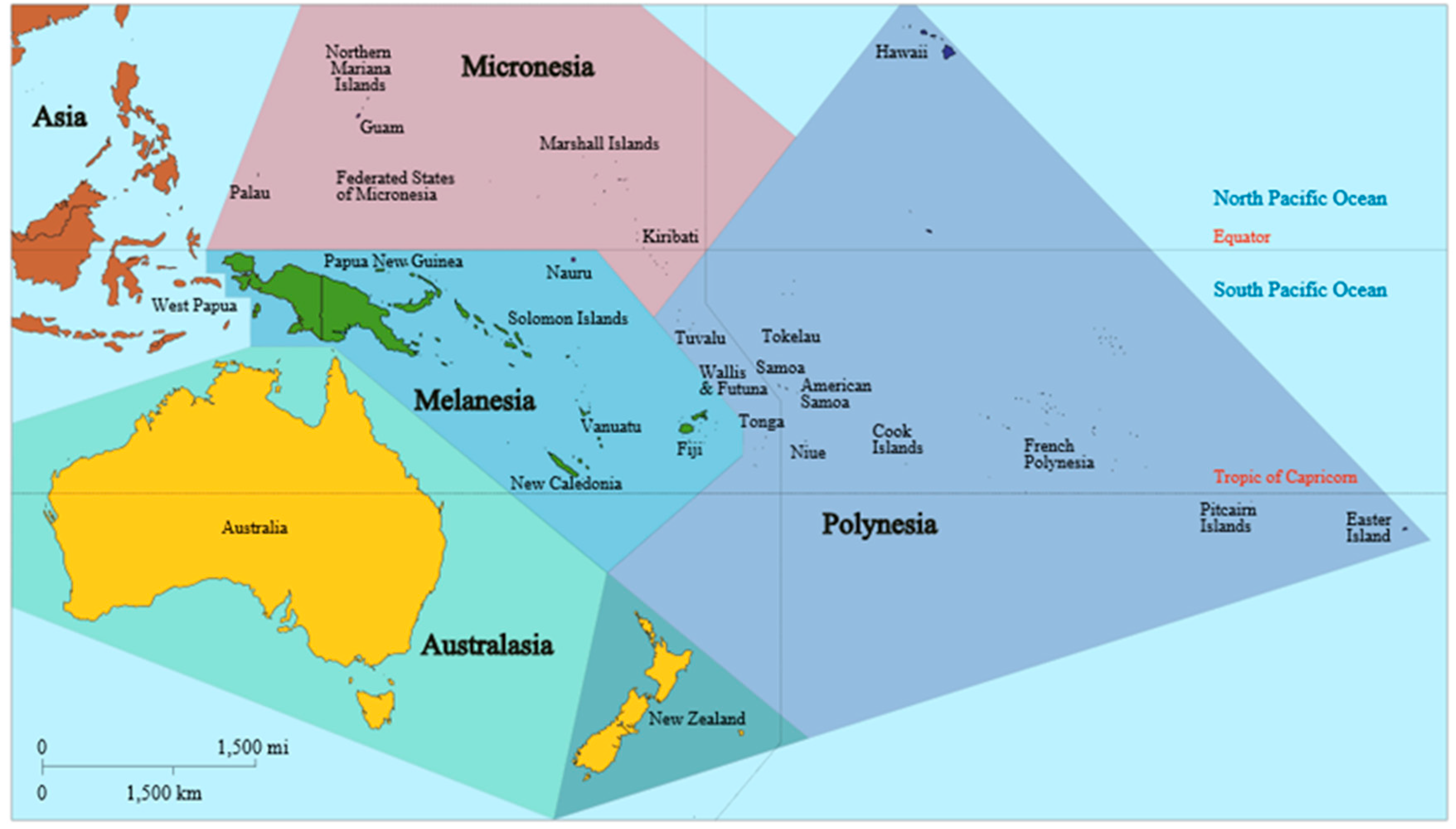
Melioidosis in Papua New Guinea and Oceania
Abstract
:1. History
2. Review of Confirmed Human Melioidosis Cases
2.1. Melioidosis Is Rare in PNG’s Urban Capital
2.2. Melioidosis Hotspot: The Balimo Region of the Western Province, PNG
2.3. Elsewhere in Oceania
3. Current Recommendations and Availability of Measures against Melioidosis
4. Awareness of Melioidosis, and Current and Future Challenges
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mirick, G.S.; Zimmerman, H.M.; Maner, G.D. Melioidosis on Guam. JAMA 1946, 130, 1063–1067. [Google Scholar] [CrossRef]
- Egerton, J. Bovine melioidosis. Aust. Vet. J. 1963, 40, 34–35. [Google Scholar] [CrossRef]
- Egerton, J.R. Melioidosis in a tree climbing kangaroo. Aust. Vet. J. 1963, 39, 243–244. [Google Scholar] [CrossRef]
- Rampling, A. Porcine melioidosis in the territory of Papua New Guinea. Aust. Vet. J. 1964, 40, 241. [Google Scholar] [CrossRef]
- Rowlands, J.B.; Curtis, P.G. A case of melioidosis in Papua and New Guinea. Med. J. Aust. 1965, 2, 494–496. [Google Scholar] [PubMed]
- Newland, R.C. Chronic melioidosis: A case in Sydney. Pathology 1969, 1, 149–152. [Google Scholar] [CrossRef] [PubMed]
- Kingston, C.W. Chronic or latent melioidosis. Med. J. Aust. 1971, 2, 618–621. [Google Scholar] [PubMed]
- Domenech, J.; Lucet, P.; Desoutter, D.; Daynès, P.; Fortineau, O.; Colas, F.; Lechapt, M. An animal health survey. Revue d’Elevage et de Médecine Vétérinaire de Nouvelle-Calédonie 1984, 2, 1–8. (In French) [Google Scholar]
- Poirier, N. Melioidosis: A Propos of a Clinical Case Observed in New Caledonia: Epidemiology, Clinical and Therapeutic Aspects. Ph.D. Thesis, Université de Rennes 1, Rennes, France, 2001. [Google Scholar]
- De Buse, P.J.; Henderson, A.; White, M. Melioidosis in a child in Papua New Guinea: Successful treatment with kanamycin and trimethoprim-sulphamethoxazole. Med. J. Aust. 1975, 2, 476–478. [Google Scholar] [PubMed]
- Lee, L.; Naraqi, S. Primary Gram-negative pneumonia in adults in Papua New Guinea. PNG Med. J. 1980, 23, 174–178. [Google Scholar]
- Currie, B. Melioidosis in Papua New Guinea: Is it less common than in tropical Australia? Trans. R. Soc. Trop. Med. Hyg. 1993, 87, 417. [Google Scholar] [CrossRef]
- Warner, J.M.; Pelowa, D.B.; Currie, B.J. Melioidosis—An uncommon but also under-recognized cause of pneumonia in Papua New Guinea. PNG Med. J. 2010, 53, 176–179. [Google Scholar]
- Barnes, D.J.; Gottlieb, T.; Naraqi, S.; Benn, R. The role of viruses and atypical organisms in the pathogenesis of adult pneumonia in Papua New Guinea. PNG Med. J. 1991, 34, 13–16. [Google Scholar]
- The National Statistical Office. Final Figures–Papua New Guinea; The National Statistical Office: Waigani, Papua New Guinea, November 2013. Available online: http://www.nso.gov.pg/index.php/document-library?view=download&fileId=65 (accessed on 1 March 2018).
- Reece, A.S. A Clinical Report: The Experience of the Balimo Health Centre with the Clinical Syndrome of Melioidosis, Including a Suggested Diagnostic, Therapeutic and Epidemiological Program; Balimo Health Centre: Balimo, Papua New Guinea, 1984. [Google Scholar]
- Warner, J.M.; Pelowa, D.B.; Currie, B.J.; Hirst, R.G. Melioidosis in a rural community of Western Province, Papua New Guinea. Trans. R. Soc. Trop. Med. Hyg. 2007, 101, 809–813. [Google Scholar] [CrossRef] [PubMed]
- Warner, J.M.; Pelowa, D.B.; Gal, D.; Rai, G.; Mayo, M.; Currie, B.J.; Govan, B.; Skerratt, L.F.; Hirst, R.G. The epidemiology of melioidosis in the Balimo region of Papua New Guinea. Epidemiol. Infect. 2008, 136, 965–971. [Google Scholar] [CrossRef] [PubMed]
- Baker, A.; Pearson, T.; Price, E.P.; Dale, J.; Keim, P.; Hornstra, H.; Greenhill, A.; Padilla, G.; Warner, J. Molecular phylogeny of Burkholderia pseudomallei from a remote region of Papua New Guinea. PLoS ONE 2011, 6, e18343. [Google Scholar] [CrossRef] [PubMed]
- Corkill, M.M.; Cornere, B. Melioidosis: A new disease to New Zealand. N. Z. Med. J. 1987, 100, 106–107. [Google Scholar] [PubMed]
- Currie, B.J. Menzies School of Health Research, Darwin, NT, Australia. Unpublished work. 2015. [Google Scholar]
- Armstrong, P.K.; Anstey, N.M.; Kelly, P.M.; Currie, B.J.; Martins, N.; Dasari, P.; Krause, V. Seroprevalence of Burkholderia pseudomallei in East Timorese refugees: Implications for healthcare in East Timor. Asian J. Trop. Med. Public Health 2005, 36, 1496. [Google Scholar]
- Le Hello, S.; Currie, B.J.; Godoy, D.; Spratt, B.G.; Mikulski, M.; Lacassin, F.; Garin, B. Melioidosis in New Caledonia. Emerg. Infect. Dis. 2005, 11, 1607–1609. [Google Scholar] [CrossRef] [PubMed]
- Estivals, M.; du Couedic, L.; Lacassin, F.; Mermond, S.; Levenes, H. Septicemia, bilateral community-acquired pneumonia and empyema due to Burkholderia pseudomallei (melioidosis) with a favorable outcome following prolonged specific antibiotic therapy. Rev. Mal. Respir. 2008, 25, 319–322. [Google Scholar] [CrossRef]
- Melot, B.; Colot, J.; Lacassin, F.; Tardieu, S.; Lapisardi, E.; Mayo, M.; Price, E.P.; Sarovinch, D.S.; Currie, B.J.; Goarant, C. Melioidosis in New Caledonia: A dominant strain in a transmission hotspot. Epidemiol. Infect. 2016, 144, 1330–1337. [Google Scholar] [CrossRef] [PubMed]
- Melioidosis Disease Found in Yap Lab Test. Available online: http://www.mvariety.com/regional-news/62874-melioidosis-disease-found-in-yap-lab-test (accessed on 1 March 2018).
- Yap-EpiNet-8 Feb 2014. Available online: https://www.facebook.com/Yap-Epi-Net-388433107955645/?hc_ref=ARRW79jS3cmeVtRnWmOoRhhoJ5PsLRvS5efkHJfMn93WMtYYLz39U7t_99JBAVohElw&fref=nf (accessed on 1 March 2018).
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Warner, J.M.; Currie, B.J. Melioidosis in Papua New Guinea and Oceania. Trop. Med. Infect. Dis. 2018, 3, 34. https://doi.org/10.3390/tropicalmed3010034
Warner JM, Currie BJ. Melioidosis in Papua New Guinea and Oceania. Tropical Medicine and Infectious Disease. 2018; 3(1):34. https://doi.org/10.3390/tropicalmed3010034
Chicago/Turabian StyleWarner, Jeffrey M., and Bart J. Currie. 2018. "Melioidosis in Papua New Guinea and Oceania" Tropical Medicine and Infectious Disease 3, no. 1: 34. https://doi.org/10.3390/tropicalmed3010034
APA StyleWarner, J. M., & Currie, B. J. (2018). Melioidosis in Papua New Guinea and Oceania. Tropical Medicine and Infectious Disease, 3(1), 34. https://doi.org/10.3390/tropicalmed3010034




