Exposure and Carriage of Pathogenic Leptospira in Livestock in St. Croix, U.S. Virgin Islands
Abstract
1. Introduction
2. Materials and Methods
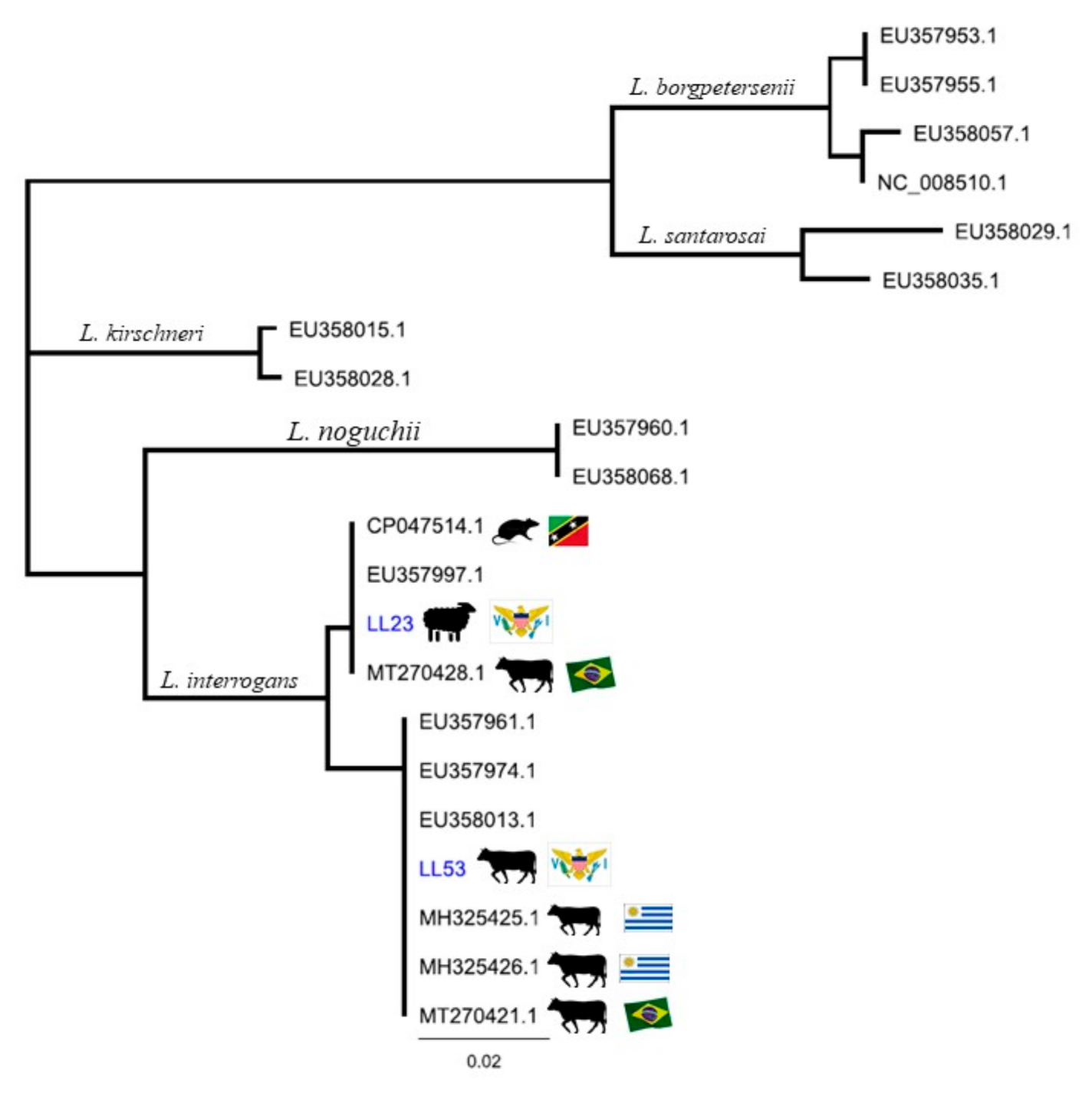
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Serogroup | Serovar | ||
|---|---|---|---|
| 1. | Australis | Australis | |
| 2. | Australis | Bratislava | |
| 3. | Autumnalis | Autumnalis | |
| 4. | Ballum | Ballum | |
| 5. | Bataviae | Bataviae | |
| 6. | Canicola | Canicola | |
| 7. | Cynopteri | Cynopteri | |
| 8. | Djasiman | Djasiman | |
| 9. | Grippotyphosa | Grippotyphosa | |
| 10. | Hebdomadis | Hebdomadis | |
| 11. | Icterohaemorrhagiae | Copenhageni | |
| 12. | Mini | Szwajizak | |
| 13. | Pomona | Pomona | |
| 14. | Pyrogenes | Pyrogenes | |
| 15. | Sejroe | Hardjo | |
| 16. | Sejroe | Undetermined (LM31) † | |
| 17. | Sejroe | Sejroe | |
| 18. | Tarassovi | Tarassovi |
Appendix B
| Specie | # | AUS † | AUT | BAL | BAT | BRA | CAN | CYN | DJA | GRI | HAR | HEB | ICT | MIN | POM | PYR | SEJ | TAR | LM31 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cattle | 51 | 100 | |||||||||||||||||
| 54 | 100 | ||||||||||||||||||
| 69 | 100 | ||||||||||||||||||
| 82 | 100 | 100 | |||||||||||||||||
| 83 | 200 | ||||||||||||||||||
| 103 | 100 | ||||||||||||||||||
| 117 | 100 | 100 | 100 | ||||||||||||||||
| Goat | 49 | 100 | |||||||||||||||||
| 100 | 200 | ||||||||||||||||||
| Pig | 56 | 100 | |||||||||||||||||
| 57 | 100 | ||||||||||||||||||
| 61 | 200 | 200 | |||||||||||||||||
| 63 | 100 | ||||||||||||||||||
| 65 | 100 | ||||||||||||||||||
| 66 | 200 | 100 | |||||||||||||||||
| 67 | 100 | ||||||||||||||||||
| 70 | 400 | 100 | 200 | 100 | |||||||||||||||
| 71 | 100 | ||||||||||||||||||
| 72 | 400 | 100 | |||||||||||||||||
| 75 | 400 | ||||||||||||||||||
| 78 | 200 | 200 | |||||||||||||||||
| 80 | 100 | 400 | |||||||||||||||||
| 81 | 400 | 400 | 200 | ||||||||||||||||
| 85 | 400 | 100 | |||||||||||||||||
| 87 | 400 | ||||||||||||||||||
| 88 | 100 | ||||||||||||||||||
| 90 | 200 | ||||||||||||||||||
| 91 | 200 | ||||||||||||||||||
| 92 | 400 | ||||||||||||||||||
| 93 | 200 | ||||||||||||||||||
| 104 | 400 | 100 | 100 | ||||||||||||||||
| 106 | 200 | ||||||||||||||||||
| 107 | 100 | ||||||||||||||||||
| 108 | 100 | 100 | |||||||||||||||||
| 109 | 100 | 400 | 200 | 800 | 100 | 200 | |||||||||||||
| 120 | 800 | 3200 | 800 | 100 | 12800 | 3200 | |||||||||||||
| 121 | 1600 | 100 | 100 | 400 | |||||||||||||||
| 123 | 200 | ||||||||||||||||||
| 124 | 200 | 400 | |||||||||||||||||
| 125 | 100 | ||||||||||||||||||
| 126 | 100 | 400 | 100 | 100 | |||||||||||||||
| Sheep | 45 | 100 | |||||||||||||||||
| 47 | 100 | 100 | |||||||||||||||||
| 48 | 100 | ||||||||||||||||||
| 59 | 200 | ||||||||||||||||||
| 102 | 100 | ||||||||||||||||||
| 110 | 100 | 800 | 800 | 3200 | 200 | 100 | 6400 | 100 | 800 |
Appendix C. secY IV Sequence for Livestock Urine Samples in St. Croix, U.S. Virgin Islands, with SNPs Highlighted in Yellow †
LL23 AGATTGGAGTTAGAGCTCAAATCTAAGAATTTTATAATAATATAAGGTGCTAATGCCAAA 60 LL53 ------GAGTTAGAGCTCAAATCTAAGAATTTTATAATAATATAAGGTGCTAATGCCAAA 54
LL23 CCTGCAAGAAACATAGCTCCGGGAAGAGTGATTCTATTTAACACTTTTTCAATGTATTCT 120 LL53 CCTGCAAGAAACATAGCTCCAGGGAGAGTGATTCTATTTAACACTTTTTCAATGTATTCT 114
LL23 TTTGTGTGAGAACCCGGACGAATTCCTGGAATGAACCCACCGTATTTTTTCAAATTCTCA 180 LL53 TTTGTGTGAGAACCCGGACGAATTCCTGGAATGAATCCACCGTATTTTTTCAAATTCTCA 174
LL23 GCCAATTCTGCAGGATTAAACTGAATCGCAATAC 214 LL53 GCCAAT---------------------------- 180
References
- Costa, F.; Hagan, J.E.; Calcagno, J.; Kane, M.; Torgerson, P.; Martinez-Silveira, M.S.; Stein, C.; Abela-Ridder, B.; Ko, A.I. Global Morbidity and Mortality of Leptospirosis: A Systematic Review. PLOS Negl. Trop. Dis. 2015, 9, e0003898. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC); National Notifiable Infectious Diseases and Conditions, United States. Annual Tables, Annual Data for 2018, Table 2h. Available online: https://wonder.cdc.gov/nndss/nndss_annual_tables_menu.asp (accessed on 3 May 2021).
- Centers for Disease Control and Prevention (CDC). Leptospirosis: Risk of Exposure. Available online: https://www.cdc.gov/leptospirosis/exposure/index.html (accessed on 3 May 2021).
- Marinova-Petkova, A.; Guendel, I.; Strysko, J.P.; Ekpo, L.L.; Galloway, R.; Yoder, J.; Kahler, A.; Artus, A.; Hoffmaster, A.R.; Bower, W.A.; et al. First Reported Human Cases of Leptospirosis in the United States Virgin Islands in the Aftermath of Hurricanes Irma and Maria, September–November 2017. Open Forum Infect. Dis. 2019, 6, ofz261. [Google Scholar] [CrossRef] [PubMed]
- Artus, A.; Cossaboom, C.; Haberling, D.; Sutherland, G.; Galloway, R.; Villarma, A.; Browne, A.S.; Carillo, M.; Delgado, D.; Doyle, J.; et al. Seroprevalence of human leptospirosis in the U.S. Virgin Islands. In Proceedings of the 11th International Leptospirosis Conference, Vancouver, BC, Canada, 8–12 July 2019. poster 33. [Google Scholar]
- Ahl, A.S.; Miller, D.A.; Bartlett, P.C. Leptospira Serology in Small Ruminants on St. Croix, U.S. Virgin Islands. Ann. New York Acad. Sci. 1992, 653, 168–171. [Google Scholar] [CrossRef]
- Everard, C.O.R.; Fraser-Chanpong, G.M.; James, A.C.; Butcher, L.V. Serological studies on leptospirosis in livestock and chickens from Grenada and Trinidad. Trans. R. Soc. Trop. Med. Hyg. 1985, 79, 859–864. [Google Scholar] [CrossRef]
- Suepaul, S.; Suepaul, S.; Carrington, C.; Carrington, C.; Campbell, M.; Campbell, M.; Borde, G.; Borde, G.; Adesiyun, A.; Adesiyun, A. Seroepidemiology of leptospirosis in livestock in Trinidad. Trop Anim. Health Prod. 2011, 43, 367–375. [Google Scholar] [CrossRef]
- Bradford, B. (Territorial Veterinarian, U.S. Virgin Islands Department of Agriculture, USVI, USA). Personal communication, 2020. [Google Scholar]
- Cole, J.R., Jr.; Sulzer, C.R.; Pursell, A.R. Improved Microtechnique for the Leptospiral Microscopic Agglutination Test. Appl. Microbiol. 1973, 25, 976–980. [Google Scholar] [CrossRef] [PubMed]
- Ellis, W.A.; Montgomery, J.; Cassells, J.A. Dihydrostreptomycin treatment of bovine carriers of Leptospira interrogans serovar hardjo. Res. Vet. Sci. 1985, 39, 292–295. [Google Scholar] [CrossRef]
- Hornsby, R.L.; Alt, D.P.; Nally, J.E. Isolation and propagation of leptospires at 37 °C directly from the mammalian host. Scientific reports 2020, 10, 9620. [Google Scholar] [CrossRef]
- Nally, J.E.; Hornsby, R.L.; Alt, D.P.; Bayles, D.; Wilson-Welder, J.H.; Palmquist, D.E.; Bauer, N.E. Isolation and characterization of pathogenic leptospires associated with cattle. Vet. Microbiol. 2018, 218, 25–30. [Google Scholar] [CrossRef]
- Galloway, R.L.; Hoffmaster, A.R. Optimization of LipL32 PCR assay for increased sensitivity in diagnosing leptospirosis. Diagn. Microbiol. Infect. Dis. 2015, 82, 199–200. [Google Scholar] [CrossRef]
- Stoddard, R.A.; Gee, J.E.; Wilkins, P.P.; McCaustland, K.; Hoffmaster, A.R. Detection of pathogenic Leptospira spp. through TaqMan polymerase chain reaction targeting the LipL32 gene. Diagn. Microbiol. Infect. Dis. 2009, 64, 247–255. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.; Engelberts, M.F.M.; Boer, K.R.; Ahmed, N.; Hartskeerl, R.A. Development and Validation of a Real-Time PCR for Detection of Pathogenic Leptospira Species in Clinical Materials. PLoS ONE 2009, 4, e7093. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, N.; Devi, S.M.; Valverde, M.d.l.A.; Vijayachari, P.; Machang’u, R.S.; Ellis, W.A.; Hartskeerl, R.A. Multilocus Sequence Typing method for identification and genotypic classification of pathogenic Leptospira species. Ann. Clin. Microbiol. Antimicrob. 2006, 5, 28. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hamond, C.; Pestana, C.P.; Medeiros, M.A.; Lilenbaum, W. Genotyping of Leptospira directly in urine samples of cattle demonstrates a diversity of species and strains in Brazil. Epidemiol. Infect. 2016, 144, 72–75. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing (3.2.1); R Foundation for Statistical Computing. Available online: http://www.r-project.org/ (accessed on 3 May 2021).
- Di Azevedo, M.I.N.; Pires, B.C.; Libonati, H.; Pinto, P.S.; Cardoso Barbosa, L.F.; Carvalho-Costa, F.A.; Lilenbaum, W. Extra-renal bovine leptospirosis: Molecular characterization of the Leptospira interrogans Sejroe serogroup on the uterus of non-pregnant cows. Vet. Microbiol. 2020, 250, 108869. [Google Scholar]
- Zarantonelli, L.; Suanes, A.; Meny, P.; Buroni, F.; Nieves, C.; Salaberry, X.; Briano, C.; Ashfield, N.; Da Silva Silveira, C.; Dutra, F.; et al. Isolation of pathogenic Leptospira strains from naturally infected cattle in Uruguay reveals high serovar diversity, and uncovers a relevant risk for human leptospirosis. PLoS Negl. Trop. Dis. 2018, 12, e0006694. [Google Scholar] [CrossRef] [PubMed]
- Rajeev, S.; Shiokawa, K.; Llanes, A.; Rajeev, M.; Restrepo, C.M.; Chin, R.; Cedeño, E.; Ellis, E. Detection and Characterization of Leptospira Infection and Exposure in Rats on the Caribbean Island of Saint Kitts. Animals 2020, 10, 350. [Google Scholar] [CrossRef] [PubMed]
- Pratt, N.; Rajeev, S. Leptospira seroprevalence in animals in the Caribbean region: A systematic review. Acta Trop. 2018, 182, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Miller, D.A.; Wilson, M.A.; Beran, G.W. Survey to estimate prevalence of Leptospira interrogans infection in mature cattle in the United States. Am. J. Vet. Res. 1991, 52, 1761–1765. [Google Scholar] [PubMed]
- Nally, J.E.; Wilson-Welder, J.H.; Hornsby, R.L.; Palmer, M.V.; Alt, D.P. Inbred Rats as a Model to Study Persistent Renal Leptospirosis and Associated Cellular Immune Responsiveness. Front. Cell. and Infect. Microbiol. 2018, 8, 66. [Google Scholar] [CrossRef] [PubMed]
- Senepol History & Development Brochure. Available online: https://www.senepolcattle.com/images/pdfs/English_Version.pdf (accessed on 3 May 2021).
| Species | Female | Male | Total Animals | Number of Farms |
|---|---|---|---|---|
| Cattle | 17 | 11 | 28 | 5 |
| Goat | 19 | 0 | 19 | 8 |
| Pig | 25 | 21 | 46 | 13 |
| Sheep | 7 | 26 | 33 | 12 |
| Total | 49 | 77 | 126 | 33 † |
| Livestock Sampled | Serogroup | % | 95% CI |
|---|---|---|---|
| Cattle (n = 28) | Ballum | 10.7% (3/28) | 2.3–28.2 |
| Sejroe | 10.7% (3/28) | 2.3–28.2 | |
| Icterohaemorrhagiae | 7.1% (2/28) | 0.8–23.5 | |
| Australis | 3.6% (1/28) | 0.1–18.4 | |
| Djasiman | 3.6% (1/28) | 0.1–18.4 | |
| Goat (n = 19) | Cynopteri | 5.3% (1/19) | 0.1–26.0 |
| Icterohaemorrhagiae | 5.3% (1/19) | 0.1–26.0 | |
| Pig (n = 46) | Australis | 56.5% (26/46) | 41.1–71.1 |
| Djasiman | 28.3% (13/46) | 16.0–43.5 | |
| Icterohaemorrhagiae | 21.7% (10/46) | 11.0–36.4 | |
| Ballum | 15.4% (4/46) | 2.4–10.8 | |
| Sejroe | 15.4% (4/46) | 2.4–10.8 | |
| Cynopteri | 6.5% (3/46) | 1.4–17.9 | |
| Grippotyphosa | 2.2% (1/46) | 0.1–11.5 | |
| Hebdomadis | 2.2% (1/46) | 0.1–11.5 | |
| Pomona | 2.2% (1/46) | 0.1–11.5 | |
| Sheep (n = 33) | Australis | 9.1% (3/33) | 1.9–24.3 |
| Autumnalis | 6.1% (2/33) | 0.7–20.2 | |
| Ballum | 6.1% (2/33) | 0.7–20.2 | |
| Canicola | 3.0% (1/33) | 0.1–15.8 | |
| Cynopteri | 3.0% (1/33) | 0.1–15.8 | |
| Djasiman | 3.0% (1/33) | 0.1–15.8 | |
| Sejroe | 3.0% (1/33) | 0.1–15.8 | |
| Hebdomadis | 3.0% (1/33) | 0.1–15.8 | |
| Icterohaemorrhagiae | 3.0% (1/33) | 0.1–15.8 | |
| Pomona | 3.0% (1/33) | 0.1–15.8 |
| Species | MAT | FAT | rt-PCR | Culture |
|---|---|---|---|---|
| Cattle | 7/28 (25.0%, 95% CI 10.7–45.0) | 0/28 | 1/28 (3.6%, 95% CI 0.1–18.4) | 0/28 |
| Goat | 2/18 (11.1%, 95% CI 1.4–34.7) | 0/16 | 1/16 (6.3%, 95% CI 0.2–30.2) | 0/16 |
| Pig | 32/46 (67.0%, 95% CI 54.3–82.3) | 0/22 | 0/25 (0%) | 0/27 |
| Sheep | 6/33 (18.2%, 95% CI 7.0–35.5) | 0/31 | 2/32 (6.3%, 95% CI 0.8–21.0) | 0/32 |
| Totals | 47/125 (37.6%, 95% CI 29.1–46.7) | 0/97 | 4/101 (4.0%, 95% CI 1.1–10.0) | 0/103 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cranford, H.M.; Taylor, M.; Browne, A.S.; Alt, D.P.; Anderson, T.; Hamond, C.; Hornsby, R.L.; LeCount, K.; Schlater, L.; Stuber, T.; et al. Exposure and Carriage of Pathogenic Leptospira in Livestock in St. Croix, U.S. Virgin Islands. Trop. Med. Infect. Dis. 2021, 6, 85. https://doi.org/10.3390/tropicalmed6020085
Cranford HM, Taylor M, Browne AS, Alt DP, Anderson T, Hamond C, Hornsby RL, LeCount K, Schlater L, Stuber T, et al. Exposure and Carriage of Pathogenic Leptospira in Livestock in St. Croix, U.S. Virgin Islands. Tropical Medicine and Infectious Disease. 2021; 6(2):85. https://doi.org/10.3390/tropicalmed6020085
Chicago/Turabian StyleCranford, Hannah M., Marissa Taylor, Andrew Springer Browne, David P. Alt, Tammy Anderson, Camila Hamond, Richard L. Hornsby, Karen LeCount, Linda Schlater, Tod Stuber, and et al. 2021. "Exposure and Carriage of Pathogenic Leptospira in Livestock in St. Croix, U.S. Virgin Islands" Tropical Medicine and Infectious Disease 6, no. 2: 85. https://doi.org/10.3390/tropicalmed6020085
APA StyleCranford, H. M., Taylor, M., Browne, A. S., Alt, D. P., Anderson, T., Hamond, C., Hornsby, R. L., LeCount, K., Schlater, L., Stuber, T., De Wilde, L., Burke-France, V. J., Ellis, E. M., Nally, J. E., & Bradford, B. (2021). Exposure and Carriage of Pathogenic Leptospira in Livestock in St. Croix, U.S. Virgin Islands. Tropical Medicine and Infectious Disease, 6(2), 85. https://doi.org/10.3390/tropicalmed6020085







