Relative Frequency of Blastocystis Subtypes 1, 2, and 3 in Urban and Periurban Human Populations of Arequipa, Peru
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Statement
2.2. Study Site
2.3. Study Population
2.4. Detection of Blastocystis
2.5. DNA Extraction and PCR Amplification
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Stensvold, C.R.; Suresh, G.K.; Tan, K.S.; Thompson, R.A.; Traub, R.J.; Viscogliosi, E.; Yoshikawa, H.; Clark, C.G. Terminology for Blastocystis subtypes—A consensus. Trends Parasitol. 2007, 23, 93–96. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, C.V.; Barreto, M.M.; Andrade, R.D.J.; Sodré, F.; D’Avila-Levy, C.M.; Peralta, J.M.; Igreja, R.P.; De Macedo, H.W.; Santos, H.L.C. Intestinal parasite infections in a rural community of Rio de Janeiro (Brazil): Prevalence and genetic diversity of Blastocystis subtypes. PLoS ONE 2018, 3, e0193860. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, H.; Tokoro, M.; Nagamoto, T.; Arayama, S.; Asih, P.B.; Rozi, I.E.; Syafruddin, D. Molecular survey of Blastocystis sp. from humans and associated animals in an Indonesian community with poor hygiene. Parasitol. Int. 2016, 65, 780–784. [Google Scholar] [CrossRef] [PubMed]
- Stensvold, C.R.; Clark, C.G. Current status of Blastocystis: A personal view. Parasitol. Int. 2016, 65, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Khademvatan, S.; Masjedizadeh, R.; Yousefi-Razin, E.; Mahbodfar, H.; Rahim, F.; Yousefi, E.; Foroutan, M. PCR-based molecular characterization of Blastocystis hominis subtypes in southwest of Iran. J. Infect. Public Health 2018, 11, 43–47. [Google Scholar] [CrossRef]
- Alfellani, M.A.; Stensvold, C.R.; Vidal-Lapiedra, A.; Onuoha, E.S.U.; Fagbenro-Beyioku, A.F.; Clark, C.G. Variable geographic distribution of Blastocystis subtypes and its potential implications. Acta Trop. 2013, 126, 11–18. [Google Scholar] [CrossRef]
- Ramírez, J.D.; Sánchez, A.; Hernández, C.; Flórez, C.; Bernal, M.C.; Giraldo, J.C.; Reyes, P.; López, M.C.; García, L.; Cooper, P.J.; et al. Geographic distribution of human Blastocystis subtypes in South America. Infect. Genet. Evol. 2016, 41, 32–35. [Google Scholar] [CrossRef]
- Skotarczak, B. Genetic diversity and pathogenicity of Blastocystis. Ann. Agric. Environ. Med. 2018, 25, 411–416. [Google Scholar] [CrossRef]
- Kaneda, Y.; Horiki, N.; Cheng, X.J.; Fujita, Y.; Maruyama, M.; Tachibana, H. Ribodemes of Blastocystis hominis isolated in Japan. Am. J. Trop. Med. Hyg. 2001, 65, 393–396. [Google Scholar] [CrossRef]
- Ramírez, J.D.; Sánchez, L.V.; Bautista, D.C.; Corredor, A.F.; Flórez, A.C.; Stensvold, C.R. Blastocystis subtypes detected in humans and animals from Colombia. Infect. Genet. Evol. 2014, 22, 223–228. [Google Scholar] [CrossRef]
- Cifre, S.; Gozalbo, M.; Ortiz, V.; Soriano, J.M.; Merino-Torres, J.F.; Trelis, M. Blastocystis subtypes and their association with Irritable Bowel Syndrome. Med. Hypotheses 2018, 116, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Di Cristanziano, V.; D’alfonso, R.; Berrilli, F.; Sarfo, F.S.; Santoro, M.; Fabeni, L.; Knops, E.; Heger, E.; Kaiser, R.; Dompreh, A.; et al. Lower prevalence of Blastocystis sp. infections in HIV positive compared to HIV negative adults in Ghana. PLoS ONE 2019, 14, e0221968. [Google Scholar] [CrossRef]
- Andersen, L.O.; Bonde, I.; Nielsen, H.B.; Stensvold, C.R. A retrospective metagenomics approach to studyingBlastocystis. FEMS Microbiol. Ecol. 2015, 91, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Andersen, L.O.; Karim, A.B.; Roager, H.M.; Vigsnæs, L.K.; Krogfelt, K.A.; Licht, T.R.; Stensvold, C.R. Associations between common intestinal parasites and bacteria in humans as revealed by qPCR. Eur. J. Clin. Microbiol. Infect. Dis. 2016, 35, 1427–1431. [Google Scholar] [CrossRef]
- Del Coco, V.F.; Molina, N.B.; Basualdo, J.A.; Córdoba, M.A. Blastocystis spp.: Avances, controversias y desafíos futuros. Rev. Argent. Microbiol. 2017, 49, 110–118. [Google Scholar] [CrossRef]
- Peña, S.; Carrasco, G.; Rojas, P.; Castillo, D.; Ozaki, L.S.; Mercado, R. Determination of subtypes of Blastocystis sp. in Chilean patients with and without inflammatory bowel syndrome, A preliminary report. Parasite Epidemiol. Control. 2020, 8, e00125. [Google Scholar] [CrossRef]
- Jiménez, P.A.; Jaimes, J.E.; Ramírez, J.D. A summary of Blastocystis subtypes in North and South America. Parasites Vectors 2019, 12, 1–9. [Google Scholar] [CrossRef]
- Ramírez, J.D.; Flórez, C.; Olivera, M.; Bernal, M.C.; Giraldo, J.C. Blastocystis subtyping and its association with intestinal parasites in children from different geographical regions of Colombia. PLoS ONE 2017, 12, e0172586. [Google Scholar] [CrossRef]
- Ibáñez-Herrera, N.; Jara, C.C.; Guerra, M.A.; Díaz-Limay, E. Prevalencia del enteroparasitismo en escolares de comunidades nativas del Alto Marañón, Amazonas, Perú. Rev. Peru Med. Exp. Salud Publica 2004, 21, 126–133. [Google Scholar] [CrossRef]
- Sánchez, L.; Gallardo, J.; Jara, C. Prevalencia de Infección por Blastocystis y Protozoarios Intestinales en Niños de “Alto Trujillo”, La Libertad, Perú. Sciéndo 2011, 14, 36–45. [Google Scholar]
- Choi, B.; Kim, B. Prevalence and Risk Factors of Intestinal Parasite Infection among Schoolchildren in the Peripheral Highland Regions of Huanuco, Peru. Osong Public Health Res. Perspect. 2017, 8, 302–307. [Google Scholar] [CrossRef] [PubMed]
- Quispe-Juli, C.U.; Coila, Y.S.C.; Moreno-Loaiza, O. Elevada prevalencia de Blastocystis spp. en niños de una escuela periurbana. An. Fac. Med. 2016, 77, 393. [Google Scholar] [CrossRef]
- Casquina-Guere, L.; Martínez-Barrios, E. Prevalencia y epidemiología del parasitismo intestinal en escolares de nivel primario de Pucchún, Camaná, Arequipa, Perú. Neotrop. Helminthol. 2011, 5, 1–9. [Google Scholar]
- Instituto Nacional de Estadística e Informática—INEI. Censos Nacionales 2017: XII de Población, VII de Vivienda y III de Comunidades Indígenas. Lima—Perú. 2018. Available online: https://www.inei.gob.pe/media/MenuRecursivo/publicaciones_digitales/Est/Lib1551/ (accessed on 11 October 2020).
- Leclerc, A.; Kaminski, M.; Lang, T. Combler le fossé en une génération: Le rapport de l’OMS sur les déterminants sociaux de la santé. Rev. D’Épidémiol. Santé Publique 2009, 57, 227–230. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, R.S.S.; Ascuña-Durand, K.; Castillo-Neyra, R.; Vásquez-Huerta, V.; Martínez-Barrios, E.; Ballón-Echegaray, J. Socio-demographic determinants associated with Blastocystis infection in Arequipa, Peru. MedRxiv 2020. [Google Scholar] [CrossRef]
- Yoshikawa, H.; Iwamasa, A. Human Blastocystis subtyping with subtype-specific primers developed from unique sequences of the SSU rRNA gene. Parasitol. Int. 2016, 65, 785–791. [Google Scholar] [CrossRef]
- Minvielle, M.C.; Pezzani, B.C.; Cordoba, M.A.; De Luca, M.M.; Apezteguia, M.C.; Basualdo, J.A. Epidemiological survey of Giardia spp. and Blastocystis hominis in an Argentinian rural community. Korean J. Parasitol. 2004, 42, 121–127. [Google Scholar] [CrossRef][Green Version]
- Noradilah, S.A.; Moktar, N.; Anuar, T.S.; Lee, I.L.; Salleh, F.M.; Manap, S.N.A.A.; Mohtar, N.S.H.M.; Azrul, S.M.; Abdullah, W.O.; Nordin, A.; et al. Molecular epidemiology of blastocystosis in Malaysia: Does seasonal variation play an important role in determining the distribution and risk factors of Blastocystis subtype infections in the Aboriginal community? Parasites Vectors 2017, 10, 1–12. [Google Scholar] [CrossRef]
- R Project. What Is R? Available online: http://www.r-project.org/about.html (accessed on 25 November 2019).
- Villamizar, X.; Higuera, A.; Herrera, G.; Vasquez-A, L.R.; Buitron, L.; Muñoz, L.M.; Gonzalez-C, F.E.; Lopez, M.C.; Giraldo, J.C.; Ramírez, J.D. Molecular and descriptive epidemiology of intestinal protozoan parasites of children and their pets in Cauca, Colombia: A cross-sectional study. BMC Infect. Dis. 2019, 19, 190. [Google Scholar] [CrossRef]
- Dogan, N.; Aydin, M.; Tuzemen, N.U.; Dinleyici, E.C.; Oguz, I.; Dogruman-Al, F. Subtype distribution of Blastocystis spp. isolated from children in Eskisehir, Turkey. Parasitol. Int. 2017, 66, 948–951. [Google Scholar] [CrossRef]
- Rojas-Velázquez, L.; Moran, P.; Serrano-Vázquez, A.; Fernández, L.D.; Pérez-Juárez, H.; Poot-Hernandez, A.C.; Portillo-Bobadilla, T.; Gonzalez, E.; Hernandez, E.; Partida-Rodriguez, O.; et al. Genetic Diversity and Distribution ofBlastocystisSubtype 3 in Human Populations, with Special Reference to a Rural Population in Central Mexico. BioMed Res. Int. 2018, 2018, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Malheiros, A.F.; Stensvold, C.R.; Braga, G.B.; Shaw, J.J.; Clark, C.G. Molecular Characterization of Blastocystis Obtained from Members of the Indigenous Tapirapé Ethnic Group from the Brazilian Amazon Region, Brazil. Am. J. Trop. Med. Hyg. 2011, 85, 1050–1053. [Google Scholar] [CrossRef] [PubMed]
- Sanpool, O.; Laymanivong, S.; Thanchomnang, T.; Rodpai, R.; Sadaow, L.; Phosuk, I.; Maleewong, W.; Intapan, P.M. Subtype identification of human Blastocystis spp. isolated from Lao People’s Democratic Republic. Acta Trop. 2017, 168, 37–40. [Google Scholar] [CrossRef] [PubMed]
- Stensvold, C.R.; Christiansen, D.B.; Olsen, K.E.P.; Nielsen, H.V. Blastocystis sp. Subtype 4 is Common in Danish Blastocystis-Positive Patients Presenting with Acute Diarrhea. Am. J. Trop. Med. Hyg. 2011, 84, 883–885. [Google Scholar] [CrossRef]
- Forsell, J.; Granlund, M.; Stensvold, C.R.; Clark, G.C.; Evengård, B. Subtype analysis of Blastocystis isolates in Swedish patients. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 1689–1696. [Google Scholar] [CrossRef][Green Version]
- Castillo-Neyra, R.; Toledo, A.M.; Arevalo-Nieto, C.; Macdonald, H.; De la Puente-León, M.; Naquira-Velarde, C.; Paz-Soldan, V.A.; Buttenheim, A.M.; Levy, M.Z. Socio-spatial heterogeneity in participation in mass dog rabies vaccination campaigns, Arequipa, Peru. PLoS Negl. Trop. Dis. 2019, 13, e0007600. [Google Scholar] [CrossRef]
- Levy, M.Z.; Bowman, N.M.; Kawai, V.; Waller, L.A.; Del Carpio, J.G.C.; Benzaquen, E.C.; Gilman, R.H.; Bern, C. Periurban Trypanosoma cruzi–infected Triatoma infestans, Arequipa, Peru. Emerg. Infect. Dis. 2006, 12, 1345–1352. [Google Scholar] [CrossRef]
- Bowman, N.M.; Kawai, V.; Levy, M.Z.; Del Carpio, J.G.C.; Cabrera, L.; Delgado, F.; Malaga, F.; Benzaquen, E.C.; Pinedo, V.V.; Steurer, F.; et al. Chagas Disease Transmission in Periurban Communities of Arequipa, Peru. Clin. Infect. Dis. 2008, 46, 1822–1828. [Google Scholar] [CrossRef]
- Bayer, A.M.; Hunter, G.C.; Gilman, R.H.; Del Carpio, J.G.C.; Naquira, C.; Bern, C.; Levy, M.Z. Chagas Disease, Migration and Community Settlement Patterns in Arequipa, Peru. PLoS Negl. Trop. Dis. 2009, 3, e567. [Google Scholar] [CrossRef]
- Koltas, I.S.; Eroglu, F. Subtype analysis of Blastocystis isolates using SSU rRNA-DNA sequencing in rural and urban population in southern Turkey. Exp. Parasitol. 2016, 170, 247–251. [Google Scholar] [CrossRef]
- Li, L.-H.; Zhou, X.-N.; Du, Z.-W.; Wang, X.-Z.; Wang, L.-B.; Jiang, J.-Y.; Yoshikawa, H.; Steinmann, P.; Utzinger, J.; Wu, Z.; et al. Molecular epidemiology of human Blastocystis in a village in Yunnan province, China. Parasitol. Int. 2007, 56, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Paulos, S.; Köster, P.C.; De Lucio, A.; Hernández-De-Mingo, M.; Cardona, G.A.; Fernández-Crespo, J.C.; Stensvold, C.R.; Carmena, D. Occurrence and subtype distribution of Blastocystis sp. in humans, dogs and cats sharing household in northern Spain and assessment of zoonotic transmission risk. Zoonoses Public Health 2018, 65, 993–1002. [Google Scholar] [CrossRef] [PubMed]
- Banhos, E.F.; Da Rocha, J.A.M.; Pimentel, M.L.; Batista, E.T.M.; Silva, L.M. Prevalence and risk factors for intestinal parasite infections in schoolchildren, in the city of Santarém, Pará State, Brazil. ABCS Health Sci. 2017, 42, 137–142. [Google Scholar] [CrossRef]
- Deng, L.; Chai, Y.; Zhou, Z.; Liu, H.; Zhong, Z.; Hu, Y.; Fu, H.; Yue, C.; Peng, G. Epidemiology of Blastocystis sp. infection in China: A systematic review. Parasite 2019, 26, 41. [Google Scholar] [CrossRef] [PubMed]
- Lepczyńska, M.; Białkowska, J.; Dzika, E.; Piskorz-Ogórek, K.; Korycińska, J. Blastocystis: How do specific diets and human gut microbiota affect its development and pathogenicity? Eur. J. Clin. Microbiol. Infect. Dis. 2017, 36, 1531–1540. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Cordón, G.; Rosales, M.J.; Valdez, R.A.; Vargas-Vásquez, F.; Cordova, O. Detección de parásitos intestinales en agua y alimentos de Trujillo, Perú. Rev. Peru Med. Exp. Salud Publica 2008, 25, 144–148. [Google Scholar]
- Tito, R.Y.; Chaffron, S.; Caenepeel, C.; Lima-Mendez, G.; Wang, J.; Vieira-Silva, S.; Falony, G.; Hildebrand, F.; Darzi, Y.; Rymenans, L.; et al. Population-level analysis of Blastocystis subtype prevalence and variation in the human gut microbiota. Gut 2018, 68, 1180–1189. [Google Scholar] [CrossRef] [PubMed]
- Sheehan, D.J.; Raucher, B.G.; McKitrick, J.C. Association of Blastocystis hominis with signs and symptoms of human disease. J. Clin. Microbiol. 1986, 24, 548–550. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Gonzalez, D.E.; Martinez-Flores, W.A.; Reyes-Gordillo, J.; Ramirez-Miranda, M.E.; Arroyo-Escalante, S.; Romero-Valdovinos, M.; Stark, D.; Souza-Saldivar, V.; Martinez-Hernandez, F.; Flisser, A.; et al. Blastocystis infection is associated with irritable bowel syndrome in a Mexican patient population. Parasitol. Res. 2012, 110, 1269–1275. [Google Scholar] [CrossRef] [PubMed]
- Shirvani, G.; Fasihi-Harandi, M.; Raiesi, O.; Bazargan, N.; Zahedi, M.J.; Sharifi, I.; Kalantari-Khandani, B.; Nooshadokht, M.; Shabandoust, H.; Mohammadi, M.A.; et al. Prevalence and Molecular Subtyping of Blastocystis from Patients with Irritable Bowel Syndrome, Inflammatory Bowel Disease and Chronic Urticaria in Iran. Acta Parasitol. 2019, 65, 90–96. [Google Scholar] [CrossRef]
- Dogruman-Al, F.; Kustimur, S.; Yoshikawa, H.; Tuncer, C.; Simsek, Z.; Tanyuksel, M.; Araz, E.; Boorom, K. Blastocystis subtypes in irritable bowel syndrome and inflammatory bowel disease in Ankara, Turkey. Mem. Instit. Oswaldo Cruz 2009, 104, 724–727. [Google Scholar] [CrossRef] [PubMed]
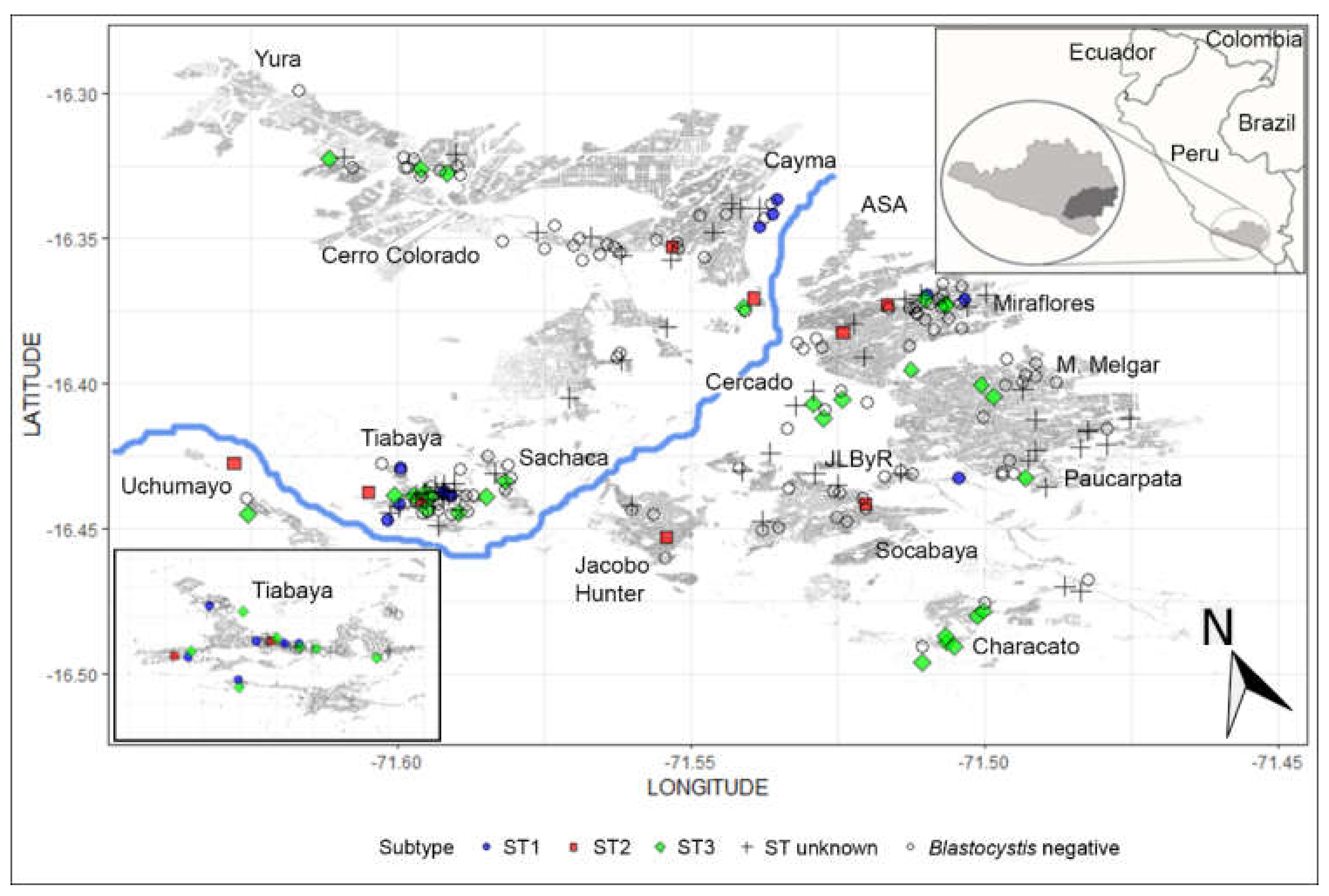
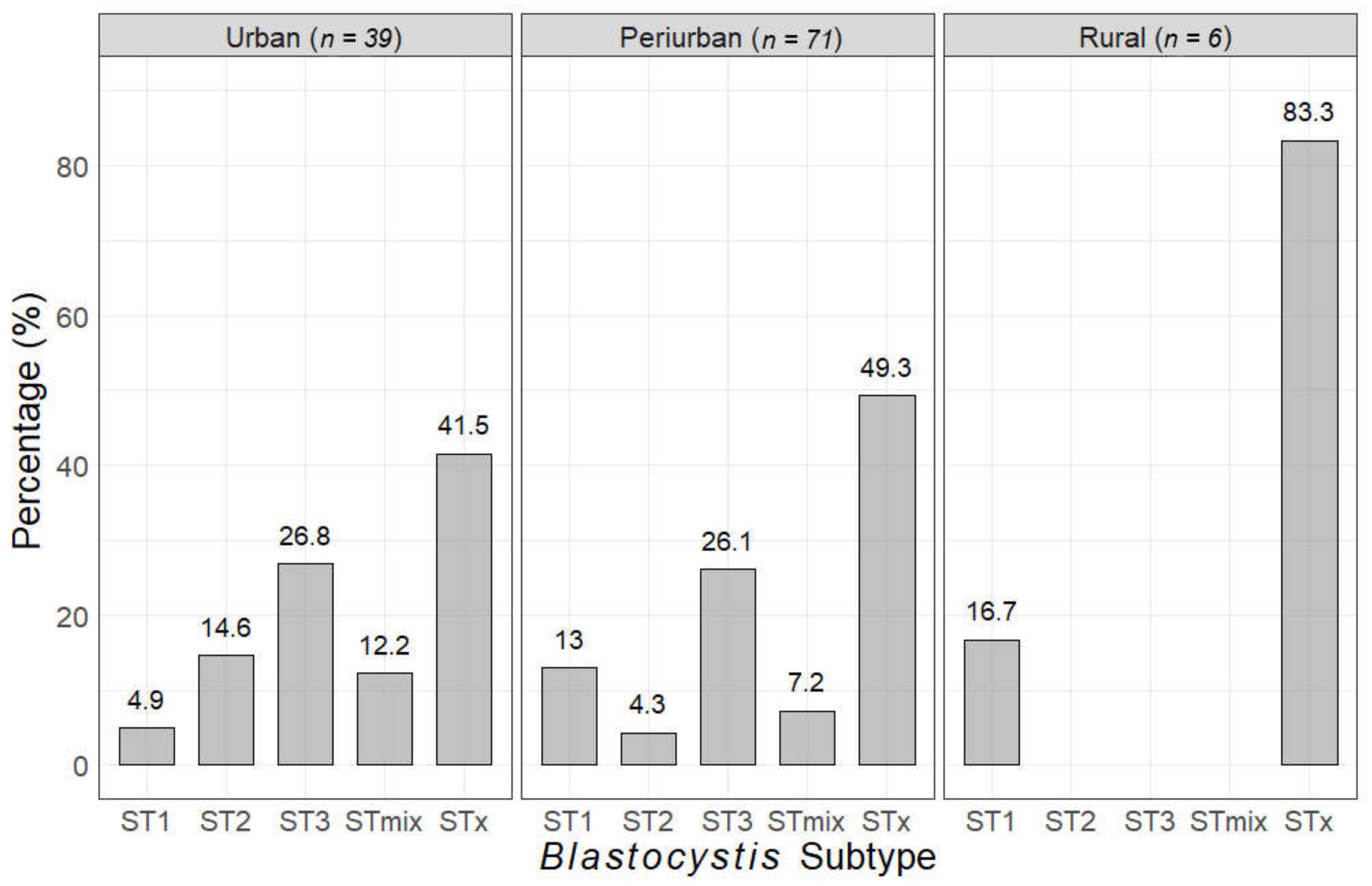
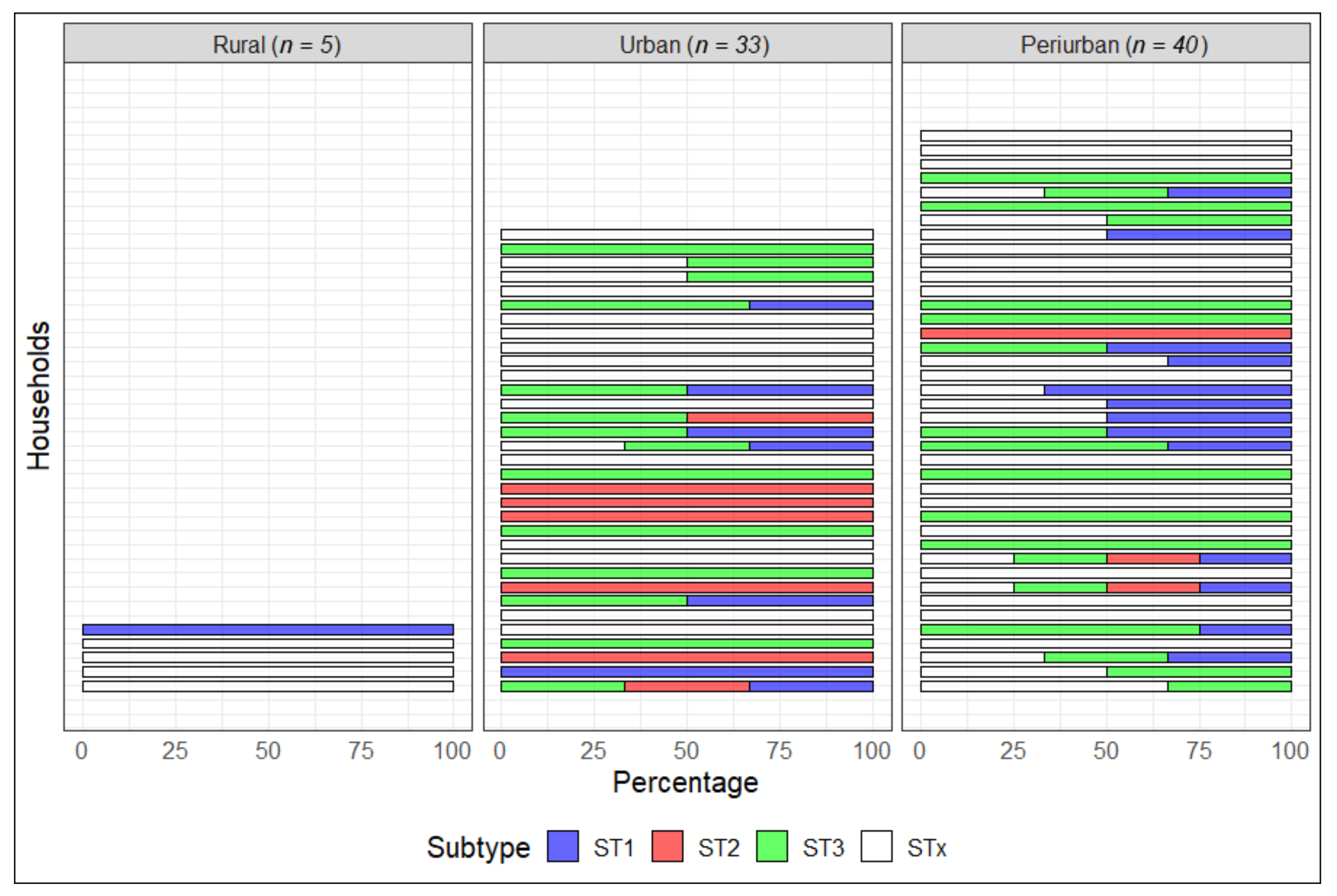
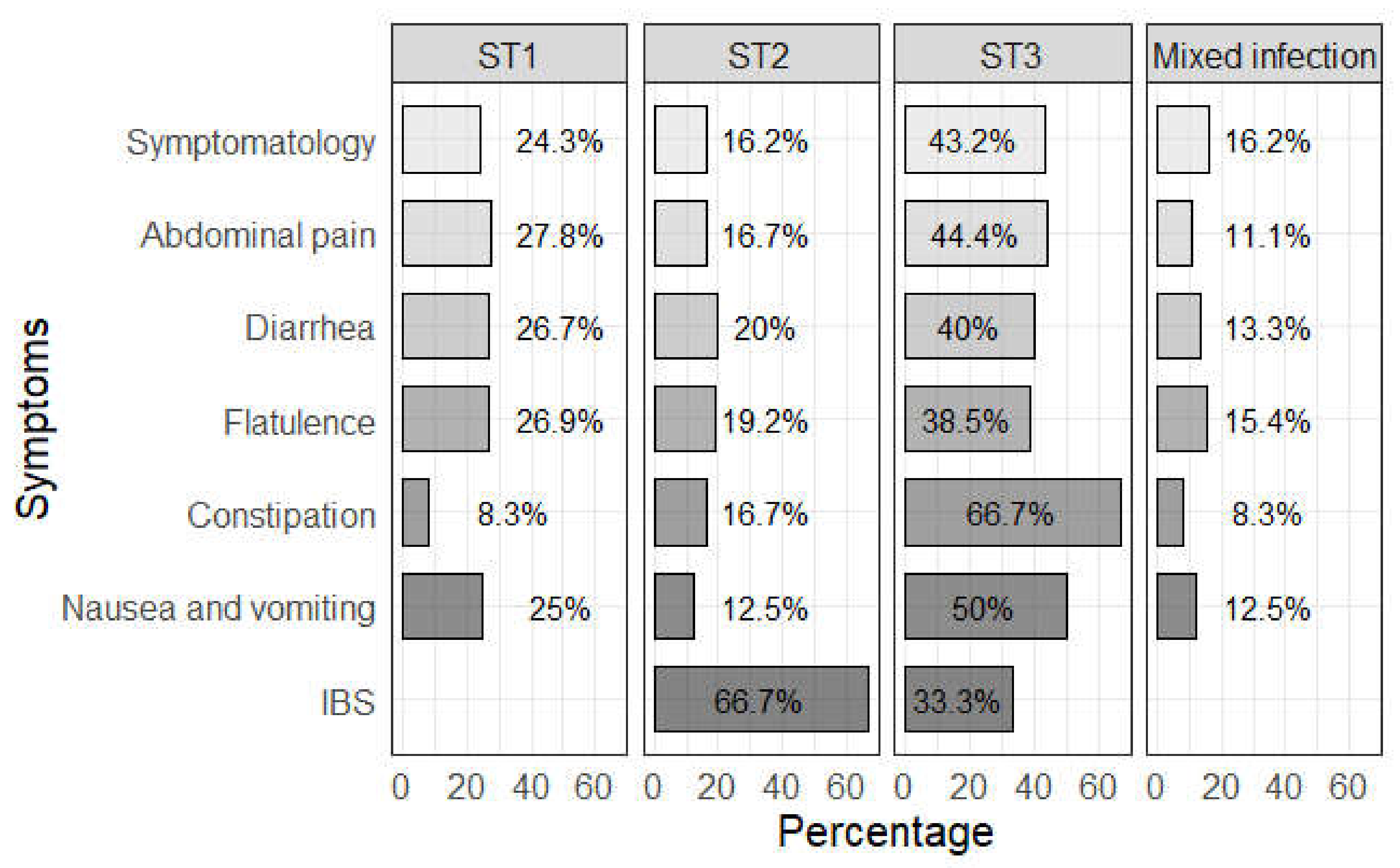
| Type of Blastocystis Infection | Blastocystis Subtype (n) | Prevalence (%) | 95% CI |
|---|---|---|---|
| Simple infection | ST1 (12) | 10.3 | 4.3–16.3 |
| ST2 (9) | 7.8 | 2.8–12.8 | |
| ST3 (29) | 25 | 17.0–33.0 | |
| Unknown (56) | 48.3 | 39.3–57.3 | |
| Mixed infection | ST1,3 (9) | 7.8 | 2.8–12.8 |
| ST1,2,3 (1) | 0.8 | 0–2.9 |
| Blastocystis Subtype | |||||
|---|---|---|---|---|---|
| Variable | ST1 | ST2 | ST3 | Mixed Infection | p-Value |
| n = 21 | n = 10 | n = 39 | n = 10 | ||
| Female | 16 (76%) | 5 (50%) | 21 (54%) | 7 (70.0%) | 0.304 † |
| Age | |||||
| Mean (SD) | 32.5 (23%) | 40.1 (23%) | 37.3 (23%) | 31 (26%) | 0.995 & |
| Median (IQR) | 33 (11–49%) | 46 (18–57%) | 33 (14–54%) | 16 (10–50%) | |
| Areas | |||||
| Urban | 6 (29%) | 7 (70%) | 16 (41%) | 5 (50%) | 0.178 † |
| Periurban | 15 (71%) | 3 (30%) | 23 (59%) | 5 (50%) | |
| Water supply | |||||
| Tap water | 20 (95%) | 10 (100%) | 36 (92%) | 10 (100%) | 1 † |
| Not tap water | 1 (5%) | 3 (8%) | |||
| Final feces disposal | |||||
| Piped sewer system | 20 (95%) | 10 (100%) | 35 (90%) | 10 (100%) | 0.761 † |
| Latrine | 1 (6%) | 4 (10%) | |||
| Presence of animals | 15 (71%) | 9 (90%) | 32 (82%) | 7 (70%) | 0.554 † |
| Presence of dogs | 12 (57%) | 8 (80%) | 26 (67%) | 6 (60%) | 0.631 † |
| Presence of cats | 5 (24%) | 2 (20%) | 12 (31%) | 2 (20%) | 0.874 † |
| Presence of guinea pigs | 3 (14%) | 1 (10%) | 10 (26%) | 2 (20%) | 0.725 † |
| Presence of rabbits | 5 (24%) | 2 (20%) | 9 (23%) | 2 (20%) | 1 † |
| Presence of poultry | 5 (24%) | 2 (20%) | 14 (36%) | 2 (20%) | 0.672 † |
| Presence of flies | 15 (71%) | 6 (60%) | 29 (74%) | 7 (70%) | 0.831 † |
| Presence of cockroaches | 5 (24%) | 1 (10%) | 9 (23%) | 2 (20%) | 0.911 † |
| Presence of rodents | 3 (14%) | 2 (20%) | 7 (18%) | 1 (10%) | 1 † |
| Place of food consumption | |||||
| House | 17 (81%) | 7 (70%) | 32 (82%) | 8 (80%) | 0.377 † |
| Restaurant | 2 (10%) | 1 (10%) | 1 (3%) | 1 (10%) | |
| House and restaurant | 2 (20%) | 4 (10%) | |||
| House and food cart | 2 (10%) | 2 (5%) | 1 (10%) | ||
| Kind of water consumption | |||||
| Boiled water | 18 (86%) | 8 (80%) | 32 (82%) | 9 (90%) | 0.737 † |
| Tap water | 1 (5%) | ||||
| Both | 2 (10%) | 2 (20%) | 7 (18%) | 1 (10%) | |
| Fresh vegetables consumption | 8 (38%) | 5 (50%) | 14 (36%) | 4 (40%) | 0.904 † |
| Fresh fruits consumption | 10 (48%) | 5 (50%) | 16 (41%) | 6 (60%) | 0.755 † |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ascuña-Durand, K.; Salazar-Sánchez, R.S.; Castillo-Neyra, R.; Ballón-Echegaray, J. Relative Frequency of Blastocystis Subtypes 1, 2, and 3 in Urban and Periurban Human Populations of Arequipa, Peru. Trop. Med. Infect. Dis. 2020, 5, 178. https://doi.org/10.3390/tropicalmed5040178
Ascuña-Durand K, Salazar-Sánchez RS, Castillo-Neyra R, Ballón-Echegaray J. Relative Frequency of Blastocystis Subtypes 1, 2, and 3 in Urban and Periurban Human Populations of Arequipa, Peru. Tropical Medicine and Infectious Disease. 2020; 5(4):178. https://doi.org/10.3390/tropicalmed5040178
Chicago/Turabian StyleAscuña-Durand, Kasandra, Renzo S. Salazar-Sánchez, Ricardo Castillo-Neyra, and Jorge Ballón-Echegaray. 2020. "Relative Frequency of Blastocystis Subtypes 1, 2, and 3 in Urban and Periurban Human Populations of Arequipa, Peru" Tropical Medicine and Infectious Disease 5, no. 4: 178. https://doi.org/10.3390/tropicalmed5040178
APA StyleAscuña-Durand, K., Salazar-Sánchez, R. S., Castillo-Neyra, R., & Ballón-Echegaray, J. (2020). Relative Frequency of Blastocystis Subtypes 1, 2, and 3 in Urban and Periurban Human Populations of Arequipa, Peru. Tropical Medicine and Infectious Disease, 5(4), 178. https://doi.org/10.3390/tropicalmed5040178





