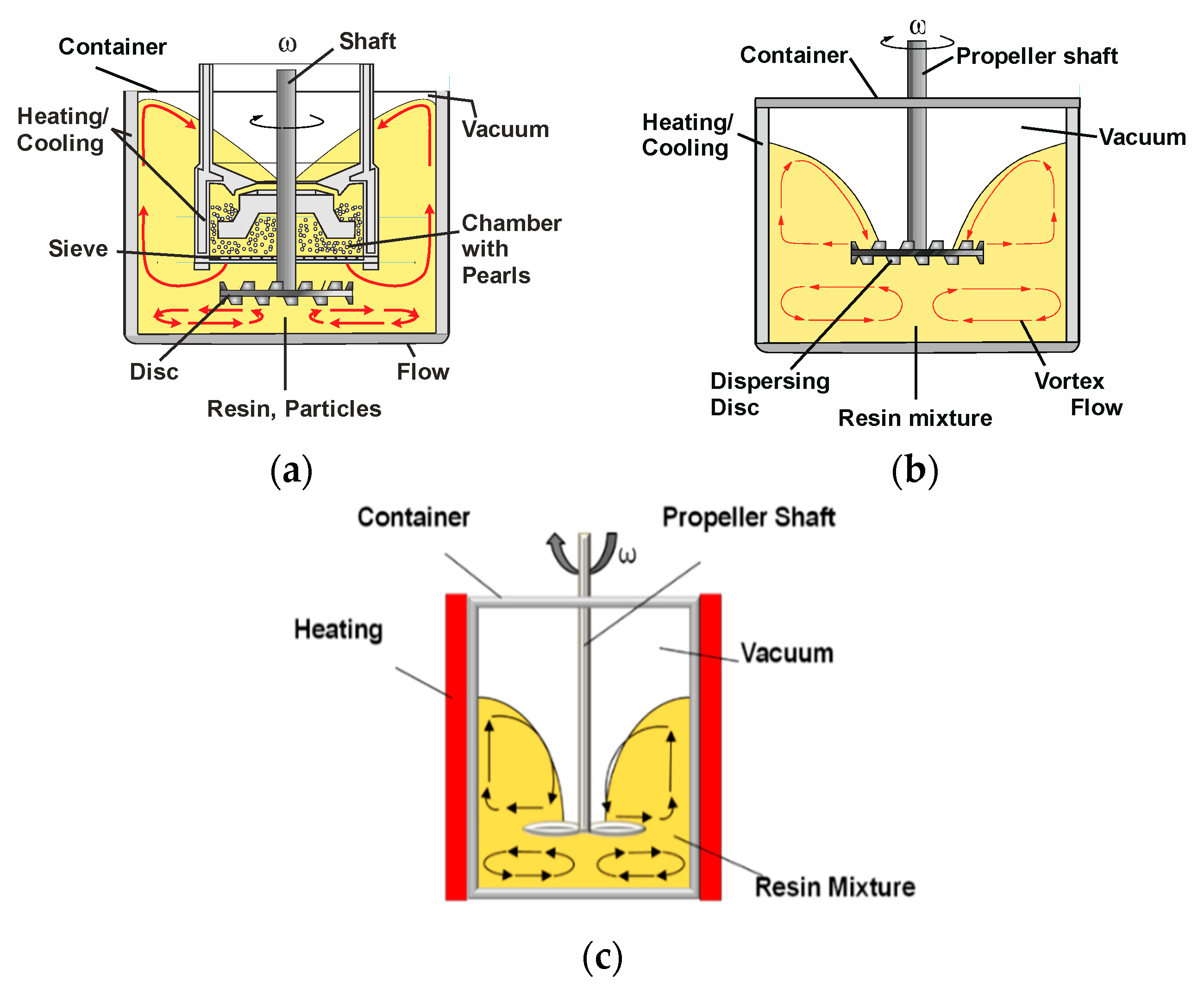
2. Materials and Methods
2.1. Differential Scanning Calorimetry
2.2. Rheometric Measurement
2.3. Dynamic-Mechanical Thermal Analysis
2.4. Mechanical Properties
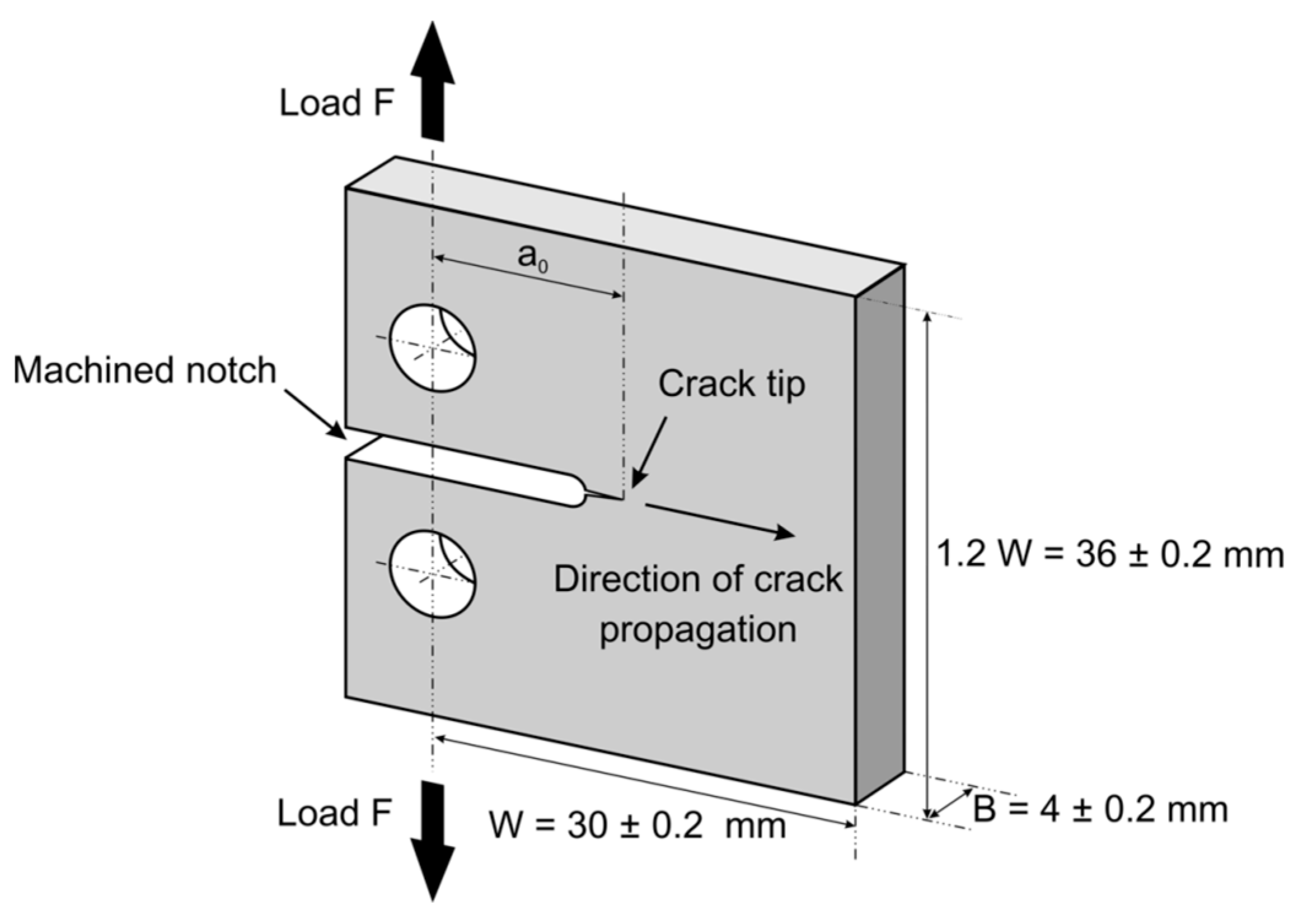
2.5. Fracture Test
2.6. Microscopy Studies
2.7. White Light Profilometry
3. Results and Discussion
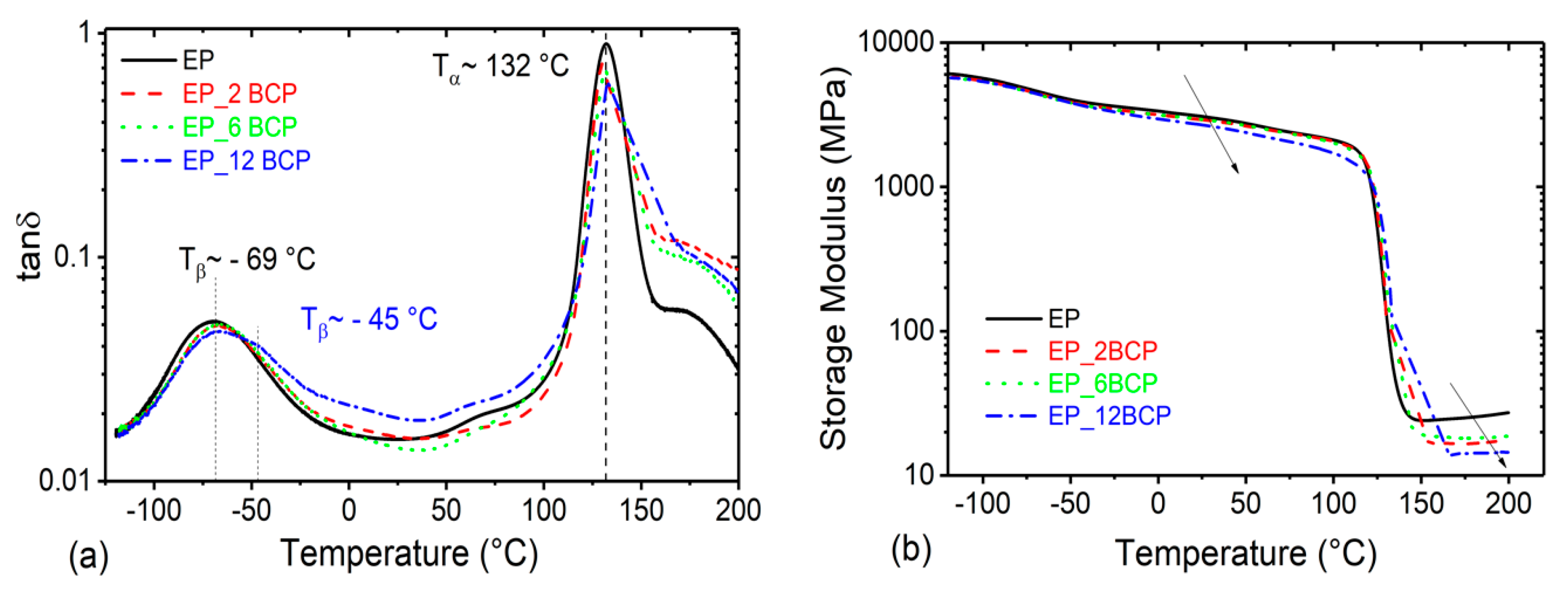
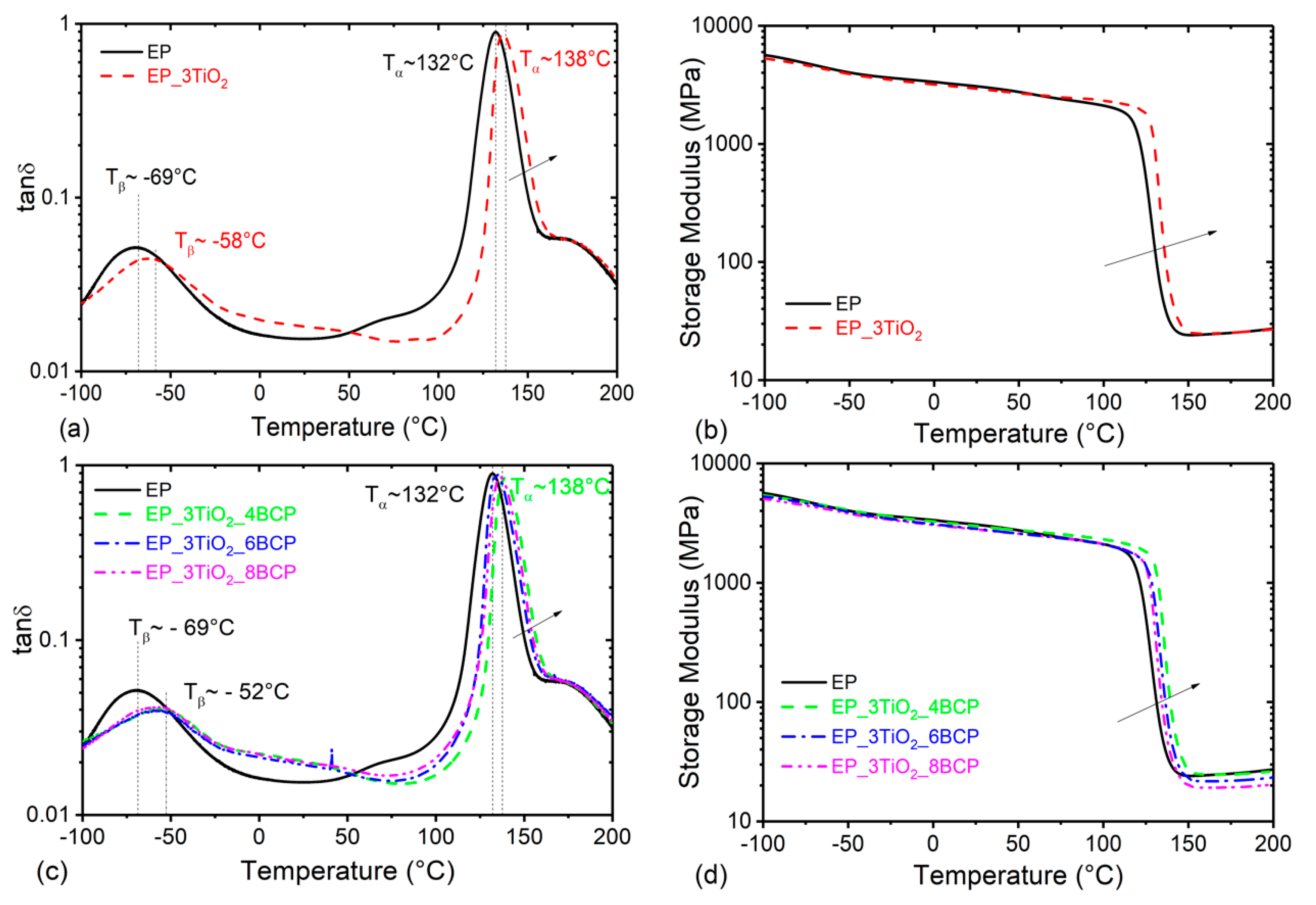
3.1. Glass Transition Temperature and Viscoelastic Properties
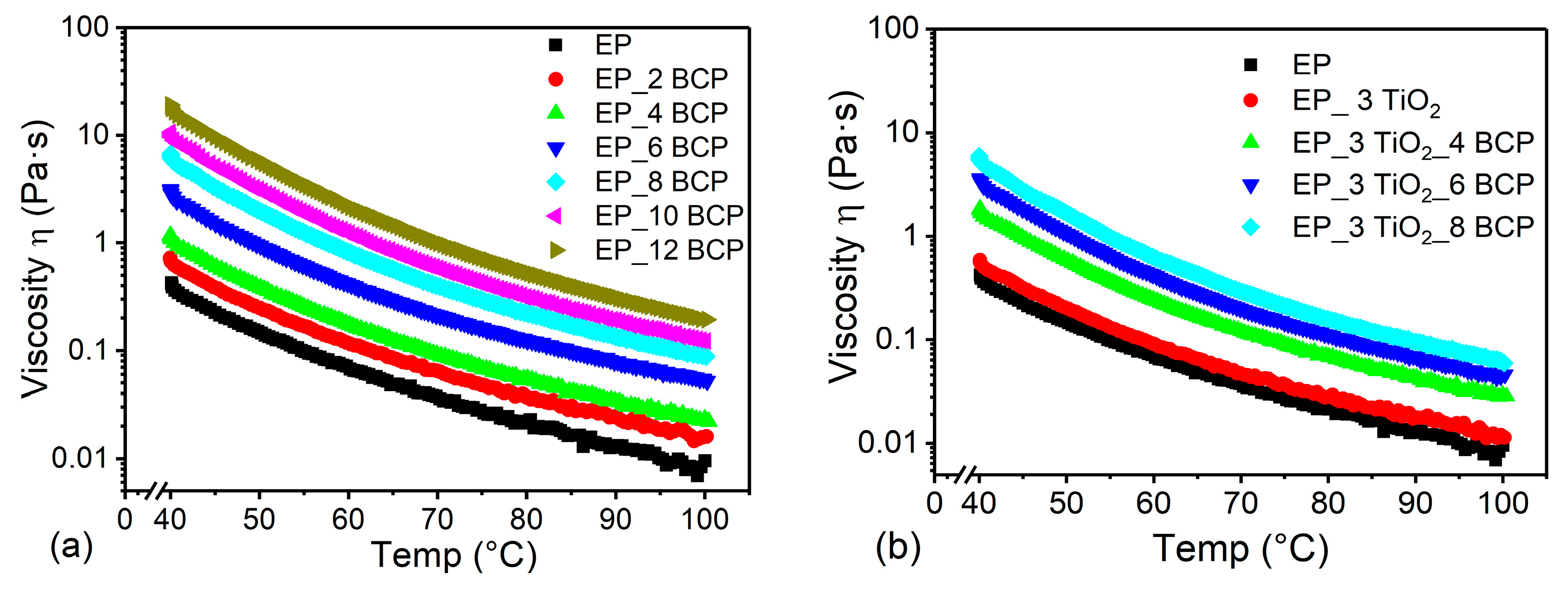
3.2. Rheology
3.3. Tensile Properties
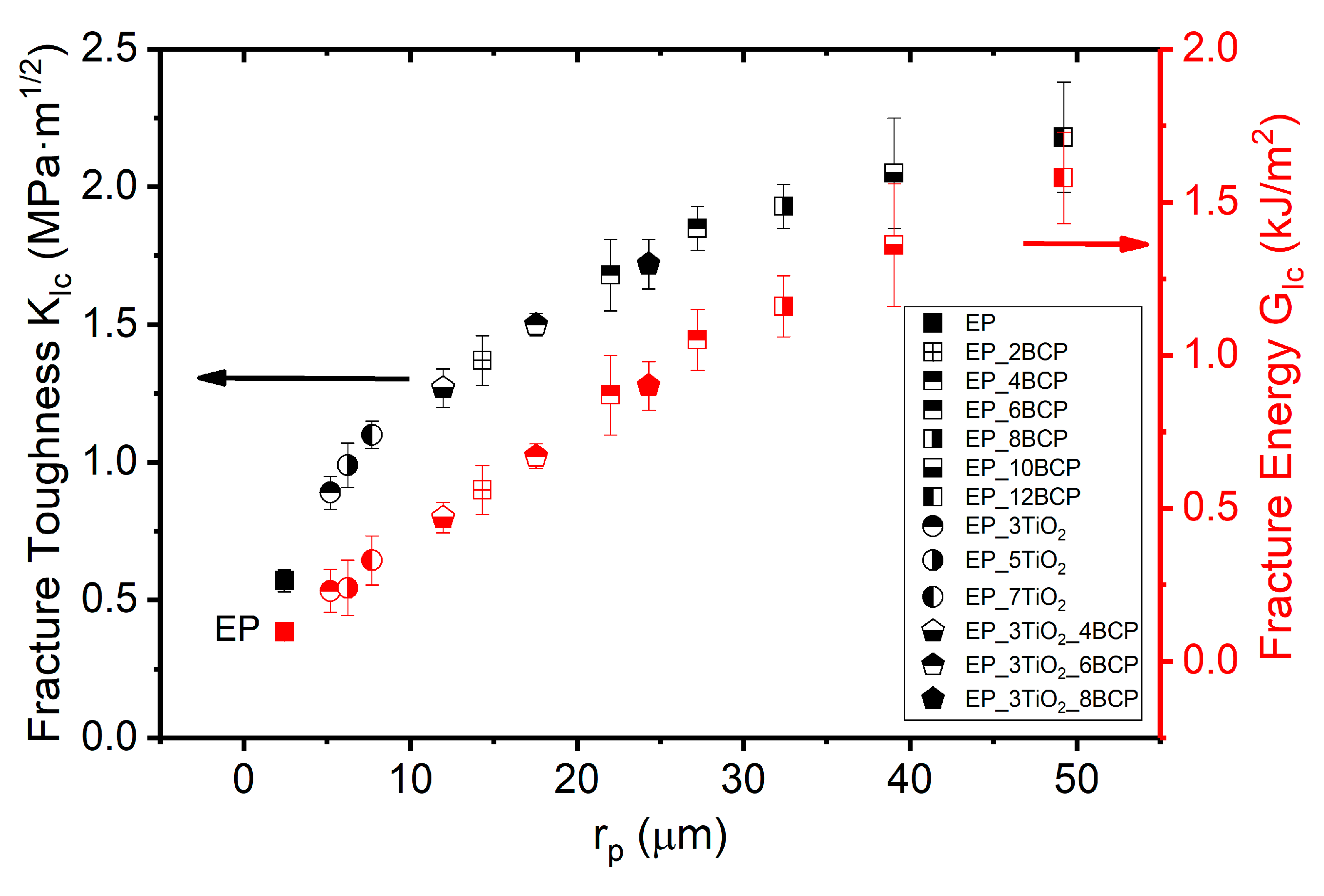
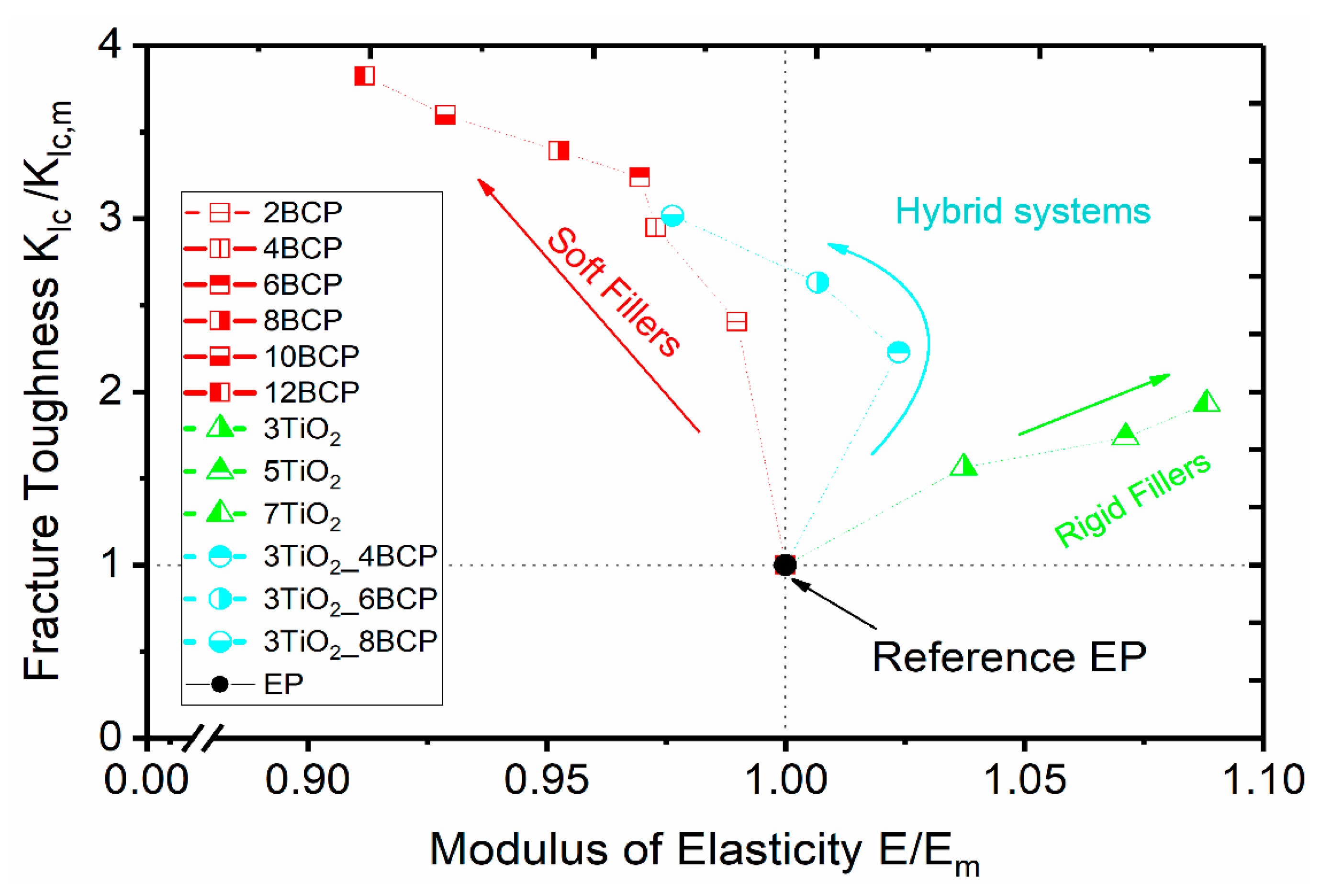
3.4. Fracture-Mechanics Properties
3.5. Fractography Studies
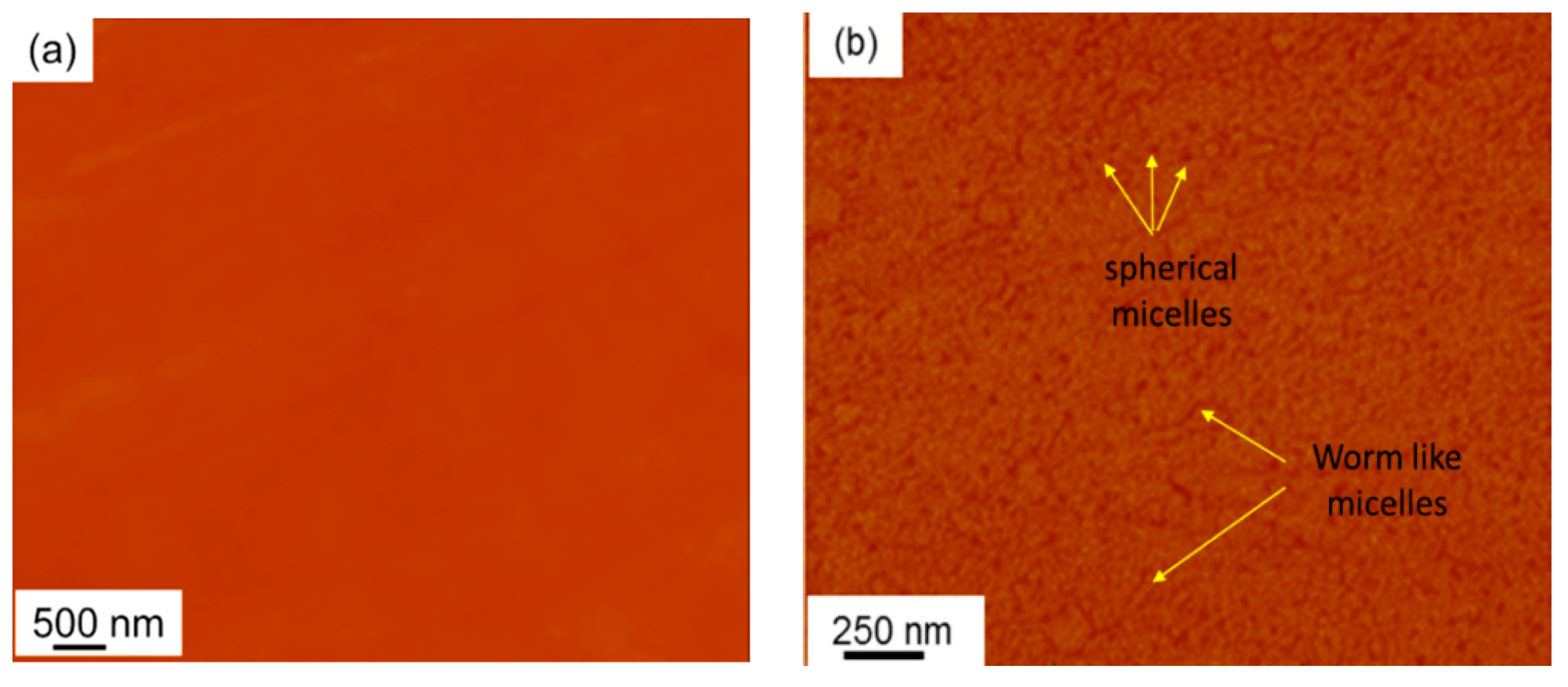
3.5.1. BCP Modified Epoxies
3.5.2. TiO2 Modified Epoxy Systems and Hybrid Composites
3.5.3. Plastic Zone Size
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kunz, S.C.; Syre, J.A.; Assink, R.A. Morphology and toughness characterization of epoxy resins modified with amine and carboxyl terminated rubbers. Polymer 1982, 23, 1897–1906. [Google Scholar] [CrossRef]
- Sultan, J.N.; McGarry, F. Effect of rubber particle size on deformation mechanisms in glassy epoxy. Polym. Eng. Sci. 1973, 13, 29–34. [Google Scholar] [CrossRef]
- Pearson, R.A.; Yee, A.F. Toughening mechanisms in elastomer-modified epoxies. J. Mater. Sci. 1989, 24, 2571–2580. [Google Scholar] [CrossRef]
- Yee, F.; Pearson, R.A. Toughening mechanisms in elastomer-modified epoxies Part 1 Mechanical studies. J. Mater. Sci. 1986, 21, 2462–2474. [Google Scholar] [CrossRef]
- Lin, K.F.; Shieh, Y.D. Core-shell particles designed for toughening the epoxy resins. II. Core-shell-particle-toughened epoxy resins. J. Appl. Polym. Sci. 1998, 70, 2313–2322. [Google Scholar] [CrossRef]
- Bates, C.M.; Bates, F.S. 50th anniversary perspective: Block polymers-pure potential. Macromolecules 2016, 50, 3–22. [Google Scholar] [CrossRef]
- Mai, Y.; Eisenberg, A. Self-assembly of block copolymers. Chem. Soc. Rev. 2012, 41, 5969–5985. [Google Scholar] [CrossRef] [PubMed]
- Lipic, P.M.; Bates, F.S.; Hillmyer, M.A. nanostructured thermosets from self-assembled amphiphilic block copolymer/epoxy resin mixtures. J. Am. Chem. Soc. 1998, 120, 8963–8970. [Google Scholar] [CrossRef]
- Maiez-Tribut, S.; Pascault, J.P.; Soule, E.R.; Borrajo, J.; Williams, R.J.J. Nanostructured epoxies based on the self-assembly of block copolymers: A new miscible block that can be tailored to different epoxy formulations. Macromolecules 2007, 40, 1268–1273. [Google Scholar] [CrossRef]
- Kishi, H.; Kunimitsu, Y.; Nakashima, Y.; Abe, T.; Imade, J.; Oshita, S. Control of nanostructures generated in epoxy matrices blended with PMMA-b-PnBA-b-PMMA triblock copolymers. Express Polym. Lett. 2015, 9, 23–25. [Google Scholar] [CrossRef]
- Yamanaka, K.; Takagi, Y.; Inoue, T. Reaction-induced phase separation in rubber-modified epoxy resins. Polymer 1989, 30, 1839–1844. [Google Scholar] [CrossRef]
- Meng, F.; Zheng, S.; Li, H.; Liang, Q.; Liu, T. Formation of ordered nanostructures in epoxy thermosets: A mechanism of reaction-induced microphase separation. Macromolecules 2006, 39, 5072–5080. [Google Scholar] [CrossRef]
- Williams, R.J.; Rozenberg, B.A.; Pascault, J.P. Reaction-induced phase separation in modified thermosetting polymers. In Polymer Analysis Polymer Physics; Springer: Berlin/Heidelberg, Germany, 1997; pp. 95–156. [Google Scholar]
- Ocando, C.; Tercjak, A.; Martín, M.D.; Ramos, J.A.; Campo, M.; Mondragon, I. Morphology development in thermosetting mixtures through the variation on chemical functionalization degree of poly(styrene-b-butadiene) diblock copolymer modifiers. Thermomechanical properties. Macromolecules 2009, 42, 6215–6224. [Google Scholar] [CrossRef]
- Fan, W.; Wang, L.; Zheng, S. Nanostructures in thermosetting blends of epoxy resin with polydimethylsiloxane-block-poly(ε-caprolactone)-block-polystyrene ABC triblock copolymer. Macromolecules 2008, 42, 327–336. [Google Scholar] [CrossRef]
- Dean, J.; Grubbs, R.B.; Saad, W.; Cook, R.; Bates, F. Mechanical properties of block copolymer vesicle and micelle modified epoxies. J. Polym. Sci. Part B Polym. Phys. 2003, 41, 2444–2456. [Google Scholar] [CrossRef]
- Kishi, H.; Kunimitsu, Y.; Nakashima, Y.; Imade, J.; Oshita, S.; Morishita, Y.; Asada, M. Relationship between the mechanical properties of epoxy/PMMA-b-PNBA-b-PMMA block copolymer blends and their three-dimensional nanostructures. Express Polym. Lett. 2017, 11, 765–777. [Google Scholar] [CrossRef]
- Redline, E.M.; Declet-Perez, C.; Bates, F.S.; Francis, L.F. Effect of block copolymer concentration and core composition on toughening epoxies. Polymer 2014, 55, 4172–4181. [Google Scholar] [CrossRef]
- Chen, J.; Taylor, A.C. Epoxy modified with triblock copolymers: Morphology, mechanical properties and fracture mechanisms. Mater. Sci. 2012, 47, 4546–4560. [Google Scholar] [CrossRef]
- Ritzenthaler, S.; Girard-Reydet, E.; Pascault, J.P. Influence of epoxy hardener on miscibility of blends of poly(methyl methacrylate) and epoxy networks. Polymer 2000, 41, 6375–6386. [Google Scholar] [CrossRef]
- Romeo, H.E.; Zucchi, I.A.; Rico, M.H.C.E.; Williams, R.J.J. From spherical micelles to hexagonally packed cylinders: The cure cycle determines nanostructures generated in block copolymer/epoxy blends. Macromolecules 2013, 46, 4854–4861. [Google Scholar] [CrossRef]
- Klingler, A.; Bajpai, A.; Wetzel, B. The effect of block copolymer and core-shell rubber hybrid toughening on morphology and fracture of epoxy-based fibre reinforced composites. Eng. Fract. Mech. 2018, 203, 81–101. [Google Scholar] [CrossRef]
- Klingler, A.; Wetzel, B. Fatigue crack propagation in triblock copolymer toughened epoxy nanocomposites. Polym. Eng. Sci. 2017, 57, 579–587. [Google Scholar] [CrossRef]
- Bajpai, A.; Alapati, A.K.; Wetzel, B. Toughening and mechanical properties of epoxy modified with block co-polymers and MWCNTs. Procedia Struct. Integr. 2016, 2, 104–111. [Google Scholar] [CrossRef]
- Chong, H.M.; Taylor, A.C. The microstructure and fracture performance of styrene-butadiene-methylmethacrylate block copolymer-modified epoxy polymers. Mater. Sci. 2013, 48, 6762–6777. [Google Scholar] [CrossRef]
- Ritzenthaler, S.; Court, F.; David, L.; Girard-Reydet, E.; Leibler, L.; Pascault, J.P. ABC triblock copolymers/epoxy—diamine blends. 1. keys to achieve nanostructured thermosets. Macromolecules 2002, 35, 6245–6254. [Google Scholar] [CrossRef]
- Carballeira, P.; Haupert, F. Toughening effects of titanium dioxide nanoparticles on TiO2/epoxy resin. Polym. Compos. 2010, 31, 1241–1246. [Google Scholar] [CrossRef]
- Wetzel, B.; Rosso, P.; Haupert, F.; Friedrich, K. Epoxy nanocomposites—Fracture and toughening mechanisms. Eng. Fract. Mech. 2006, 73, 2375–2398. [Google Scholar] [CrossRef]
- Lee, J.; Yee, A.F. Fracture of glass bead/epoxy composites: On micro-mechanical deformations. Polymer 2000, 41, 8363–8373. [Google Scholar] [CrossRef]
- Hsieh, T.H.; Kinloch, A.J.; Masania, K.; Taylor, A.C.; Sprenger, S. The mechanisms and mechanics of the toughening of epoxy polymers modified with silica nanoparticles. Polymer 2010, 51, 6284–6294. [Google Scholar] [CrossRef]
- Liang, Y.L.; Pearson, R.A. The toughening mechanism in hybrid epoxy-silica-rubber nanocomposites. Polymer 2010, 51, 4880–4890. [Google Scholar] [CrossRef]
- Hsieh, T.H.; Kinloch, A.J.; Masania, K.; Lee, J.S.; Taylor, A.C.; Sprenger, S. The toughness of epoxy polymers and fibre composites modified with rubber microparticles and silica nanoparticles. J. Mater. Sci. 2010, 45, 1193–1210. [Google Scholar] [CrossRef]
- Maxwell, D.; Young, R.J.; Kinloch, A.J. Hybrid particulate-filled epoxy-polymers. J. Mater. Sci. Lett. 1984, 3, 9–12. [Google Scholar] [CrossRef]
- Kinloch, J.; Maxwell, D.; Young, R.J. Micromechanisms of crack propagation in hybrid-particulate composites. J. Mater. Sci. Lett. 1985, 4, 1276–1279. [Google Scholar] [CrossRef]
- Durig, J. Comparisons of epoxy technology for protective coatings and linings in wasterwater facilities. In Proceedings of the Industrial Protective Coatings Conference and Exhibit, Houston, TX, USA, 14–18 November 1999. [Google Scholar]
- H. Inc. Product Specifications for the EPON862 and Technical Data Sheet; H. Inc.: Lansing, MI, USA, 2005. [Google Scholar]
- Evonik Resource Efficiency GmbH. Product information AEROXIDE® TiO2 P 25; Evonik Resource Efficiency GmbH: Darmstadt, Germany, 2016. [Google Scholar]
- Carballeira, P.; Haupert, F. Toughening effects of titanium dioxide nanoparticles on TiO2 /epoxy resin nanocomposites. Soc. Plast. Eng. 2009, 31, 1241–1246. [Google Scholar] [CrossRef]
- Wetzel, B. Mechanische Eigenschaften von Nanoverbundwerkstoffen aus Epoxydharz und keramischen Nanopartikeln; PhD, IVW Schriftenreihe Band 69; Technische Universität Kaiserslautern: Kaiserslautern, Germany, 2006; ISBN 3-934930-65-4. [Google Scholar]
- Moore, D. Application of Fracture Mechanics to Polymers, Adhesives and Composites; Elsevier Science: Amsterdam, The Netherlands, 2003. [Google Scholar]
- ISO 13586:2000(E): Plastics—Determination of Fracture Toughness (GIc and KIc)—Linear Elastic Fracture Mechanics (LEFM) Approach; ISO (The International Organization for Standardization): Geneva, Switzerland, 2000.
- Kinloch, J. Stresses in Adhesive Joints. In Adhesion and Adhesives: Science and Technology; Springer: London, UK, 1987; p. 209. [Google Scholar]
- Baller, J.; Becker, N.; Ziehmer, M.; Thomassey, M.; Zielinski, B.; Müller, U.; Sanctuary, R. Interactions between silica nanoparticles and an epoxy resin before and during network formation. Polymer 2009, 50, 3211–3219. [Google Scholar] [CrossRef]
- Arkema. Technical Data Sheet—Nanostrength® Epoxy Application; Arkema: Paris, France, 2013. [Google Scholar]
- Nielsen, L.; Landel, R.F. Mechanical Properties of Polymers and Composites; Marcel Dekker, Inc.: New York, NY, USA, 1994. [Google Scholar]
- Hull, D. Fractography: Observing, Measuring and Interpreting Fracture Structure Topography; Cambridge University Press: Cambridge, UK, 1999. [Google Scholar]
- Bagheri, R.; Pearson, R.A. Role of plastic cavitation in rubber toughened epoxies. Polymer 1996, 37, 5597–5600. [Google Scholar] [CrossRef]
- Wu, J.; Thio, Y.S.; Bates, F.S. Structure and properties of PBO-PEO diblock copolymer modified epoxy. J. Polym. Sci. Part B Polym. Phys. 2005, 43, 1950–1965. [Google Scholar] [CrossRef]
- Kinloch, J.; Young, R.J. Fracture Behaviour of Polymers; Elsevier Applied Science: New York, NY, USA, 1983. [Google Scholar]


| Series | BCP (wt %) | BCP (vol %) | TiO2 (wt %) | TiO2 (vol %) |
|---|---|---|---|---|
| EP | 0 | 0 | 0 | 0 |
| EP_2 BCP | 2 | 2.14 | 0 | 0 |
| EP_4 BCP | 4 | 4.26 | 0 | 0 |
| EP_6 BCP | 6 | 6.39 | 0 | 0 |
| EP_8 BCP | 8 | 8.50 | 0 | 0 |
| EP_10 BCP | 10 | 10.62 | 0 | 0 |
| EP_12 BCP | 12 | 12.72 | 0 | 0 |
| EP_3 TiO2 | 0 | 0 | 3 | 0.82 |
| EP_5 TiO2 | 0 | 0 | 5 | 1.40 |
| EP_7 TiO2 | 0 | 0 | 7 | 1.98 |
| EP_3 TiO2_4 BCP | 4 | 4.36 | 3 | 0.82 |
| EP_3 TiO2_6 BCP | 6 | 7.32 | 3 | 0.81 |
| EP_3 TiO2_8 BCP | 8 | 9.72 | 3 | 0.81 |
| Sample | Tg (°C) DSC | Tg (°C) (tanδ Peak) | Sample | Tg (°C) DSC | Tg (°C) (tanδ Peak) |
|---|---|---|---|---|---|
| EP | 134 | 132 | EP | 134 | 132 |
| EP_2 BCP | 135 | 132 | EP_3 TiO2 | 136 | 138 |
| EP_4 BCP | 134 | N/A | EP_5 TiO2 | 136 | N/A |
| EP_6 BCP | 136 | 132 | EP_7 TiO2 | 137 | N/A |
| EP_8 BCP | 135 | N/A | EP_3 TiO2_4 BCP | 135 | 138 |
| EP_10 BCP | 134 | N/A | EP_3 TiO2_6 BCP | 136 | 134 |
| EP_12 BCP | 133 | 132 | EP_3 TiO2_8 BCP | 135 | 136 |
| Systems | Et (MPa) | σm (MPa) | εm (%) | KIc (MPa·m1/2) | GIc (kJ/m2) |
|---|---|---|---|---|---|
| EP | 2950 (±75.8) | 84.0 (±0.7) | 6.3 (±0.4) | 0.57 (±0.04) | 0.096 (±0.03) |
| EP_2 BCP | 2920 (±35.2) | 83.4 (±0.8) | 6.7 (±0.4) | 1.37 (±0.09) | 0.56 (±0.08) |
| EP_4 BCP | 2870 (±39.4) | 82.5 (±4.9) | 5.9 (±1.4) | 1.68 (±0.13) | 0.87 (±0.13) |
| EP_6 BCP | 2860 (±22.7) | 81.7 (±1.7) | 5.6 (±0.7) | 1.85 (±0.08) | 1.05 (±0.10) |
| EP_8 BCP | 2810 (±36.1) | 78.1(±4.3) | 5.0 (±0.5) | 1.93 (±0.08) | 1.16 (±0.10) |
| EP_10 BCP | 2740 (±43.2) | 75.6 (±1.7) | 4.8 (±0.1) | 2.05 (±0.28) | 1.36 (±0.34) |
| EP_12 BCP | 2690 (±17.6) | 71.2 (±1.0) | 4.5 (±0.2) | 2.18(±0.30) | 1.58 (±0.45) |
| Systems | Et (MPa) | σm (MPa) | εm (%) | KIc (MPa·m1/2) | GIc (kJ/m2) |
|---|---|---|---|---|---|
| EP | 2950 (±75.8) | 84.0 (±0.70) | 6.3 (±0.40) | 0.57 (±0.04) | 0.096 (±0.03) |
| EP_3 TiO2 | 3060 (±13.8) | 89.9 (± 0.63) | 6.3 (±0.54) | 0.89 (±0.06) | 0.23 (±0.12) |
| EP_5 TiO2 | 3160 (±26.2) | 91.3 (± 0.37) | 6.5 (±0.19) | 0.99 (±0.08) | 0.24 (±0.16) |
| EP_7 TiO2 | 3210 (±17.6) | 91.3 (±0.28) | 6.5 (±0.12) | 1.10 (±0.05) | 0.33 (±0.03) |
| EP_3TiO2_4 BCP | 3020 (±28.8) | 84.6 (±0.87) | 6.2 (±0.75) | 1.27 (±0.07) | 0.47 (±0.05) |
| EP_3TiO2_6 BCP | 2970 (±16.7) | 82.5 (±0.23) | 6.3 (±0.53) | 1.50 (±0.04) | 0.67 (±0.04) |
| EP_3TiO2_8 BCP | 2880 (±14.7) | 80.4 (±0.37) | 6.4 (±0.04) | 1.72 (±0.17) | 0.90 (±0.17) |
| System | Surface Roughness (µm) |
|---|---|
| EP | 0.157 |
| EP_2 BCP | 0.183 |
| EP_4 BCP | 0.201 |
| EP_6 BCP | 0.223 |
| EP_8 BCP | 0.235 |
| EP_10 BCP | 0.245 |
| EP_12 BCP | 0.251 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bajpai, A.; Alapati, A.K.; Klingler, A.; Wetzel, B. Tensile Properties, Fracture Mechanics Properties and Toughening Mechanisms of Epoxy Systems Modified with Soft Block Copolymers, Rigid TiO2 Nanoparticles and Their Hybrids. J. Compos. Sci. 2018, 2, 72. https://doi.org/10.3390/jcs2040072
Bajpai A, Alapati AK, Klingler A, Wetzel B. Tensile Properties, Fracture Mechanics Properties and Toughening Mechanisms of Epoxy Systems Modified with Soft Block Copolymers, Rigid TiO2 Nanoparticles and Their Hybrids. Journal of Composites Science. 2018; 2(4):72. https://doi.org/10.3390/jcs2040072
Chicago/Turabian StyleBajpai, Ankur, Arun Kumar Alapati, Andreas Klingler, and Bernd Wetzel. 2018. "Tensile Properties, Fracture Mechanics Properties and Toughening Mechanisms of Epoxy Systems Modified with Soft Block Copolymers, Rigid TiO2 Nanoparticles and Their Hybrids" Journal of Composites Science 2, no. 4: 72. https://doi.org/10.3390/jcs2040072
APA StyleBajpai, A., Alapati, A. K., Klingler, A., & Wetzel, B. (2018). Tensile Properties, Fracture Mechanics Properties and Toughening Mechanisms of Epoxy Systems Modified with Soft Block Copolymers, Rigid TiO2 Nanoparticles and Their Hybrids. Journal of Composites Science, 2(4), 72. https://doi.org/10.3390/jcs2040072







