Clinical Implementation of Exercise Guidelines for Cancer Patients: Adaptation of ACSM’s Guidelines to the Italian Model
Abstract
:1. Introduction
2. Medical Treatment Considerations and Contraindications
2.1. Surgery
2.2. Surgical Complications: Lymphedema
2.3. Radiotherapy (RT) Induced Pathology
2.4. Chemotherapy (CT) Induced Pathology
2.5. Cancer Related Fatigue (CRF)
2.6. Cancer Induced Peripheral Neuropathy (CIPN)
2.7. Metabolic Disturbances
3. Contraindications for Participating in Exercise Programs among Cancer Patients
3.1. Medical Assessment
3.2. Other Assessments
3.3. Exercise Guidelines
3.4. Exercise Intensity
3.5. Practical Application
3.5.1. Modality
3.5.2. Anaerobic-Resistance Training
3.5.3. Intensity
3.5.4. Flexibility
3.5.5. Nutrition
- Loss of appetite
- Sore mouth or throat
- Dry mouth
- Dental and gum problems
- Changes in taste or smell
- Nausea
- Vomiting
- Diarrhea
- Constipation
- Cancer related fatigue
- Depression
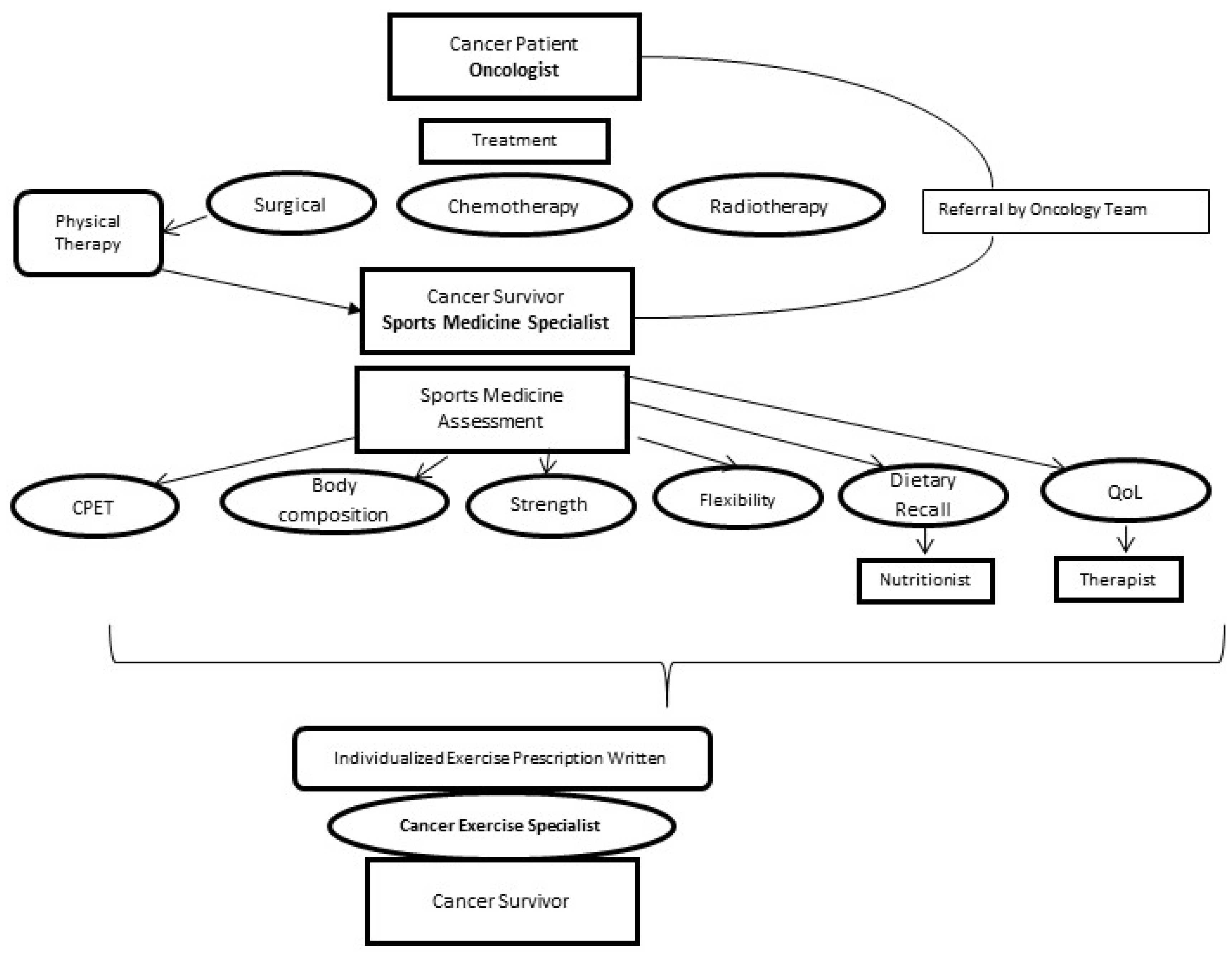
3.6. How to Approach the Patient Flow Chart, Italian Model
4. Conclusions
Author Contributions
Conflicts of Interest
References
- Stewart, B.W.; Wild, C.P. (Eds.) World Cancer Report 2014; International Agency for Research on Cancer: Lyon, France, 2014.
- Hewitt, M.; Greenfield, S.; Stovall, E. (Eds.) From Cancer Patient to Cancer Survivor: Lost in Transition; National Academies Press: Washington, DC, USA, 2006.
- Jones, L.W.; Eves, N.D.; Haykowsky, S.; Freeland, S.J.; Mackey, J.R. Exercise intolerance in cancer and the role of exercise therapy to reverse dysfunction. Lancet Oncol. 2009, 10, 598–605. [Google Scholar] [CrossRef]
- Gould, D.W.; Lahart, I.; Carmichael, A.R.; Koutedakis, Y.; Metsios, G.S. Cancer cachexia prevention via physical exercise: Molecular mechanisms. J. Cachexia Sarcopenia Muscle 2012, 4, 111–124. [Google Scholar] [CrossRef] [PubMed]
- Hanson, E.D.; Wagoner, C.W.; Anderson, T.; Battaglini, C.L. The independent effects of strength training in cancer survivors: A systematic review. Curr. Oncol. Rep. 2016, 18, 31–49. [Google Scholar] [CrossRef] [PubMed]
- Hofman, M.; Ryan, J.L.; Figueroa-Moseley, C.D.; Jean-Pierre, P.; Morrow, G.R. Cancer related fatigue: The scale of the problem. Oncologist 2007, 12, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Bao, T.; Basal, C.; Seluzicki, C.; Li, S.Q.; Seidman, A.D.; Mao, J.J. Long-term chemotherapy-induced peripheral neuropathy among breast cancer survivors: Prevalence, risk factors, and fall risk. Breast Cancer Res. Treat. 2016, 159, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Minami, M.; Matsumoto, S.; Horiuchi, H. Cardiovascular side-effects of modern cancer therapy. Circ. J. 2010, 74, 1779–1786. [Google Scholar] [CrossRef] [PubMed]
- Stefani, L.; Petri, C.; Mascherini, G.; Galanti, G. Lifestyle intervention in surviving cancer patients. J. Func. Morphol. Kinesiol. 2016, 1, 48–53. [Google Scholar] [CrossRef]
- Albrecht, T.A.; Taylor, A.G. Physical activity in patients with advanced-stage cancer: A systematic review of the literature. Clin. J. Oncol. Nurs. 2012, 16, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Conn, V.S.; Hafdahl, A.R.; Porock, D.C.; McDaniel, R.; Nielsen, P.J. A meta-analysis of exercise interventions among people treated for cancer. Support. Care Cancer 2006, 14, 699–712. [Google Scholar] [CrossRef] [PubMed]
- Speck, R.M.; Courneya, K.S.; Masse, L.C.; Duval, S.; Schmitz, K.H. An update of controlled physical activity trials in cancer survivors: A systematic review and meta-analysis. J. Cancer Surviv. 2010, 4, 87–100. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, K.H.; Courneya, K.S.; Matthews, C.; Demark-Wahnefried, W.; Galvão, D.A.; Pinto, B.M.; Irwin, M.L.; Wolin, K.Y.; Segal, R.J.; Lucia, A.; et al. American College of Sports Medicine roundtable on exercise guidelines for cancer survivors. Med. Sci. Sports Exerc. 2010, 42, 1409–1426. [Google Scholar] [CrossRef] [PubMed]
- Wonders, K.Y.; Reigle, B.S.; Drury, D.G. Treatment strategies for chemotherapy-induced peripheral neuropathy: Potential role of exercise. Oncol. Rev. 2010, 4, 117–125. [Google Scholar] [CrossRef]
- Yancik, R. Population aging and cancer: A cross-national concern. Cancer J. 2005, 11, 437–441. [Google Scholar] [CrossRef] [PubMed]
- Liberman, K.; Forti, L.N.; Beyer, I.; Bautmans, I. The effects of exercise on muscle strength, body composition, physical functioning and the inflammatory profile of older adults: A systematic review. Curr. Opin. Clin. Nutr. Metab. Care 2016, 20, 30–53. [Google Scholar] [CrossRef] [PubMed]
- Meneses-Echavez, J.F.; Correa-Bautista, J.E.; Gonzalez-Jimenez, E.; Schmidt Rio-Valle, J.; Elkins, M.R.; Lobelo, F.; Ramirez-Velez, R. The effect of exercise training on mediators of inflammation in breast cancer survivors: A systematic review with meta-analysis. Cancer Epidemiol. Biomark. Prev. 2016, 25, 1009–1017. [Google Scholar] [CrossRef] [PubMed]
- Van Blarigan, E.L.; Meyerhardt, J.A. Role of physical activity and diet after colorectal cancer diagnosis. J. Clin. Oncol. 2015, 33, 1825–1834. [Google Scholar] [CrossRef] [PubMed]
- DiSipio, T.; Rye, S.; Newman, B.; Hayes, S. Incidence of unilateral arm lymphedema after breast cancer: A systematic review and meta-analysis. Lancet Oncol. 2013, 14, 500–515. [Google Scholar] [CrossRef]
- NLN Medical Advisory Committee, National Lymphedema Network. Position Statement on the Diagnosis and Treatment of Lymphedema. Available online: www.lymphnet.org (accessed on 1 January 2017).
- Ezzo, J.; Manheimer, E.; McNeely, M.L.; Howell, D.M.; Weiss, R.; Johansson, K.I.; Bao, T.; Bily, L.; Tuppo, C.M.; Williams, A.F.; et al. Manual lymphatic drainage for lymphedema following breast cancer treatment. Cochrane Database Syst. Rev. 2015. [Google Scholar] [CrossRef]
- Schmitz, K.H.; Ahmed, R.L.; Troxel, A.B.; Cheville, A.; Lewis-Grant, L.; Smith, R.; Bryan, C.J.; Williams-Smith, C.T.; Chittams, J. Weight lifting for women at risk for breast cancer-related lymphedema: A randomized trial. JAMA 2010, 304, 2699–2705. [Google Scholar] [CrossRef] [PubMed]
- Getz, C.L.; Phillips, J.; Skirven, T.M.; Osterman, A.L.; Fedorczyk, J.M.; Adadio, P.C. Adhesive capsulitis. In Rehabilitation of the Hand and Upper Extremity, 6th ed.; Elsevier Mosby: Philadelphia, PA, USA, 2011; Chapter 89. [Google Scholar]
- Taunk, N.K.; Haffty, B.G.; Kostis, J.B.; Goyal, S. Radiation-induced heart disease: Pathologic abnormalities and putative mechanisms. Front. Oncol. 2015, 5, 39. [Google Scholar] [CrossRef] [PubMed]
- Zinzani, P.L.; Gherlinzoni, F.; Piovaccari, G.; Frezza, G.; Bendandi, M.; Ferretti, R.M.; Barbieri, E.; Fiacchini, M.; Babini, L.; Magnani, B.; et al. Cardiac injury as late toxicity of mediastinal radiation therapy for Hodgkin’s disease patients. Haematologica 1996, 81, 132–137. [Google Scholar] [PubMed]
- Madan, R.; Benson, R.; Sharma, D.N.; Julka, P.K.; Rath, G.K. Radiation induced heart disease: Pathogenesis, management and review literature. J. Egypt. Natl. Cancer Inst. 2015, 4, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Slamon, D.J.; Clark, G.M.; Wong, S.G.; Levin, W.J.; Ullrich, A.; McGuire, W.L. Human breast cancer: Correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science 1987, 235, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Stefani, L.; Pedrizzetti, G.; Galanti, G. Clinical application of 2D speckle tracking strain for assessing cardio-toxicity in oncology. J. Func. Morphol. Kinesiol. 2016, 1, 343–354. [Google Scholar] [CrossRef]
- Altena, R.; Perik, P.J.; van Veldhuisen, D.J.; de Vries, E.G.; Gietema, J.A. Cardiovascular toxicity caused by cancer treatment: Strategies for early detection. Lancet Oncol. 2009, 10, 391–399. [Google Scholar] [CrossRef]
- Fiuza-Luces, C.; Garatachea, N.; Berger, N.A.; Lucia, A. Exercise is the real polypill. Physiology 2013, 28, 330–358. [Google Scholar] [CrossRef] [PubMed]
- Mustian, K.M.; Morrow, G.R.; Carroll, J.K.; Figueroa-Moseley, C.D.; Jean-Pierre, P.; Williams, G.C. Integrative nonpharmacologic behavioral interventions for the management of cancer-related fatigue. Oncologist 2007, 12, 52–67. [Google Scholar] [CrossRef] [PubMed]
- Morten, Q.; Lis Adamsen, L.; Rorth, M.; Laursen, J.H.; Christensen, K.B.; Langer, S.W. The impact of a multidimensional exercise intervention on physical and functional capacity, anxiety, and depression in patients with advanced-stage lung cancer undergoing chemotherapy integrative cancer therapy. Integr. Cancer Ther. 2015, 14, 341–349. [Google Scholar]
- Balducci, S.; Iacobellis, G.; Parisi, L.; di Biase, N.; Calandriello, E.; Leonetti, F.; Fallucca, F. Exercise training can modify the natural history of diabetic peripheral neuropathy. J. Diabetes Complicat. 2006, 20, 216–223. [Google Scholar] [CrossRef] [PubMed]
- Stubblefield, M.D.; Burstein, H.J.; Burton, A.W.; Custodio, C.M.; Deng, G.E.; Ho, M.; Junck, L.; Morris, G.S.; Paice, J.A.; Tummala, S.; et al. NCCN task force report: Management of neuropathy in cancer. J. Natl. Compr. Cancer Netw. 2009, 7, 1–26. [Google Scholar]
- Park, J.S.; Kim, S.; Hoke, A. An exercise regimen prevents development paclitaxel induced peripheral neuropathy in a mouse model. J. Peripher. Nerv. Syst. 2015, 20, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Coller, H.A. Is cancer a metabolic disease? Am. J. Pathol. 2014, 184, 4–17. [Google Scholar] [CrossRef] [PubMed]
- McNeely, M.L.; Peddle, C.J.; Parilment, M.; Courneya, K.S. Cancer rehabilitation: Recommendations for integrating exercise programming in a clinical setting. Curr. Cancer Ther. Rev. 2006, 2, 251–260. [Google Scholar] [CrossRef]
- McTiernan, A. (Ed.) Cancer Prevention and Management through Exercise and Weight Control; Taylor and Francis: Boca Raton, FL, USA, 2006.
- Schmitz, K.; Courneya, K.; Matthews, C.; Demark-Wahnefried, W.; Galvao, D.A.; Pinto, B.M.; Irwin, M.L.; Wolin, K.Y.; Segal, R.J.; Lucia, A.; et al. ACSM’s Guidelines for Exercise Testing and Prescription, 9th ed.; Wolters Kluwer: Baltimore, MD, USA, 2014. [Google Scholar]
- Ware, J.E., Jr.; Sherbourne, C.D. The MOS 36-item short-form health survey (SF-36): I. Conceptual framework and item selection. Med. Care 1992, 30, 473–483. [Google Scholar] [CrossRef] [PubMed]
- Cella, D.F.; Tulsky, D.S.; Gray, G.; Sarafian, B.; Linn, E.; Bonomi, A.; Silberman, M.; Yellen, S.B.; Winicour, P.; Brannon, J.; et al. The functional assessment of cancer therapy scale: Development and validation of the general measure. J. Clin. Oncol. 1993, 11, 570–579. [Google Scholar] [PubMed]
- Jones, L.W.; Eves, N.D.; Mackey, J.R.; Peddle, C.J.; Haykowsky, M.; Joy, A.A.; Courenya, K.S.; Tankel, K.; Spratlin, J.; Reiman, T. Safety and feasibility of cardiopulmonary exercise testing in patients with advance cancer. Lung Cancer 2007, 55, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Van Waart, H.; Stuiverm, M.M.; van Harten, W.H.; Geleijn, E.; Kieffer, J.M.; Buffart, L.M.; de Maaker-Berkof, M.; Boven, E. Effect of low-intensity physical activity and moderate—To high-intensity physical exercise during adjuvant chemotherapy on physical fitness, fatigue, and chemotherapy completion rates: Results of the PACES randomized clinical trial. JCO 2014, 59, 1081–1091. [Google Scholar] [CrossRef] [PubMed]
- Lakoski, S.G.; Willis, B.L.; Barlow, C.E.; Leonard, D.; Gao, A.; Radford, N.B.; Farrell, S.W.; Douglas, P.S.; Berry, J.D.; DeFina, L.F.; et al. Midlife cardiorespiratory fitness, incident cancer, and survival after cancer in men: The cooper center longitudinal study. JAMA Oncol. 2015, 1, 231–237. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Health and Human Services. Physical Activity Guidelines for Americans; Department of Health and Human Services: Washington, DC, USA, 2008.
- Tanaka, H.; Monahan, K.G.; Seals, D.S. Age—Predicted maximal heart rate revisited. J. Am. Coll. Cardiol. 2001, 37, 153–156. [Google Scholar] [CrossRef]
- Scharhaq-Rosenberger, F.; Kuehl, R.; Klassen, O.; Schommer, K.; Schmidt, M.E.; Ulrich, C.M.; Wiskemann, J.; Steindorf, K. Exercise training intensity prescription in breast cancer survivors: Validity of current practice and specific recommendations. J. Cancer Survivorship 2015, 9, 612–619. [Google Scholar] [CrossRef] [PubMed]
- Klika, R.J.; Callahan, K.E.; Drum, S.N. Effects of individualized 12 weeks exercise training enhance aerobic capacity of cancer survivors. Phys. Sportsmed. 2009, 37, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Nelson, M.E.; Rejeski, W.J.; Blair, S.N.; Duncan, P.W.; Judge, J.O.; King, A.C.; Macera, C.A.; Castaneda-Sceppa, C. Physical activity and public health in older adults: Recommendation from the American College of Sports Medicine and the American Heart Association. Med. Sci. Sports Exerc. 2007, 8, 1435–1445. [Google Scholar] [CrossRef] [PubMed]
- Holmes, M.D.; Chen, Y.; Feskanich, D.; Kroenke, C.H.; Colditz, G.A. Physical activity and survival after breast cancer diagnosis. JAMA 2005, 293, 2479–2486. [Google Scholar] [CrossRef] [PubMed]
- McGill, S. Core training: Evidence translating to better performance and injury prevention. Strength Cond. J. 2010, 32, 33–46. [Google Scholar] [CrossRef]
- Eustruch, R.; Ros, E.; Salas-Salvado, J.; Covas, M.I.; Corella, D.; Aros, F.; Gomez-Gracia, E.; Ruiz-Gutierrez, V.; Fiol, M.; Lapetra, J.; et al. Primary prevention of cardiovascular disease with a Mediterranean diet. N. Engl. J. Med. 2013, 368, 1279–1290. [Google Scholar] [CrossRef] [PubMed]
- Shai, I.; Schwarzfuchs, D.; Henkin, Y.; Shahar, D.R.; Witkow, S.; Greenberg, I.; Golan, R.; Fraser, D.; Bolotin, A.; Vardi, H.; et al. Weight loss with a low-carbohydrate, Mediterranean, or low-fat diet. NEJM 2008, 359, 229–241. [Google Scholar] [CrossRef] [PubMed]
- Bjelakovic, G.; Nikolova, D.; Gluud, L.L.; Simonetti, R.G.; Gluud, C. Antioxidant supplements for prevention of mortality in healthy participants and patients with various diseases. Cochrane Database Syst. Rev. 2012. [Google Scholar] [CrossRef]

| Cancer Treatment | Physical Changes | Psychological and Behavioral Changes | ||
|---|---|---|---|---|
| Surgery Radiation Chemotherapy Immunotherapy Hormone Therapy Steroid Therapy | ↓ | pulmonary function | ↓ | decreased exercise/physical activity |
| ↓ | cardiac function | ↑ | physical symptoms and pain | |
| ↓ | muscle mass | ↑ | depression | |
| ↑ | fat mass | ↓ | cognitive function | |
| ↑ | body weight or body mass index | ↓ | quality of life (multiple domains) | |
| ↓ | decreased muscle strength/power | |||
| ↑ | inflammation | |||
| ↓ | immune function | |||
| ↓ | bone health | |||
| ↑ | trauma and scarring | |||
| ↑ | lymphedema | |||
| Clinical Cancer Complications and Acute Conditions | Contraindications to Exercise Testing and Training | Precautions Requiring Modification and/or Physician Approval |
|---|---|---|
| Factors Related to Cancer Treatment | No exercise on days of intravenous chemotherapy (recommendation changing) | Caution if on treatments that affect the lung and/or heart: recommend medically supervised exercise testing and training |
| No exercise before blood draw | Mouth sores/ulcerations: avoid mouthpiece for maximal testing: use face mask | |
| Severe tissue reaction to radiation therapy | Lymphedema: wear appropriate compression garments | |
| Hematologic | Platelet Count < 50,000 | Platelets > 50,000–150,000: avoid tests or exercise (contact sports) that increase risk of bleeding |
| Hemoglobin level < 10.0 g/dL | White blood cells > 3000–4000: ensure proper sterilization of equipment | |
| Absolute Neutrophil Count < 0.5 × 109/L | Hemoglobin > 10 g/dL (11.5–13.5 g/dL): caution with maximal tests | |
| Avoid activities that may increase the risk of bacterial infection (swimming) | ||
| Musculoskeletal | Extreme fatigue/muscle weakness | Any pain or cramping: investigate |
| Bone, back or neck pain | Osteopenia: avoid high-impact exercise if risk of fracture | |
| Severe cachexia (loss of >35% premorbid weight) | Loss of muscle mass limits exercise to mild intensity | |
| Karnofsky performance status score <60%; Poor functional status: avoid exercise testing | Cachexia: multidisciplinary approach to exercise | |
| Systemic | Acute infections | May indicate systemic infection and should be investigated. Avoid high intensity exercise |
| Febril illness: fever > 100 F | Avoid exercise until asymptomatic for >48 h | |
| General Malaise | ||
| Gastrointestinal | Severe Nausea | Compromised fluid and/or food intake: recommend multidisciplinary approach/consultation with nutritionist |
| Dehydration | Ensure adequate nutrition with electrolyte drinks and water (avoid hyponatremia) | |
| Vomiting or diarrhea within 24–36 h | Avoid exercise | |
| Poor nutrition: inadequate fluid and/or intake | Avoid exercise | |
| Cardiovascular | Chest pain | Exercise is contraindicated (refer to physician) |
| Resting HR > 100 bpm or < 50 bpm | Caution: recommend medically supervised exercise testing and training | |
| Resting SBP > 145 mmHg and/or DBP > 95 mmHg | Caution with exercise | |
| Resting SBP < 85 mmHg | Caution with exercise | |
| Irregular HR | Exercise is contraindicated (refer to physician) | |
| Swelling of ankles | Lymphedema: wear appropriate compression garments | |
| Pulmonary | Dyspnea | Mild to moderate dyspnea: avoid maximal tests |
| Cough, wheezing | Avoid activities that require significant oxygen transport (high intensity X) | |
| Chest pain increased by deep breath | Avoid exercise | |
| Neurologic | Ataxia/Dizziness/peripheral Sensory Neuropathy | Avoid activities that require significant balance and coordination (treadmill) |
| Significant decline in cognitive performance | Ensure patient is able to understand and follow instructions | |
| Disorientation | Use well supported positions for exercise | |
| Blurred vision | Avoid activities that require significant balance and coordination |
| Intensity of Exercise | MET Level | Heart Rate Reserve (HRR) | Rating of Perceived Exertion | Exercise Examples |
|---|---|---|---|---|
| Light | 1.1–3.0 | 35%–50% | 1–3 | Standing, light walking, washing dishes, doing laundry, cooking, light calisthenics |
| Moderate | 3.0–6.0 | 50%–70% | 4–6 | Walking at a moderate pace, bicycling 8–14.5 kph, water aerobics, weight training, dancing, recreational swimming, gardening and yard work, moderate home repair or housework |
| Vigorous | 6.0+ | 70%–85% * | 7–8 | Walking briskly (>8 kph), Jogging, running, bicycling, backpacking, aerobic dancing, vigorous calisthenics, circuit weight training, tennis, most competitive sports, lap swimming for fitness, heavy gardening and housework, occupational work with heavy loads |
| Date | Physical Exercise | Exercise Program |
|---|---|---|
| Monday | Aerobic Training | Continuous moderate intensity (40%–60% HRR) for 30 min |
| Tuesday | Resistance Training | Gym, free weights or body weight exercises with balance and agility work |
| Wednesday | Aerobic Training | Intermittent moderate-to-vigorous intensity (70%–85% HRR) for 30 min total exercise time |
| Thursday | Resistance Training | Gym, free weights or body weight exercises with balance and agility work |
| Friday | Aerobic Training | Continuous moderate intensity (40%–60% HRR) for 30 min |
| Saturday | Aerobic Training | Continuous moderate intensity (40%–60% HRR) for 30 min |
| Sunday | Day Off | - |
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stefani, L.; Galanti, G.; Klika, R. Clinical Implementation of Exercise Guidelines for Cancer Patients: Adaptation of ACSM’s Guidelines to the Italian Model. J. Funct. Morphol. Kinesiol. 2017, 2, 4. https://doi.org/10.3390/jfmk2010004
Stefani L, Galanti G, Klika R. Clinical Implementation of Exercise Guidelines for Cancer Patients: Adaptation of ACSM’s Guidelines to the Italian Model. Journal of Functional Morphology and Kinesiology. 2017; 2(1):4. https://doi.org/10.3390/jfmk2010004
Chicago/Turabian StyleStefani, Laura, Giorgio Galanti, and Riggs Klika. 2017. "Clinical Implementation of Exercise Guidelines for Cancer Patients: Adaptation of ACSM’s Guidelines to the Italian Model" Journal of Functional Morphology and Kinesiology 2, no. 1: 4. https://doi.org/10.3390/jfmk2010004
APA StyleStefani, L., Galanti, G., & Klika, R. (2017). Clinical Implementation of Exercise Guidelines for Cancer Patients: Adaptation of ACSM’s Guidelines to the Italian Model. Journal of Functional Morphology and Kinesiology, 2(1), 4. https://doi.org/10.3390/jfmk2010004








