National Antimicrobial Consumption: Analysis of Central Warehouses Supplies to In-Patient Care Health Facilities from 2017 to 2019 in Uganda
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Period
2.2. Study Setting
2.2.1. General Setting
| Levels of Health Care | Type of Service | Services Provided | 1 NMS | 2 JMS |
|---|---|---|---|---|
| National Referral Hospital (NRH) | Tertiary(specialized) | Inpatient; Outpatient care | 2 | |
| Regional Referral Hospital (RRH) | Tertiary | Inpatient; Outpatient care | 18 | 4 |
| General Hospital (GH) | Secondary | Inpatient; Outpatient care | 50 | 97 |
| Health centre (HC) IV | Secondary | Inpatient; Outpatient care | 179 | 92 |
| Health centre III | Primary | Outpatient care only | ||
| Health centre II | Primary | Outpatient care only | ||
| Total | 249 | 193 |
2.2.2. Specific Setting
2.3. Study Sites
2.4. Study Procedure, Data Sources, and Variables
2.5. Data Collection, Sources, and Variables
2.6. Data Analysis and Statistics
3. Results
3.1. Antimicrobial Products Supplied by NMS and JMS in Uganda from 2017 to 2019
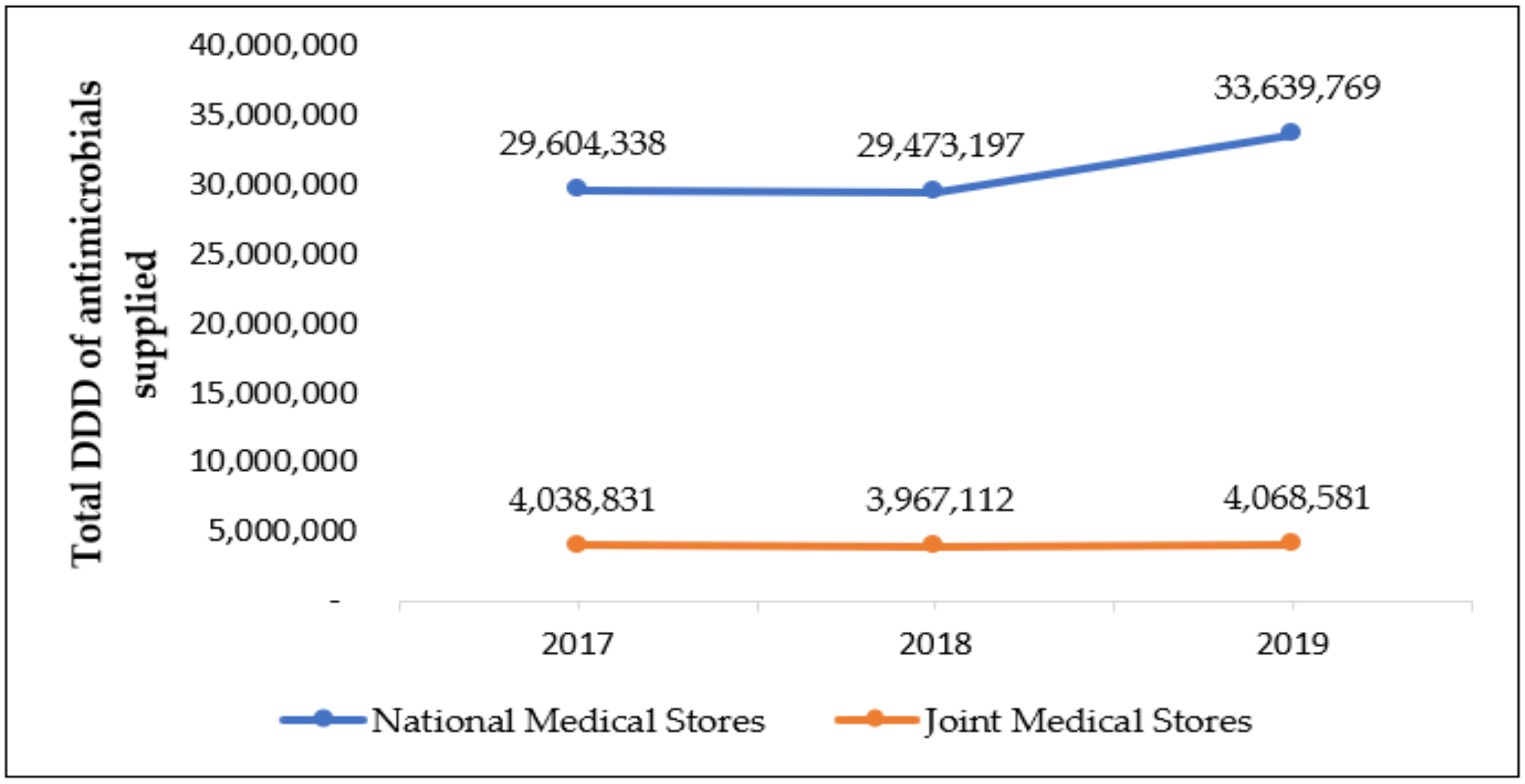
3.2. Total Defined Daily Doses of Antimicrobials per Year from 2017 to 2019
3.3. Top 10 Antimicrobials Supplied by NMS and JMS to Health Facilities from 2017 to 2019
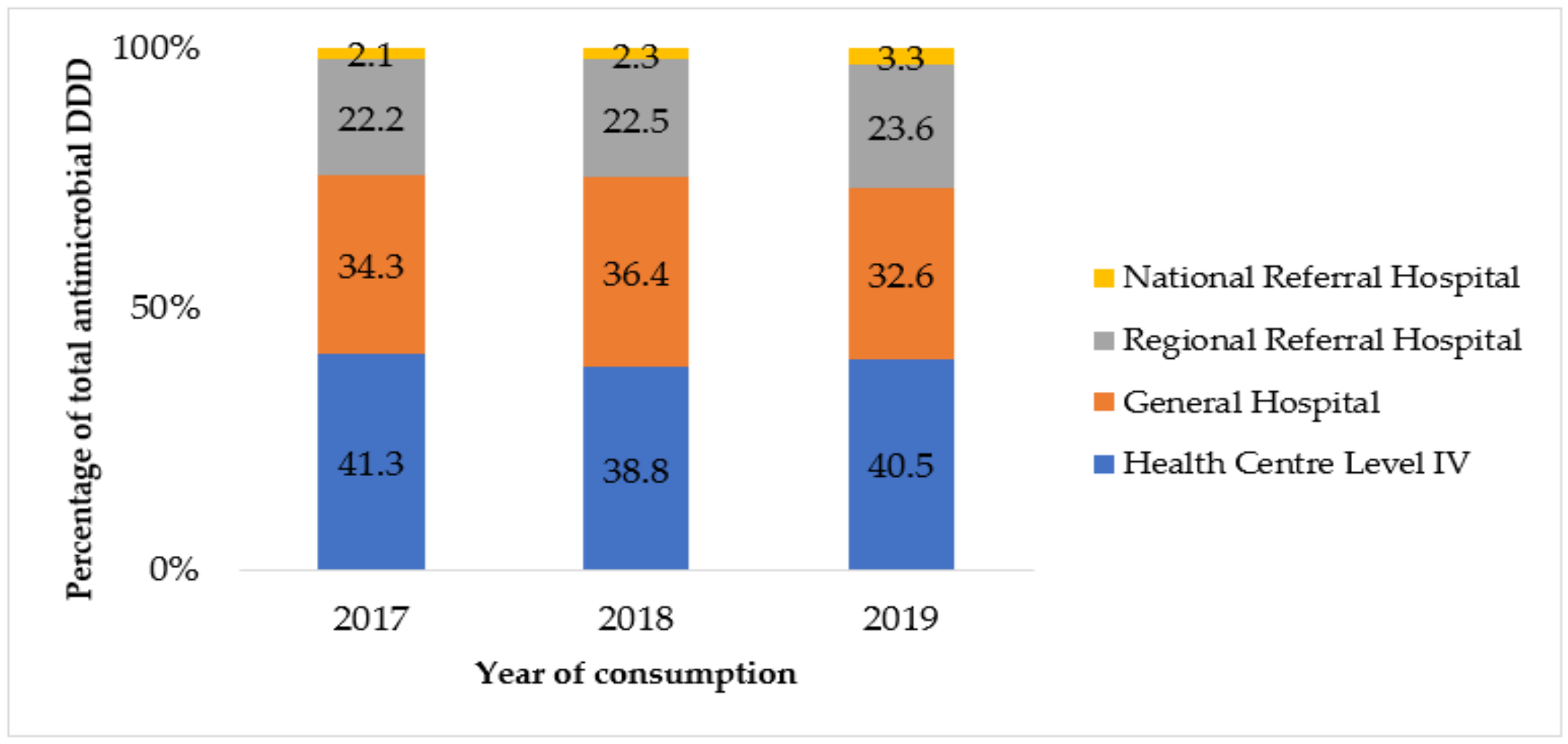
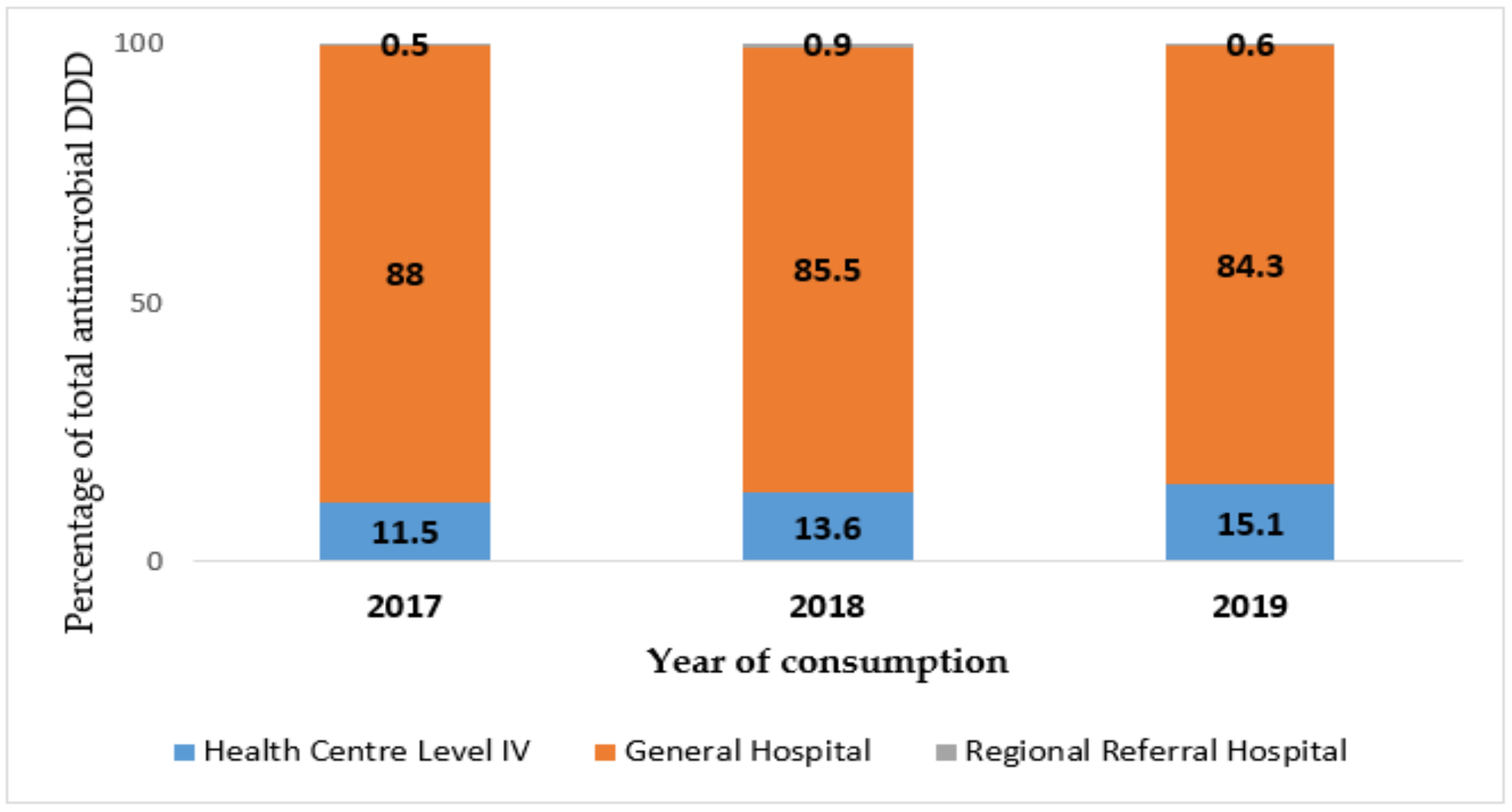
3.4. Analysis of Total DDD Supplied to Each Level of Care of Health Facilities per Year
3.5. Antimicrobial Consumption by AWaRe Categorization
3.6. Antimicrobial Consumption Density at Different Levels of Care Using Hospital Activity Units
3.7. Analysis of Antimicrobial Consumption by Pharmacological Subgroup
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Product Name | Pack Size | Paediatric | ** AWaRe | Warehouse | Total DDD 2017–2019 | |
|---|---|---|---|---|---|---|
| Nitroimidazole derivatives (P01AB) | ||||||
| 1 | Metronidazole tablet 200 mg tablet | 100, 1000 | No | Access | Both | 14,055,650 |
| 2 | Metronidazole oral suspension 200 mg/5 mL | 100 mL bottle | Yes | Access | Both | 95,602 |
| 3 | Metronidazole rectal suppositories 500 mg | 10 supp | No | Access | NMS | 4950 |
| 4 | Metronidazole infusion, 500 mg/100 mL | 100 mL bottle | No | Access | Both | 938,357 |
| 5 | Ornidazole injection, bottle 500 mg/100 mL | 100 mL bottle | No | Access | Both | 7475 |
| 6 | Tinidazole tablet 500 mg | 100 | No | Access | NMS | 28,725 |
| 7 | Secnidazole tablet 1 G | 2 | No | Access | JMS | 7806 |
| Tetracyclines (J01A) | ||||||
| 8 | Doxycycline capsules 100 mg | 100 | No | Access | Both | 12,533,400 |
| Amphenicols (J01B) | ||||||
| 9 | Chloramphenicol capsule 250 mg capsule | 100, 1000 | No | Access | Both | 124,920 |
| 10 | Chloramphenicol oral suspension 125 mg/5 mL | 100 mL bottle | Yes | Access | JMS | 3004 |
| 11 | Chloramphenicol injection, vial 1 G | 50 vials | No | Access | Both | 93,706 |
| Penicillins, betalactams (J01C) | ||||||
| 12 | Amoxicillin capsule 250 mg | 1000, 100 | No | Access | Both | 31,689,473 |
| 13 | Amoxicillin dispersable tablets 125 mg | 20, 100 | Yes | Access | Both | 50,427 |
| 14 | Amoxicillin dispersable tablets 250 mg | 10, 20, 100 | Yes | Access | NMS | 2,110,359 |
| 15 | Amoxicillin powder for suspension 125 mg/5 mL | 100 mL bottle | Yes | Access | JMS | 78,260 |
| 16 | Amoxicillin clavulanate injection 1.2 G | 1 vial | No | Access | Both | 1356 |
| 17 | Amoxicillin clavulanate tablet 1000 mg | 10, 14 | No | Access | JMS | 4672 |
| 18 | Amoxicillin clavulanate syrup 156 mg/5 mL | 100 mL bottle | Yes | Access | Both | 937 |
| 19 | Amoxicillin clavulanate tablet 228.5 mg tablet | 10 | Yes | Access | JMS | 552 |
| 20 | Amoxicillin clavulanate syrup 228.5 mg/5 mL | 100 mL bottle | Yes | Access | JMS | 57,798 |
| 21 | Amoxicillin clavulanate tablet 625 mg tablet | 14, 15, 16, 20 | No | Access | Both | 66,615 |
| 22 | Amoxicillin/clavulanic acid tablet 375 mg | 10, 20, 100 | No | Access | JMS | 43,710 |
| 23 | Ampicillin injection, vial 500 mg | 100 mL bottle | No | Access | Both | 290,074 |
| 24 | Ampicillin oral suspension 125 mg/5 mL | 100 mL bottle | Yes | Access | JMS | 3021 |
| 25 | Ampicillin capsules 250 mg | 100 | No | Access | JMS | 613 |
| 26 | Ampicillin/cloxacillin injection, vial 250 mg/250 mg | 1, 100 vials | No | Access | NMS | 2,871,400 |
| 27 | Ampicillin/cloxacillin capsules 250 mg/250 mg | 100, 200 | No | Access | NMS | 4,438,100 |
| 28 | Ampicillin/cloxacillin oral suspension 125 mg/5 mL | 100 mL bottle | Yes | Access | JMS | 121,550 |
| 29 | Benzathine benzylpenicillin injection, vial 2.4 mu/1.44 g | 10, 50 vials | No | Access | Both | 89,388 |
| 30 | Benzylpenicillin injection, vial 1 MU/600 mg | 10, 50 vials | No | Access | Both | 483,027 |
| 31 | Cloxacillin capsules 250 mg | 100 | No | Access | Both | 338,363 |
| 32 | Cloxacillin injection, vial 500 mg | 50 vials | No | Access | Both | 127,688 |
| 33 | Phenoxymethylpenicillin tablets 250 mg | 100 | No | Access | JMS | 84,763 |
| 34 | Piperacillin-tazobactam injection, vial 4.5 g | 1 vial | No | Watch | Both | 63,730 |
| 35 | Procaine benzylpenicillin injection, vial 4 MU | 10, 50 | No | Access | NMS | 24,160 |
| 36 | Flucloxacillin/amoxicillin oral suspension 250 mg | 80 mL bottle | No | Access | Both | * |
| 37 | Flucloxacillin/amoxicillin capsules 250/250 mg | 16 | No | Access | Both | * |
| 38 | Flucloxacillin/amoxicillin injection, vial 500 mg/500 mg | 1 vial | No | Access | Both | * |
| Carbapenems (J01DH) | ||||||
| 39 | Imipenem/cilastatin injection, vial 500/500 mg | 1 vial | No | Watch | NMS | 3 |
| 40 | Meropenem injection, vial 500 mg | 1 vial | No | Watch | NMS | 21,391 |
| Cephalosporins (J01D) | ||||||
| 41 | Cefixime tablets 200 mg | 10, 12, 20, 120 | No | Watch | Both | 491,015 |
| 42 | Cefixime capsules 400 mg | 5, 6, 100 | No | Watch | Both | 44,489 |
| 43 | Cefixime oral suspension 50 mg/5 mL | 100 mL bottle | Yes | Watch | JMS | 37,513 |
| 44 | Cefixime oral suspension 100 mg/5 mL | 50, 60 mL bottle | Yes | Watch | JMS | 7112 |
| 45 | Cefotaxime injection, vial 1 G | 1 vial | No | Watch | Both | 9563 |
| 46 | Cefotaxime injection, vial 500 mg | 1 vial | No | Watch | NMS | 34,718 |
| 47 | Cefpodoxime tablets 200 mg | 10 | No | Watch | JMS | 3140 |
| 48 | Cefpodoxime suspension 50 mg/5 mL | 100 mL bottle | Yes | Watch | JMS | 455 |
| 49 | Ceftriaxone injection, vial 1 G | 1 vial | No | Watch | Both | 3,536,638 |
| 50 | Ceftriaxone injection, vial 500 mg | 1 vial | No | Watch | JMS | 1303 |
| 51 | Ceftriaxone + sulbactam injection, vial 1.5 G | 1 vial | No | Watch | JMS | 5297 |
| 52 | Cefuroxime tablets 250 mg | 10 | No | Watch | JMS | 9660 |
| 53 | Cefuroxime tablets 500 mg | 10, 100 | No | Watch | Both | 830,940 |
| 54 | Cefuroxime injection, vial 1.5 G | 1 vial | No | Watch | NMS | 650 |
| 55 | Cefuroxime injection, vial 750 mg | 1 vial | No | Watch | NMS | 250 |
| 56 | Cefuroxime suspension 125 mg/5 mL | 50, 70, 100 mL bottle | Yes | Watch | Both | 44,180 |
| 57 | Cephalexin capsules 250 mg | 100 | No | Access | JMS | 156,250 |
| 58 | Cephalexin oral suspension 125 mg/5 mL | 100 mL bottle | Yes | Access | JMS | 21,318 |
| Sulphonamides and Trimethoprim (J01E) | ||||||
| 59 | Cotrimoxazole tablet 120 mg | 100, 1000 | Yes | Access | Both | 35,904 |
| 60 | Cotrimoxazole tablet 480 mg | 100, 1000 | No | Access | Both | 4,902,800 |
| 61 | Cotrimoxazole tablet 960 mg | 100, 1000 | No | Access | Both | 6,474,350 |
| 62 | Cotrimoxazole oral suspension 240 mg/5 mL | 60, 100 mL bottle | Yes | Access | JMS | 14,593 |
| 63 | Cotrimoxazole injection, vial 80 mg/400 mg | 1 vial | No | Access | NMS | 100 |
| Macrolides and Lincosamides (J01F) | ||||||
| 64 | Azithromycin tablet 250 mg | 180, 500 | No | Watch | NMS | 791,218 |
| 65 | Azithromycin tablet 500 mg | 3, 6, 30, 90 | No | Watch | Both | 2,009,970 |
| 66 | Azithromycin oral suspension 200 mg/5 mL | 15 mL bottle | Yes | Watch | JMS | 30,712 |
| 67 | Erythromycin oral suspension 125 mg/5 mL | 100 mL bottle | Yes | Watch | Both | 102,613 |
| 68 | Erythromycin tablet 250 mg | 100, 1000 | No | Watch | Both | 2,895,650 |
| 69 | Clarithromycin tablet 500 mg | 7, 14 | No | Watch | Both | 150,451 |
| 70 | Clindamycin capsules 150 mg | 50 | No | Access | JMS | 4794 |
| Aminoglycosides (J01G) | ||||||
| 71 | Amikacin Injection vial 500 mg/2 mL | 1 vial | No | Access | NMS | 16,612 |
| 72 | Gentamicin injection, ampoule 80 mg/2 mL | 100 vials | No | Access | Both | 1,171,049 |
| 73 | Kanamycin injection, ampoule 1 G | 10, 50 vials | No | Watch | NMS | 19,740 |
| 74 | Streptomycin injection, vial 1 G | 50, 100 vials | No | Watch | NMS | 69,600 |
| Fluoroquinolones (J01M) | ||||||
| 75 | Ciprofloxacin tablet 500 mg | 10, 100 | No | Watch | Both | 9,671,035 |
| 76 | Ciprofloxacin tablet 750 mg | 20 | No | Watch | JMS | 10,125 |
| 77 | Ciprofloxacin tablet 250 mg | 100 | No | Watch | JMS | 11,075 |
| 78 | Ciprofloxacin injection, 200 mg/100 mL | 100 mL bottle | No | Watch | Both | 49,688 |
| 79 | Levofloxacin tablet 250 mg | 100 | No | Watch | NMS | 841,500 |
| 80 | Levofloxacin tablet 500 mg | 5, 10 tablets | No | Watch | Both | 136,010 |
| 81 | Levofloxacin tablet 750 mg | 10 | No | Watch | JMS | 11,775 |
| 82 | Levofloxacin injection, bottle 500 mg/150 mL | 150 mL bottle | No | Watch | Both | 12,221 |
| 83 | Levofloxacin injection, bottle 750 mg/100 mL | 100 mL bottle | No | Watch | NMS | 1860 |
| 84 | Moxifloxacin tablet 400 mg | 100 | No | Watch | NMS | 181,600 |
| 85 | Nalidixic acid tablet 500 mg | 100, 1000 | No | Watch | Both | 19,975 |
| 86 | Ofloxacin injection, bottle 200 mg/100 mL | 200 mL bottle | No | Watch | NMS | 4730 |
| 87 | Ofloxacin tablet 200 mg | 100 | No | Watch | JMS | 432 |
| 88 | Pefloxacin tablet 400 mg | 100 | No | Watch | JMS | 3950 |
| 89 | Ofloxacin and ornidazole tablets 200/500 mg | 100 | No | Watch | JMS | 13,110 |
| Glycopeptides, Nitrofuran, Linezolid (J01X) | ||||||
| 90 | Linezolid tablet 600 mg | 10, 20, 100 | No | Reserve | NMS | 13,845 |
| 91 | Nitrofurantoin tablet 100 mg | 100, 1000 | No | Access | Both | 1,352,050 |
| 92 | Vancomycin injection, vial 500 mg | 1 vial | No | Watch | NMS | 128 |
References
- Mcewen, S.A.; Collignon, P.J. Antimicrobial Resistance: A One Health Perspective. In Antimicrobial Resistance in Bacteria from Livestock and Companion Animals; American Society for Microbiology: Washington, DC, USA, 2018; pp. 1–26. [Google Scholar] [CrossRef]
- Kallen, M.C.; Natsch, S.; Opmeer, B.C.; Hulscher, M.E.J.L.; Schouten, J.A.; Prins, J.M. How to measure quantitative antibiotic use in order to support antimicrobial stewardship in acute care hospitals: A retrospective observational study. Eur. J. Clin. Microbiol. Infect. Dis. 2019, 38, 347–355. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, J.I.M.O. Tackling Drug Resistant Infections Globally: Final Report and Recommendations; Welcome Trust-HM Government: London, UK, 2016. [Google Scholar]
- World Bank. Drug-Resistant Infections: A Threat to Our Economic Future; World Bank: Washington, DC, USA, 2017. [Google Scholar]
- UNAS. Antibiotic Resistance in Uganda: Situational Analysis and Recommendations; Uganda National Academy of Sciences: Kampala, Uganda, 2015; ISBN 9789970424108. [Google Scholar]
- Kajumbula, H.; Fujita, A.W.; Mbabazi, O.; Najjuka, C.; Izale, C.; Akampurira, A.; Aisu, S.; Lamorde, M.; Walwema, R.; Bahr, N.C.; et al. Antimicrobial drug resistance in blood culture isolates at a tertiary hospital, Uganda. Emerg. Infect. Dis. 2018, 24, 174–175. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Action Plan on Antimicrobial Resistance; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- Ministry of Health Uganda. Antimicrobial Resistance National Action Plan; Ministry of Health: Kampala, Uganda, 2018.
- Holmes, A.H.; Moore, L.S.P.; Sundsfjord, A.; Steinbakk, M.; Regmi, S.; Karkey, A.; Guerin, P.J.; Piddock, L.J.V. Understanding the mechanisms and drivers of antimicrobial resistance. Lancet 2016, 387, 176–187. [Google Scholar] [CrossRef]
- Almagor, J.; Temkin, E.; Benenson, I.; Fallach, N. The impact of antibiotic use on transmission of resistant bacteria in hospitals: Insights from an agent-based model. PLoS ONE 2018, 13, e0197111. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Methodology for a Global Programme on Surveillance of Antimicrobial Consumption; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- World Health Organization. WHO Model List of Essential Medicines; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Sharland, M.; Pulcini, C.; Harbarth, S.; Zeng, M.; Gandra, S.; Mathur, S.; Magrini, N.; 21st WHO Expert Committee on Selection and Use of Essential Medicines. Classifying antibiotics in the WHO Essential Medicines List for optimal use-be AWaRe. Lancet Infect. Dis. 2018, 18, 18–20. [Google Scholar] [CrossRef]
- Ministry of Health Uganda. Health Sector Strategic Plan III 2010/11–2014/15; Ministry of Health: Kampala, Uganda, 2014.
- UBOS, Uganda Bureau of Statistics National Population Projections by Single Age and Sex; Uganda Bureau of Statistics: Kampala, Uganda, 2017; ISBN 9781626239777.
- Ugandan Ministry of Health. The Uganda Malaria Reduction Strategic Plan 2014–2020; Ministry of Health: Kampala, Uganda, 2014; pp. 1–83.
- National Medical Stores. FY 2019-20—NMS Annual Performance Report; National Medical Stores: Kampala, Uganda, 2020.
- Joint Medical Stores Uganda. 2019/2020 Annual Report; Joint Medical Stores Uganda: Kampala, Uganda, 2020. [Google Scholar]
- Bank of Uganda. Major Forex Exchange Rates. Available online: https://www.bou.or.ug/bou/bouwebsite/bouwebsitecontent/ExchangeRates/scripts.MajorExchangeRates/index.jsp?year=2020&month=Jul&day=01 (accessed on 6 April 2021).
- World Health Organization. GLASS Guide for National Surveillance Systems for Monitoring Antimicrobial Consumption in Hospitals; World Health Organization: Geneva, Switzerland, 2020; ISBN 9789240000421. [Google Scholar]
- World Health Organization. WHO Report on Surveillance of Antibiotic Consumption, Early Implementation 2016 to 2018; World Health Organization: Geneva, Switzerland, 2018; ISBN 9789241514880. [Google Scholar]
- Ministry of Health Uganda. Annual Pharmaceutical Sector Performance Report 2015–2016; Ministry of Health: Kampala, Uganda, 2017.
- Kiguba, R.; Karamagi, C.; Bird, S.M. Extensive antibiotic prescription rate among hospitalized patients in Uganda: But with frequent missed-dose days. J. Antimicrob. Chemother. 2016, 71, 1697–1706. [Google Scholar] [CrossRef] [PubMed]
- Boyles, T.H.; Oxon, B.M.B.; Sa, C.I.D.; Naicker, V.; Rawoot, N.; Raubenheimer, P.J.; Chb, M.B.; Eick, B.; Chb, M.B.; Mendelson, M.; et al. Sustained reduction in antibiotic consumption in a South African public sector hospital: Four-year outcomes from the Groote Schuur Hospital antibiotic stewardship programme. S. Afr. Med. J. 2017, 107, 115–118. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Draft Thirteenth General Programmae of Work, 2019–2023; WHO Press: Geneva, Switzerland, 2018; Volume 50. [Google Scholar]
- Ministry of Health Uganda. Essential Medicines and Health Supplies List for Uganda (EMHSLU) 2016; Ministry of Health: Kampala, Uganda, 2016.
- Dijk, K.H.; Van, L. The world medicines situation: Rational use of medicines. World Med. Situat. 2011, 2, 24–30. [Google Scholar]
- Obakiro, S.B.; Kiyimba, K.; Paasi, G.; Napyo, A.; Anthierens, S.; Waako, P.; Van Royen, P.; Iramiot, J.S.; Goossens, H.; Kostyanev, T. Prevalence of antibiotic-resistant bacteria among patients in two tertiary hospitals in Eastern Uganda. J. Glob. Antimicrob. Resist. 2021, 25, 82–86. [Google Scholar] [CrossRef] [PubMed]
- Najjuka, C.F.; Kateete, D.P.; Kajumbula, H.M.; Joloba, M.L.; Essack, S.Y. Antimicrobial susceptibility profiles of Escherichia coli and Klebsiella pneumoniae isolated from outpatients in urban and rural districts of Uganda. BMC Res. Notes 2016, 9, 1–14. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Antimicrobial Resistance and Use Surveillance System (GLASS) Report; World Health Organization: Geneva, Switzerland, 2020; ISBN 9789240005587. [Google Scholar]
| 2017 | 2018 | 2019 | ||||
|---|---|---|---|---|---|---|
| Ranking | Antibiotic | % of DDD | Antibiotic | % of DDD | Antibiotic | % of DDD |
| National Medical Stores | ||||||
| 1 | Amoxicillin | 38.7% | Amoxicillin | 38.3% | Amoxicillin | 26.8% |
| 2 | Metronidazole | 15.6% | Metronidazole | 17.4% | Cotrimoxazole | 19.9% |
| 3 | Doxycycline | 14.7% | Doxycycline | 12.1% | Metronidazole | 12.2% |
| 4 | Ciprofloxacin | 10.4% | Ciprofloxacin | 10.1% | Doxycycline | 9.8% |
| 5 | Cotrimoxazole | 9.0% | Cotrimoxazole | 6.3% | Ampicillin-Cloxacillin | 8.6% |
| 6 | Ceftriaxone | 2.8% | Ceftriaxone | 4.2% | Ciprofloxacin | 7.2% |
| 7 | Erythromycin | 2.2% | Erythromycin | 2.9% | Azithromycin | 5.3% |
| 8 | Gentamicin | 1.5% | Ampicillin - Cloxacillin | 2.3% | Erythromycin | 2.2% |
| 9 | Ampicillin-Cloxacillin | 1.4% | Gentamicin | 1.4% | Ceftriaxone | 2.1% |
| 10 | Levofloxacin | 1.1% | Levofloxacin | 1.2% | Nitrofurantoin | 1.8% |
| Joint Medical Stores | ||||||
| 1 | Amoxicillin | 15.8% | Amoxicillin | 18.1% | Amoxicillin | 18.5% |
| 2 | Ciprofloxacin | 12.4% | Doxycycline | 11.6% | Doxycycline | 9.8% |
| 3 | Doxycycline | 10.2% | Metronidazole | 10.4% | Metronidazole | 9.7% |
| 4 | Metronidazole | 9.8% | Ciprofloxacin | 10.2% | Ampicillin-Cloxacillin | 9.6% |
| 5 | Ampicillin-Cloxacillin | 8.2% | Ampicillin-Cloxacillin | 7.4% | Ciprofloxacin | 9.1% |
| 6 | Azithromycin | 7.8% | Erythromycin | 6.5% | Azithromycin | 7.2% |
| 7 | Erythromycin | 6.9% | Ceftriaxone | 6.3% | Ceftriaxone | 7.0% |
| 8 | Ceftriaxone | 5.8% | Azithromycin | 6.3% | Erythromycin | 5.5% |
| 9 | Gentamycin | 3.1% | Cloxacillin | 3.5% | Cloxacillin | 3.3% |
| 10 | Cefixime | 3.0% | Gentamycin | 3.0% | Nitrofurantoin | 3.3% |
| AWaRe | Total DDD | |||||
|---|---|---|---|---|---|---|
| 2017 sum | % | 2018 sum | % | 2019 sum | % | |
| National Medical Stores | ||||||
| Access | 24,282,698.7 | 82.0 | 23,620,557.4 | 80.1 | 26,980,972.2 | 80.2 |
| Watch | 5,317,560.6 | 17.9 | 5,842,719.4 | 19.8 | 6,651,496.5 | 19.8 |
| Reserve | 450.0 | 0.001 | 6095.0 | 0.0 | 7300.0 | 0.02 |
| Other | 3629.0 | 0.012 | 3825.0 | 0.01 | ||
| Joint Medical Stores | ||||||
| Access | 2,495,735.4 | 61.8 | 2,603,915.4 | 65.6 | 2,681,477.7 | 65.9 |
| Watch | 1,543,095.2 | 38.2 | 1,363,196.3 | 34.4 | 1,387,103.6 | 34.1 |
| Level of Care | Average DDD/100 Patient Days | Average DDD/100 Admissions | ||||
|---|---|---|---|---|---|---|
| 2017 | 2018 | 2019 | 2017 | 2018 | 2019 | |
| National Referral Hospital | 99.9 | 113.3 | 191.1 | 1259.2 | 1417.9 | 2079.9 |
| Regional Referral Hospital | 570.6 | 559.3 | 523.9 | 2302.3 | 2435.4 | 2004.9 |
| General Hospital | 978.2 | 1124.4 | 1080.9 | 3090.9 | 3340.1 | 3101.7 |
| Health Centre Level IV | 3536.9 | 2852.5 | 4044.5 | 4382.6 | 4305.8 | 3776.7 |
| ATC * | Level: Pharmacological Class | Percentage of DDD | ||
|---|---|---|---|---|
| 2017 | 2018 | 2019 | ||
| National Medical Stores | ||||
| J01A | Tetracyclines | 14.7 | 12.1 | 9.8 |
| J01B | Amphenicols | 0.3 | 0.2 | 0.1 |
| J01C | Beta-Lactam antibacterials, Penicillins | 40.8 | 41.6 | 36.2 |
| J01D | Other beta-lactam antibacterials | 3.7 | 4.8 | 4.0 |
| J01E | Sulfonamides and trimethoprim | 9.0 | 6.3 | 19.9 |
| J01F | Macrolides | 2.6 | 3.2 | 7.8 |
| J01G | Aminoglycosides | 1.6 | 1.4 | 0.1 |
| J01M | Quinolone antibacterials | 11.5 | 11.7 | 7.9 |
| J01X | Other antibacterials | 15.8 | 18.6 | 14.1 |
| Joint Medical Store | ||||
| J01A | Tetracyclines | 10.2 | 11.6 | 9.8 |
| J01B | Amphenicols | 0.3 | 0.3 | 0.4 |
| J01C | Beta-Lactam antibacterials, Penicillins | 33.1 | 34.4 | 37.1 |
| J01D | Other beta-lactam antibacterials | 11.6 | 11.5 | 11.4 |
| J01E | Sulfonamides and trimethoprim | 1.5 | 1.6 | 1.7 |
| J01F | Macrolides | 14.8 | 12.8 | 12.7 |
| J01G | Aminoglycosides | 3.1 | 3.0 | 2.3 |
| J01M | Quinolone antibacterials | 13.1 | 11.4 | 11.0 |
| J01X | Other antibacterials | 11.9 | 18.6 | 13.1 |
| P01A | Antiprotozoals against amoebiasis | 0.3 | 0.3 | 0.3 |
| J01A | Tetracyclines | 10.2 | 11.6 | 9.8 |
| J01B | Amphenicols | 0.3 | 0.3 | 0.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Namugambe, J.S.; Delamou, A.; Moses, F.; Ali, E.; Hermans, V.; Takarinda, K.; Thekkur, P.; Nanyonga, S.M.; Koroma, Z.; Mwoga, J.N.; et al. National Antimicrobial Consumption: Analysis of Central Warehouses Supplies to In-Patient Care Health Facilities from 2017 to 2019 in Uganda. Trop. Med. Infect. Dis. 2021, 6, 83. https://doi.org/10.3390/tropicalmed6020083
Namugambe JS, Delamou A, Moses F, Ali E, Hermans V, Takarinda K, Thekkur P, Nanyonga SM, Koroma Z, Mwoga JN, et al. National Antimicrobial Consumption: Analysis of Central Warehouses Supplies to In-Patient Care Health Facilities from 2017 to 2019 in Uganda. Tropical Medicine and Infectious Disease. 2021; 6(2):83. https://doi.org/10.3390/tropicalmed6020083
Chicago/Turabian StyleNamugambe, Juliet Sanyu, Alexandre Delamou, Francis Moses, Engy Ali, Veerle Hermans, Kudakwashe Takarinda, Pruthu Thekkur, Stella Maris Nanyonga, Zikan Koroma, Joseph Ngobi Mwoga, and et al. 2021. "National Antimicrobial Consumption: Analysis of Central Warehouses Supplies to In-Patient Care Health Facilities from 2017 to 2019 in Uganda" Tropical Medicine and Infectious Disease 6, no. 2: 83. https://doi.org/10.3390/tropicalmed6020083
APA StyleNamugambe, J. S., Delamou, A., Moses, F., Ali, E., Hermans, V., Takarinda, K., Thekkur, P., Nanyonga, S. M., Koroma, Z., Mwoga, J. N., Akello, H., Imi, M., & Kitutu, F. E. (2021). National Antimicrobial Consumption: Analysis of Central Warehouses Supplies to In-Patient Care Health Facilities from 2017 to 2019 in Uganda. Tropical Medicine and Infectious Disease, 6(2), 83. https://doi.org/10.3390/tropicalmed6020083






