Recent Advances in Radioisotope Imaging Technology for Plant Science Research in Japan
Abstract
:1. Introduction
2. RI Imaging Technologies for Plant Science
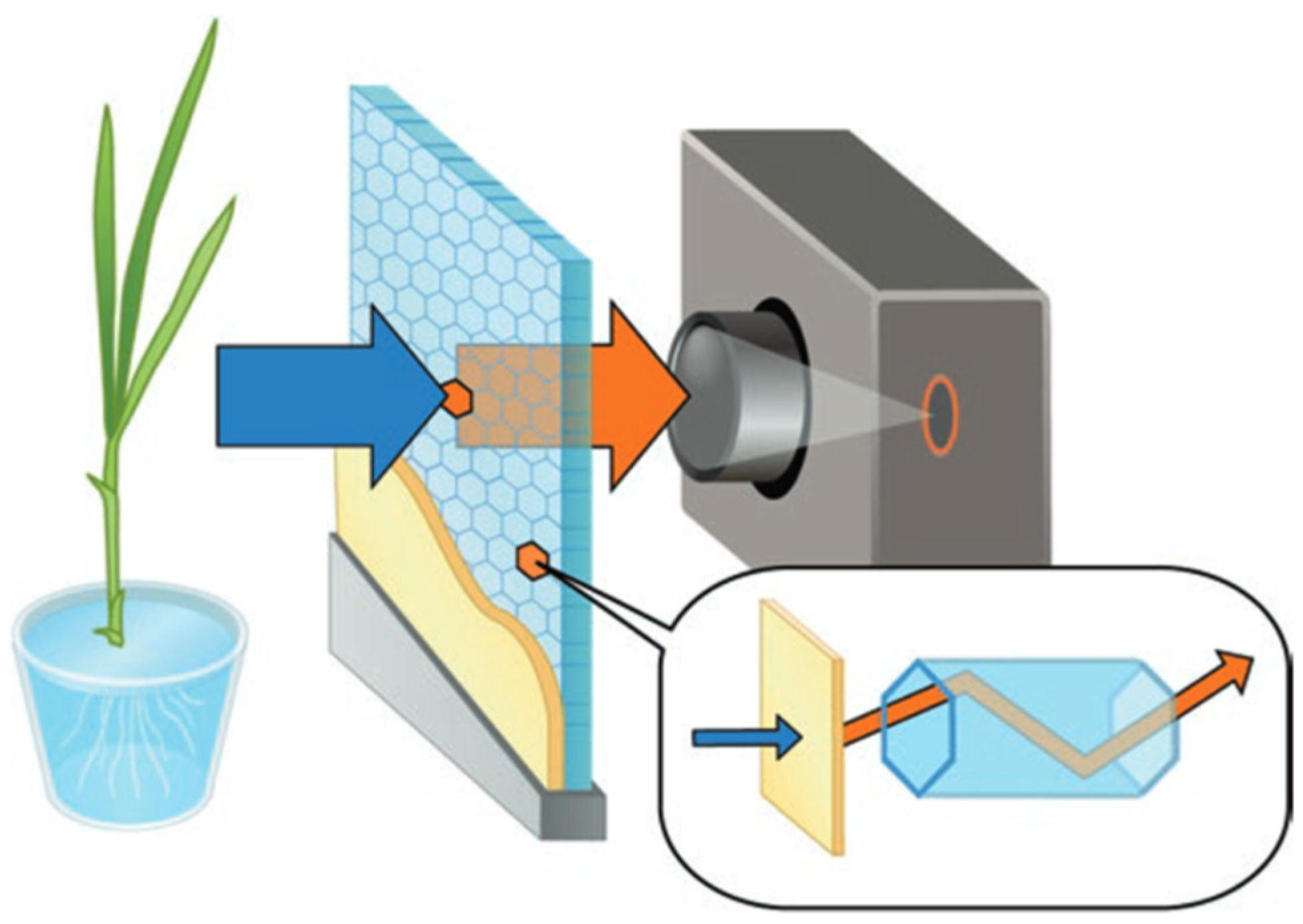
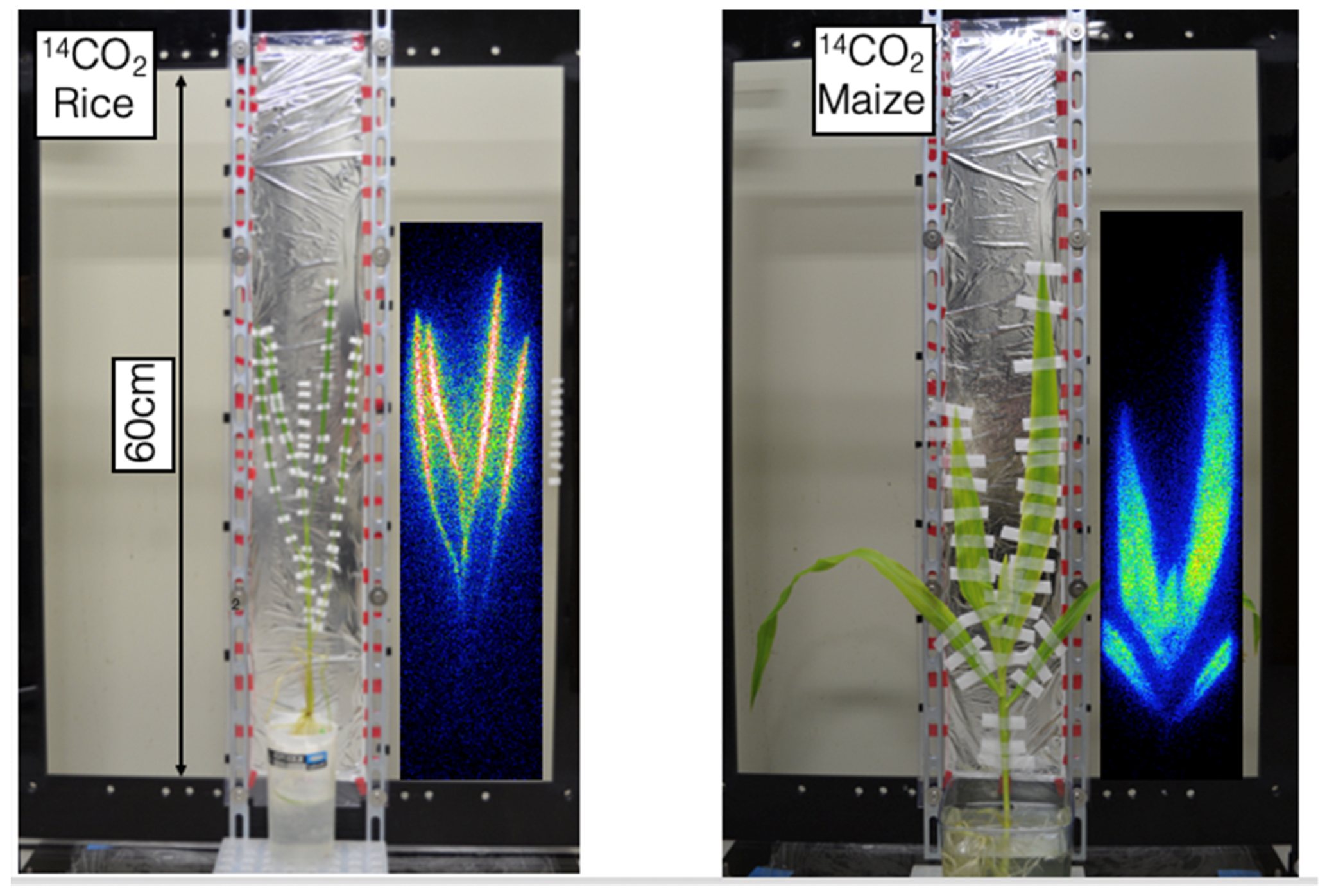
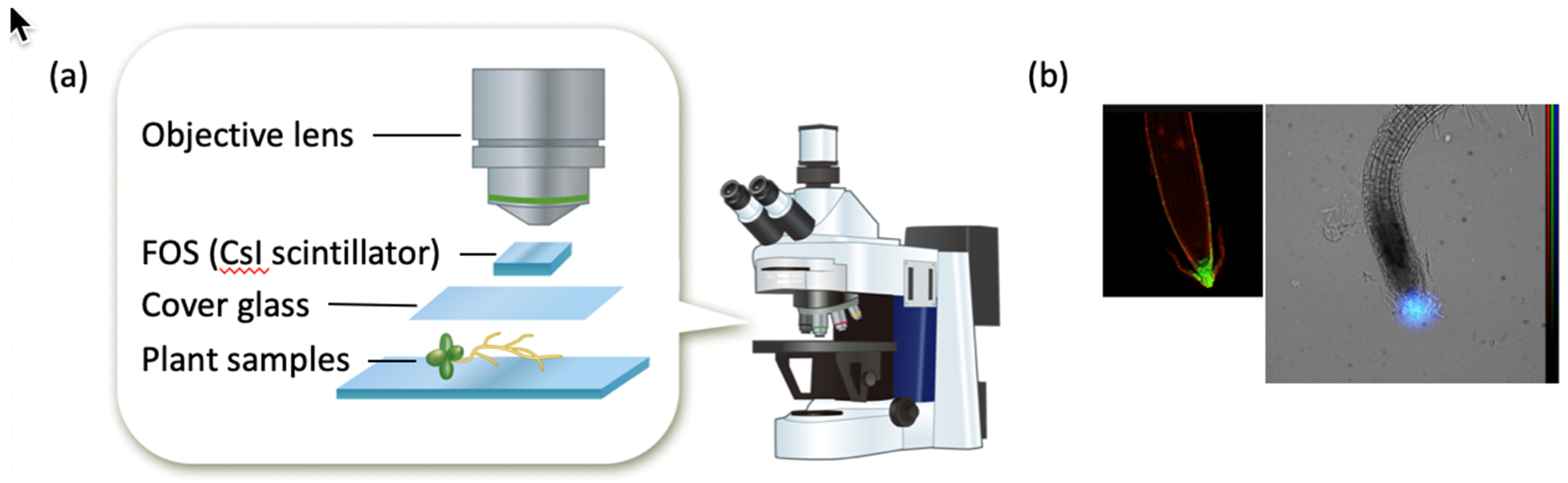
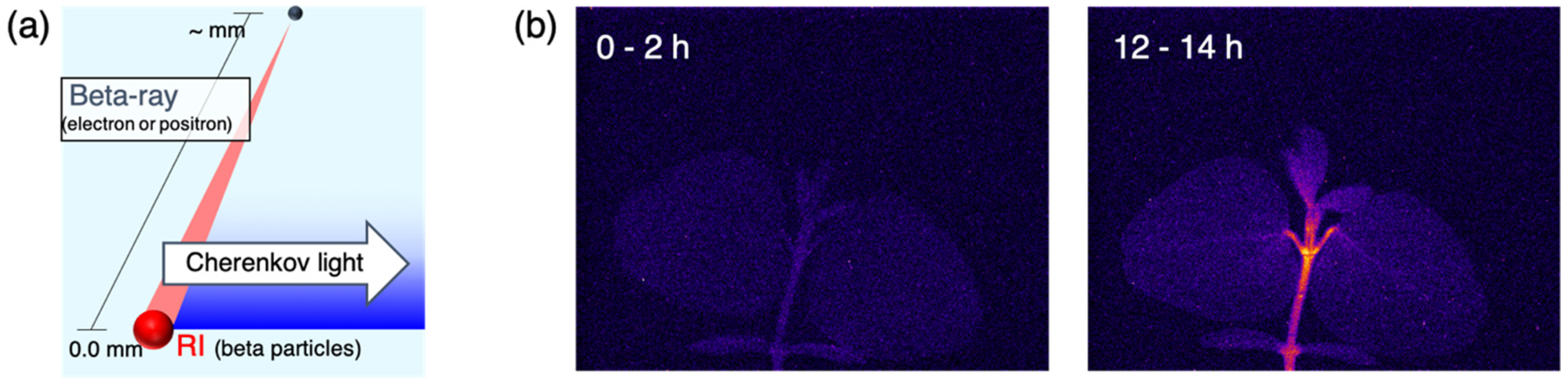
2.1. Beta-Rays Imaging
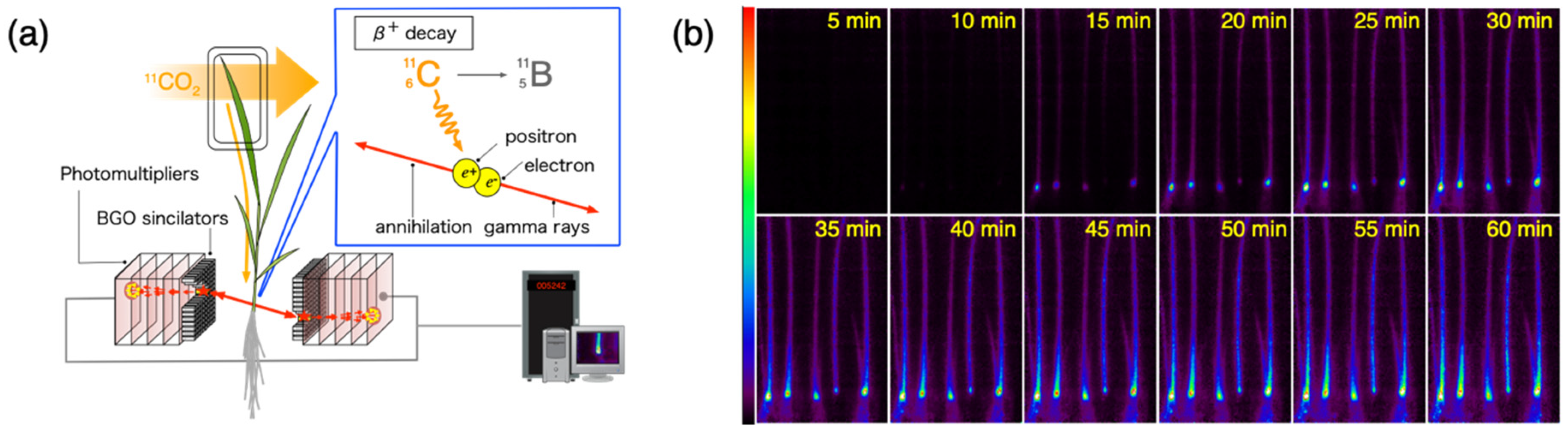
2.2. Positron Imaging
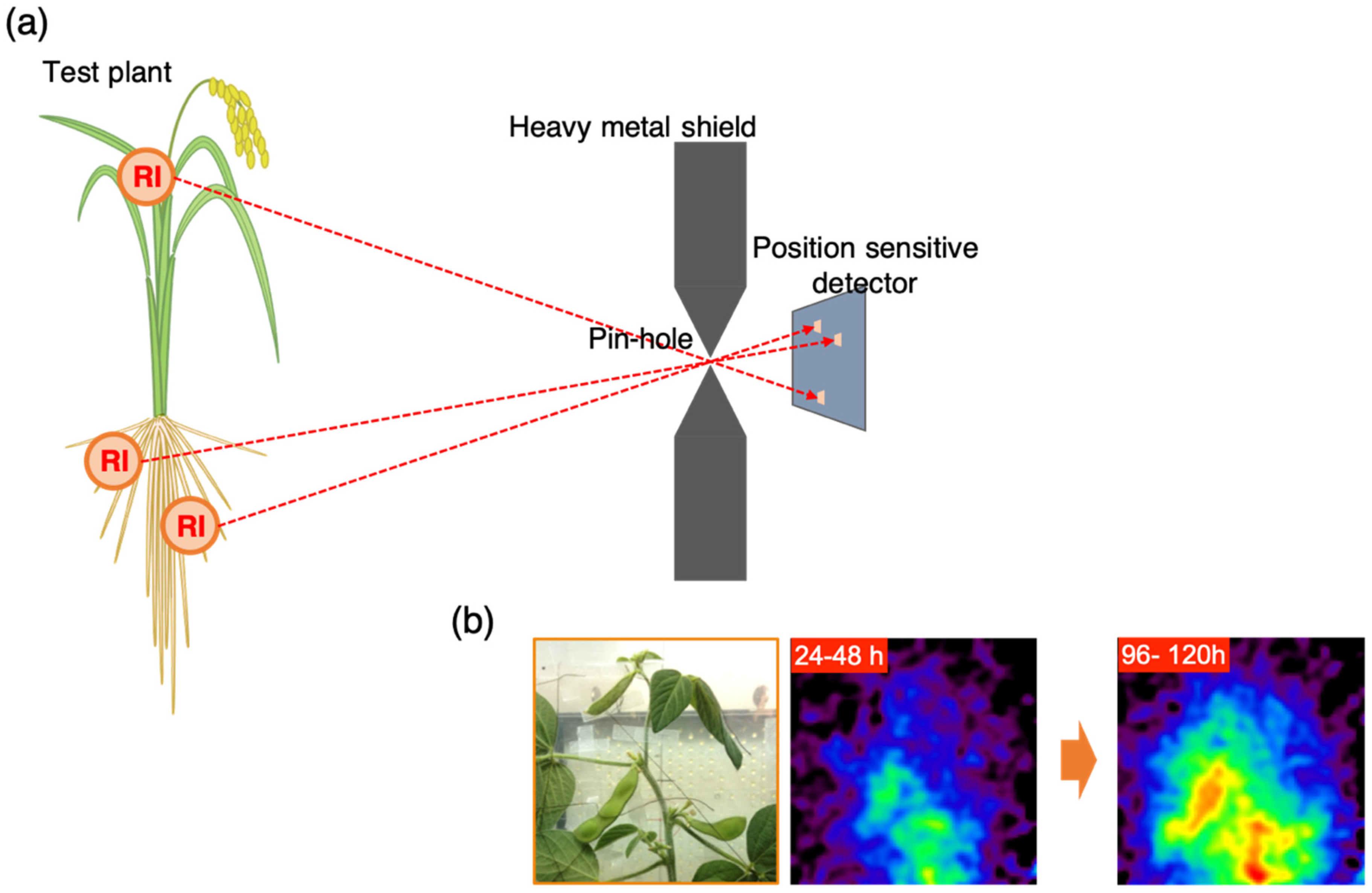
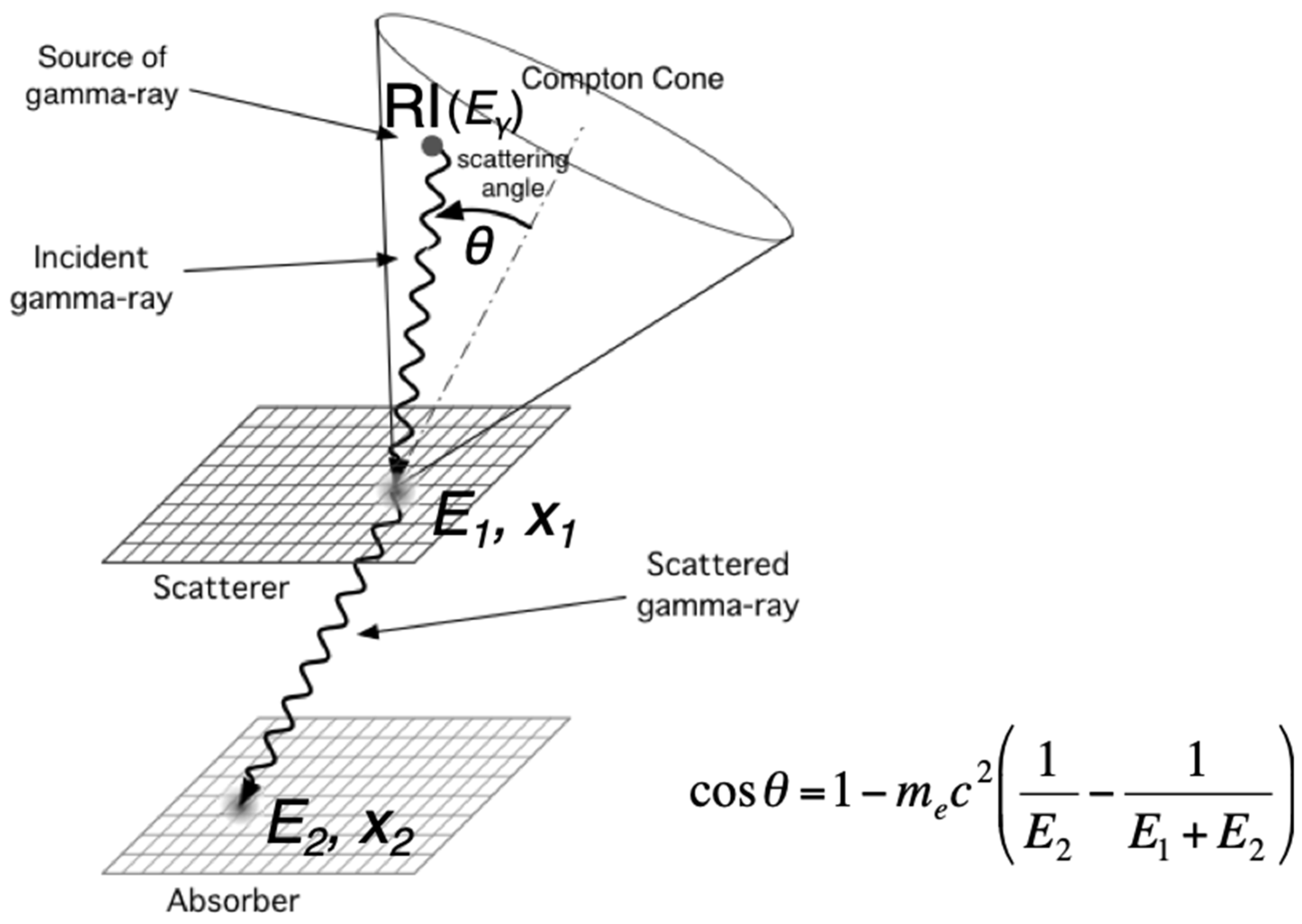
2.3. Gamma Camera Imaging
2.4. Prospective Imaging Techniques
3. Conclusions
Funding
Conflicts of Interest
References
- Rennie, E.A.; Turgeon, R. A comprehensive picture of phloem loading strategies. Proc. Natl. Acad. Sci. USA 2009, 106, 14162–14167. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hubeau, M.; Mincke, J.; Vanhove, C.; Gorel, A.P.; Fayolle, A.; Epila, J.; Leroux, O.; Vandenberghe, S.; Steppe, K. 11C-Autoradiographs to Image Phloem Loading. Front. For. Glob. Chang. 2019, 2. [Google Scholar] [CrossRef]
- Hubeau, M.; Steppe, K. Plant-PET Scans: In Vivo Mapping of Xylem and Phloem Functioning. Trends Plant. Sci. 2015, 20, 676–685. [Google Scholar] [CrossRef] [PubMed]
- Schaart, D.R.; van Dam, H.T.; Seifert, S.; Vinke, R.; Dendooven, P.; Löhner, H.; Beekman, F.J. A novel, SiPM-array-based, monolithic scintillator detector for PET. Phys. Med. Biol. 2009, 54, 3501–3512. [Google Scholar] [CrossRef] [PubMed]
- Kanno, S.; Rai, H.; Ohya, T.; Hayashi, Y.; Tanoi, K.; Nakanishi, T.M. Real-time imaging of radioisotope labeled compounds in a living plant. J. Radioanal. Nucl. Chem. 2007, 272, 565–570. [Google Scholar] [CrossRef]
- Nakanishi, T.M.; Yamawaki, M.; Kannno, S.; Nihei, N.; Masuda, S.; Tanoi, K. Real-time imaging of ion uptake from root to above-ground part of the plant using conventional beta-ray emitters. J. Radioanal. Nucl. Chem. 2009, 282, 265. [Google Scholar] [CrossRef]
- Kanno, S.; Arrighi, J.-F.; Chiarenza, S.; Bayle, V.; Berthomé, R.; Péret, B.; Javot, H.; Delannoy, E.; Marin, E.; Nakanishi, T.M.; et al. A novel role for the root cap in phosphate uptake and homeostasis. eLife 2016, 5, e14577. [Google Scholar] [CrossRef] [PubMed]
- Sugita, R.; Kobayashi, N.I.; Hirose, A.; Tanoi, K.; Nakanishi, T.M. Evaluation ofin vivodetection properties of 22Na, 65Zn, 86Rb, 109Cd and 137Cs in plant tissues using real-time radioisotope imaging system. Phys. Med. Biol. 2014, 59, 837–851. [Google Scholar] [CrossRef] [PubMed]
- Sugita, R.; Kobayashi, N.I.; Hirose, A.; Ohmae, Y.; Tanoi, K.; Nakanishi, T.M. Nondestructive real-time radioisotope imaging system for visualizing 14C-labeled chemicals supplied as CO2 in plants using Arabidopsis thaliana. J. Radioanal. Nucl. Chem. 2013, 298, 1411–1416. [Google Scholar] [CrossRef]
- Sugita, R.; Sugahara, K.; Kobayashi, N.I.; Hirose, A.; Nakanishi, T.M.; Furuta, E.; Sensui, M.; Tanoi, K. Evaluation of plastic scintillators for live imaging of 14C-labeled photosynthate movement in plants. J. Radioanal. Nucl. Chem. 2018, 318, 579–584. [Google Scholar] [CrossRef]
- Sugita, R.; Kobayashi, N.I.; Hirose, A.; Saito, T.; Iwata, R.; Tanoi, K.; Nakanishi, T.M. Visualization of Uptake of Mineral Elements and the Dynamics of Photosynthates in Arabidopsis by a Newly Developed Real-Time Radioisotope Imaging System (RRIS). Plant. Cell Physiol. 2016, 57, 743–753. [Google Scholar] [CrossRef] [PubMed]
- Sugita, R.; Kobayashi, N.I.; Saito, T.; Hirose, A.; Iwata, R.; Tanoi, K.; Nakanishi, T.M. Quantitative Analysis of 28Mg in Arabidopsis using Real-time Radioisotope Imaging System(RRIS). Radioisotopes 2014, 63, 227–237. [Google Scholar] [CrossRef]
- Sugita, R.; Kobayashi, N.I.; Hirose, A.; Iwata, R.; Suzuki, H.; Tanoi, K.; Nakanishi, T.M. Visualization of how light changes affect ion movement in rice plants using a real-time radioisotope imaging system. J. Radioanal. Nucl. Chem. 2017, 312, 717–723. [Google Scholar] [CrossRef]
- Yamawaki, M.; Kanno, S.; Ishibashi, H.; Noda, A.; Hirose, A.; Tanoi, K.; Nakanishi, T.M. The development of real-time RI imaging system for plant under light environment. J. Radioanal. Nucl. Chem. 2009, 282, 275. [Google Scholar] [CrossRef]
- Nussaume, L.; Kanno, S.; Javot, H.; Marin, E.; Nakanishi, T.M.; Thibaud, M.-C. Phosphate Import in Plants: Focus on the PHT1 Transporters. Front. Plant. Sci. 2011, 2. [Google Scholar] [CrossRef] [PubMed]
- Sugahara, K.; Sugita, R.; Kobayashi, N.I.; Hirose, A.; Nakanishi, T.M.; Furuta, E.; Sensui, M.; Tanoi, K. Plastic Scintillators Enable Live Imaging of 32P-labelled Phosphorus Movement in Large Plants. Radioisotopes 2019, 68, 73–82. [Google Scholar] [CrossRef]
- Kanno, S.; Cuyas, L.; Javot, H.; Bligny, R.; Gout, E.; Dartevelle, T.; Hanchi, M.; Nakanishi, T.M.; Thibaud, M.-C.; Nussaume, L. Performance and Limitations of Phosphate Quantification: Guidelines for Plant Biologists. Plant. Cell Physiol. 2016, 57, 690–706. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kanno, S.; Yamawaki, M.; Ishibashi, H.; Kobayashi, N.I.; Hirose, A.; Tanoi, K.; Nussaume, L.; Nakanishi, T.M. Development of real-time radioisotope imaging systems for plant nutrient uptake studies. Philos. Trans. R. Soc. B Biol. Sci. 2012, 367, 1501–1508. [Google Scholar] [CrossRef] [PubMed]
- Aramaki, T.; Sugita, R.; Hirose, A.; Kobayashi, N.I.; Tanoi, K.; Nakanishi, T.M. Application of 42K to Arabidopsis Tissues Using Real-Time Radioisotope Imaging System(RRIS). Radioisotopes 2015, 64, 169–176. [Google Scholar] [CrossRef]
- Hirose, A.; Yamawaki, M.; Kanno, S.; Igarashi, S.; Sugita, R.; Ohmae, Y.; Tanoi, K.; Nakanishi, T.M. Development of a 14C detectable real-time radioisotope imaging system for plants under intermittent light environment. J. Radioanal. Nucl. Chem. 2013, 296, 417–422. [Google Scholar] [CrossRef]
- Kobayashi, N.I.; Tanoi, K.; Kanno, S.; Nakanishi, T.M. Analysis of the Iron Movement in the Root Tip Part Using Real-time Imaging System. Radioisotopes 2012, 61, 121–128. [Google Scholar] [CrossRef] [Green Version]
- Yamawaki, M.; Hirose, A.; Kanno, S.; Ishibashi, H.; Noda, A.; Tanoi, K.; Nakanishi, T.M. Evaluation of 109Cd Detection Performance of a Real-Time RI Imaging System for Plant Research. Radioisotopes 2010, 59, 155–162. [Google Scholar] [CrossRef]
- Sugita, R.; Kobayashi, N.I.; Hirose, A.; Tanoi, K.; Nakanishi, T.M. Visualization of Ion Transport in Plants. In Agricultural Implications of the Fukushima Nuclear Accident (III): After 7 Years; Nakanishi, T.M., O’Brien, M., Tanoi, K., Eds.; Springer: Singapore, 2019; pp. 221–231. [Google Scholar] [Green Version]
- McKay, R.M.L.; Palmer, G.R.; Ma, X.P.; Layzell, D.B.; McKee, B.T.A. The use of positron emission tomography for studies of long-distance transport in plants: uptake and transport of 18F. Plant. Cell Environ. 1988, 11, 851–861. [Google Scholar] [CrossRef]
- Kume, T.; Matsuhashi, S.; Shimazu, M.; Ito, H.; Fujimura, T.; Adachi, K.; Uchida, H.; Shigeta, N.; Matsuoka, H.; Osa, A.; et al. Uptake and transport of positron-emitting tracer (18F) in plants. Appl. Radiat. Isot. 1997, 48, 1035–1043. [Google Scholar] [CrossRef]
- Fujimaki, S.; Suzui, N.; Ishioka, N.S.; Kawachi, N.; Ito, S.; Chino, M.; Nakamura, S. Tracing cadmium from culture to spikelet: Non-invasive imaging and quantitative characterization of absorption, transport and accumulation of cadmium in an intact rice plant. Plant. Physiol. 2010, 152, 1796–1806. [Google Scholar] [CrossRef]
- Ishikawa, S.; Suzui, N.; Ito-Tanabata, S.; Ishii, S.; Igura, M.; Abe, T.; Kuramata, M.; Kawachi, N.; Fujimaki, S. Real-time imaging and analysis of differences in cadmium dynamics in rice cultivars (Oryza sativa) using positron-emitting107Cd tracer. BMC Plant. Biol. 2011, 11, 172. [Google Scholar] [CrossRef] [PubMed]
- Fontanili, L.; Lancilli, C.; Suzui, N.; Dendena, B.; Yin, Y.-G.; Ferri, A.; Ishii, S.; Kawachi, N.; Lucchini, G.; Fujimaki, S.; et al. Kinetic Analysis of Zinc/Cadmium Reciprocal Competitions Suggests a Possible Zn-Insensitive Pathway for Root-to-Shoot Cadmium Translocation in Rice. Rice 2016, 9, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Nakamura, S.-I.; Suzui, N.; Nagasaka, T.; Komatsu, F.; Ishioka, N.S.; Ito-Tanabata, S.; Kawachi, N.; Rai, H.; Hattori, H.; Chino, M.; et al. Application of glutathione to roots selectively inhibits cadmium transport from roots to shoots in oilseed rape. J. Exp. Bot. 2013, 64, 1073–1081. [Google Scholar] [CrossRef]
- Yoshihara, T.; Suzui, N.; Ishii, S.; Kitazaki, M.; Yamazaki, H.; Kitazaki, K.; Kawachi, N.; Yin, Y.-G.; Ito-Tanabata, S.; Hashida, S.-N.; et al. A kinetic analysis of cadmium accumulation in a Cd Hyper-accumulator Fern, Athyrium Yokoscense and tobacco plants. Plant. Cell Environ. 2014, 37, 1086–1096. [Google Scholar] [CrossRef]
- Hu, P.; Yin, Y.-G.; Ishikawa, S.; Suzui, N.; Kawachi, N.; Fujimaki, S.; Igura, M.; Yuan, C.; Huang, J.; Li, Z.; et al. Nitrate facilitates cadmium uptake, transport and accumulation in the hyperaccumulator Sedum plumbizincicola. Environ. Sci. Pollut. Res. 2013, 20, 6306–6316. [Google Scholar] [CrossRef]
- Fujimaki, S.; Maruyama, T.; Suzui, N.; Kawachi, N.; Miwa, E.; Higuchi, K. Base to Tip and Long-Distance Transport of Sodium in the Root of Common Reed [Phragmites australis (Cav.) Trin. ex Steud.] at Steady State Under Constant High-Salt Conditions. Plant. Cell Physiol. 2015, 56, 943–950. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suzuki, M.; Takahashi, M.; Tsukamoto, T.; Watanabe, S.; Matsuhashi, S.; Yazaki, J.; Kishimoto, N.; Kikuchi, S.; Nakanishi, H.; Mori, S. Biosynthesis and secretion of mugineic acid family phytosiderophores in zinc-deficient barley. Plant. J. 2006, 48, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Suzui, N.; Yin, Y.-G.; Ishii, S.; Sekimoto, H.; Kawachi, N. Visualization of zinc dynamics in intact plants using positron imaging of commercially available 65Zn. Plant. Methods 2017, 13, 40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wongkaew, A.; Nakamura, S.-I.; Suzui, N.; Yin, Y.-G.; Ishii, S.; Kawachi, N.; Kojima, K.; Sekimoto, H.; Yokoyama, T.; Ohkama-Ohtsu, N. Elevated glutathione synthesis in leaves contributes to zinc transport from roots to shoots in Arabidopsis. Plant. Sci. 2019, 283, 416–423. [Google Scholar] [CrossRef] [PubMed]
- Kawachi, N.; Yin, Y.-G.; Suzui, N.; Ishii, S.; Yoshihara, T.; Watabe, H.; Yamamoto, S.; Fujimaki, S. Imaging of radiocesium uptake dynamics in a plant body by using a newly developed high-resolution gamma camera. J. Environ. Radioact. 2016, 151, 461–467. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamamoto, S.; Kamada, K.; Yoshikawa, A. Use of YAP(Ce) in the development of high spatial resolution radiation imaging detectors. Radiat. Meas. 2018, 119, 184–191. [Google Scholar] [CrossRef]
- Ando, K.; Yamaguchi, M.; Yamamoto, S.; Toshito, T.; Kawachi, N. Development of a low-energy X-ray camera for the imaging of secondary electron bremsstrahlung x-ray emitted during proton irradiation for range estimation. Phys. Med. Biol. 2017, 62, 5006–5020. [Google Scholar] [CrossRef] [PubMed]
- Kabuki, S.; Hattori, K.; Kohara, R.; Kunieda, E.; Kubo, A.; Kubo, H.; Miuchi, K.; Nakahara, T.; Nagayoshi, T.; Nishimura, H.; et al. Development of Electron Tracking Compton Camera using micro pixel gas chamber for medical imaging. Nucl. Instrum. Methods Phys. Res. Sect. A: Accel. Spectrom. Assoc. Equip. 2007, 580, 1031–1035. [Google Scholar] [CrossRef] [Green Version]
- Takeda, S.; Aono, H.; Okuyama, S.; Ishikawa, S.; Odaka, H.; Watanabe, S.; Kokubun, M.; Takahashi, T.; Nakazawa, K.; Tajima, H.; et al. Experimental Results of the Gamma-Ray Imaging Capability With a Si/CdTe Semiconductor Compton Camera. IEEE Trans. Nucl. Sci. 2009, 56, 783–790. [Google Scholar] [CrossRef]
- Alnaaimi, M.A.; Royle, G.J.; Ghoggali, W.; Banoqitah, E.; Cullum, I.; Speller, R.D. Performance evaluation of a pixellated Ge Compton camera. Phys. Med. Biol. 2011, 56, 3473–3486. [Google Scholar] [CrossRef]
- Motomura, S.; Kanayama, Y.; Hiromura, M.; Fukuchi, T.; Ida, T.; Haba, H.; Watanabe, Y.; Enomoto, S. Improved imaging performance of a semiconductor Compton camera GREI makes for a new methodology to integrate bio-metal analysis and molecular imaging technology in living organisms. J. Anal. At. Spectrom. 2013, 28, 934–939. [Google Scholar] [CrossRef]
- Kishimoto, A.; Kataoka, J.; Taya, T.; Tagawa, L.; Mochizuki, S.; Ohsuka, S.; Nagao, Y.; Kurita, K.; Yamaguchi, M.; Kawachi, N.; et al. First demonstration of multi-color 3-D in vivo imaging using ultra-compact Compton camera. Sci. Rep. 2017, 7, 2110. [Google Scholar] [CrossRef] [PubMed]
- Nieves-Cordones, M.; Mohamed, S.; Tanoi, K.; Kobayashi, N.I.; Takagi, K.; Vernet, A.; Guiderdoni, E.; Périn, C.; Sentenac, H.; Véry, A.-A. Production of low-Cs+ rice plants by inactivation of the K+ transporter OsHAK1 with the CRISPR-Cas system. Plant. J. 2017, 92, 43–56. [Google Scholar] [CrossRef] [PubMed]
- Rai, H.; Yokoyama, S.; Satoh-Nagasawa, N.; Furukawa, J.; Nomi, T.; Ito, Y.; Fujimura, S.; Takahashi, H.; Suzuki, R.; Yousra, E.; et al. Cesium Uptake by Rice Roots Largely Depends Upon a Single Gene, HAK1, Which Encodes a Potassium Transporter. Plant. Cell Physiol. 2017, 58, 1486–1493. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamamoto, S.; Ogata, Y.; Kawachi, N.; Suzui, N.; Yin, Y.-G.; Fujimaki, S. Ultra-high resolution of radiocesium distribution detection based on Cherenkov light imaging. Nucl. Instrum. Methods Phys. Res. Sect. A: Accel. Spectrom. Assoc. Equip. 2015, 777, 102–109. [Google Scholar] [CrossRef]
- Kurita, K.; Suzui, N.; Yin, Y.-G.; Ishii, S.; Watabe, H.; Yamamoto, S.; Kawachi, N. Development of a Cherenkov light imaging system for studying the dynamics of radiocesium in plants. J. Nucl. Sci. Technol. 2017, 54, 662–667. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suzui, N.; Tanoi, K.; Furukawa, J.; Kawachi, N. Recent Advances in Radioisotope Imaging Technology for Plant Science Research in Japan. Quantum Beam Sci. 2019, 3, 18. https://doi.org/10.3390/qubs3030018
Suzui N, Tanoi K, Furukawa J, Kawachi N. Recent Advances in Radioisotope Imaging Technology for Plant Science Research in Japan. Quantum Beam Science. 2019; 3(3):18. https://doi.org/10.3390/qubs3030018
Chicago/Turabian StyleSuzui, Nobuo, Keitaro Tanoi, Jun Furukawa, and Naoki Kawachi. 2019. "Recent Advances in Radioisotope Imaging Technology for Plant Science Research in Japan" Quantum Beam Science 3, no. 3: 18. https://doi.org/10.3390/qubs3030018




