Vector-Borne Tularemia: A Re-Emerging Cause of Cervical Lymphadenopathy
Abstract
1. Introduction
2. Pathogenesis of Tularemia
3. Transmission of Tularemia
4. Tularemia as a Tick-Borne Disease
5. Epidemiology of Tularemia
6. Clinical Presentation of Tularemia
7. Complications of Tularemia
8. Diagnosing Tularemia
9. Differential Diagnosis of Cervical Lymphadenopathy after a Tick Bite
10. Treatment of Tularemia
11. Case Example
11.1. Patient History
11.2. Clinical Examination
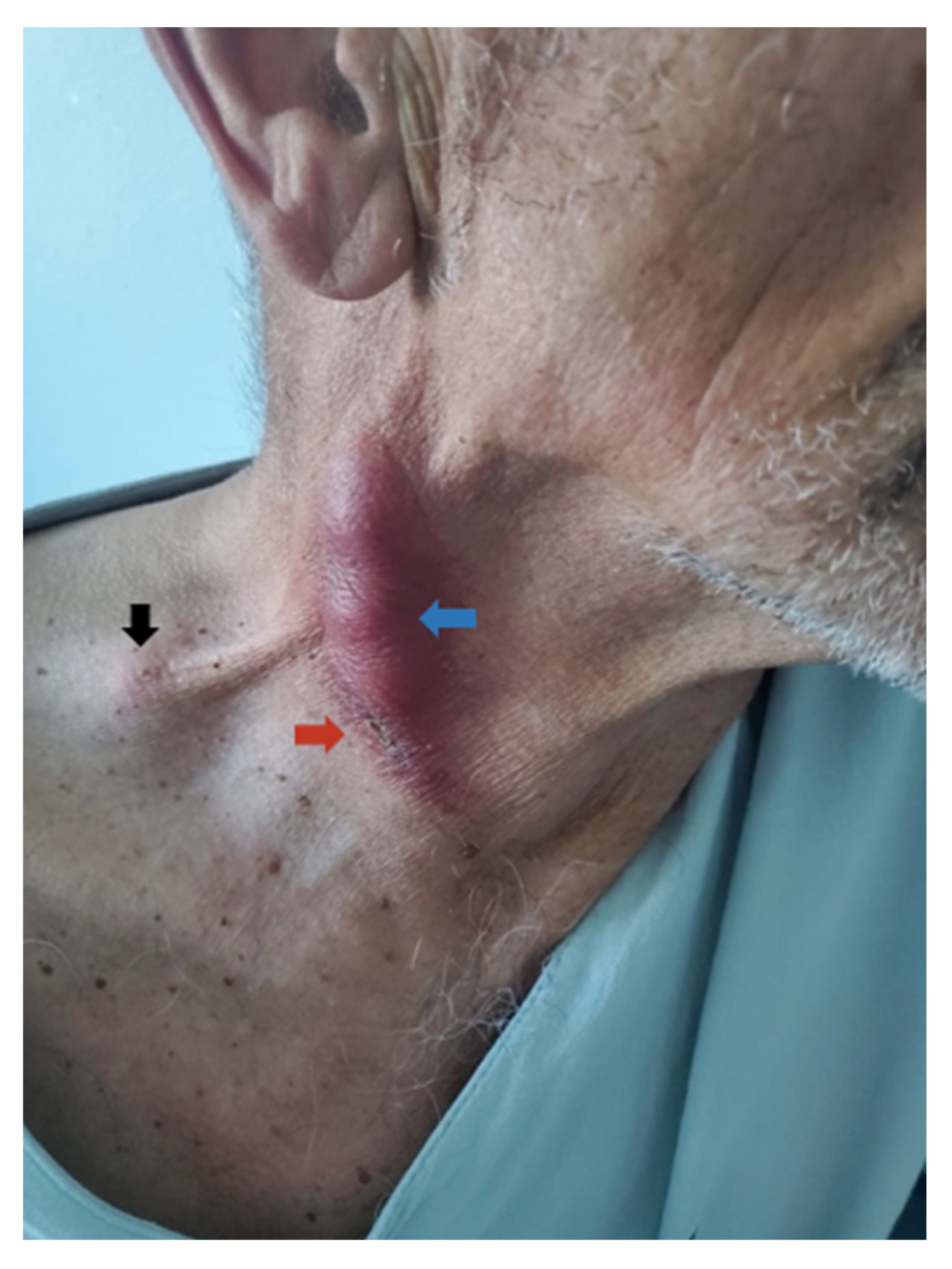
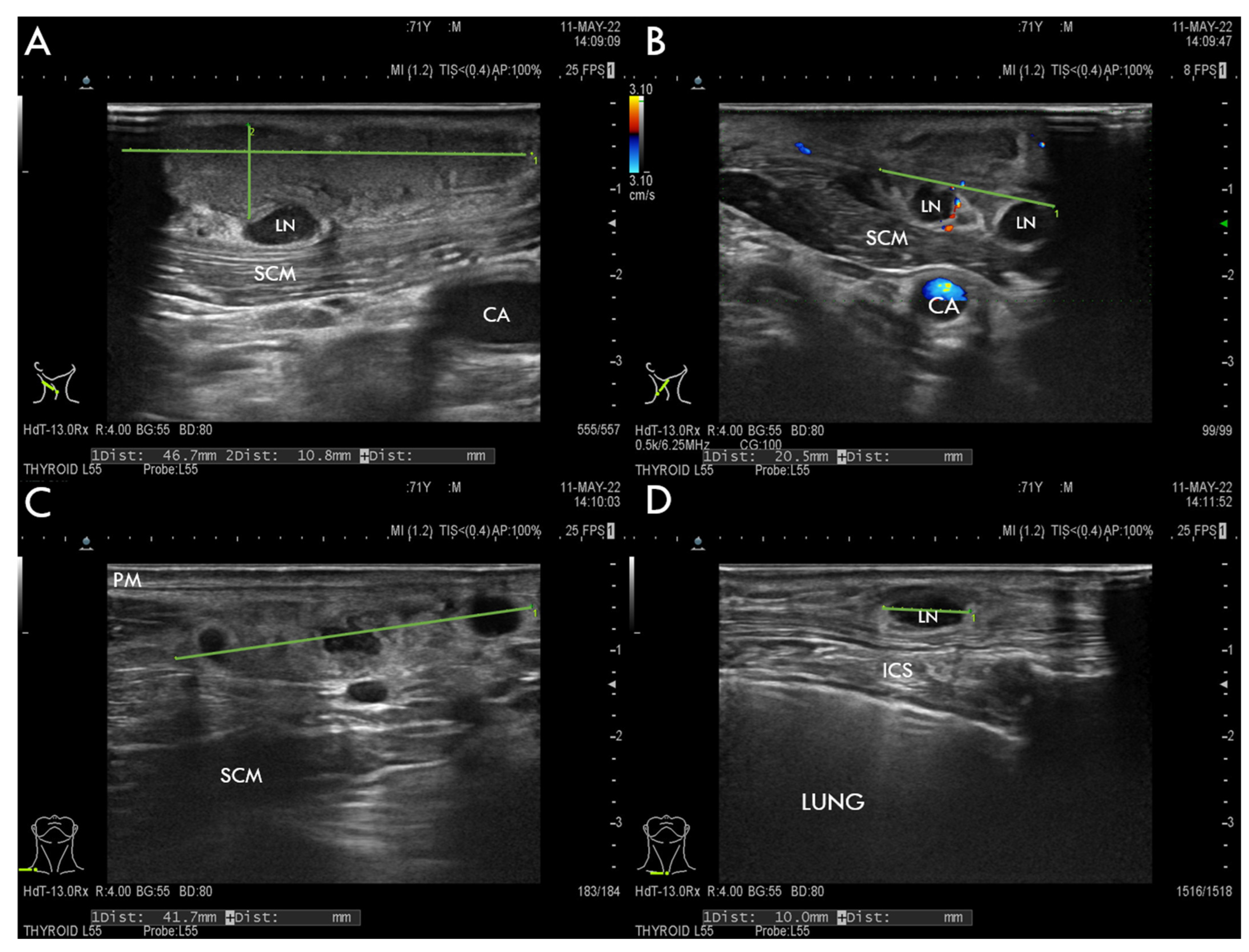
11.3. Imaging Studies
11.4. Laboratory Tests Results and Infectious Disease Specialist Consultation
11.5. Surgical Treatment
11.6. Biopsy, Histopathology, and Microbiology Findings
11.7. On-Ward Management
11.8. Follow-Up and Outcome
12. Case Report Discussion
13. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sjöstedt, A. Tularemia: History, Epidemiology, Pathogen Physiology, and Clinical Manifestations. Ann. N. Y. Acad. Sci. 2007, 1105, 1–29. [Google Scholar] [CrossRef]
- Männikkö, N. Etymologia: Francisella Tularensis. Emerg. Infect. Dis. 2011, 17, 799. [Google Scholar] [CrossRef]
- Rojko, T.; Korva, M.; Lotrič-Furlan, S.; Strle, F.; Avšič-Županc, T. Cluster of Ulceroglandular Tularemia Cases in Slovenia. Ticks Tick-Borne Dis. 2016, 7, 1193–1197. [Google Scholar] [CrossRef]
- Hestvik, G.; Warns-Petit, E.; Smith, L.A.; Fox, N.J.; Uhlhorn, H.; Artois, M.; Hannant, D.; Hutchings, M.R.; Mattsson, R.; Yon, L.; et al. The Status of Tularemia in Europe in a One-Health Context: A Review. Epidemiol. Infect. 2015, 143, 2137–2160. [Google Scholar] [CrossRef]
- Dennis, D.T.; Inglesby, T.V.; Henderson, D.A.; Bartlett, J.G.; Ascher, M.S.; Eitzen, E.; Fine, A.D.; Friedlander, A.M.; Hauer, J.; Layton, M.; et al. Tularemia as a Biological Weapon: Medical and Public Health Management. JAMA 2001, 285, 2763–2773. [Google Scholar] [CrossRef]
- Stupak, H.D.; Scheuller, M.C.; Schindler, D.N.; Ellison, D.E. Tularemia of the Head and Neck: A Possible Sign of Bioterrorism. Ear. Nose. Throat J. 2003, 82, 263–265. [Google Scholar] [CrossRef]
- Prokšová, M.; Bavlovič, J.; Klimentová, J.; Pejchal, J.; Stulík, J. Tularemia—Zoonosis Carrying a Potential Risk of Bioterrorism. Epidemiol. Mikrobiol. Imunol. Cas. Spol. Epidemiol. Mikrobiol. Ceske Lek. Spol. JE Purkyne 2019, 68, 82–89. [Google Scholar]
- Kingry, L.C.; Petersen, J.M. Comparative Review of Francisella Tularensis and Francisella Novicida. Front. Cell. Infect. Microbiol. 2014, 4, 35. [Google Scholar] [CrossRef]
- Select Agents and Toxins List | Federal Select Agent Program. Available online: https://www.selectagents.gov/sat/list.htm (accessed on 25 July 2022).
- Johansson, A.; Celli, J.; Conlan, W.; Elkins, K.L.; Forsman, M.; Keim, P.S.; Larsson, P.; Manoil, C.; Nano, F.E.; Petersen, J.M.; et al. Objections to the Transfer of Francisella Novicida to the Subspecies Rank of Francisella Tularensis. Int. J. Syst. Evol. Microbiol. 2010, 60, 1717–1718. [Google Scholar] [CrossRef]
- Busse, H.-J.; Huber, B.; Anda, P.; Escudero, R.; Scholz, H.C.; Seibold, E.; Splettstoesser, W.D.; Kämpfer, P. Objections to the Transfer of Francisella Novicida to the Subspecies Rank of Francisella Tularensis—Response to Johansson et al. Int. J. Syst. Evol. Microbiol. 2010, 60, 1718–1720. [Google Scholar] [CrossRef]
- Ellis, J.; Oyston, P.C.F.; Green, M.; Titball, R.W. Tularemia. Clin. Microbiol. Rev. 2002, 15, 631–646. [Google Scholar] [CrossRef]
- Maurin, M.; Gyuranecz, M. Tularaemia: Clinical Aspects in Europe. Lancet Infect. Dis. 2016, 16, 113–124. [Google Scholar] [CrossRef]
- Celli, J.; Zahrt, T.C. Mechanisms of Francisella Tularensis Intracellular Pathogenesis. Cold Spring Harb. Perspect. Med. 2013, 3, a010314. [Google Scholar] [CrossRef]
- Molins, C.R.; Delorey, M.J.; Yockey, B.M.; Young, J.W.; Belisle, J.T.; Schriefer, M.E.; Petersen, J.M. Virulence Difference between the Prototypic Schu S4 Strain (A1a) and Francisella Tularensis A1a, A1b, A2 and Type B Strains in a Murine Model of Infection. BMC Infect. Dis. 2014, 14, 67. [Google Scholar] [CrossRef]
- Strehl, J.; Schoerner, C.; Hartmann, A.; Agaimy, A. Tularemia lymphadenitis. An emerging differential diagnosis of necrotizing granulomatous cervical lymphadenitis. Der Pathologe 2014, 35, 166–172. [Google Scholar] [CrossRef]
- Tully, B.G.; Huntley, J.F. Mechanisms Affecting the Acquisition, Persistence and Transmission of Francisella Tularensis in Ticks. Microorganisms 2020, 8, E1639. [Google Scholar] [CrossRef]
- Mörner, T. The Ecology of Tularaemia. Rev. Sci. Tech. Int. Off. Epizoot. 1992, 11, 1123–1130. [Google Scholar] [CrossRef]
- Gurycová, D. First Isolation of Francisella Tularensis Subsp. Tularensis in Europe. Eur. J. Epidemiol. 1998, 14, 797–802. [Google Scholar] [CrossRef]
- Sinclair, R.; Boone, S.A.; Greenberg, D.; Keim, P.; Gerba, C.P. Persistence of Category A Select Agents in the Environment. Appl. Environ. Microbiol. 2008, 74, 555–563. [Google Scholar] [CrossRef] [PubMed]
- Bahuaud, O.; Le Brun, C.; Lemaignen, A. Host Immunity and Francisella Tularensis: A Review of Tularemia in Immunocompromised Patients. Microorganisms 2021, 9, 2539. [Google Scholar] [CrossRef]
- Green, T.W.; Eigelsbach, H.T. Immunity in Tularemia: Report of Two Cases of Proved Reinfection. Arch. Intern. Med. 1950, 85, 777–782. Available online: https://jamanetwork.com/journals/jamainternalmedicine/article-abstract/554748 (accessed on 28 June 2022). [CrossRef]
- Svensson, K.; Larsson, P.; Johansson, D.; Byström, M.; Forsman, M.; Johansson, A. Evolution of Subspecies of Francisella Tularensis. J. Bacteriol. 2005, 187, 3903–3908. [Google Scholar] [CrossRef]
- Whipp, M.J.; Davis, J.M.; Lum, G.; de Boer, J.; Zhou, Y.; Bearden, S.W.; Petersen, J.M.; Chu, M.C.; Hogg, G. Characterization of a Novicida-like Subspecies of Francisella Tularensis Isolated in Australia. J. Med. Microbiol. 2003, 52, 839–842. [Google Scholar] [CrossRef]
- Leelaporn, A.; Yongyod, S.; Limsrivanichakorn, S.; Yungyuen, T.; Kiratisin, P. Francisella Novicida Bacteremia, Thailand. Emerg. Infect. Dis. 2008, 14, 1935–1937. [Google Scholar] [CrossRef]
- Biškup, U.G.; Kogoj, R.; Korva, M.; Knap, N.; Cerar, T.; Knapič, T.; Petrovec, M.; Avšič-Županc, T. Characterization of Tularemia Cases in Slovenia with Multiple-Locus Variable-Number Tandem Repeat Analysis. Vector Borne Zoonotic Dis. Larchmt. N 2021, 21, 351–357. [Google Scholar] [CrossRef]
- Foley, J.E.; Nieto, N.C. Tularemia. Vet. Microbiol. 2010, 140, 332–338. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Guidelines on Tularaemia; World Health Organization: Geneva, Switzerland, 2007; p. 115. [Google Scholar]
- Telford, S.R.; Goethert, H.K. Ecology of Francisella Tularensis. Annu. Rev. Entomol. 2020, 65, 351–372. [Google Scholar] [CrossRef]
- Porast Tularemije (Zajčje Mrzlice). Available online: https://www.nijz.si/sl/porast-tularemije-zajcje-mrzlice (accessed on 28 June 2022).
- Tularaemia. Available online: https://www.ecdc.europa.eu/en/tularaemia (accessed on 28 June 2022).
- Kugeler, K.J.; Mead, P.S.; Janusz, A.M.; Staples, J.E.; Kubota, K.A.; Chalcraft, L.G.; Petersen, J.M. Molecular Epidemiology of Francisella Tularensis in the United States. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2009, 48, 863–870. [Google Scholar] [CrossRef] [PubMed]
- Zellner, B.; Huntley, J.F. Ticks and Tularemia: Do We Know What We Don’t Know? Front. Cell. Infect. Microbiol. 2019, 9, 146. [Google Scholar] [CrossRef]
- Parker, R.R.; Spencer, R.R.; Francis, E. Tularæmia: XI. Tularæmia Infection in Ticks of the Species Dermacentor Andersoni Stiles in the Bitterroot Valley, Mont. Public Health Rep. 1896–1970 1924, 39, 1057–1073. [Google Scholar] [CrossRef]
- Yeni, D.K.; Büyük, F.; Ashraf, A.; Shah, M.S.U.D. Tularemia: A Re-Emerging Tick-Borne Infectious Disease. Folia Microbiol. 2021, 66, 1–14. [Google Scholar] [CrossRef]
- CDC Tularemia Home|CDC. Available online: https://www.cdc.gov/tularemia/index.html (accessed on 29 June 2022).
- Rosenberg, R.; Lindsey, N.P.; Fischer, M.; Gregory, C.J.; Hinckley, A.F.; Mead, P.S.; Paz-Bailey, G.; Waterman, S.H.; Drexler, N.A.; Kersh, G.J.; et al. Vital Signs: Trends in Reported Vectorborne Disease Cases—United States and Territories, 2004–2016. Morb. Mortal. Wkly. Rep. 2018, 67, 496–501. [Google Scholar] [CrossRef] [PubMed]
- Petersen, J.M.; Schriefer, M.E. Tularemia: Emergence/Re-Emergence. Vet. Res. 2005, 36, 455–467. [Google Scholar] [CrossRef]
- Maurin, M.; Pelloux, I.; Brion, J.P.; Del Banõ, J.-N.; Picard, A. Human Tularemia in France, 2006–2010. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2011, 53, e133–e141. [Google Scholar] [CrossRef]
- Gurycová, D.; Tináková, K.; Výrosteková, V.; Gacíková, E. The incidence of tularemia in Slovakia in 1997–2008. Epidemiol. Mikrobiol. Imunol. Cas. Spol. Epidemiol. Mikrobiol. Ceske Lek. Spol. JE Purkyne 2010, 59, 39–44. [Google Scholar]
- Dlugaiczyk, J.; Harrer, T.; Zwerina, J.; Traxdorf, M.; Schwarz, S.; Splettstoesser, W.; Geissdörfer, W.; Schoerner, C. Oropharyngeal Tularemia--a Differential Diagnosis of Tonsillopharyngitis and Cervical Lymphadenitis. Wien. Klin. Wochenschr. 2010, 122, 110–114. [Google Scholar] [CrossRef]
- Dryselius, R.; Hjertqvist, M.; Mäkitalo, S.; Lindblom, A.; Lilja, T.; Eklöf, D.; Lindström, A. Large Outbreak of Tularaemia, Central Sweden, July to September 2019. Euro Surveill. Bull. Eur. Sur Mal. Transm. Eur. Commun. Dis. Bull. 2019, 24, 1900603. [Google Scholar] [CrossRef]
- Zajmi, D.; Berisha, M.; Kalaveshi, A.; Begolli, I.; Ramadani, N.; Hoxha, R. Epidemiological Characteristics of Tularemia in Kosova in the Period 2006–2011. Mater. Socio-Med. 2013, 25, 220–222. [Google Scholar] [CrossRef]
- Grunow, R.; Kalaveshi, A.; Kühn, A.; Mulliqi-Osmani, G.; Ramadani, N. Surveillance of Tularaemia in Kosovo *, 2001 to 2010. Eurosurveillance 2012, 17, 20217. [Google Scholar] [CrossRef]
- Factsheet on Tularaemia. Available online: https://www.ecdc.europa.eu/en/tularaemia/facts (accessed on 29 June 2022).
- Schulze, C.; Heuner, K.; Myrtennäs, K.; Karlsson, E.; Jacob, D.; Kutzer, P.; Große, K.; Forsman, M.; Grunow, R. High and Novel Genetic Diversity of Francisella Tularensis in Germany and Indication of Environmental Persistence. Epidemiol. Infect. 2016, 144, 3025–3036. [Google Scholar] [CrossRef]
- Appelt, S.; Faber, M.; Köppen, K.; Jacob, D.; Grunow, R.; Heuner, K. Francisella Tularensis Subspecies Holarctica and Tularemia in Germany. Microorganisms 2020, 8, 1448. [Google Scholar] [CrossRef]
- European Food Safety Authority; European Centre for Disease Prevention and Control. The European Union One Health 2020 Zoonoses Report. EFSA J. 2021, 19, e06971. [Google Scholar] [CrossRef]
- Pojav Tularemije V Severno-Primorski Regiji. Available online: https://www.nijz.si/sl/pojav-tularemije-v-severno-primorski-regiji (accessed on 29 June 2022).
- Tomažič, J.; Tularemija, R.M. Infekcijske Bolezni; Združenje Za Infektologijo, Slovensko Zdravniško Društvo: Ljubljana, Slovenia, 2014; pp. 517–518. [Google Scholar]
- Tärnvik, A.; Berglund, L. Tularaemia. Eur. Respir. J. 2003, 21, 361–373. [Google Scholar] [CrossRef]
- Dixon, M.K.; Dayton, C.L.; Anstead, G.M. Parinaud’s Oculoglandular Syndrome: A Case in an Adult with Flea-Borne Typhus and a Review. Trop. Med. Infect. Dis. 2020, 5, 126. [Google Scholar] [CrossRef]
- Plourde, P.J.; Embree, J.; Friesen, F.; Lindsay, G.; Williams, T. Glandular Tularemia with Typhoidal Features in a Manitoba Child. CMAJ Can. Med. Assoc. J. 1992, 146, 1953–1955. [Google Scholar]
- Craft, D.; Kijek, M.T. Sentinel Level Clinical Laboratory Guidelines for Suspected Agents of Bioterorrism and Emerging Infectious Diseases. Available online: https://asm.org/ASM/media/Policy-and-Advocacy/LRN/Sentinel%20Files/tularemia.pdf (accessed on 18 June 2022).
- Luotonen, J.; Syrjala, H.; Jokinen, K.; Sutinen, S.; Salminen, A. Tularemia in Otolaryngologic Practice: An Analysis of 127 Cases. Arch. Otolaryngol. -Head Neck Surg. 1986, 112, 77–80. [Google Scholar] [CrossRef]
- Lübbert, C.; Taege, C.; Seufferlein, T.; Grunow, R. Prolonged course of tick-borne ulceroglandular tularemia in a 20-year-old patient in Germany—Case report and review of the literature. Dtsch. Med. Wochenschr. 1946 2009, 134, 1405–1410. [Google Scholar] [CrossRef]
- Bryant, K.A.; Marshall, G.S. Clinical Manifestations of Tick-Borne Infections in Children. Clin. Diagn. Lab. Immunol. 2000, 7, 523–527. [Google Scholar] [CrossRef]
- Hanke, C.A.; Otten, J.-E.; Berner, R.; Serr, A.; Splettstoesser, W.; von Schnakenburg, C. Ulceroglandular Tularemia in a Toddler in Germany after a Mosquito Bite. Eur. J. Pediatr. 2009, 168, 937–940. [Google Scholar] [CrossRef]
- Kukla, R.; Kračmarová, R.; Ryšková, L.; Bavlovič, J.; Pellantová, V.; Bolehovská, R.; Fajfr, M.; Pavlík, I.; Boštík, P. Francisella Tularensis Caused Cervical Lymphadenopathy in Little Children after a Tick Bite: Two Case Reports and a Short Literature Review. Ticks Tick-Borne Dis. 2022, 13, 101893. [Google Scholar] [CrossRef]
- Wills, P.I.; Gedosh, E.A.; Nichols, D.R. Head and Neck Manifestations of Tularemia. Laryngoscope 1982, 92, 770–773. [Google Scholar] [CrossRef]
- Atmaca, S.; Bayraktar, C.; Cengel, S.; Koyuncu, M. Tularemia Is Becoming Increasingly Important as a Differential Diagnosis in Suspicious Neck Masses: Experience in Turkey. Eur. Arch. Oto-Rhino-Laryngol. Off. J. Eur. Fed. Oto-Rhino-Laryngol. Soc. EUFOS Affil. Ger. Soc. Oto-Rhino-Laryngol. Head Neck Surg. 2009, 266, 1595–1598. [Google Scholar] [CrossRef]
- Borde, J.P.; Zange, S.; Antwerpen, M.H.; Georgi, E.; von Buttlar, H.; Kern, W.V.; Rieg, S. Five Cases of Vector-Borne Francisella Tularensis Holarctica Infections in South-Western Germany and Genetic Diversity. Ticks Tick-Borne Dis. 2017, 8, 808–812. [Google Scholar] [CrossRef]
- Penn, R.L.; Kinasewitz, G.T. Factors Associated with a Poor Outcome in Tularemia. Arch. Intern. Med. 1987, 147, 265–268. [Google Scholar] [CrossRef]
- Maurin, M. 78—Tularemia. In Hunter’s Tropical Medicine and Emerging Infectious Diseases, 10th ed.; Ryan, E.T., Hill, D.R., Solomon, T., Aronson, N.E., Endy, T.P., Eds.; Elsevier: London, UK, 2020; pp. 630–635. ISBN 978-0-323-55512-8. [Google Scholar]
- Nemmour, A.; Bakri, A.; Fischer, C.A.; Brand, Y. Paediatric Oropharyngeal Tularaemia Requiring Surgical Intervention. BMJ Case Rep. 2019, 12, e229754. [Google Scholar] [CrossRef]
- Alsan, M.M.; Lin, H.W. Tularemia Presenting as a Cervical Abscess. Otolaryngol. -Head Neck Surg. Off. J. Am. Acad. Otolaryngol. -Head Neck Surg. 2010, 143, 311–312. [Google Scholar] [CrossRef][Green Version]
- Kaeppler, M.; Kapoor, R.; Shah, N.; Katbamna, B.; Wantz, M.; Kott, A. Tick-Borne Illness and Infective Endocarditis: A Rare Case of Tularemia. CASE Cardiovasc. Imaging Case Rep. 2020, 4, 78–81. [Google Scholar] [CrossRef]
- Frischknecht, M.; Meier, A.; Mani, B.; Joerg, L.; Kim, O.C.-H.; Boggian, K.; Strahm, C. Tularemia: An Experience of 13 Cases Including a Rare Myocarditis in a Referral Center in Eastern Switzerland (Central Europe) and a Review of the Literature. Infection 2019, 47, 683–695. [Google Scholar] [CrossRef]
- Evans, M.E.; Gregory, D.W.; Schaffner, W.; McGee, Z.A. Tularemia: A 30-Year Experience with 88 Cases. Medicine 1985, 64, 251–269. [Google Scholar] [CrossRef]
- Pérez-Castrillón, J.L.; Bachiller-Luque, P.; Martín-Luquero, M.; Mena-Martín, F.J.; Herreros, V. Tularemia Epidemic in Northwestern Spain: Clinical Description and Therapeutic Response. Clin. Infect. Dis. 2001, 33, 573–576. [Google Scholar] [CrossRef]
- CDC. Managing Laboratory Exposures to Tularemia|CDC. Available online: https://www.cdc.gov/tularemia/laboratoryexposure/index.html (accessed on 29 June 2022).
- Maurin, M. Francisella Tularensis, Tularemia and Serological Diagnosis. Front. Cell. Infect. Microbiol. 2020, 10, 512090. [Google Scholar] [CrossRef]
- Tärnvik, A.; Chu, M.C. New Approaches to Diagnosis and Therapy of Tularemia. Ann. N. Y. Acad. Sci. 2007, 1105, 378–404. [Google Scholar] [CrossRef] [PubMed]
- Grunow, R.; Splettstoesser, W.; McDonald, S.; Otterbein, C.; O’Brien, T.; Morgan, C.; Aldrich, J.; Hofer, E.; Finke, E.-J.; Meyer, H. Detection of Francisella Tularensis in Biological Specimens Using a Capture Enzyme-Linked Immunosorbent Assay, an Immunochromatographic Handheld Assay, and a PCR. Clin. Diagn. Lab. Immunol. 2000, 7, 86–90. [Google Scholar] [CrossRef]
- Oztoprak, N.; Celebi, G.; Hekimoglu, K.; Kalaycioglu, B. Evaluation of Cervical Computed Tomography Findings in Oropharyngeal Tularaemia. Scand. J. Infect. Dis. 2008, 40, 811–814. [Google Scholar] [CrossRef] [PubMed]
- Anand, N.; Deochand, O.; Murphy, R. Imaging Findings of Ulceroglandular Tularemia. J. Radiol. Case Rep. 2017, 11, 1–6. [Google Scholar] [CrossRef]
- Strle, F.; Maraspin, V.; Pleterski-Rigler, D.; Lotric-Furlan, S.; Ruzić-Sabljić, E.; Jurca, T.; Cimperman, J. Treatment of Borrelial Lymphocytoma. Infection 1996, 24, 80–84. [Google Scholar] [CrossRef]
- What Is Borrelial Lymphocytoma in Lyme Disease? Available online: https://www.medscape.com/answers/330178-101037/what-is-borrelial-lymphocytoma-in-lyme-disease (accessed on 29 June 2022).
- Bonekamp, D.; Horton, K.M.; Hruban, R.H.; Fishman, E.K. Castleman Disease: The Great Mimic. Radiogr. Rev. Publ. Radiol. Soc. N. Am. Inc. 2011, 31, 1793–1807. [Google Scholar] [CrossRef]
- Fanous, A.; Morcrette, G.; Fabre, M.; Couloigner, V.; Galmiche-Rolland, L. Diagnostic Approach to Congenital Cystic Masses of the Neck from a Clinical and Pathological Perspective. Dermatopathology 2021, 8, 342–358. [Google Scholar] [CrossRef]
- Asano, S. Granulomatous Lymphadenitis. J. Clin. Exp. Hematop. JCEH 2012, 52, 1–16. [Google Scholar] [CrossRef]
- Gestin, B.; Valade, E.; Thibault, F.; Schneider, D.; Maurin, M. Phenotypic and Genetic Characterization of Macrolide Resistance in Francisella Tularensis Subsp. Holarctica Biovar I. J. Antimicrob. Chemother. 2010, 65, 2359–2367. [Google Scholar] [CrossRef]
- EMA. Disabling and Potentially Permanent Side Effects Lead to Suspension or Restrictions of Quinolone Fluoroquinolone Antibiotics. Available online: https://www.ema.europa.eu/en/news/disabling-potentially-permanent-side-effects-lead-suspension-restrictions-quinolone-fluoroquinolone (accessed on 29 June 2022).
- Forti, G.; Benincori, C. Doxycycline and the Teeth. Lancet Lond. Engl. 1969, 1, 782. [Google Scholar] [CrossRef]
- Rothweiler, R.; Fuessinger, M.A.; Schmelzeisen, R.; Metzger, M.C. Lymph Node Abscess Caused by Francisella Tularensis—A Rare Differential Diagnosis for Cervical Lymph Node Swelling: A Case Report. J. Med. Case Rep. 2019, 13, 247. [Google Scholar] [CrossRef] [PubMed]
- Theissing, J.; Rudack, C. ENT-Head and Neck Surgery: Essential Procedures; Thieme Medical Publishers: New York, NY, USA, 2010; pp. 264–265. [Google Scholar]
- Jessica, L.V.; Severin, D.D.M.; Chu, M.C.; Petersen, J.M. Development of a Multitarget Real-Time TaqMan PCR Assay for Enhanced Detection of Francisella Tularensis in Complex Specimens. J. Clin. Microbiol. 2003, 41, 5492–5499. [Google Scholar] [CrossRef]
- Fulop, M.J.; Leslie, D.L.; Titball, R.W. A Rapid, Highly Sensitive Method for the Detection of Francisella Tularensis in Clinical Samples Using the Polymerase Chain Reaction. Am. J. Trop. Med. Hyg. 1996, 54, 364–366. [Google Scholar] [CrossRef]
| Disease Severity | Treatment Regimen |
|---|---|
| Severe to moderate infection | ● Streptomycin 7.5 mg–1 g IM or IV, twice daily, 7–10 days OR |
| ● Gentamicin or tobramycin 5 mg/kg IV, once or twice daily, 10 days | |
| Children: | |
| ● Gentamicin 2.5 mg/kg IV, three times daily, with OR without ciprofloxacin in 10–15 mg/kg orally, twice daily | |
| Mild infection | ● Ciprofloxacin 400 mg IV or 750 mg orally, twice a day, 14–21 days OR |
| ● Doxycycline 100 mg orally or IV, twice a day, 14–21 days | |
| Children: | |
| ● above 8 years old: doxycycline 2.2 mg/kg orally, twice daily | |
| ● 1–10 years old: ciprofloxacin 10–15 mg/kg orally, twice daily | |
| Hematogenous meningitis | ● Aminoglycoside + chloramphenicol 50–100 mg/kg/day IV in 4 divided doses |
| Pregnancy | ● Streptomycin or chloramphenicol 15 mg/kg, four times a day, 14 days |
| Prophylaxis for aerosol exposure | ● Doxycycline 100 mg orally, twice daily, 14 days OR |
| ● Ciprofloxacin 500 mg, orally, twice daily, 14 days |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Troha, K.; Božanić Urbančič, N.; Korva, M.; Avšič-Županc, T.; Battelino, S.; Vozel, D. Vector-Borne Tularemia: A Re-Emerging Cause of Cervical Lymphadenopathy. Trop. Med. Infect. Dis. 2022, 7, 189. https://doi.org/10.3390/tropicalmed7080189
Troha K, Božanić Urbančič N, Korva M, Avšič-Županc T, Battelino S, Vozel D. Vector-Borne Tularemia: A Re-Emerging Cause of Cervical Lymphadenopathy. Tropical Medicine and Infectious Disease. 2022; 7(8):189. https://doi.org/10.3390/tropicalmed7080189
Chicago/Turabian StyleTroha, Kaja, Nina Božanić Urbančič, Miša Korva, Tatjana Avšič-Županc, Saba Battelino, and Domen Vozel. 2022. "Vector-Borne Tularemia: A Re-Emerging Cause of Cervical Lymphadenopathy" Tropical Medicine and Infectious Disease 7, no. 8: 189. https://doi.org/10.3390/tropicalmed7080189
APA StyleTroha, K., Božanić Urbančič, N., Korva, M., Avšič-Županc, T., Battelino, S., & Vozel, D. (2022). Vector-Borne Tularemia: A Re-Emerging Cause of Cervical Lymphadenopathy. Tropical Medicine and Infectious Disease, 7(8), 189. https://doi.org/10.3390/tropicalmed7080189








