CD40, a Novel Inducer of Purinergic Signaling: Implications to the Pathogenesis of Experimental Diabetic Retinopathy
Abstract
1. Introduction
2. CD40 is Required for Development of Experimental DR
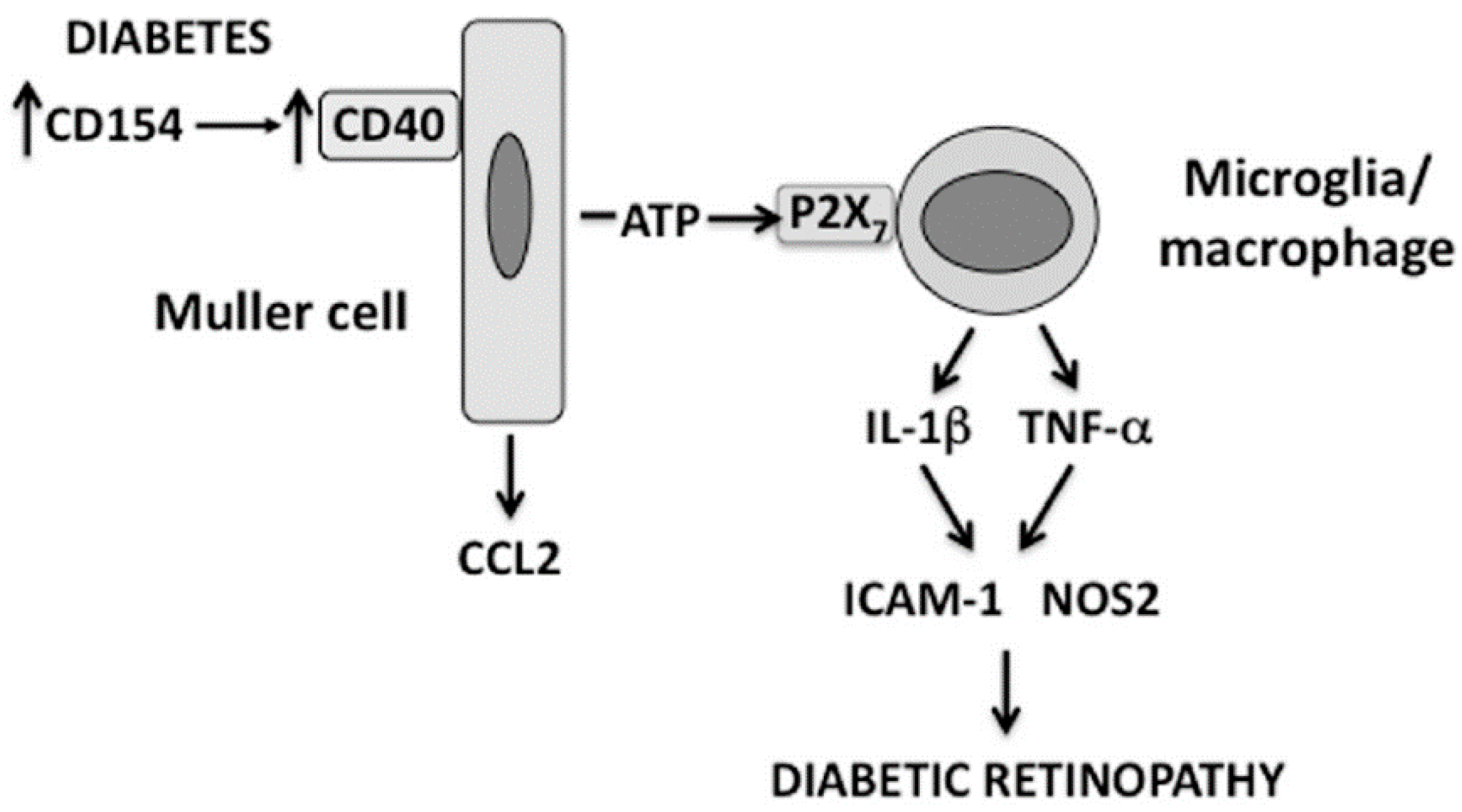
3. CD40 is a Novel Inducer of Purinergic Signaling: CD40 Induces ATP Release by Müller Cells Triggering P2X7-Driven TNF-α/IL-1β Production by Macrophages/Microglia
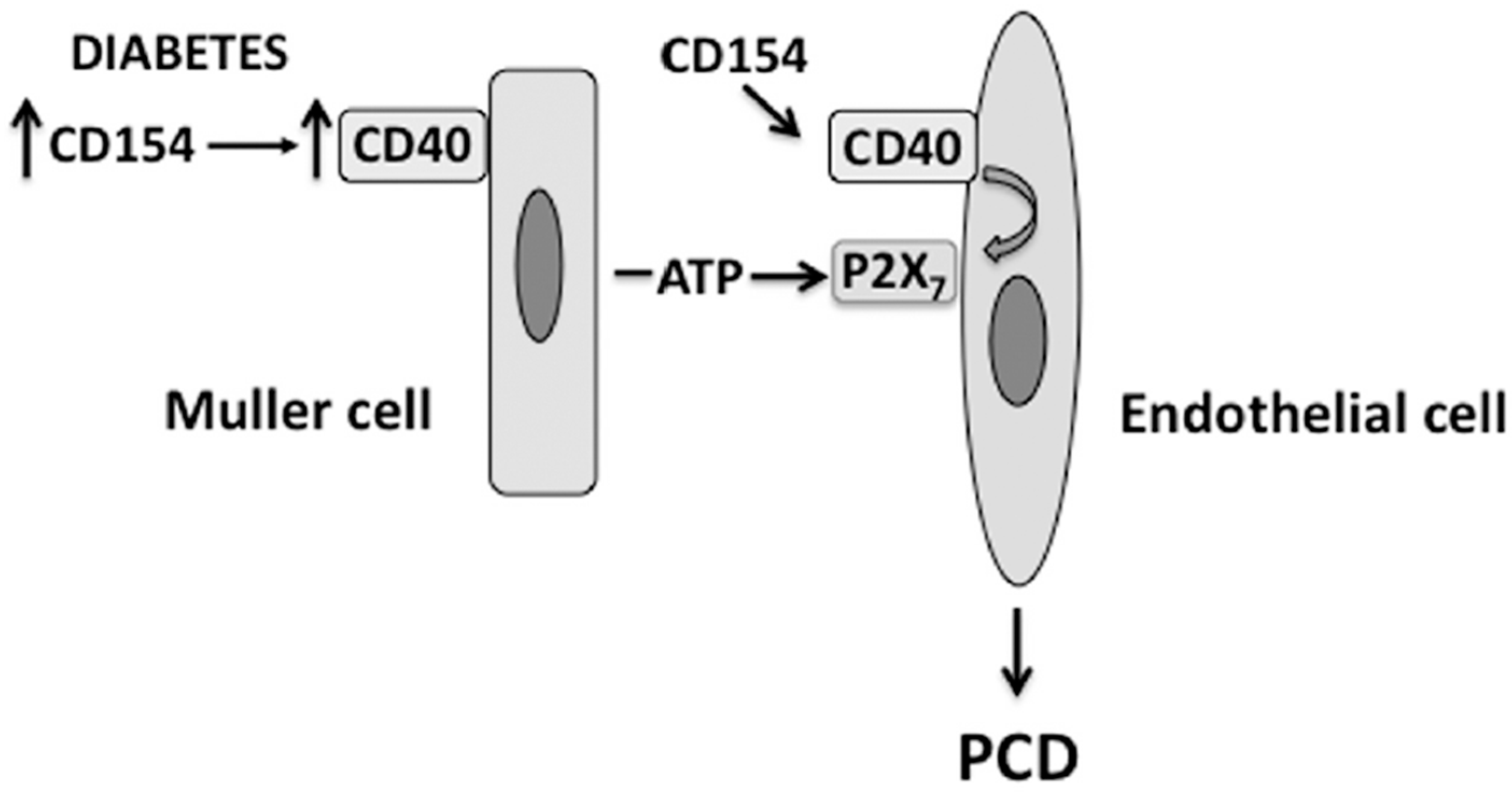
4. The CD40-ATP-P2X7 Pathway Mediates Programmed Cell Death (PCD) of Retinal Endothelial Cells
Acknowledgments
Conflicts of Interest
References
- World Health Organization Global Report on Diabetes; World Health Organization: Geneva, Switzerland, 2016.
- Yau, J.W.; Rogers, S.L.; Kawasaki, R.; Lamoureux, E.L.; Kowalski, J.W.; Bek, T.; Chen, S.J.; Dekker, J.M.; Fletcher, A.; Grauslund, J.; et al. Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care 2012, 35, 556–564. [Google Scholar] [CrossRef] [PubMed]
- Kempen, J.H.; O’Colmain, B.J.; Leske, M.C.; Haffner, S.M.; Klein, R.; Moss, S.E.; Taylor, H.R.; Hamman, R.F. The prevalence of diabetic retinopathy among adults in the United States. Arch. Ophthalmol. 2004, 122, 552–563. [Google Scholar] [PubMed]
- Mizutani, M.; Kern, T.S.; Lorenzi, M. Accelerated death of retinal microvascular cells in human and experimental diabetic retinopathy. J. Clin. Invest. 1996, 97, 2883–2890. [Google Scholar] [CrossRef] [PubMed]
- Kohner, E.M.; Henkind, P. Correlation of fluorescein angiogram and retinal digest in diabetic retinopathy. Am. J. Ophthalmol. 1970, 69, 403–414. [Google Scholar] [CrossRef]
- Brownlee, M. Biochemistry and molecular cell biology of diabetic complications. Nature 2001, 414, 813–820. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Kern, T.S. Inflammation in diabetic retinopathy. Prog. Retin. Eye Res. 2011, 30, 343–358. [Google Scholar] [CrossRef] [PubMed]
- McLeod, D.S.; Lefer, D.J.; Merges, C.; Lutty, G.A. Enhanced expression of intracellular adhesion molecule-1 and P-selectin in the diabetic human retina and choroid. Am. J. Pathol. 1995, 147, 642–653. [Google Scholar] [PubMed]
- Miyamoto, K.; Khorsrof, S.; Bursell, S.E.; Rohan, R.; Murata, T.; Clermont, A.C.; Aiello, L.P.; Ogura, Y.; Adamis, A.P. Prevention of leukostasis and vascular leakage in streptozotocin-induced diabetic retinopathy via intercellular adhesion molecule-1 inhibition. Proc. Natl. Acad. Sci. USA 1999, 96, 10836–10841. [Google Scholar] [CrossRef] [PubMed]
- Joussen, A.M.; Poulaki, V.; Le, M.L.; Koizumi, K.; Esser, C.; Janicki, H.; Schraermeyer, U.; Kociok, N.; Fauser, S.; Kirchhof, B.; et al. A central role for inflammation in the pathogenesis of diabetic retinopathy. FASEB J. 2004, 18, 1450–1452. [Google Scholar] [CrossRef] [PubMed]
- El-Asrar, A.M.A.; Desmet, S.; Meersschaert, A.; Dralands, L.; Missotten, L.; Geboes, K. Expression of the inducible isoform of nitric oxide synthase in the retinas of huma subjects with diabetes mellitus. Am. J. Ophthalmol. 2001, 132, 551–556. [Google Scholar] [CrossRef]
- Du, Y.; Smith, M.A.; Miller, C.M.; Kern, T.S. Diabetes-induced nitrative stress in the retina, and correction by aminoguanidine. J. Neurochem. 2002, 80, 771–779. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Du, Y.; Miller, C.; Gubitosi-Klug, R.A.; Kern, T.S.; Ball, S.; Berkowitz, B.A. Critical role of inducible nitric oxide synthase in degeneration of retinal capillaries in mice with streptozotocin-induced diabetes. Diabetologia 2007, 50, 1987–1996. [Google Scholar] [CrossRef] [PubMed]
- Leal, E.C.; Manivannan, A.; Hosoya, K.-I.; Terasaki, T.; Cunha-Vaz, J.; Ambrósio, A.F.; Forrester, J.V. Inducible nitric oxide synthase isoform is a key mediator of leukostasis and blood-retinal barrier breakdown in diabetic retinopathy. Investig. Ophthalmol. Vis. Sci. 2007, 48, 5257–5265. [Google Scholar] [CrossRef] [PubMed]
- El-Asrar, A.M.A.; Van Damme, J.O.; Put, W.; Veckeneer, M.; Dralands, L.; Billiau, A.; Missotten, L. Monocyte chemotactic protein-1 in proliferative vitroretinal disorders. Am. J. Ophthalmol. 1997, 123, 599–606. [Google Scholar] [CrossRef]
- Zhang, S.X.; Wang, J.J.; Gao, G.; Shao, C.; Mott, R.; Ma, J.X. Pigmented epithelium-derived factor (PEDF) is an endogenous antiinflammatory factor. FASEB J. 2006, 20, 323–325. [Google Scholar] [PubMed]
- Mitamura, Y.; Takeuchi, S.; Matsuda, A.; Tagawa, Y.; Mizue, Y.; Nishihira, J. Monocyte chemotactic protein-1 in the vitreous of patients with proliferative diabetic retinopathy. Ophthalmologica 2001, 215, 415–418. [Google Scholar] [CrossRef] [PubMed]
- Portillo, J.-A.; Lopez Corcino, Y.; Dubyak, G.R.; Kern, T.S.; Matsuyama, S.; Subauste, C.S. Ligation of CD40 in human retinal Muller cells induces P2X7 receptor-dependent death of retinal endothelial cells. Investig. Ophthalmol. Vis. Sci. 2016, 57, 6278–6286. [Google Scholar] [CrossRef] [PubMed]
- Portillo, J.-A.; Schwartz, I.; Zarini, S.; Bapputy, R.; Kern, T.S.; Gubitosi-Klug, R.A.; Murphy, R.C.; Subauste, M.C.; Subauste, C.S. Pro-inflammatory responses induced by CD40 in retinal endothelial and Muller cells are inhibited by blocking CD40-TRAF2,3 or CD40-TRAF6 signaling. Investig. Ophthalmol. Vis. Sci. 2014, 55, 8590–8597. [Google Scholar] [CrossRef] [PubMed]
- Portillo, J.-A.C.; Greene, J.A.; Okenka, G.; Miao, Y.; Sheibani, N.; Kern, T.S.; Subauste, C.S. CD40 promotes the development of early diabetic retinopathy. Diabetologia 2014, 57, 2222–2231. [Google Scholar] [CrossRef] [PubMed]
- Portillo, J.-A.C.; Lopez Corcino, Y.; Miao, Y.; Tang, J.; Sheibani, N.; Kern, T.S.; Dubyak, G.R.; Subauste, C.S. CD40 in retinal Muller cells induces P2X7-dependent cytokine expression in macrophages/microglia in diabetic mice and development of early experimental diabetic retinopathy in mice. Diabetes 2017, 66, 483–493. [Google Scholar] [CrossRef] [PubMed]
- Grewal, I.S.; Flavell, R.A. CD40 and CD154 in cell-mediated immunity. Ann. Rev. Immunol. 1998, 16, 111–135. [Google Scholar] [CrossRef] [PubMed]
- Van Kooten, C.; Banchereau, J. CD40-CD40 ligand. J. Leuk. Biol. 2000, 67, 2–17. [Google Scholar]
- Andrade, R.M.; Wessendarp, M.; Gubbels, M.J.; Striepen, B.; Subauste, C.S. CD40 induces macrophage anti-Toxoplasma gondii activity by triggering autophagy-dependent fusion of pathogen-containing vacuoles and lysosomes. J. Clin. Invest. 2006, 116, 2366–2377. [Google Scholar] [CrossRef] [PubMed]
- Portillo, J.-A.C.; Okenka, G.; Kern, T.S.; Subauste, C.S. Identification of primary retinal cells and ex vivo identification of pro-inflammatory molecules in retinal cells using flow cytometry. Mol. Vis. 2009, 15, 1383–1389. [Google Scholar] [PubMed]
- Varo, N.; Vincent, D.; Libby, P.; Nuzzo, R.; Calle-Pascual, A.L.; Bernal, M.R.; Fernández-Cruz, A.; Veves, A.; Jarolim, P.; Varo, J.J.; et al. Elevated plasma levels of the atherogenic mediator soluble CD40 ligand in diabetic patients. A novel target of thiazolidinediones. Circulation 2003, 107, 2644–2649. [Google Scholar] [CrossRef] [PubMed]
- Yngen, M.; Ostenson, C.-G.; Hu, H.M.; Li, N.; Hjemdahl, P.; Wallen, N.H. Enhanced P-selectin expression and increased soluble CD40 ligand in patients with type 1 diabetes mellitus and microangiopathy: Evidence for platelet hyperactivity and chronic inflammation. Diabetologia 2004, 47, 537–540. [Google Scholar] [CrossRef] [PubMed]
- Cipollone, F.; Chiarelli, F.; Davi, G.; Ferri, C.; Desideri, G.; Fazia, M.; Iezzi, A.; Santilli, F.; Pini, B.; Cuccurullo, C.; et al. Enhanced soluble CD40 ligand contributes to endothelial cell dysfunction in vitro and monocyte activation in patients with diabetes mellitus: Effect of improved metabolic control. Diabetologia 2005, 48, 1216–1224. [Google Scholar] [CrossRef] [PubMed]
- Kiener, P.A.; Moran-Davis, P.; Rankin, B.M.; Wahl, A.F.; Aruffo, A.; Hollenbaugh, D. Stimulation of CD40 with purified soluble gp39 induces proinflammatory responses in human monocytes. J. Immunol. 1995, 155, 4917–4925. [Google Scholar] [PubMed]
- Portillo, J.-A.C.; Greene, J.A.; Schwartz, I.; Subauste, M.C.; Subauste, C.S. Blockade of CD40-TRAF2,3 or CD40-TRAF6 intercations is sufficient to impair pro-inflammatory responses in human aortic endothelial cells and human aortic smooth muscle cells. Immunology 2015, 144, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Portillo, J.-A.C.; Muniz-Feliciano, L.; Subauste, M.C.; Heinzel, F.P.; Subauste, C.S. CD40 and TNF-a synergize to induce nitric oxide synthase in macrophages. Immunology 2012, 135, 140–150. [Google Scholar] [CrossRef] [PubMed]
- Subauste, C.; Portillo, J.A.; Zheng, L.; Okenka, G.; Van Grol, J.; Kern, T. CD40 mediates retinal inflammation and neuro-vascular degeneration. J. Immunol. 2008, 181, 8719–8726. [Google Scholar]
- Abcouwer, S.F. Muller Cell-Microglia Cross Talk Drives Neuroinflammation in Diabetic Retinopathy. Diabetes 2017, 66, 261–263. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Xu, X.; Elliot, M.H.; Zhu, M.; Le, Y.-Z. Muller cell-derived VEGF is essential for diabetes-induced retinal inflammation and vascular leakage. Diabetes 2010, 59, 2297–2305. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.-P.; Sun, H.-L.; Wu, L.-M.; Guo, X.-J.; Dou, H.-L.; Tso, M.O.; Zhao, L.; Li, S.M. Baicalein reduces inflammatory process in a rodent model of diabetic retinopathy. Investig. Ophthalmol. Vis. Sci. 2009, 50, 2319–2327. [Google Scholar] [CrossRef] [PubMed]
- Monif, M.; Reid, C.A.; Powell, K.L.; Smart, M.L.; Williams, D.A. The P2X7 receptor drives microglial activation and proliferation: A trophic role for P2X7R pore. J. Neurosci. 2009, 29, 3781–3791. [Google Scholar] [CrossRef] [PubMed]
- Klaus, G.G.; Choi, M.S.; Holman, M. Properties of mouse CD40. Ligation of CD40 activates B cells via a Ca(++)-dependent, FK506-sensitive pathway. Eur. J. Immunol. 1994, 24, 3229–3232. [Google Scholar] [CrossRef] [PubMed]
- Lazaar, A.L.; Amrani, Y.; Hsu, J.; Panettieri, R.A., Jr.; Fanslow, W.C.; Albelda, S.M.; Puré, E. CD40-mediated signal transduction in human airway smooth muscle. J. Immunol. 1998, 161, 3120–3127. [Google Scholar] [PubMed]
- Du, Y.; Miller, C.M.; Kern, T.S. Hyperglycemia increases mitochondrial superoxide in retina and retinal cells. Free Radic. Biol. Med. 2003, 35, 1491–1499. [Google Scholar] [CrossRef] [PubMed]
- Kowluru, R.A.; Odenbach, S. Effect of rlong-term administration of alpha-lipoic acid on retinal capillary cell death and the development of retinopathy in diabetic rats. Diabetes 2004, 53, 3233–3238. [Google Scholar] [CrossRef] [PubMed]
- El-Remessy, A.B.; Al-Shabrawey, M.; Khalifa, Y.; Tsai, N.T.; Caldwell, R.B.; Liou, G.I. Neuroprotective and blood-retinal barrier-preserving effects of cannabidiol in experimental diabetes. Am. J. Pathol. 2006, 168, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Busik, J.V.; Mohr, S.; Grant, M.B. Hyperglycemia-induced reactive oxygen species toxicity to endothelial cells is dependent on paracrine mediators. Diabetes 2008, 57, 1952–1965. [Google Scholar] [CrossRef] [PubMed]
- Behl, Y.; Krothapalli, P.; Desta, T.; DiPiazza, A.; Roy, S.; Graves, D.T. Diabetes-enhanced tumor necrosis factor-alpha production promotes apoptosis and the loss of retinal microvascular cells in type 1 and type 2 models of diabetic retinopathy. Am. J. Pathol. 2008, 172, 1411–1418. [Google Scholar] [CrossRef] [PubMed]
- Joussen, A.M.; Doehmen, S.; Le, M.L.; Koizumi, K.; Radetzky, S.; Krohne, T.U.; Poulaki, V.; Semkova, I.; Kociok, N. TNF-a mediated apoptosis plays an important role in the development of early diabetic retinopathy and long-term histopathological alterations. Mol. Vis. 2009, 15, 1418–1428. [Google Scholar] [PubMed]
- Vincent, J.A.; Mohr, S. Inhibition of caspase-1/interleukin-1b signaling prevents degeneration of retinal capillaries in diabetes and galactosemia. Diabetes 2007, 56, 224–230. [Google Scholar] [CrossRef] [PubMed]
- Di Virgilio, F.; Chiozzi, P.; Falzoni, S.; Ferrari, D.; Sanz, J.M.; Enketaraman, V.; Baricordi, O.R. Cytolytic P2X purinoceptors. Cell Death Differ. 1998, 5, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, T. Enhancement of of P2X7-induced pore formation and apoptosis: An early effect of diabetes on the retinal microvasculature. Investig. Ophthalmol. Vis. Sci. 2004, 45, 1026–1032. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, T. Role of P2X7 receptors in the development of diabetic retinopathy. World J. Diabetes 2014, 5, 141–145. [Google Scholar] [PubMed]
- Solini, A.; Chiozzi, P.; Falzoni, S.; Morelli, A.; Fellin, R.; Di Virgilio, F. High glucose modulates P2X7 receptor-mediated function in human primary fibroblasts. Diabetologia 2000, 43, 1248–1256. [Google Scholar] [CrossRef] [PubMed]
- Solini, A.; Chiozzi, P.; Morelli, A.; Adinolfi, E.; Rizzo, R.; Baricordi, O.R.; di Virgilio, F. Enhanced P2X7 activity in human fibroblasts from diabetic patients: A possible pathogenetic mechanism for vascular damage in diabetes. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 1240–1245. [Google Scholar] [CrossRef] [PubMed]
- Mukundan, L.; Bishop, G.A.; Head, K.Z.; Zhang, L.; Wahl, L.; Suttles, J. TNF receptor-associated factor 6 is an essential mediator of CD40-activated proinflammatory pathways in monocytes and macrophages. J. Immunol. 2005, 174, 1081–1090. [Google Scholar] [CrossRef] [PubMed]
- Mackey, M.F.; Wang, Z.; Eichelberg, K.; Germain, R.N. Distinct contributions of different CD40 TRAF binding sites to CD154-induced dendritic cell maturation and IL-12 secretion. Eur. J. Immunol. 2003, 33, 779–789. [Google Scholar] [CrossRef] [PubMed]
- Greene, J.A.; Portillo, J.-A.; Lopez Corcino, Y.; Subauste, C.S. CD40-TRAF signaling upregulates CXC3L1 and TNF-a in human aortic endothelial cells but not in retinal endothelial cells. PLoS ONE 2015, 10, e0144133. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Subauste, C.S. CD40, a Novel Inducer of Purinergic Signaling: Implications to the Pathogenesis of Experimental Diabetic Retinopathy. Vision 2017, 1, 20. https://doi.org/10.3390/vision1030020
Subauste CS. CD40, a Novel Inducer of Purinergic Signaling: Implications to the Pathogenesis of Experimental Diabetic Retinopathy. Vision. 2017; 1(3):20. https://doi.org/10.3390/vision1030020
Chicago/Turabian StyleSubauste, Carlos S. 2017. "CD40, a Novel Inducer of Purinergic Signaling: Implications to the Pathogenesis of Experimental Diabetic Retinopathy" Vision 1, no. 3: 20. https://doi.org/10.3390/vision1030020
APA StyleSubauste, C. S. (2017). CD40, a Novel Inducer of Purinergic Signaling: Implications to the Pathogenesis of Experimental Diabetic Retinopathy. Vision, 1(3), 20. https://doi.org/10.3390/vision1030020




