WIPO Re:Search: Catalyzing Public-Private Partnerships to Accelerate Tropical Disease Drug Discovery and Development
Abstract
1. Introduction
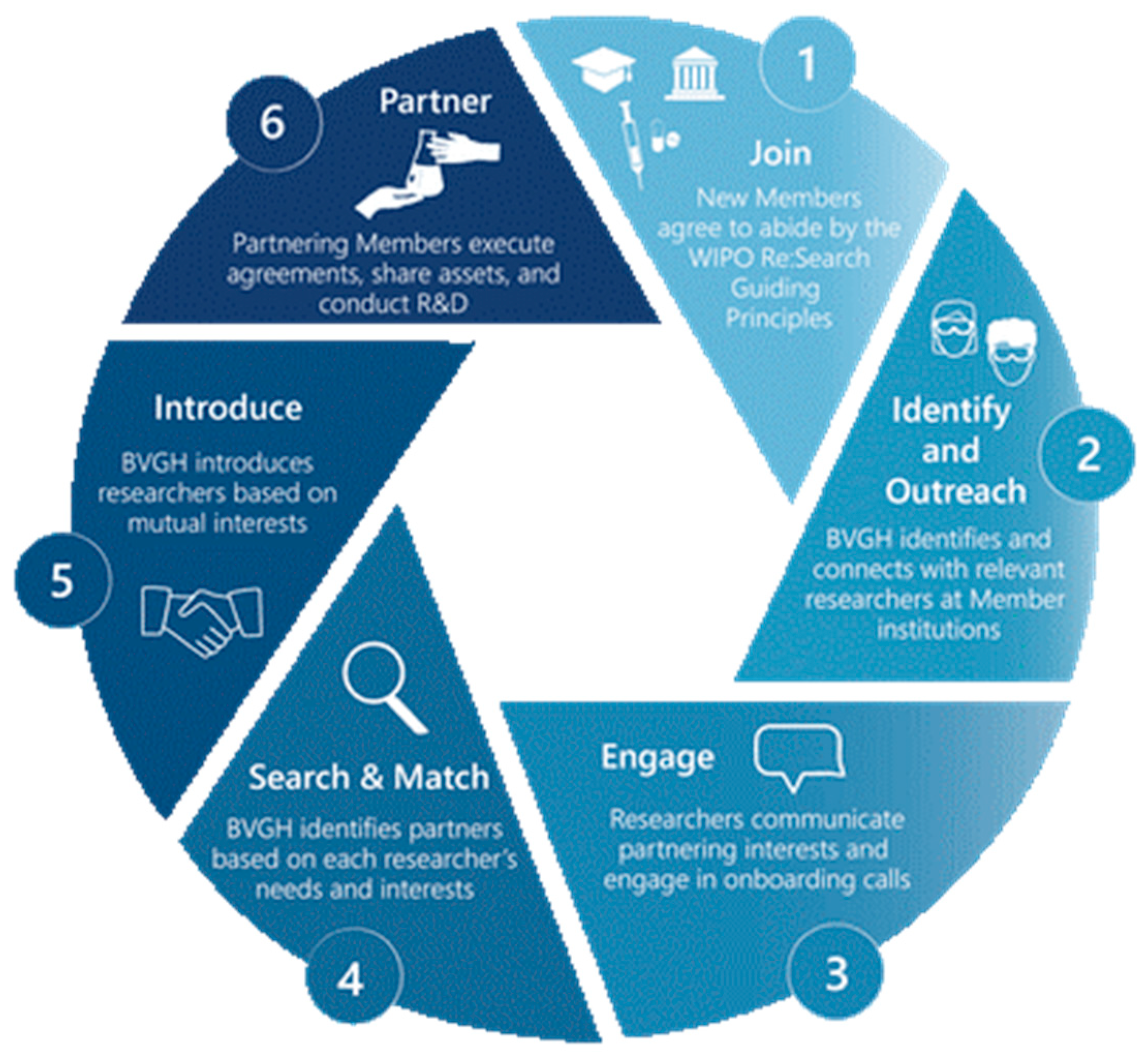
2. WIPO Re:Search: Advancing Product R&D for Tropical Diseases and Other Diseases of Poverty Through a Novel Public-Private Partnership Approach
3. Case Studies: Leveraging Pharmaceutical Company Assets to Drive Tropical Disease R&D at Academic/Non-Profit Entities and Smaller Companies
3.1. Malaria
3.2. Schistosomiasis
3.3. Dengue
3.4. Onchocerciasis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Neglected Tropical Diseases. Available online: https://www.who.int/neglected_diseases/diseases/en/ (accessed on 22 February 2019).
- GBD 2017 DALYs and HALE Collaborators. Global, regional, and national disability-adjusted life-years (DALYs) for 359 diseases and injuries and healthy life expectancy (HALE) for 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1859–1922. [Google Scholar] [CrossRef]
- World Malaria Report 2018; World Health Organization: Geneva, Switzerland, 2018; Licence: CC BY-NC-SA 3.0 IGO; Available online: https://apps.who.int/iris/bitstream/handle/10665/275867/9789241565653-eng.pdf (accessed on 19 March 2019).
- United Nations Sustainable Development Goals. Available online: https://www.un.org/sustainabledevelopment/ (accessed on 21 February 2019).
- World Health Statistics 2018: Monitoring Health for the SDGs, Sustainable Development Goals; World Health Organization: Geneva, Switzerland, 2018; Licence: CC BY-NC-SA 3.0 IGO; Available online: https://apps.who.int/iris/bitstream/handle/10665/272596/9789241565585-eng.pdf (accessed on 19 March 2019).
- Reaching a Billion. Ending Neglected Tropical Diseases: A Gateway to Universal Health Coverage. Fifth Progress Report on the London Declaration on NTDs. Uniting to Combat NTDs 2017. Available online: https://unitingtocombatntds.org/wp-content/themes/tetloose/app/staticPages/fifthReport/files/fifth_progress_report_english.pdf (accessed on 19 March 2019).
- Vale, N.; Gouveia, M.J.; Rinaldi, G.; Brindley, P.J.; Gartner, F.; Correia da Costa, J.M. Praziquantel for schistosomiasis: Single-drug metabolism revisited, mode of action, and resistance. Antimicrob. Agents Chemother 2017, 61, e02582-16. [Google Scholar] [CrossRef] [PubMed]
- Cioli, D.; Pica-Mattoccia, L.; Basso, A.; Guidi, A. Schistosomiasis control: Praziquantel forever? Mol. Biochem. Parasitol. 2014, 195, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Wilder-Smith, A.; Ooi, E.E.; Horstick, O.; Wills, B. Dengue. Lancet 2019, 393, 350–363. [Google Scholar] [CrossRef]
- Freitas-Junior, L.H.; Chatelain, E.; Kim, H.A.; Siqueira-Neto, J.L. Visceral leishmaniasis treatment: What do we have, what do we need and how to deliver it? Int. J. Parasitol. Drugs Drug Resist. 2012, 2, 11–19. [Google Scholar] [CrossRef] [PubMed]
- G-FINDER 2018; Policy Cures Research: Sydney, Australia, 2019; Available online: https://www.policycuresresearch.org/wp-content/uploads/Y11_G-FINDER_Full_report_Reaching_new_heights.pdf (accessed on 19 March 2019).
- Health Product Research and Development Fund: A Proposal for Financing and Operation; World Health Organization Special Programme for Research and Training in Tropical Diseases: Geneva, Switzerland, 2016; Available online: https://apps.who.int/iris/bitstream/handle/10665/204522/9789241510295_eng.pdf (accessed on 19 March 2019).
- Furuse, Y. Analysis of research intensity on infectious disease by disease burden reveals which infectious diseases are neglected by researchers. Proc. Natl. Acad. Sci. USA 2019, 116, 478–483. [Google Scholar] [CrossRef] [PubMed]
- Pedrique, B.; Strub-Wourgaft, N.; Some, C.; Olliaro, P.; Trouiller, P.; Ford, N.; Pécoul, B.; Bradol, J.-H. The drug and vaccine landscape for neglected diseases (2000–11): A systematic assessment. Lancet Glob. Health 2013, 1, e371–e379. [Google Scholar] [CrossRef]
- Klug, D.M.; Gelb, M.H.; Pollastri, M.P. Repurposing strategies for tropical disease drug discovery. Bioorg. Med. Chem. Lett. 2016, 26, 2569–2576. [Google Scholar] [CrossRef] [PubMed]
- WIPO Re:Search Guiding Principles. Available online: http://www.wipo.int/export/sites/www/research/docs/guiding_principles.pdf (accessed on 23 February 2019).
- WIPO Re:Search Members. Available online: https://bvgh.org/wp-content/uploads/2019/01/1-10-2019-WIPO-ReSearch-Member-List.pdf (accessed on 18 March 2019).
- Accelerating R&D for Neglected Diseases through Global Collaborations: WIPO Re:Search Partnership Stories 2013–2015; BIO Ventures for Global Health: Seattle, WA, USA, 2016; Available online: https://bvgh.org/wp-content/uploads/2017/07/2013-2015-WIPO-ReSearch-Storybook.pdf (accessed on 19 March 2019).
- 2018 BVGH Partnership Hub Annual Report; BIO Ventures for Global Health: Seattle, WA, USA, 2019; Available online: https://bvgh.org/wp-content/uploads/2019/02/BVGH-Partnership-Hub-Annual-Report-2018-v3.pdf (accessed on 19 March 2019).
- 2018 Access to Medicine Index; Access to Medicine Foundation: Amsterdam, The Netherlands, 2018. Available online: https://accesstomedicinefoundation.org/media/uploads/downloads/5c1a81b1d525a_Access-to-Medicine-Index-2018.pdf (accessed on 19 March 2019).
- DNDi Target Product Profiles. Available online: https://www.dndi.org/diseases-projects/target-product-profiles/ (accessed on 18 March 2019).
- MMV Target Product Profiles and Target Candidate Profiles. Available online: https://www.mmv.org/research-development/information-scientists/target-product-profiles-target-candidate-profiles (accessed on 18 March 2019).
- WIPO Re:Search Collaboration Pipeline, 2011–2019. Available online: https://bvgh.org/wp-content/uploads/2019/03/Mar.-2019-WIPO-ReSearch-Pipeline.pdf (accessed on 18 March 2019).
- Boddey, J.A.; Hodder, A.N.; Gunther, S.; Gilson, P.R.; Patsiouras, H.; Kapp, E.A.; Pearce, J.A.; de Koning-Ward, T.F.; Simpson, R.J.; Crabb, B.S.; et al. An aspartyl protease directs malaria effector proteins to the host cell. Nature 2010, 463, 627–631. [Google Scholar] [CrossRef] [PubMed]
- Sleebs, B.E.; Lopaticki, S.; Marapana, D.S.; O’Neill, M.T.; Rajasekaran, P.; Gazdik, M.; Gunther, S.; Whitehead, L.W.; Lowes, K.N.; Barfod, L.; et al. Inhibition of Plasmepsin V activity demonstrates its essential role in protein export, PfEMP1 display, and survival of malaria parasites. PLoS Biol. 2014, 12, e1001897. [Google Scholar] [CrossRef] [PubMed]
- Boddey, J.A.; Carvalho, T.G.; Hodder, A.N.; Sargeant, T.J.; Sleebs, B.E.; Marapana, D.; Lopaticki, S.; Nebl, T.; Cowman, A.F. Role of plasmepsin V in export of diverse protein families from the Plasmodium falciparum exportome. Traffic 2013, 14, 532–550. [Google Scholar] [CrossRef] [PubMed]
- Boddey, J.A.; (Walter and Eliza Hall Institute of Medical Research, Parkville, VIC, Australia); Cowman, A.F.; (Walter and Eliza Hall Institute of Medical Research, Parkville, VIC, Australia); Fine, J.B.; (MSD, Kenilworth, NJ, USA); Olsen, D.B.; (MSD, Kenilworth, NJ, USA); Sleebs, B.E.; (Walter and Eliza Hall Institute of Medical Research, Parkville, VIC, Australia). Personal communication, 2019.
- Cowman, A.F.; (Walter and Eliza Hall Institute of Medical Research, Parkville, VIC, Australia); Jackson, P.; (Johnson & Johnson, San Diego, CA, USA); Sleebs, B.E.; (Walter and Eliza Hall Institute of Medical Research, Parkville, VIC, Australia). Personal communication, 2019.
- Trampuz, A.; Jereb, M.; Muzlovic, I.; Prabhu, R.M. Clinical review: Severe malaria. Crit. Care 2003, 7, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Idro, R.; Marsh, K.; John, C.C.; Newton, C.R. Cerebral malaria: Mechanisms of brain injury and strategies for improved neurocognitive outcome. Pediatr. Res. 2010, 68, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Asada, M.; (Eisai Co., Ltd., Tsukuba-shi, Ibaraki, Japan); Craig, A.G.; (Liverpool School of Tropical Medicine, Liverpool, England). Personal communication, 2019.
- Rojo-Arreola, L.; Long, T.; Asarnow, D.; Suzuki, B.M.; Singh, R.; Caffrey, C.R. Chemical and genetic validation of the statin drug target to treat the helminth disease, schistosomiasis. PLoS ONE 2014, 9, e87594. [Google Scholar] [CrossRef] [PubMed]
- Caffrey, C.R.; (University of California, San Diego, CA, USA); Fine, J.B.; (MSD, Kenilworth, NJ, USA); Olsen, D.B.; (MSD, Kenilworth, NJ, USA). Personal communication, 2019.
- World Health Organization: Dengue and Severe Dengue. Available online: https://www.who.int/news-room/fact-sheets/detail/dengue-and-severe-dengue (accessed on 24 February 2019).
- Souza, D.G.; Fagundes, C.T.; Sousa, L.P.; Amaral, F.A.; Souza, R.S.; Souza, A.L.; Kroon, E.G.; Sachs, D.; Cunha, F.Q.; Bukin, E.; et al. Essential role of platelet-activating factor receptor in the pathogenesis of Dengue virus infection. Proc. Natl. Acad. Sci. USA 2009, 106, 14138–14143. [Google Scholar] [CrossRef] [PubMed]
- Herz, J.; (60P Pharmaceuticals, Washington, DC, USA); Loock, D.; (60P Pharmaceuticals, Washington, DC, USA); Reiche, S.; (Pfizer, New York, NY, USA). Personal communication, 2019.
- World Health Organization: Onchocerciasis. Available online: https://www.who.int/en/news-room/fact-sheets/detail/onchocerciasis (accessed on 27 February 2019).
- Basanez, M.G.; Pion, S.D.; Boakes, E.; Filipe, J.A.; Churcher, T.S.; Boussinesq, M. Effect of single-dose ivermectin on Onchocerca volvulus: A systematic review and meta-analysis. Lancet Infect. Dis. 2008, 8, 310–322. [Google Scholar] [CrossRef]
- Plaisier, A.P.; van Oortmarssen, G.J.; Remme, J.; Habbema, J.D. The reproductive lifespan of Onchocerca volvulus in West African savanna. Acta Trop. 1991, 48, 271–284. [Google Scholar] [CrossRef]
- Twum-Danso, N.A. Loa loa encephalopathy temporally related to ivermectin administration reported from onchocerciasis mass treatment programs from 1989 to 2001: Implications for the future. Filaria J. 2003, 2 (Suppl. 1), S7. [Google Scholar] [CrossRef]
- Gardon, J.; Gardon-Wendel, N.; Demanga, N.; Kamgno, J.; Chippaux, J.P.; Boussinesq, M. Serious reactions after mass treatment of onchocerciasis with ivermectin in an area endemic for Loa loa infection. Lancet 1997, 350, 18–22. [Google Scholar] [CrossRef]
- Kamgno, J.; Pion, S.D.; Chesnais, C.B.; Bakalar, M.H.; D’Ambrosio, M.V.; Mackenzie, C.D.; Nana-Djeunga, H.C.; Gounoue-Kamkumo, R.; Njitchouang, G.R.; Nwane, P.; et al. A test-and-not-treat strategy for onchocerciasis in Loa loa-endemic areas. N. Engl. J. Med. 2017, 377, 2044–2052. [Google Scholar] [CrossRef] [PubMed]
- Cho-Ngwa, F.; (University of Buea, Cameroon); Hartmann, A.; (Merck KGaA, Darmstadt, Germany); Mrak, N.; (Merck KGaA, Darmstadt, Germany). Personal communication, 2019.
| Parasitic Diseases | Bacterial Diseases | Viral Diseases | Other Conditions |
|---|---|---|---|
| Chagas disease | Buruli ulcer | Dengue | Podoconiosis |
| Cysticercosis | Leprosy | Rabies | Snakebite |
| Dracunculiasis | Trachoma | ||
| Echinococcosis | Tuberculosis | ||
| Foodborne trematodiases 1 | Yaws | ||
| Human African trypanosomiasis | |||
| Leishmaniasis | |||
| Lymphatic filariasis | |||
| Malaria | |||
| Onchocerciasis | |||
| Schistosomiasis | |||
| Soil-transmitted helminthiases |
| Advancing Business and Corporate Social Responsibility Objectives |
|
| Accelerating R&D Programs |
|
| Expanding Global Brand Awareness and Visibility |
|
| Increasing Scientific Recognition |
|
| Providing Additional Value for LMIC Organizations and Researchers |
|
| # | Criterion | Rationale |
|---|---|---|
| 1 | Advanced stage of development (typically preclinical or later) | Advanced-stage collaborations have higher chances of successfully completing all phases of development. |
| 2 | Addresses an unmet medical need | Such projects have greater likelihood of receiving funding or being transferred to an appropriate partner, such as a PDP or company, for continued development. |
| 3 | Novel target or approach | Such collaborations have greater potential to lead to innovative products that address unmet needs. |
| 4 | Aligns with published target product profiles (TPPs) or target candidate profiles (TCPs) (e.g., [21,22]) | TPPs and TCPs define experts’ standards and expected needs for on-the-ground product use. By aligning with TPP or TCP criteria, a product is more likely to meet experts’ standards and be accessible and acceptable for use in endemic regions. |
| 5 | Exemplifies the use of IP as an enabler of innovation, or demonstrates a company’s commitment to improving access to medicines | WIPO Re:Search has two core goals: to accelerate product development for diseases of poverty, and to demonstrate that IP is not a barrier to access to medicines. Projects that fulfill this criterion exemplify the latter goal. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manner, C.K.; Graef, K.M.; Dent, J. WIPO Re:Search: Catalyzing Public-Private Partnerships to Accelerate Tropical Disease Drug Discovery and Development. Trop. Med. Infect. Dis. 2019, 4, 53. https://doi.org/10.3390/tropicalmed4010053
Manner CK, Graef KM, Dent J. WIPO Re:Search: Catalyzing Public-Private Partnerships to Accelerate Tropical Disease Drug Discovery and Development. Tropical Medicine and Infectious Disease. 2019; 4(1):53. https://doi.org/10.3390/tropicalmed4010053
Chicago/Turabian StyleManner, Cathyryne K., Katy M. Graef, and Jennifer Dent. 2019. "WIPO Re:Search: Catalyzing Public-Private Partnerships to Accelerate Tropical Disease Drug Discovery and Development" Tropical Medicine and Infectious Disease 4, no. 1: 53. https://doi.org/10.3390/tropicalmed4010053
APA StyleManner, C. K., Graef, K. M., & Dent, J. (2019). WIPO Re:Search: Catalyzing Public-Private Partnerships to Accelerate Tropical Disease Drug Discovery and Development. Tropical Medicine and Infectious Disease, 4(1), 53. https://doi.org/10.3390/tropicalmed4010053





