Sustainable Elimination of Schistosomiasis in Ethiopia—A Five-Year Follow-Up Study
Abstract
:1. Introduction
2. Methods and Materials
Statistical Analysis
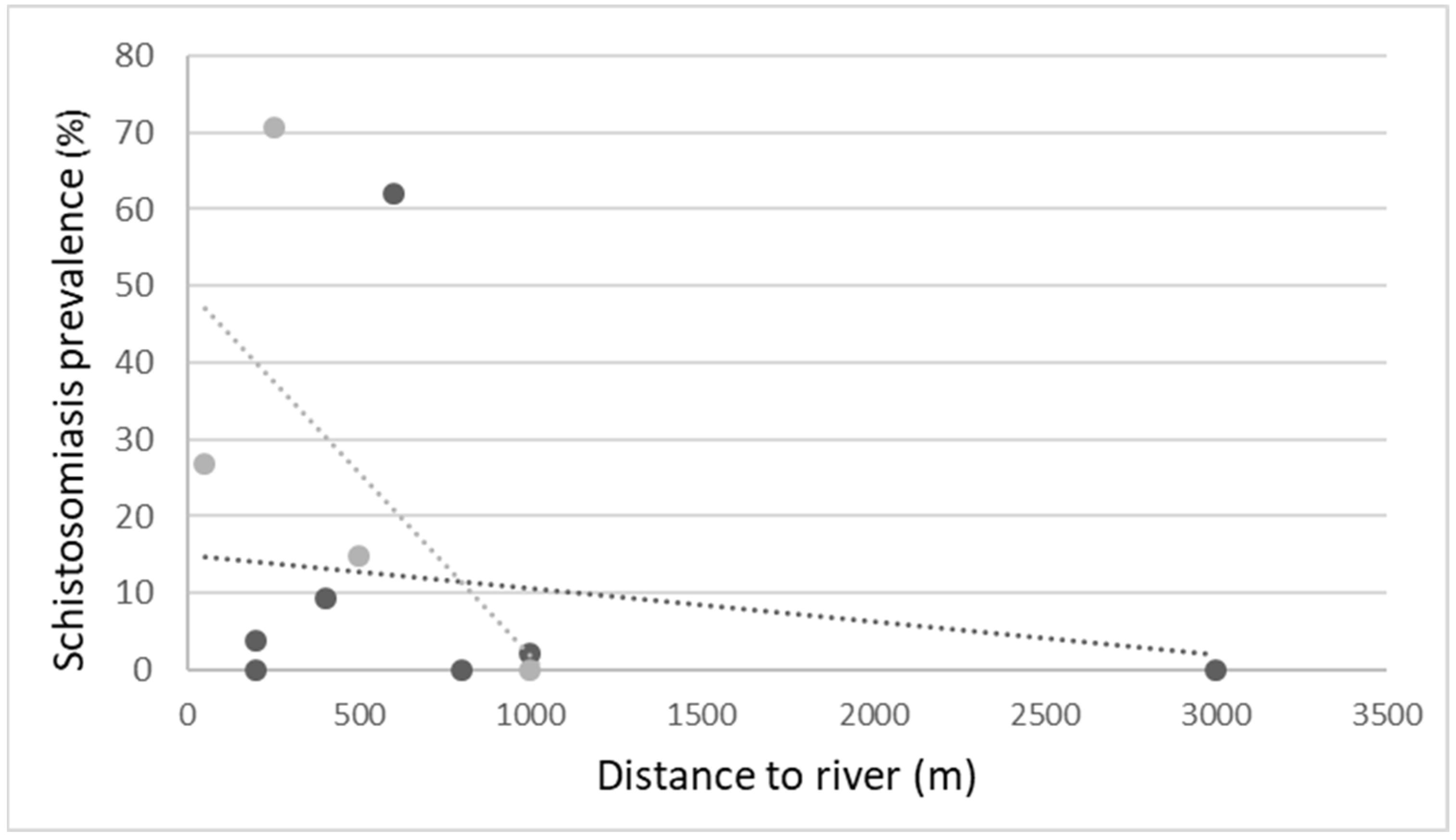
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- GBD 2017 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1789–1858. [Google Scholar] [CrossRef]
- Anto, F.; Asoala, V.; Adjuik, M.; Anyorigiya, T.; Oduro, A.; Akazili, J.; Akweongo, P.; Ayivor, P.; Bimi, L.; Hodgson, A. Water contact activities and prevalence of schistosomiasis infection among school-age children in communities along an irrigation scheme in rural Northern Ghana. J. Bacteriol. Parasitol. 2013, 4, 2. [Google Scholar] [CrossRef]
- Deribe, K.; Meribo, K.; Gebre, T.; Hailu, A.; Ali, A.; Aseffa, A.; Davey, G. The burden of neglected tropical diseases in Ethiopia, and opportunities for integrated control and elimination. Parasites Vectors 2012, 5, 240. [Google Scholar] [CrossRef] [PubMed]
- Webster, J.P.; Molyneux, D.H.; Hotez, P.J.; Fenwick, A. The contribution of mass drug administration to global health: Past, present and future. Philos. Trans. R. Soc. B Biol. Sci. 2014, 369, 20130434. [Google Scholar] [CrossRef] [PubMed]
- Montresor, A.; Engels, D.; Ramsan, M.; Foum, A.; Savioli, L. Field test of the ‘dose pole’ for praziquantel in Zanzibar. Trans. R. Soc. Trop. Med. Hyg. 2002, 96, 323–324. [Google Scholar] [CrossRef]
- Mengitsu, B.; Shafi, O.; Kebede, B.; Kebede, F.; Worku, D.T.; Herero, M.; French, M.; Kebede, B.; Mackenzie, C.; Martindale, S.; et al. Ethiopia and its steps to mobilize resources to achieve 2020 elimination and control goals for neglected tropical diseases: Spider webs joined can tie a lion. Int. Health 2016, 8 (Suppl. S1), i34–i52. [Google Scholar] [CrossRef]
- Ouattara, M.; Bassa, F.K.; Diakité, N.R.; Hattendorf, J.; Coulibaly, J.T.; Yao, P.K.; Tian-Bi, Y.N.; Konan, C.K.; Assaré, R.K.; Koné, N.; et al. Effectiveness of Four Different Interventions against Schistosoma haematobium in a Seasonal Transmission Setting of Côte d’Ivoire: A Cluster Randomized Trial. Clin. Infect. Dis. 2021, 74, 2181–2190. [Google Scholar] [CrossRef]
- Crellen, T.; Walker, M.; Lamberton, P.H.; Kabatereine, N.B.; Tukahebwa, E.M.; Cotton, J.A.; Webster, J.P. Reduced efficacy of praziquantel against Schistosoma mansoni is associated with multiple rounds of mass drug administration. Clin. Infect. Dis. 2016, 63, 1151–1159. [Google Scholar]
- Ross, A.G.; Olveda, R.M.; Chy, D.; Olveda, D.U.; Li, Y.; Harn, D.A.; Gray, D.J.; McManus, D.P.; Tallo, V.; Chau, T.N.; et al. Can mass drug administration lead to the sustainable control of schistosomiasis? J. Infect. Dis. 2015, 211, 283–289. [Google Scholar] [CrossRef]
- King, C.H.; Bertsch, D. Historical perspective: Snail control to prevent schistosomiasis. PLoS Negl. Trop. Dis. 2015, 9, e0003657. [Google Scholar] [CrossRef]
- Hanson, C.; Weaver, A.; Zoerhoff, K.L.; Kabore, A.; Linehan, M.; Doherty, A.; Engels, D.; Savioli, L.; Ottesen, E.A. Integrated implementation of programs targeting neglected tropical diseases through preventive chemotherapy: Identifying best practices to roll out programs at national scale. Am. J. Trop. Med. Hyg. 2012, 86, 508. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.G.; Chau, T.N.; Inobaya, M.T.; Olveda, R.M.; Li, Y.; Harn, D.A. A new global strategy for the elimination of schistosomiasis. Int. J. Infect. Dis. 2017, 54, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Ayalew, A.; Debebe, T.; Worku, A. Prevalence and risk factors of intestinal parasites among Delgi school children, North Gondar, Ethiopia. J. Parasitol. Vector Biol. 2011, 3, 75–81. [Google Scholar]
- Woldegerima, E.; Bayih, A.G.; Tegegne, Y.; Aemero, M.; Jejaw Zeleke, A. Prevalence and reinfection rates of Schistosoma mansoni and praziquantel efficacy against the parasite among primary school children in Sanja Town, Northwest Ethiopia. J. Parasitol. Res. 2019, 2019, 3697216. [Google Scholar] [CrossRef]
- Deol, A.K.; Fleming, F.M.; Calvo-Urbano, B.; Walker, M.; Bucumi, V.; Gnandou, I.; Tukahebwa, E.M.; Jemu, S.; Mwingira, U.J.; Alkohlani, A.; et al. Schistosomiasis—assessing progress toward the 2020 and 2025 global goals. N. Engl. J. Med. 2019, 381, 2519–2528. [Google Scholar] [CrossRef] [PubMed]
- Federal Democratic Republic of Ethiopia, Ministry of Health (FMoH). Second Edition of National Master Plan on Neglected Tropical Diseases (NTDs), 2015–2019; Federal Ministry of Health: Addis Ababa, Ethiopia, 2016.
- Ali, J.; Polland, A.; Adlerstein, D.; Gziabher, Y.G.; Sabar, G.; Liss, Y.; Bentwich, Z. Deworming school children in Ethiopia: The importance of a comprehensive approach. Open J. Trop. Med. 2019, 3, 001–006. [Google Scholar]
- Schistosomiasis, W.H. Progress Report 2001–2011 and Strategic Plan 2012–2020; World Health Organization: Geneva, Switzerland, 2013. [Google Scholar]
- World Health Organization. Helminth Control in School-Age Children: A Guide for Managers of Control Programmes; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Assaré, R.K.; N’Tamon, R.N.; Bellai, L.G.; Koffi, J.A.; Mathieu, T.B.; Ouattara, M.; Hürlimann, E.; Coulibaly, J.T.; Diabaté, S.; N’Goran, E.K.; et al. Characteristics of persistent hotspots of Schistosoma mansoni in western Côte d’Ivoire. Parasites Vectors 2020, 13, 337. [Google Scholar] [CrossRef]
- Lessler, J.; Azman, A.S.; McKay, H.S.; Moore, S.M. What is a hotspot anyway? Am. J. Trop. Med. Hyg. 2017, 96, 1270. [Google Scholar] [CrossRef]
- Kittur, N.; Campbell, C.H., Jr.; Binder, S.; Shen, Y.; Wiegand, R.E.; Mwanga, J.R.; Kinung’hi, S.M.; Musuva, R.M.; Odiere, M.R.; Matendechero, S.H.; et al. Discovering, defining, and summarizing persistent hotspots in SCORE studies. Am. J. Trop. Med. Hyg. 2020, 103 (Suppl. S1), 24. [Google Scholar] [CrossRef]
- Ratcliffe, J.W. Analyst biases in KAP surveys: A cross-cultural comparison. Stud. Fam. Plan. 1976, 7, 322–330. [Google Scholar] [CrossRef]
- Endris, M.; Tekeste, Z.; Lemma, W.; Kassu, A. Comparison of the Kato-Katz, wet mount, and formol-ether concentration diagnostic techniques for intestinal helminth infections in Ethiopia. Int. Sch. Res. Not. 2013, 2013, 180439. [Google Scholar] [CrossRef]
- World Health Organization. Basic Laboratory Methods in Medical Parasitology; World Health Organization: Geneva, Switzerland, 1991. [Google Scholar]
- World Health Organization. Prevention and Control of Schistosomiasis and Soil-Transmitted Helminthiasis, 21 February 2003 ed.; World Health Organization: Geneva, Switzerland, 2002. [Google Scholar]
- Freeman, M.C.; Ellis, A.S.; Ogutu, E.A.; Caruso, B.A.; Linabarger, M.; Micek, K.; Muga, R.; Girard, A.W.; Wodnik, B.K.; Arriola, K.J. Impact of a demand-side integrated WASH and nutrition community-based care group intervention on behavioural change: A randomised controlled trial in western Kenya. BMJ Glob. Health 2020, 5, e002806. [Google Scholar] [CrossRef] [PubMed]
- Gryseels, B.; Polman, K.; Clerinx, J.; Kestens, L. Human schistosomiasis. Lancet 2006, 368, 1106–1118. [Google Scholar] [CrossRef]
- Birrie, H.; Tedla, S.; Tilahun, G.; Kloos, H.; Eshete, H. Schistosomiasis and its distribution in Ethiopia and Eritrea. Schistosomiasis Ethiop. Eritrea 1998, 2, 29–86. [Google Scholar]
- Ahmed, A.M.; El Tash, L.A.; Mohamed, E.Y.; Adam, I. High levels of Schistosoma mansoni infections among schoolchildren in central Sudan one year after treatment with praziquantel. J. Helminthol. 2012, 86, 228–232. [Google Scholar] [CrossRef] [PubMed]
- Alemu, M.; Hailu, A.; Bugssa, G. Prevalence of intestinal schistosomiasis and soil-transmitted helminthiasis among primary schoolchildren in Umolante district, South Ethiopia. Clin. Med. Res. 2014, 3, 174–180. [Google Scholar] [CrossRef]
- Turner, K.M.; Rousseau, N.; Croot, L.; Duncan, E.; Yardley, L.; O’Cathain, A.; Hoddinott, P. Understanding successful development of complex health and healthcare interventions and its drivers from the perspective of developers and wider stakeholders: An international qualitative interview study. BMJ Open 2019, 9, e028756. [Google Scholar] [CrossRef] [Green Version]
| Semi-Urban Schools | Urban Schools | Total n = 624 | p-Value | |||
|---|---|---|---|---|---|---|
| ≤500 m from River n = 202 | >500 m from River n = 40 | ≤500 m from River n = 165 | >500 m from River | |||
| Male | 102 (50.5%) | 20 (50%) | 83 (50.3%) | 110 50.7%) | 315 (50.5%) | 0.99 |
| Female | 100 (49.5%) | 20 (50%) | 82 (49.7%) | 107 (49.3%) | 309 (49.5%) | |
| Grade | 4 | 4 | 3 | 4 | 4 | 0.015 |
| Age, years | 10.7 ± 2.5 | 10.9 ± 2.1 | 9.6 ± 2.0 | 10.1 ± 2.2 | 10.2 ± 2.3 | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gal, L.B.; Bruck, M.; Tal, R.; Baum, S.; Ali, J.M.; Weldegabriel, L.L.; Sabar, G.; Golan, R.; Bentwich, Z. Sustainable Elimination of Schistosomiasis in Ethiopia—A Five-Year Follow-Up Study. Trop. Med. Infect. Dis. 2022, 7, 218. https://doi.org/10.3390/tropicalmed7090218
Gal LB, Bruck M, Tal R, Baum S, Ali JM, Weldegabriel LL, Sabar G, Golan R, Bentwich Z. Sustainable Elimination of Schistosomiasis in Ethiopia—A Five-Year Follow-Up Study. Tropical Medicine and Infectious Disease. 2022; 7(9):218. https://doi.org/10.3390/tropicalmed7090218
Chicago/Turabian StyleGal, Lotte Ben, Michal Bruck, Robyn Tal, Sarit Baum, Jemal Mahdi Ali, Lemlem Legesse Weldegabriel, Galia Sabar, Rachel Golan, and Zvi Bentwich. 2022. "Sustainable Elimination of Schistosomiasis in Ethiopia—A Five-Year Follow-Up Study" Tropical Medicine and Infectious Disease 7, no. 9: 218. https://doi.org/10.3390/tropicalmed7090218
APA StyleGal, L. B., Bruck, M., Tal, R., Baum, S., Ali, J. M., Weldegabriel, L. L., Sabar, G., Golan, R., & Bentwich, Z. (2022). Sustainable Elimination of Schistosomiasis in Ethiopia—A Five-Year Follow-Up Study. Tropical Medicine and Infectious Disease, 7(9), 218. https://doi.org/10.3390/tropicalmed7090218






