Treat and Extend Treatment Interval Patterns with Anti-VEGF Therapy in nAMD Patients
Abstract
1. Introduction
2. Materials and Methods
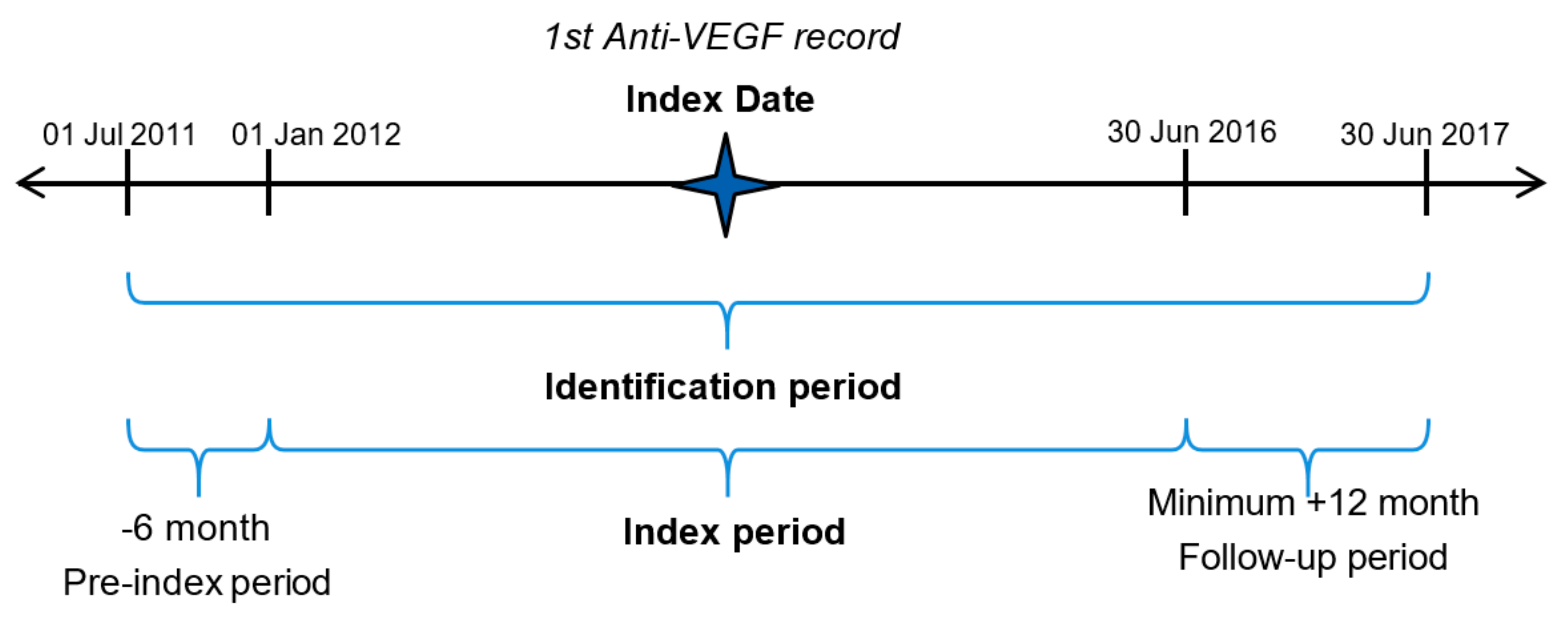
2.1. Study Design
2.2. Study Participants
2.3. Study Objectives
2.4. Outcome Measurements
2.5. Statistical Analysis
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wong, W.L.; Su, X.; Li, X.; Cheung, C.M.G.; Klein, R.; Cheng, C.-Y.; Wong, T.Y. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: A systematic review and meta-analysis. Lancet Glob. Health 2014, 2, e106–e116. [Google Scholar] [CrossRef]
- Kovach, J.L.; Schwartz, S.G.; Flynn, H.W., Jr.; Scott, I.U. Anti-VEGF Treatment Strategies for Wet AMD. J. Ophthalmol. 2012, 2012, 786–870. [Google Scholar] [CrossRef] [PubMed]
- Chakravarthy, U.; Harding, S.P.; A Rogers, C.; Downes, S.M.; Lotery, A.J.; A Culliford, L.; Reeves, B.C. Alternative treatments to inhibit VEGF in age-related choroidal neovascularisation: 2-year findings of the IVAN randomised controlled trial. Lancet 2013, 382, 1258–1267. [Google Scholar] [CrossRef]
- Martin, D.F.; Maguire, M.G.; Fine, S.L.; Ying, G.-S.; Jaffe, G.J.; Grunwald, J.E.; Toth, C.; Redford, M.; Ferris, F.L. Ranibizumab and bevacizumab for treatment of neovascular age-related macular degeneration: Two-year results. Ophthalmology 2012, 119, 1388–1398. [Google Scholar] [CrossRef] [PubMed]
- Rosenfeld, P.J.; Brown, D.M.; Heier, J.S.; Boyer, D.S.; Kaiser, P.K.; Chung, C.Y.; Kim, R.Y. Ranibizumab for neovascular age-related macular degeneration. N. Engl. J. Med. 2006, 355, 1419–1431. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.M.; Kaiser, P.K.; Michels, M.; Soubrane, G.; Heier, J.S.; Kim, R.Y.; Sy, J.P.; Schneider, S. Ranibizumab versus verteporfin for neovascular age-related macular degeneration. N. Engl. J. Med. 2006, 355, 1432–1444. [Google Scholar] [CrossRef]
- Brown, D.M.; Michels, M.; Kaiser, P.K.; Heier, J.S.; Sy, J.P.; Ianchulev, T. Ranibizumab versus verteporfin photodynamic therapy for neovascular age-related macular degeneration: Two-year results of the ANCHOR study. Ophthalmology 2009, 116, 57–65.e5. [Google Scholar] [CrossRef]
- Chen, Y.; Han, F. Profile of ranibizumab: Efficacy and safety for the treatment of wet age-related macular degeneration. Ther. Clin. Risk. Manag. 2012, 8, 343–351. [Google Scholar] [CrossRef]
- Heier, J.S.; Brown, D.M.; Chong, V.; Korobelnik, J.-F.; Kaiser, P.K.; Nguyen, Q.D.; Kirchhof, B.; Ho, A.; Ogura, Y.; Yancopoulos, G.D.; et al. Intravitreal aflibercept (VEGF trap-eye) in wet age-related macular degeneration. Ophthalmology 2012, 119, 2537–2548. [Google Scholar] [CrossRef]
- Arnold, J.J.; Campain, A.; Barthelmes, D.; Simpson, J.M.; Guymer, R.H.; Hunyor, A.P.; McAllister, I.L.; Essex, R.W.; Morlet, N.; Gillies, M.C.; et al. Two-year outcomes of “treat and extend” intravitreal therapy for neovascular age-related macular degeneration. Ophthalmology 2015, 122, 1212–1219. [Google Scholar] [CrossRef]
- Gillies, M.C.; Campain, A.; Barthelmes, D.; Simpson, J.M.; Arnold, J.J.; Guymer, R.H.; McAllister, I.L.; Essex, R.W.; Morlet, N.; Hunyor, A.P.; et al. Long-Term Outcomes of Treatment of Neovascular Age-Related Macular Degeneration: Data from an Observational Study. Ophthalmology 2015, 122, 1837–1845. [Google Scholar] [CrossRef] [PubMed]
- Johnston, R.L.; Carius, H.-J.; Skelly, A.; Ferreira, A.; Milnes, F.; Mitchell, P. A Retrospective Study of Ranibizumab Treatment Regimens for Neovascular Age-Related Macular Degeneration (nAMD) in Australia and the United Kingdom. Adv. Ther. 2017, 34, 703–712. [Google Scholar] [CrossRef] [PubMed]
- Writing Committee for the UK Age-Related Macular Degeneration EMR Users Group. The neovascular age-related macular degeneration database: Multicenter study of 92 976 ranibizumab injections: Report 1: Visual acuity. Ophthalmology 2014, 121, 1092–1101. [Google Scholar] [CrossRef] [PubMed]
- Berg, K.; Pedersen, T.R.; Sandvik, L.; Bragadottir, R. Comparison of ranibizumab and bevacizumab for neovascular age-related macular degeneration according to LUCAS treat-and-extend protocol. Ophthalmology 2015, 122, 146–152. [Google Scholar] [CrossRef] [PubMed]
- Berg, K.; Hadzalic, E.; Gjertsen, I.; Forsaa, V.; Berger, L.H.; Kinge, B.; Henschien, H.; Fossen, K.; Markovic, S.; Pedersen, T.R.; et al. Ranibizumab or Bevacizumab for Neovascular Age-Related Macular Degeneration According to the Lucentis Compared to Avastin Study Treat-and-Extend Protocol: Two-Year Results. Ophthalmology 2016, 123, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Wykoff, C.C.; Croft, D.E.; Brown, D.M.; Wang, R.; Payne, J.F.; Clark, L.; Abdelfattah, N.S.; Sadda, S.R.; TREX-AMD Study Group. Prospective Trial of Treat-and-Extend versus Monthly Dosing for Neovascular Age-Related Macular Degeneration: TREX-AMD 1-Year Results. Ophthalmology 2015, 122, 2514–2522. [Google Scholar] [CrossRef] [PubMed]
- Gillies, M.C.; Hunyor, A.P.; Arnold, J.J.; Guymer, R.H.; Wolf, S.; Ng, P.; Pecheur, F.L.; McAllister, I.L. Effect of Ranibizumab and Aflibercept on Best-Corrected Visual Acuity in Treat-and-Extend for Neovascular Age-Related Macular Degeneration: A Randomized Clinical Trial. JAMA Ophthalmol. 2019, 137, 372–379. [Google Scholar] [CrossRef] [PubMed]
- Abedi, F.; Wickremasinghe, S.; Islam, A.F.; Inglis, K.M.; Guymer, R.H. Anti-VEGF treatment in neovascular age-related macular degeneration: A treat-and-extend protocol over 2 years. Retina 2014, 34, 1531–1538. [Google Scholar] [CrossRef] [PubMed]
- Engelbert, M.; Zweifel, S.A.; Freund, K.B. “Treat and extend” dosing of intravitreal antivascular endothelial growth factor therapy for type 3 neovascularization/retinal angiomatous proliferation. Retina 2009, 29, 1424–1431. [Google Scholar] [CrossRef]
- Engelbert, M.; Zweifel, S.A.; Freund, K.B. Long-term follow-up for type 1 (subretinal pigment epithelium) neovascularization using a modified “treat and extend” dosing regimen of intravitreal antivascular endothelial growth factor therapy. Retina 2010, 30, 1368–1375. [Google Scholar] [CrossRef]
- Gupta, O.P.; Shienbaum, G.; Patel, A.H.; Fecarotta, C.; Kaiser, R.S.; Regillo, C.D. A treat and extend regimen using ranibizumab for neovascular age-related macular degeneration clinical and economic impact. Ophthalmology 2010, 117, 2134–2140. [Google Scholar] [CrossRef] [PubMed]
- Oubraham, H.; Cohen, S.Y.; Samimi, S.; Marotte, D.; Bouzaher, I.; Bonicel, P.; Fajnkuchen, F.; Tadayoni, R. Inject and extend dosing versus dosing as needed: A comparative retrospective study of ranibizumab in exudative age-related macular degeneration. Retina 2011, 31, 26–30. [Google Scholar] [CrossRef] [PubMed]
- Essex, R.W.; Nguyen, V.; Walton, R.; Arnold, J.J.; McAllister, I.L.; Guymer, R.H.; Morlet, N.; Young, S.; Barthelmes, D.; Gillies, M.C.; et al. Treatment Patterns and Visual Outcomes during the Maintenance Phase of Treat-and-Extend Therapy for Age-Related Macular Degeneration. Ophthalmology 2016, 123, 2393–2400. [Google Scholar] [CrossRef] [PubMed]
- Novartis Pharmaceuticals Australia Pty Limited. LUCENTIS ranibizumab. Produce Information for Inclusion in the Australian Register of Therapeutic Goods (the ARTG). Updated 22 August 2017. Available online: http://www.guildlink.com.au/gc/ws/nv/pi.cfm?product=nvplucin10316 (accessed on 22 August 2019).
- Public Policy Committee, International Society of Pharmacoepidemiology. Guidelines for good pharmacoepidemiology practice (GPP). Pharmacoepidemiol. Drug Saf. 2016, 25, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Vandenbroucke, J.P.; Von Elm, E.; Altman, D.G.; Gøtzsche, P.C.; Mulrow, C.D.; Pocock, S.J.; Poole, C.; Schlesselman, J.J.; Egger, M. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE): Explanation and elaboration. Int. J. Surg. 2014, 12, 1500–1524. [Google Scholar] [CrossRef] [PubMed]
- World Medical Association. World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. JAMA 2013, 310, 2191–2194. [Google Scholar] [CrossRef] [PubMed]
- Gregori, N.Z.; Feuer, W.; Rosenfeld, P.J. Novel method for analyzing snellen visual acuity measurements. Retina 2010, 30, 1046–1050. [Google Scholar] [CrossRef] [PubMed]
- Spaide, R. Ranibizumab according to need: A treatment for age-related macular degeneration. Am. J. Ophthalmol. 2007, 143, 679–680. [Google Scholar] [CrossRef]
- Skelly, A.; Carius, H.J.; Bezlyak, V.; Chen, F.K. Dispensing Patterns of Ranibizumab and Aflibercept for the Treatment of Neovascular Age-Related Macular Degeneration: A Retrospective Cohort Study in Australia. Adv. Ther. 2017, 34, 2585–2600. [Google Scholar] [CrossRef]
- Gillies, M.C.; Walton, R.; Simpson, J.M.; Arnold, J.J.; Guymer, R.H.; McAllister, I.L.; Hunyor, A.P.; Essex, R.W.; Morlet, N.; Barthelmes, D.; et al. Time to initial clinician-reported inactivation of neovascular age-related macular degeneration treated primarily with ranibizumab. Ophthalmology 2015, 122, 589–594.e1. [Google Scholar] [CrossRef] [PubMed]
- Ackermann, B.; Neubert, H.; Hughes, N.; Garofolo, F.; Abberley, L.; Alley, S.C.; Brown-Augsburger, P.; Bustard, M.; Chen, L.-Z.; Heinrich, J.; et al. 2015 White Paper on recent issues in bioanalysis: Focus on new technologies and biomarkers (Part 2—Hybrid LBA/LCMS and input from regulatory agencies). Bioanalysis 2015, 7, 3019–3034. [Google Scholar] [CrossRef] [PubMed]
- Sherman, R.E.; Anderson, S.A.; Pan, G.J.D.; Gray, G.W.; Gross, T.; Hunter, N.L.; LaVange, L.; Marinac-Dabic, D.; Marks, P.W.; Robb, M.A.; et al. Real-World Evidence—What Is It and What Can It Tell Us? N. Engl. J. Med. 2016, 375, 2293–2297. [Google Scholar] [CrossRef] [PubMed]
| Injection Treatment Pattern | Days from Previous Injection Lower Limit | Days from Previous Injection Upper Limit |
|---|---|---|
| 4-week interval | 22 | 34 |
| 6-week interval | 35 | 48 |
| 8-week interval | 49 | 69 |
| 12-week interval | 70 | 97 |
| 16-week interval | 98 | 125 |
| 20+-week interval | 126 | ≥180 * |
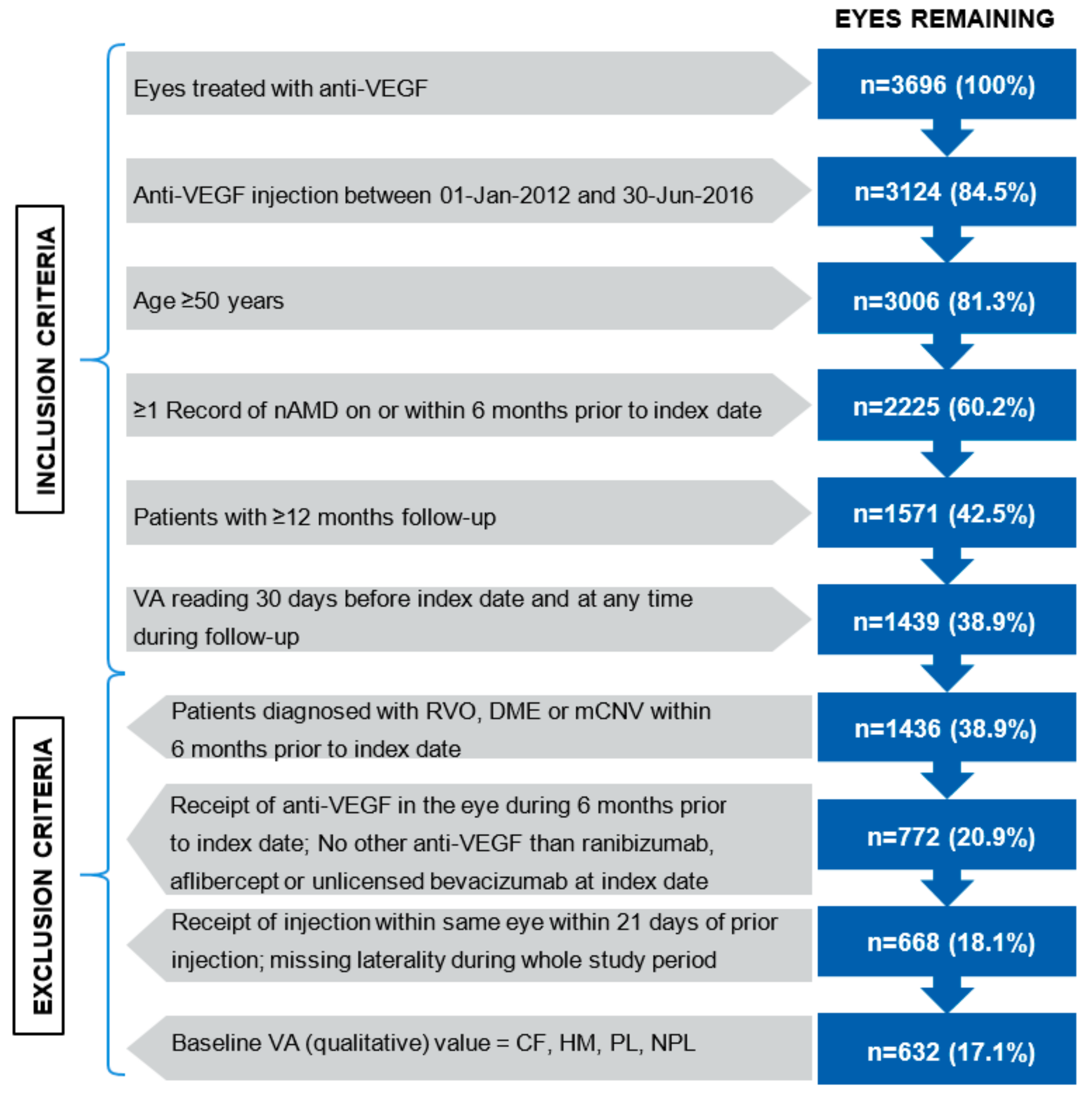
| Characteristics | Overall Cohort N = 555 Patients, n = 632 * Eyes |
|---|---|
| Age in database (patient) | |
| Mean (SD) | 78 (7.8) |
| Gender (n, %) | |
| Female | 254 (62.4%) |
| Eyes treated (patient; n, %) | |
| Unilateral | 478 (86.1%) |
| Bilateral | 77 (13.9%) |
| Eyes treated (patient, with laterality specified; n, %) | |
| Left | 217 (39.1%) |
| Right | 261 (47.0%) |
| Both | 77 (13.9%) |
| VA study eye letters | |
| Mean (SD) | 55.9 (17.1) |
| Central retinal thickness (n, %) | 289 (45.7%) |
| Mean (µm, SD) | 331 (107) |
| Year of baseline visit | |
| 2012 | 214 (33.9%) |
| 2013 | 174 (27.5%) |
| 2014 | 141 (22.3%) |
| 2015 | 82 (13.0%) |
| 2016 | 21 (3.3%) |
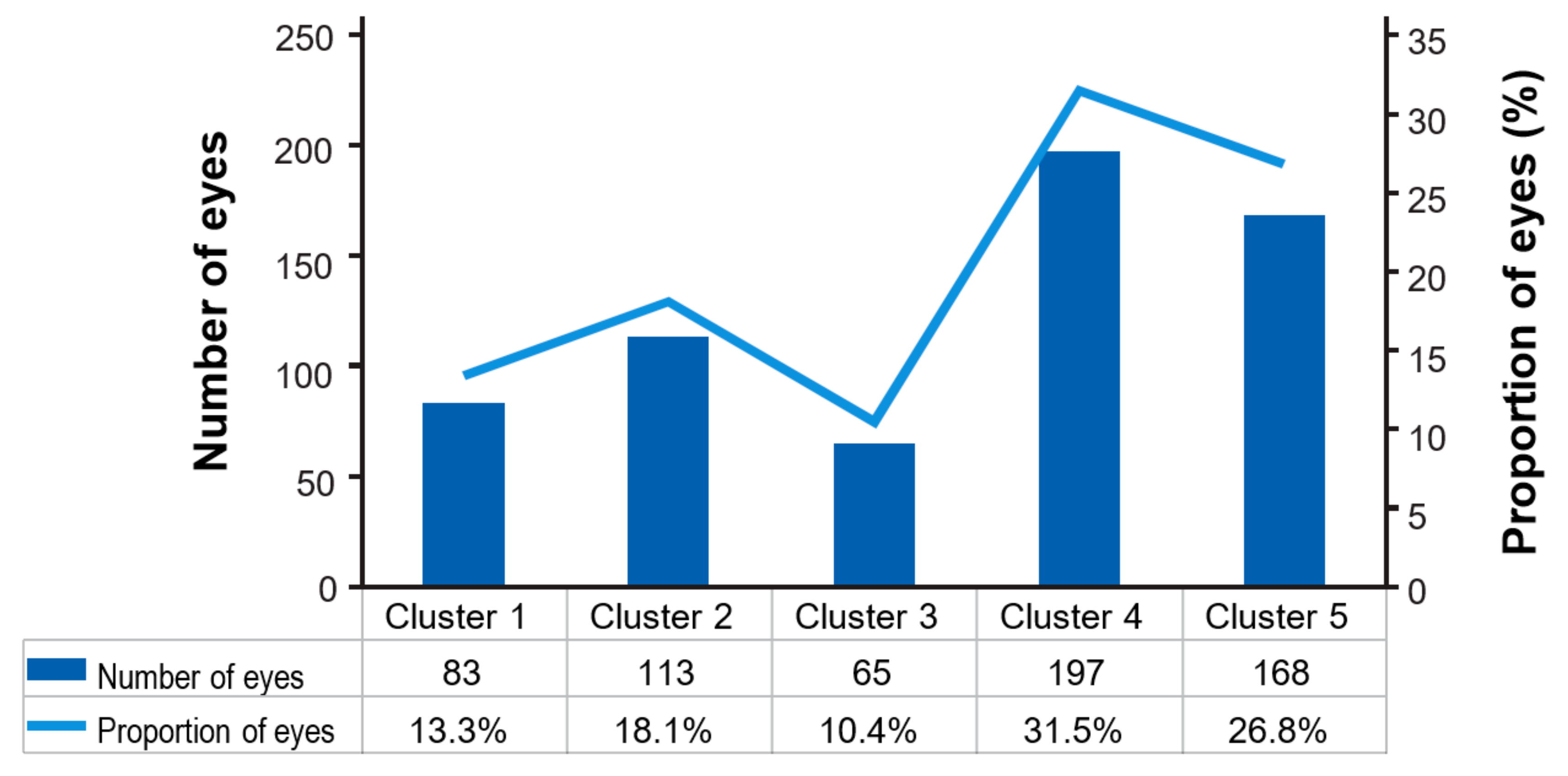
| Clusters (n = 632 * Eyes) | Description |
|---|---|
| Cluster 1 | Eyes with a record of interval of ≥12 weeks § that remain on this interval during follow-up visits |
| Cluster 2 | Eyes with a record of interval of ≥12 weeks § after which a shorter treatment interval occurred |
| Cluster 3 | Eyes with a record of consecutive (≥2) 8-week intervals without a record of a 12-week interval and without a shorter treatment occurring after these intervals |
| Cluster 4 | Eyes with a record of an 8-week interval, after which at least one shorter treatment interval occurs. Subsequent intervals may be of shorter or longer duration |
| Cluster 5 | Eyes with all intervals below 8 weeks or with a record of a single 8-week interval at the end of the study period |
| Characteristics | Overall Cohort n = 555 Patients, n = 632 * Eyes | Cluster 1 n = 79 Patients, n = 83 Eyes | Cluster 2 n = 107 Patients, n = 113 Eyes | Cluster 3 n = 63 Patients, n = 65 Eyes | Cluster 4 n = 182 Patients, n = 197 Eyes | Cluster 5 n = 154 Patients, n = 168 Eyes | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Baseline age (years; SD) | 78.0 (7.8) | 78.3 (8.5) | 78.2 (7.7) | 79.4 (7.7) | 77.5 (8.1) | 78.0 (7.3) | ||||||
| Baseline VA (letters; n, %) | 632 (100.0%) | 83 (100%) | 113 (100%) | 65 (100%) | 197 (100%) | 168 (100%) | ||||||
| Mean (SD) | 55.9 (17.1) | 53.7 (19.6) | 57.3 (16.3) | 56.3 (14.7) | 56.1 (17.0) | 54.8 (17.1) | ||||||
| VA stabilised § (n, %) | 496 (78.5%) | 63 (75.9%) | 84 (74.3%) | 51 (78.5%) | 154 (78.2%) | 139 (82.7%) | ||||||
| Median (Q1; Q3) time to VA stabilisation (days) | 118.5 (56.5–276.5) | 92.0 (35.0–346.0) | 196.5 (80.5–341.0) | 138.0 (48.0–268.0) | 114.0 (56.0–296.0) | 94.0 (57.0–223.0) | ||||||
| Mean (SD) change in VA at stabilisation (letters) | 6.7 (15.7) | 5.3 (20.7) | 6.9 (18.2) | 8.1 (15.4) | 7.0 (13.5) | 6.4 (14.4) | ||||||
| Mean (SD) VA at stabilisation (letters) | 63.4 (16.3) | 60.1 (22.0) | 65.3 (14.5) | 65.2 (14.3) | 64.3 (13.9) | 61.7 (17.1) | ||||||
| Loss of letters at stabilisation (n, %) | ||||||||||||
| ≥15 letters | 23 (3.6) | 6 (7.2) | 5 (4.4) | 1 (1.5) | 6 (3.0) | 5 (3.0) | ||||||
| 10–14 letters | 26 (4.1) | 2 (2.4) | 5 (4.4) | 3 (4.6) | 5 (2.5) | 11 (6.5) | ||||||
| 5–9 letters | 29 (4.6) | 3 (3.6) | 7 (6.2) | 1 (1.5) | 10 (5.1) | 8 (4.8) | ||||||
| 1–4 letters | 38 (6.0) | 4 (4.8) | 6 (5.3) | 2 (3.1) | 11 (5.6) | 15 (8.9) | ||||||
| No change in VA at stabilisation (n, %) | 76 (12.0%) | 10 (12.0%) | 11 (9.7%) | 8 (12.3%) | 27 (13.7%) | 17 (10.1%) | ||||||
| VA gain at stabilisation (n, %) | ||||||||||||
| 1–4 letters | 48 (7.6) | 7 (8.4) | 5 (4.4) | 1 (1.5) | 17 (8.6%) | 17 (10.1) | ||||||
| 5–9 letters | 71 (11.2) | 9 (10.8) | 9 (8.0) | 14 (21.5) | 21 (10.7) | 17 (10.1) | ||||||
| 10–14 letters | 52 (8.2) | 5 (6.0) | 9 (8.0) | 7 (10.8) | 21 (10.7) | 10 (6.0) | ||||||
| ≥15 letters | 133 (21.0) | 17 (20.5) | 27 (23.9) | 14 (21.5) | 36 (18.3) | 39 (23.2) | ||||||
| Change in VA from baseline to: | n, % | Mean (SD) | n, % | Mean (SD) | n, % | Mean (SD) | n, % | Mean (SD) | n, % | Mean (SD) | n, % | Mean (SD) |
| Month 3 | 544 (86.1) | 5.9 (14.8) | 71 (85.5) | 6.9 (17.4) | 82 (72.6) | 3.5 (18.5) | 63 (96.9) | 8.1 (12.0) | 181 (91.9) | 6.4 (14.2) | 142 (84.5 | 5.6 (12.8) |
| Month 6 | 546 (86.4%) | 6.3 (15.5) | 67 (80.7%) | 7.3 (17.4) | 88 (77.9%) | 4.3 (18.1) | 62 (95.4%) | 7.2 (13.8) | 182 (92.4%) | 7.1 (14.7) | 141 (83.9%) | 6.0 (14.9) |
| Month 12 | 505 (79.9%) | 5.8 (16.7) | 53 (63.9%) | 4.7 (17.6) | 94 (83.2%) | 6.3 (17.1) | 52 (80.0%) | 6.5 (15.5) | 171 (86.8%) | 6.7 (15.1) | 129 (76.8%) | 4.9 (19.0) |
| Month 24 | 358 (56.6%) | 3.2 (19.8) | 40 (48.2%) | 4.5 (23.2) | 66 (58.4%) | 2.1 (21.0) | 37 (56.9%) | 9.4 (15.2) | 116 (58.9%) | 3.9 (19.4) | 98 (58.3%) | 0.4 (19.2) |
| Characteristics | Overall cohort N = 555 Patients, n = 632 * Eyes | Cluster 1 N = 79 Patients, n = 83 Eyes | Cluster 2 N = 107 Patients, n = 113 Eyes | Cluster 3 N = 63 Patients, n = 65 Eyes | Cluster 4 N = 182 Patients, n = 197 Eyes | Cluster 5 N = 154 Patients, n = 168 Eyes |
|---|---|---|---|---|---|---|
| Eyes with follow-up in Year 1 (n, %) | 632 (100.0%) | 83 (100.0%) | 113 (100.0%) | 65 (100.0%) | 197 (100.0%) | 168 (100.0%) |
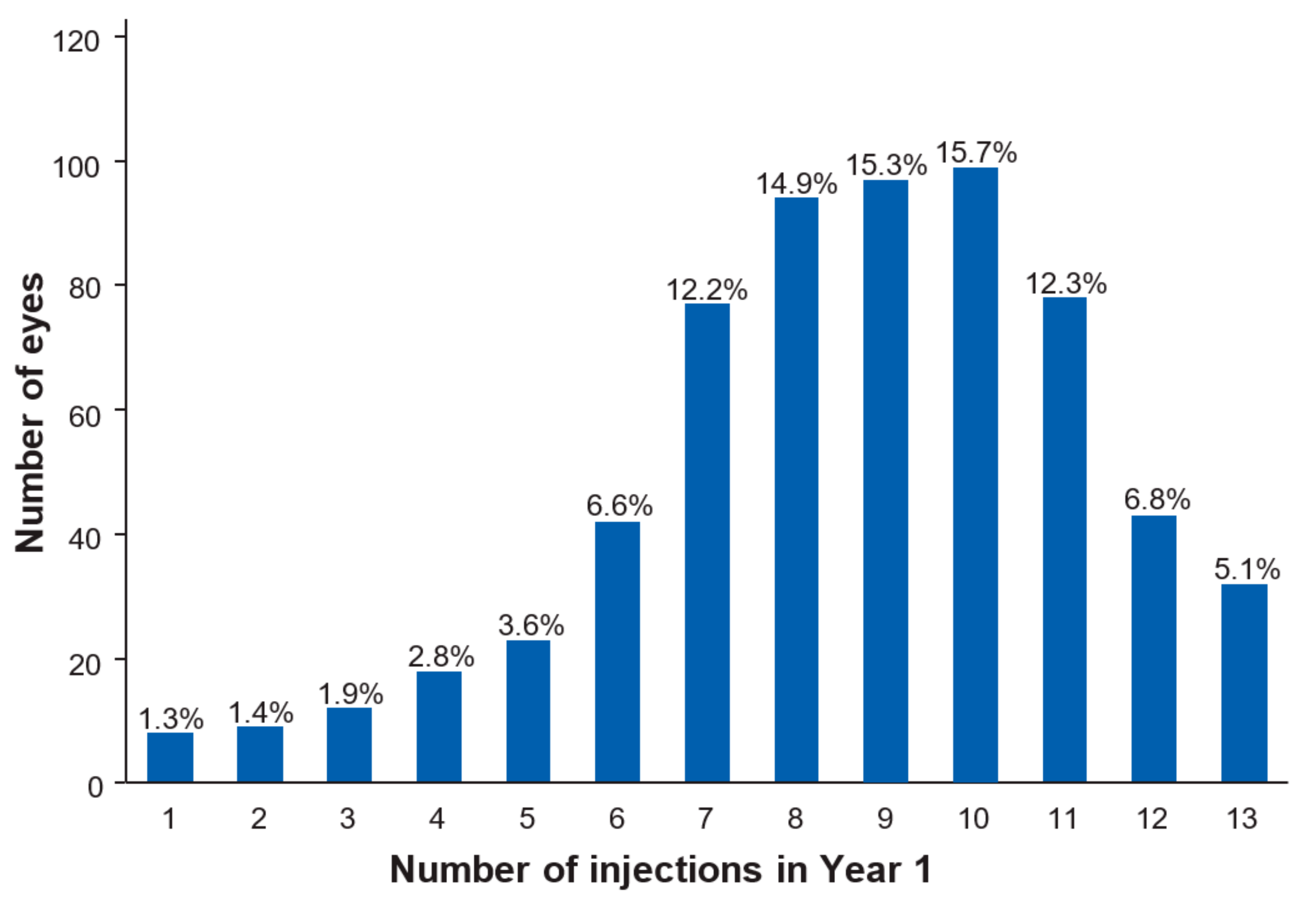
| Number of injections (mean, SD) | 8.6 (2.6) | 5.7 (2.1) | 7.3 (1.6) | 8.3 (1.1) | 9.2 (1.6) | 10.7 (2.4) |
| Number of visits (mean, SD) | 9.7 (2.6) | 6.9 (1.7) | 8.9 (2.6) | 9.0 (1.9) | 10.0 (2.3) | 11.5 (1.9) |
| Eyes with follow-up in Year 2 (n, %) | 632 (100.0%) | 83 (100.0%) | 113 (100.0%) | 65 (100.0%) | 197 (100.0%) | 168 (100.0%) |
| Number of injections (mean, SD) | 4.9 (2.9) | 2.8 (2.1) | 4.6 (2.3) | 4.7 (1.8) | 5.5 (2.9) | 5.8 (3.2) |
| Number of visits (mean, SD) | 5.8 (3.2) | 3.7 (2.3) | 5.9 (30.0) | 5.3 (2.4) | 6.4 (3.4) | 6.5 (3.1) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Skelly, A.; Bezlyak, V.; Liew, G.; Kap, E.; Sagkriotis, A. Treat and Extend Treatment Interval Patterns with Anti-VEGF Therapy in nAMD Patients. Vision 2019, 3, 41. https://doi.org/10.3390/vision3030041
Skelly A, Bezlyak V, Liew G, Kap E, Sagkriotis A. Treat and Extend Treatment Interval Patterns with Anti-VEGF Therapy in nAMD Patients. Vision. 2019; 3(3):41. https://doi.org/10.3390/vision3030041
Chicago/Turabian StyleSkelly, Adrian, Vladimir Bezlyak, Gerald Liew, Elisabeth Kap, and Alexandros Sagkriotis. 2019. "Treat and Extend Treatment Interval Patterns with Anti-VEGF Therapy in nAMD Patients" Vision 3, no. 3: 41. https://doi.org/10.3390/vision3030041
APA StyleSkelly, A., Bezlyak, V., Liew, G., Kap, E., & Sagkriotis, A. (2019). Treat and Extend Treatment Interval Patterns with Anti-VEGF Therapy in nAMD Patients. Vision, 3(3), 41. https://doi.org/10.3390/vision3030041




