A Statistically Rigorous Approach to Experimental Design of Vertical Living Walls for Green Buildings
Abstract
:1. Introduction
2. Materials and Methods
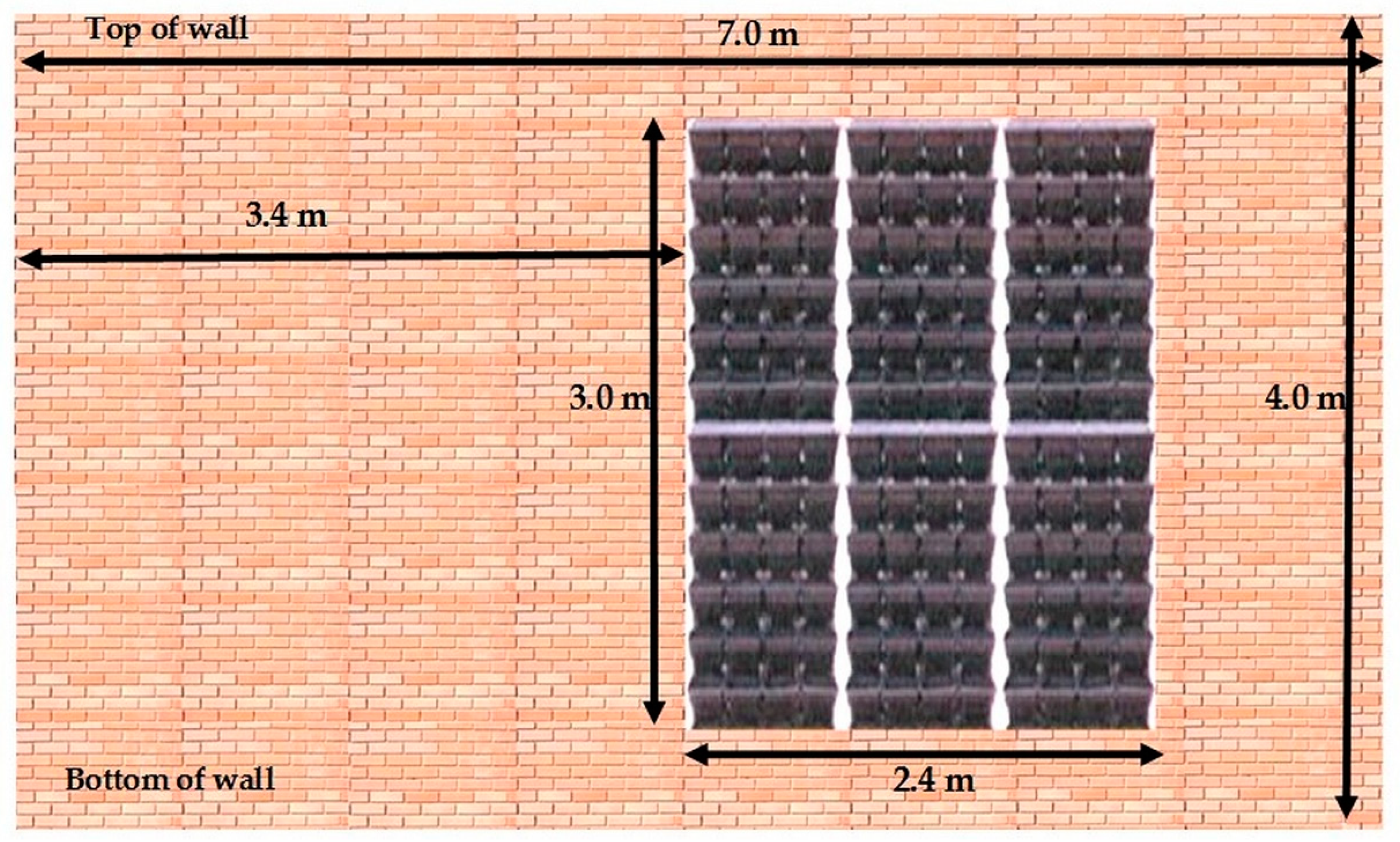
2.1. Experimental Setup
2.1.1. Plant Selection
2.1.2. Soil Substrates
2.1.3. Irrigation
2.1.4. Arrangement of Living Wall Pots
2.2. Statistical Analysis
3. Results and Discussions
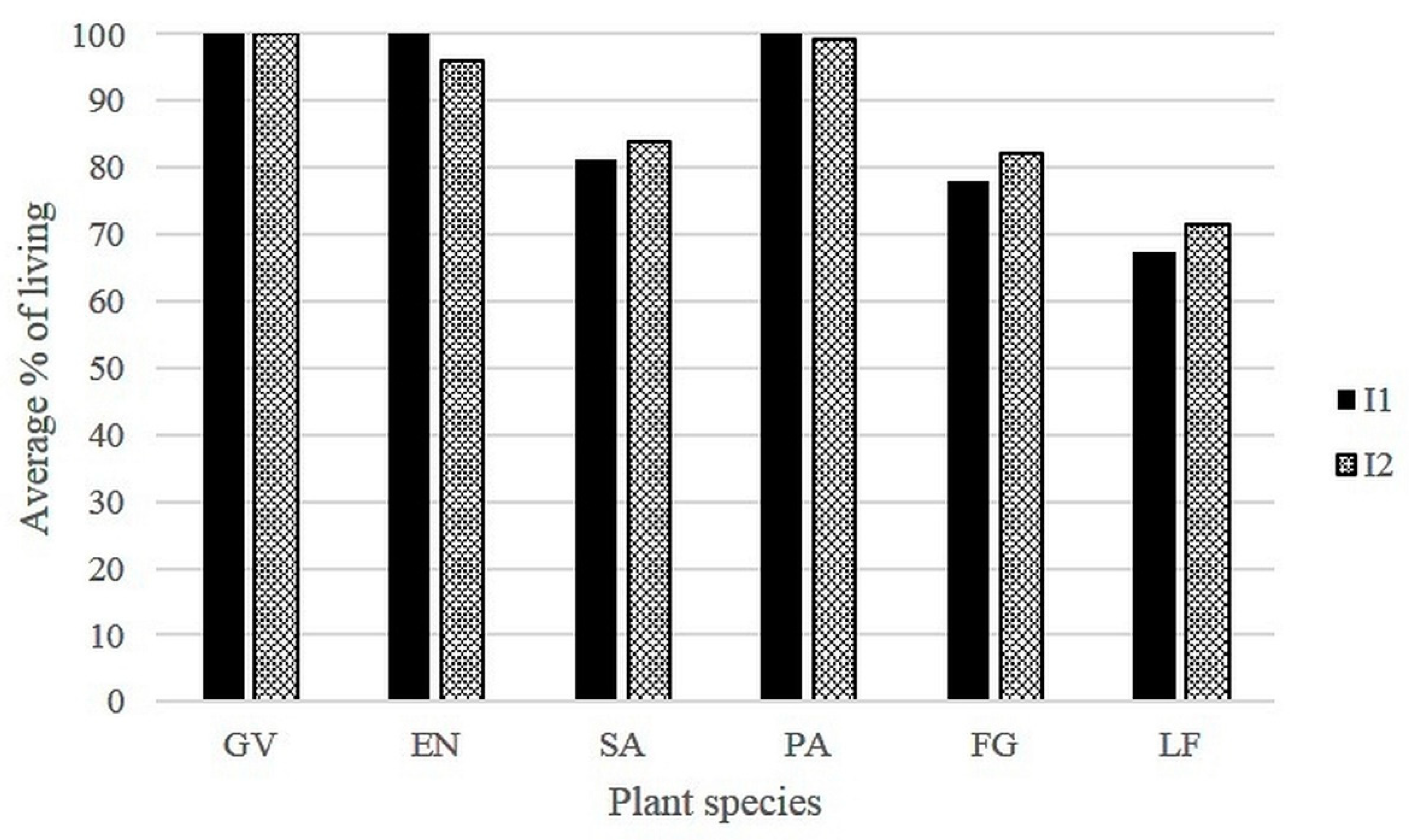
3.1. Plant Health and Survival
3.2. Plant Height
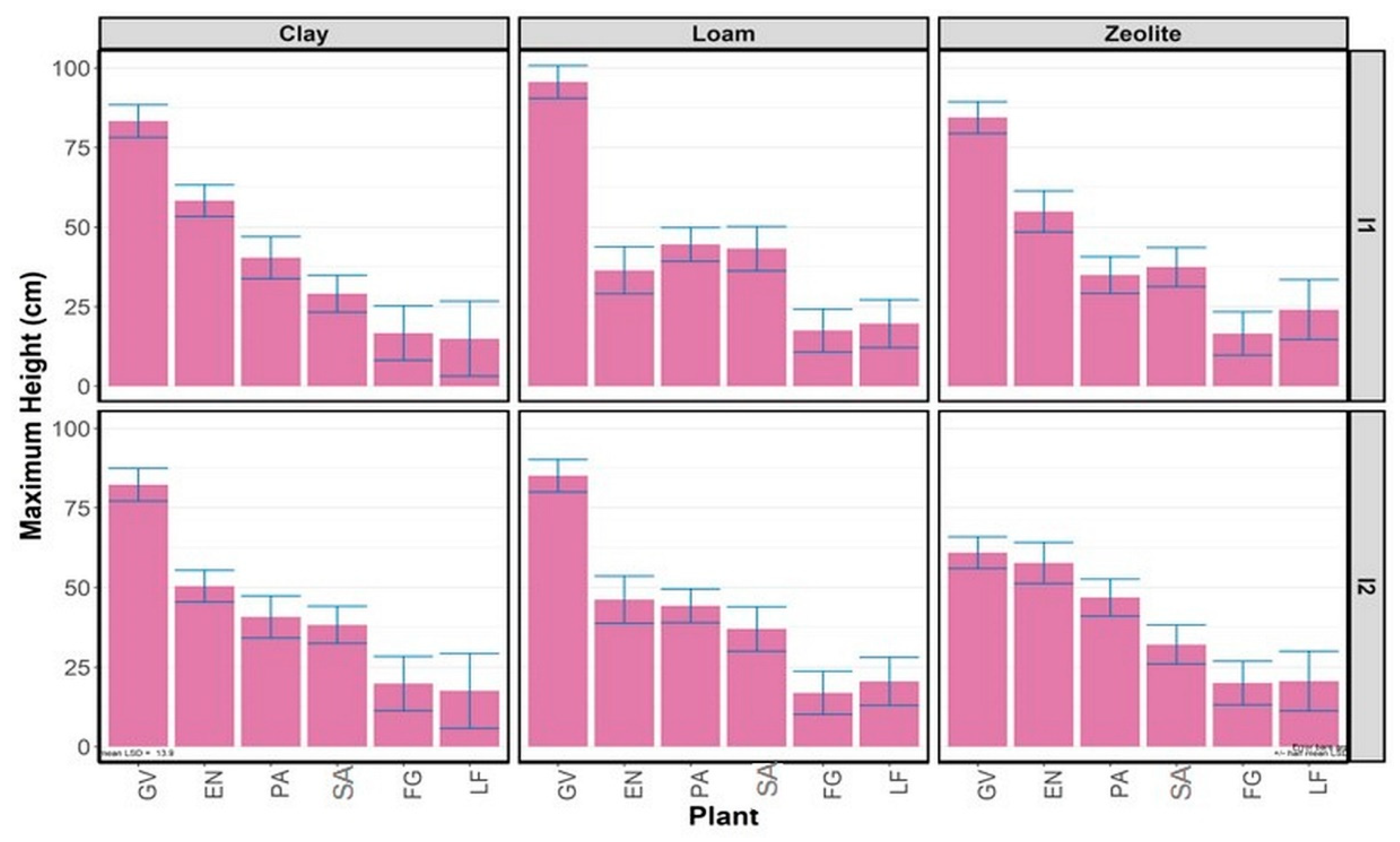
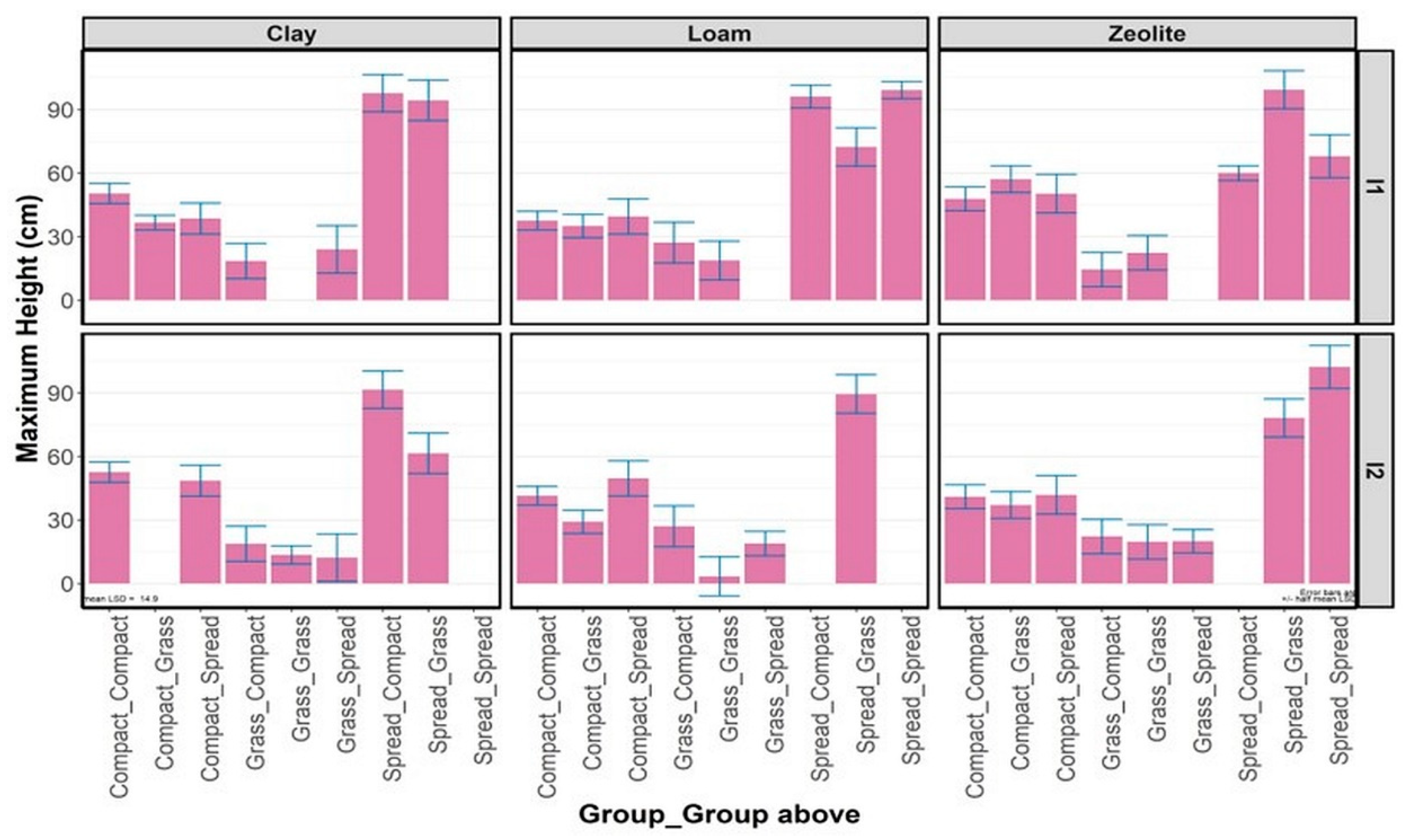
3.2.1. Maximum Plant Height
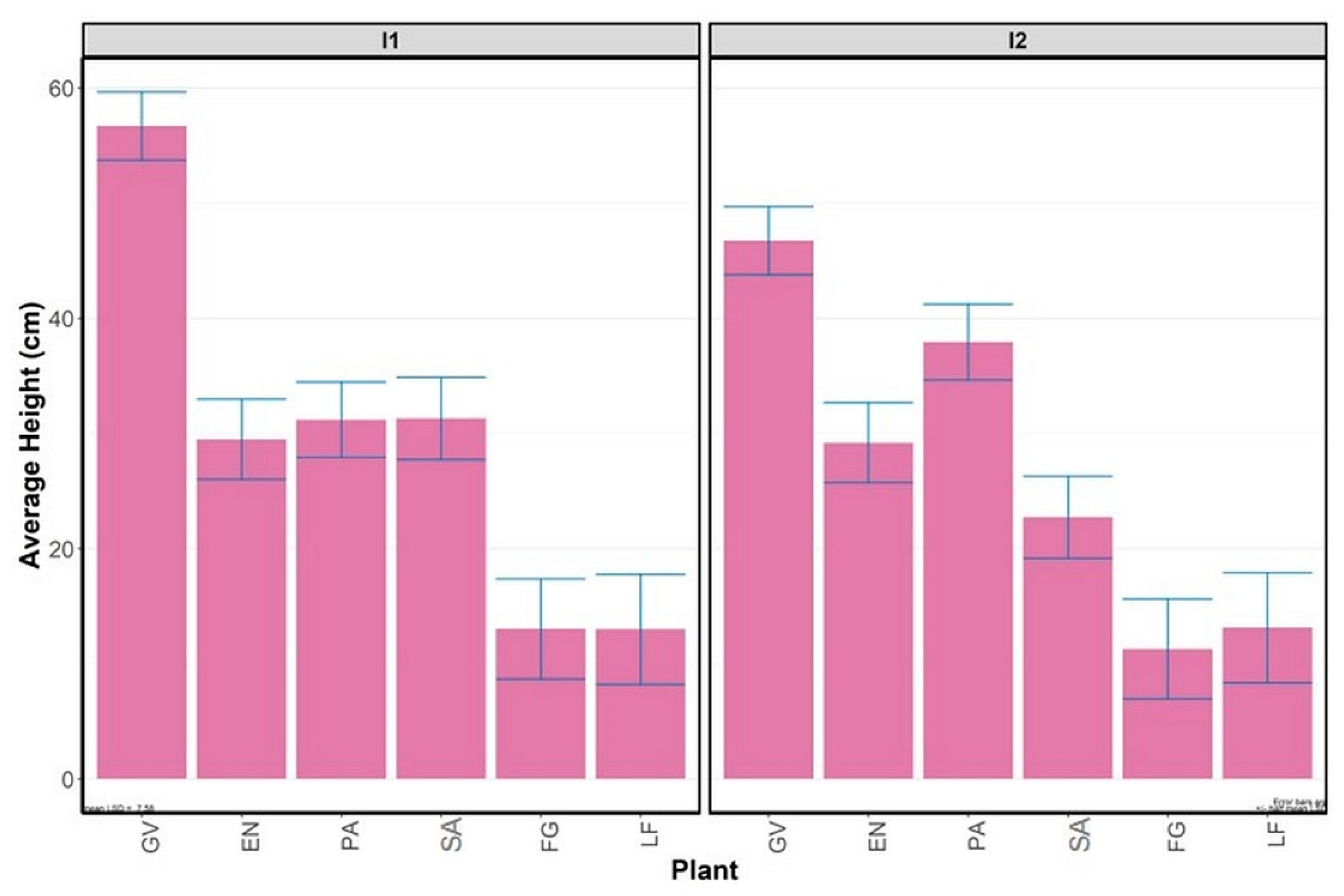
3.2.2. Average Plant Height
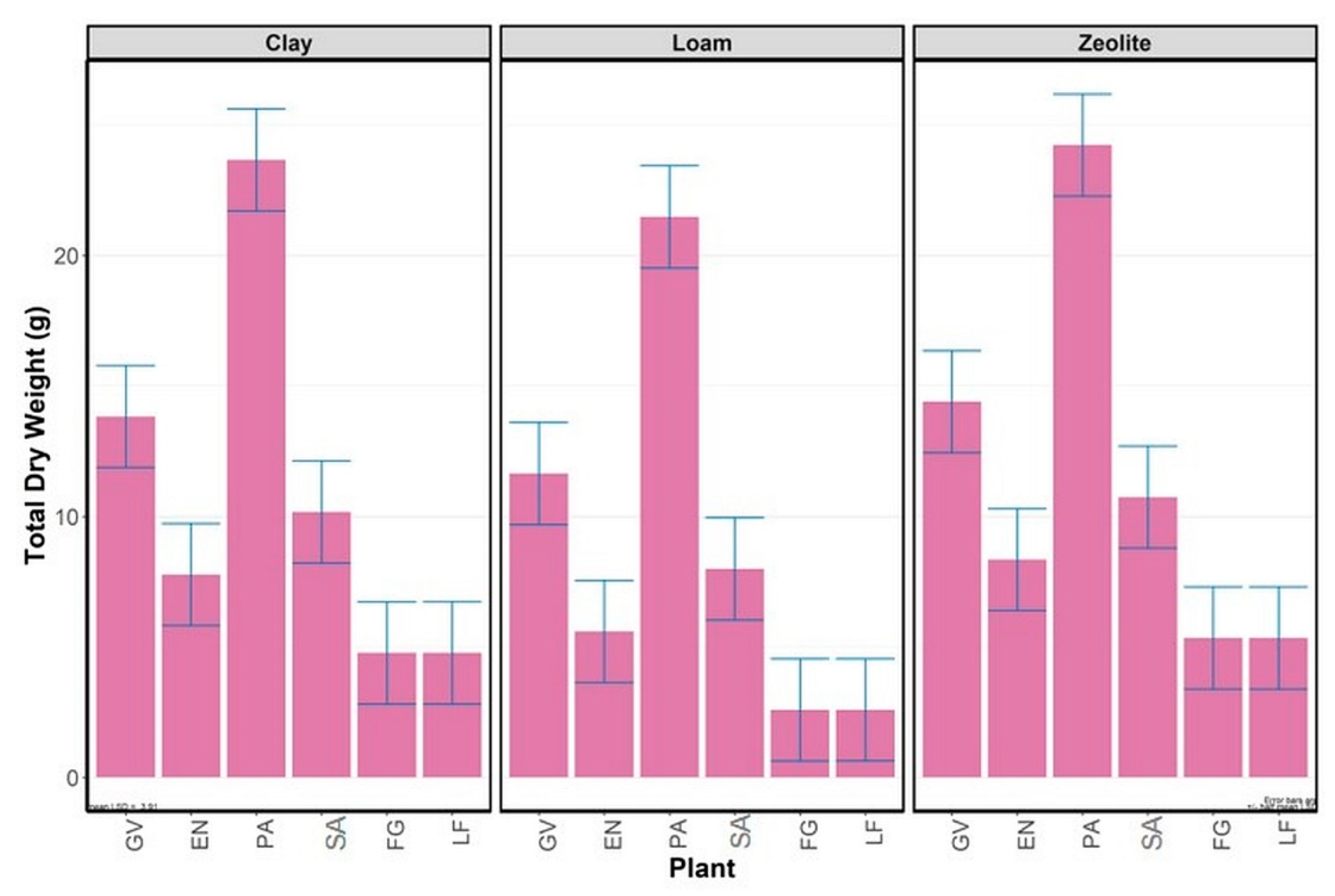
3.3. Dry Weight
3.4. Additional Observations
3.4.1. Issues
3.4.2. Wall Ecology
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Aleksandrowicz, O.; Vuckovic, M.; Kiesel, K.; Mahdavi, A. Current trends in urban heat island mitigation research: Observations based on a comprehensive research repository. Urban Clim. 2017, 21, 1–26. [Google Scholar] [CrossRef]
- Soltani, A.; Sharifi, E. Daily variation of urban heat island effect and its correlations to urban greenery: A case study of Adelaide. Front. Archit. Res. 2017, 6, 529–538. [Google Scholar] [CrossRef]
- Li, D.; Liao, W.; Rigden, A.J.; Liu, X.; Wang, D.; Malyshev, S.; Shevliakova, E. Urban heat island: Aerodynamics or imperviousness? Sci. Adv. 2019, 5, eaau4299. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Köhler, M. Green facades—A view back and some visions. Urban Ecosyst. 2008, 11, 423–436. [Google Scholar] [CrossRef]
- Safikhani, T.; Abdullah, A.M.; Ossen, D.R.; Baharvand, M. A review of energy characteristic of vertical greenery systems. Renew. Sustain. Energy Rev. 2014, 40, 450–462. [Google Scholar] [CrossRef]
- Medl, A.; Stangl, R.; Florineth, F. Vertical greening systems—A review on recent technologies and research advancement. Build. Environ. 2017, 125, 227–239. [Google Scholar] [CrossRef]
- Manso, M.; Castro-Gomes, J. Green wall systems: A review of their characteristics. Renew. Sustain. Energy Rev. 2015, 41, 863–871. [Google Scholar] [CrossRef]
- Othman, A.R.; Sahidin, N. Vertical Greening Façade as Passive Approach in Sustainable Design. Procedia Soc. Behav. Sci. 2016, 222, 845–854. [Google Scholar] [CrossRef]
- Bustami, R.A.; Belusko, M.; Ward, J.; Beecham, S. Vertical greenery systems: A systematic review of research trends. Build. Environ. 2018, 146, 226–237. [Google Scholar] [CrossRef]
- Pérez, G.; Rincón, L.; Vila, A.; González, J.M.; Cabeza, L.F. Green vertical systems for buildings as passive systems for energy savings. Appl. Energy 2011, 88, 4854–4859. [Google Scholar] [CrossRef]
- Razzaghmanesh, M.; Razzaghmanesh, M. Thermal performance investigation of a living wall in a dry climate of Australia. Build. Environ. 2017, 112, 45–62. [Google Scholar] [CrossRef]
- Scarpa, M.; Mazzali, U.; Peron, F. Modeling the energy performance of living walls: Validation against field measurements in temperate climate. Energy Build. 2014, 79, 155–163. [Google Scholar] [CrossRef]
- Koyama, T.; Yoshinaga, M.; Maeda, K.I.; Yamauchi, A. Transpiration cooling effect of climber greenwall with an air gap on indoor thermal environment. Ecol. Eng. 2015, 83, 343–353. [Google Scholar] [CrossRef]
- Wong, N.H.; Kwang Tan, A.Y.; Chen, Y.; Sekar, K.; Tan, P.Y.; Chan, D.; Chiang, K.; Wong, N.C. Thermal evaluation of vertical greenery systems for building walls. Build. Environ. 2010, 45, 663–672. [Google Scholar] [CrossRef]
- Chen, Q.; Li, B.; Liu, X. An experimental evaluation of the living wall system in hot and humid climate. Energy Build. 2013, 61, 298–307. [Google Scholar] [CrossRef]
- Susorova, I.; Angulo, M.; Bahrami, P.; Stephens, B. A model of vegetated exterior facades for evaluation of wall thermal performance. Build. Environ. 2013, 67, 1–13. [Google Scholar] [CrossRef]
- Basher, H.S.; Ahmad, S.S.; Rahman, A.M.A.; Zaman, N.Q. The use of edible vertical greenery system to improve thermal performance in tropical climate. J. Mech. Eng. 2016, 13, 57–66. [Google Scholar]
- de Jesus, M.P.; Lourenço, J.M.; Arce, R.M.; Macias, M. Green façades and in situ measurements of outdoor building thermal behaviour. Build. Environ. 2017, 119, 11–19. [Google Scholar] [CrossRef]
- Cuce, E. Thermal regulation impact of green walls: An experimental and numerical investigation. Appl. Energy 2017, 194, 247–254. [Google Scholar] [CrossRef]
- Coma, J.; Pérez, G.; de Gracia, A.; Burés, S.; Urrestarazu, M.; Cabeza, L.F. Vertical greenery systems for energy savings in buildings: A comparative study between green walls and green facades. Build. Environ. 2017, 111, 228–237. [Google Scholar] [CrossRef] [Green Version]
- Cheng, C.Y.; Cheung, K.K.S.; Chu, L.M. Thermal performance of a vegetated cladding system on facade walls. Build. Environ. 2010, 45, 1779–1787. [Google Scholar] [CrossRef]
- Tudiwer, D.; Korjenic, A. The effect of living wall systems on the thermal resistance of the façade. Energy Build. 2017, 135, 10–19. [Google Scholar] [CrossRef]
- He, Y.; Yu, H.; Ozaki, A.; Dong, N.; Zheng, S. An investigation on the thermal and energy performance of living wall system in Shanghai area. Energy Build. 2017, 140, 324–335. [Google Scholar] [CrossRef]
- Madre, F.; Clergeau, P.; Machon, N.; Vergnes, A. Building biodiversity: Vegetated façades as habitats for spider and beetle assemblages. Glob. Ecol. Conserv. 2015, 3, 222–233. [Google Scholar] [CrossRef]
- Cameron, R.W.F.; Taylor, J.E.; Emmett, M.R. What’s ‘cool’ in the world of green façades? How plant choice influences the cooling properties of green walls. Build. Environ. 2014, 73, 198–207. [Google Scholar] [CrossRef]
- Dunnett, N.; Kingsbury, N. Planting Green Roofs and Living Walls; Timber Press Inc.: Portland, OR, USA, 2008; ISBN 13:978-0-88192-911-9. [Google Scholar]
- Perini, K.; Rosasco, P. Cost–benefit analysis for green façades and living wall systems. Build. Environ. 2013, 70, 110–121. [Google Scholar] [CrossRef]
- Mårtensson, L.-M.; Wuolo, A.; Fransson, A.-M.; Emilsson, T. Plant performance in living wall systems in the Scandinavian climate. Ecol. Eng. 2014, 71, 610–614. [Google Scholar] [CrossRef] [Green Version]
- Devecchi, M.; Merlo, F.; Vigetti, A.; Larcher, F. The cultivation of mediterranean aromatic plants on green walls. Acta Hortic. 2013, 999, 243–247. [Google Scholar] [CrossRef]
- Taylor, J.E.; Cameron, R.W.F.; Emmett, M.R. The role of shrubs and climbers on improving thermal performance of brick walls during winter. Acta Hortic. 2014, 1108, 353–359. [Google Scholar] [CrossRef]
- Larcher, F.; Merlo, F.; Devecchi, M. The use of Mediterranean shrubs in green living walls. Agronomic evaluation of Myrtus communis L. Acta Hortic. 2013, 990, 495–500. [Google Scholar] [CrossRef]
- Mårtensson, L.-M.; Fransson, A.-M.; Emilsson, T. Exploring the use of edible and evergreen perennials in living wall systems in the Scandinavian climate. Urban For. Urban Green. 2016, 15, 84–88. [Google Scholar] [CrossRef]
- Nagle, L.; Echols, S.; Tamminga, K. Food production on a living wall: Pilot study. J. Green Build. 2017, 12, 23–38. [Google Scholar] [CrossRef]
- Cakir, A.; Yalcinalp, E.; Dogan, E.; Meral, A. Determination of the Suitability of Some American grapevine rootstocks as a new edible landscape component of vertical gardens. Sustainability 2017, 9, 1275. [Google Scholar] [CrossRef]
- Hasan, M.M.; Karim, A.; Brown, R.J.; Perkins, M.; Joyce, D. Estimation of Energy saving of Commercial building by living wall and green facade in Subtropical climate of Australia. In Proceedings of the 7th International Green Energy Conference & the 1st DNL Conference on Clean Energy, Dalian, China, 28–30 May 2012. [Google Scholar]
- Wong, N.H.; Tan, A.Y.K.; Tan, P.Y.; Sia, A.; Wong, N.C. Perception Studies of Vertical Greenery Systems in Singapore. J. Urban Plan. Dev. 2010, 136, 330–338. [Google Scholar] [CrossRef]
- Blanc, P.; Lalot, V. The Vertical Garden: From Nature to the City; W.W. Norton: New York, NY, USA, 2008; ISBN 9780393732597. [Google Scholar]
- Pérez-Urrestarazu, L.; Blasco-Romero, A.; Fernández-Cañero, R. Media and social impact valuation of a living wall: The case study of the Sagrado Corazon hospital in Seville (Spain). Urban For. Urban Green. 2017, 24, 141–148. [Google Scholar] [CrossRef]
- Veisten, K.; Smyrnova, Y.; Klæboe, R.; Hornikx, M.; Mosslemi, M.; Kang, J. Valuation of green walls and green roofs as soundscape measures: Including monetised amenity values together with noise-attenuation values in a cost-benefit analysis of a green wall affecting courtyards. Int. J. Environ. Res. Public Health 2012, 9, 3770–3778. [Google Scholar] [CrossRef] [PubMed]
- Ottelé, M.; Perini, K.; Fraaij, A.L.A.; Haas, E.M.; Raiteri, R. Comparative life cycle analysis for green façades and living wall systems. Energy Build. 2011, 43, 3419–3429. [Google Scholar] [CrossRef]
- Pan, L.; Chu, L.M. Energy saving potential and life cycle environmental impacts of a vertical greenery system in Hong Kong: A case study. Build. Environ. 2015, 96, 293–300. [Google Scholar] [CrossRef]
- Natarajan, M.; Rahimi, M.; Sen, S.; Mackenzie, N.; Imanbayev, Y. Living wall systems: Evaluating life-cycle energy, water and carbon impacts. Urban Ecosyst. 2014, 18, 1–11. [Google Scholar] [CrossRef]
- Pérez-Urrestarazu, L.; Egea, G.; Franco-Salas, A.; Fernández-Cañero, R. Irrigation Systems Evaluation for Living Walls. J. Irrig. Drain. Eng. 2014, 140, 04013024. [Google Scholar] [CrossRef]
- Kottek, M.; Grieser, J.; Beck, C.; Rudolf, B.; Rubel, F. World Map of the Köppen-Geiger climate classification updated. Meteorol. Z. 2006, 15, 259–263. [Google Scholar] [CrossRef]
- ASHRAE. ASHRAE Standard 169-2013 Climatic Data for Building Design Standards; American Society of Heating, Refrigerating and Air-Conditioning Engineers: Atlanta, GA, USA, 2013; ISBN 1041-2336. [Google Scholar]
- Bureau of Meteorology (BOM). Adelaide (Kent Town) Monthly Climate Statistics. Available online: http://www.bom.gov.au/jsp/ncc/cdio/cvg/av?p_stn_num=023090&p_prim_element_index=0&p_comp_element_index=0&redraw=null&p_display_type=full_statistics_table&normals_years=1981-2010&tablesizebutt=normal (accessed on 3 February 2016).
- Maxim Integrated DS1923 iButton Hygrochron Temperature/Humidity Logger with 8KB Data-Log Memory—Maxim. Available online: https://www.maximintegrated.com/en/products/ibutton/data-loggers/DS1923.html (accessed on 8 July 2019).
- Elmich Pte Ltd. VersiWallTM GP Tray Planter. Available online: https://elmich.com.au/wp-content/uploads/2014/11/Elmich-VersiWall-AU-28-01-16.pdf (accessed on 5 July 2016).
- Jørgensen, L.; Dresbøll, D.B.; Thorup-Kristensen, K. Root growth of perennials in vertical growing media for use in green walls. Sci. Hortic. 2014, 166, 31–41. [Google Scholar] [CrossRef]
- Weinmaster, M. Are Green Walls a “Green” as They Look? An Introduction to the Various Technologies and Ecological Benefits of Green Walls. J. Green Build. 2009, 4, 3–18. [Google Scholar] [CrossRef]
- Reuter, G. Improvement of sandy soils by clay-substrate application. Appl. Clay Sci. 1994, 9, 107–120. [Google Scholar] [CrossRef]
- Meisl, T.; Dubský, M.; Šrámek, F.; Nečas, T. The effect of clay amendment on substrate properties and growth of woody plants. Acta Univ. Agric. Silvic. Mendel. Brun. 2012, 60, 163–170. [Google Scholar] [CrossRef]
- Kolar, M.; Dubský, M.; Šrámek, F.; Pintar, M. The Effect of Natural Zeolite in Peat Based Growing Media on Pelargonium zonale Plants. Eur. J. Hortic. Sci. 2010, 75, 226–230. [Google Scholar]
- Bunt, A.C. Media and Mixes for Container-Grown Plants: A Manual on the Preparation and Use of Growing Media for Pot Plants; Springer: Dordrecht, The Netherlands, 1988; ISBN 978-94-011-7904-1. [Google Scholar]
- A Package for the Computer Generation of Experimental Designs 2013, CycDesigN 5.1; VSN International: Hemel Hempstead, UK, 2013.
- Pérez-Harguindeguy, N.; Diaz, S.; Garnier, E.; Lavorel, S.; Poorter, H.; Jaureguiberry, P.; Bret-Harte, M.S.S.; Cornwell, W.K.K.; Craine, J.M.M.; Gurvich, D.E.E.; et al. New Handbook for standardized measurment of plant functional traits worldwide. Aust. J. Bot. 2013, 61, 167–234. [Google Scholar] [CrossRef]
- Farrell, C.; Mitchell, R.E.; Szota, C.; Rayner, J.P.; Williams, N.S.G. Green roofs for hot and dry climates: Interacting effects of plant water use, succulence and substrate. Ecol. Eng. 2012, 49, 270–276. [Google Scholar] [CrossRef]
- Butler, D.G.; Cullis, B.R.; Gilmour, A.R.; Gogel, B.J. Analysis of Mixed Models for S Language Environments: ASReml-R Reference Manual; DPI Publications: Brisbane, Australia, 2009. [Google Scholar]
- Brien, C.J. asremlPlus: Augments the Use of ASReml-R in Fitting Mixed Models 2017. Available online: http://cran.at.r-project.org/package=asremlPlus (accessed on 28 September 2018).
- R Development Core Team. R: A Language and Environment for Statistical Computing 2017; R Development Core Team: St. Louis, MO, USA, 2017. [Google Scholar]

| Column Row | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | EN-Z-I1 | LF-Z-I1 | PA-Z-I2 | EN-L-I1 | FG-C-I2 | SA-C-I1 | PA-L-I1 | FG-L-I2 | SA-L-I2 | EN-L-I2 | GV-Z-I1 | SA-C-I2 |
| 2 | GV-C-I2 | GV-C-I1 | PA-C-I2 | FG-L-I1 | PA-L-I2 | PA-Z-I2 | LF-L-I1 | EN-C-I1 | SA-Z-I2 | SA-L-I2 | PA-Z-I1 | SA-C-I1 |
| 3 | LF-Z-I2 | GV-Z-I2 | EN-Z-I2 | SA-Z-I1 | EN-L-I1 | FG-L-I1 | FG-C-I1 | PA-C-I2 | LF-L-I2 | GV-L-I1 | SA-C-I2 | GV-C-I1 |
| 4 | FG-C-I2 | EN-Z-I1 | LF-L-I1 | PA-L-I2 | LF-C-I1 | GV-L-I2 | FG-Z-I1 | PA-L-I1 | GV-Z-I2 | EN-C-I1 | EN-L-I2 | FG-L-I1 |
| 5 | SA-C-I1 | PA-C-I2 | SA-L-I1 | FG-C-I1 | GV-L-I2 | EN-Z-I1 | EN-C-I2 | SA-Z-I2 | GV-L-I1 | LF-Z-I2 | GV-C-I2 | LF-Z-I1 |
| 6 | FG-L-I2 | EN-L-I1 | GV-Z-I1 | LF-C-I2 | EN-C-I2 | LF-Z-I1 | PA-C-I2 | LF-Z-I2 | FG-Z-I1 | SA-Z-I2 | SA-L-I1 | EN-L-I2 |
| 7 | FG-L-I1 | SA-C-I1 | LF-C-I2 | GV-C-I1 | FG-Z-I1 | SA-Z-I2 | GV-C-I2 | GV-L-I2 | FG-Z-I2 | EN-L-I1 | LF-L-I2 | PA-Z-I1 |
| 8 | EN-C-I1 | SA-L-I1 | FG-Z-I2 | SA-C-I2 | PA-Z-I1 | SA-L-I2 | GV-Z-I1 | LF-C-I1 | EN-C-I2 | GV-Z-I2 | FG-Z-I1 | FG-L-I2 |
| 9 | PA-C-I1 | LF-C-I2 | PA-L-I1 | PA-Z-I1 | SA-Z-I1 | PA-L-I2 | GV-L-I1 | EN-Z-I2 | EN-Z-I1 | FG-C-I2 | LF-L-I1 | GV-C-I2 |
| 10 | PA-Z-I2 | LF-L-I2 | EN-C-I1 | LF-L-I1 | FG-C-I1 | LF-Z-I2 | FG-L-I2 | PA-C-I1 | SA-L-I1 | SA-Z-I1 | FG-C-I2 | EN-C-I2 |
| 11 | SA-Z-I1 | PA-L-I2 | GV-L-I1 | EN-Z-I2 | FG-Z-I2 | PA-C-I1 | PA-Z-I2 | SA-L-I2 | FG-C-I1 | LF-C-I1 | LF-C-I2 | GV-L-I2 |
| 12 | EN-L-I2 | SA-C-I2 | LF-L-I2 | GV-Z-I2 | GV-Z-I1 | LF-C-I1 | EN-Z-I2 | GV-C-I1 | PA-L-I1 | PA-C-I1 | LF-Z-I1 | FG-Z-I2 |
| Plant Species | Clay | Loam | Zeolite | Total | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| I1 | I2 | Total | I1 | I2 | Total | I1 | I2 | Total | ||
| GV | 4 | 4 | 8 | 4 | 4 | 8 | 4 | 4 | 8 | 24 |
| EN | 4 | 4 | 8 | 2 | 3 | 5 | 4 | 3 | 7 | 20 |
| PA | 3 | 4 | 7 | 4 | 4 | 8 | 4 | 3 | 7 | 22 |
| SA | 4 | 3 | 7 | 2 | 3 | 5 | 3 | 3 | 6 | 18 |
| FG | 1 | 4 | 5 | 2 | 3 | 5 | 2 | 3 | 5 | 15 |
| LF | 1 | 3 | 4 | 2 | 2 | 4 | 1 | 2 | 3 | 11 |
| Total | 17 | 22 | 39 | 16 | 19 | 35 | 18 | 18 | 36 | 110 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bustami, R.A.; Brien, C.; Ward, J.; Beecham, S.; Rawlings, R. A Statistically Rigorous Approach to Experimental Design of Vertical Living Walls for Green Buildings. Urban Sci. 2019, 3, 71. https://doi.org/10.3390/urbansci3030071
Bustami RA, Brien C, Ward J, Beecham S, Rawlings R. A Statistically Rigorous Approach to Experimental Design of Vertical Living Walls for Green Buildings. Urban Science. 2019; 3(3):71. https://doi.org/10.3390/urbansci3030071
Chicago/Turabian StyleBustami, Rosmina A., Chris Brien, James Ward, Simon Beecham, and Robyn Rawlings. 2019. "A Statistically Rigorous Approach to Experimental Design of Vertical Living Walls for Green Buildings" Urban Science 3, no. 3: 71. https://doi.org/10.3390/urbansci3030071
APA StyleBustami, R. A., Brien, C., Ward, J., Beecham, S., & Rawlings, R. (2019). A Statistically Rigorous Approach to Experimental Design of Vertical Living Walls for Green Buildings. Urban Science, 3(3), 71. https://doi.org/10.3390/urbansci3030071






