Room Temperature Humidity Sensor Based on Single β-Ga2O3 Nanowires †
1. Summary
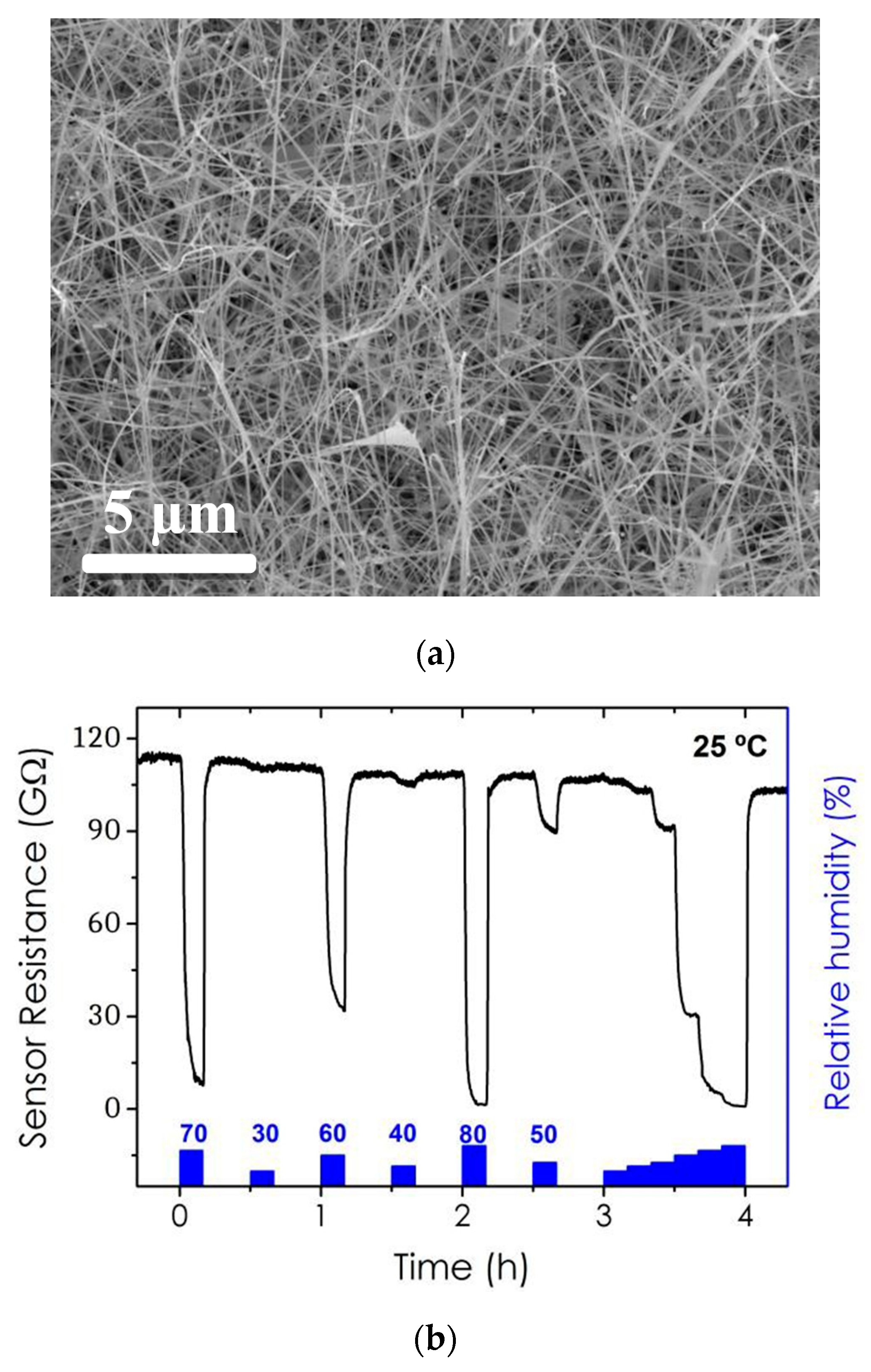
2. Motivation and Results
References
- Fleischer, M.; Meixner, H. Gallium oxide thin films: A new material for high-temperature oxygen sensors. Sens. Act. B Chem. 1991, 4, 437–441. [Google Scholar] [CrossRef]
- Park, S.; Kim, H.; Jin, C.; Lee, C. Synthesis, structure, and room-temperature gas sensing of multiple- networked Pd-doped Ga2O3 nanowires. J. Korean Phys. Soc. 2012, 60, 1560–1564. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Domènech-Gil, G.; Peiró, I.; López-Aymerich, E.; Moreno, M.; Pellegrino, P.; Gràcia, I.; Cané, C.; Barth, S.; Romano-Rodríguez, A. Room Temperature Humidity Sensor Based on Single β-Ga2O3 Nanowires. Proceedings 2018, 2, 958. https://doi.org/10.3390/proceedings2130958
Domènech-Gil G, Peiró I, López-Aymerich E, Moreno M, Pellegrino P, Gràcia I, Cané C, Barth S, Romano-Rodríguez A. Room Temperature Humidity Sensor Based on Single β-Ga2O3 Nanowires. Proceedings. 2018; 2(13):958. https://doi.org/10.3390/proceedings2130958
Chicago/Turabian StyleDomènech-Gil, Guillem, Irmina Peiró, Elena López-Aymerich, Mauricio Moreno, Paolo Pellegrino, Isabel Gràcia, Carles Cané, Sven Barth, and Albert Romano-Rodríguez. 2018. "Room Temperature Humidity Sensor Based on Single β-Ga2O3 Nanowires" Proceedings 2, no. 13: 958. https://doi.org/10.3390/proceedings2130958
APA StyleDomènech-Gil, G., Peiró, I., López-Aymerich, E., Moreno, M., Pellegrino, P., Gràcia, I., Cané, C., Barth, S., & Romano-Rodríguez, A. (2018). Room Temperature Humidity Sensor Based on Single β-Ga2O3 Nanowires. Proceedings, 2(13), 958. https://doi.org/10.3390/proceedings2130958



