Biochar Applied with Inorganic Nitrogen Improves Soil Carbon, Nitrate and Ammonium Content of a Sandy Loam Temperate Soil
Abstract
:1. Introduction
2. Materials and Methods
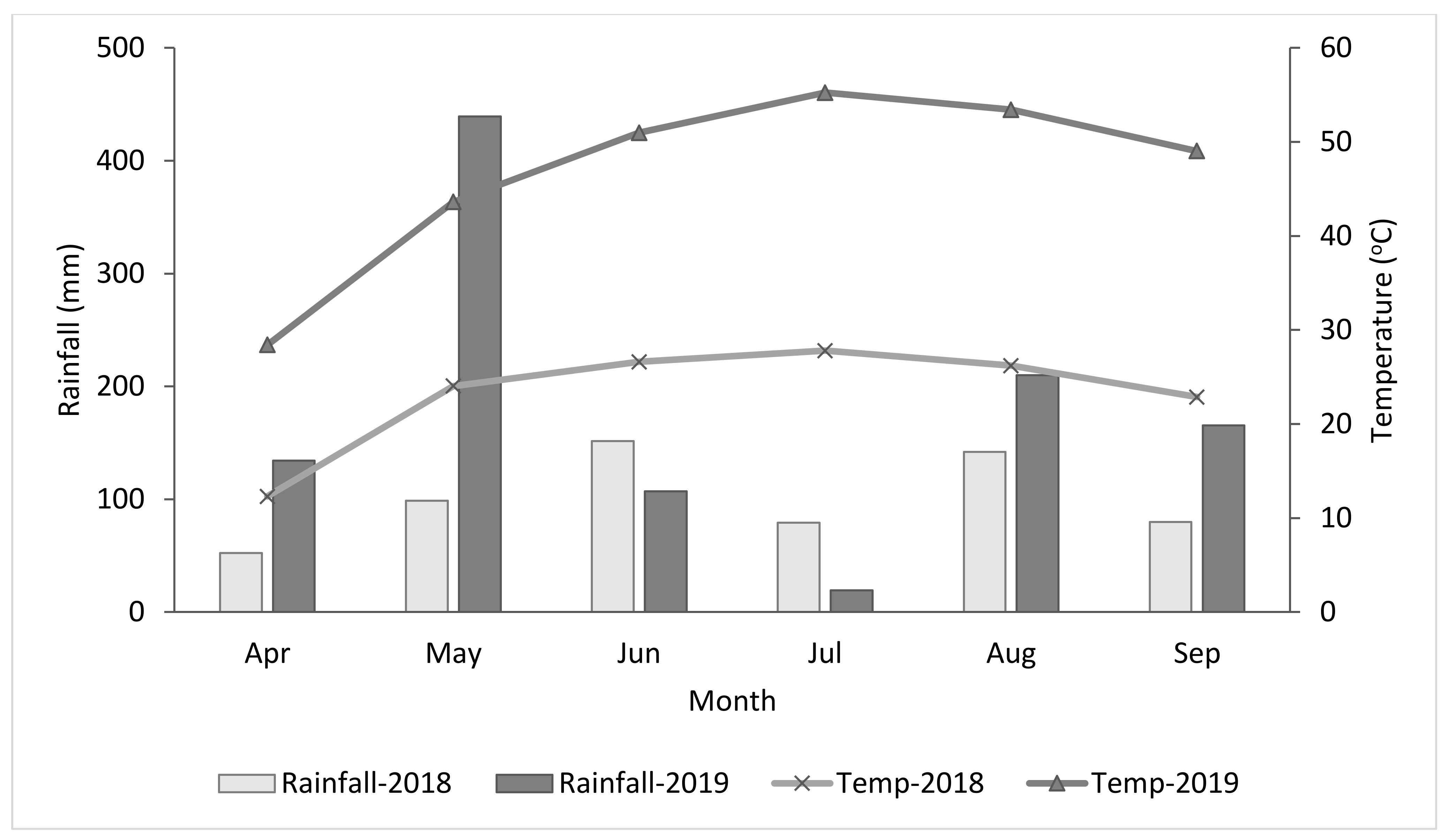
2.1. Experimental Sites and Design
2.2. Data Collection and Analysis
2.3. Statistical Analysis
3. Results
3.1. Soil Nitrate N
3.2. Soil Ammonium N
3.3. Soil Organic C
3.4. Total Soil N
4. Discussion
4.1. Soil Inorganic N
4.2. Soil Organic C
4.3. Total Soil N
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chan, K.Y.; Zwieten, L.V.; Meszaros, I.; Downie, A.; Joseph, S. Agronomic Values of Green Waste Biochar as a Soil Amendment. Soil Res. 2007, 45, 629–634. [Google Scholar] [CrossRef]
- Woolf, D.; Amonette, J.E.; Street-Perrott, F.A.; Lehmann, J.; Joseph, S. Sustainable biochar to mitigate global climate change. Nat. Commun. 2010, 1, 56. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crombie, K.; Mašek, O.; Sohi, S.P.; Brownsort, P.; Cross, A. The Effect of Pyrolysis Conditions on Biochar Stability as Determined by Three Methods. GCB Bioenergy 2013, 5, 122–131. [Google Scholar] [CrossRef] [Green Version]
- Spigarelli, B.P.; Kawatra, S.K. Opportunities and Challenges in Carbon Dioxide Capture. J. CO2 Util. 2013, 1, 69–87. [Google Scholar] [CrossRef]
- Jindo, K.; Mizumoto, H.; Sawada, Y.; Sonoki, T. Physical and Chemical Characterization of Biochars Derived from Different Agricultural Residues. Biogeosciences 2014, 11, 6613–6621. [Google Scholar] [CrossRef] [Green Version]
- Stavins, R.N. The Costs of Carbon Sequestration: A Revealed-Preference Approach. Am. Econ. Rev. 2017, 89, 994–1009. [Google Scholar] [CrossRef] [Green Version]
- Singh, B.P.; Hatton, B.J.; Singh, B.; Cowie, A.L.; Kathuria, A. Influence of Biochars on Nitrous Oxide Emission and Nitrogen Leaching from Two Contrasting Soils. J. Environ. Qual. 2010, 39, 1224. [Google Scholar] [CrossRef]
- Atkinson, C.J.; Fitzgerald, J.D.; Hipps, N.A. Potential Mechanisms for Achieving Agricultural Benefits from Biochar Application to Temperate Soils: A Review. Plant Soil 2010, 337, 1–18. [Google Scholar] [CrossRef]
- Aula, L.; Macnack, N.; Omara, P.; Mullock, J.; Raun, W.R. Effect of Fertilizer Nitrogen on Soil Organic Carbon, Total Nitrogen, and Soil pH in Long-Term Continuous Winter Wheat (Triticum aestivum L.). Commun. Soil Sci. Plant Anal. 2016, 47, 863–874. [Google Scholar] [CrossRef]
- Wardle, D.A.; Nilsson, M.C.; Zackrisson, O. Response to Comment on Fire-Derived Charcoal Causes Loss of Forest Humus. Science 2008, 321, 1295. [Google Scholar] [CrossRef] [Green Version]
- Jones, D.L.; Murphy, D.V.; Khalid, M.; Ahmad, W.; Edwards-Jones, G.; DeLuca, T.H. Short-Term Biochar-Induced Increase in Soil CO2 Release Is Both Biotically and Abiotically Mediated. Soil Biol. Biochem. 2011, 43, 1723–1731. [Google Scholar] [CrossRef]
- Cross, A.; Sohi, S.P. The priming potential of biochar products in relation to labile carbon contents and soil organic matter status. Soil Boil. Biochem. 2011, 43, 2127–2134. [Google Scholar] [CrossRef]
- Kimetu, J.M.; Lehmann, J. Stability and Stabilization of Biochar and Green Manure in Soil with Different Organic Carbon Contents. Soil Res. 2010, 48, 577–585. [Google Scholar] [CrossRef]
- Jones, C.M.; Kammen, D.M. Quantifying Carbon Footprint Reduction Opportunities for U.S. Households and Communities. Environ. Sci. Technol. 2011, 45, 4088–4095. [Google Scholar] [CrossRef] [PubMed]
- Howarth, R.W. Nitrogen in Freshwater Systems and Estuaries Nitrogen. In Reference Module in Earth Systems and Environmental Sciences; Elsevier: Amsterdam, The Netherlands, 2014. [Google Scholar] [CrossRef]
- Prommer, J.; Wanek, W.; Hofhansl, F.; Trojan, D.; Offre, P.; Urich, T.; Schleper, C.; Sassmann, S.; Kitzler, B.; Soja, G.; et al. Biochar decelerates soil organic nitrogen cycling but stimulates soil nitrification in a temperate arable field trial. PLoS ONE 2014, 9, 86388. [Google Scholar] [CrossRef] [PubMed]
- Bai, S.H.; Reverchon, F.; Xu, C.Y.; Xu, Z.; Blumfield, T.J.; Zhao, H.; Van Zwieten, L.; Wallace, H.M. Wood biochar increases nitrogen retention in field settings mainly through abiotic processes. Soil Biol. Biochem. 2015, 90, 232–240. [Google Scholar] [CrossRef]
- Raun, W.R.; Johnson, G.V. Improving Nitrogen Use Efficiency for Cereal Production. Agron. J. 1999, 91, 357–363. [Google Scholar] [CrossRef] [Green Version]
- Fageria, N.K.; Baligar, V.C. Enhancing nitrogen use efficiency in crop plants. Adv. Agron. 2005, 88, 97–185. [Google Scholar] [CrossRef]
- Omara, P.; Aula, L.; Oyebiyi, F.B.; Raun, W.R. World Cereal Nitrogen Use Efficiency Trends: Review and Current Knowledge. Agrosyst. Geosci. Environ. 2019, 2, 1–8. [Google Scholar] [CrossRef]
- Uzoma, K.C.; Inoue, M.; Andry, H.; Fujimaki, H.; Zahoor, A.; Nishihara, E. Effect of cow manure biochar on maize productivity under sandy soil condition. Soil Use Manag. 2011, 27, 205–212. [Google Scholar] [CrossRef]
- Jiang, T.Y.; Jiang, J.; Xu, R.K.; Li, Z. Adsorption of Pb (II) on variable charge soils amended with rice-straw derived biochar. Chemosphere 2012, 89, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Bruun, E.W.; Petersen, C.T.; Hansen, E.; Holm, J.K.; Hauggaard-Nielsen, H. Biochar amendment to coarse sandy subsoil improves root growth and increases water retention. Soil Use Manag. 2014, 30, 109–118. [Google Scholar] [CrossRef]
- Lawrinenko, M.; Laird, D.A. Anion exchange capacity of biochar. Green Chem. 2015, 17, 4628–4636. [Google Scholar] [CrossRef] [Green Version]
- Agegnehu, G.; Bass, A.M.; Nelson, P.N.; Bird, M.I. Benefits of biochar, compost and biochar–compost for soil quality, maize yield and greenhouse gas emissions in a tropical agricultural soil. Sci. Total Environ. 2016, 543, 295–306. [Google Scholar] [CrossRef] [PubMed]
- Soil Survey Staff. Natural Resources Conservation Service, United States Department of Agriculture. Web Soil Survey. Available online: http://websoilsurvey.sc.egov.usda.gov/ (accessed on 24 June 2020).
- Schepers, J.S.; Francis, D.D.; Thompson, M.T. Simultaneous determination of total C, total N and 15N on soil and plant material. Commun. Soil Sci. Plant Anal. 1989, 20, 949–959. [Google Scholar] [CrossRef]
- SAS Institute Inc. Base SAS 9.4 Procedures Guide: Statistical Procedures, 2nd ed.; SAS Institute Inc.: Cary, NC, USA, 2013. [Google Scholar]
- Nogueira, M.C.S. Orthogonal contrasts: Definitions and concepts. Sci. Agric. 2004, 61, 118–124. [Google Scholar] [CrossRef] [Green Version]
- Abdi, H.; Williams, L.J. Contrast analysis. Encycl. Res. Des. 2010, 1, 243–251. [Google Scholar]
- Yao, Y.; Gao, B.; Zhang, M.; Inyang, M.; Zimmerman, A.R. Effect of Biochar Amendment on Sorption and Leaching of Nitrate, Ammonium, and Phosphate in a Sandy Soil. Chemosphere 2012, 89, 1467–1471. [Google Scholar] [CrossRef]
- Libutti, A.; Mucci, M.; Francavilla, M.; Monteleone, M. Effect of biochar amendment on nitrate recovery in a silty clay loam soil. Ital. J. Agron. 2016, 11, 273–276. [Google Scholar] [CrossRef] [Green Version]
- Gao, S.; Hoffman-Krull, K.; Bidwell, A.L.; DeLuca, T.H. Locally produced wood biochar increases nutrient retention and availability in agricultural soils of the San Juan Islands, USA. Agric. Ecosyst. Environ. 2016, 233, 43–54. [Google Scholar] [CrossRef]
- Zheng, H.; Wang, Z.; Deng, X.; Herbert, S.; Xing, B. Impacts of adding biochar on nitrogen retention and bioavailability in agricultural soil. Geoderma 2013, 206, 32–39. [Google Scholar] [CrossRef]
- Libutti, A.; Cammerino, A.R.B.; Francavilla, M.; Monteleone, M. Soil amendment with biochar affects water drainage and nutrient losses by leaching: Experimental evidence under field-grown conditions. Agronomy 2019, 9, 758. [Google Scholar] [CrossRef] [Green Version]
- Liang, B.; Lehmann, J.; Solomon, D.; Kinyangi, J.; Grossman, J.; O’Neill, B.; Skjemstad, J.O.; Thies, J.; Luizão, F.J.; Petersen, J.; et al. Black carbon increases cation exchange capacity in soils. Soil Sci. Soc. Am. J. 2006, 70, 1719–1730. [Google Scholar] [CrossRef] [Green Version]
- Laird, D.A.; Fleming, P.; Davis, D.D.; Horton, R.; Wang, B.; Karlen, D.L. Impact of Biochar Amendments on the Quality of a Typical Midwestern Agricultural Soil. Geoderma 2010, 158, 443–449. [Google Scholar] [CrossRef] [Green Version]
- Liu, S.; Zhang, Y.; Zong, Y.; Hu, Z.; Wu, S.; Zhou, J.; Jin, Y.; Zou, J. Response of soil carbon dioxide fluxes, soil organic carbon and microbial biomass carbon to biochar amendment: A meta-analysis. GCB Bioenergy 2016, 8, 392–406. [Google Scholar] [CrossRef]
- Zhang, M.; Cheng, G.; Feng, H.; Sun, B.; Zhao, Y.; Chen, H.; Chen, J.; Dyck, M.; Wang, X.; Zhang, J.; et al. Effects of straw and biochar amendments on aggregate stability, soil organic carbon, and enzyme activities in the Loess Plateau, China. Environ. Sci. Pollut. Res. 2017, 24, 10108–10120. [Google Scholar] [CrossRef]
- Lorenz, K.; Lal, R. Biochar application to soil for climate change mitigation by soil organic carbon sequestration. J. Plant Nutri. Soil Sci. 2014, 177, 651–670. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Xiong, Z.; Kuzyakov, Y. Biochar stability in soil: Meta-analysis of decomposition and priming effects. GCB Bioenergy 2016, 8, 512–523. [Google Scholar] [CrossRef] [Green Version]
- Omara, P.; Aula, L.; Eickhoff, E.M.; Dhillon, J.S.; Lynch, T.; Wehmeyer, G.B.; Raun, W.R. Influence of No-Tillage on Soil Organic Carbon, Total Soil Nitrogen, and Winter Wheat (Triticum aestivum L.) Grain Yield. Int. J. Agron. 2019, 2019, 9632969. [Google Scholar] [CrossRef] [Green Version]
| Biochar/ Site | pH | K | Ca | Mg | Mn | Fe | BD | TP | TN | TOC |
|---|---|---|---|---|---|---|---|---|---|---|
| Unit | mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | g cm−1 | mg kg−1 | g kg−1 | g kg−1 | |
| Biochar | 7.4 | 612 | 4128 | 1225 | 234 | 595 | 0.48 | 4.53 | 5.9 | 876.7 |
| LCB | 5.7 | 349 | 804 | 207 | x | x | x | 12 | 0.8 | 9.1 |
| Efaw | 5.6 | 153 | 1466 | 354 | x | x | x | 13 | 0.7 | 6.8 |
| Treatment | N Rate | Biochar | NO3−–N at Efaw | NO3−–N at LCB | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| kg ha−1 | t ha−1 | mg kg−1 | mg kg−1 | |||||||
| 2018 | 2019 | 2018 | 2019 | |||||||
| mean | ±S.E | Mean | ±S.E | mean | ±S.E | mean | ±S.E | |||
| 1 | 0 | 0 | 4.24 | 0.42 | 4.05 | 0.24 | 2.13 | 0.24 | 3.71 | 0.71 |
| 2 | 50 | 0 | 4.76 | 0.14 | 4.81 | 0.54 | 2.66 | 0.17 | 3.94 | 0.21 |
| 3 | 100 | 0 | 4.94 | 0.23 | 5.33 | 0.11 | 2.76 | 0.18 | 4.39 | 0.17 |
| 4 | 150 | 0 | 6.38 | 0.22 | 5.88 | 0.18 | 1.96 | 0.06 | 5.11 | 0.28 |
| 5 | 0 | 5 | 4.45 | 0.70 | 4.05 | 0.02 | 2.15 | 0.17 | 3.95 | 0.09 |
| 6 | 0 | 10 | 3.80 | 0.39 | 4.22 | 0.03 | 2.14 | 0.06 | 4.00 | 0.11 |
| 7 | 0 | 15 | 4.02 | 0.63 | 4.18 | 0.12 | 2.24 | 0.26 | 4.09 | 0.07 |
| 8 | 50 | 5 | 5.01 | 0.35 | 4.58 | 0.04 | 2.98 | 0.03 | 4.67 | 0.70 |
| 9 | 100 | 10 | 5.29 | 0.32 | 4.88 | 0.17 | 3.87 | 0.26 | 5.68 | 0.21 |
| 10 | 150 | 15 | 4.86 | 0.85 | 5.58 | 0.01 | 3.27 | 0.12 | 6.98 | 0.88 |
| Pr > F | 0.0534 | <0.0001 | <0.0001 | 0.001 | ||||||
| C.V, % | 17.3 | 7.7 | 11.5 | 16.6 | ||||||
| Contrasts | F | Pr > F | F | Pr > F | F | Pr > F | F | Pr > F | ||
| 2 vs. 8 | 0.17 | 0.6840 | 0.43 | 0.5222 | 2.13 | 0.1702 | 1.11 | 0.3134 | ||
| 3 vs. 9 | 0.35 | 0.5640 | 1.63 | 0.2262 | 25.02 | 0.0003 | 3.42 | 0.0891 | ||
| 4 vs. 10 | 6.45 | 0.0259 | 0.74 | 0.4067 | 34.60 | <0.0001 | 7.18 | 0.0200 | ||
| 2, 3 & 4 vs. 8, 9 & 10 | 0.78 | 0.3944 | 2.60 | 0.1326 | 50.79 | <0.0001 | 10.39 | 0.0073 | ||
| Treatment | N Rate | Biochar | NH4+–N at Efaw | NH4+–N at LCB | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| kg ha−1 | t ha−1 | mg kg−1 | mg kg−1 | |||||||
| 2018 | 2019 | 2018 | 2019 | |||||||
| mean | ±S.E | Mean | ±S.E | mean | ±S.E | mean | ±S.E | |||
| 1 | 0 | 0 | 17.63 | 0.59 | 4.21 | 0.07 | 24.43 | 1.23 | 4.06 | 0.03 |
| 2 | 50 | 0 | 18.53 | 1.23 | 5.01 | 0.27 | 24.42 | 1.13 | 4.19 | 0.16 |
| 3 | 100 | 0 | 19.33 | 1.22 | 5.15 | 0.29 | 24.25 | 0.78 | 4.24 | 0.14 |
| 4 | 150 | 0 | 20.72 | 1.25 | 5.51 | 0.38 | 26.32 | 0.86 | 4.43 | 0.09 |
| 5 | 0 | 5 | 19.70 | 2.47 | 3.87 | 0.32 | 24.76 | 2.27 | 4.10 | 0.01 |
| 6 | 0 | 10 | 19.03 | 0.81 | 4.26 | 0.10 | 23.60 | 0.61 | 4.11 | 0.05 |
| 7 | 0 | 15 | 19.26 | 3.53 | 4.27 | 0.06 | 23.98 | 1.11 | 4.09 | 0.06 |
| 8 | 50 | 5 | 20.17 | 1.53 | 4.40 | 0.11 | 25.90 | 1.44 | 4.22 | 0.16 |
| 9 | 100 | 10 | 21.15 | 2.08 | 4.49 | 0.15 | 28.15 | 0.67 | 4.59 | 0.17 |
| 10 | 150 | 15 | 20.07 | 2.54 | 4.82 | 0.18 | 30.52 | 1.38 | 5.21 | 0.15 |
| Pr > F | 0.9268 | 0.0009 | 0.016 | <0.0001 | ||||||
| C.V, % | 14.9 | 8.3 | 8.4 | 4.6 | ||||||
| Contrasts | F | Pr > F | F | Pr > F | F | Pr > F | F | Pr > F | ||
| 2 vs. 8 | 0.46 | 0.5121 | 3.09 | 0.1044 | 0.93 | 0.3546 | 0.02 | 0.8881 | ||
| 3 vs. 9 | 0.56 | 0.4676 | 3.51 | 0.0854 | 6.45 | 0.0260 | 3.02 | 0.1078 | ||
| 4 vs. 10 | 0.07 | 0.7953 | 3.95 | 0.0702 | 7.47 | 0.0182 | 14.31 | 0.0026 | ||
| 2, 3 & 4 vs. 8, 9 & 10 | 0.45 | 0.5156 | 10.52 | 0.0070 | 12.96 | 0.0036 | 10.70 | 0.0067 | ||
| Treatment | N Rate | Biochar | Soil Organic C at Efaw | Soil Organic C at LCB | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| kg ha−1 | t ha−1 | g kg−1 | g kg−1 | |||||||
| 2018 | 2019 | 2018 | 2019 | |||||||
| mean | ±S.E | Mean | ±S.E | Mean | ±S.E | mean | ±S.E | |||
| 1 | 0 | 0 | 6.76 | 0.35 | 6.86 | 0.43 | 9.14 | 0.98 | 8.53 | 0.19 |
| 2 | 50 | 0 | 7.05 | 0.10 | 7.25 | 0.44 | 8.24 | 0.21 | 8.23 | 0.21 |
| 3 | 100 | 0 | 6.76 | 0.37 | 7.13 | 0.13 | 8.40 | 0.16 | 8.58 | 0.16 |
| 4 | 150 | 0 | 6.61 | 0.25 | 7.34 | 0.31 | 8.76 | 0.12 | 10.39 | 0.37 |
| 5 | 0 | 5 | 7.79 | 0.06 | 8.48 | 1.13 | 10.23 | 1.60 | 10.48 | 0.46 |
| 6 | 0 | 10 | 8.69 | 1.03 | 10.09 | 0.55 | 9.79 | 0.19 | 9.32 | 0.73 |
| 7 | 0 | 15 | 9.37 | 0.20 | 7.03 | 0.36 | 10.46 | 1.38 | 11.18 | 1.40 |
| 8 | 50 | 5 | 7.39 | 0.38 | 7.55 | 0.63 | 9.64 | 0.09 | 9.58 | 0.57 |
| 9 | 100 | 10 | 9.22 | 0.96 | 9.37 | 0.58 | 10.91 | 0.71 | 10.11 | 0.25 |
| 10 | 150 | 15 | 9.58 | 0.64 | 11.01 | 0.99 | 12.23 | 1.04 | 12.72 | 0.72 |
| Pr > F | 0.0016 | 0.0007 | 0.0758 | 0.0015 | ||||||
| C.V, % | 11.8 | 13.2 | 14.9 | 10.9 | ||||||
| Contrasts | F | Pr > F | F | Pr > F | F | Pr > F | F | Pr > F | ||
| 2 vs. 8 | 0.21 | 0.6542 | 0.14 | 0.7147 | 3.50 | 0.0858 | 5.21 | 0.0415 | ||
| 3 vs. 9 | 10.87 | 0.0064 | 7.50 | 0.0180 | 11.22 | 0.0058 | 6.65 | 0.0241 | ||
| 4 vs. 10 | 15.79 | 0.0018 | 20.11 | 0.0007 | 21.51 | 0.0006 | 5.76 | 0.0335 | ||
| 2, 3 & 4 vs. 8, 9 & 10 | 19.92 | 0.0008 | 19.24 | 0.0009 | 28.27 | 0.0002 | 17.58 | 0.0012 | ||
| Treatment | N Rate | Biochar | Total Soil N at Efaw | Total Soil N at LCB | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| kg ha−1 | t ha−1 | g kg−1 | g kg−1 | |||||||
| 2018 | 2019 | 2018 | 2019 | |||||||
| mean | ±S.E | Mean | ±S.E | mean | ±S.E | mean | ±S.E | |||
| 1 | 0 | 0 | 0.71 | 0.01 | 0.79 | 0.04 | 0.80 | 0.03 | 0.79 | 0.03 |
| 2 | 50 | 0 | 0.82 | 0.06 | 0.74 | 0.05 | 0.76 | 0.04 | 0.77 | 0.04 |
| 3 | 100 | 0 | 0.70 | 0.05 | 0.80 | 0.06 | 0.75 | 0.08 | 0.75 | 0.02 |
| 4 | 150 | 0 | 0.68 | 0.01 | 0.73 | 0.04 | 0.78 | 0.00 | 0.81 | 0.06 |
| 5 | 0 | 5 | 0.78 | 0.02 | 0.79 | 0.04 | 0.73 | 0.03 | 0.85 | 0.03 |
| 6 | 0 | 10 | 0.72 | 0.04 | 0.81 | 0.04 | 0.84 | 0.04 | 0.77 | 0.01 |
| 7 | 0 | 15 | 0.73 | 0.01 | 0.72 | 0.03 | 0.78 | 0.04 | 0.87 | 0.00 |
| 8 | 50 | 5 | 0.71 | 0.02 | 0.83 | 0.04 | 0.82 | 0.05 | 0.81 | 0.05 |
| 9 | 100 | 10 | 0.75 | 0.04 | 0.77 | 0.04 | 0.82 | 0.03 | 0.84 | 0.03 |
| 10 | 150 | 15 | 0.71 | 0.06 | 0.80 | 0.05 | 0.82 | 0.05 | 0.78 | 0.02 |
| Pr > F | 0.3316 | 0.6854 | 0.6466 | 0.2424 | ||||||
| C.V, % | 8.8 | 9.7 | 9 | 7.2 | ||||||
| Contrasts | F | Pr > F | F | Pr > F | F | Pr > F | F | Pr > F | ||
| 2 vs. 8 | 2.77 | 0.1221 | 1.62 | 0.2268 | 0.85 | 0.3740 | 0.39 | 0.5462 | ||
| 3 vs. 9 | 0.49 | 0.4954 | 0.16 | 0.6990 | 1.39 | 0.2609 | 2.63 | 0.1309 | ||
| 4 vs. 10 | 0.30 | 0.5921 | 1.32 | 0.2730 | 0.37 | 0.5533 | 0.38 | 0.5512 | ||
| 2, 3 & 4 vs. 8, 9 & 10 | 0.06 | 0.8170 | 1.37 | 0.2647 | 2.45 | 0.1432 | 0.88 | 0.3654 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Omara, P.; Aula, L.; Otim, F.; Obia, A.; Souza, J.L.B.; Arnall, D.B. Biochar Applied with Inorganic Nitrogen Improves Soil Carbon, Nitrate and Ammonium Content of a Sandy Loam Temperate Soil. Nitrogen 2022, 3, 90-100. https://doi.org/10.3390/nitrogen3010007
Omara P, Aula L, Otim F, Obia A, Souza JLB, Arnall DB. Biochar Applied with Inorganic Nitrogen Improves Soil Carbon, Nitrate and Ammonium Content of a Sandy Loam Temperate Soil. Nitrogen. 2022; 3(1):90-100. https://doi.org/10.3390/nitrogen3010007
Chicago/Turabian StyleOmara, Peter, Lawrence Aula, Fred Otim, Alfred Obia, Joao Luis Bigatao Souza, and Daryl Brain Arnall. 2022. "Biochar Applied with Inorganic Nitrogen Improves Soil Carbon, Nitrate and Ammonium Content of a Sandy Loam Temperate Soil" Nitrogen 3, no. 1: 90-100. https://doi.org/10.3390/nitrogen3010007
APA StyleOmara, P., Aula, L., Otim, F., Obia, A., Souza, J. L. B., & Arnall, D. B. (2022). Biochar Applied with Inorganic Nitrogen Improves Soil Carbon, Nitrate and Ammonium Content of a Sandy Loam Temperate Soil. Nitrogen, 3(1), 90-100. https://doi.org/10.3390/nitrogen3010007








