LC–NMR for Natural Product Analysis: A Journey from an Academic Curiosity to a Robust Analytical Tool
Abstract
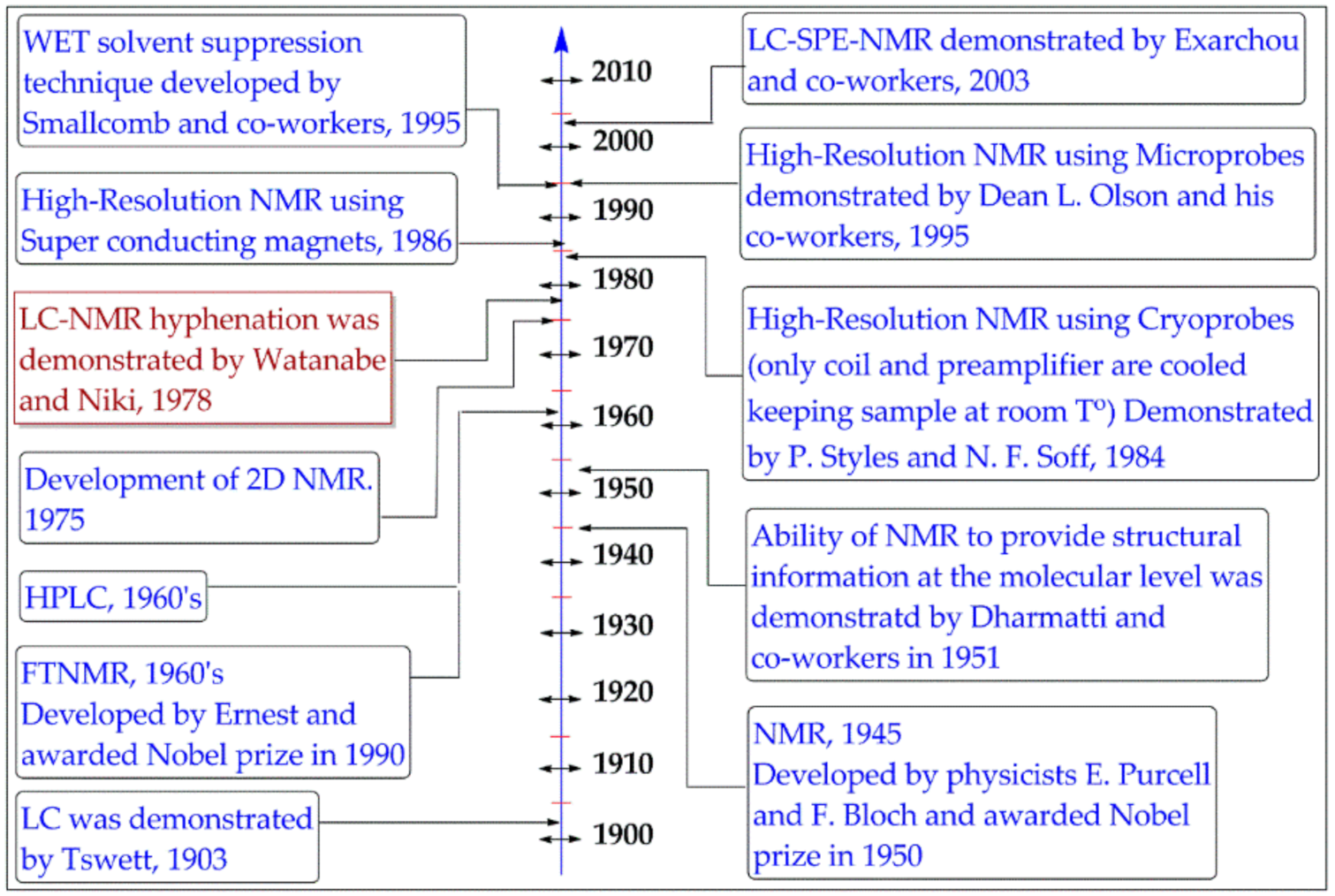
1. Introduction
2. LC–NMR Instrumentation and Principal Modes of Operations
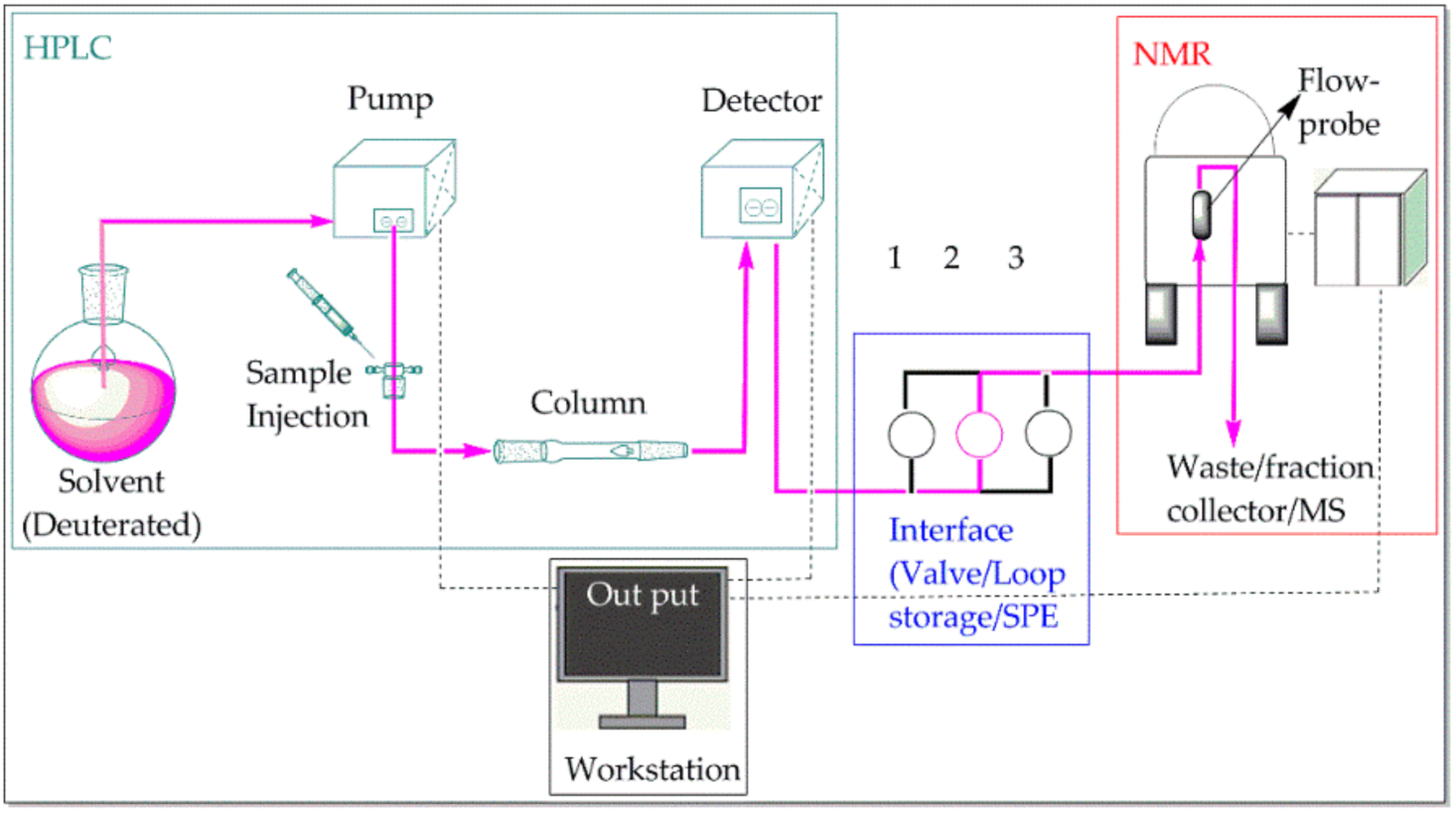
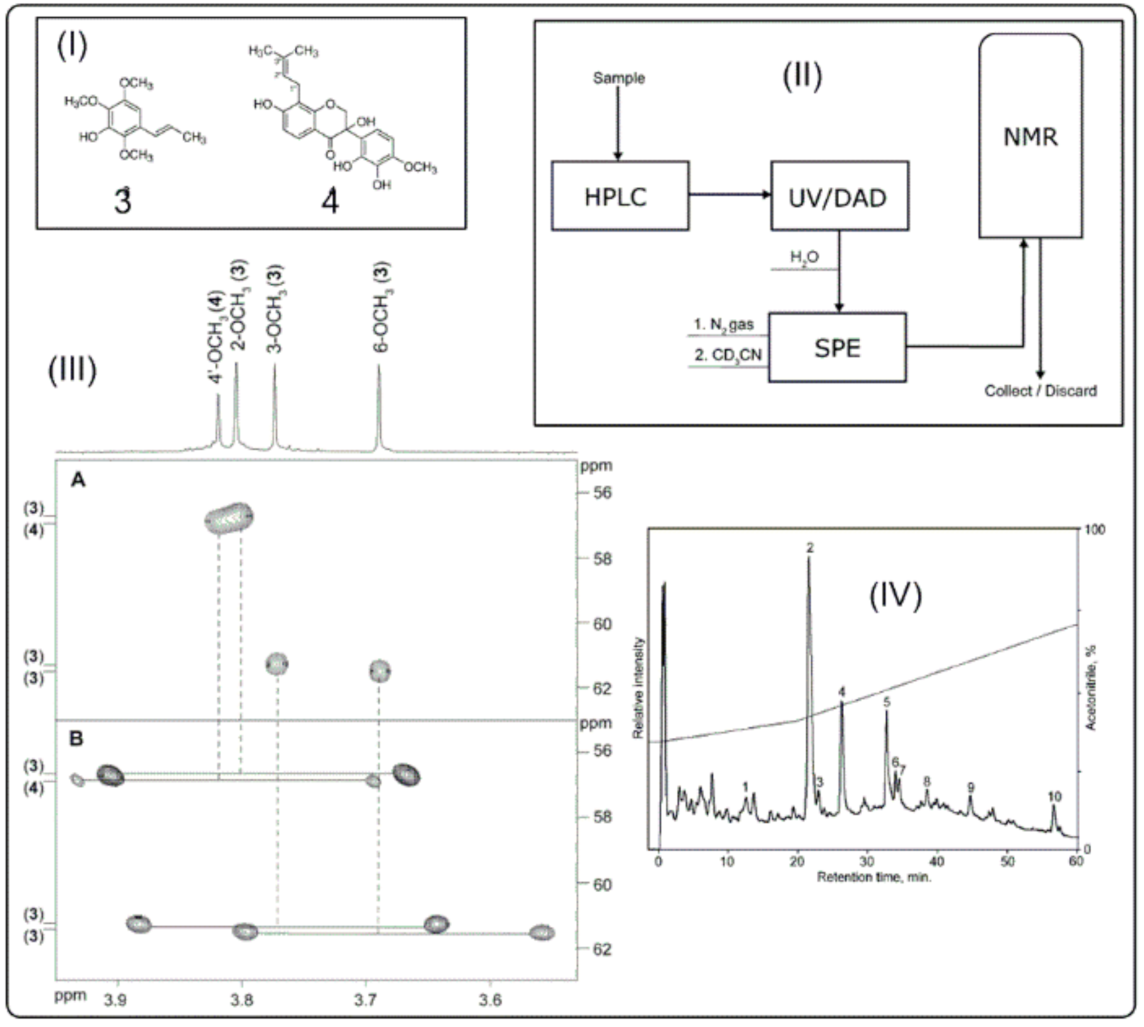
2.1. LC–NMR Instrumentation
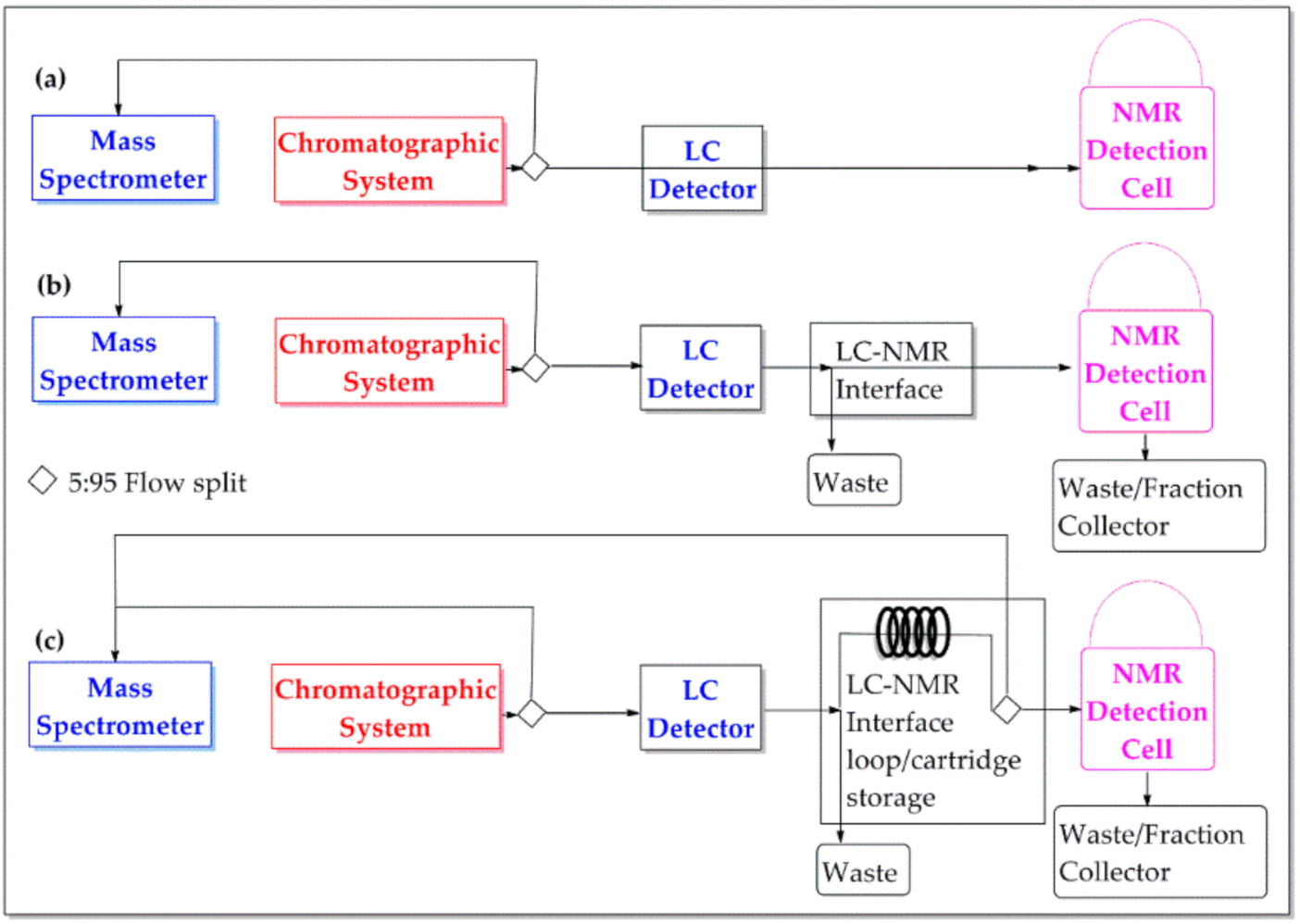
2.2. LC–NMR Principal Modes of Operation
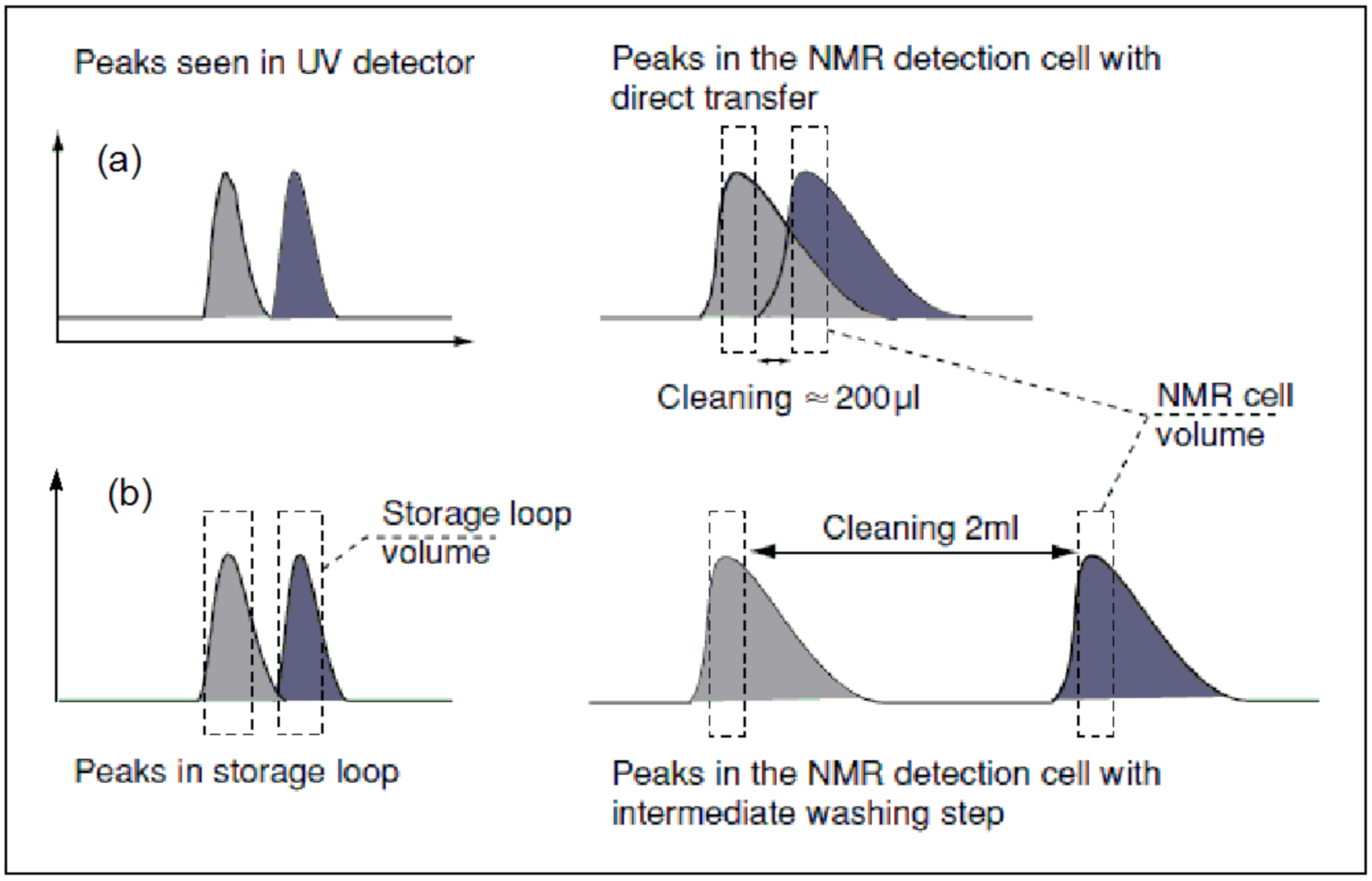
2.2.1. Online-Flow Mode
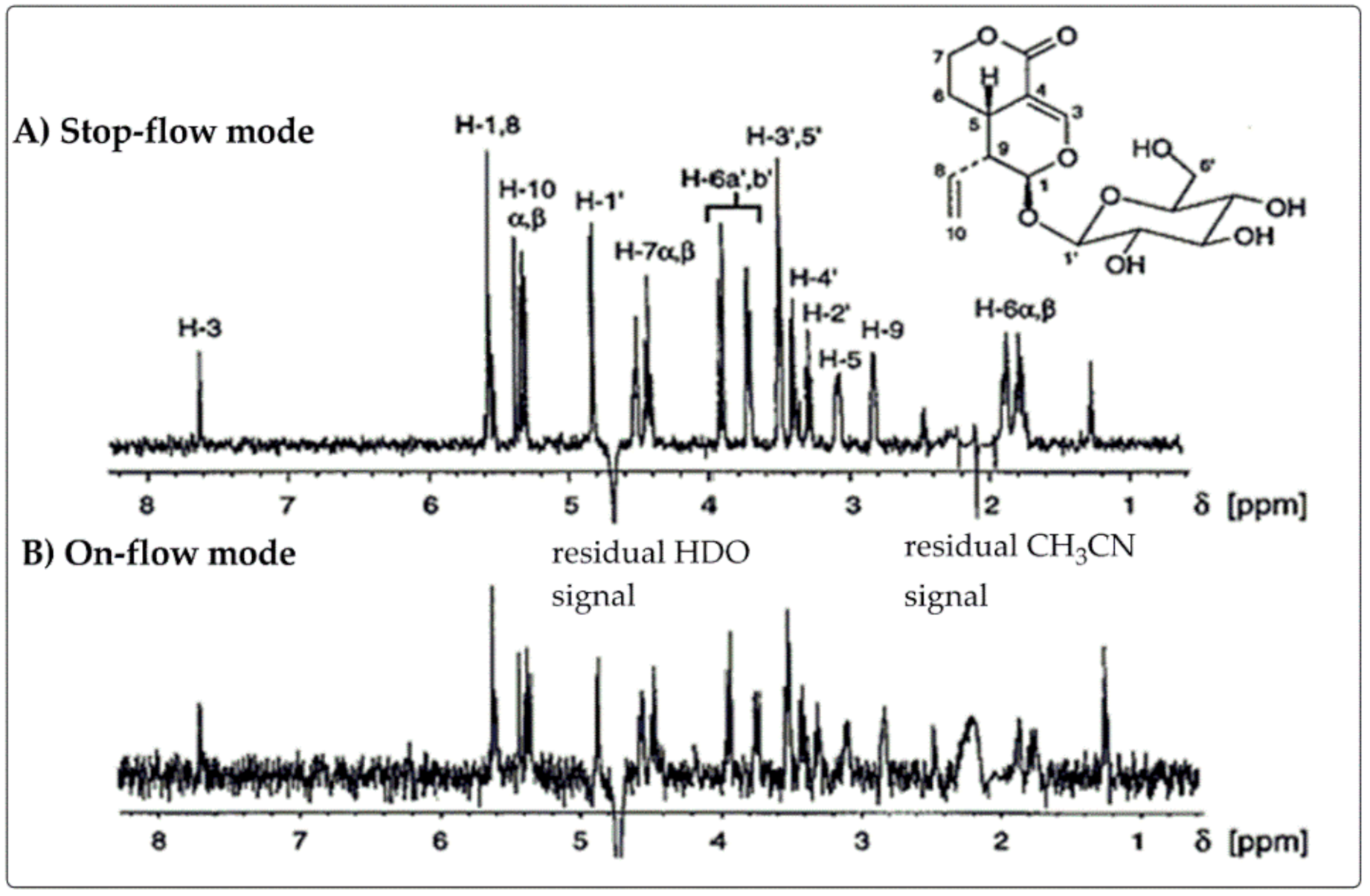
2.2.2. Stop-Flow Mode
2.2.3. Loop/Cartridge Storage Mode
3. Technological Progresses and Limitations of LC–NMR
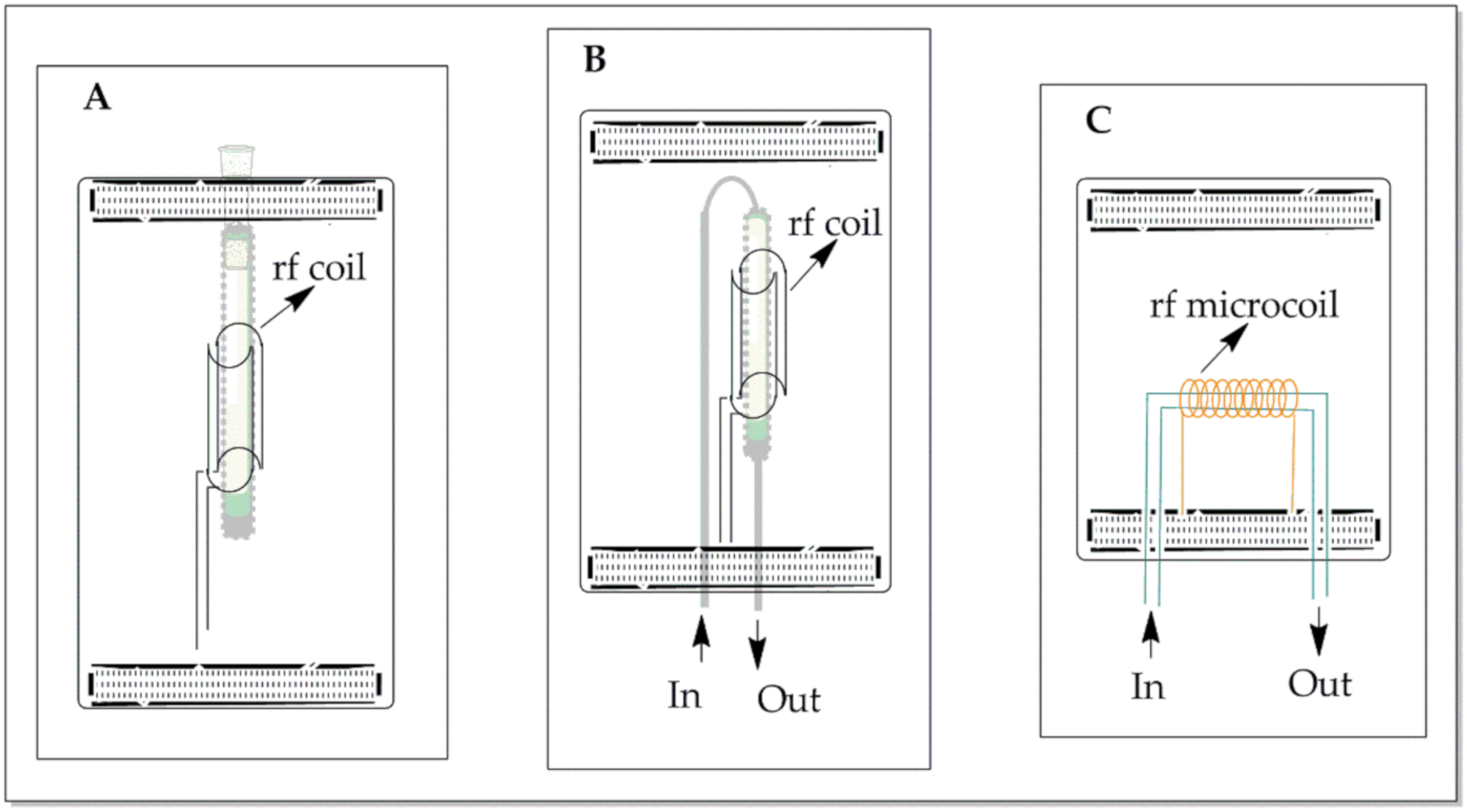
3.1. NMR Flow-Probe Design
3.2. Solvent Suppression
4. Application of LC–NMR in Natural Product Identification
5. Conclusions and Future Outlooks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Wolfender, J.; Rodriguez, S.; Hostettmann, K.; Hiller, W. Liquid Chromatography/Ultra Violet/Mass Spectrometric and Liquid Chromatography/Nuclear Magnetic Resonance Spectroscopic Analysis of Crude Extracts of Gentianaceae Species. Phytochem. Anal. 1997, 8, 97–104. [Google Scholar] [CrossRef]
- Ettre, L.S. Key Moments in the Evolution of Liquid Chromatography. J. Chromatogr. A 1990, 535, 3–12. [Google Scholar] [CrossRef]
- Purcell, H.C.; Torrey, R.V.; Pound, E.M. Resonance Absorption by Nuclear Magnetic Moments in a Solid. Phys. Rev. 1945, 69, 37–38. [Google Scholar] [CrossRef]
- Bloch, F. Nuclear Induction. Phy. Rev. 1946, 70, 460–474. [Google Scholar] [CrossRef]
- Arnold, J.T.; Dharmatti, S.S.; Packard, M.E. Chemical Effects on Nuclear Induction Signals from Organic Compounds. J. Chem. Phys. 1951, 19, 507. [Google Scholar] [CrossRef]
- Wolfender, J.; Rodriguez, S.; Hostettmann, K. Liquid Chromatography Coupled to Mass Spectrometry and Nuclear Magnetic Resonance Spectroscopy for the Screening of Plant Constituents. J. Chromatogr. A 1998, 794, 299–316. [Google Scholar] [CrossRef]
- Bringmann, G.; Messer, K.; Wohlfarth, M.; Dumbuya, K.; Ru, M. HPLC-CD On-Line Coupling in Combination with HPLC-NMR and HPLC-MS/MS for the Determination of the Full Absolute Stereostructure of New Metabolites in Plant Extracts. Anal. Chem. 1999, 71, 2678–2686. [Google Scholar] [CrossRef]
- Albert, K.; Bayer, E. High-Performance Liquid Chromatography-Nuclear Magnetic Resonance on-Line Coupling. TrAC-Trends Anal. Chem. 1988, 7, 283–288. [Google Scholar] [CrossRef]
- Garo, E.; Wolfender, J.L.; Hostettmann, K.; Hiller, W.; Antus, S.; Mavi, S. Prenylated Flavanones from Monotes Engleri: On-Line Structure Elucidation by LC/UV/NMR. Helv. Chim. Acta 1998, 81, 754–763. [Google Scholar] [CrossRef]
- Vogler, B.; Klaiber, I.; Roos, G.; Walter, C.U.; Hiller, W.; Sandor, P.; Kraus, W. Combination of LC-MS and LC-NMR as a Tool for the Structure Determination of Natural Products. J. Nat. Prod. 1998, 61, 75–78. [Google Scholar] [CrossRef]
- Albert, K.; Dachtler, M.; Glaser, T.; Ha, H.; Lacker, T.; Braumann, U. On-Line Coupling of Separation Techniques to NMR. J. High. Resol. Chromatogr. 1999, 22, 135–143. [Google Scholar] [CrossRef]
- Daniel, L.; Norwood, J.O.; Mullis, T.N.F. Hyphenated Techniques. In HPLC Method Development for Pharmaceutical Analysis; Ahuja, S., Rasmussen, H., Eds.; Elsevier: Chapel Hill, NC, USA, 2007; pp. 189–235. [Google Scholar]
- Takashi Tokunaga, M.O. Recent Progress in LC-NMR. Sumitomo Kagaku 2010, 2, 40–48. [Google Scholar]
- Gonnella, N.C. LC-NMR: Expanding the Limits of Structure Elucidation; Grinberg, N., Ed.; Taylor & Francis Group: Boca Raton, FL, USA, 2013; Volume 105. [Google Scholar]
- Wolfender, J.L.; Ndjoko, K.; Hostettmann, K. The Potential of LC-NMR in Phytochemical Analysis. Phytochem. Anal. 2001, 12, 2–22. [Google Scholar] [CrossRef]
- Albert, K. On-Line LC–NMR and Related Techniques; Albert, K., Ed.; John Wiley & Sons: Chichester, UK, 2002. [Google Scholar]
- Gil, A.M.; Duarte, I.F.; Godejohann, M.; Braumann, U.; Maraschin, M.; Spraul, M. Characterization of the Aromatic Composition of Some Liquid Foods by Nuclear Magnetic Resonance Spectrometry and Liquid Chromatography with Nuclear Magnetic Resonance and Mass Spectrometric Detection. Anal. Chim. Acta 2003, 488, 35–51. [Google Scholar] [CrossRef]
- Graça, G.; Duarte, I.F.; Goodfellow, J.B.; Carreira, I.M.; Couceiro, A.B.; Domingues, M.D.R.; Spraul, M.; Tseng, L.H.; Gil, A.M. Metabolite Profiling of Human Amniotic Fluid by Hyphenated Nuclear Magnetic Resonance. Anal. Chem. 2008, 80, 6085–6092. [Google Scholar] [CrossRef]
- Huang, X.; Powers, R.; Tymiak, A.; Espina, R.; Roongta, V. Introduction to NMR and Its Application in Metabolite Structure Determination. In Drug Metabolism in Drug Design and Development: Basic Concepts and Practice; Zhang, D., Zhu, M., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2008; pp. 369–405. [Google Scholar]
- Exarchou, V.; Godejohann, M.; van Beek, T.A.; Gerothanassis, I.P.; Vervoort, J. LC-UV-Solid-Phase Extraction-NMR-MS Combined with a Cryogenic Flow Probe and Its Application to the Identification of Compounds Present in Greek Oregano. Anal. Chem. 2003, 75, 6288–6294. [Google Scholar] [CrossRef]
- Lambert, M.; Stærk, D.; Honore, S.; Sairafianpour, M.; Jaroszewski, J.W. Rapid Extract Dereplication Using HPLC-SPE-NMR: Analysis of Isoflavonoids from Smirnowia Iranica. J. Nat. Prod. 2005, 68, 1500–1509. [Google Scholar] [CrossRef]
- Watanabe, N.; Niki, E. Direct-Coupling of FT-NMR to High Performance Liquid Chromatography. Proc. Jpn. Acad. Ser. B 1978, 54, 194–199. [Google Scholar] [CrossRef]
- Bayer, E.; Albert, K.; Nleder, M.; Grom, E. On-Line Coupling of Liquid Chromatography and High-Field Nuclear Magnetic Resonance Spectrometry. Anal. Chem. 1982, 54, 1747–1750. [Google Scholar] [CrossRef]
- Smallcombe, S.H.; Patt, S.L.; Keifer, P.A. WET Solvent Suppression and Its Applications to LC NMR and High-Resolution NMR Spectroscopy. J. Magn. Res. Ser. A 1995, 2, 295–303. [Google Scholar] [CrossRef]
- Bayer, E.; Albert, K.; Nieder, M.; Grom, E.; Keller, T. On-Line Coupling of High-Performance Liquid Chromato-Graphy and Nuclear Magnetic Resonance. J. Chromatogr. A 1979, 186, 497–507. [Google Scholar] [CrossRef]
- Webb, A.G. Microcoil Nuclear Magnetic Resonance Soectroscopy. In NMR Spectroscopy in Pharmaceutical Analysis; Holzgrabe, U., Wawer, I., Diehl, B., Eds.; Elsevier: Köln, Germany, 2008; pp. 84–127. [Google Scholar]
- Kovacs, H.; Moskau, D.; Spraul, M. Cryogenically Cooled Probes-a Leap in NMR Technology. Prog. Nucl. Magn. Reson. Spectrosc. 2005, 46, 131–155. [Google Scholar] [CrossRef]
- Mukhopadhyay, R. Liquid NMR Probes: Oh so Many Choices. Anal. Chem. 2007, 7959–7963. [Google Scholar] [CrossRef]
- Dorn, H.C. 1H-NMR: A New Detector for Liquid Chromatography. Anal. Chem. 1984, 56, 747A–758A. [Google Scholar] [CrossRef]
- Bobzin, S.C.; Yang, S.; Kasten, T.P. Application of Liquid Chromatography-Nuclear Magnetic Resonance Spectroscopy to the Identification of Natural Products. J. Chromatogr. B 2000, 748, 259–267. [Google Scholar] [CrossRef]
- Wolfender, J.; Queiroz, E.F.; Hostettmann, K. Phytochemistry in the Microgram Domain—A LC–NMR Perspective. Magn. Reson. Chem. 2005, 43, 697–709. [Google Scholar] [CrossRef]
- Olson, D.L.; Norcross, J.A.; Neil-johnson, M.O.; Molitor, P.F.; Detlefsen, D.J.; Wilson, A.G.; Peck, T.L. Microflow NMR: Concepts and Capabilities. Anal. Chem. 2004, 76, 2966–2974. [Google Scholar] [CrossRef]
- Olson, D.L.; Peck, T.L.; Webb, A.G.; Magin, R.L.; Sweedler, J.V. High-Resolution Microcoil ’H-NMR for Mass-Limited, Nanoliter-Volume Samples. Science 2000, 270, 1967–1970. [Google Scholar] [CrossRef]
- Jones, C.J.; Larive, C.K. Could Smaller Really Be Better? Current and Future Trends in High-Resolution Microcoil NMR Spectroscopy. Anal. Bioanal. Chem. 2012, 402, 61–68. [Google Scholar] [CrossRef]
- Jansma, A.; Chuan, T.; Albrecht, R.W.; Olson, D.L.; Peck, T.L.; Geierstanger, B.H. Automated Microflow NMR: Routine Analysis of Five-Microliter Samples. Anal. Chem. 2005, 77, 6509–6515. [Google Scholar] [CrossRef][Green Version]
- Exarchou, V.; Krucker, M.; Van Beek, T.A.; Vervoort, J.; Gerothanassis, I.P.; Albert, K. LC-NMR Coupling Technology: Recent Advancements and Applications in Natural Products Analysis. Magn. Reson. Chem. 2005, 43, 681–687. [Google Scholar] [CrossRef] [PubMed]
- Hwang, T.L.; Shaka, A.J. Water Suppression That Works: Excitation Sculpting Using Arbitrary Waveforms and Pulsed Field Gradients. J. Magn. Res. 1995, 112, 275–279. [Google Scholar] [CrossRef]
- Xiao, H.B.; Krucker, M.; Putzbach, K.; Albert, K. Capillary Liquid Chromatography-Microcoil 1H Nuclear Magnetic Resonance Spectroscopy and Liquid Chromatography-Ion Trap Mass Spectrometry for on-Line Structure Elucidation of Isoflavones in Radix Astragali. J. Chromatogr. A 2005, 1067, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Sturm, S.; Seger, C. Liquid Chromatography-Nuclear Magnetic Resonance Coupling as Alternative to Liquid Chromatography-Mass Spectrometry Hyphenations: Curious Option or Powerful and Complementary Routine Tool? J. Chromatogr. A 2012, 1259, 50–61. [Google Scholar] [CrossRef] [PubMed]
- Wolfender, J.-L.; Queiroz, E.F.; Hostettmann, K. Bioactive Natural Products: Detection, Isolation, and Structural Determination, 2nd ed.; Colegate, S.M., Molyneux, R.J., Eds.; Taylor & Francis Group: Boca Raton, FL, USA, 2008. [Google Scholar]
- Spring, O.; Buschmann, H.; Vogler, B.; Schilling, E.E.; Spraul, M.; Hoffmann, M. Sesquiterpene Lactone Chemistry of Zaluzania Grayana from On-Line LC-NMR Measurements. Phytochemistry 1995, 39, 609–612. [Google Scholar] [CrossRef]
- Ndjoko, K.; Wolfender, J.-L.; Roder, E.; Hostettmann, K. Determination of Pyrrolizidine Alkaloids in Senecio Species by Liquid Chromatography/Thermospray-Mass Spectrometry and Liquid Chromatography/Nuclear Magnetic Resonance Spectroscopy. Planta Med. 1998, 65, 562–566. [Google Scholar] [CrossRef] [PubMed]
- Ioset, J.; Wolfender, J.; Marston, A.; Gupta, M.P.; Hostettmann, K. Identification of Two Isomeric Meroterpenoid Naphthoquinones from Cordia Linnaei by Liquid Chromatography-Mass Spectrometry and Liquid Chromatography-Nuclear Magnetic Resonance Spectroscopy. Phytochem. Anal. 1999, 10, 137–142. [Google Scholar] [CrossRef]
- Fouseki, M.M.; Damianakos, H.; Karikas, G.A.; Roussakis, C.; Gupta, M.P.; Chinou, I. Chemical constituents from Cordia alliodora and C. colloccoca (Boraginaceae) and their biological activities. Fitoterapia 2016, 115, 9–14. [Google Scholar] [CrossRef]
- Spring, O.; Heil, N.; Voglert, B. Sesquiterpene Lactones and Flavanones in Scalesia Species. Phytochemistry 1997, 46, 1369–1373. [Google Scholar] [CrossRef]
- Bringmann, G.; Ruckert, M.; Messer, K.; Schupp, O.; Louis, A.M. Acetogenic Isoquinoline Alkaloids CXXI. Use of on-Line High-Performance Liquid Chromatography-Nuclear Magnetic Resonance Spectrometry Coupling in Phytochemical Screening Studies: Rapid Identification of Metabolites in Dioncophyllum Thollonii. J. Chem. Phys. 1999, 837, 267–272. [Google Scholar]
- Bringmann, G.; Gu, C.; Schlauer, J.; Ru, M. HPLC-NMR On-Line Coupling Including the ROESY Technique: Direct Characterization of Naphthylisoquinoline Alkaloids in Crude Plant Extracts. Anal. Chem. 1998, 70, 2805–2811. [Google Scholar] [CrossRef]
- Zhao, Y.; Nookandeh, A.; Schneider, B.; Sun, X.; Schmitt, B.; Stockigt, J. Lignans from Torreya Jackii Identified by Stopped-Flow High-Performance Liquid Chromatography-Nuclear Magnetic Resonance Spectroscopy. J. Chromatogr. A 1999, 837, 83–91. [Google Scholar] [CrossRef]
- Renukappa, T.; Roos, G.; Klaiber, I.; Vogler, B.; Kraus, W. Application of High-Performance Liquid Chromatography Coupled to Nuclear Magnetic Resonance Spectrometry, Mass Spectrometry and Bioassay for the Determination of Active Saponins from Bacopa Monniera Wettst. J. Chromatogr. A 1999, 847, 109–116. [Google Scholar] [CrossRef]
- Kleinwächter, P.; Martin, K.; Groth, I.; Dornberger, K. Use of Coupled HPLC/1H NMR and HPLC/ESI-MS for the Detection and Identification of (2E,4Z)-Decadienoic Acid from a New Agromyces Species. J. High Resol. Chromatogr. 2000, 23, 609–612. [Google Scholar] [CrossRef]
- Schneider, B.; Zhao, Y.; Blitzke, T.; Schmitt, B.; Nookandeh, A. Taxane Analysis by High Performance Liquid Chromatography-Nuclear Magnetic Resonance Spectroscopy of Taxus Species. Phytochem. Anal. 1998, 9, 237–244. [Google Scholar] [CrossRef]
- Strohschein, S.; Rentel, C.; Lacker, T.; Bayer, E.; Albert, K. Separation and Identification of Tocotrienol Isomers by HPLC-MS and HPLC-NMR Coupling. Anal. Chem. 1999, 71, 1780–1785. [Google Scholar] [CrossRef] [PubMed]
- Ho-ltzel, A.; Schlotterbeck, G.; Albert, K.; Bayer, E. Separation and Characterisation of Hop Bitter Acids by HPLC-1H NMR Coupling. Chromatographia 1996, 42, 499–500. [Google Scholar] [CrossRef]
- Wilson, I.D.; Morgan, E.D.; Lafont, R.; Wright, B. High-Performance Liquid Chromatography Coupled to Nuclear Magnetic Resonance Spectroscopy Application to the Ecdysteroids of Silene Otites. J. Chromatogr. A 1998, 799, 333–336. [Google Scholar] [CrossRef]
- Dachtler, M.; Glaser, T.; Kohler, K.; Albert, K. Combined HPLC-MS and HPLC-NMR On-Line Coupling for the Separation and Determination of Lutein and Zeaxanthin Stereoisomers in Spinach and in Retina. Anal. Chem. 2001, 73, 667–674. [Google Scholar] [CrossRef]
- Vilegas, W.; Vilegas, J.H.Y.; Dachtler, M.; Glaser, T.; Albert, K. Application of On-Line C30 RP-HPLC-NMR for the Analysis of Flavonoids from Leaf Extract of Maytenus Aquifolium. Phytochem. Anal. 2000, 321, 317–321. [Google Scholar] [CrossRef]
- Santos, L.C.; Dachtler, M.; Andrade, F.D.P.; Albert, K.; Vilegas, W. Application of HPLC-NMR Coupling Using C30 Phase in the Separation and Identification of Flavonoids of Taxonomic Relevance. Fresenius J. Anal. Chem. 2000, 368, 540–541. [Google Scholar] [CrossRef] [PubMed]
- Pusecker, K.; Albert, K.; Bayer, E. Investigation of Hop and Beer Bitter Acids by Coupling of High-Performance Liquid Chromatography to Nuclear Magnetic Resonance Spectroscopy. J. Chromatogr. A 1999, 836, 245–252. [Google Scholar] [CrossRef]
- Abel, C.B.L.; Lindon, J.C.; Noble, D.; Rudd, B.A.M.; Sidebottom, P.J.; Nicholson, J.K. Characterization of Metabolites in Intact Streptomyces Citricolor Culture Supernatants Using High-Resolution Nuclear Magnetic Resonance and Directly Coupled High-Pressure Liquid Chromatography-Nuclear Magnetic Resonance Spectroscopy. Anal. Biochem. 1999, 270, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Sandvoss, M.; Weltring, A.; Preiss, A.; Levsen, K.; Wuensch, G. Combination of Matrix Solid-Phase Dispersion Extraction and Direct on-Line Liquid Chromatography-Nuclear Magnetic Resonance Spectroscopy-Tandem Mass Spectrometry as a New Efficient Approach for the Rapid Screening of Natural Products. J. Chromatogr. A 2001, 917, 75–86. [Google Scholar] [CrossRef]
- Sandvoss, M.; Pham, H.; Levsen, K.; Preiss, A.; Mügge, C.; Wünsch, G. Isolation and Structural Elucidation of Steroid Oligoglycosides from the Starfish Asterias Rubens by Means of Direct Online LC-NMR-MS Hyphenation and One- and Two-Dimensional NMR Investigations. Eur. J. Org. Chem. 2000, 2000, 1253–1262. [Google Scholar] [CrossRef]
- Cavin, A.; Potterat, O.; Wolfender, J.; Hostettmann, K.; Dyatmyko, W. Use of on-Flow LC/1H NMR for the Study of an Antioxidant Fraction from Orophea Enneandra and Isolation of a Polyacetylene, Lignans, and a Tocopherol Derivative. J. Nat. Prod. 1998, 61, 1497–1501. [Google Scholar] [CrossRef]
- Kang, S.W.; Kim, M.C.; Kim, C.Y.; Jung, S.H.; Um, B.H. The Rapid Identification of Isoflavonoids from Belamcanda Chinensis by LC-NMR and LC-MS. Chem. Pharm. Bull. 2008, 56, 1452–1454. [Google Scholar] [CrossRef]
- Horník, Š.; Sajfrtová, M.; Karban, J.; Sýkora, J.; Březinová, A.; Wimmer, Z. LC-NMR Technique in the Analysis of Phytosterols in Natural Extracts. J. Anal. Methods Chem. 2013, 2013, 526818. [Google Scholar] [CrossRef]
- Acevedo de la Cruz, A.; Hilbert, G.; Rivière, C.; Mengin, V.; Ollat, N.; Bordenave, L.; Decroocq, S.; Delaunay, J.C.; Delrot, S.; Mérillon, J.M.; et al. Anthocyanin Identification and Composition of Wild Vitis spp. Accessions by Using LC-MS and LC-NMR. Anal. Chim. Acta 2012, 732, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Schaller, F.; Wolfender, J.L.; Hostettmann, K.; Mavi, S. New Antifungal “quinone Methide” Diterpenes from Bobgunnia Madagascariensis and Study of Their Interconversion by LC/NMR. Helv. Chim. Acta 2001, 84, 222–229. [Google Scholar] [CrossRef]
- Bieri, S.; Varesio, E.; Veuthey, J.; Muñoz, O.; Tseng, L.; Braumann, U.; Spraul, M.; Christen, P. Identification of Isomeric Tropane Alkaloids from Schizanthus Grahamii by HPLC-NMR with Loop Storage and HPLC-UV-MS/SPE-NMR Using a Cryogenic Flow Probe. Phytochem. Anal. 2006, 17, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Schefer, A.B.; Braumann, U.; Tseng, L.; Spraul, M.; Soares, M.G.; Fernandes, B.; Maria, F.G.F.; Vieira, P.C.; Ferreira, A.G. Application of High-Performance Liquid Chromatography-Nuclear Magnetic Resonance Coupling to the Identification of Limonoids from Mahogany Tree (Switenia Macrophylla, Meliaceae) by Stopped-Flow 1D and 2D NMR Spectroscopy. J. Chromatogr. A 2006, 1128, 152–163. [Google Scholar] [CrossRef] [PubMed]
- Gavidia, I.; Seitz, H.U.; Pérez-Bermúdez, P.; Vogler, B. LC-NMR Applied to the Characterisation of Cardiac Glycosides from Three Micropropagated Isoplexis Species. Phytochem. Anal. 2002, 13, 266–271. [Google Scholar] [CrossRef] [PubMed]
- Queiroz, E.F.; Wolfender, J.L.; Raoelison, G.; Hostettmann, K. Determination of the Absolute Configuration of 6-Alkylated α-Pyrones from Ravensara Crassifolia by LC-NMR. Phytochem. Anal. 2003, 14, 34–39. [Google Scholar] [CrossRef]
- Guilet, D.; Guntern, A.; Ioset, J.R.; Queiroz, E.F.; Ndjoko, K.; Foggin, C.M.; Hostettmann, K. Absolute Configuration of a Tetrahydrophenanthrene from Heliotropium Ovalifolium by LC-NMR of Its Mosher Esters. J. Nat. Prod. 2003, 66, 17–20. [Google Scholar] [CrossRef]
- Iwasa, K.; Takahashi, T.; Nishiyama, Y.; Moriyasu, M.; Sugiura, M.; Takeuchi, A.; Tode, C.; Tokuda, H.; Takeda, K. Online Structural Elucidation of Alkaloids and Other Constituents in Crude Extracts and Cultured Cells of Nandina Domestica by Combination of LC-MS/MS, LC-NMR, and LC-CD Analyses. J. Nat. Prod. 2008, 71, 1376–1385. [Google Scholar] [CrossRef]
- Kenny, O.; Smyth, T.J.; Hewage, C.M.; Brunton, N.P.; McLoughlin, P. 4-Hydroxyphenylacetic Acid Derivatives of Inositol from Dandelion (Taraxacum Officinale) Root Characterised Using LC-SPE-NMR and LC-MS Techniques. Phytochemistry 2014, 98, 197–203. [Google Scholar] [CrossRef]
- Tatsis, E.C.; Boeren, S.; Exarchou, V.; Troganis, A.N.; Vervoort, J.; Gerothanassis, I.P. Identification of the Major Constituents of Hypericum Perforatum by LC/SPE/NMR and/or LC MS. Phytochemistry 2007, 68, 383–393. [Google Scholar] [CrossRef]
- Clarkson, C.; Madikane, E.V.; Hansen, S.H.; Smith, P.J.; Jaroszewski, J.W. HPLC-SPE-NMR Characterization of Sesquiterpenes in an Antimycobacterial Fraction from Warburgia Salutaris. Planta Med. 2007, 73, 578–584. [Google Scholar] [CrossRef]
- Lam, S.; Wang, J.; Chen, C.; Lee, S. Investigation of Flavonoid Glycosides from Neolitsea Sericea Var. Aurata via the General Method and HPLC-SPE-NMR. J. Chin. Chem. Soc. 2008, 55, 449–455. [Google Scholar] [CrossRef]
- Lee, S.; Lai, Y.; Chen, C.; Tseng, L.; Wang, C. Characterization of Isoquinoline Alkaloids from Neolitsea Sericea Var. Aurata by HPLC-SPE-NMR. J. Nat. Prod. 2007, 70, 637–642. [Google Scholar] [CrossRef] [PubMed]
- Cao, M.; Muganga, R.; Nistor, I.; Tits, M.; Angenot, L.; Frederich, M. LC-SPE-NMR-MS Analysis of Strychnos Usambarensis Fruits from Rwanda. Phytochem. Lett. 2012, 5, 170–173. [Google Scholar] [CrossRef]
- Capistrano, I.R.; Wouters, A.; Foubert, K.; Baldé, A.M.; Apers, S.; Lardon, F.; Pieters, L.; Exarchou, V. Phytochemical Characterisation of a Cytotoxic Stem Bark Extract of Steganotaenia Araliacea and Identification of a Protoflavanone by LC-SPE-NMR. Phytochem. Lett. 2015, 12, 119–124. [Google Scholar] [CrossRef]
- Xu, Y.J.; Foubert, K.; Dhooghe, L.; Lemière, F.; Maregesi, S.; Coleman, C.M.; Zou, Y.; Ferreira, D.; Apers, S.; Pieters, L. Rapid Isolation and Identification of Minor Natural Products by LC-MS, LC-SPE-NMR and ECD: Isoflavanones, Biflavanones and Bisdihydrocoumarins from Ormocarpum Kirkii. Phytochemistry 2012, 79, 121–128. [Google Scholar] [CrossRef] [PubMed]

| Solvent Suppression Method | Operating Setup | Benefits | Drawbacks |
|---|---|---|---|
| Presaturation | Irradiation of solvent signal for a period of time with a continuous wave RF field | Easy setup and effective for eliminating single solvent signal | Not easy to suppress multiple solvent peaks simultaneously |
| Excitation sculpting | Extraction of the solvent signals with selective pulses | Could be automated and multiple solvent signals can be suppressed | The pulse sequences need to be optimized and could suppress analyte signals that overlap with the solvent |
| Watergate excitation technique (WET) | The solvent signals are suppressed at the beginning of the experiment | Could be automated and multiple solvent peaks can be suppressed | Suppresses analyte signals that overlap with the solvent. |
| Mode of Operation | Plant Species | Plant Parts | Class of Compounds Analyzed | Reference |
|---|---|---|---|---|
| On-flow | Orophea enneandra | Leaf | Lignans, Tocopherol, Polyacetylene | [62] |
| Iris domestica | Root | Isoflavonoids | [63] | |
| Ancistrocladus guineensis | Leaf | Alkaloids | [47] | |
| E. vacciniifolium | Bark | Crude alkaloid | [31] | |
| Urtica dioica | Root | Phytosterols | [64] | |
| Vitis vinifera, Vitis amurensis, Vitis cinerea and Vitis X champinii | Grape barry skin | Anthocyanin composition | [65] | |
| Bobgunnia madagascariensis | Root bark | new antifungal constituents | [66] | |
| Stop-flow (including loop storage) | Monotes engleri | Leaf | Prenylated flavanones | [9] |
| Schizanthus grahamii | Stem-bark | Tropane alkaloids | [67] | |
| Swietenia macrophylla | Seed | Limonoids | [68] | |
| E. vacciniifolium | Bark | Crude alkaloid | [31] | |
| Isoplexis species | Seed | glycosides | [69] | |
| Ravensara crassifolia | Aerial parts | 6-alkylated a-pyrones | [70] | |
| Heliotropiu ovalifolium | Aerial parts | tetrahydrophenanthrene | [71] | |
| Schizanthus grahamii | Stem-bark | Isomeric tropane alkaloids | [67] | |
| Nandina domestic | Stem part | Alkaloids, crude extracts and cultured cells | [72] | |
| Vitis species | Grape barry skin | Anthocyanin composition | [65] | |
| HPLC–SPE–NMR | Taraxacum officinale | Root | 4-hydroxyphenylacetic acid derivatives of inositol | [73] |
| Hypericum perforatum | Aerial parts | Naphtodianthrones, Phloroglucinols, Flavonoids, Phenolic acids | [74] | |
| Warburgia salutaris | Bark | Sequisterpenes | [75] | |
| Schizanthus grahamii | Stem-bark | Isomeric tropane alkaloids | [67] | |
| Smirnowia iranica | Root | Isoflavonoids | [21] | |
| Neolitsea sericea | Leaf | Flavonoid glycosides | [76] | |
| Neolitsea sericea | Leaf | Isoquinoline alkaloids | [77] | |
| Strychnos usambarensis G. | Fruit | Akagerine, Palicoside | [78] | |
| Steganotaenia araliacea | Stem-bark | Cytotoxic stem bark extract | [79] | |
| Ormocarpum kirki | Root | Minor natural products | [80] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gebretsadik, T.; Linert, W.; Thomas, M.; Berhanu, T.; Frew, R. LC–NMR for Natural Product Analysis: A Journey from an Academic Curiosity to a Robust Analytical Tool. Sci 2021, 3, 6. https://doi.org/10.3390/sci3010006
Gebretsadik T, Linert W, Thomas M, Berhanu T, Frew R. LC–NMR for Natural Product Analysis: A Journey from an Academic Curiosity to a Robust Analytical Tool. Sci. 2021; 3(1):6. https://doi.org/10.3390/sci3010006
Chicago/Turabian StyleGebretsadik, Tesfay, Wolfgang Linert, Madhu Thomas, Tarekegn Berhanu, and Russell Frew. 2021. "LC–NMR for Natural Product Analysis: A Journey from an Academic Curiosity to a Robust Analytical Tool" Sci 3, no. 1: 6. https://doi.org/10.3390/sci3010006
APA StyleGebretsadik, T., Linert, W., Thomas, M., Berhanu, T., & Frew, R. (2021). LC–NMR for Natural Product Analysis: A Journey from an Academic Curiosity to a Robust Analytical Tool. Sci, 3(1), 6. https://doi.org/10.3390/sci3010006








