Comparative Yield of Tuberculosis during Active Case Finding Using GeneXpert or Smear Microscopy for Diagnostic Testing in Nepal: A Cross-Sectional Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Case-Finding Strategies
2.1.1. Close-Contact Tracing
2.1.2. TB Camps
2.1.3. Screening at Hospital OPD
Ethical Approval
2.2. Statistical Analysis
2.2.1. Screening and Diagnosis of TB Cases
2.2.2. Comparison of Yield from GeneXpert vs. Smear Microscopy
2.2.3. Additionality in District Level TB Case Notification Rates
3. Results
3.1. Screening and Diagnosis of TB Cases
3.2. Comparison of Yield from GeneXpert vs. Smear Microscopy
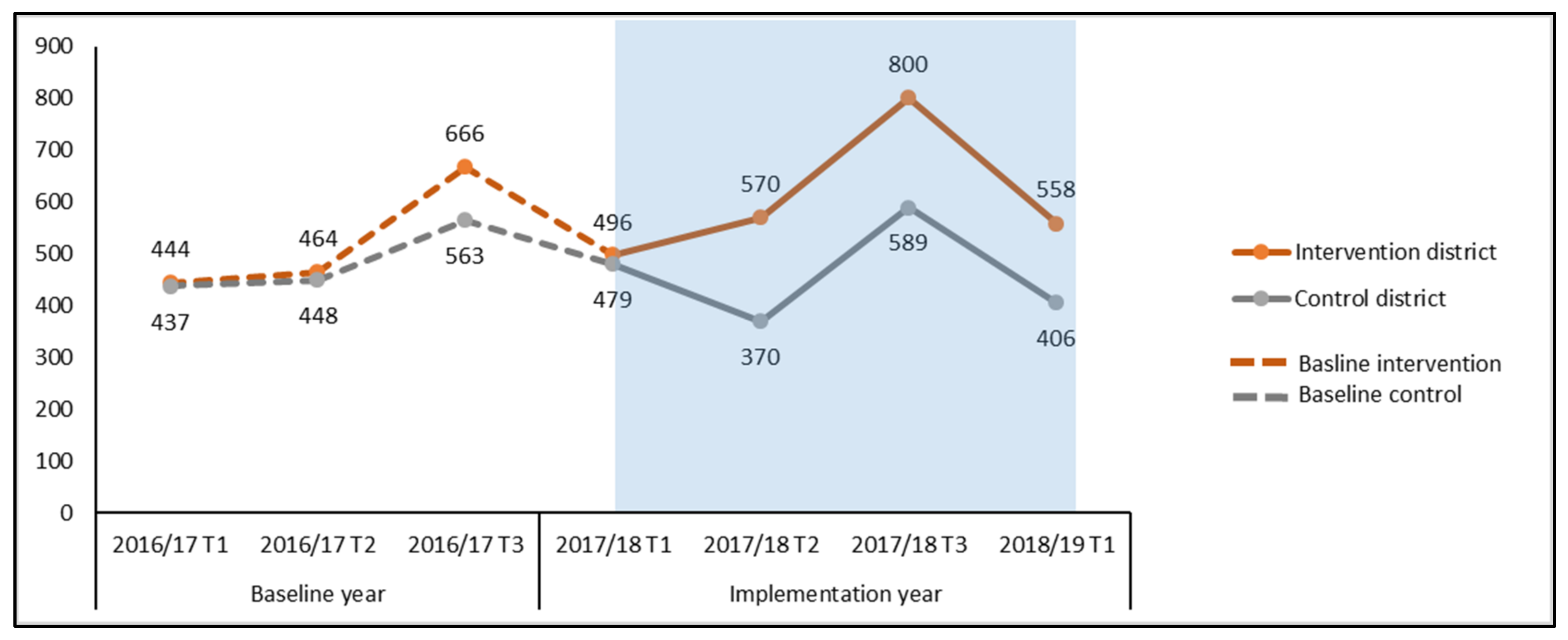
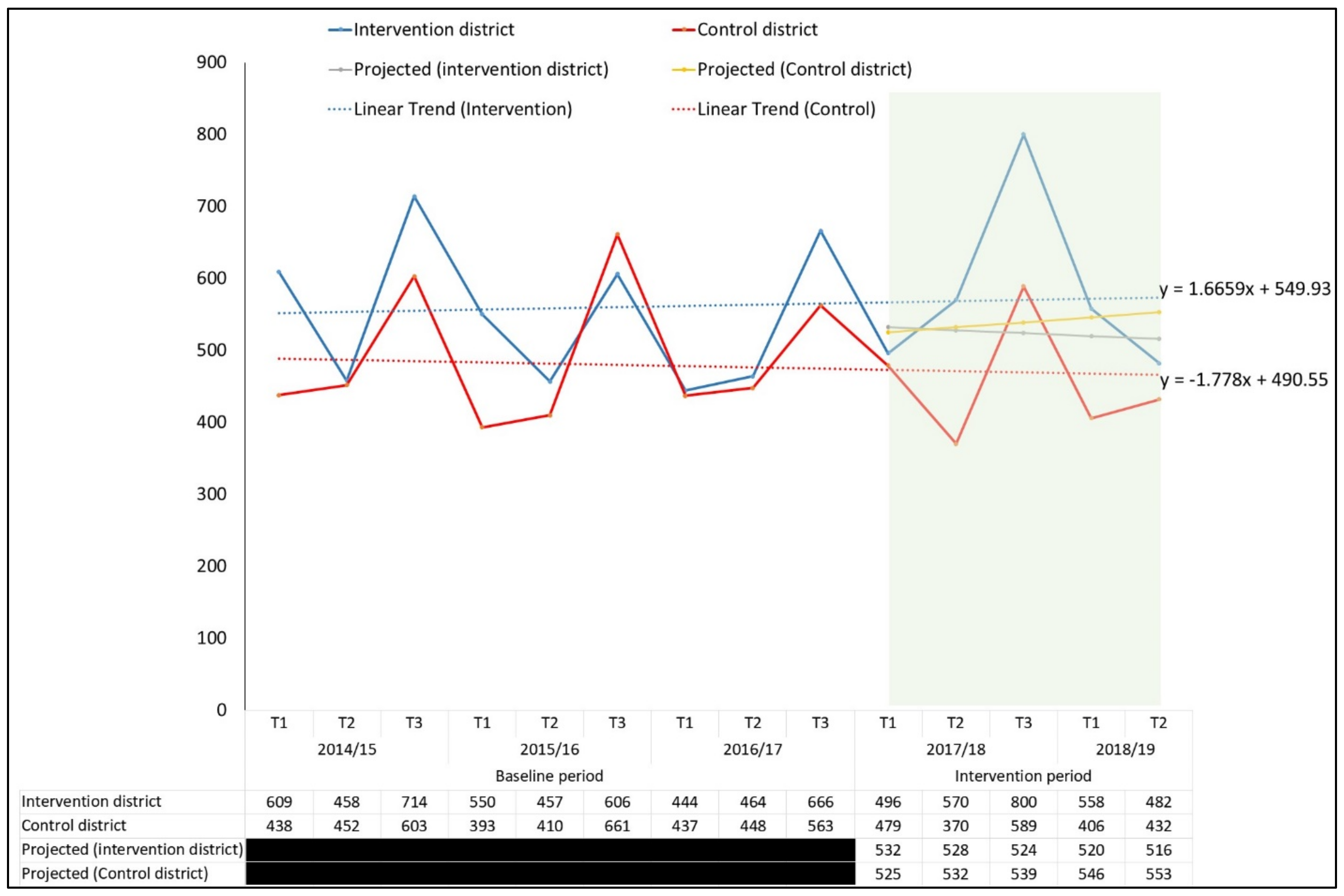
3.3. Additionality in Case Notification
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization (WHO). Global Tuberculosis Report [Internet]. Geneva. 2020. Available online: https://apps.who.int/iris/bitstream/handle/10665/336069/9789240013131-eng.pdf?ua=1 (accessed on 20 October 2020).
- Stop TB Partnership. The Global Plan to End TB 2016–2020: The Paradigm Shift [Internet]. 2015. Available online: http://www.stoptb.org/assets/documents/global/plan/GlobalPlanToEndTB_TheParadigmShift_2016-2020_StopTBPartnership.pdf (accessed on 28 January 2020).
- World Health Organization (WHO). Systematic Screening for Active Tuberculosis: An Operational Guide [Internet]. 2015. Available online: https://apps.who.int/iris/bitstream/handle/10665/181164/9789241549172_eng.pdf;jsessionid=1FD961395F5ADC73389399DE78D91BD9?sequence=1 (accessed on 6 August 2020).
- Yuen, C.M.; Amanullah, F.; Dharmadhikari, A.; Nardell, E.A.; Seddon, J.A.; Vasilyeva, I.; Zhao, Y.; Keshavjee, S.; Becerra, M.C. Turning off the tap: Stopping tuberculosis transmission through active case-finding and prompt effective treatment. Lancet 2015, 386, 2334–2343. [Google Scholar] [CrossRef]
- World Health Organization. Report of the 16th Meeting of the Strategic and Technical Advisory Group for Tuberculosis [Internet]. Geneva. 2016. Available online: https://www.challengetb.org/reportfiles/stag_tb_report_2016.pdf (accessed on 24 September 2020).
- National Tuberculosis Control Center. Nepal National TB Prevalence Survey Brief [Internet]. 2020. Available online: https://nepalntp.gov.np/wp-content/uploads/2020/03/NEPAL-NATIONAL-TB-PREVALENCE-SURVEY-BRIEF-March-24-2020.pdf (accessed on 19 April 2020).
- National Tuberculosis Control Center. Annual Report 2075/76 (2018/19) [Internet]. 2019. Available online: https://nepalntp.gov.np/wp-content/uploads/2020/04/NTP-Annual-Report-2075-76-2018-19.pdf (accessed on 6 August 2020).
- Laohasiriwong, W.; Mahato, R.K.; Koju, R.; Vaeteewootacharn, K. Delay for First Consultation and Its Associated Factors among New Pulmonary Tuberculosis Patients of Central Nepal. Tuberc. Res. Treat. 2016, 2016, 4583871. [Google Scholar] [CrossRef] [PubMed]
- Mahato, R.K.; Laohasiriwong, W.; Vaeteewootacharn, K. Major Delays in the Diagnosis and Management of Tuberculosis Patients in Nepal. J. Clin. Diagnostic. Res. 2015, 9, 5–9. [Google Scholar] [CrossRef] [PubMed]
- National Tuberculosis Center. National Strategic Plan for Tuberculosis Prevention, Care and Control [Internet]. Kathmandu. 2016. Available online: https://nepalntp.gov.np/wp-content/uploads/2018/01/NSP-report-english-revised.pdf (accessed on 28 January 2020).
- National Tuberculosis Centre. Laboratory Manual for Sputum Microscopy; National Tuberculosis Centre: Madhyapur Thimi, Nepal, 2017. [Google Scholar]
- Cepheid. Release or Treat in under Two Hours. Xpert® MTB/RIF. Accurate Detection of MTBC and Rifampin Resistance Mutations. 2015. Available online: https://www.cepheid.com/PackageInsertFiles/Xpert-MTB-RIF-ENGLISH-Package-Insert-301-1404-Rev-F.pdf (accessed on 10 February 2021).
- World Health Organization. Systematic Screening for Active Tuberculosis: Principles and Recommendations [Internet]. 2013. Available online: https://www.who.int/tb/publications/Final_TB_Screening_guidelines.pdf (accessed on 1 February 2021).
- National Tuberculosis Control Center. National Tuberculosis Management Guidelines 2019 [Internet]. 2019. Available online: http://nepalntp.gov.np/wp-content/uploads/2019/10/National-Tuberculosis-Management-Guidelines-2019_Nepal.pdf (accessed on 16 August 2020).
- Creswell, J.; Sahu, S.; Blok, L.; Bakker, M.I.; Stevens, R.; Ditiu, L. A multi-site evaluation of innovative approaches to increase tuberculosis case notification: Summary results. PLoS ONE 2014, 9, e94465. [Google Scholar]
- The Britain Nepal Medical Trust. TB REACH Wave 2 Report; The Britain Nepal Medical Trust: Kathmandu, Nepal, 2014. [Google Scholar]
- World Health Organisation. Chest Radiography in Tuberculosis. Summary of Current WHO Recommendations and Guidance on Programmatic Approaches. WHO Libr Cat Data [Internet]. 2016. Available online: https://apps.who.int/iris/bitstream/handle/10665/252424/9789241511506-eng.pdf?sequence=1 (accessed on 24 September 2020).
- Qin, Z.Z.; Sander, M.S.; Rai, B.; Titahong, C.N.; Sudrungrot, S.; Laah, S.N.; Adhikari, L.M.; Carter, E.J.; Puri, L.; Codlin, A.J.; et al. Using artificial intelligence to read chest radiographs for tuberculosis detection: A multi-site evaluation of the diagnostic accuracy of three deep learning systems. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Gurung, S.C.; Dixit, K.; Rai, B.; Caws, M.; Paudel, P.R.; Dhital, R.; Acharya, A.S.; Budhathoki, G.; Malla, D.; Levy, J.W.; et al. The role of active case finding in reducing patient incurred catastrophic costs for tuberculosis in Nepal. Infect. Dis. Poverty 2019, 8, 99. [Google Scholar] [CrossRef] [PubMed]
- Gurung, S.C.; Rai, B.; Dixit, K.; Worrall, E.; Paudel, P.R.; Dhital, R.; Sah, M.K.; Pandit, R.N.; Prasad Aryal, T.; Majhi, G.; et al. Active case finding reduces household costs for people affected by tuberculosis: The IMPACT TB longitudinal costing survey in Nepal. Health Policy Plan. 2020. [Google Scholar] [CrossRef] [PubMed]
- Kranzer, K.; Afnan-Holmes, H.; Tomlin, K.; Golub, J.E.; Shapiro, A.E.; Schaap, A.; Corbett, E.L.; Lönnroth, K.; Glynn, J.R. The benefits to communities and individuals of screening for active tuberculosis disease: A systematic review. Int. J. Tuberc. Lung Dis. 2013, 17, 432–446. [Google Scholar] [CrossRef] [PubMed]
- Scott, L.E.; McCarthy, K.; Gous, N.; Nduna, M.; Van Rie, A.; Sanne, I.; Venter, W.F.; Duse, A.; Stevens, W. Comparison of Xpert MTB/RIF with other nucleic acid technologies for diagnosing pulmonary tuberculosis in a high HIV prevalence setting: A prospective study. PLoS Med. 2011, 8, 8–11. [Google Scholar] [CrossRef] [PubMed]
- Trébucq, A.; Enarson, D.A.; Chiang, C.Y.; Van Deun, A.; Harries, A.D.; Boillot, F.; Detjen, A.; Fujiwara, P.I.; Graham, S.M.; Monedero, I.; et al. Xpert® MTB/RIF for national tuberculosis programmes in low-income countries: When, where and how? Int. J. Tuberc. Lung Dis. 2011, 15, 1567–1571. [Google Scholar] [CrossRef] [PubMed]
- Biermann, O.; Lönnroth, K.; Caws, M.; Viney, K. Factors influencing active tuberculosis case-finding policy development and implementation: A scoping review. BMJ Open 2019, 9, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Biermann, O.; Atkins, S.; Lönnroth, K.; Caws, M.; Viney, K. “Power plays plus push”: Experts’ insights into the development and implementation of active tuberculosis case-finding policies globally, a qualitative study. BMJ Open 2020, 10, e036285. [Google Scholar] [CrossRef] [PubMed]
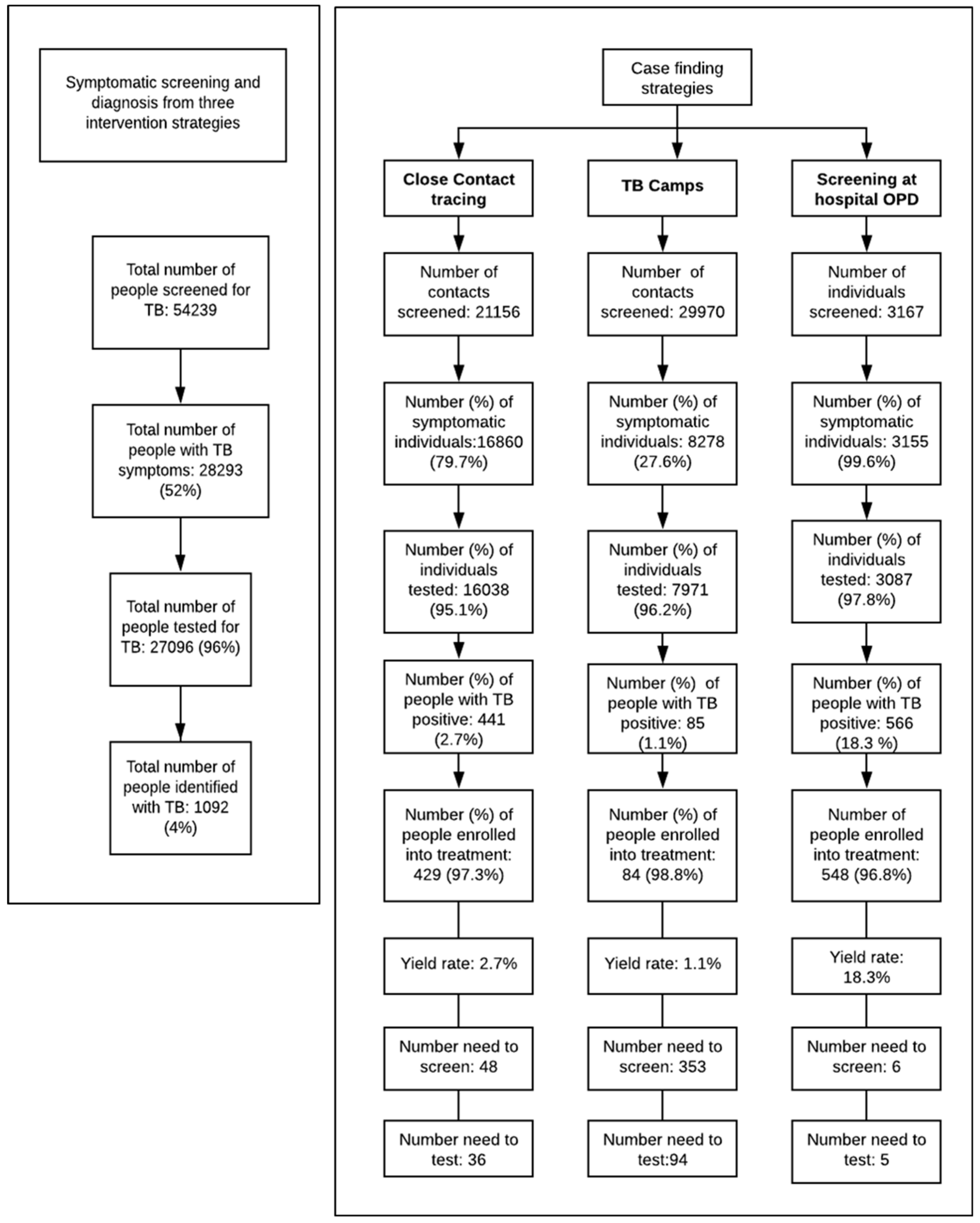
| Indicators | Districts Diagnosing TB with GeneXpert | Districts Diagnosing TB with Smear Microscopy | |
|---|---|---|---|
| Total number of people screened | 32,616 | 11,202 | |
| Total number of people with TB symptoms | 16,060 | 6,575 | |
| Total number of people tested for TB | 15,637 | 6,309 | |
| Total number of people diagnosed with TB positive | 859 | 120 | |
| Yield rate (%) | Contact Tracing | 3.2 (273/8567) | 2.4 (103/4246) |
| TB Camps | 1.4 (58/4106) | 0.82 (17/2063) | |
| Three interventions combined | 5.5 (859/15,637) | 2 (120/6309) | |
| Numbers needed to screen (NNS) | 38 | 93 | |
| Numbers needed to test (NNT) | 18 | 53 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gurung, S.C.; Dixit, K.; Rai, B.; Dhital, R.; Paudel, P.R.; Acharya, S.; Budhathoki, G.; Malla, D.; Levy, J.W.; Lönnroth, K.; et al. Comparative Yield of Tuberculosis during Active Case Finding Using GeneXpert or Smear Microscopy for Diagnostic Testing in Nepal: A Cross-Sectional Study. Trop. Med. Infect. Dis. 2021, 6, 50. https://doi.org/10.3390/tropicalmed6020050
Gurung SC, Dixit K, Rai B, Dhital R, Paudel PR, Acharya S, Budhathoki G, Malla D, Levy JW, Lönnroth K, et al. Comparative Yield of Tuberculosis during Active Case Finding Using GeneXpert or Smear Microscopy for Diagnostic Testing in Nepal: A Cross-Sectional Study. Tropical Medicine and Infectious Disease. 2021; 6(2):50. https://doi.org/10.3390/tropicalmed6020050
Chicago/Turabian StyleGurung, Suman Chandra, Kritika Dixit, Bhola Rai, Raghu Dhital, Puskar Raj Paudel, Shraddha Acharya, Gangaram Budhathoki, Deepak Malla, Jens W. Levy, Knut Lönnroth, and et al. 2021. "Comparative Yield of Tuberculosis during Active Case Finding Using GeneXpert or Smear Microscopy for Diagnostic Testing in Nepal: A Cross-Sectional Study" Tropical Medicine and Infectious Disease 6, no. 2: 50. https://doi.org/10.3390/tropicalmed6020050
APA StyleGurung, S. C., Dixit, K., Rai, B., Dhital, R., Paudel, P. R., Acharya, S., Budhathoki, G., Malla, D., Levy, J. W., Lönnroth, K., Ramsay, A., Basnyat, B., Thapa, A., Mishra, G., Subedi, B., Shah, M. K., Shrestha, A., & Caws, M. (2021). Comparative Yield of Tuberculosis during Active Case Finding Using GeneXpert or Smear Microscopy for Diagnostic Testing in Nepal: A Cross-Sectional Study. Tropical Medicine and Infectious Disease, 6(2), 50. https://doi.org/10.3390/tropicalmed6020050









