Structural Alterations of Histone Proteins in DNA-Damaged Cells Revealed by Synchrotron Radiation Circular Dichroism Spectroscopy: A New Piece of the DNA-Damage-Response Puzzle
Abstract
:1. Introduction
2. CD Spectroscopy
2.1. CD and Its Notation
2.2. Advantages of CD Spectroscopy Using Synchrotron Radiation
2.3. Experimental Procedure for CD Spectroscopy
2.3.1. Sample Preparation
2.3.2. The Sample Cell
2.3.3. Measurements and Analyses of CD Spectra
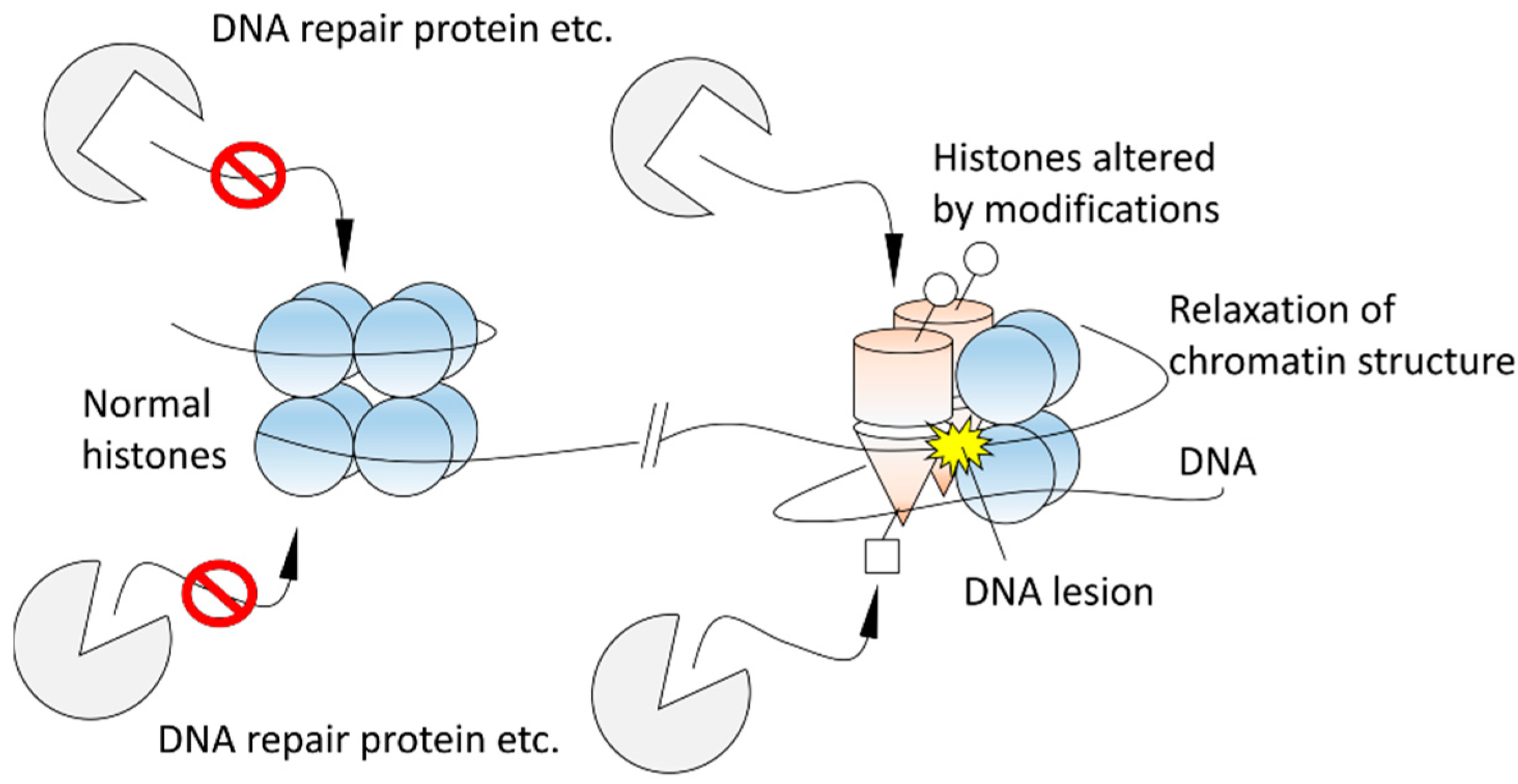
3. Secondary Structure Analyses of Histones Extracted from X-Irradiated Cells
3.1. Experimental Procedure
3.2. Absorption and CD Spectra of Histones
3.3. Secondary Structure Alterations of Histones
4. Future Perspectives
Funding
Acknowledgments
Conflicts of Interest
References
- De Bont, R.; van Larebeke, N. Endogenous DNA damage in humans: A review of quantitative data. Mutagenesis 2004, 19, 169–185. [Google Scholar] [CrossRef] [Green Version]
- Cadet, J.; Sage, E.; Douki, T. Ultraviolet radiation-mediated damage to cellular DNA. Mutat. Res. 2005, 571, 3–17. [Google Scholar] [CrossRef]
- Hunt, C.R.; Ramnarain, D.; Horikoshi, N.; Iyengar, P.; Pandita, R.K.; Shay, J.W.; Pandita, T.K. Histone modifications and DNA double-strand break repair after exposure to ionizing radiations. Radiat. Res. 2013, 179, 383–392. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kanaar, R.; Hoeijmakers, J.H.J.; van Gent, D.C. Molecular mechanisms of DNA double-strand break repair. Trends Cell Biol. 1998, 8, 483–489. [Google Scholar] [CrossRef]
- Price, B.D.; D’Andrea, A.D. Chromatin remodeling at DNA double-strand breaks. Cell 2013, 152, 1344–1354. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Attikum, H.; Gasser, S.M. Crosstalk between histone modifications during the DNA damage response. Trends Cell Biol. 2009, 19, 207–217. [Google Scholar] [CrossRef]
- Pandita, T.K.; Richardson, C. Chromatin remodeling finds its place in the DNA double-strand break response. Nucleic Acids Res. 2009, 37, 1363–1377. [Google Scholar] [CrossRef]
- Lieber, M.R. The mechanism of double-strand DNA break repair by the nonhomologous DNA end-joining pathway. Annu. Rev. Biochem. 2010, 79, 181–211. [Google Scholar] [CrossRef] [Green Version]
- Symington, L.S.; Gautier, J. Double-strand break end resection and repair pathway choice. Annu. Rev. Genet. 2011, 45, 247–271. [Google Scholar] [CrossRef]
- Gong, F.; Miller, K.M. Mammalian DNA repair: HATs and HDACs make their mark through histone acetylation. Mutat. Res. 2013, 750, 23–30. [Google Scholar] [CrossRef]
- Uziel, T.; Lerenthal, Y.; Moyal, L.; Andegeko, Y.; Mittelman, L.; Shiloh, Y. Requirement of the MRN complex for ATM activation by DNA damage. EMBO J. 2003, 22, 5612–5621. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burma, S.; Chen, B.P.; Murphy, M.; Kurimasa, A.; Chen, D.J. ATM phosphorylates histone H2AX in response to DNA double-strand breaks. J. Biol. Chem. 2001, 276, 42462–42467. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stewart, G.S.; Wang, B.; Bignell, C.R.; Taylor, A.M.R.; Elledge, S.J. MDC1 is a mediator of the mammalian DNA damage checkpoint. Nature 2003, 421, 961–966. [Google Scholar] [CrossRef] [PubMed]
- Lou, Z.; Minter-Dykhouse, K.; Franco, S.; Gostissa, M.; Rivera, M.A.; Celeste, A.; Manis, J.P.; van Deursen, J.; Nussenzweig, A.; Paull, T.T.; et al. MDC1 maintains genomic stability by participating in the amplification of ATM-dependent DNA damage signals. Moll. Cell 2006, 21, 187–200. [Google Scholar] [CrossRef] [PubMed]
- Rogakou, E.P.; Pilch, D.R.; Orr, A.H.; Ivanova, V.S.; Bonner, W.M. DNA double-stranded breaks induce histone H2AX phosphorylation on serine 139. J. Biol. Chem. 1998, 273, 5858–5868. [Google Scholar] [CrossRef] [Green Version]
- Ikura, T.; Tashiro, S.; Kakino, A.; Shima, H.; Jacob, N.; Amunugama, R.; Yoder, K.; Izumi, S.; Kuraoka, I.; Tanaka, K.; et al. DNA damage-dependent acetylation and ubiquitination of H2AX enhances chromatin dynamics. Mol. Cell. Biol. 2007, 27, 7028–7040. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huen, M.S.Y.; Grant, R.; Manke, I.; Minn, K.; Yu, X.; Yaffe, M.B.; Chen, J. RNF8 transduces the DNA-damage signal via histone ubiquitylation and checkpoint protein assembly. Cell 2007, 131, 901–914. [Google Scholar] [CrossRef] [Green Version]
- Matsuoka, S.; Ballif, B.A.; Smogorzewska, A.; McDonald III, E.R.; Hurov, K.E.; Luo, J.; Bakalarski, C.E.; Zhao, Z.; Solimini, N.; Lerenthal, Y.; et al. ATM and ATR substrate analysis reveals extensive protein networks responsive to DNA damage. Science 2007, 316, 1160–1166. [Google Scholar] [CrossRef] [Green Version]
- Kolas, N.K.; Chapman, J.R.; Nakada, S.; Ylanko, J.; Chahwan, R.; Sweeney, F.D.; Panier, S.; Mendez, M.; Wildenhain, J.; Thomson, T.M.; et al. Orchestration of the DNA-damage response by the RNF8 ubiquitin ligase. Science 2007, 318, 1637–1640. [Google Scholar] [CrossRef] [Green Version]
- Plans, V.; Scheper, J.; Soler, M.; Loukili, N.; Okano, Y.; Thomson, T.M. The RING finger protein RNF8 recruits UBC13 for lysine 63-based self polyubiquitylation. J. Cell. Biochem. 2006, 97, 572–582. [Google Scholar] [CrossRef]
- Mailand, N.; Bekker-Jensen, S.; Faustrup, H.; Melander, F.; Bartek, J.; Lukas, C.; Lukas, J. RNF8 ubiquitylates histones at DNA double-strand breaks and promotes assembly of repair proteins. Cell 2007, 131, 887–900. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stewart, G.S.; Panier, S.; Townsend, K.; Al-Hakim, A.K.; Kolas, N.K.; Miller, E.S.; Nakada, S.; Ylanko, J.; Olivarius, S.; Mendez, M.; et al. The RIDDLE syndrome protein mediates a ubiquitin-dependent signaling cascade at sites of DNA damage. Cell 2009, 136, 420–434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, B.; Matsuoka, S.; Ballif, B.A.; Zhang, D.; Smogorzewska, A.; Gygi, S.P.; Elledge, S.J. Abraxas and RAP80 from a BRCA1 protein complex required for the DNA damage response. Science 2007, 316, 1194–1198. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sobhian, B.; Shao, G.; Lilli, D.R.; Culhane, A.C.; Moreau, L.A.; Xia, B.; Livingston, D.M.; Greenberg, R.A. RAP80 targets BRCA1 to specific ubiquitin structures at DNA damage sites. Science 2007, 316, 1198–1202. [Google Scholar] [CrossRef] [Green Version]
- Kim, H.; Chen, J.; Yu, X. Ubiquitin-binding protein RAP80 mediates BRCA1-dependent DNA damage response. Science 2007, 316, 1202–1205. [Google Scholar] [CrossRef]
- Smedon, M.J. DNA repair and the role of chromatin structure. Curr. Opin. Cell Biol. 1991, 3, 422–428. [Google Scholar] [CrossRef]
- Soria, G.; Polo, S.E.; Almouzni, G. Prime, repair, restore: The active role of chromatin in the DNA damage response. Moll. Cell. 2012, 46, 722–734. [Google Scholar] [CrossRef] [Green Version]
- Goldstein, M.; Derheimer, F.A.; Tait-Mulder, J.; Kastan, M.B. Nucleolin mediates nucleosome disruption critical DNA double-strand repair. Proc. Natl. Acad. Sci. USA 2013, 110, 16874–16879. [Google Scholar] [CrossRef] [Green Version]
- Adler, A.J.; Fasman, G.D.; Wangh, L.J.; Allfrey, V.G. Altered conformational effects of naturally acetylated histone f2al (IV) in f2al-deoxyribonucleic acid complexes. Circular dichroism studies. J. Biol. Chem. 1974, 249, 2911–2914. [Google Scholar]
- Prevelige, P.E., Jr.; Fasman, G.D. Structural studies of acetylated and control inner core histones. Biochem. 1987, 26, 2944–2955. [Google Scholar] [CrossRef]
- Wang, X.; Moore, S.C.; Laszckzak, M.; Ausió, J. Acetylation increases the α-helical content of the histone tails of the nucleosome. J. Biol. Chem. 2000, 275, 35013–35020. [Google Scholar] [CrossRef] [Green Version]
- Yang, D.; Arya, G. Structure and binding of the H4 histone tail and the effects of lysine 16 acetylation. Phys. Chem. Chem. Phys. 2011, 13, 2911–2921. [Google Scholar] [CrossRef] [PubMed]
- Izumi, Y.; Matsuo, K.; Fujii, K.; Yokoya, A.; Taniguchi, M.; Namatamae, H. Circular dichroism spectroscopic study on structural alterations of histones induced by post-translational modifications in DNA damage responses: Lysine-9 methylation of H3. J. Radiat. Res. 2018, 59, 108–115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Izumi, Y.; Matsuo, K.; Namatame, H. Structural analysis of lysine-4 methylated histone H3 proteins using synchrotron radiation circular dichroism spectroscopy. Chirality 2018, 30, 536–540. [Google Scholar] [CrossRef] [PubMed]
- Izumi, Y.; Matsuo, K. Sample volume reduction using the Schwarzschild objective for a circular dichroism spectrophotometer and an application to the structural analysis of lysine-36 trimethylated histone H3 protein. Molecules 2018, 23, 2865. [Google Scholar] [CrossRef] [Green Version]
- Izumi, Y.; Yamamoto, S.; Fujii, K.; Yokoya, A. Secondary structure alterations of histones H2A and H2B in X-irradiated human cancer cells: Altered histones persist in cells for at least 24 hours. Radiat. Res. 2015, 184, 554–558. [Google Scholar] [CrossRef]
- Izumi, Y.; Fujii, K.; Wien, F.; Houée-Lévin, C.; Lacombe, S.; Salado-Leza, D.; Porcel, E.; Masoud, R.; Yamamoto, S.; Réfrégiers, M.; et al. Structure change from β-strand and turn to α-helix in histone H2A–H2B induced by DNA damage response. Biophys. J. 2016, 111, 69–78. [Google Scholar] [CrossRef] [Green Version]
- Izumi, Y.; Fujii, K.; Yamamoto, S.; Matsuo, K.; Namatame, H.; Taniguchi, M.; Yokoya, A. DNA damage response induces structural alterations in histone H3–H4. J. Radiat. Res. 2017, 58, 59–65. [Google Scholar] [CrossRef] [Green Version]
- Miles, A.J.; Wallace, B.A. Synchrotron radiation circular dichroism spectroscopy of proteins and applications in structural and functional genomics. Chem. Soc. Rev. 2006, 35, 39–51. [Google Scholar] [CrossRef]
- Matsuo, K.; Gekko, K. Vacuum-ultraviolet circular dichroism study of saccharides by synchrotron radiation spectrometry. Carbohydr. Res. 2004, 339, 591–597. [Google Scholar] [CrossRef]
- Matsuo, K.; Namatame, H.; Taniguchi, M.; Gekko, K. Vacuum-ultraviolet electronic circular dichroism study of methyl α-d-glucopyranoside in aqueous solution by time-dependent density functional theory. J. Phys. Chem. A 2012, 116, 9996–10003. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, K.; Gekko, K. Construction of a synchrotron-radiation vacuum-ultraviolet circular-dichroism spectrophotometer and its application to the structural analysis of biomolecules. Bull. Chem. Soc. Jpn. 2013, 86, 675–689. [Google Scholar] [CrossRef] [Green Version]
- Miles, A.J.; Hoffmann, S.V.; Tao, Y.; Janes, R.W.; Wallace, B.A. Synchrotron radiation circular dichroism (SRCD) spectroscopy: New beamlines and new applications in biology. Spectroscopy 2007, 21, 245–255. [Google Scholar] [CrossRef]
- Tao, Y.; Huang, Y.; Gao, Z.; Zhuang, H.; Zhou, A.; Tan, Y.; Li, D.; Sun, S. Developing VUV spectroscopy for protein folding and material luminescence on beamline 4B8 at the Beijing Synchrotron Radiation Facility. J. Synchrotron Rad. 2009, 16, 857–863. [Google Scholar] [CrossRef]
- Liu, S.-H.; Lin, Y.-H.; Huang, L.-J.; Luo, S.-W.; Tsai, W.-L.; Chiang, S.-Y.; Fung, H.-S. Design and construction of a compact end-station at NSRRC for circular-dichroism spectra in the vacuum-ultraviolet region. J. Synchrotron Rad. 2010, 17, 761–768. [Google Scholar] [CrossRef]
- Hussain, R.; Jávorfi, T.; Siligardi, G. Circular dichroism beamline B23 at the Diamond Light Source. J. Synchrotron Rad. 2012, 19, 132–135. [Google Scholar] [CrossRef]
- Réfrégiers, M.; Wien, F.; Ta, H.-P.; Premvardhan, L.; Bac, S.; Jamme, F.; Rouam, V.; Lagarde, B.; Polack, F.; Giorgetta, J.-L.; et al. DISCO synchrotron-radiation circular-dichroism endstation at SOLEIL. J. Synchrotron Rad. 2012, 19, 831–835. [Google Scholar] [CrossRef] [Green Version]
- Reichardt, G.; Bahrdt, J.; Schmidt, J.-S.; Gudat, W.; Ehresmann, A.; Müller-Albrecht, R.; Molter, H.; Schmoranzer, H.; Martins, M.; Schwentner, N.; et al. A 10 m-normal incidence monochromator at the quasi-periodic undulator U125-2 at BESSY II. Nucl. Instr. Methods Phys. Res. A 2001, 467–468, 462–465. [Google Scholar] [CrossRef]
- Bürck, J.; Roth, S.; Windisch, D.; Wadhwani, P.; Moss, D.; Ulrich, A.S. UV-CD12: Synchrotron radiation circular dichroism beamline at ANKA. J. Synchrotron Rad. 2015, 22, 844–852. [Google Scholar] [CrossRef] [Green Version]
- Matsuo, K.; Sakai, K.; Matsushima, S.; Fukuyama, T.; Gekko, K. Optical cell with a temperature-control unit for a vacuum-ultraviolet circular dichroism spectrophotometer. Anal. Sci. 2003, 19, 129–132. [Google Scholar] [CrossRef] [Green Version]
- Sreerama, N.; Venyaminov, S.Y.; Woody, R.W. Estimation of the number of α-helical and β-strand segments in proteins using circular dichroism spectroscopy. Protein Sci. 1999, 8, 370–380. [Google Scholar] [CrossRef] [PubMed]
- Sreerama, N.; Woody, R.W. Estimation of protein secondary structure from circular dichroism spectra: Comparison of CONTIN, SELCON, and CDSSTR methods with an expanded reference set. Anal. Biochem. 2000, 287, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Whitmore, L.; Wallace, B.A. Protein secondary structure analyses from circular dichroism spectroscopy: Methods and reference databases. Biopolymers 2004, 89, 392–400. [Google Scholar] [CrossRef] [PubMed]
- Micsonai, A.; Wien, F.; Kernya, L.; Lee, Y.-H.; Goto, Y.; Réfrégiers, M.; Kardos, J. Accurate secondary structure prediction and fold recognition for circular dichroism spectroscopy. Proc. Natl. Acad. Sci. USA 2015, 112, E3095–E3103. [Google Scholar] [CrossRef] [Green Version]
- Thomson, A.C.; Attwood, D.T.; Gullikson, E.M.; Howells, M.R.; Kortright, J.B.; Robinson, A.L.; Underwood, J.H. X-ray Data Booklet, 2nd ed.; Thomson, A.C., Vaughan, D., Eds.; Lawrence Berkeley National Laboratory: Berkeley, CA, USA, 2001. [Google Scholar]
- von Sontag, C. Free-Radical-Induced DNA Damage and Its Repair; Springer-Verlag: Berlin/Heidelberg, Germany, 2006. [Google Scholar]
- Greenfield, N.; Fasman, G.D. Computed circular dichroism spectra for the evaluation of protein conformation. Biochemistry 1969, 8, 4108–4116. [Google Scholar] [CrossRef]
- Wien, F.; Miles, A.J.; Lees, J.G.; Hoffmann, V.; Wallace, B.A. VUV irradiation effects on proteins in high-flux synchrotron radiation circular dichroism spectroscopy. J. Synchrotron Rad. 2005, 12, 517–523. [Google Scholar] [CrossRef] [Green Version]
- Kobayashi, J.; Fujimoto, H.; Sato, J.; Hayashi, I.; Burma, S.; Matsuura, S.; Chen, D.J.; Komatsu, K. Nucleolin participates in DNA double-strand break-induced damage response through MDC1-dependent pathway. PLoS ONE 2012, 7, e49245. [Google Scholar] [CrossRef] [Green Version]
- Banáth, J.P.; MacPhail, S.H.; Olive, P.L. Radiation sensitivity, H2AX phosphorylation, and kinetics of repair of DNA strand breaks in irradiated cervical cancer cell lines. Cancer Res. 2004, 64, 7144–7149. [Google Scholar] [CrossRef] [Green Version]
- Kühne, M.; Riballo, E.; Rief, N.; Rothkamm, K.; Jeggo, P.A.; Löbrich, M. A double-strand break repair defect in ATM-deficient cells contributes to radiosensitivity. Cancer Res. 2004, 64, 500–508. [Google Scholar] [CrossRef] [Green Version]
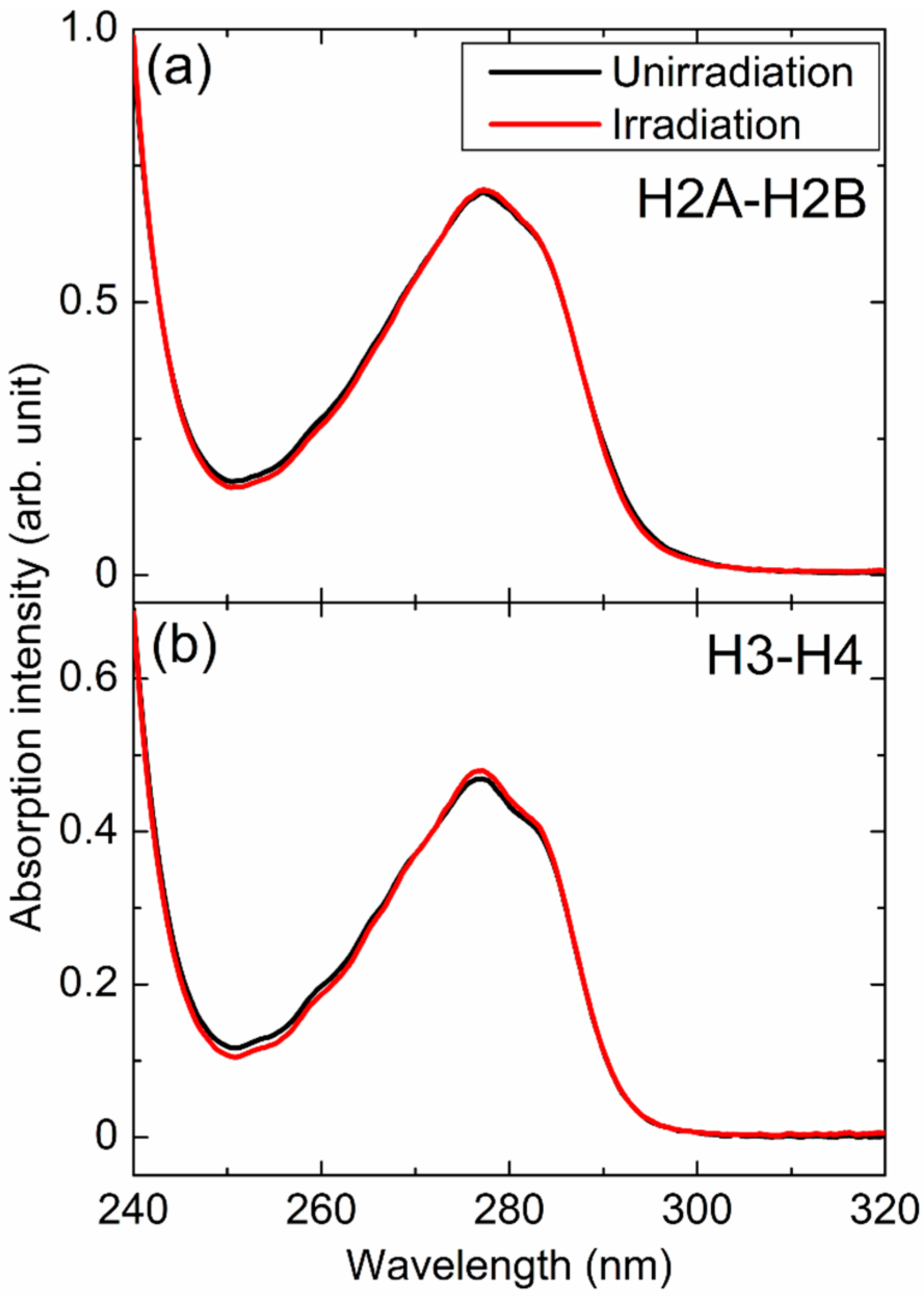
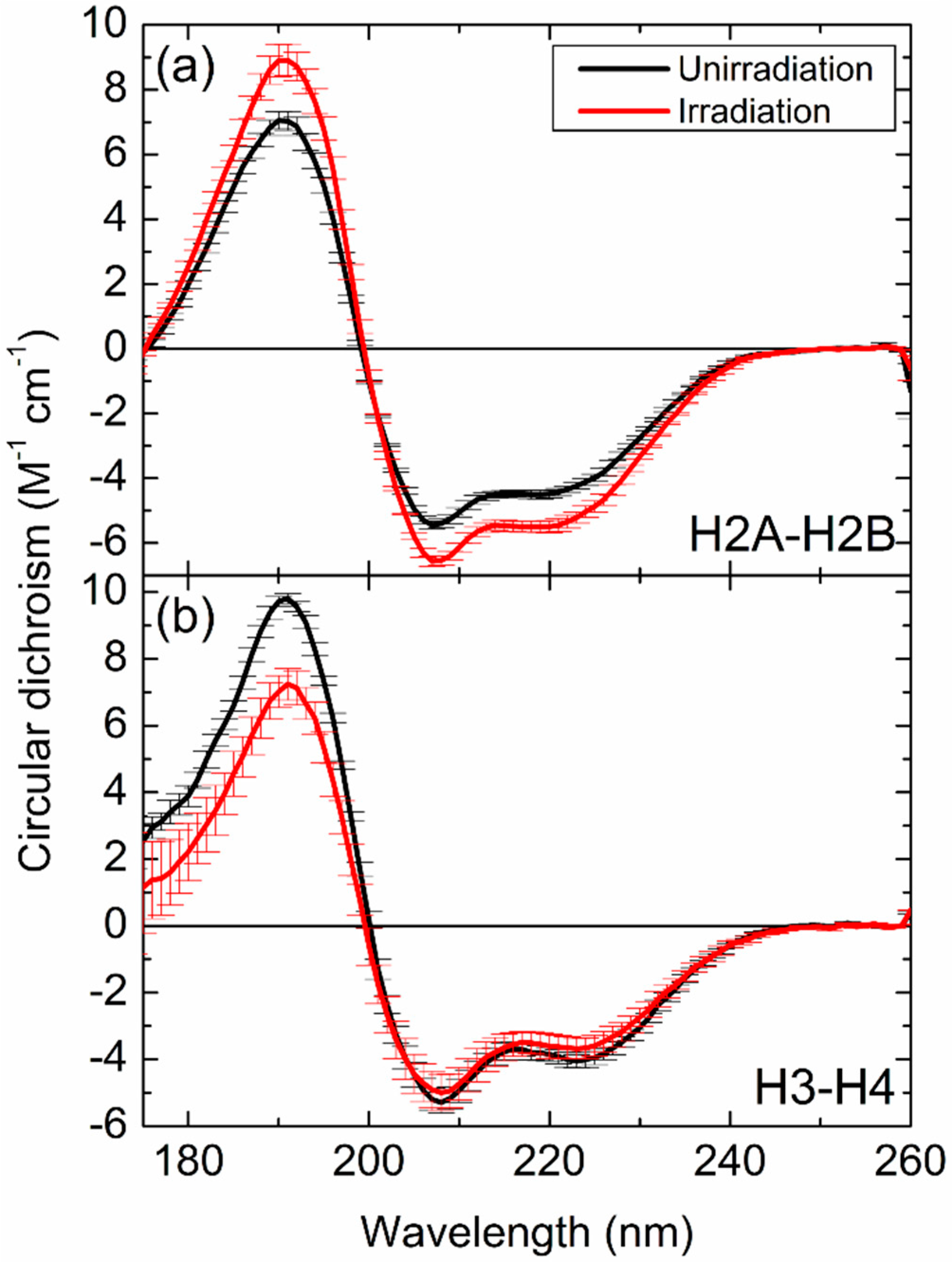
| Structure (%) | H2A–H2B (U) | H2A–H2B (I) | H3–H4 (U) | H3–H4 (I) |
|---|---|---|---|---|
| α-Helix | 33.5 ± 1.9 | 42.7 ± 0.6 | 61.6 ± 0.6 | 48.3 ± 0.8 |
| β-Strand | 14.6 ± 2.0 | 8.0 ± 1.2 | 1.9 ± 0.7 | 8.0 ± 0.3 |
| Turn | 12.7 ± 0.1 | 9.8 ± 0.4 | 18.2 ± 1.6 | 19.1 ± 0.4 |
| Unordered | 39.3 ± 0.1 | 39.5 ± 0.2 | 17.8 ± 2.5 | 26.0 ± 1.9 |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Izumi, Y. Structural Alterations of Histone Proteins in DNA-Damaged Cells Revealed by Synchrotron Radiation Circular Dichroism Spectroscopy: A New Piece of the DNA-Damage-Response Puzzle. Quantum Beam Sci. 2019, 3, 23. https://doi.org/10.3390/qubs3040023
Izumi Y. Structural Alterations of Histone Proteins in DNA-Damaged Cells Revealed by Synchrotron Radiation Circular Dichroism Spectroscopy: A New Piece of the DNA-Damage-Response Puzzle. Quantum Beam Science. 2019; 3(4):23. https://doi.org/10.3390/qubs3040023
Chicago/Turabian StyleIzumi, Yudai. 2019. "Structural Alterations of Histone Proteins in DNA-Damaged Cells Revealed by Synchrotron Radiation Circular Dichroism Spectroscopy: A New Piece of the DNA-Damage-Response Puzzle" Quantum Beam Science 3, no. 4: 23. https://doi.org/10.3390/qubs3040023
APA StyleIzumi, Y. (2019). Structural Alterations of Histone Proteins in DNA-Damaged Cells Revealed by Synchrotron Radiation Circular Dichroism Spectroscopy: A New Piece of the DNA-Damage-Response Puzzle. Quantum Beam Science, 3(4), 23. https://doi.org/10.3390/qubs3040023





