Semantic Expectation Effects on Object Detection: Using Figure Assignment to Elucidate Mechanisms
Abstract
1. Introduction
2. The Present Experiments
3. Materials and Methods
3.1. Participants
3.1.1. Control Experiments
3.1.2. Study 1: Invalid DSC Labels
3.1.3. Study 2: Invalid SSC Labels
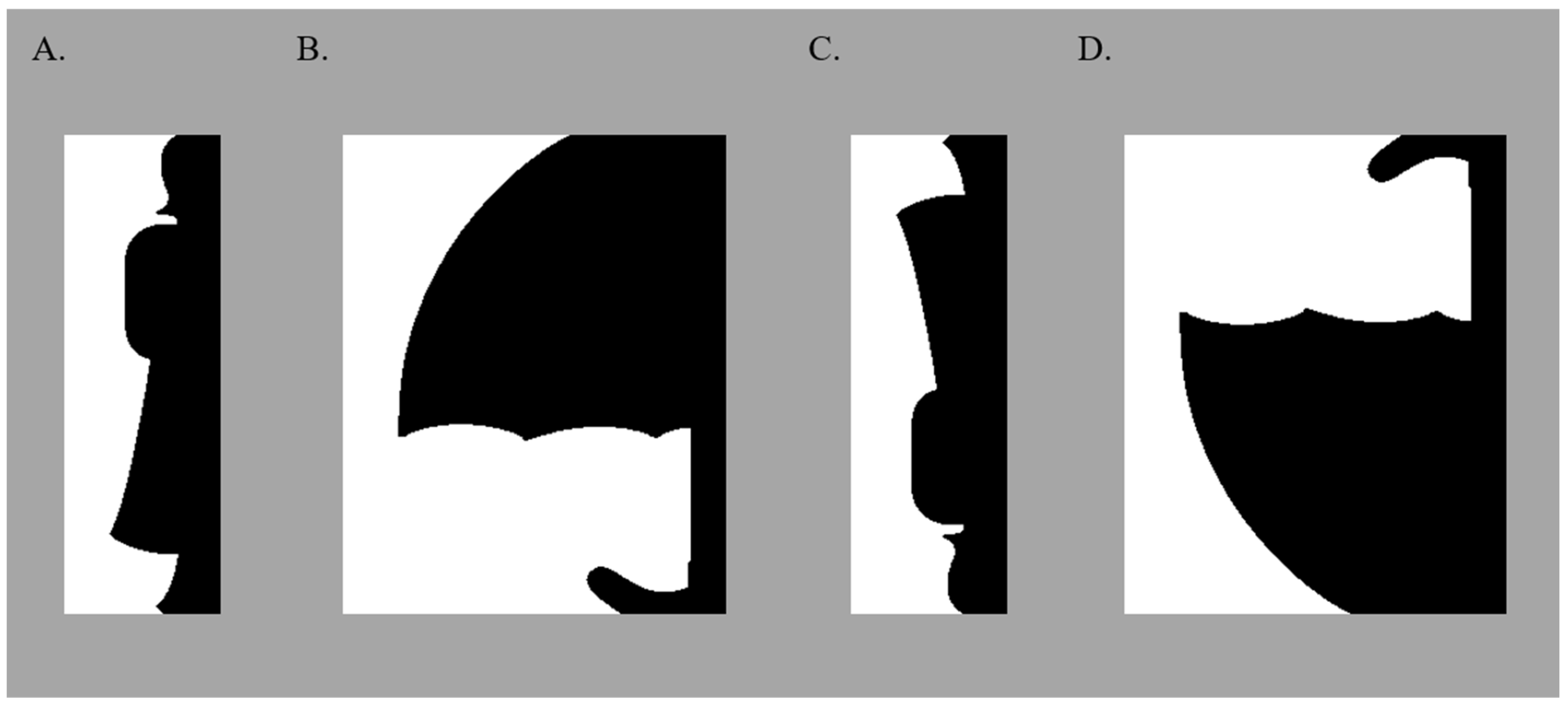
3.2. Apparatus and Stimuli
3.3. Test Displays
3.4. Labels
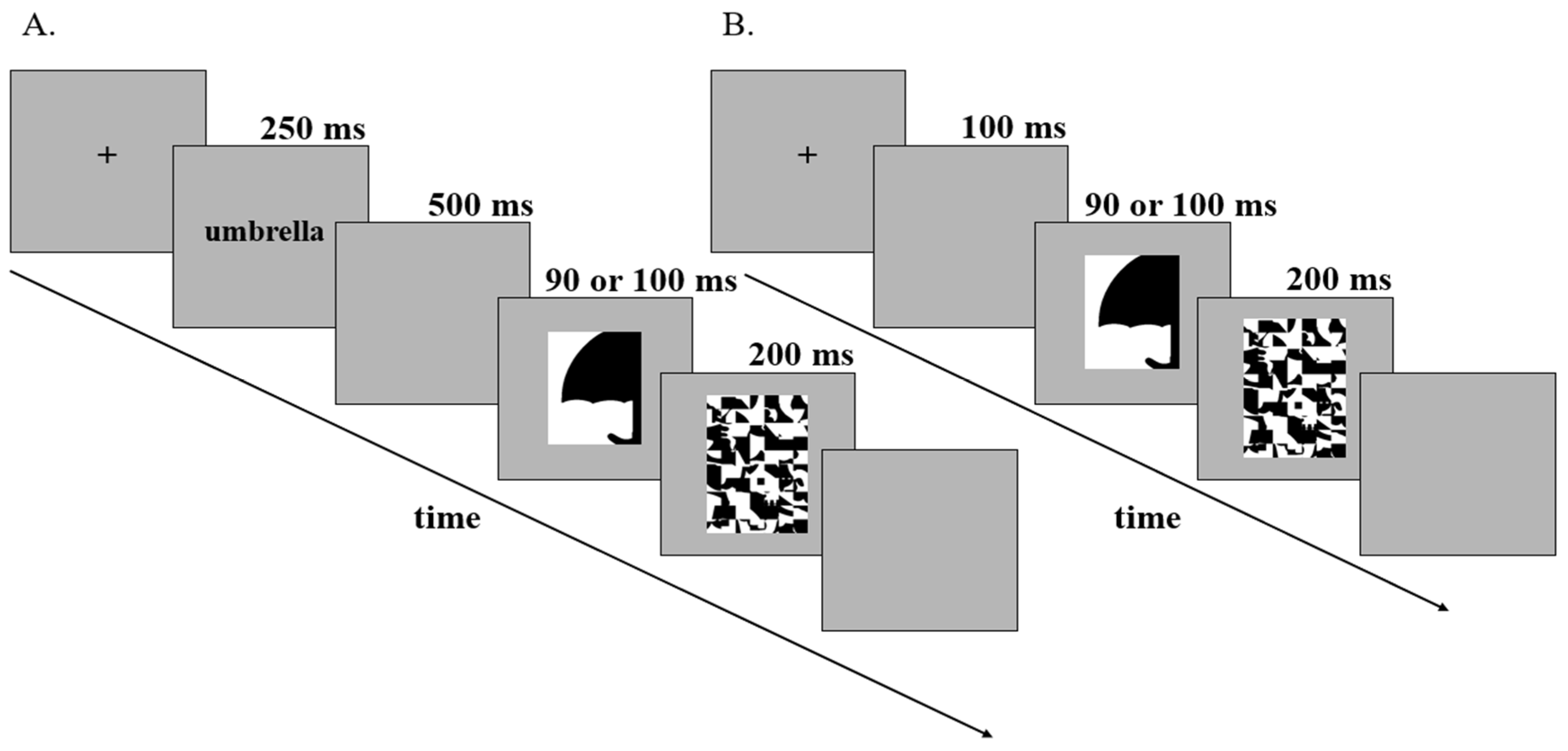
3.5. Design and Procedure
3.6. Trial Structure
4. Data Analysis Methods
5. Results
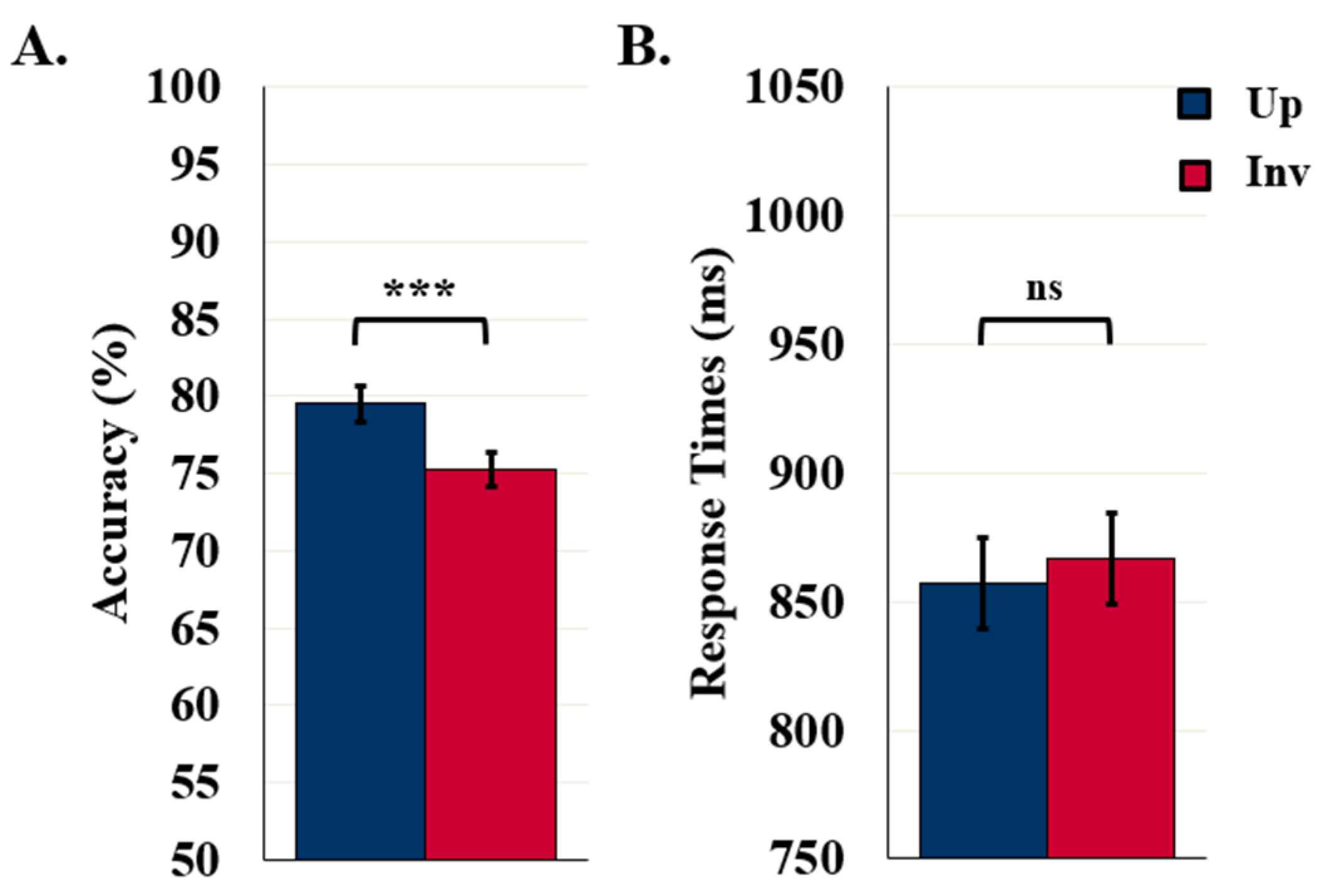
5.1. Control Experiments: 90- and 100-ms Display Durations
5.1.1. Detection Accuracy
5.1.2. Response Times
5.2. Study 1: Invalid DSC Labels
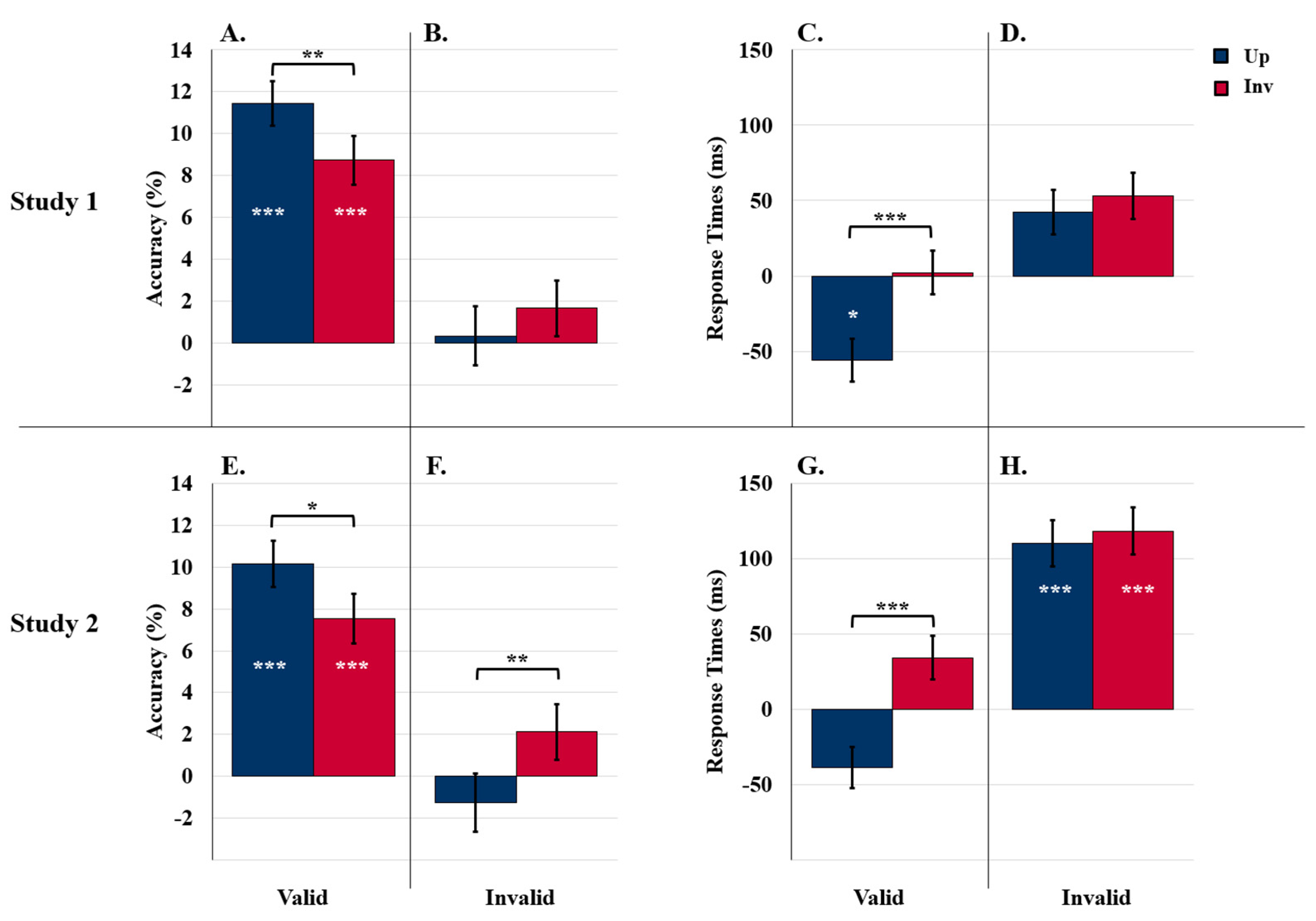
5.2.1. Detection Accuracy
5.2.2. Response Times
5.2.3. Comparison of Study 1 Results to Control Results
Accuracy
Response Times
5.2.4. Study 1 Summary
5.3. Study 2: Invalid SSC Labels
5.3.1. Detection Accuracy
5.3.2. Response Times
5.3.3. Comparison of Study 2 Results to Control Results
Accuracy
Response Times
6. Comparing Study 1 and Study 2
6.1. Detection Accuracy
6.2. Response Time
7. Discussion
8. Open Questions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
List of Stimuli and Labels
| Stimulus/Valid Label | Study 1: Invalid DSC Label | Study 2: Invalid SSC Label |
| anchor | spider | candle |
| axe | ant | mop |
| bell | fish | book |
| boot | frog | oven |
| bulb | clam | rake |
| butterfly | submarine | blueberry |
| cow | toy | log |
| dog | bed | hay |
| eagle | robot | peach |
| elephant | envelope | broccoli |
| face | door | fire |
| faucet | gopher | podium |
| flower | wallet | turkey |
| foot | cake | corn |
| guitar | turkey | needle |
| hand | road | fish |
| horse | paper | lemon |
| house | honey | watch |
| hydrant | piranha | spatula |
| kettle | jaguar | zipper |
| lamp | bush | drum |
| leaf | glue | swan |
| owl | rag | pea |
| palm tree | blue jean | rain drop |
| pig | hat | ivy |
| pineapple | cardboard | alligator |
| rhino | pedal | mango |
| snowman | pelican | band-aid |
| toilet | tongue | button |
| train | river | radio |
| tree | film | goat |
| trumpet | lettuce | compass |
| umbrella | squirrel | envelope |
| watering can | electric eel | baseball bat |
| wineglass | persimmon | thumbtack |
| woman | money | shark |
References
- Block, N. If perception is probabilistic, why does it not seem probabilistic? Philos. Trans. R. Soc. B Biol. Sci. 2018, 373, 20170341. [Google Scholar] [CrossRef] [PubMed]
- Fodor, J. Observation reconsidered. Philos. Sci. 1984, 51, 23–43. [Google Scholar] [CrossRef]
- Gottschaldt, K. Gestalt factors and repetition. In A Source Book of Gestalt Psychology; Kegan Paul, Trench, Trubner & Company: London, UK, 1938. [Google Scholar] [CrossRef]
- Koffka, K. Principles of Gestalt Psychology, International Library of Psychology. In Philosophy and Scientific Method; Routledge: London, UK, 1935. [Google Scholar] [CrossRef]
- Köhler, W. Gestalt Psychology; New American Library: New York, NY, USA, 1929. [Google Scholar]
- Peterson, M.A.; Cacciamani, L.; Mojica, A.J.; Sanguinetti, J.L. Meaning can be accessed for the ground side of a figure. Gestalt. Theory 2012, 34, 297–314. [Google Scholar]
- Pinna, B. New Gestalt principles of perceptual organization: An extension from grouping to shape and meaning. Gestalt Theory 2010, 32, 11. [Google Scholar]
- Rosenthal, V. Microgenesis, Immediate Experience and Visual Processes in Reading. In Seeing, Thinking and Knowing; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2004; pp. 221–243. [Google Scholar] [CrossRef]
- Sander, F. Structure, totality of experience, and Gestalt. In Psychologies of 1930; Clark University Press: Worcester, MA, USA, 1930; pp. 188–204. [Google Scholar]
- Wertheimer, M. A brief introduction to gestalt, identifying key theories and principles. Psychol. Forsch. 1923, 4, 301–350. [Google Scholar] [CrossRef]
- Peterson, M.A.; Harvey, E.M.; Weidenbacher, H.J. Shape recognition contributions to figure-ground reversal: Which route counts? J. Exp. Psychol. Hum. Percept. Perform. 1991, 17, 1075. [Google Scholar] [CrossRef]
- Peterson, M.A. Past experience and meaning affect object detection: A hierarchical Bayesian approach. In Psychology of Learning and Motivation; Academic Press: Cambridge, MA, USA, 2019; Volume 70, pp. 223–257. [Google Scholar] [CrossRef]
- Costello, P.; Jiang, Y.; Baartman, B.; McGlennen, K.; He, S. Semantic and subword priming during binocular suppression. Conscious. Cogn. 2009, 18, 375–382. [Google Scholar] [CrossRef]
- Lupyan, G. Linguistically modulated perception and cognition: The label-feedback hypothesis. Front. Psychol. 2012, 3, 54. [Google Scholar] [CrossRef]
- Lupyan, G.; Ward, E.J. Language can boost otherwise unseen objects into visual awareness. Proc. Natl. Acad. Sci. USA 2013, 110, 14196–14201. [Google Scholar] [CrossRef]
- Pinto, Y.; van Gaal, S.; de Lange, F.P.; Lamme, V.A.; Seth, A.K. Expectations accelerate entry of visual stimuli into awareness. J. Vis. 2015, 15, 13. [Google Scholar] [CrossRef]
- Stein, T.; Peelen, M.V. Content-specific expectations enhance stimulus detectability by increasing perceptual sensitivity. J. Exp. Psychol. Gen. 2015, 144, 1089. [Google Scholar] [CrossRef] [PubMed]
- Lupyan, G.; Clark, A. Words and the world: Predictive coding and the language-perception-cognition interface. Curr. Dir. Psychol. Sci. 2015, 24, 279–284. [Google Scholar] [CrossRef]
- Clarke, A. Neural dynamics of visual and semantic object processing. In Psychology of Learning and Motivation; Academic Press: Cambridge, MA, USA, 2019; Volume 70, pp. 71–95. [Google Scholar] [CrossRef]
- Clarke, A.; Tyler, L.K. Object-specific semantic coding in human perirhinal cortex. J. Neurosci. 2014, 34, 4766–4775. [Google Scholar] [CrossRef] [PubMed]
- Clarke, A.; Tyler, L.K. Understanding what we see: How we derive meaning from vision. Trends Cogn. Sci. 2015, 19, 677–687. [Google Scholar] [CrossRef] [PubMed]
- Liuzzi, A.G.; Bruffaerts, R.; Vandenberghe, R. The medial temporal written word processing system. Cortex 2019, 119, 287–300. [Google Scholar] [CrossRef]
- Martin, C.B.; Douglas, D.; Newsome, R.N.; Man, L.L.; Barense, M.D. Integrative and distinctive coding of visual and conceptual object features in the ventral visual stream. Elife 2018, 7, e31873. [Google Scholar] [CrossRef]
- Gayet, S.; Van der Stigchel, S.; Paffen, C.L. Breaking continuous flash suppression: Competing for consciousness on the pre-semantic battlefield. Front. Psychol. 2014, 5, 460. [Google Scholar] [CrossRef]
- Jolicoeur, P. The time to name disoriented natural objects. Mem. Cogn. 1985, 13, 289–303. [Google Scholar] [CrossRef]
- Hochberg, J. Art and perception. Handb. Percept. 1978, 10, 225–258. [Google Scholar]
- Jiang, Y.; Costello, P.; He, S. Processing of invisible stimuli: Advantage of upright faces and recognizable words in overcoming interocular suppression. Psychol. Sci. 2007, 18, 349–355. [Google Scholar] [CrossRef]
- Stein, T.; Hebart, M.N.; Sterzer, P. Breaking continuous flash suppression: A new measure of unconscious processing during interocular suppression? Front. Hum. Neurosci. 2011, 5, 167. [Google Scholar] [CrossRef] [PubMed]
- Sterzer, P.; Stein, T.; Ludwig, K.; Rothkirch, M.; Hesselmann, G. Neural processing of visual information under interocular suppression: A critical review. Front. Psychol. 2014, 5, 453. [Google Scholar] [CrossRef]
- Yang, E.; Blake, R. Deconstructing continuous flash suppression. J. Vis. 2012, 12, 8. [Google Scholar] [CrossRef] [PubMed]
- Hochberg, J. Perception. In Experimental Psychology; Kling, J.W., Riggs, L.A., Eds.; Winston: New York, NY, USA, 1971. [Google Scholar]
- Skocypec, R.; Peterson, M. Do Semantic Expectations Arising from Masked Word Primes Aid Object Detection at the Earliest Level? Now You See It, Now You Don’t. J. Vis. 2018, 18, 1321. [Google Scholar] [CrossRef]
- Skocypec, R.M.; Peterson, M.A. The Influence of Semantics on Figure Assignment: Unmasked Primes, Masked Primes, and Context. J. Vis. 2019, 19, 35b. [Google Scholar] [CrossRef]
- Peterson, M.A.; Gibson, B.S. The initial identification of figure-ground relationships: Contributions from shape recognition processes. Bull. Psychon. Soc. 1991, 29, 199–202. [Google Scholar] [CrossRef]
- Peterson, M.A.; Gibson, B.S. Must figure-ground organization precede object recognition? An assumption in peril. Psychol. Sci. 1994, 5, 253–259. [Google Scholar] [CrossRef]
- Gibson, B.S.; Peterson, M.A. Does orientation-independent object recognition precede orientation-dependent recognition? Evidence from a cuing paradigm. J. Exp. Psychol. Hum. Percept. Perform. 1994, 20, 299. [Google Scholar] [CrossRef]
- Barense, M.D.; Ngo, J.K.; Hung, L.H.; Peterson, M.A. Interactions of memory and perception in amnesia: The figure–ground perspective. Cereb. Cortex 2012, 22, 2680–2691. [Google Scholar] [CrossRef][Green Version]
- Gerhardstein, P.; Kovacs, I.; Ditre, J.; Feher, A. Detection of contour continuity and closure in three-month-olds. Vis. Res. 2004, 44, 2981–2988. [Google Scholar] [CrossRef]
- Peterson, M.A.; De Gelder, B.; Rapcsak, S.Z.; Gerhardstein, P.C.; Bachoud-Lévi, A.C. Object memory effects on figure assignment: Conscious object recognition is not necessary or sufficient. Vis. Res. 2000, 40, 1549–1567. [Google Scholar] [CrossRef]
- Sasia, B.; Cacciamani, L. High-definition transcranial direct current stimulation of the lateral occipital cortex influences figure-ground perception. Neuropsychologia 2021, 155, 107792. [Google Scholar] [CrossRef] [PubMed]
- Ashbridge, E.; Perrett, D.I.; Oram, M.W.; Jellema, T. Effect of image orientation and size on object recognition: Responses of single units in the macaque monkey temporal cortex. Cogn. Neuropsychol. 2000, 17, 13–34. [Google Scholar] [CrossRef] [PubMed]
- Oram, M.W. Integrating neuronal coding into cognitive models: Predicting reaction time distributions. Netw. Comput. Neural Syst. 2005, 16, 377–400. [Google Scholar] [CrossRef][Green Version]
- Perrett, D.I.; Oram, M.W.; Ashbridge, E. Evidence accumulation in cell populations responsive to faces: An account of generalisation of recognition without mental transformations. Cognition 1998, 67, 111–145. [Google Scholar] [CrossRef]
- Schendan, H.E. Memory influences visual cognition across multiple functional states of interactive cortical dynamics. In Psychology of Learning and Motivation; Academic Press: Cambridge, MA, USA, 2019; Volume 71, pp. 303–386. [Google Scholar] [CrossRef]
- Schendan, H.E.; Kutas, M. Time course of processes and representations supporting visual object identification and memory. J. Cogn. Neurosci. 2003, 15, 111–135. [Google Scholar] [CrossRef]
- Schendan, H.E.; Kutas, M. Neurophysiological evidence for the time course of activation of global shape, part, and local contour representations during visual object categorization and memory. J. Cogn. Neurosci. 2007, 19, 734–749. [Google Scholar] [CrossRef]
- Tyler, L.K.; Chiu, S.; Zhuang, J.; Randall, B.; Devereux, B.J.; Wright, P.; Clarke, A.; Taylor, K.I. Objects and Categories: Feature Statistics and Object Processing in the Ventral Stream. J. Cogn. Neurosci. 2013, 25, 1723–1735. [Google Scholar] [CrossRef]
- Cacciamani, L.; Mojica, A.J.; Sanguinetti, J.L.; Peterson, M.A. Semantic access occurs outside of awareness for the ground side of a figure. Atten. Percept. Psychophys. 2014, 76, 2531–2547. [Google Scholar] [CrossRef]
- Cohen, M.A.; Konkle, T.; Rhee, J.Y.; Nakayama, K.; Alvarez, G.A. Processing multiple visual objects is limited by overlap in neural channels. Proc. Natl. Acad. Sci. USA 2014, 111, 8955–8960. [Google Scholar] [CrossRef]
- Zachariou, V.; Del Giacco, A.C.; Ungerleider, L.G.; Yue, X. Bottom-up processing of curvilinear visual features is sufficient for animate/inanimate object categorization. J. Vis. 2018, 18, 3. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Ralph, M.A.L.; Rogers, T.T. A unified model of human semantic knowledge and its disorders. Nat. Hum. Behav. 2017, 1, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Desimone, R.; Duncan, J. Neural mechanisms of selective visual attention. Annu. Rev. Neurosci. 1995, 18, 193–222. [Google Scholar] [CrossRef] [PubMed]
- Duncan, J.; Humphreys, G.W.; Ward, R. Competitive brain activity in visual attention. Curr. Opin. Neurobiol. 1997, 7, 255–261. [Google Scholar] [CrossRef]
- Forster, K.I.; Forster, J.C. DMDX: A Windows display program with millisecond accuracy. Behav. Res. Methods Instrum. Comput. 2003, 35, 116–124. [Google Scholar] [CrossRef]
- Flowers, C.S.; Orsten-Hooge, K.D.; Jannuzi, B.G.; Peterson, M.A. Normative data for an expanded set of stimuli for testing high-level influences on object perception: OMEFA-II. PLoS ONE 2020, 15, e0224471. [Google Scholar] [CrossRef]
- Stokes, M.; Thompson, R.; Nobre, A.C.; Duncan, J. Shape-specific preparatory activity mediates attention to targets in human visual cortex. Proc. Natl. Acad. Sci. USA 2009, 106, 19569–19574. [Google Scholar] [CrossRef]
- Bracci, S.; Ritchie, J.B.; Kalfas, I.; de Beeck, H.P.O. The ventral visual pathway represents animal appearance over animacy, unlike human behavior and deep neural networks. J. Neurosci. 2019, 39, 6513–6525. [Google Scholar] [CrossRef]
- Carlson, T.A.; Schrater, P.; He, S. Patterns of activity in the categorical representations of objects. J. Cogn. Neurosci. 2003, 15, 704–717. [Google Scholar] [CrossRef]
- Grootswagers, T.; Robinson, A.K.; Carlson, T.A. The representational dynamics of visual objects in rapid serial visual processing streams. NeuroImage 2019, 188, 668–679. [Google Scholar] [CrossRef]
- Huth, A.G.; Nishimoto, S.; Vu, A.T.; Gallant, J.L. A continuous semantic space describes the representation of thousands of object and action categories across the human brain. Neuron 2012, 76, 1210–1224. [Google Scholar] [CrossRef] [PubMed]
- Kriegeskorte, N.; Mur, M.; Ruff, D.; Kiani, R.; Bodurka, J.; Esteky, H.; Tanaka, K.; Bandettini, P.A. Matching categorical object representations in inferior temporal cortex of man and monkey. Neuron 2008, 60, 1126–1141. [Google Scholar] [CrossRef] [PubMed]
- Plaut, D.C. Graded modality-specific specialisation in semantics: A computational account of optic aphasia. Cogn. Neuropsychol. 2002, 19, 603–639. [Google Scholar] [CrossRef] [PubMed]
- Rogers, T.T.; Patterson, K. Object categorization: Reversals and explanations of the basic-level advantage. J. Exp. Psychol. Gen. 2007, 136, 451. [Google Scholar] [CrossRef]
- Rosch, E.; Mervis, C.B. Family resemblances: Studies in the internal structure of categories. Cogn. Psychol. 1975, 7, 573–605. [Google Scholar] [CrossRef]
- Rosch, E.; Mervis, C.B.; Gray, W.D.; Johnson, D.M.; Boyes-Braem, P. Basic objects in natural categories. Cogn. Psychol. 1976, 8, 382–439. [Google Scholar] [CrossRef]
- Avital-Cohen, R.; Gronau, N. The asymmetric mixed-category advantage in visual working memory: An attentional, not perceptual (face-specific) account. J. Exp. Psychol. Hum. Percept. Perform. 2021, 47, 852. Available online: https://psycnet.apa.org/doi/10.1037/xhp0000921 (accessed on 1 March 2022). [CrossRef]
- Brooks, J.L.; Palmer, S.E. Cue competition affects temporal dynamics of edge-assignment in human visual cortex. J. Cogn. Neurosci. 2011, 23, 631–644. [Google Scholar] [CrossRef]
- Peterson, M.A.; Enns, J.T. The edge complex: Implicit perceptual memory for cross-edge competition leading to figure assignment. Percept. Psychophys. 2005, 14, 727–740. [Google Scholar] [CrossRef]
- Peterson, M.A.; Lampignano, D.W. Implicit memory for novel figure-ground displays includes a history of cross-border competition. J. Exp. Psychol. Hum. Percept. Perform. 2003, 29, 808. [Google Scholar] [CrossRef][Green Version]
- Kok, P.; Jehee, J.F.; De Lange, F.P. Less is more: Expectation sharpens representations in the primary visual cortex. Neuron 2012, 75, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Hindy, N.C.; Ng, F.Y.; Turk-Browne, N.B. Linking pattern completion in the hippocampus to predictive coding in visual cortex. Nat. Neurosci. 2016, 19, 665–667. [Google Scholar] [CrossRef] [PubMed]
- Boutonnet, B.; Lupyan, G. Words jump-start vision: A label advantage in object recognition. J. Neurosci. 2015, 35, 9329–9335. [Google Scholar] [CrossRef] [PubMed]
- Trujillo, L.T.; Allen, J.J.; Schnyer, D.M.; Peterson, M.A. Neurophysiological evidence for the influence of past experience on figure–ground perception. J. Vis. 2010, 10, 5. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sanguinetti, J.L.; Allen, J.J.; Peterson, M.A. The ground side of an object: Perceived as shapeless yet processed for semantics. Psychol. Sci. 2014, 25, 256–264. [Google Scholar] [CrossRef] [PubMed]
- Sanguinetti, J.L.; Trujillo, L.T.; Schnyer, D.M.; Allen, J.J.; Peterson, M.A. Increased alpha band activity indexes inhibitory competition across a border during figure assignment. Vis. Res. 2016, 126, 120–130. [Google Scholar] [CrossRef]
- De Lange, F.P.; Heilbron, M.; Kok, P. How do expectations shape perception? Trends Cogn. Sci. 2018, 22, 764–779. [Google Scholar] [CrossRef]
- Regier, T.; Xu, Y. The Sapir-Whorf hypothesis and inference under uncertainty. Wiley Interdiscip. Rev. Cogn. Sci. 2017, 8, e1440. [Google Scholar] [CrossRef]
- Lupyan, G.; Rahman, R.A.; Boroditsky, L.; Clark, A. Effects of Language on Visual Perception. Trends Cogn. Sci. 2020, 24, 930–944. [Google Scholar] [CrossRef]
- Kong, D.; Soon, C.S.; Chee, M.W. Reduced visual processing capacity in sleep deprived persons. Neuroimage 2011, 55, 629–634. [Google Scholar] [CrossRef]
- Louca, M.; Short, M.A. The effect of one night’s sleep deprivation on adolescent neurobehavioral performance. Sleep 2014, 37, 1799–1807. [Google Scholar] [CrossRef] [PubMed]
- Brysbaert, M.; New, B. Moving beyond Kučera and Francis: A critical evaluation of current word frequency norms and the introduction of a new and improved word frequency measure for American English. Behav. Res. Methods 2009, 41, 977–990. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Skocypec, R.M.; Peterson, M.A. Semantic Expectation Effects on Object Detection: Using Figure Assignment to Elucidate Mechanisms. Vision 2022, 6, 19. https://doi.org/10.3390/vision6010019
Skocypec RM, Peterson MA. Semantic Expectation Effects on Object Detection: Using Figure Assignment to Elucidate Mechanisms. Vision. 2022; 6(1):19. https://doi.org/10.3390/vision6010019
Chicago/Turabian StyleSkocypec, Rachel M., and Mary A. Peterson. 2022. "Semantic Expectation Effects on Object Detection: Using Figure Assignment to Elucidate Mechanisms" Vision 6, no. 1: 19. https://doi.org/10.3390/vision6010019
APA StyleSkocypec, R. M., & Peterson, M. A. (2022). Semantic Expectation Effects on Object Detection: Using Figure Assignment to Elucidate Mechanisms. Vision, 6(1), 19. https://doi.org/10.3390/vision6010019




