3D-Printed, Pocket-Size Diffusion Cells for Skin Permeation Investigation †
Abstract
:1. Introduction
2. Materials and Methods
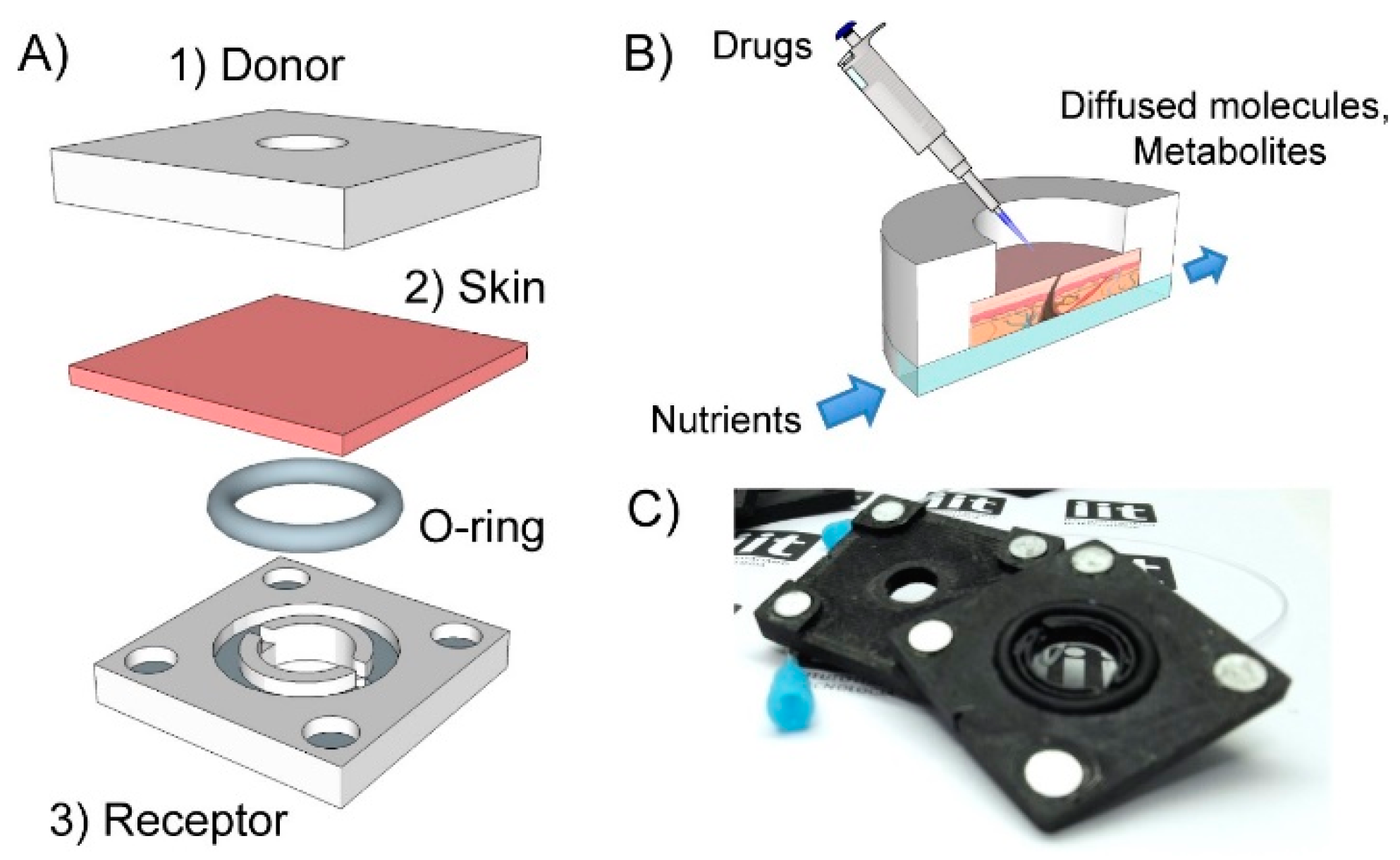
2.1. Fabrication of the 3D-PDC
2.2. Skin Tissue Preparation
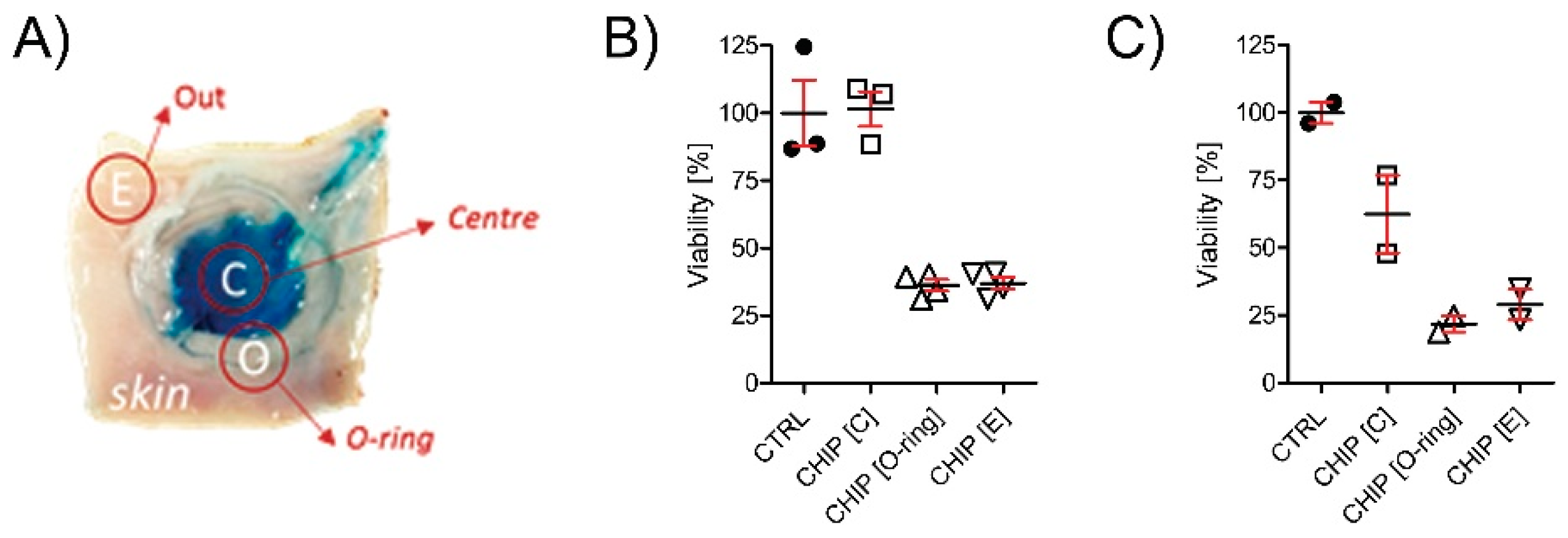
2.3. In Vitro Viability Test
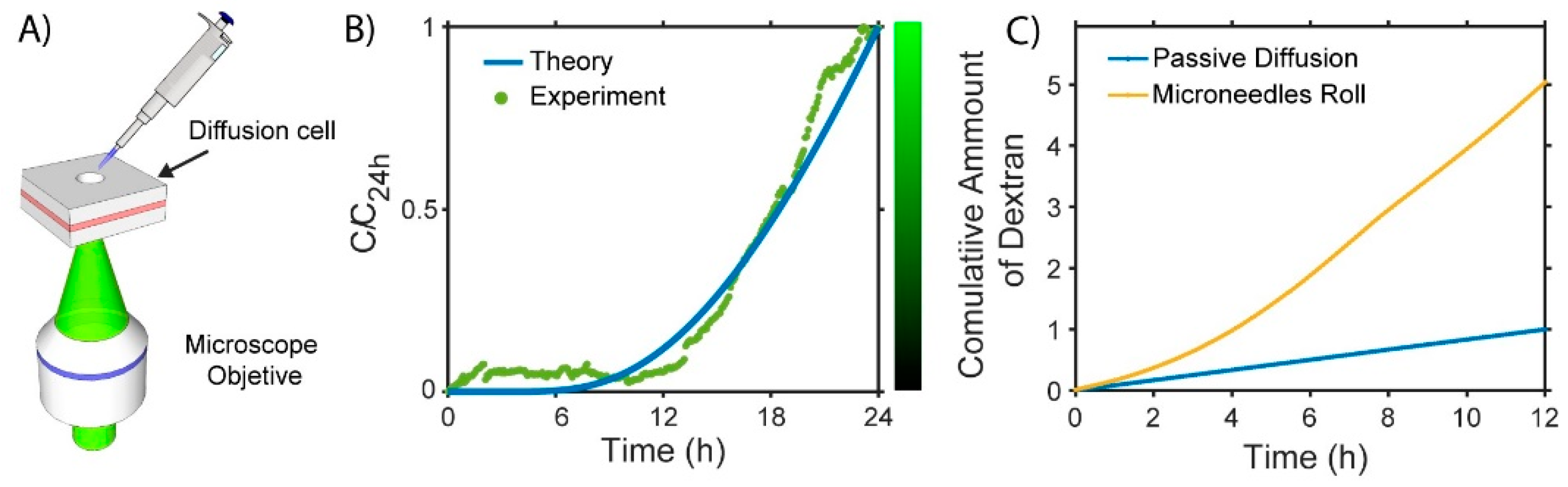
2.4. Diffusivity Analysis
3. Discussion
4. Conclusions and Future Perspectives
Conflicts of Interest
References
- Romgens, A.M.; Bader, D.L.; Bouwstra, J.A.; Baaijens, F.T.P.; Oomens, C.W.J. Diffusion profile of macromolecules within and between human skin layers for (trans)dermal drug delivery. J. Mech. Behav. Biomed. Mat. 2015, 50, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Cornelissen, L.H.; Bronneberg, D.; Oomens, C.W.J.; Baaijens, F.T.P. Diffusion measurements in epidermal tissues with fluorescent recovery after photobleaching. Skin Res. Tech. 2008, 14, 462–467. [Google Scholar] [CrossRef] [PubMed]
- Larese, F.F.; D’Agostin, F.; Crosera, M.; Adami, G.; Renzi, N.; Bovenzi, M.; Maina, G. Human skin penetration of silver nanoparticles through intact and damaged skin. Toxicology 2009, 255, 33–37. [Google Scholar] [CrossRef] [PubMed]
- Franz, T.J. Percutaneous absorption. On the relevance of in vitro data. J. Investig. Dermatol. 1975, 64, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Bartosova, L.; Bajgar, J. Transdermal drug delivery in vitro using diffusion cells. Curr. Med. Chem. 2012, 19, 4671–4677. [Google Scholar] [CrossRef] [PubMed]
- Ng, S.F.; Rouse, J.S.; Sanderson, F.D.; Meidan, V.; Eccleston, G.M. Validation of a static Franz diffusion cell system for in vitro permeation studies. AAPS PharmSciTech 2010, 11, 1432–1441. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suarato, G.; Spanò, R.; Bertorelli, R.; Diaspro, A.; Athanassiou, A.; Surdo, S. 3D-Printed, Pocket-Size Diffusion Cells for Skin Permeation Investigation. Proceedings 2018, 2, 945. https://doi.org/10.3390/proceedings2130945
Suarato G, Spanò R, Bertorelli R, Diaspro A, Athanassiou A, Surdo S. 3D-Printed, Pocket-Size Diffusion Cells for Skin Permeation Investigation. Proceedings. 2018; 2(13):945. https://doi.org/10.3390/proceedings2130945
Chicago/Turabian StyleSuarato, Giulia, Raffaele Spanò, Rosalia Bertorelli, Alberto Diaspro, Athanassia Athanassiou, and Salvatore Surdo. 2018. "3D-Printed, Pocket-Size Diffusion Cells for Skin Permeation Investigation" Proceedings 2, no. 13: 945. https://doi.org/10.3390/proceedings2130945
APA StyleSuarato, G., Spanò, R., Bertorelli, R., Diaspro, A., Athanassiou, A., & Surdo, S. (2018). 3D-Printed, Pocket-Size Diffusion Cells for Skin Permeation Investigation. Proceedings, 2(13), 945. https://doi.org/10.3390/proceedings2130945






