Effects of Transcranial Direct Current Stimulation on Cycling Time Trial Performance and Prefrontal Cortex Activation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Participants
2.3. Anthropometric and Body Composition Measurements
2.4. Exercise Testing and Screening
2.5. Time Trial Task
2.6. Blood Lactate and Heart Rate
2.7. Halo Sport Procedures
2.8. Functional Infrared Spectroscopy Recording Procedures
2.9. Muscle Oxygenation Procedures
2.10. Statistical Analysis
3. Results
3.1. Time to Completion
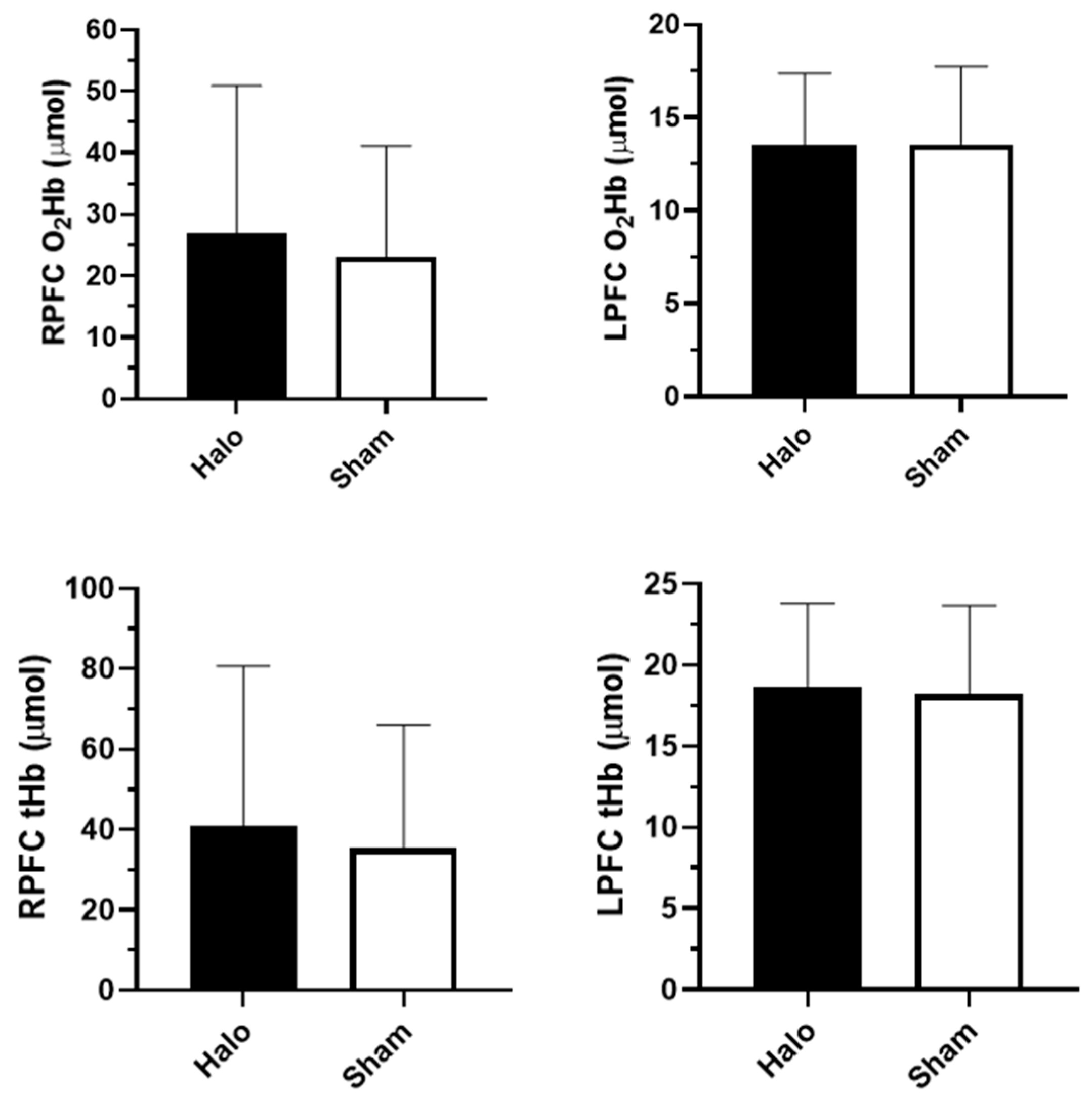
3.2. Prefrontal Cortex Oxygenation
3.3. Muscle Oxygenation
3.4. Heart Rate, Blood Lactate and Rating of Perceived Exertion
| Characteristic | Halo | Sham |
|---|---|---|
| BL (mmol/L) | 10.66 ± 2.46 | 9.98 ± 2.88 |
| RPE | 16.20 ± 1.99 | 15.95 ± 2.03 |
| HR (bpm) | 163.5 ± 17.2 | 160.9 ± 20.1 |
| Time (min) | 17.58 ± 1.88 | 17.67 ± 1.92 |
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Amann, M. Central and peripheral fatigue: Interaction during cycling exercise in humans. Med. Sci. Sports Exerc. 2011, 43, 2039–2045. [Google Scholar] [CrossRef]
- Neyroud, D.; Vallotton, A.; Millet, G.Y.; Kayser, B.; Place, N. The effect of muscle fatigue on stimulus intensity requirements for central and peripheral fatigue quantification. Eur. J. Appl. Physiol. 2014, 114, 205–215. [Google Scholar] [CrossRef] [Green Version]
- Schillings, M.L.; Hoefsloot, W.; Stegeman, D.F.; Zwarts, M.J. Relative contributions of central and peripheral factors to fatigue during a maximal sustained effort. Eur. J. Appl. Physiol. 2003, 90, 562–568. [Google Scholar] [CrossRef]
- Taylor, J.L.; Amann, M.; Duchateau, J.; Meeusen, R.; Rice, C.L. Neural contributions to muscle fatigue: From the brain to the muscle and back again. Med. Sci. Sports Exerc. 2016, 48, 2294. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Angius, L.; Hopker, J.G.; Marcora, S.M.; Mauger, A.R. The effect of transcranial direct current stimulation of the motor cortex on exercise-induced pain. Eur. J. Appl. Physiol. 2015, 115, 2311–2319. [Google Scholar] [CrossRef] [PubMed]
- Rossini, P.M.; Burke, D.; Chen, R.; Cohen, L.G.; Daskalakis, Z.; Di Iorio, R.; Di Lazzaro, V.; Ferreri, F.; Fitzgerald, P.B.; George, M.S.; et al. Non-invasive electrical and magnetic stimulation of the brain, spinal cord, roots and peripheral nerves: Basic principles and procedures for routine clinical and research application: An updated report from an I.F.C.N. Committee. Clin. Neurophysiol. 2015, 126, 1071–1107. [Google Scholar] [CrossRef]
- Huang, L.; Deng, Y.; Zheng, X.; Liu, Y. Transcranial direct current stimulation with halo sport enhances repeated sprint cycling and cognitive performance. Front. Physiol. 2019, 10, 1–7. [Google Scholar] [CrossRef]
- Baldari, C.; Buzzachera, C.F.; Vitor-Costa, M.; Gabardo, J.M.; Bernardes, A.G.; Altimari, L.R.; Guidetti, L. Effects of transcranial direct current stimulation on psychophysiological responses to maximal incremental exercise test in recreational endurance runners. Front. Psychol. 2018, 9, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mesquita, P.H.C.; Lage, G.M.; Franchini, E.; Romano-Silva, M.A.; Albuquerque, M.R. Bi-hemispheric anodal transcranial direct current stimulation worsens taekwondo-related performance. Hum. Mov. Sci. 2019, 66, 578–586. [Google Scholar] [CrossRef]
- Okano, A.H.; Fontes, E.B.; Montenegro, R.A.; De Tarso Veras Farinatti, P.; Cyrino, E.S.; Li, L.M.; Bikson, M.; Noakes, T.D. Brain stimulation modulates the autonomic nervous system, rating of perceived exertion and performance during maximal exercise. Br. J. Sports Med. 2015, 49, 1213–1218. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rooks, C.R.; Thom, N.J.; McCully, K.K.; Dishman, R.K. Effects of incremental exercise on cerebral oxygenation measured by near-infrared spectroscopy: A systematic review. Prog. Neurobiol. 2010, 92, 134–150. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, A.; Sparling, P.B. Endurance exercise selectively impairs prefrontal-dependent cognition. Brain Cogn. 2004, 55, 516–524. [Google Scholar] [CrossRef]
- Fox, E.L. A simple, accurate technique for predicting maximal aerobic power. J. Appl. Physiol. 1973, 35, 914–916. [Google Scholar] [CrossRef]
- Uchida, M.C.; Teixeira, L.F.M.; Godoi, V.J.; Marchetti, P.H.; Conte, M.; Coutts, A.J.; Bacurau, R.F.P. Does the timing of measurement alter session-RPE in boxers? J. Sports Sci. Med. 2014, 13, 59. [Google Scholar] [PubMed]
- Park, S.B.; Sung, D.J.; Kim, B.; Kim, S.J.; Han, J.K. Transcranial Direct Current Stimulation of motor cortex enhances running performance. PLoS ONE 2019, 14, e0211902. [Google Scholar] [CrossRef] [Green Version]
- Smith, K.J.; Billaut, F. Influence of cerebral and muscle oxygenation on repeated-sprint ability. Eur. J. Appl. Physiol. 2010, 109, 989–999. [Google Scholar] [CrossRef]
- Orihuela-Espina, F.; Leff, D.R.; James, D.R.C.; Darzi, A.W.; Yang, G.Z. Quality control and assurance in functional near infrared spectroscopy (fNIRS) experimentation. Phys. Med. Biol. 2010, 55, 3701–3724. [Google Scholar] [CrossRef]
- Herold, F.; Wiegel, P.; Scholkmann, F.; Müller, N. Applications of Functional Near-Infrared Spectroscopy (fNIRS) Neuroimaging in Exercise–Cognition Science: A Systematic, Methodology-Focused Review. J. Clin. Med. 2018, 7, 466. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Billaut, F.; Buchheit, M. Repeated-sprint performance and vastus lateralis oxygenation: Effect of limited O 2 availability. Scand. J. Med. Sci. Sports 2013, 23, e185–e193. [Google Scholar] [CrossRef]
- Paquette, M.; Bieuzen, F.; Billaut, F. Sustained muscle deoxygenation vs sustained high VO2 during high-intensity interval training in sprint canoe-kayak. Front. Sport. Act. Living 2019, 1, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodriguez, R.F.; Townsend, N.E.; Aughey, R.J.; Billaut, F. Influence of averaging method on muscle deoxygenation interpretation during repeated-sprint exercise. Scand. J. Med. Sci. Sports 2018, 28, 2263–2271. [Google Scholar] [CrossRef]
- Ferrari, M.; Muthalib, M.; Quaresima, V. The use of near-infrared spectroscopy in understanding skeletal muscle physiology: Recent developments. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2011, 369, 4577–4590. [Google Scholar] [CrossRef] [PubMed]
- McManus, C.J.; Collison, J.; Cooper, C.E. Performance comparison of the MOXY and PortaMon near-infrared spectroscopy muscle oximeters at rest and during exercise. J. Biomed. Opt. 2018, 23, 15007. [Google Scholar] [CrossRef] [PubMed]
- Barwood, M.J.; Butterworth, J.; Goodall, S.; House, J.R.; Laws, R.; Nowicky, A.; Corbett, J. The effects of direct current stimulation on exercise performance, pacing and perception in temperate and hot environments. Brain Stimul. 2016, 9, 842–849. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lattari, E.; de Oliveira, B.S.; Oliveira, B.R.R.; de Mello Pedreiro, R.C.; Machado, S.; Neto, G.A.M. Effects of transcranial direct current stimulation on time limit and ratings of perceived exertion in physically active women. Neurosci. Lett. 2018, 662, 12–16. [Google Scholar] [CrossRef]
- Angius, L.; Mauger, A.R.; Hopker, J.; Pascual-Leone, A.; Santarnecchi, E.; Marcora, S.M. Bilateral extracephalic transcranial direct current stimulation improves endurance performance in healthy individuals. Brain Stimul. 2018, 11, 108–117. [Google Scholar] [CrossRef] [Green Version]
- Vitor-Costa, M.; Okuno, N.M.; Bortolotti, H.; Bertollo, M.; Boggio, P.S.; Frengi, F.; Altimari, L.R. Improving Cycling Performance: Transcranial Direct Current Stimulation Increases Time to Exhaustion in Cycling. PLoS ONE 2015, 10, e0144916. [Google Scholar] [CrossRef]
- Thomas, R.; Stephane, P. Prefrontal cortex oxygenation and neuromuscular responses to exhaustive exercise. Eur. J. Appl. Physiol. 2008, 102, 153–163. [Google Scholar] [CrossRef]
- Muthalib, M.; Kan, B.; Nosaka, K.; Perrey, S. Effects of transcranial direct current stimulation of the motor cortex on prefrontal cortex activation during a neuromuscular fatigue task: An fNIRS study. In Oxygen Transport to Tissue XXXV; Springer: Berlin/Heidelberg, Germany, 2013; pp. 73–79. [Google Scholar]
- Holgado, D.; Zandonai, T.; Ciria, L.F.; Zabala, M.; Hopker, J.; Sanabria, D. Transcranial direct current stimulation (tDCS) over the left prefrontal cortex does not affect time-trial self-paced cycling performance: Evidence from oscillatory brain activity and power output. PLoS ONE 2019, 14, e0210873. [Google Scholar] [CrossRef] [Green Version]
- Belardinelli, R.; Barstow, T.J.; Porszasz, J.; Wasserman, K. Changes in skeletal muscle oxygenation during incremental exercise measured with near infrared spectroscopy. / Modifications de l’ oxygenation du muscle squelettique lors d’ un exercice progressif, mesurees par spectroscopie infrarouge. Eur. J. Appl. Physiol. Occup. Physiol. 1995, 70, 487–492. [Google Scholar] [CrossRef]


| Age (Years) | Height (cm) | Weight (kg) | BMI (kg/m2) | Body Fat (%) | Estimated VO2 Max (mL/kg/min) |
|---|---|---|---|---|---|
| 25.5 ± 7.8 | 181.7 ± 5.9 | 85.9 ± 12.3 | 25.7 ± 3.0 | 15.3 ± 7.0 | 41.5 ± 5.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Auten, A.; Cavey, K.; Reed, J.; Dolgener, F.; Moriarty, T. Effects of Transcranial Direct Current Stimulation on Cycling Time Trial Performance and Prefrontal Cortex Activation. Sci 2021, 3, 32. https://doi.org/10.3390/sci3030032
Auten A, Cavey K, Reed J, Dolgener F, Moriarty T. Effects of Transcranial Direct Current Stimulation on Cycling Time Trial Performance and Prefrontal Cortex Activation. Sci. 2021; 3(3):32. https://doi.org/10.3390/sci3030032
Chicago/Turabian StyleAuten, Abi, Kristina Cavey, Jacob Reed, Forrest Dolgener, and Terence Moriarty. 2021. "Effects of Transcranial Direct Current Stimulation on Cycling Time Trial Performance and Prefrontal Cortex Activation" Sci 3, no. 3: 32. https://doi.org/10.3390/sci3030032
APA StyleAuten, A., Cavey, K., Reed, J., Dolgener, F., & Moriarty, T. (2021). Effects of Transcranial Direct Current Stimulation on Cycling Time Trial Performance and Prefrontal Cortex Activation. Sci, 3(3), 32. https://doi.org/10.3390/sci3030032







