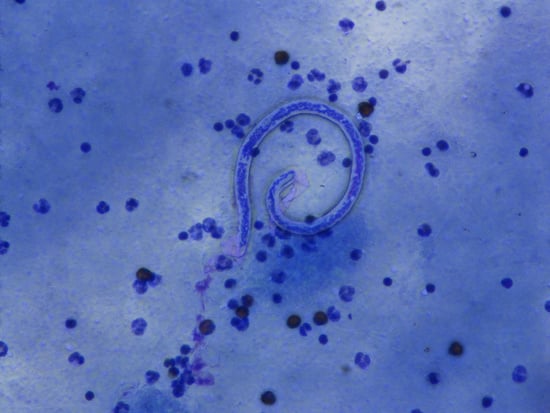
Triple-Drug Treatment Is Effective for Lymphatic Filariasis Microfilaria Clearance in Samoa
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Setting
2.2. Study Design
2.3. Ethics Approval and Consent
2.4. Treatment of Mf-Positive Persons
2.5. Blood Samples and Laboratory Methods
- Venous blood samples in heparin or EDTA were used on the same day of collection or (rarely) stored at 4 °C until the next morning. They were used for:
- (a)
- membrane filters;
- (b)
- three-line thick blood films on slides (60 uL);
- (c)
- collection of plasma (for potential pharmacokinetic assays) by centrifuging the remaining sample at >1000× g for 10 min.
- Fingerprick blood samples were used on the day of collection or the next day after storage at 4 °C to:
- (a)
- test for circulating filarial antigen (Ag) using Alere Filariasis Test Strips (FTS);
- (b)
- prepare dried blood spots on TropBio filter papers (Cellabs) for future testing for anti-filarial antibodies (Ab);
- (c)
- prepare three-line thick blood films on slides (60 uL per slide).
2.6. Procedure for Membrane Filters
2.7. Filariasis Test Strips
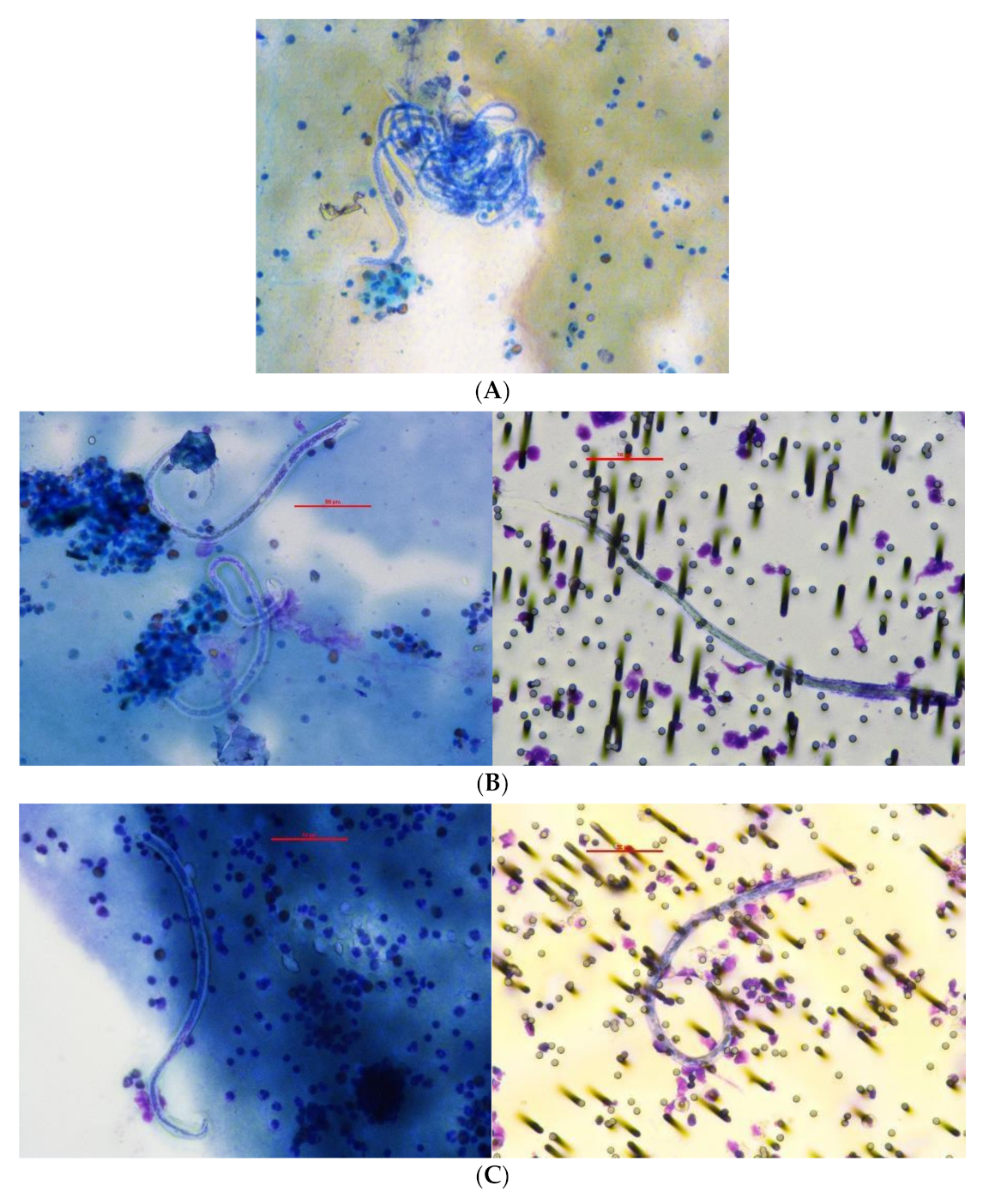
2.8. Blood Slides
2.9. Staining of Filters and Slides
2.10. Statistical Analysis
3. Results
3.1. Study Participants
3.2. MDA Participation in 2018
3.3. Antigen and Mf Results at Baseline in 2019
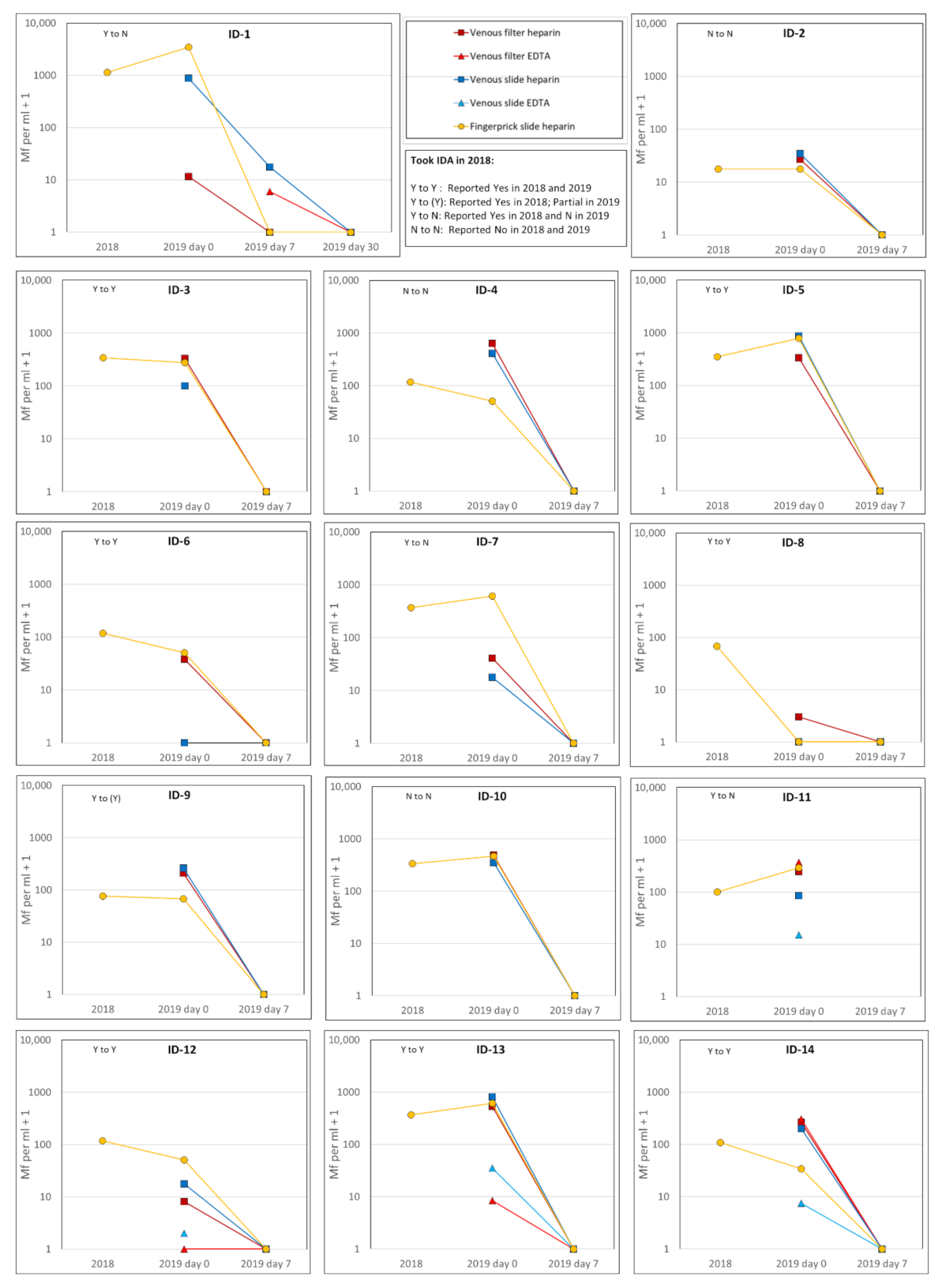
3.4. Mf Counts in Study Participants Before and After Treatment
- ID-6 who was Mf-negative on the venous heparin slide but positive on all other sample and specimen types available;
- ID-8 who was Mf-positive with 2 Mf/mL on the venous filter but negative on all other sample and specimen types available.
3.5. Pharmacokinetics
3.6. Comparions of Mf Density in Different Sample and Specimen Types
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ramaiah, K.D.; Ottesen, E.A. Progress and Impact of 13 Years of the Global Programme to Eliminate Lymphatic Filariasis on Reducing the Burden of Filarial Disease. PLoS Negl. Trop. Dis. 2014, 8. [Google Scholar] [CrossRef] [PubMed]
- Local Burden of Disease Neglected Tropical Diseases Collaborators. The global distribution of lymphatic filariasis, 2000–2018: A geospatial analysis. Lancet Glob. Health 2020, 8, e1186–e1194. [Google Scholar] [CrossRef]
- Malecela, M.; Gyapong, J.; Ramaiah, K.D.; Molyneux, D. Two decades of public health achievements in lymphatic filariasis (2000–2020): Reflections, progress and future challenges. Int. Health 2020, 13 (Suppl. 1), S1–S2. [Google Scholar] [CrossRef]
- Yajima, A.; Ichimori, K. Progress in the elimination of lymphatic filariasis in the Western Pacific Region: Successes and challenges. Int. Health 2021, 13 (Suppl. 1), S10–S16. [Google Scholar] [CrossRef]
- WHO. Global programme to eliminate lymphatic filariasis: Progress report, 2019. Wkly. Epidemiol. Rec. 2020, 43, 509–524. [Google Scholar]
- Lau, C.L.; Won, K.Y.; Becker, L.; Soares Magalhaes, R.J.; Fuimaono, S.; Melrose, W.; Lammie, P.J.; Graves, P.M. Seroprevalence and spatial epidemiology of Lymphatic Filariasis in American Samoa after successful mass drug administration. PLoS Negl. Trop. Dis. 2014, 8, e3297. [Google Scholar] [CrossRef]
- Lau, C.L.; Sheridan, S.; Ryan, S.; Roineau, M.; Andreosso, A.; Fuimaono, S.; Tufa, J.; Graves, P.M. Detecting and confirming residual hotspots of lymphatic filariasis transmission in American Samoa 8 years after stopping mass drug administration. PLoS Negl. Trop. Dis. 2017, 11, e0005914. [Google Scholar] [CrossRef]
- Sheel, M.; Sheridan, S.; Gass, K.; Won, K.; Fuimaono, S.; Kirk, M.; Gonzales, A.; Hedtke, S.M.; Graves, P.M.; Lau, C.L. Identifying residual transmission of lymphatic filariasis after mass drug administration: Comparing school-based versus community-based surveillance—American Samoa, 2016. PLoS Negl. Trop. Dis. 2018, 12, e0006583. [Google Scholar] [CrossRef]
- Lau, C.L.; Meder, K.; Mayfield, H.J.; Kearns, T.; McPherson, B.; Naseri, T.; Thomsen, R.; Hedtke, S.M.; Sheridan, S.; Gass, K.; et al. Lymphatic filariasis epidemiology in Samoa in 2018: Geographic clustering and higher antigen prevalence in older age groups. PLoS Negl. Trop. Dis. 2020, 14, e0008927. [Google Scholar] [CrossRef]
- WHO. Guideline—Alternative Mass Drug Administration Regimens to Eliminate Lymphatic Filariasis; World Health Organization/Department of Control of Neglected Tropical Diseases: Geneva, Switzerland, 2017. [Google Scholar]
- Thomsen, E.K.; Sanuku, N.; Baea, M.; Satofan, S.; Maki, E.; Lombore, B.; Schmidt, M.S.; Siba, P.M.; Weil, G.J.; Kazura, J.W.; et al. Efficacy, Safety, and Pharmacokinetics of Coadministered Diethylcarbamazine, Albendazole, and Ivermectin for Treatment of Bancroftian Filariasis. Clin. Infect. Dis. 2016, 62, 334–341. [Google Scholar] [CrossRef]
- Ottesen, E.A.; Horton, J. Setting the stage for a Global Programme to Eliminate Lymphatic Filariasis: The first 125 years (1875–2000). Int. Health 2020, 13 (Suppl. 1), S3–S9. [Google Scholar] [CrossRef]
- Weil, G.J.; Jacobson, J.A.; King, J.D. A triple-drug treatment regimen to accelerate elimination of lymphatic filariasis: From conception to delivery. Int. Health 2020, 13 (Suppl. 1), S60–S64. [Google Scholar] [CrossRef]
- Ichimori, K.; Tupuimalagi-Toelupe, P.; Iosia, V.T.; Graves, P.M. Wuchereria bancrofti filariasis control in Samoa before PacELF (Pacific Programme to Eliminate Lymphatic Filariasis). Trop. Med. Health 2007, 35, 261–269. [Google Scholar] [CrossRef][Green Version]
- Ichimori, K.; Graves, P.M. Overview of PacELF-the Pacific Programme for the Elimination of Lymphatic Filariasis. Trop. Med. Health 2017, 45, 34. [Google Scholar] [CrossRef]
- World Health Organization—Regional Office for the Western Pacific. The PacELF Way: Towards the Elimination of Lymphatic Filariasis from the Pacific, 1999–2005; WHO Western Pacific Region: Manila, Philippines, 2006. [Google Scholar]
- Willis, G.A.; Mayfield, H.J.; Kearns, T.; Naseri, T.; Thomsen, R.; Gass, K.; Sheridan, S.; Graves, P.M.; Lau, C.L. A community survey of coverage and adverse events following country-wide triple-drug mass drug administration for lymphatic filariasis elimination, Samoa 2018. PLoS Negl. Trop. Dis. 2020, 14, e0008854. [Google Scholar] [CrossRef]
- Samoa Bureau of Statistics. Census—Population and Demography. 2016. Available online: https://sbs.gov.ws/populationanddemography (accessed on 1 February 2021).
- Wylie, J.P. Detection of microfilariae by a filter technique. J. Am. Vet. Med. Assoc. 1970, 156, 1403–1405. [Google Scholar]
- Chularerk, P.; Desowitz, R.S. A simplified membrane filtration technique for the diagnosis of microfilaremia. J. Parasitol. 1970, 56, 623–624. [Google Scholar] [CrossRef]
- Dickerson, J.W.; Eberhard, M.L.; Lammie, P.J. A technique for microfilarial detection in preserved blood using nuclepore filters. J. Parasitol. 1990, 76, 829–833. [Google Scholar] [CrossRef]
- Desowitz, R.S.; Hitchcock, J.C. Hyperendemic bancroftian filariasis in the Kingdom of Tonga: The application of the membrane filter concentration technique to an age-stratified blood survey. Am. J. Trop. Med. Hyg. 1974, 23, 877–879. [Google Scholar] [CrossRef]
- Desowitz, R.S.; Southgate, B.A. Studies of filariasis in the Pacific. 2. The persistence of microfilaraemia in diethylcarbamazine treated populations of Fiji and Western Samoa: Diagnostic application of the membrane-filtration technique. Southeast Asian J. Trop. Med. Public Health 1973, 4, 179–183. [Google Scholar]
- Desowitz, R.S.; Jenkins, C.; Anian, G. Bancroftian Filariasis in an Isolated Hunter Gatherer Shifting Horticulturist Group in Papua-New-Guinea. Bull. World Health Organ. 1993, 71, 55–58. [Google Scholar]
- Vinkeles Melchers, N.V.S.; Coffeng, L.E.; de Vlas, S.J.; Stolk, W.A. Standardisation of lymphatic filariasis microfilaraemia prevalence estimates based on different diagnostic methods: A systematic review and meta-analysis. Parasit. Vectors. 2020, 13, 302. [Google Scholar] [CrossRef]
- Eberhard, M.L.; Roberts, I.M.; Lammie, P.J.; Lowrie, R.C., Jr. Comparative densities of Wuchereria bancrofti microfilaria in paired samples of capillary and venous blood. Trop. Med. Parasitol. 1988, 39, 295–298. [Google Scholar]
- Dickerson, J.W.; Eberhard, M.L.; Lammie, P.J.; Roberts, J.M. Further evidence of a skewed distribution of microfilariae in capillary blood. Trop. Med. Parasitol. 1989, 40, 472–473. [Google Scholar]
- Yoeli, M. Observations of agglutination and thigmotaxis of microfilariae in bancroftian filariasis. Trans. R. Soc. Trop. Med. Hyg. 1957, 51, 132–136. [Google Scholar] [CrossRef]
| Weight Range (kg) | Number of Ivermectin Tablets (3 mg) | Number of DEC Tablets (100 mg) | Number of Albendazole Tablets (400 mg) | Total Number of Tablets |
|---|---|---|---|---|
| <15 kg (or 2–4 years old) | 0 | 1 | 1 | 2 |
| 15–23 kg | 1 | 1 | 1 | 3 |
| 24–38 kg | 2 | 2 | 1 | 5 |
| 39–53 kg | 3 | 3 | 1 | 7 |
| 54–68 kg | 4 | 4 | 1 | 9 |
| 69–83 kg | 5 | 5 | 1 | 11 |
| 84–98 kg | 6 | 6 | 1 | 13 |
| 99–124 kg | 7 | 7 | 1 | 15 |
| >124 kg | 8 | 8 | 1 | 17 |
| Household Visit | Timing | Activities | Venous Blood Samples | Fingeprick Blood Samples |
|---|---|---|---|---|
| 1 | Day 0 |
| Heparin & EDTA * samples for:
| Heparin samples for:
|
| 2 | Day 0, 3 h post- treatment |
| Heparin & EDTA samples for:
| Not collected |
| 3 | Day 7 |
| Heparin & EDTA samples for:
| Heparin samples for:
|
| 4 | Day 30 (only visited if Mf-positive on Day 7) |
| Heparin & EDTA samples for:
| Heparin samples for:
|
| Participant ID Number | Age (Years) | Gender | Reported (in 2018) Taking 2018 MDA | Reported (in 2019) Taking 2018 MDA |
|---|---|---|---|---|
| 1 | 59 | M | Yes | No |
| 2 | 46 | F | No | No |
| 3 | 50 | F | Yes | Yes |
| 4 | 44 | F | No | No |
| 5 | 60 | M | Yes | Yes |
| 6 | 52 | F | Yes | Yes |
| 7 | 20 | F | Yes | No |
| 8 | 5 | F | Yes | Yes |
| 9 | 44 | M | Yes | Incomplete 1 |
| 10 | 64 | F | No | No |
| 11 | 34 | F | Yes | No |
| 12 | 74 | M | Yes | Yes |
| 13 | 58 | M | Yes | Yes |
| 14 | 50 | M | Yes | Yes |
| No blood samples: interview only or missing in 2019 | ||||
| 15 | 27 | M | Yes | Unknown |
| 16 | 73 | M | Yes | No |
| 17 | 41 | F | No | No |
| 18 | 31 | F | Yes | No |
| MDA Participation in 2018 | ||||||
|---|---|---|---|---|---|---|
| Group | Status | n | Reported in 2018 | Reported in 2019 | ||
| Mf-positive participants identified in 2018 | Ag pos + Mf pos | 14 | Yes | 11 | Yes * | 8 |
| No | 3 | No | 6 | |||
| Family members | Ag pos + Mf neg | 5 | Yes | 4 | Yes | 4 |
| No | 1 | No | 1 | |||
| Ag neg + Mf neg | 22 | Yes | 20 | Yes | 20 | |
| No | 2 | No | 2 | |||
| All family members | 27 | Yes | 24 | Yes | 24 | |
| No | 3 | No | 3 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Graves, P.M.; Sheridan, S.; Scott, J.; Amosa-Lei Sam, F.; Naseri, T.; Thomsen, R.; King, C.L.; Lau, C.L. Triple-Drug Treatment Is Effective for Lymphatic Filariasis Microfilaria Clearance in Samoa. Trop. Med. Infect. Dis. 2021, 6, 44. https://doi.org/10.3390/tropicalmed6020044
Graves PM, Sheridan S, Scott J, Amosa-Lei Sam F, Naseri T, Thomsen R, King CL, Lau CL. Triple-Drug Treatment Is Effective for Lymphatic Filariasis Microfilaria Clearance in Samoa. Tropical Medicine and Infectious Disease. 2021; 6(2):44. https://doi.org/10.3390/tropicalmed6020044
Chicago/Turabian StyleGraves, Patricia M., Sarah Sheridan, Jessica Scott, Filipina Amosa-Lei Sam, Take Naseri, Robert Thomsen, Christopher L. King, and Colleen L. Lau. 2021. "Triple-Drug Treatment Is Effective for Lymphatic Filariasis Microfilaria Clearance in Samoa" Tropical Medicine and Infectious Disease 6, no. 2: 44. https://doi.org/10.3390/tropicalmed6020044
APA StyleGraves, P. M., Sheridan, S., Scott, J., Amosa-Lei Sam, F., Naseri, T., Thomsen, R., King, C. L., & Lau, C. L. (2021). Triple-Drug Treatment Is Effective for Lymphatic Filariasis Microfilaria Clearance in Samoa. Tropical Medicine and Infectious Disease, 6(2), 44. https://doi.org/10.3390/tropicalmed6020044