Rabies in the Caribbean: A Situational Analysis and Historic Review
Abstract
1. Introduction
2. Materials and Methods
3. Results
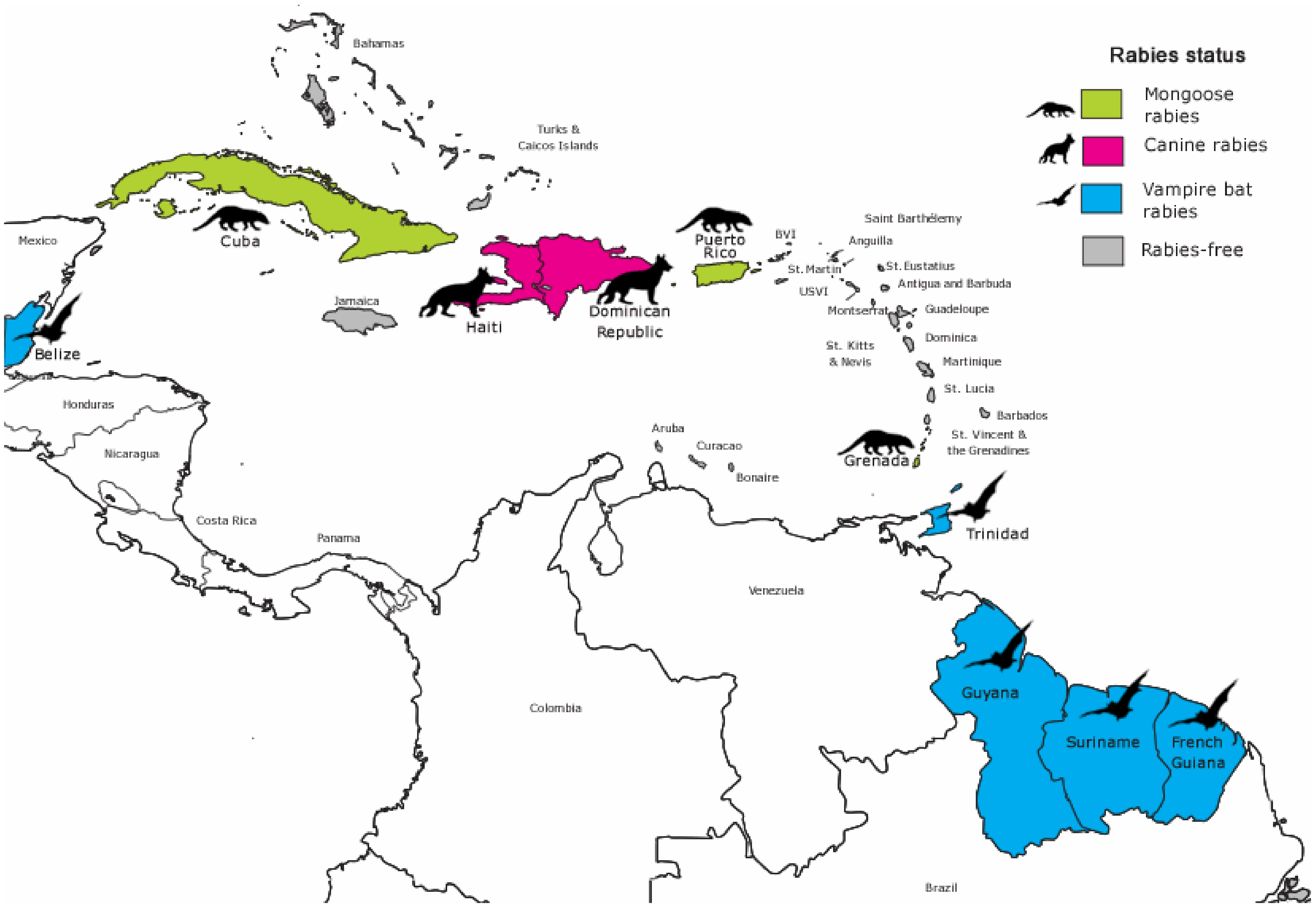
3.1. Rabies Status and Main Animal Reservoir
3.2. Risk of Rabies Introduction
3.3. Rabies Case Burden
3.3.1. Animal Rabies
3.3.2. Human Rabies
3.4. Rabies Case Definitions, Protocols, and Legislation
3.5. Rabies Virus Exposure Incidents
3.6. National Agencies with Responsibility for Rabies Surveillance
3.7. Rabies Surveillance Programs in Animal Populations
3.7.1. General
3.7.2. Wildlife
3.7.3. Domestic Animals
3.8. Animal Rabies Vaccination
3.9. Human Pre-Exposure and Post-Exposure Prophylaxis (PEP)
3.10. Animal Reservoir Population Control Programs
3.11. Rabies Laboratory Diagnostics
3.12. National Import Health Regulations Related to Rabies
4. Discussion
4.1. Rabies Status and Reservoir Hosts
4.2. Risk of Introduction
4.3. Surveillance and Reporting of Animal Rabies
4.4. Surveillance and Reporting of Human Rabies
4.5. Human Exposures and Public Health Risk
4.6. Rabies Diagnostic Capacity
4.7. Prevention and Control Measures
4.7.1. Animal Vaccination
4.7.2. Human Pre-Exposure and Post-Exposure Prophylaxis (PEP)
4.7.3. Reservoir Host Population Control Programs
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dietzgen, R.G.; Calisher, C.H.; Kurath, G.; Kuzmin, I.V.; Rodriguez, L.L.; Stone, D.M.; Tesh, R.B.; Tordo, N.; Walker, P.J.; Wetzel, T.; et al. Virus taxonomy: Ninth Report of The International Committee on Taxonomy of Viruses; Elsevier: Oxford, UK, 2011; pp. 686–714. [Google Scholar]
- Banyard, A.C.; Evans, J.S.; Luo, T.R.; Fooks, A.R. Lyssaviruses and bats: Emergence and zoonotic threat. Viruses 2014, 6, 2974–2990. [Google Scholar] [CrossRef] [PubMed]
- Manning, S.E.; Rupprecht, C.E.; Fishbein, D.; Hanlon, C.A.; Lumlertdacha, B.; Guerra, M.; Meltzer, M.I.; Dhankhar, P.; Vaidya, S.A.; Jenkins, S.R.; et al. Human rabies prevention--United States, 2008: Recommendations of the advisory committee on immunization practices. MMWR 2008, 57, 1–28. [Google Scholar] [PubMed]
- Rupprecht, C.E.; Willoughby, R.; Slate, D. Current and future trends in the prevention, treatment and control of rabies. Expert Rev. Anti-Infect. 2006, 4, 1021–1038. [Google Scholar] [CrossRef] [PubMed]
- Hanlon, C.A.; Niezgoda, M.; Rupprecht, C.E. Rabies in terrestrial animals. In Rabies; Jackson, A.C., Wunner, W.H., Eds.; Academic Press Publications: Cambridge, UK, 2007; pp. 201–258. [Google Scholar]
- Woldehiwet, Z. Rabies: Recent developments. Res. Vet. Sci. 2002, 73, 17–25. [Google Scholar] [CrossRef]
- Udow, S.J.; Marrie, R.A.; Jackson, A.C. Clinical features of dog- and bat-acquired rabies in humans. Clin. Infect. Dis. 2013, 57, 689–696. [Google Scholar] [CrossRef] [PubMed]
- Nadin-Davis, S.A.; Real, L.A. Molecular phylogenetics of the lyssaviruses-insights from a coalescent approach. Adv. Virus Res. 2011, 79, 203–238. [Google Scholar] [PubMed]
- Velasco-Villa, A.; Mauldin, M.R.; Shi, M.; Escobar, L.E.; Gallardo-Romero, N.F.; Damon, I.; Olson, V.A.; Streicker, D.G.; Emerson, G. The history of rabies in the western hemisphere. Antiviral Res. 2017, 146, 221–232. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.S.; Orciari, L.A.; Yager, P.A.; Seidel, H.D.; Warner, C.K. Epidemiologic and historical relationships among 87 rabies virus isolates as determined by limited sequence analysis. J. Infect. Dis. 1992, 166, 296–307. [Google Scholar] [CrossRef] [PubMed]
- Vos, A.; Nunan, C.; Bolles, D.; Muller, T.; Fooks, A.R.; Tordo, N.; Baer, G.M. Historical review: The occurence of rabies in pre-Colombian Central America: A historical search. Epidemiol. Infect. 2011, 139, 1445–1452. [Google Scholar] [CrossRef] [PubMed]
- Badrane, H.; Tordo, N. Host switching in Lyssavirus history from the chiroptera to the carnivora orders. J. Virol. 2001, 75, 8096–8104. [Google Scholar] [CrossRef] [PubMed]
- Kuzmina, N.A.; Kuzmin, I.V.; Ellison, J.A.; Taylor, S.T.; Bergman, D.T.; Dew, B.; Rupprecht, C.E. A reassessment of the evolutionary timescale of bat rabies viruses based upon glycoprotein gene sequences. Virus Genes 2013, 47, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Acha, P.N. A review of rabies prevention and control in the Americas, 1970–1980. Overall status of rabies. Bull. Off. Int. Epiz. 1981, 93, 9–52. [Google Scholar]
- Baer, G.M. Vampire bat and bovine paralytic rabies. In The Natural History of Rabies, 2nd ed.; Baer, G.M., Ed.; CRC Press Inc.: Boca Raton, FL, USA, 1991; pp. 389–403. [Google Scholar]
- Pastoret, P.P.; Van Gucht, S.; Brochier, B. Eradicating rabies at source. Rev. Sci. Tech. Off. Int. Epiz. 2014, 33, 509–519. [Google Scholar]
- Pawan, J.L. The transmission of paralytic rabies in Trinidad by the vampire bat (Desmodus rotundus murinus Wagner 1840). Ann. Trop. Med. Parasitol. 1936, 30, 101–129. [Google Scholar] [CrossRef]
- Acha, P.N. Epidemiology of paralytic bovine rabies and bat rabies. Bulll. Off. Int. Epizoot. 1967, 67, 343–382. [Google Scholar]
- Nehaul, B.B.G. Rabies transmitted by bats in British Guiana. Am. J. Trop. Med. Hyg. 1955, 4, 550–553. [Google Scholar] [CrossRef] [PubMed]
- Schneider, M.C.; Romijn, P.C.; Uieda, W.; Tamayo, H.; daSilva, D.F.; Leanes, L.F. Rabies transmitted by vampire bats to humans: An emerging zoonotic disease in Latin America? Rev. Panam. Salud Publica 2009, 25, 260–269. [Google Scholar] [CrossRef] [PubMed]
- Schneider, M.C.; Belotto, A.J.; Ade, M.P.; Hendrickx, S.; Leanes, L.F.; Rodrigues, M.J.F.; Medina, G.; Correa, E. Current status of human rabies transmitted by dogs in Latin America. Cad. Saude Publica 2009, 23, 2049–2063. [Google Scholar] [CrossRef]
- Escobar, L.E.; Peterson, A.T.; Favi, M.; Yung, V.; Medina-Vogel, G. Bat borne rabies in Latin America. Rev. Inst. Med. Trop. Sao Paulo 2015, 57, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Vigilato, M.A.N.; Clavijo, A.; Knobl, T.; Silva, H.M.T.; Cosivi, O.; Schneider, M.C.; Leanes, L.F.; Belotto, A.; Espinal, M.A. Progress towards eliminating canine rabies: Policies and perspectives from Latin America and the Caribbean. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2013, 368, 20120143. [Google Scholar] [CrossRef] [PubMed]
- Belotto, A.; Leanes, L.F.; Schneider, M.C.; Tamayo, H.; Correa, E. Overview of rabies in the Americas. Virus Res. 2005, 111, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Vigilato, M.A.N.; Cosivi, O.; Knobi, T.; Clavijo, A.; Silva, H.M.T. Rabies update for Latin America and the Caribbean. Emerg. Infect. Dis. 2013, 19, 678–679. [Google Scholar] [CrossRef] [PubMed]
- Clavijo, A.; Del-Rio-Vilas, V.J.; Mayen, F.L.; Yadon, Z.E.; Belotto, A.J.; Vigilato, M.A.N.; Schneider, M.C.; Cosivi, O. Perspective piece: Gains and future road map for the elimination of dog-transmitted rabies in the Americas. Am. J. Trop. Med. Hyg. 2013, 89, 1040–1042. [Google Scholar] [CrossRef] [PubMed]
- Del-Rio-Vilas, V.J.; Freire-de-Carvalho, M.H.; Vigilato, M.A.; Rocha, F.; Vokaty, A.; Pompei, J.A.; Molina Flores, B.; Fenelon, N.; Cosivi, O. Tribulations of the last mile: Sides from a regional program. Front. Vet. Sci. 2017, 4, 4. [Google Scholar] [CrossRef] [PubMed]
- Pan American Health Organization. PANAFTOSA. Rabies: Pending Challenges―An Unfinished Process. 16th Meeting of Rabies Program Directors in the Americas. REDIPRA 16. Available online: http://www.panaftosa.org/redipra16/index.php?option=com_content&view=article&id=49&Itemid=54&lang=en (accessed on 27 February 2018).
- Freire-de-Carvalho, M.; Vigilato, M.A.N.; Pompei, J.A.; Rocha, F.; Vokaty, A.; Molina-Flores, B.; Cosivi, O.; Del Rio Vilas, V.J. Rabies in the Americas: 1998-2014. PLoS Negl. Trop. Dis. 2018, 12, e0006271. [Google Scholar] [CrossRef] [PubMed]
- Pan American Health Organization. Regional Information System for Epidemiological Surveillance of Rabies (SIRVERA). Available online: http://sivera.panaftosa.org.br/login (assessed on 8 July 2018).
- Ma, X.; Monroe, B.P.; Cleaton, J.M.; Orciari, L.A.; Yager, P.; Li, Y.; Kirby, J.D.; Blanton, J.D.; Petersen, B.W.; Wallace, R.M. Rabies surveillance in the United States during 2016. J. Am. Vet. Med. Assoc. 2018, 252, 945–957. [Google Scholar] [CrossRef] [PubMed]
- Velasco-Villa, A.; Escobar, L.E.; Sanchez, A.; Shi, M.; Streicker, D.G.; Gallardo-Romero, N.F.; Vargas-Pino, F.; Gutierrez-Cedillo, V.; Damon, I.; Emerson, G. Successful strategies implemented towards the elimination of canine rabies in the western hemisphere. Antivir. Res. 2017, 143, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Wallace, R.; Reses, H.; Franka, R.; Dilius, P.; Fenelon, N.; Orciari, L.; Etheart, M.; Destine, A.; Crowdis, K.; Blanton, J.D.; et al. Establishment of a canine rabies burden in Haiti through the implementation of a novel surveillance program. PLoS Negl. Trop. Dis. 2015, 9, e0004245. [Google Scholar] [CrossRef] [PubMed]
- Lefrancois, T.; Petit-Sinturel, M.; Kalloo, M.; Shaw, J.; Herbert-Hackshaw, K.; Trotman, M.; Gongora, V. Caribvet: A model for surveillance of zoonotic diseases. Int. J. Infect. Dis. 2010, 14, e185. [Google Scholar] [CrossRef]
- World Health Organization (WHO). World Health Organization (WHO) Expert Consultation on Rabies; WHO: Geneva, Switzerland, 2018. [Google Scholar]
- Muir, P.; Roome, A. Indigenous rabies in the UK. Lancet 2005, 365, 2175. [Google Scholar] [CrossRef]
- World Organization for Animal Health (OIE). Rabies, Belize. In World Animal Health Information Database (WAHID). Available online: http://www.oie.int/wahis_2/public/wahid.php/Diseaseinformation/statusdetail (accessed on 21 November 2017).
- Meynard, J.B.; Flamand, C.; Dupuy, C.; Mahamat, A.; Eltges, F.; Queuche, F.; Renner, J.; Fontanella, J.M.; Hommel, D.; Dussart, P.; et al. First human rabies cases in French Guiana, 2008: Epidemiological investigation and control. PLoS Negl. Trop. Dis. 2012, 6, e1537. [Google Scholar] [CrossRef] [PubMed]
- Berger, F.; Desplanches, N.; Baillargeaux, S.; Joubert, M.; Miller, M.; Ribadeau-Dumas, F.; Spiegel, A.; Bourhy, H. Rabies risk: Difficulties encountered during management of grouped cases of bat bites in 2 isolated villages in French Guiana. PLoS Negl. Trop. Dis. 2013, 7, e2258. [Google Scholar] [CrossRef] [PubMed]
- Seetahal, J.F.R.; Velasco-Villa, A.; Allicock, O.M.; Adesiyun, A.A.; Bissessar, J.; Amour, K.; Phillip-Hosein, A.; Marston, D.A.; McElhinney, L.M.; Shi, M.; et al. Evolutionary history and phylogeography of rabies viruses associated with outbreaks in Trinidad. PLoS Negl. Trop. Dis. 2013, 7, e2365. [Google Scholar] [CrossRef] [PubMed]
- Cruz, R.B. Situation of Rabies in Cuba. In Proceedings of the The XVI International Conference on Rabies in the Americas, Ottawa, ON, Canada, 16–21 October 2005. [Google Scholar]
- Nadin Davis, S.A.; Torres, G.; Ribas, M.; Guzman, M.; Cruz De La Paz, R.; Morales, M.; Wandeler, A.I. A molecular epidemiological study of rabies in Cuba. Epidemiol. Infect. 2006, 134, 1313–1324. [Google Scholar] [CrossRef] [PubMed]
- Constantine, D.G. Bat Rabies and Other Lyssavirus Infections; United States Geological Survey National Wildlife Health Center: Reston, VA, USA, 2009. [Google Scholar]
- Goodwin, G.G.; Greenhall, A. A review of the bats of Trinidad and Tobago: Descriptions, rabies infection and ecology. Amer. Muse. Nat. Hist. 1961, 122, 187–302. [Google Scholar]
- Tierkel, E.S.; Arbona, G.; Rivera, A.; de Juan, A. Mongoose rabies in Puerto Rico. Public Health Rep. 1952, 67, 274–278. [Google Scholar] [CrossRef] [PubMed]
- Everard, C.O.R.; Everard, J.D. Mongoose rabies in the Caribbean. Ann. N. Y. Acad. Sci. 1992, 653, 356–366. [Google Scholar] [CrossRef] [PubMed]
- Everard, C.O.R.; Murray, D.; Gilbert, P.K. Rabies in Grenada. Trans. R. Soc. Trop. Med. Hyg. 1972, 66, 878–888. [Google Scholar] [CrossRef]
- Jonkers, A.H.; Alexis, F.; Loregnard, R. Mongoose rabies in Grenada. West Indian Med. J. 1969, 18, 167–170. [Google Scholar] [PubMed]
- Everad, C.O.R.; Everard, J.D. Mongoose rabies in Grenada. In Population Dynamics of Rabies in Wildlife; Bacon, P.J., Ed.; Nat. Acad. Press: London, UK, 1985; pp. 43–69. [Google Scholar]
- Espeut, W.B. On the acclimatization of the Indian mongoose in Jamaica. Proc. Zool. Soc. Lond. 1882, 28, 712–714. [Google Scholar]
- Hinton, H.E.; Dunn, A.M.S. Mongooses: Their Natural History and Behavior; UC Press: Berkeley, CA, USA, 1967. [Google Scholar]
- Horst, G.R.; Hoagland, D.B.; Kilpatrick, C.W. The mongoose in the West Indies: The biogeography and population biology of an introduced species. In Biogeography of the West Indies: Patterns and Perspectives, 2nd ed.; Woods, C.A., Sergile, F.E., Eds.; CRC Press: Boca Raton, FL, USA, 2001; pp. 409–424. [Google Scholar]
- Nadin Davis, S.A.; Velez, J.; Malaga, C.; Wandeler, A.I. A molecular epidemiological study of rabies in Puerto Rico. Virus Res. 2008, 131, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Zieger, U.; Marston, D.A.; Sharma, R.; Chikweto, A.; Tiwari, K.; Sayyid, M.; Louison, B.; Hooman, G.; Voller, K.; Breed, A.C.; et al. The phylogeography of rabies in Grenada, West Indies, and implication for control. PLoS Negl. Trop. Dis. 2014, 8, e3251. [Google Scholar] [CrossRef] [PubMed]
- InterAmerican Institute for Coorporation on Agriculture (IICA). A Proposal to Modernize the Agricultural Health Services of Belize; IICA: Belize, CA, USA, 1996; p. 3. [Google Scholar]
- Cordero, Y.; de los Angeles, R.M.; Beatriz, C.H.; Cintra, Y.; Tejero, Y.; Gonzalez, J.; Daileny, P.; Castilho, J. Epidemiology and Molecular Characterization of Rabies in Cuba from 2003 to 2016. In Proceedings of the XXVIII Rabies in the Americas Conference, Calgary, AB, Canada, 22–26 October 2017; p. 17. [Google Scholar]
- Zieger, U.; Cheetham, S.; Santana, S.E.; Leiser-miller, L.; Matthew-Belmar, V.; Hooman, G.; Fooks, A.R. Natural exposure of bats in Grenada to rabies virus. Infect. Ecol. Epidemiol. 2017, 7, 1332935. [Google Scholar] [CrossRef] [PubMed]
- Price, J.L.; Everard, C.O.R. Rabies virus and antibody in bats in Grenada and Trinidad. J. Wildl. Dis. 1977, 131–134. [Google Scholar] [CrossRef]
- Speer, K.A.; Petronio, B.J.; Simmons, N.B.; Richey, R.; Magrini, K.; Soto-Centeno, J.A.; Reed, D.L. Population struture of a widespread bat (Tadarida brasiliensis) in an island system. Ecol. Evol. 2017, 7, 7585–7598. [Google Scholar] [CrossRef] [PubMed]
- Millien, M.F. Update of the Rabies Situation in Haiti. In Proceedings of the Regional Conference on Surveillance and Research on Vector-borne and Emerging Animal and Zoonotic Diseases in the Caribbean, Guadeloupe, France, 21 April 2015. [Google Scholar]
- Everard, C.O.R.; James, A.C.; Da Breo, S. Ten years of rabies surveillance in Grenada, 1968–1977. Bull. Pan. Am. Health Organ. 1979, 13, 342–353. [Google Scholar] [PubMed]
- World Health Organization (WHO). WHO Wkly Epidemiol. Rec.; WHO: Geneva, Switzerland, 1976; pp. 381–382. [Google Scholar]
- Dyer, H.; Yager, P.; Orciari, L.; Greenburg, L.; Wallace, R.; Hanlon, C.A.; Blanton, J.D. Rabies surveillance in the United States during 2013. J. Am. Vet. Med. Assoc. 2014, 245, 1111–1123. [Google Scholar] [CrossRef] [PubMed]
- Krebs, J.W.; Smith, J.S.; Rupprecht, C.E.; Childs, J.E. Rabies surveillance in the United States during 1998. J. Am. Vet. Med. Assoc. 1999, 215, 1786–1798. [Google Scholar] [PubMed]
- Everard, C.O.R.; Everard, J.D. Mongoose rabies. Rev. Infect. Dis. 1988, 10, 610–614. [Google Scholar] [CrossRef]
- Organizacion Panamericana de la Salud (OPS). Republica Dominicana: Elimination of Dog-Transmitted Rabies in Latin America: Situation Analysis; OPS: Washington, WA, USA, 2004; pp. 58–59. [Google Scholar]
- Gannon, M.R.; Kurta, A.; Rodriguez-Duran, A.; Willig, M.R. Bats of Puerto Rico: An Island Focus and A Caribbean Perspective; Texas Tech University Press: Lubbock, TX, USA, 2005. [Google Scholar]
- Fenton, M.B.; Bernard, E.; Hollis, L.; Johnston, D.S.; Lausen, C.L.; Ratcliffe, J.M.; Riskin, D.K.; Taylor, J.R.; Zigouris, J. The bat fauna of Lamanai, Belize: Roosts and tropical roles. J. Trop. Ecol. 2001, 17, 511–524. [Google Scholar] [CrossRef]
- Willig, M.R.; Presley, S.J.; Bloch, C.P.; Genoways, H.H. Macroecology of Caribbean bats: Effects of area, elevation, latitude and hurricane-induced disturbance. In Island Bats: Evolution, Ecology and Conservation; Fleming, T.H., Racey, P.A., Eds.; The University of Chicago Press Ltd.: Chicago, IL, USA, 2009; pp. 216–264. [Google Scholar]
- Lim, B.K.; Emngstrom, M.D. Species diversity of bats (Mammalia: Chiroptera) in Iwokrama forest, Guyana, and the Guianan subregion: Implications for conservation. Biodivers. Conserv. 2001, 10, 613–657. [Google Scholar]
- Shapley, R.L.; Wilson, D.E.; Warren, A.N.; Barnett, A.A. Bats of the Potaro Plateau region, Western Guyana. Mammalia 2005, 69, 375–394. [Google Scholar] [CrossRef]
- Genoways, H.H.; Phillips, C.J.; Baker, R.J. Bats of the Antillean island of Grenada: A new zoogeographic perspective. Mammal. Pap. Univ. Nebraska State Mus. 1998, 98, 1–28. [Google Scholar]
- Flanders, J.; Yohe, L.; Rossiter, S.; Davalos, L.M. Field Key to the Bats of Hispanola. 2014. Available online: https://seis.bristol.ac.uk/~bzjrf/JonFlanders/Field_key_bats_Hispaniola.pdf (accessed on 21 November 2017).
- Gomes, G.A.; Reid, F.A. Bats of Trinidad and Tobago: A Field Guide and Natural History; Trinibats: Port of Spain, Trinidad, 2015. [Google Scholar]
- Moratelli, R.; Wilson, D.E.; Novaes, R.L.M.; Helgen, K.M.; Gutierrez, E.E. Caribbean Myotis (Chiroptera, Vespertilionidae), with description of a new species from Trinidad and Tobago. J. Mammal. 2017, 98, 1–15. [Google Scholar] [CrossRef]
- Seetahal, J.F.R.; Sanchez-Vazquez, M.J.; Vokaty, A.; Carrington, C.V.F.; Mahabir, R.; Adesiyun, A.A.; Rupprecht, C.E. Unpublished work. 2018.
- Pumo, D.E.; Goldin, E.Z.; Elliot, B.; Phillips, C.J.; Genoways, H.H. Mitochondrial DNA polymorphism in three Antillean island populations of the fruit bats, Artibeus jamaicensis. Mol. Biol. Evol. 1988, 5, 79–89. [Google Scholar] [PubMed]
- Butcher, L.V. The present status of paralytic rabies (bat transmitted) in Trinidad. West Indian Med. J. 1958, 7, 17–20. [Google Scholar] [PubMed]
- Pan American Health Organization (PAHO). Country volume: Belize. In Health in the Americas; PAHO: Washington, DC, USA, 2002; pp. 64–75. [Google Scholar]
- Pan American Health Organization (PAHO). Country volume: Guyana. In Health in the Americas; PAHO: Washington, DC, USA, 2007; pp. 395–411. [Google Scholar]
- Fielding, W.J.; Ostberg, A. Animal Welfare in the Wider Caribbean in 2008, with Emphasis on Dogs; The Pegasus Foundation: Concord, NH, USA, 2008. [Google Scholar]
- Berger, S. Rabies. In Infectious Diseases of Haiti; Gideon Informatics Inc.: Los Angeles, CA, USA, 2014; pp. 274–277. [Google Scholar]
- Millien, M.F.; Pierre-Louis, J.B.; Wallace, R.; Caldas, E.; Rwangabgoba, J.M.; Poncelet, J.L. Control of dog mediated human rabies in Haiti: No time to spare. PLoS Negl. Trop. Dis. 2015, 9, 0003806. [Google Scholar] [CrossRef] [PubMed]
- Beran, G.W. Rabies and infections by rabies related viruses. In Handbook of Zoonoses, 2nd ed.; Beran, G.W., Ed.; CRC Press: Boca Raton, FL, USA, 2014; p. 313. [Google Scholar]
- Hadad, J.H. Country volume: Cuba. In Health in the Americas; Pan American Health Organization (PAHO): Washington, DC, USA, 2012. [Google Scholar]
- Centers for Disease Control and Prevention (CDC). Rabies in Puerto Rico: First Case of Human Rabies Caused by Mongoose bite in Puerto Rico. Available online: http://www.cdc.gov/worldrabiesday/puerto-rico.html (accessed on 16 October 2016).
- Dupuy, C.; Berger, F.; Baudrimont, X.; Martrenchar, A.; Moutou, F.; Spiegel, A.; Desplanches, N.; Krieger, N. Situation de la rage animale en Guyane. Bull. Epid. Santé Anim. Alim. 2011, 43, 26–30. [Google Scholar]
- Rabies-French Guiana (02): (CY) Canine, OIE. Available online: http://www.promedmail.org/direct.php?id=3624448 (accessed on 4 September 2015).
- Pan American Health Organization (PAHO). Report of the IX Meeting of Directors of National Programs for Rabies Control in Latin America REDIPRA, Santa Cruz de la Sierra, Bolivia, 7–9 October 2002; PAHO: Santa Cruz de la Sierra, Bolivia, 2003; Available online: http://bvs1.panaftosa.org.br/local/file/textoc/IX_Redipra_ing.pdf (accessed on 29 June 2018).
- Mani, R.S.; Madhusudana, S.N. Laboratory diagnosis of human rabies: Recent advances. Sci. World J. 2013, 2013, 569–712. [Google Scholar] [CrossRef] [PubMed]
- The 14th Meeting of Directors of National Programs for Rabies Control in Latin America (REDIPRA), Lima (Peru), 2013. Available online: https://rabiesalliance.org/resource/14th-redipra-lima-peru (accessed on 25 November 2013).
- World Organization for Animal Health (OIE). Vaccine Banks. Available online: http://www.oie.int/support-to-oie-members/vaccine-bank/ (accessed on 25 July 2017).
- Seetahal, J.F.R.; Vokaty, A.; Carrington, C.V.F.; Adesiyun, A.A.; Mahabir, R.; Hinds, A.; Rupprecht, C.E. The history of rabies in Trinidad: Epidemiology and control measures. Trop. Med. Infect. Dis. 2017, 2, 27. [Google Scholar] [CrossRef]
- Coleman, P.G.; Dye, C. Immunization coverage required to prevent outbreaks of dog rabies. Vaccine 1996, 14, 185–186. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Report of the rabies global conference. In Global Elimination of Dog-Mediated Human Rabies; World Health Organization (WHO) and World Organization for Animal Health (OIE): Geneva, Switzerland, 2016. [Google Scholar]
- Thomas, D.; Delgado, A.; Louison, B.; Lefrancois, T.; Shaw, J. Examining dog owners’ beliefs regarding rabies vaccination during government-funded vaccine clinics in Grenada to improve vaccine coverage rates. Prev. Vet. Med. 2013, 110, 563–569. [Google Scholar] [CrossRef] [PubMed]
- Fielding, W.J.; Gall, M.; Green, D.B.; Eller, W.S. Care of dogs and attitudes of dog owners in Port-au-Prince, the Republic of Haiti. J. Appl. Anim. Welf. Sci. 2012, 15, 126–253. [Google Scholar] [CrossRef] [PubMed]
- Director of Agriculture of Trinidad and Tobago. Extracts from the administration reports of the director of agriculture for the years 1923–1948. Caribb. Med. J. 1959, 21, 172–184. [Google Scholar]
- Berger, S. Rabies. In Infectious Diseases of the Dominican Republic; Gideon Informatics Inc.: Los Angeles, CA, USA, 2015; pp. 274–277. [Google Scholar]
- Stoner-Duncan, B.; Streicker, D.G.; Tedeschi, C.M. Vampire bats and rabies: Towards an ecological solution to a public health problem. PLoS Negl. Trop. Dis. 2014, 8, e2867. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.R.; Berentsen, A.R.; Ellis, C.; Davis, A.; Vercauteren, K.C. Estimates of small Indian mongoose densities: Implications for rabies management. J. Wildl. Manag. 2016, 80, 37–47. [Google Scholar] [CrossRef]
- Styezynski, A.; Tran, C.; Dirlikov, E.; Zapara, M.R.; Ryff, K.; Peterson, B.; Cruz Sanchez, A.; Mayshack, M.; Martinez, L.; Condori, R.; et al. Human rabies - Puerto Rico, 2015. MMWR Morb. Mort Wkly Rep. 2017, 65, 1474–1476. [Google Scholar] [CrossRef] [PubMed]
- King, A.A.; Fooks, A.R.; Aubert, M.; Wandeler, A.I. Historical Perspective of Rabies in Europe and the Mediterranean Basin; OIE: Paris, France, 2005; p. 362. [Google Scholar]
| Location | Main Animal Reservoir | Mean # Cases per Year | Main Animals Affected |
|---|---|---|---|
| Belize | Vampire bat | 1–5 | Cattle |
| Cuba | Mongoose | 16–20 | Dogs, cats, mongoose |
| Dominican Republic | Dog | >20 | n.r. |
| French Guiana | Vampire bat | 0–1 | Cattle, dogs, bats |
| Grenada | Mongoose | 1–5 | Mongoose, cattle, small ruminants, dogs, cats |
| Guyana | Vampire bat | 1–5 | Cattle |
| Haiti | Dog | >20 | Dogs, cats |
| Puerto Rico | Mongoose | >20 | Mongoose |
| Suriname | Vampire bat | 0 a | Bats a,b |
| Trinidad | Vampire bat | 6–10 | Cattle, small ruminants |
| Location | Years since Last Reported Case * | Mean # Human Cases per Year * | Causative Variant of Last Human Case | Pre-Exposure Vaccination | Biologics Available for Post-Exposure Prophylaxis |
|---|---|---|---|---|---|
| Belize | >20 | 0 | Canine | Yes | vaccine |
| Cuba | 1–5 | 0–1 | Mongoose | Yes | vaccine and RIG |
| Dominican Republic | <1 | 0–1 | Canine | Yes | vaccine |
| French Guiana | 6–10 | 0–1 | Bat | Yes | vaccine and RIG |
| Grenada | >20 | 0 | Mongoose | Yes | vaccine and RIG |
| Guyana | n.c.r. | 0 | n.c.r. | Yes | vaccine |
| Haiti | <1 | 6–10 # | Canine | Yes | vaccine and RIG |
| Puerto Rico | 1–5 | 0–1 | Mongoose | Yes | vaccine and RIG |
| Suriname | 11–20 | 0–1 | Bat | Yes | vaccine |
| Trinidad | >20 | 0 | Bat | Yes | vaccine |
| Location | Vaccination Requirement | Main Animals Vaccinated | Periodicity | Estimated Population Coverage |
|---|---|---|---|---|
| Belize | recommended ## | Dogs and cats | annually | 80% dogs |
| Cuba | recommended ## | Dogs | annually | >90% dogs ¶ |
| Dominican Republic | recommended | Dogs and cats | n.r. | 80% dogs and cats |
| French Guiana | mandatory by legislation | Ruminants, dogs and cats, equine | biennially | No data available |
| Grenada | recommended ## | Ruminants, dogs and cats, equine | annually | 40% dogs ***; 4% cats; 31% goats; 22% sheep; 4% cattle |
| Guyana | recommended | Bovine | annually | 10% bovine |
| Haiti | recommended ## | Dogs | annually | 40—50% dogs |
| Puerto Rico | recommended | Dogs and cats, ruminants, equine | annually | No data available |
| Suriname | Not conducted | - | - | - |
| Trinidad | mandatory by legislation ## | Ruminants, equine | triennial | 70% bovine; <40% goat and sheep; <40% equine |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Seetahal, J.F.R.; Vokaty, A.; Vigilato, M.A.N.; Carrington, C.V.F.; Pradel, J.; Louison, B.; Sauers, A.V.; Roopnarine, R.; Arrebato, J.C.G.; Millien, M.F.; et al. Rabies in the Caribbean: A Situational Analysis and Historic Review. Trop. Med. Infect. Dis. 2018, 3, 89. https://doi.org/10.3390/tropicalmed3030089
Seetahal JFR, Vokaty A, Vigilato MAN, Carrington CVF, Pradel J, Louison B, Sauers AV, Roopnarine R, Arrebato JCG, Millien MF, et al. Rabies in the Caribbean: A Situational Analysis and Historic Review. Tropical Medicine and Infectious Disease. 2018; 3(3):89. https://doi.org/10.3390/tropicalmed3030089
Chicago/Turabian StyleSeetahal, Janine F. R., Alexandra Vokaty, Marco A. N. Vigilato, Christine V. F. Carrington, Jennifer Pradel, Bowen Louison, Astrid Van Sauers, Rohini Roopnarine, Jusayma C. González Arrebato, Max F. Millien, and et al. 2018. "Rabies in the Caribbean: A Situational Analysis and Historic Review" Tropical Medicine and Infectious Disease 3, no. 3: 89. https://doi.org/10.3390/tropicalmed3030089
APA StyleSeetahal, J. F. R., Vokaty, A., Vigilato, M. A. N., Carrington, C. V. F., Pradel, J., Louison, B., Sauers, A. V., Roopnarine, R., Arrebato, J. C. G., Millien, M. F., James, C., & Rupprecht, C. E. (2018). Rabies in the Caribbean: A Situational Analysis and Historic Review. Tropical Medicine and Infectious Disease, 3(3), 89. https://doi.org/10.3390/tropicalmed3030089







