Prediction Error Stabilization and Long-Term Standard Results with a Monofocal Intraocular Lens
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects and Procedures
2.2. Intraocular Lens and Surgery
2.3. Statistical Analysis
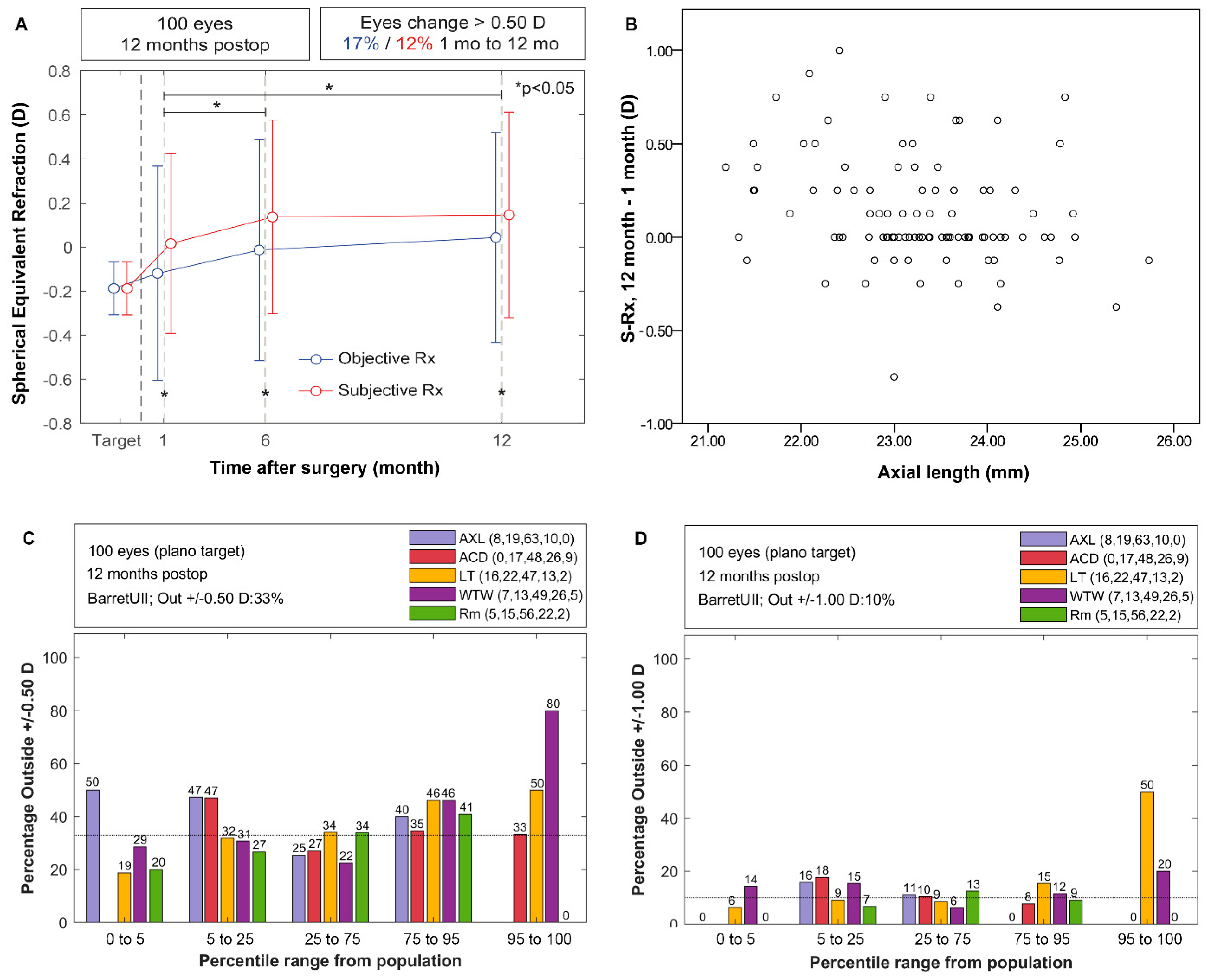
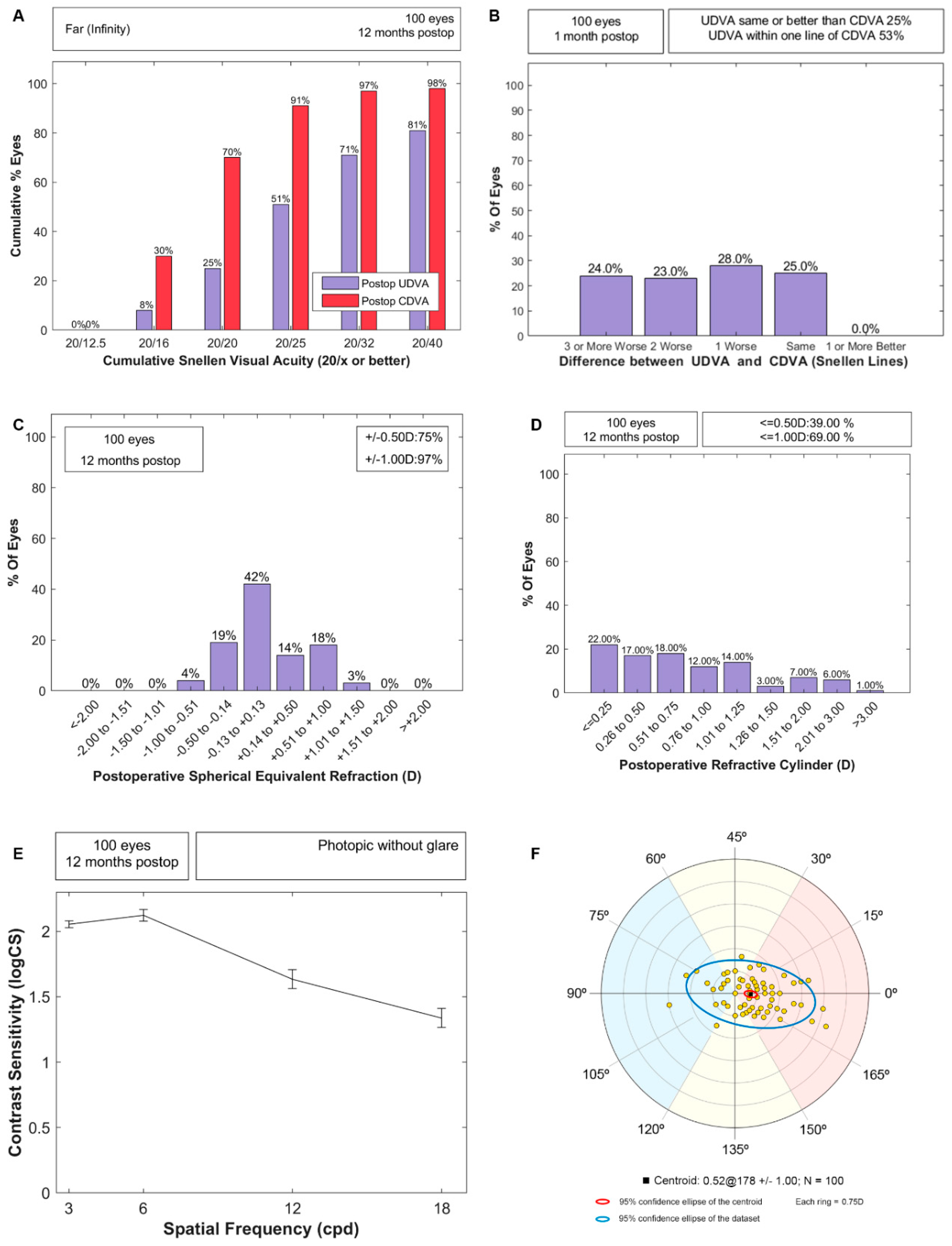
3. Results
3.1. Prediction Error
3.2. Standard Results
3.2.1. Safety
3.2.2. Efficacy and Predictability
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Szakáts, I.; Sebestyén, M.; Tóth, É.; Purebl, G. Dry Eye Symptoms, Patient-Reported Visual Functioning, and Health Anxiety Influencing Patient Satisfaction After Cataract Surgery. Curr. Eye Res. 2017, 42, 832–836. [Google Scholar] [CrossRef] [PubMed]
- Kinard, K.; Jarstad, A.; Olson, R.J. Correlation of Visual Quality with Satisfaction and Function in a Normal Cohort of Pseudophakic Patients. J. Cataract. Refract. Surg. 2013, 39, 590–597. [Google Scholar] [CrossRef]
- Kirwan, C.; Nolan, J.M.; Stack, J.; Moore, T.C.B.; Beatty, S. Determinants of Patient Satisfaction and Function Related to Vision Following Cataract Surgery in Eyes with No Visually Consequential Ocular Co-Morbidity. Graefe’s Arch. Clin. Exp. Ophthalmol. 2015, 253, 1735–1744. [Google Scholar] [CrossRef]
- Hawker, M.J.; Madge, S.N.; Baddeley, P.A.; Perry, S.R. Refractive Expectations of Patients Having Cataract Surgery. J. Cataract. Refract. Surg. 2005, 31, 1970–1975. [Google Scholar] [CrossRef] [PubMed]
- Hoffer, K.J. Accuracy of Ultrasound Intraocular Lens Calculation. Arch. Ophthalmol. 1981, 10, 1819–1823. [Google Scholar] [CrossRef] [PubMed]
- Holladay, J.T.; Wilcox, R.R.; Koch, D.D.; Wang, L. Review and Recommendations for Univariate Statistical Analysis of Spherical Equivalent Prediction Error for IOL Power Calculations. J. Cataract. Refract. Surg. 2021, 47, 65–77. [Google Scholar] [CrossRef]
- Hoffer, K.J.; Savini, G. Update on Intraocular Lens Power Calculation Study Protocols the Better Way to Design and Report Clinical Trials. Ophthalmology 2021, 128, e115–e120. [Google Scholar] [CrossRef]
- Wang, L.; Koch, D.D.; Hill, W.; Abulafia, A. Pursuing Perfection in Intraocular Lens Calculations: III. Criteria for Analyzing Outcomes. J. Cataract. Refract. Surg. 2017, 43, 999–1002. [Google Scholar] [CrossRef]
- Hoffer, K.J.; Aramberri, J.; Haigis, W.; Olsen, T.; Savini, G.; Shammas, H.J.; Bentow, S. Protocols for Studies of Intraocular Lens Formula Accuracy. Am. J. Ophthalmol. 2015, 160, 403–405.e1. [Google Scholar] [CrossRef]
- Reinstein, D.Z.; Archer, T.J.; Srinivasan, S.; Mamalis, N.; Kohnen, T.; Dupps, W.J.; Randleman, J.B. Standard for Reporting Refractive Outcomes of Intraocular Lens–Based Refractive Surgery. J. Cataract. Refract. Surg. 2017, 43, 435–439. [Google Scholar] [CrossRef]
- Fernández, J.; Rodríguez-Vallejo, M.; Piñero, D.P. Re: Hoffer et al. Update on Intraocular Lens Power Calculation Study Protocols: The Better Way to Design and Report Clinical Trials. Ophthalmology 2020, 128, e17–e18. [Google Scholar] [CrossRef] [PubMed]
- Vounotrypidis, E.; Haralanova, V.; Muth, D.R.; Wertheimer, C.; Shajari, M.; Wolf, A.; Priglinger, S.; Mayer, W.J. Accuracy of SS-OCT Biometry Compared with Partial Coherence Interferometry Biometry for Combined Phacovitrectomy with Internal Limiting Membrane Peeling. J. Cataract. Refract. Surg. 2019, 45, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Satou, T.; Shimizu, K.; Tsunehiro, S.; Igarashi, A.; Kato, S.; Koshimizu, M.; Niida, T. Development of a New Intraocular Lens Power Calculation Method Based on Lens Position Estimated with Optical Coherence Tomography. Sci. Rep. 2020, 10, 6501. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, K.; Ogawa, S.; Yoshida, M.; Yoshimura, K. Influence of Patient Age on Intraocular Lens Power Prediction Error. Am. J. Ophthalmol. 2016, 170, 232–237. [Google Scholar] [CrossRef][Green Version]
- Fernández, J.; Rodríguez-Vallejo, M.; Poyales, F.; Burguera, N.; Garzón, N. New Method to Assess the Accuracy of Intraocular Lens Power Calculation Formulas According to Ocular Biometric Parameters. J. Cataract. Refract. Surg. 2020, 46, 849–856. [Google Scholar] [CrossRef]
- Rodríguez-Vallejo, M. Refractive Analysis. 1.0.5. A Matlab Toolbox for the Analysis of Refractive Results in Anterior Segment Surgery. Available online: https://www.test-eye.com/en/refractive-analysis (accessed on 22 November 2021).
- Caglar, C.; Batur, M.; Eser, E.; Demir, H.; Yaşar, T. The Stabilization Time of Ocular Measurements after Cataract Surgery. Semin. Ophthalmol. 2016, 32, 1–6. [Google Scholar] [CrossRef]
- Carracedo, G.; Carpena-Torres, C.; Serramito, M.; Batres-Valderas, L.; Gonzalez-Bergaz, A. Comparison Between Aberrometry-Based Binocular Refraction and Subjective Refraction. Transl. Vis. Sci. Technol. 2018, 7, 11. [Google Scholar] [CrossRef]
- Koeppl, C.; Findl, O.; Kriechbaum, K.; Sacu, S.; Drexler, W. Change in IOL Position and Capsular Bag Size with an Angulated Intraocular Lens Early after Cataract Surgery. J. Cataract. Refract. Surg. 2005, 31, 348–353. [Google Scholar] [CrossRef]
- Petternel, V.; Menapace, R.; Findl, O.; Kiss, B.; Wirtitsch, M.; Rainer, G.; Drexler, W. Effect of Optic Edge Design and Haptic Angulation on Postoperative Intraocular Lens Position Change. J. Cataract. Refract. Surg. 2004, 30, 52–57. [Google Scholar] [CrossRef]
- Wirtitsch, M.G.; Findl, O.; Menapace, R.; Kriechbaum, K.; Koeppl, C.; Buehl, W.; Drexler, W. Effect of Haptic Design on Change in Axial Lens Position after Cataract Surgery. J. Cataract. Refract. Surg. 2004, 30, 45–51. [Google Scholar] [CrossRef]
- Hienert, J.; Hirnschall, N.; Ruiss, M.; Ullrich, M.; Zwickl, H.; Findl, O. Prospective Study to Compare Axial Position Stability Following Fellow-Eye Implantation of Two Distinct Intraocular Lens Designs. J. Cataract. Refract. Surg. 2021, 47, 999–1005. [Google Scholar] [CrossRef]
- Cabeza-Gil, I.; Pérez-Gracia, J.; Remón, L.; Calvo, B. Effect of Haptic Geometry in C-Loop Intraocular Lenses on Optical Quality. J. Mech. Behav. Biomed. Mater. 2021, 114, 104165. [Google Scholar] [CrossRef]
- Melles, R.B.; Holladay, J.T.; Chang, W.J. Accuracy of Intraocular Lens Calculation Formulas. Ophthalmology 2018, 125, 169–178. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Lee, S.H.; Kim, N.R.; Chin, H.S.; Jung, J.W. Accuracy of Intraocular Lens Power Calculation Formulas Using a Swept-Source Optical Biometer. PLoS ONE 2020, 15, e0227638. [Google Scholar] [CrossRef]
- Donachie, P.H.J.; Sparrow, J.M.; Buchan, J.C. National Ophthalmology Database Audit. Feasibility Study of Post-Cataract Posterior Capsule Opacification 2021. Available online: https://www.nodaudit.org.uk/u/docs/20/rijbxkcubs/RCOphth%20NOD%20PCO%20Report%202021.pdf (accessed on 22 November 2021).
- Burguera-Giménez, N.; García-Lázaro, S.; España-Gregori, E.; Gallego-Pinazo, R.; Burguera-Giménez, N.; Rodríguez-Vallejo, M.; Jonna, G. Multimodal Evaluation of Visual Function in Geographic Atrophy versus Normal Eyes. Clin. Ophthalmol. 2020, 14, 1533–1545. [Google Scholar] [CrossRef]
- García-Pérez, J.L.; Gros-Otero, J.; Sánchez-Ramos, C.; Blázquez, V.; Contreras, I. Short Term Visual Outcomes of a New Trifocal Intraocular Lens. BMC Ophthalmol. 2017, 17, 72. [Google Scholar] [CrossRef]
- Rementería-Capelo, L.A.; Contreras, I.; García-Perez, J.L.; Carrillo, V.; Gros-Otero, J.; Ruiz-Alcocer, J. Tolerance to Residual Refractive Errors After Trifocal and Trifocal Toric Intraocular Lens Implantation. Eye Contact Lens Sci. Clin. Pract. 2020, 47, 213–218. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Vallejo, M.; Remón, L.; Monsoriu, J.A.; Furlan, W.D. Designing a New Test for Contrast Sensitivity Function Measurement with IPad. J. Optom. 2015, 8, 101–108. [Google Scholar] [CrossRef]
- Li, S.; Hu, Y.; Guo, R.; Shao, Y.; Zhao, J.; Zhang, J.; Wang, J. The Effects of Different Shapes of Capsulorrhexis on Postoperative Refractive Outcomes and the Effective Position of the Intraocular Lens in Cataract Surgery. BMC Ophthalmol. 2019, 19, 59. [Google Scholar] [CrossRef] [PubMed]
| Variable | Mean ± SD | Median [IQR] | Range [Min–Max] |
|---|---|---|---|
| Age | 73 ± 7 | 74 (10) | 52–90 |
| Anterior Chamber Depth (mm) | 3.25 ± 0.38 | 3.27 (0.49) | 2.46–4.57 |
| Axial Length (mm) | 23.28 ± 0.93 | 23.29 (1.23) | 21.19–25.73 |
| Average Corneal Power (D) | 43.91 ± 1.38 | 43.87 (1.90) | 41.10–47.82 |
| Corneal Astigmatism (D) | 0.79 ± 0.59 | 0.69 (0.60) | 0.02–3.74 |
| Corneal diameter (mm) | 11.97 ± 0.43 | 12 (0.58) | 10.64–13.10 |
| Lens Thickness (mm) | 4.46 ± 0.45 | 4.5 (0.64) | 3.31–5.57 |
| Intraocular Lens Power (D) | 22.48 ± 2.63 | 22.5 (3) | 14–31 |
| Target Refraction (D) | −0.19 ± 0.12 | −0.17 (0.19) | −0.52–0.06 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Macías-Murelaga, B.; Garay-Aramburu, G.; Bergado-Mijangos, R.; Coello-Ojeda, D.; Ozaeta, I.; Garcia-Gómez, P.J.; Garrido-Fierro, J.; Rodríguez-Vallejo, M.; Fernández, J. Prediction Error Stabilization and Long-Term Standard Results with a Monofocal Intraocular Lens. Vision 2022, 6, 5. https://doi.org/10.3390/vision6010005
Macías-Murelaga B, Garay-Aramburu G, Bergado-Mijangos R, Coello-Ojeda D, Ozaeta I, Garcia-Gómez PJ, Garrido-Fierro J, Rodríguez-Vallejo M, Fernández J. Prediction Error Stabilization and Long-Term Standard Results with a Monofocal Intraocular Lens. Vision. 2022; 6(1):5. https://doi.org/10.3390/vision6010005
Chicago/Turabian StyleMacías-Murelaga, Beatríz, Gonzaga Garay-Aramburu, Roberto Bergado-Mijangos, Daniel Coello-Ojeda, Itziar Ozaeta, Pio Jésus Garcia-Gómez, Jesús Garrido-Fierro, Manuel Rodríguez-Vallejo, and Joaquín Fernández. 2022. "Prediction Error Stabilization and Long-Term Standard Results with a Monofocal Intraocular Lens" Vision 6, no. 1: 5. https://doi.org/10.3390/vision6010005
APA StyleMacías-Murelaga, B., Garay-Aramburu, G., Bergado-Mijangos, R., Coello-Ojeda, D., Ozaeta, I., Garcia-Gómez, P. J., Garrido-Fierro, J., Rodríguez-Vallejo, M., & Fernández, J. (2022). Prediction Error Stabilization and Long-Term Standard Results with a Monofocal Intraocular Lens. Vision, 6(1), 5. https://doi.org/10.3390/vision6010005








