An Integrative Explainable Artificial Intelligence Approach to Analyze Fine-Scale Land-Cover and Land-Use Factors Associated with Spatial Distributions of Place of Residence of Reported Dengue Cases
Abstract
1. Introduction
2. Materials and Methods
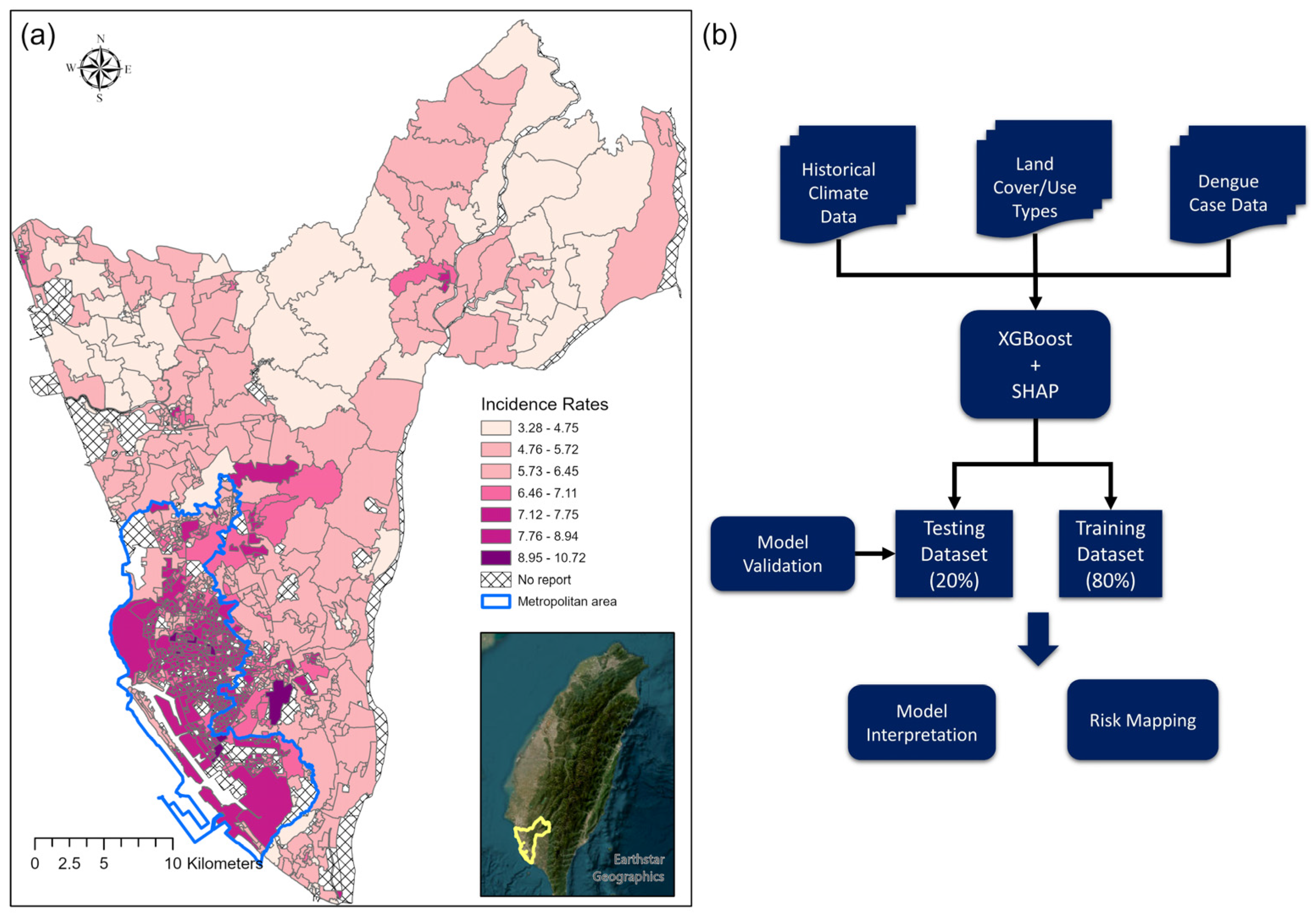
2.1. Study Area
2.2. Dengue Data
2.3. Landscape-Level Variables
2.4. Modeling Approaches
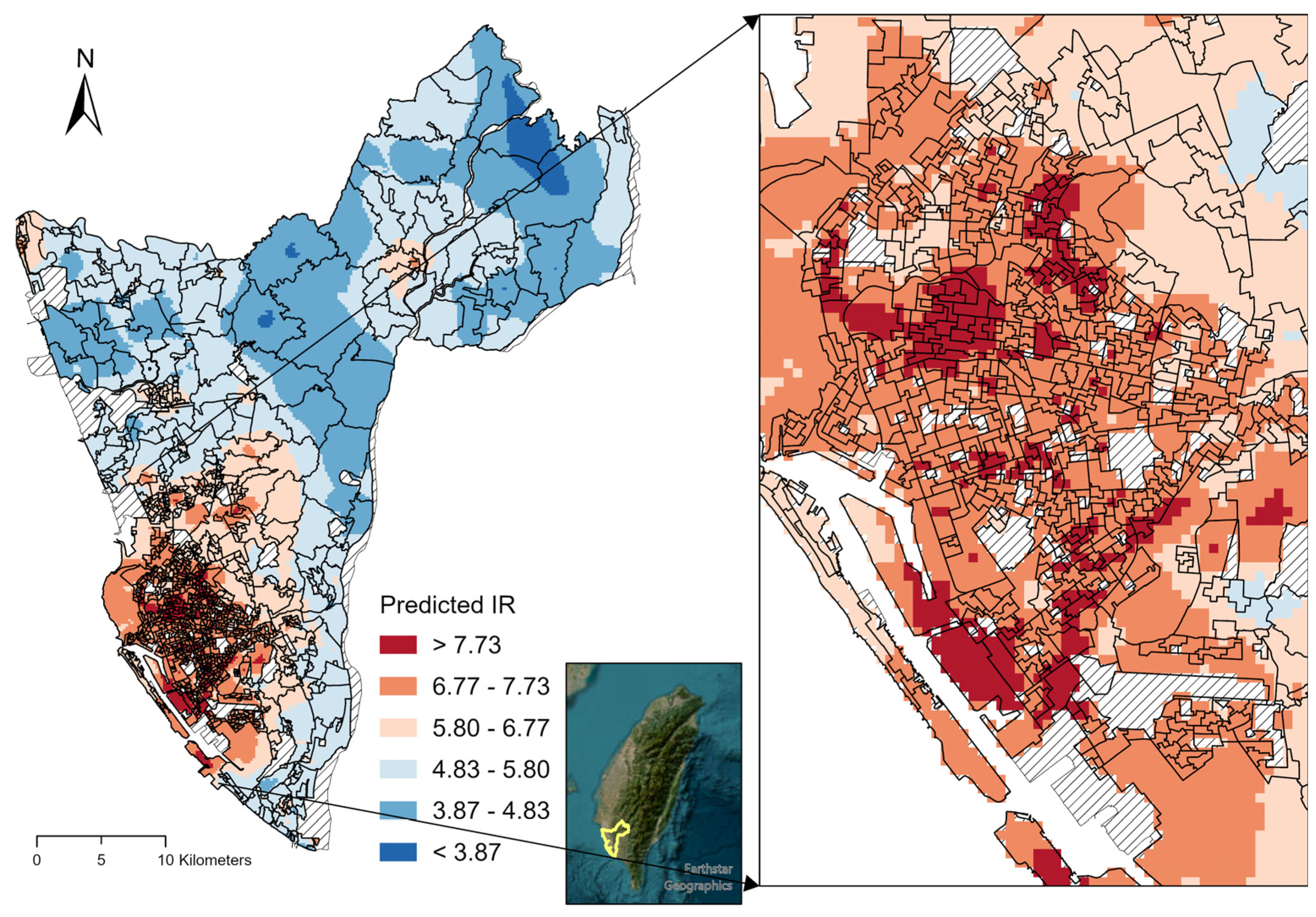
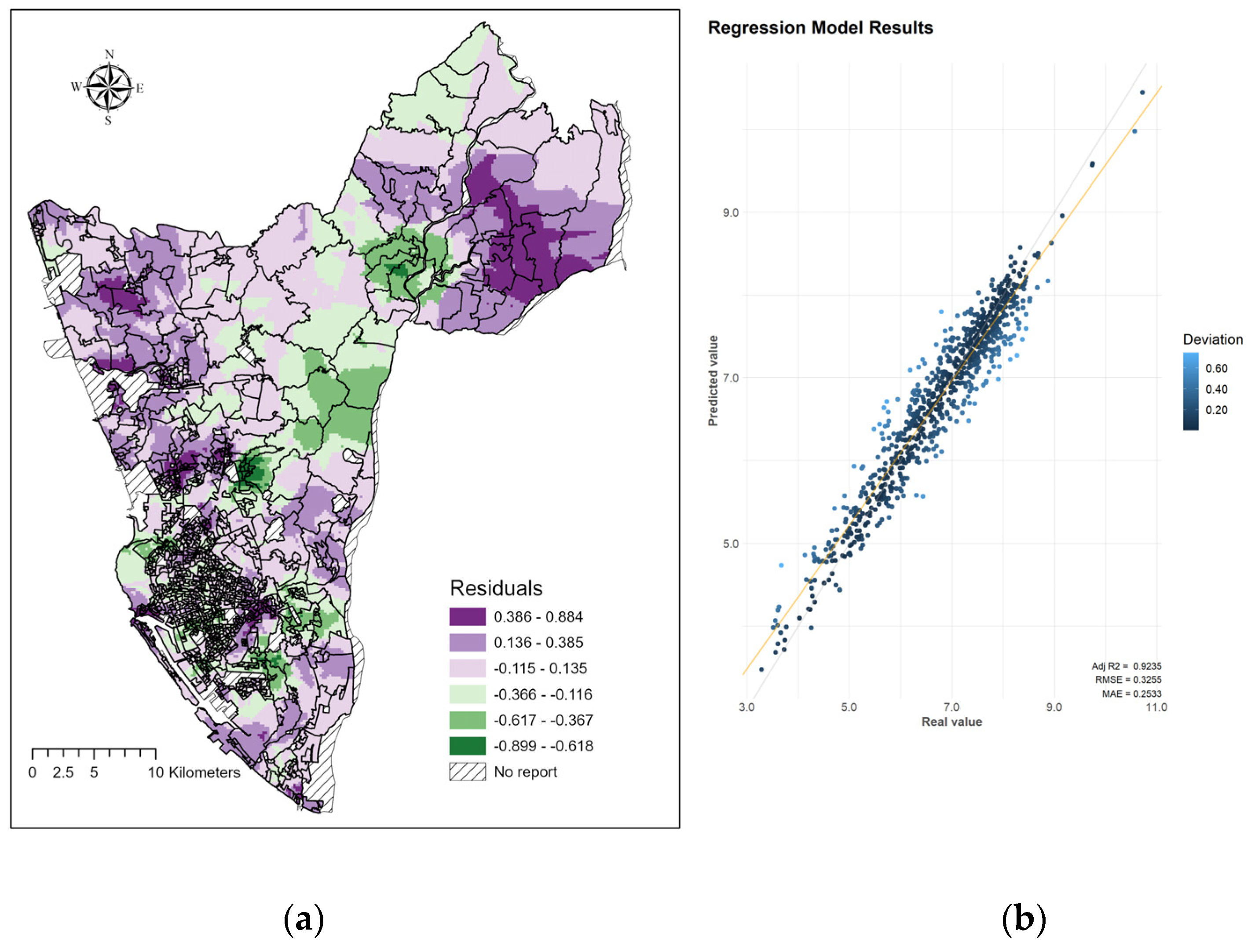
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ryan, S.J.; Carlson, C.J.; Mordecai, E.A.; Johnson, L.R. Global expansion and redistribution of Aedes-borne virus transmission risk with climate change. PLoS Negl. Trop. Dis. 2019, 13, e0007213. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, S.; Gething, P.W.; Brady, O.J.; Messina, J.P.; Farlow, A.W.; Moyes, C.L.; Drake, J.M.; Brownstein, J.S.; Hoen, A.G.; Sankoh, O.; et al. The global distribution and burden of dengue. Nature 2013, 496, 504–507. [Google Scholar] [CrossRef] [PubMed]
- Brady, O.J.; Gething, P.W.; Bhatt, S.; Messina, J.P.; Brownstein, J.S.; Hoen, A.G.; Moyes, C.L.; Farlow, A.W.; Scott, T.W.; Hay, S.I. Refining the global spatial limits of dengue virus transmission by evidence-based consensus. PLoS Negl. Trop. Dis. 2012, 6, e1760. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Dengue Guidelines for Diagnosis, Treatment, Prevention and Control, New Edition; WHO Press: Geneva, Switzerland, 2009. [Google Scholar]
- Simmons, C.P.; Farrar, J.J.; Nguyen, V.; Wills, B. Dengue. N. Engl. J. Med. 2012, 366, 1423–1432. [Google Scholar] [CrossRef]
- Stanaway, J.D.; Shepard, D.S.; Undurraga, E.A.; Halasa, Y.A.; Coffeng, L.E.; Brady, O.J.; Hay, S.I.; Bedi, N.; Bensenor, I.M.; Castaneda-Orjuela, C.A.; et al. The global burden of dengue: An analysis from the Global Burden of Disease Study 2013. Lancet Infect. Dis. 2016, 16, 712–723. [Google Scholar] [CrossRef]
- Paz-Bailey, G.; Adams, L.; Wong, J.M.; Poehling, K.A.; Chen, W.H.; McNally, V.; Atmar, R.L.; Waterman, S.H. Dengue vaccine: Recommendations of the Advisory Committee on Immunization Practices, United States, 2021. MMWR Recomm. Rep. 2021, 70, 1–16. [Google Scholar] [CrossRef]
- Thomson, M.C.; Stanberry, L.R. Climate change and vectorborne diseases. N. Engl. J. Med. 2022, 387, 1969–1978. [Google Scholar] [CrossRef]
- Dostal, T.; Meisner, J.; Munayco, C.; Garcia, P.J.; Carcamo, C.; Perez Lu, J.E.; Morin, C.; Frisbie, L.; Rabinowitz, P.M. The effect of weather and climate on dengue outbreak risk in Peru, 2000–2018: A time-series analysis. PLoS Negl. Trop. Dis. 2022, 16, e0010479. [Google Scholar] [CrossRef]
- Nguyen, V.H.; Tuyet-Hanh, T.T.; Mulhall, J.; Minh, H.V.; Duong, T.Q.; Chien, N.V.; Nhung, N.T.T.; Lan, V.H.; Minh, H.B.; Cuong, D.; et al. Deep learning models for forecasting dengue fever based on climate data in Vietnam. PLoS Negl. Trop. Dis. 2022, 16, e0010509. [Google Scholar] [CrossRef]
- Nguyen, L.T.; Le, H.X.; Nguyen, D.T.; Ho, H.Q.; Chuang, T.W. Impact of climate variability and abundance of mosquitoes on dengue transmission in central Vietnam. Int. J. Environ Res. Public Health 2020, 17, 2453. [Google Scholar] [CrossRef]
- Akter, R.; Hu, W.; Gatton, M.; Bambrick, H.; Naish, S.; Tong, S. Different responses of dengue to weather variability across climate zones in Queensland, Australia. Environ Res. 2020, 184, 109222. [Google Scholar] [CrossRef] [PubMed]
- Lowe, R.; Coelho, C.A.; Barcellos, C.; Carvalho, M.S.; Catao Rde, C.; Coelho, G.E.; Ramalho, W.M.; Bailey, T.C.; Stephenson, D.B.; Rodo, X. Evaluating probabilistic dengue risk forecasts from a prototype early warning system for Brazil. Elife 2016, 5, e11285. [Google Scholar] [CrossRef]
- Wang, Y.; Wei, Y.; Li, K.; Jiang, X.; Li, C.; Yue, Q.; Zee, B.C.; Chong, K.C. Impact of extreme weather on dengue fever infection in four Asian countries: A modelling analysis. Environ Int. 2022, 169, 107518. [Google Scholar] [CrossRef] [PubMed]
- Carreto, C.; Gutierrez-Romero, R.; Rodriguez, T. Climate-driven mosquito-borne viral suitability index: Measuring risk transmission of dengue, chikungunya and Zika in Mexico. Int. J. Health Geogr. 2022, 21, 15. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Liu, T.; Lin, H.; Zhu, G.; Zeng, W.; Li, X.; Zhang, B.; Song, T.; Deng, A.; Zhang, M.; et al. Weather variables and the El Nino Southern Oscillation may drive the epidemics of dengue in Guangdong Province, China. Sci. Total Environ. 2018, 624, 926–934. [Google Scholar] [CrossRef]
- Chuang, T.W.; Chaves, L.F.; Chen, P.J. Effects of local and regional climatic fluctuations on dengue outbreaks in southern Taiwan. PLoS ONE 2017, 12, e0178698. [Google Scholar] [CrossRef]
- Munoz, E.; Poveda, G.; Arbelaez, M.P.; Velez, I.D. Spatiotemporal dynamics of dengue in Colombia in relation to the combined effects of local climate and ENSO. Acta Trop. 2021, 224, 106136. [Google Scholar] [CrossRef]
- Temenos, A.; Tzortzis, I.N.; Kaselimi, M.; Rallis, I.; Doulamis, A.; Doulamis, N. Novel insights in spatial epidemiology utilizing explainable AI (XAI) and remote sensing. Remote Sens. 2022, 14, 3074. [Google Scholar] [CrossRef]
- Kopczewska, K. Spatial machine learning: New opportunities for regional science. Ann. Reg. Sci. 2022, 68, 713–755. [Google Scholar] [CrossRef]
- Shortliffe, E.H.; Sepulveda, M.J. Clinical decision support in the era of artificial intelligence. JAMA 2018, 320, 2199–2200. [Google Scholar] [CrossRef]
- Xu, J.S.; Wang, A.; Schmidt, N.; Adams, M.; Hatzopoulou, M. A gradient boost approach for predicting near-road ultrafine particle concentrations using detailed traffic characterization. Environ. Pollut. 2020, 265, 114777. [Google Scholar] [CrossRef] [PubMed]
- Wong, K.C.Y.; Xiang, Y.; Yin, L.Y.; So, H.C. Uncovering clinical risk factors and predicting severe COVID-19 cases using UK biobank data: Machine learning approach. Jmir. Public Health Surveill. 2021, 7, e29544. [Google Scholar] [CrossRef] [PubMed]
- Department of Household Registration, Ministry of the Interior (Taiwan). Available online: https://www.ris.gov.tw/app/portal (accessed on 1 February 2023).
- Ng, K.C.; Chaves, L.F.; Tsai, K.H.; Chuang, T.W. Increased adult Aedes aegypti and Culex quinquefasciatus (Diptera: Culicidae) Abundance in a dengue transmission hotspot, compared to a coldspot, within Kaohsiung City, Taiwan. Insects 2018, 9, 98. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.; Chen, C.D.; Shih, C.M.; Lee, T.C.; Wu, M.T.; Wu, D.C.; Chen, Y.H.; Hung, C.H.; Wu, M.C.; Huang, C.C.; et al. Time-lagging interplay effect and excess risk of meteorological/mosquito parameters and petrochemical gas explosion on dengue incidence. Sci. Rep. 2016, 6, 35028. [Google Scholar] [CrossRef]
- Chuang, T.W.; Ng, K.C.; Nguyen, T.L.; Chaves, L.F. Epidemiological characteristics and space-time analysis of the 2015 dengue outbreak in the metropolitan region of Tainan City, Taiwan. Int. J. Environ Res. Public Health 2018, 15, 396. [Google Scholar] [CrossRef]
- National Land Surveying and Mapping Center, Ministry of the Interior, R.O.C. (Taiwan). Available online: https://www.nlsc.gov.tw/en/cp.aspx?n=2101 (accessed on 31 October 2021).
- Spellerberg, I.F.; Fedor, P.J. A tribute to Claude Shannon (1916-2001) and a plea for more rigorous use of species richness, species diversity and the Shannon-Wiener’ Index. Glob. Ecol. Biogeogr. 2003, 12, 177–179. [Google Scholar] [CrossRef]
- Romero-Vega, L.M.; Piche-Ovares, M.; Soto-Garita, C.; Barantes Murillo, D.F.; Chaverri, L.G.; Alfaro-Alarcon, A.; Corrales-Aguilar, E.; Troyo, A. Seasonal changes in the diversity, host preferences and infectivity of mosquitoes in two arbovirus-endemic regions of Costa Rica. Parasit. Vectors 2023, 16, 34. [Google Scholar] [CrossRef]
- Friedman, J.H. Greedy function approximation: A gradient boosting machine. Ann. Stat. 2001, 29, 1189–1232. [Google Scholar] [CrossRef]
- Lundberg, S.M.; Erion, G.; Chen, H.; DeGrave, A.; Prutkin, J.M.; Nair, B.; Katz, R.; Himmelfarb, J.; Bansal, N.; Lee, S.I. From local explanations to global understanding with explainable AI for trees. Nat. Mach. Intell. 2020, 2, 56–67. [Google Scholar] [CrossRef]
- Lundberg, S.M.; Lee, S.I. Consistent feature attribution for tree ensembles. arXiv 2017, arXiv:1706.06060. [Google Scholar]
- Lundberg, S.M.; Erion, G.G.; Lee, S.I. Consistent individualized feature attribution for tree ensembles. arXiv 2018, arXiv:1802.03888. [Google Scholar]
- Lundberg, S.M.; Lee, S.I. A unified approach to interpreting model predictions. arXiv 2017, arXiv:1705.07874. [Google Scholar]
- Li, Z. Extracting spatial effects from machine learning model using local interpretation method: An example of SHAP and XGBoost. Comput. Environ. Urban Syst. 2022, 96, 101845. [Google Scholar] [CrossRef]
- Iban, M.C. An explainable model for the mass appraisal of residences: The application of tree-based Machine Learning algorithms and interpretation of value determinants. Habitat Int. 2022, 128, 102660. [Google Scholar] [CrossRef]
- Barboza, L.A.; Chou-Chen, S.W.; Vasquez, P.; Garcia, Y.E.; Calvo, J.G.; Hidalgo, H.G.; Sanchez, F. Assessing dengue fever risk in Costa Rica by using climate variables and machine learning techniques. PLoS Negl. Trop. Dis. 2023, 17, e0011047. [Google Scholar] [CrossRef] [PubMed]
- You, S.H.; Chen, S.C.; Huang, Y.H.; Tsai, H.C. Dengue meteorological determinants during epidemic and non-epidemic periods in Taiwan. Trop. Med. Infect. Dis. 2022, 7, 408. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.L.; Lippi, C.A.; Stewart-Ibarra, A.M.; Ayala, E.B.; Mordecai, E.A.; Sippy, R.; Heras, F.H.; Blackburn, J.K.; Ryan, S.J. Household and climate factors influence Aedes aegypti presence in the arid city of Huaquillas, Ecuador. PLoS Negl. Trop. Dis. 2021, 15, e0009931. [Google Scholar] [CrossRef]
- Pless, E.; Saarman, N.P.; Powell, J.R.; Caccone, A.; Amatulli, G. A machine-learning approach to map landscape connectivity in Aedes aegypti with genetic and environmental data. Proc. Natl. Acad. Sci. USA 2021, 118, e2003201118. [Google Scholar] [CrossRef]
- Benitez-Valladares, D.; Kroeger, A.; Tejeda, G.S.; Hussain-Alkhateeb, L. Validation of the early warning and response system (EWARS) for dengue outbreaks: Evidence from the national vector control program in Mexico. PLoS Negl. Trop. Dis. 2021, 15, e0009261. [Google Scholar] [CrossRef]
- Benedum, C.M.; Shea, K.M.; Jenkins, H.E.; Kim, L.Y.; Markuzon, N. Weekly dengue forecasts in Iquitos, Peru; San Juan, Puerto Rico; and Singapore. PLoS Negl. Trop. Dis. 2020, 14, e0008710. [Google Scholar] [CrossRef]
- Rahman, M.S.; Pientong, C.; Zafar, S.; Ekalaksananan, T.; Paul, R.E.; Haque, U.; Rocklov, J.; Overgaard, H.J. Mapping the spatial distribution of the dengue vector Aedes aegypti and predicting its abundance in northeastern Thailand using machine-learning approach. One Health 2021, 13, 100358. [Google Scholar] [CrossRef] [PubMed]
- Benitez, E.M.; Luduena-Almeida, F.; Frias-Cespedes, M.; Almiron, W.R.; Estallo, E.L. Could land cover influence Aedes aegypti mosquito populations? Med. Vet. Entomol. 2020, 34, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, C.; Chiaravalloti-Neto, F.; de Oliveira Lage, M.; Quintanilha, J.A.; Parra, M.C.; Dibo, M.R.; Favaro, E.A.; Guirado, M.M.; Nogueira, M.L. Remote sensing for risk mapping of Aedes aegypti infestations: Is this a practical task? Acta Trop. 2020, 205, 105398. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Ren, H.; Zheng, L.; Cao, W.; Zhang, A.; Zhuang, D.; Lu, L.; Jiang, H. Ecological niche modeling identifies fine-scale areas at high risk of dengue fever in the Pearl River Delta, China. Int. J. Environ Res. Public Health 2017, 14, 619. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Cao, W.; Ren, H.; Ji, Z.; Jiang, H. Spatiotemporal responses of dengue fever transmission to the road network in an urban area. Acta Trop. 2018, 183, 8–13. [Google Scholar] [CrossRef]
- Chen, T.; Guestrin, C. XGBoost: A scalable tree boosting system. In Proceedings of the 22nd ACM SIGKDD International Conference on Knowledge Discovery and Data Mining, San Francisco, CA, USA, 13–17 August 2016; pp. 785–794. [Google Scholar]
- Phang, W.K.; Hamid, M.H.A.; Jelip, J.; Mudin, R.N.; Chuang, T.W.; Lau, Y.L.; Fong, M.Y. Predicting Plasmodium knowlesi transmission risk across Peninsular Malaysia using machine learning-based ecological niche modeling approaches. Front. Microbiol 2023, 14, 357. [Google Scholar] [CrossRef]
- Lorenz, C.; Castro, M.C.; Trindade, P.M.P.; Nogueira, M.L.; de Oliveira Lage, M.; Quintanilha, J.A.; Parra, M.C.; Dibo, M.R.; Favaro, E.A.; Guirado, M.M.; et al. Predicting Aedes aegypti infestation using landscape and thermal features. Sci. Rep. 2020, 10, 21688. [Google Scholar] [CrossRef]
- Cromwell, E.A.; Stoddard, S.T.; Barker, C.M.; Van Rie, A.; Messer, W.B.; Meshnick, S.R.; Morrison, A.C.; Scott, T.W. The relationship between entomological indicators of Aedes aegypti abundance and dengue virus infection. PLoS Negl. Trop. Dis. 2017, 11, e0005429. [Google Scholar] [CrossRef]
- Wen, T.H.; Lin, M.H.; Fang, C.T. Population Movement and Vector-Borne Disease Transmission: Differentiating Spatial–Temporal Diffusion Patterns of Commuting and Noncommuting Dengue Cases. Ann. Assoc. Am. Geogr. 2012, 102, 1026–1037. [Google Scholar] [CrossRef]
- Lim, J.T.; Dickens, B.S.L.; Chew, L.Z.X.; Choo, E.L.W.; Koo, J.R.; Aik, J.; Ng, L.C.; Cook, A.R. Impact of sars-cov-2 interventions on dengue transmission. PLoS Negl. Trop. Dis. 2020, 14, e0008719. [Google Scholar] [CrossRef]
- Lim, J.T.; Chew, L.Z.X.; Choo, E.L.W.; Dickens, B.S.L.; Ong, J.; Aik, J.; Ng, L.C.; Cook, A.R. Increased dengue transmissions in Singapore attributable to SARS-CoV-2 social distancing measures. J. Infect. Dis. 2021, 223, 399–402. [Google Scholar] [CrossRef] [PubMed]
- Mohammad, M.H.; Palmer José, H.; María, A.R.; Anusua, S.; Ana, C.S.C.; Mohammad, Y.E. Concurrent epidemics of dengue and COVID-19 in Peru: Which way forward? Lancet Reg. Health Am. 2022, 12, 100277. [Google Scholar]
- Chang, S.F.; Huang, J.H.; Shu, P.Y. Characteristics of dengue epidemics in Taiwan. J. Formos. Med. Assoc. 2012, 111, 297–299. [Google Scholar] [CrossRef] [PubMed]


| LULC Types | Mean | Standard Deviation | Max | Min |
|---|---|---|---|---|
| F010102 (Dry Crops) | 2.98% | 6.85% | 58.31% | 0 |
| F030303 (General Roads) | 21.10% | 10.00% | 47.60% | 0.91% |
| F010103 (Fruit Tree) | 3.39% | 7.74% | 56.83% | 0 |
| F010402 (Agriculture Storage Facility) | 0.10% | 0.21% | 2.08% | 0 |
| F090801 (Unused Land) | 4.20% | 5.10% | 38% | 0 |
| House Density | 8643.1 | 7008.6 | 69,215.2 | 3.0 |
| F050201 (Residential Area) | 26.20% | 16.20% | 67.90% | 0 |
| F050301 (Manufacturing) | 1.98% | 5.92% | 80% | 0 |
| F050202 (Industrial Area) | 0.06% | 0.17% | 4.13 | 0 |
| Shannon’s Diversity Index | 1.9 | 0.4 | 3.0 | 0.28 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, H.; Nguyen, T.-N.; Chuang, T.-W. An Integrative Explainable Artificial Intelligence Approach to Analyze Fine-Scale Land-Cover and Land-Use Factors Associated with Spatial Distributions of Place of Residence of Reported Dengue Cases. Trop. Med. Infect. Dis. 2023, 8, 238. https://doi.org/10.3390/tropicalmed8040238
Yang H, Nguyen T-N, Chuang T-W. An Integrative Explainable Artificial Intelligence Approach to Analyze Fine-Scale Land-Cover and Land-Use Factors Associated with Spatial Distributions of Place of Residence of Reported Dengue Cases. Tropical Medicine and Infectious Disease. 2023; 8(4):238. https://doi.org/10.3390/tropicalmed8040238
Chicago/Turabian StyleYang, Hsiu, Thi-Nhung Nguyen, and Ting-Wu Chuang. 2023. "An Integrative Explainable Artificial Intelligence Approach to Analyze Fine-Scale Land-Cover and Land-Use Factors Associated with Spatial Distributions of Place of Residence of Reported Dengue Cases" Tropical Medicine and Infectious Disease 8, no. 4: 238. https://doi.org/10.3390/tropicalmed8040238
APA StyleYang, H., Nguyen, T.-N., & Chuang, T.-W. (2023). An Integrative Explainable Artificial Intelligence Approach to Analyze Fine-Scale Land-Cover and Land-Use Factors Associated with Spatial Distributions of Place of Residence of Reported Dengue Cases. Tropical Medicine and Infectious Disease, 8(4), 238. https://doi.org/10.3390/tropicalmed8040238







