Osteoclast Formation within a Human Co-Culture System on Bone Material as an In Vitro Model for Bone Remodeling Processes
Abstract
1. Introduction
2. Materials and Methods
2.1. Isolation of Human Monocytes
2.2. Induction of Osteoclastogenesis
2.3. Activated Cell Scanning via Flow Cytometry
2.4. hMSCs Isolation and Culture
2.5. Induction of Osteoblastic Differentiation of hMSCs
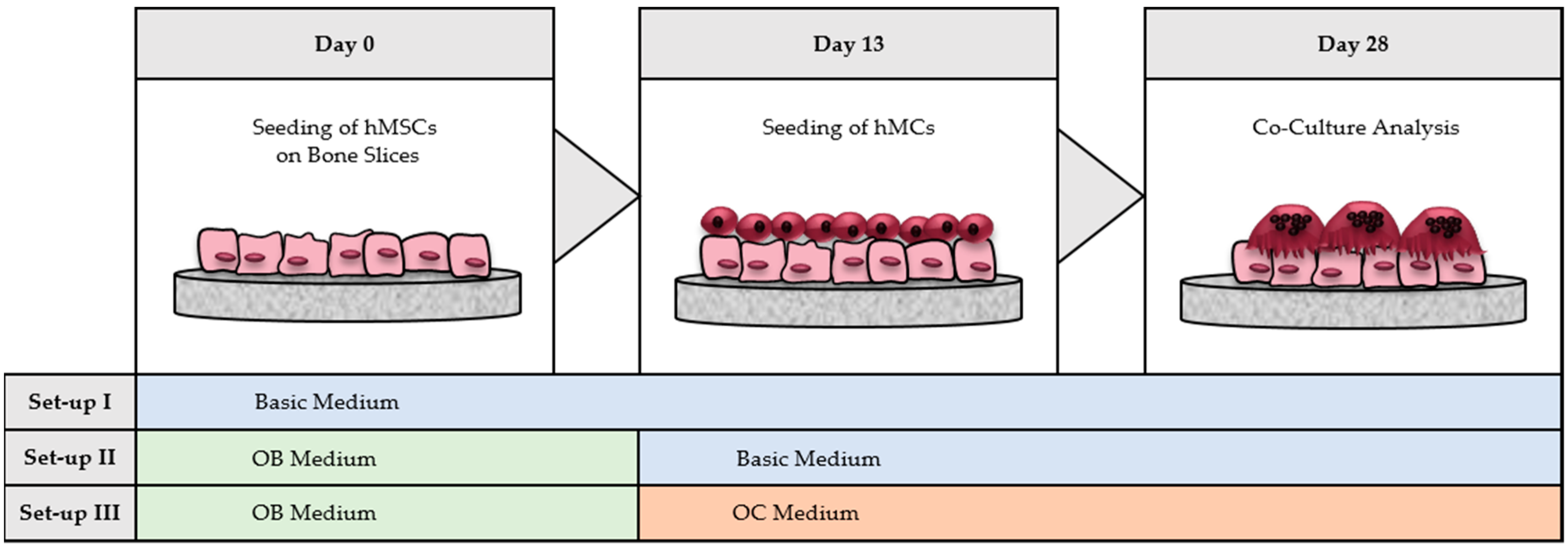
2.6. Co-Culture of hMSCs and hMCs on TCPS and Cortical Bone Slices
- hMSCs with hMCs: the complete co-culture was done in basic medium without adding differentiation factors.
- hOBs with hMCs: hMSCs were differentiated with OB medium until day 13. After hMCs were seeded on top of the generated hOBs, the co-culture was kept in basic medium.
- hOBs with hOCs: hMSCs were differentiated with OB medium until day 13. After seeding hMCs on top of hOBs, the co-culture was performed in OC medium by addition of osteoclastogenic differentiation factors (M-CSF and RANKL).
2.7. Fluorescence Staining
2.8. Vybrant™ Cell Labeling
2.9. Scanning Electron Microscopy (SEM)
2.10. Statistics
3. Results
3.1. Characterization of Human Monocytes
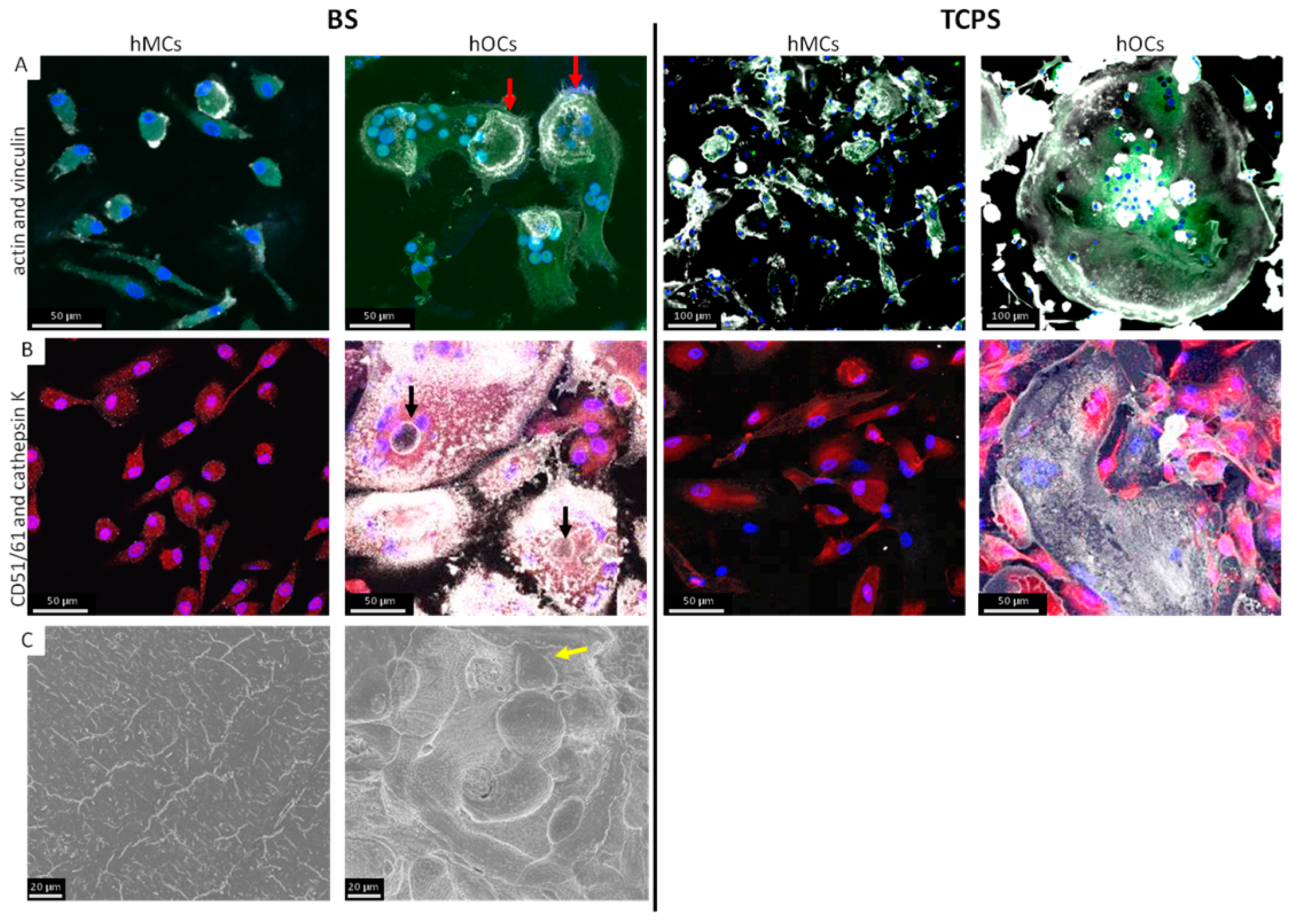
3.2. Comparison of hMCs and hOCs on TCPS and BS
3.3. Characterization of the Co-Culture Set-Ups on BS
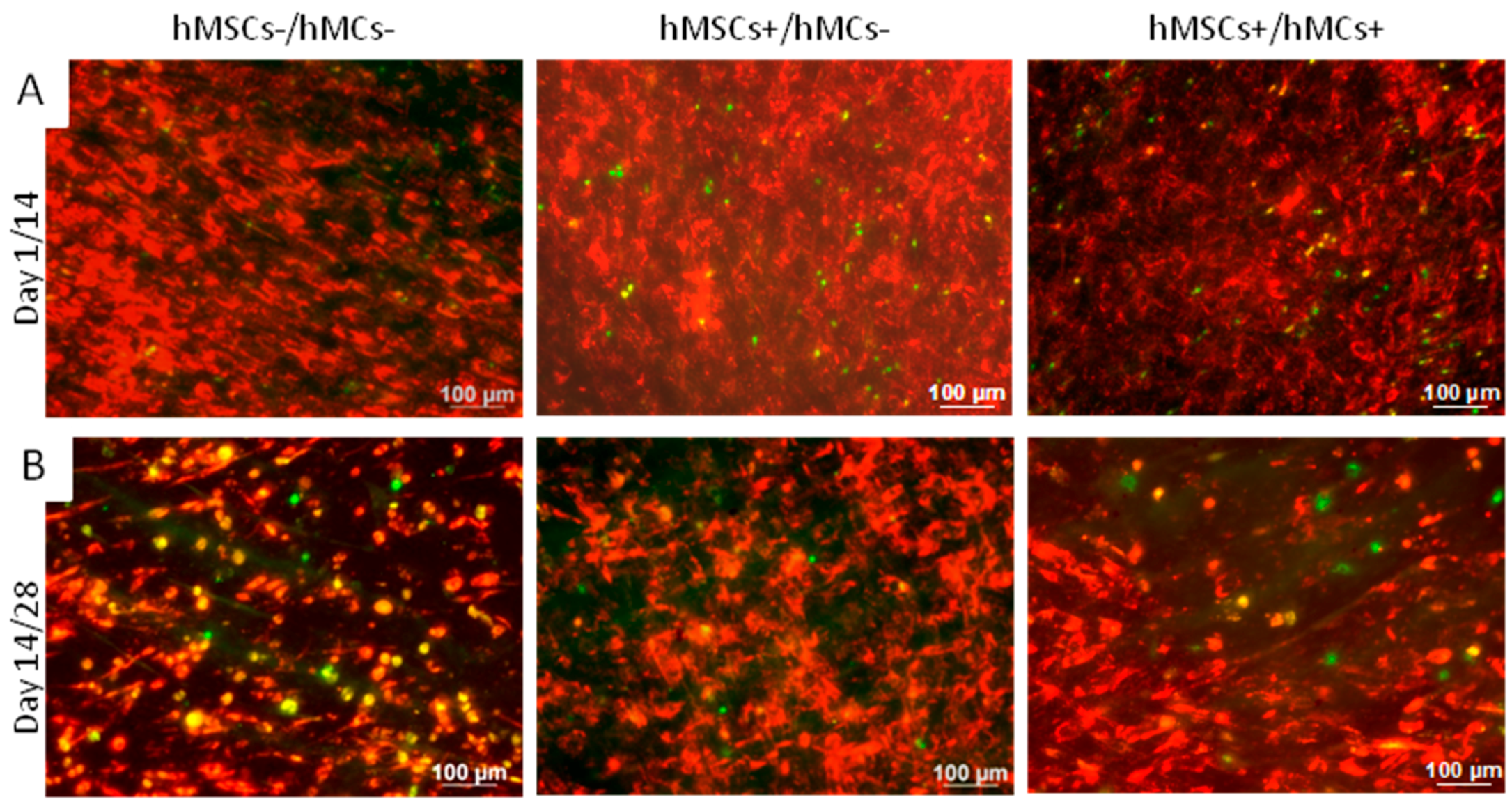
3.3.1. Distribution of hMCs and hMSC on BS in Our Co-Culture Set-Ups
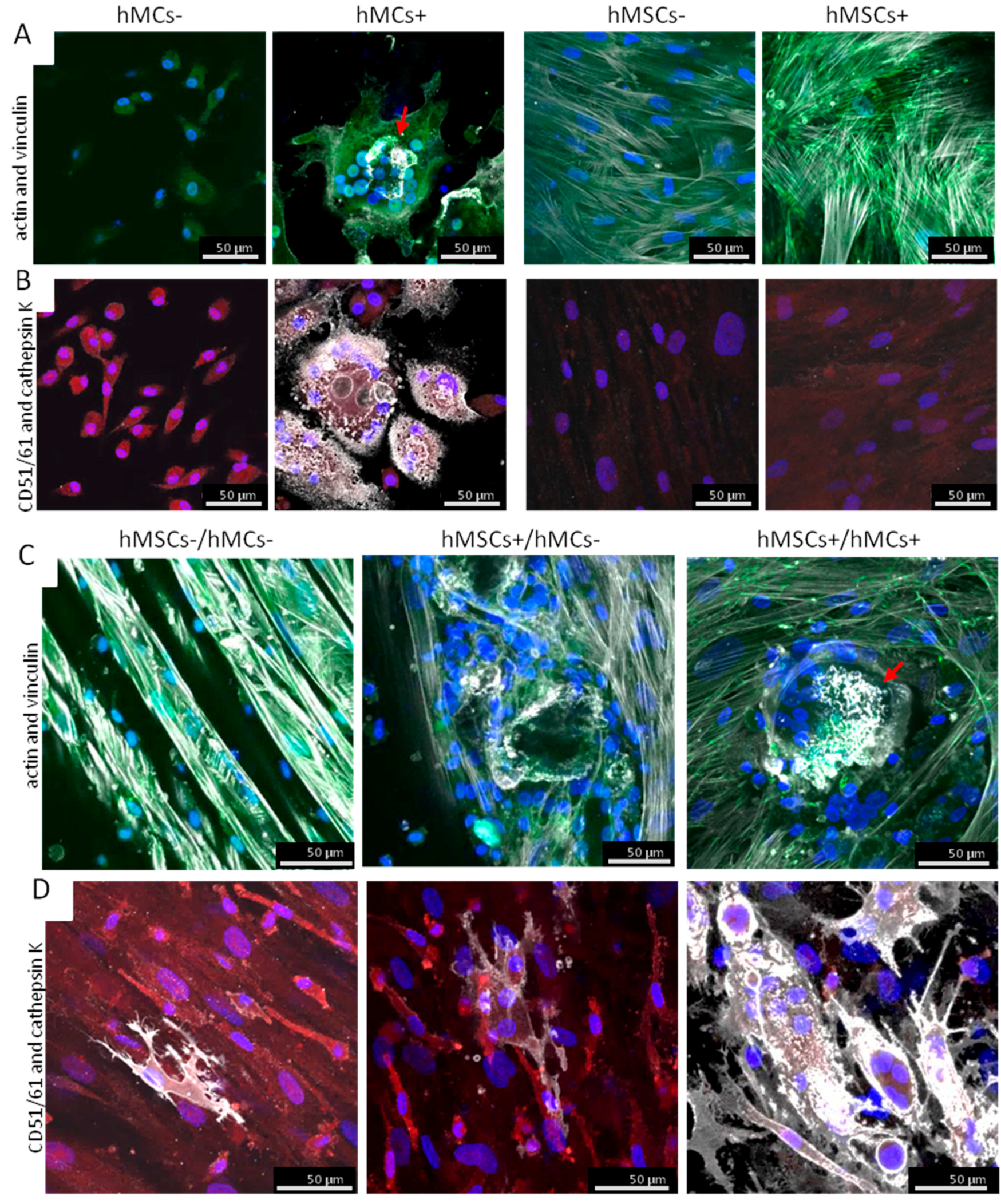
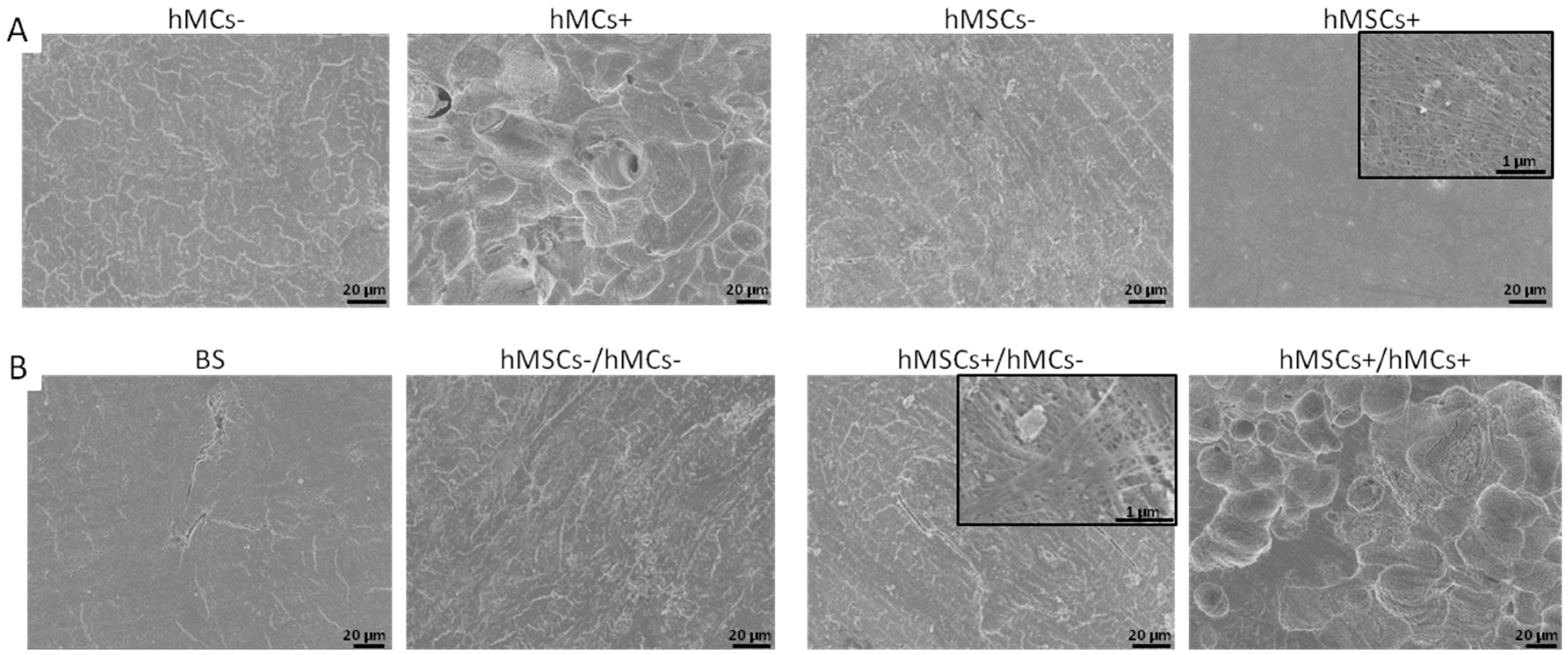
3.3.2. Morphological Analysis of the Osteoclastogenesis on BS in Co-Culture Set-Ups
3.3.3. Analysis of Osteoclast-Specific Bone Resorption Activity in Co-Culture Set-Ups on BS
4. Discussion
4.1. Characterization of Isolated hMCs
4.2. Comparison of Osteoclast Generation on TCPS and BS
4.3. Different Co-Culture Set-Ups in Comparison
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Salgado, A.J.; Coutinho, O.P.; Reis, R.L. Bone tissue engineering: State of the art and future trends. Macromol. Biosci. 2004, 4, 743–765. [Google Scholar] [CrossRef] [PubMed]
- Hadjidakis, D.J.; Androulakis, I.I. Bone remodeling. Ann. N. Y. Acad. Sci. 2006, 1092, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Roodman, G.D. Cell biology of the osteoclast. Exp. Hematol. 1999, 27, 1229–1241. [Google Scholar] [CrossRef]
- Roodman, G.D. Regulation of osteoclast differentiation. Ann. N. Y. Acad. Sci. 2006, 1068, 100–109. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, T. Nakashima, H. Takayanagi, Osteocyte control of osteoclastogenesis. Bone 2013, 54, 258–263. [Google Scholar] [CrossRef] [PubMed]
- Suda, T.; Takahashi, N.; Udagawa, N.; Jimi, E.; Gillespie, M.T.; Martin, T.J. Modulation of osteoclast differentiation and function by the new members of the tumor necrosis factor receptor and ligand families. Endocr. Rev. 1999, 20, 345–357. [Google Scholar] [CrossRef] [PubMed]
- Cody, J.J.; Rivera, A.A.; Liu, J.; Liu, J.M.; Douglas, J.T.; Feng, X. A simplified method for the generation of human osteoclasts in vitro. Int. J. Biochem. Mol. Biol. 2011, 2, 183–189. [Google Scholar] [PubMed]
- Avnet, S.; Lamolinara, A. Effects of antisense mediated inhibition of cathepsin K on human osteoclasts obtained from peripheral blood. J. Orthop. Res. 2006, 24, 1699–1708. [Google Scholar] [CrossRef] [PubMed]
- Bronner, F.; Farach-Carson, M.C.; Rubin, J. Bone Resorption; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2006. [Google Scholar]
- Kleinhans, C.; Schmid, F.F.; Schmid, F.V.; Kluger, P.J. Comparison of osteoclastogenesis and resorption activity of human osteoclasts on tissue culture polystyrene and on natural extracellular bone matrix in 2D and 3D. J. Biotechnol. 2015, 205, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Mercatali, L.; Spadazzi, C.; Miserocchi, G.; Liverani, C.; De, A.V.; Bongiovanni, A.; Recine, F.; Amadori, D.; Ibrahim, T. Development of a Human Preclinical Model of Osteoclastogenesis from Peripheral Blood Monocytes Co-cultured with Breast Cancer Cell Lines. J. Vis. Exp. 2017. [CrossRef] [PubMed]
- Penolazzi, L.; Lolli, A.; Sardelli, L.; Angelozzi, M.; Lambertini, E.; Trombelli, L.; Ciarpella, F.; Vecchiatini, R.; Piva, R. Establishment of a 3D-dynamic osteoblasts-osteoclasts co-culture model to simulate the jawbone microenvironment in vitro. Life Sci. 2016, 152, 82–93. [Google Scholar] [CrossRef] [PubMed]
- Iwamoto, Y.; Nishikawa, K.; Imai, R.; Furuya, M.; Uenaka, M.; Ohta, Y.; Morihana, T.; Itoi-Ochi, S.; Penninger, J.M.; Katayama, I.; et al. Intercellular Communication between Keratinocytes and Fibroblasts Induces Local Osteoclast Differentiation: A Mechanism Underlying Cholesteatoma-Induced Bone Destruction. Mol. Cell. Biol. 2016, 36, 1610–1620. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.K.; Zhang, M.C. IL-23 promotes osteoclastogenesis in osteoblast-osteoclast co-culture system. Genet. Mol. Res. 2014, 13, 4673–4679. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, A.; Rotwein, P. Selective signaling by Akt1 controls osteoblast differentiation and osteoblast-mediated osteoclast development. Mol. Cell. Biol. 2012, 32, 490–500. [Google Scholar] [CrossRef] [PubMed]
- Yao, Z.; Xing, L.; Qin, C.; Schwarz, E.M.; Boyce, B.F. Osteoclast precursor interaction with bone matrix induces osteoclast formation directly by an interleukin-1-mediated autocrine mechanism. J. Biol. Chem. 2008, 283, 9917–9924. [Google Scholar] [CrossRef] [PubMed]
- Guihard, P.; Danger, Y.; Brounais, B.; David, E.; Brion, R.; Delecrin, J.; Richards, C.; Chevalier, S.; Redini, F.; Heymann, D.; et al. Induction of osteogenesis in mesenchymal stem cells by activated monocytes/macrophages depends on Oncostatin M signaling. Stem Cells 2012, 30, 762–772. [Google Scholar] [CrossRef] [PubMed]
- Nicolaidou, V.; Wong, M.M.; Redpath, A.N.; Ersek, A.; Baban, D.F.; Williams, L.M.; Cope, A.P.; Horwood, N.J. Monocytes induce STAT3 activation in human mesenchymal stem cells to promote osteoblast formation. PLoS ONE 2012, 7, e39871. [Google Scholar] [CrossRef] [PubMed]
- Pirraco, R.P.; Reis, R.L.; Marques, A.P. Effect of monocytes/macrophages on the early osteogenic differentiation of hBMSCs. J. Tissue Eng. Regen. Med. 2013, 7, 392–400. [Google Scholar] [CrossRef] [PubMed]
- Zachos, C.; Steubesand, N.; Seekamp, A.; Fuchs, S.; Lippross, S. Co-cultures of programmable cells of monocytic origin and mesenchymal stem cells do increase osteogenic differentiation. J. Orthop. Res. 2014, 32, 1264–1270. [Google Scholar] [CrossRef] [PubMed]
- Heinemann, C.; Heinemann, S.; Worch, H.; Hanke, T. Development of an osteoblast/osteoclast co-culture derived by human bone marrow stromal cells and human monocytes for biomaterials testing. Eur. Cell Mater. 2011, 21, 80–93. [Google Scholar] [CrossRef] [PubMed]
- Heinemann, S.; Heinemann, C.; Wenisch, S.; Alt, V.; Worch, H.; Hanke, T. Calcium phosphate phases integrated in silica/collagen nanocomposite xerogels enhance the bioactivity and ultimately manipulate the osteoblast/osteoclast ratio in a human co-culture model. Acta Biomater. 2013, 9, 4878–4888. [Google Scholar] [CrossRef] [PubMed]
- Kleinhans, C.; Barz, J.; Wurster, S.; Willig, M.; Oehr, C.; Müller, M.; Walles, H.; Hirth, T.; Kluger, P.J. Ammonia plasma treatment of polystyrene surfaces enhances proliferation of primary human mesenchymal stem cells and human endothelial cells. Biotechnol. J. 2013, 8, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Massey, H.M.; Flanagan, A.M. Human osteoclasts derive from CD14-positive monocytes. Br. J. Haematol. 1999, 106, 167–170. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, G.C.; Malakellis, M.; Collier, F.M.; Cameron, P.U.; Holloway, W.R.; Gough, T.J.; Gregorio-King, C.; Kirkland, M.A.; Myers, D.E. Induction of osteoclasts from CD14-positive human peripheral blood mononuclear cells by receptor activator of nuclear factor kappaB ligand (RANKL). Clin. Sci. Lond. 2000, 99, 133–140. [Google Scholar] [CrossRef]
- Walter, G.J.; Evans, H.G.; Menon, B.; Gullick, N.J.; Kirkham, B.W.; Cope, A.P.; Geissmann, F.; Taams, L.S. Interaction with activated monocytes enhances cytokine expression and suppressive activity of human CD4+CD45RO+CD25+CD127low regulatory T cells. Arthritis Rheum. 2013, 65, 627–638. [Google Scholar] [CrossRef] [PubMed]
- Quinn, J.M.W.; Fujikawa, Y.; McGee, J.O.D.; Athanasou, N.A. Rodent osteoblast-like cells support osteoclastic differentiation of human cord blood monocytes in the presence of M-CSF and 1,25 dihydroxyvitamin D3. Int. J. Biochem. Cell Biol. 1997, 29, 173–179. [Google Scholar] [CrossRef]
- Heinemann, E.; Siggelkow, H.; Ponce, L.M.; Viereck, V.; Wiese, K.G.; Peters, J.H. Alkaline phosphatase expression during monocyte differentiation. Overlapping markers as a link between monocytic cells, dendritic cells, osteoclasts and osteoblasts. Immunobiology 2000, 202, 68–81. [Google Scholar] [CrossRef]
- Meyer, U.; Meyer, T.; Handschel, J.; Wiesmann, H. Fundamentals of Tissue Engineering and Regenerative Medicine; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2009. [Google Scholar]
- Sekine, C.; Koyanagi, A.; Koyama, N.; Hozumi, K.; Chiba, S.; Yagita, H. Differential regulation of osteoclastogenesis by Notch2/Delta-like 1 and Notch1/Jagged1 axes. Arthritis Res. Ther. 2012, 14, R45. [Google Scholar] [CrossRef] [PubMed]
- Udagawa, N.; Takahashi, N.; Jimi, E.; Matsuzaki, K.; Tsurukai, T.; Itoh, K.; Nakagawa, N.; Yasuda, H.; Goto, M.; Tsuda, E.; et al. Osteoblasts/stromal cells stimulate osteoclast activation through expression of osteoclast differentiation factor/RANKL but not macrophage colony-stimulating factor. Bone 1999, 25, 517–523. [Google Scholar] [CrossRef]
- Bernhardt, A.; Schumacher, M.; Gelinsky, M. Formation of osteoclasts on calcium phosphate bone cements and polystyrene depends on monocyte isolation conditions. Tissue Eng. Part C Methods 2014, 21, 160–170. [Google Scholar] [CrossRef] [PubMed]
- Capasso, R.; Sambri, I.; Cimmino, A.; Salemme, S.; Lombardi, C.; Acanfora, F.; Satta, E.; Puppione, D.L.; Perna, A.F.; Ingrosso, D. Homocysteinylated albumin promotes increased monocyte-endothelial cell adhesion and up-regulation of MCP1, Hsp60 and ADAM17. PLoS ONE 2012, 7, e31388. [Google Scholar] [CrossRef] [PubMed]
- Babaei, S.; Fekete, N.; Hoesli, C.A.; Girard-Lauriault, P.-L. Adhesion of Human Monocytes to Oxygen- and Nitrogen- containing Plasma Polymers: Effect of Surface Chemistry and Protein Adsorption. Colloids Surfaces B Biointerfaces 2017, 162, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Saunders, E.; Johnson, P. Modulation of immune cell signalling by the leukocyte common tyrosine phosphatase, CD45. Cell. Signal. 2010, 22, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Hermann, P.; Armant, M.; Brown, E.; Rubio, M.; Ishihara, H.; Ulrich, D.; Caspary, R.G.; Lindberg, F.P.; Armitage, R.; Maliszewski, C.; et al. The vitronectin receptor and its associated CD47 molecule mediates proinflammatory cytokine synthesis in human monocytes by interaction with soluble CD23. J. Cell Biol. 1999, 144, 767–775. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, N.; Ejiri, S.; Yanagisawa, S.; Ozawa, H. Regulation of osteoclast polarization. Odontology 2007, 95, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Ciobanasu, C.; Faivre, B.; Le Clainche, C. Actin dynamics associated with focal adhesions. Int. J. Cell Biol. 2012, 2012, 941292. [Google Scholar] [CrossRef] [PubMed]
- Takito, J.; Otsuka, H.; Inoue, S.; Kawashima, T.; Nakamura, M. Symmetrical retrograde actin flow in the actin fusion structure is involved in osteoclast fusion. Biol. Open 2017, 6, 1104–1114. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, N.; Udagawa, N.; Kobayashi, Y.; Suda, T. Generation of osteoclasts in vitro; assay of osteoclast activity. Methods Mol. Med. 2007, 135, 285–301. [Google Scholar] [PubMed]
- Saltel, F.; Destaing, O.; Bard, F. Apatite-mediated actin dynamics in resorbing osteoclasts. Mol. Biol. 2004, 15, 5231–5241. [Google Scholar] [CrossRef] [PubMed]
- Song, R.L.; Liu, X.Z.; Zhu, J.Q.; Zhang, J.M.; Gao, Q.; Zhao, H.Y.; Sheng, A.Z.; Yuan, Y.; Gu, J.H.; Zou, H.; et al. New roles of filopodia and podosomes in the differentiation and fusion process of osteoclasts. Genet. Mol. Res. 2014, 13, 4776–4787. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Nagase, Y.; Hirose, J.; Tokuyama, N.; Yasui, T.; Kadono, Y.; Ueki, K.; Kadowaki, T.; Nakamura, K.; Tanaka, S. Regulation of bone resorption and sealing zone formation in osteoclasts occurs through protein kinase B-mediated microtubule stabilization. J. Bone Miner. Res. 2013, 28, 1191–1202. [Google Scholar] [CrossRef] [PubMed]
- Bernhardt, A.; Thieme, S.; Domaschke, H.; Springer, A.; Rösen-Wolff, A.; Gelinsky, M. Crosstalk of osteoblast and osteoclast precursors on mineralized collagen--towards an in vitro model for bone remodeling. J. Biomed. Mater. Res. A 2010, 95, 848–856. [Google Scholar] [CrossRef] [PubMed]
- Mbalaviele, G.; Jaiswal, N.; Meng, A.; Cheng, L.; Van Den Bos, C.; Thiede, M. Human mesenchymal stem cells promote human osteoclast differentiation from CD34+ bone marrow hematopoietic progenitors. Endocrinology 1999, 140, 3736–3743. [Google Scholar] [CrossRef] [PubMed]
- Heinemann, C.; Heinemann, S.; Bernhardt, A. In vitro osteoclastogenesis on textile chitosan scaffold. Eur. Cell Mater. 2010, 19, 96–106. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, N.; Akatsu, T.; Udagawa, N.; Sasaki, T.; Yamaguchi, A.; Moseley, J.M.; Martin, T.J.; Suda, T. Osteoblastic cells are involved in osteoclast formation. Endocrinology 1988, 123, 2600–2602. [Google Scholar] [CrossRef] [PubMed]
- Jäger, M.; Fischer, J.; Dohrn, W.; Li, X.; Ayers, D.C.; Czibere, A.; Prall, W.C.; Lensing-Höhn, S.; Krauspe, R. Dexamethasone modulates BMP-2 effects on mesenchymal stem cells in vitro. J. Orthop. Res. 2008, 26, 1440–1448. [Google Scholar] [CrossRef] [PubMed]
- Turgeman, G.; Pittman, D.D.; Mueller, R.; Gowda Kurkalli, B.; Zhou, S.; Pelled, G.; Peyser, A.; Zilberman, Y.; Moutsatsos, I.K.; Gazit, D. Engineered human mesenchymal stem cells: a novel platform for skeletal cell mediated gene therapy. J. Gene Med. 2001, 3, 240–251. [Google Scholar] [CrossRef]
- Artmann, G.M.; Minger, S.; Hescheler, J. Stem Cell Engineering: Principles and Applications; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2010. [Google Scholar]
- Shugg, R.P.P.; Thomson, A.; Tanabe, N.; Kashishian, A.; Steiner, B.H.; Puri, K.D.; Pereverzev, A.; Lannutti, B.J.; Jirik, F.R.; Dixon, S.J.; et al. Effects of isoform-selective phosphatidylinositol 3-kinase inhibitors on osteoclasts: actions on cytoskeletal organization, survival and resorption. J. Biol. Chem. 2013, 288, 35346–35357. [Google Scholar] [CrossRef] [PubMed]
- Schulze, S.; Wehrum, D.; Dieter, P.; Hempel, U. A supplement-free osteoclast-osteoblast co-culture for pre-clinical application. J. Cell. Physiol. 2017, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Lutter, A.-H.; Hempel, U.; Wolf-Brandstetter, C.; Garbe, A.I.; Goettsch, C.; Hofbauer, L.C.; Jessberger, R.; Dieter, P. A novel resorption assay for osteoclast functionality based on an osteoblast-derived native extracellular matrix. J. Cell. Biochem. 2010, 109, 1025–1032. [Google Scholar] [PubMed]
- Battiston, K.G.; Cheung, J.W.C.; Jain, D.; Santerre, J.P. Biomaterials in co-culture systems: Towards optimizing tissue integration and cell signaling within scaffolds. Biomaterials 2014, 35, 4465–4476. [Google Scholar] [CrossRef] [PubMed]
| Aliqout | Antibody Combination |
|---|---|
| 1 | Autofluorescence (undyed control) |
| 2 | 51/61 FITC, IgG1-PE, IgG2a-APC |
| 3 | IgG1 FITC, 45-PE, IgG2a-APC |
| 4 | IgG1 FITC, IgG1-PE, CD14-APC |
| 5 | 51-61 FITC, 45-PE, CD14-APC |
| Antibody/Fluorophore | Product Code | Manufacturer | Registered Office |
|---|---|---|---|
| Purified Mouse IgG3, Isotype Clone MG3-35 | 401302 | BioLegend Biozol | Eching (D) |
| Maus IgG1 | X0931 | DAKO GmbH | Glostrup (DK) |
| Rabbit IgG, monoclonal isotype control SP137 | ab128142 | Abcam | Cambridge (USA) |
| Antibody-Diluent | S302283-2 | Acris GmbH | Herford (D) |
| Actin staining | |||
| Alexa Fluor 546-Phalloidin | A22283 | Invitrogen | Karlsruhe (D) |
| Vinculin staining | |||
| Monoclonal Anti-Vinculin-FITC antibody | F7053-500UL | Sigma-Aldrich | Steinheim (D) |
| CD51/61 staining | |||
| Anti-CD51 + CD61 antibody [23C6] | ab34226 | Abcam | Cambridge (USA) |
| Alexa Fluor F(ab′)2 488 Fragment of Goat Anti-Mouse IgG (H+L) Antibody | A-11017 | Life technologies | Darmstadt (D) |
| Cathepsin K | |||
| Anti-Cathepsin K antibody | ab19027 | Abcam | Cambridge (USA) |
| Alexa Fluor F(ab′)2 555 | A-21430 | Life technologies | Darmstadt (D) |
| DAPI (4′,6-Diamidin-2-phenylindol) | 18860.01 | Serva Electrophoresis GmbH | Heidelberg (D) |
| CD14 (%) | CD45 (%) | CD51/61 (%) | |
|---|---|---|---|
| DGC | 21.00 ± 13.35 | 96.66 ± 0.98 | 0.22 ± 0.12 |
| 1.5 h PA | 34.60 ± 9.37 | 95.52 ± 1.35 | 0.25 ± 0.12 |
| 1.5 h PA and 7 days cultured | 37.43 ± 2.52 | 99.42 ± 0.66 | 0.40 ± 0.53 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schmid, F.V.; Kleinhans, C.; Schmid, F.F.; Kluger, P.J. Osteoclast Formation within a Human Co-Culture System on Bone Material as an In Vitro Model for Bone Remodeling Processes. J. Funct. Morphol. Kinesiol. 2018, 3, 17. https://doi.org/10.3390/jfmk3010017
Schmid FV, Kleinhans C, Schmid FF, Kluger PJ. Osteoclast Formation within a Human Co-Culture System on Bone Material as an In Vitro Model for Bone Remodeling Processes. Journal of Functional Morphology and Kinesiology. 2018; 3(1):17. https://doi.org/10.3390/jfmk3010017
Chicago/Turabian StyleSchmid, Franziska V., Claudia Kleinhans, Freia F. Schmid, and Petra J. Kluger. 2018. "Osteoclast Formation within a Human Co-Culture System on Bone Material as an In Vitro Model for Bone Remodeling Processes" Journal of Functional Morphology and Kinesiology 3, no. 1: 17. https://doi.org/10.3390/jfmk3010017
APA StyleSchmid, F. V., Kleinhans, C., Schmid, F. F., & Kluger, P. J. (2018). Osteoclast Formation within a Human Co-Culture System on Bone Material as an In Vitro Model for Bone Remodeling Processes. Journal of Functional Morphology and Kinesiology, 3(1), 17. https://doi.org/10.3390/jfmk3010017




