Cyclotron Production and Purification of 83Sr as a 90Sr Substitute for Positron Emission Tomography (PET)
Abstract
1. Introduction
2. Materials and Methods
2.1. General
2.1.1. Target Disks
2.1.2. Ion Chromatography
2.1.3. Activity Determination and γ-Spectrometry
2.1.4. Chemicals and Materials
2.1.5. Irradiation Facility—Isochronous Cyclotron U-120M
2.2. Preparation of the RbCl Targets
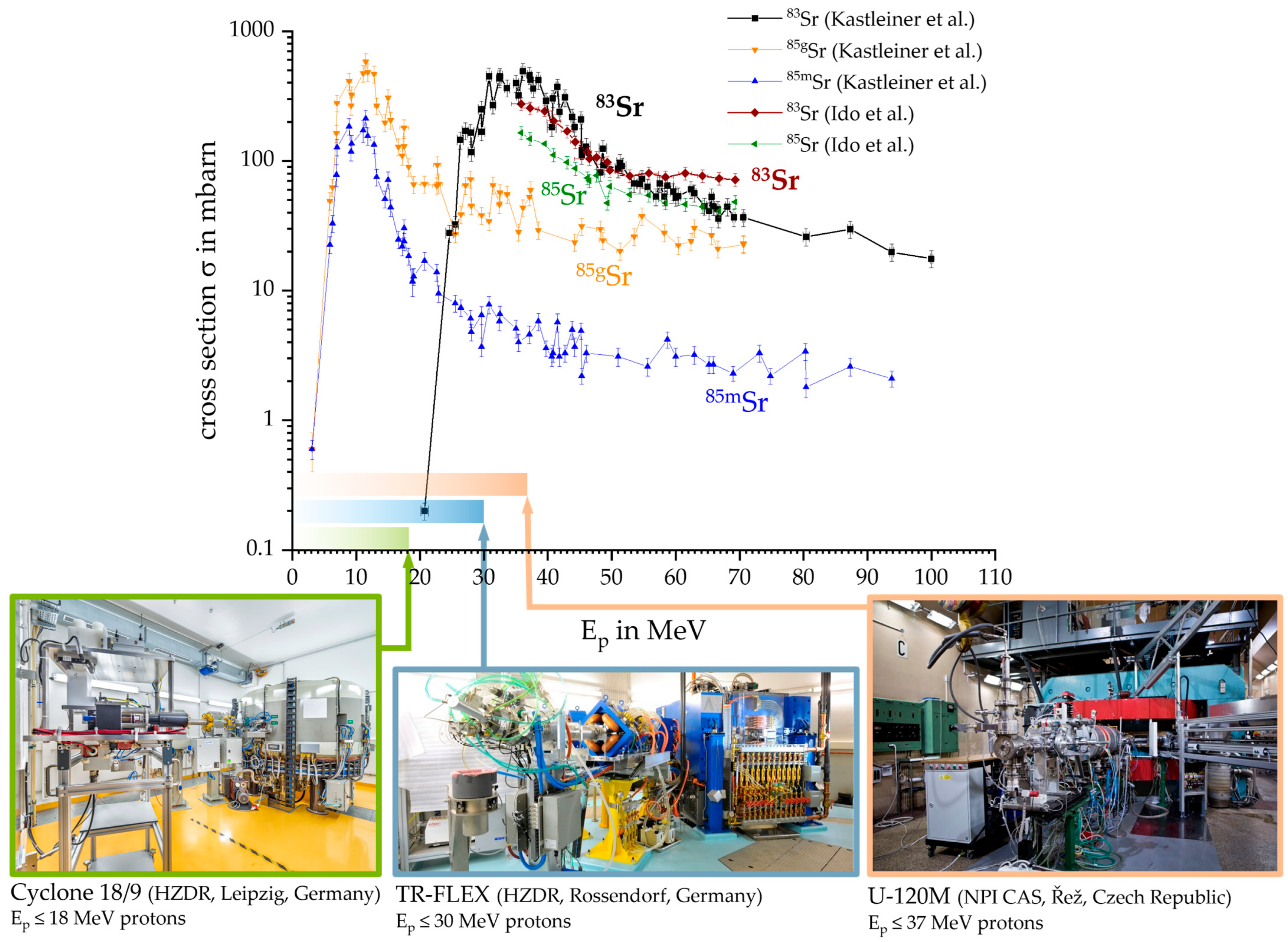
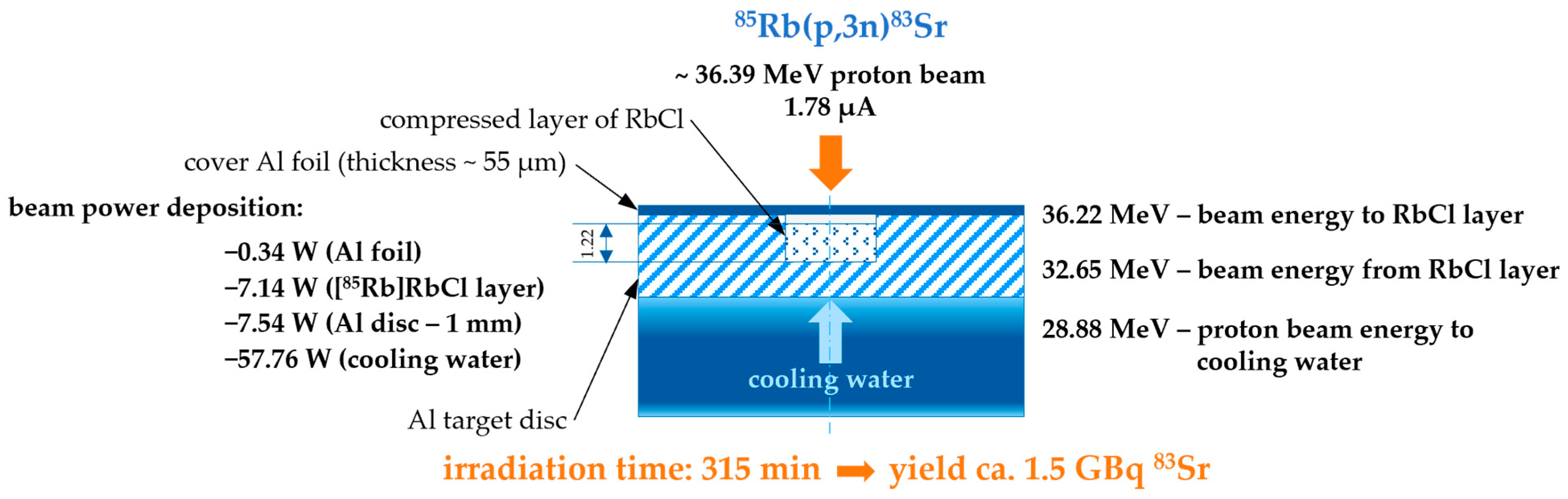
2.3. Cyclotron Production of 83Sr
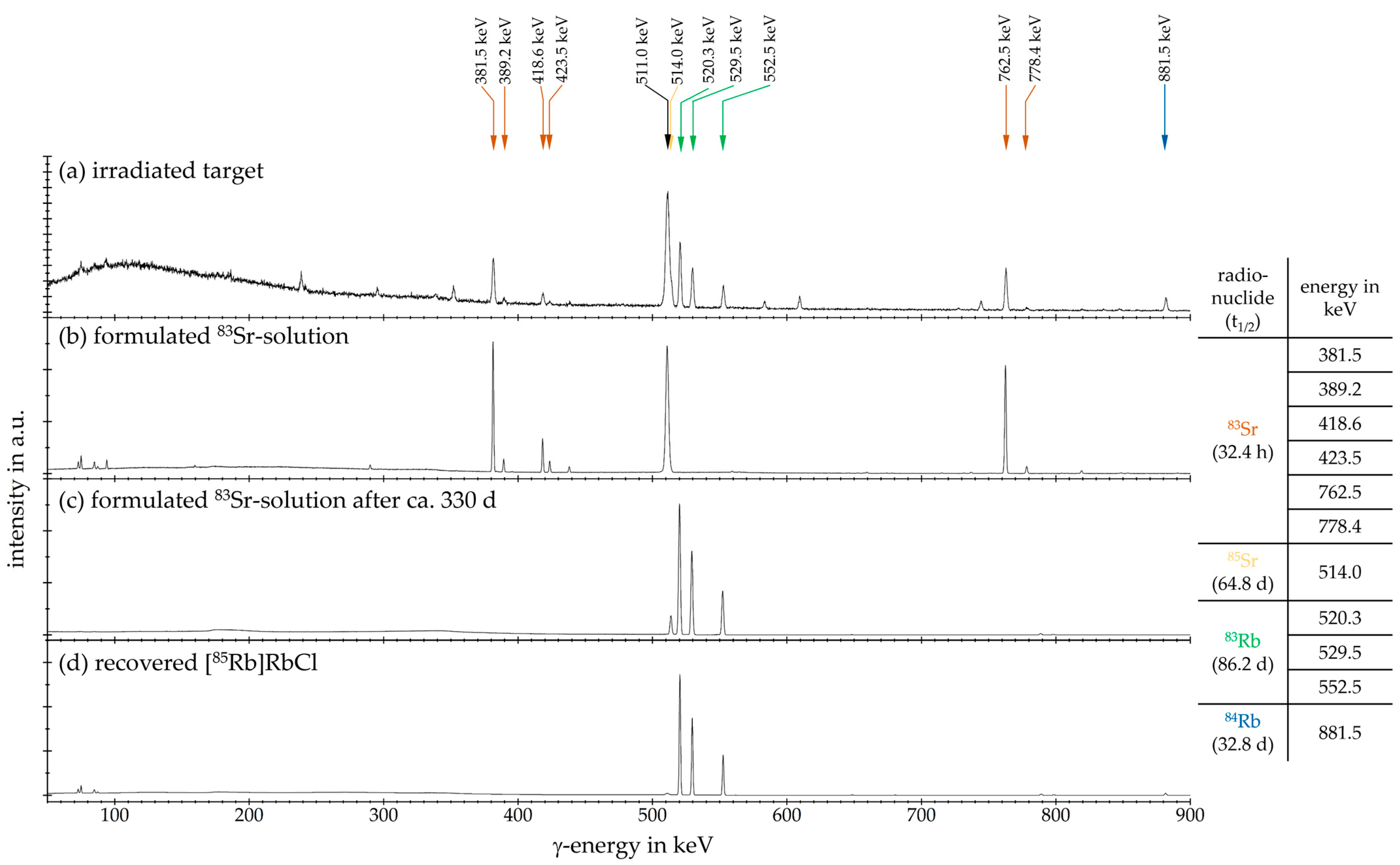
2.4. Purification of 83Sr
2.5. Target Material Recovery
2.5.1. Non-Radioactive Test with natRbNO3
2.5.2. Recovery of 85Rb Target Material
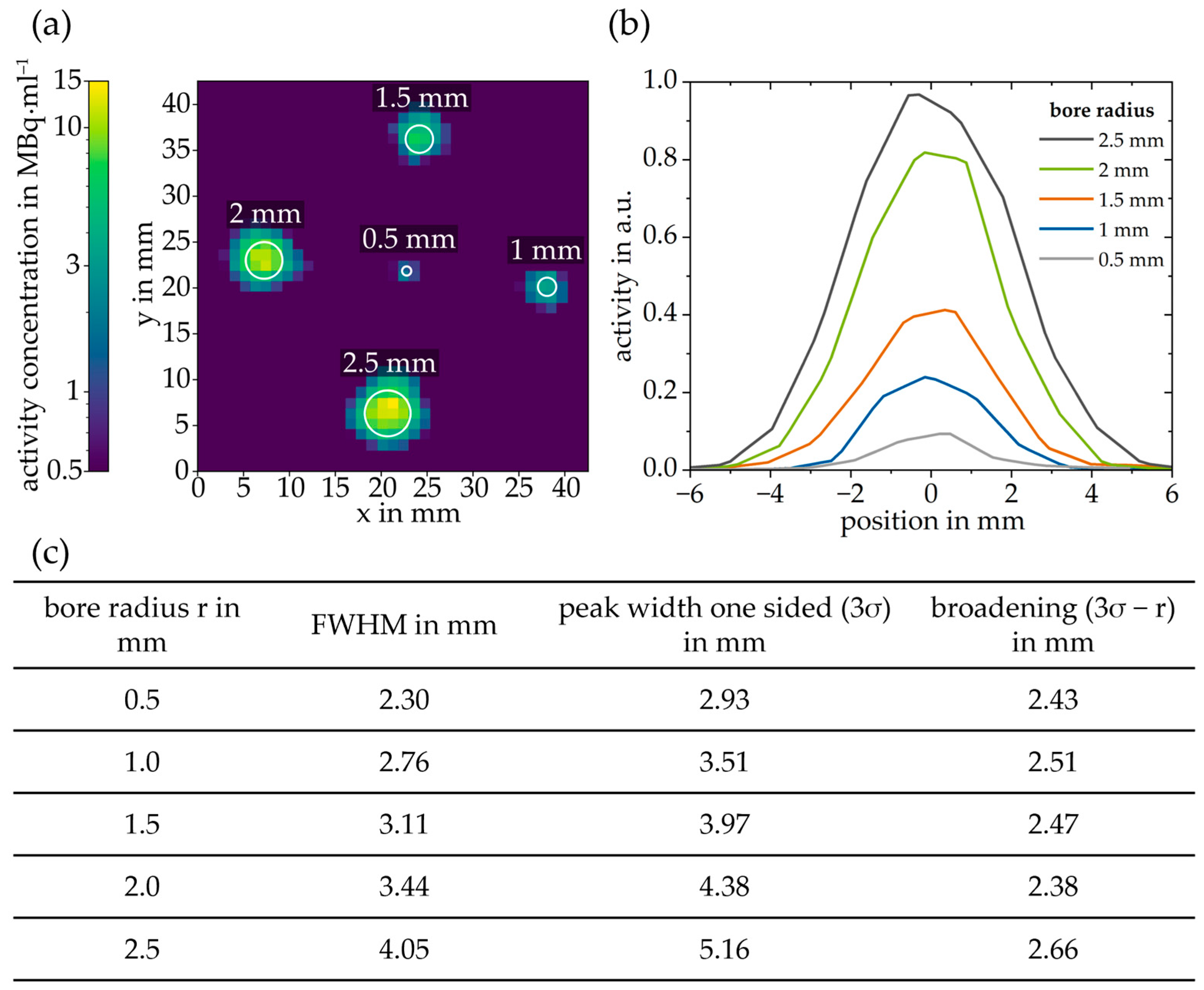
2.6. Positron Emission Tomography—Phantom
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Robinson, C.; Shaw, S.; Lloyd, J.R.; Graham, J.; Morris, K. Phosphate (Bio)mineralization Remediation of 90Sr-Contaminated Groundwaters. ACS ES&T Water 2023, 3, 3223–3234. [Google Scholar] [CrossRef]
- Zhou, Z.; Ren, H.; Zhou, L.; Wang, P.; Lou, X.; Zou, H.; Cao, Y. Recent Development on Determination of Low-Level 90Sr in Environmental and Biological Samples: A Review. Molecules 2023, 28, 90. [Google Scholar] [CrossRef]
- Tamma, A.A.; Lejcuś, K.; Fiałkiewicz, W.; Marczak, D. Advancing Phytoremediation: A Review of Soil Amendments for Heavy Metal Contamination Management. Sustainability 2025, 17, 5688. [Google Scholar] [CrossRef]
- Yan, A.; Wang, Y.; Tan, S.N.; Yusof, M.L.M.; Ghosh, S.; Chen, Z. Phytoremediation: A Promising Approach for Revegetation of Heavy Metal-Polluted Land. Front. Plant Sci. 2020, 11, 359. [Google Scholar] [CrossRef] [PubMed]
- Basu, S.; Mccutchan, E. Nuclear Data Sheets for A = 90. Nucl. Data Sheets 2020, 165, 1–329. [Google Scholar] [CrossRef]
- Singh, B. Nuclear Data Sheets for A = 80. Nucl. Data Sheets 2005, 105, 223–418. [Google Scholar] [CrossRef]
- Shamsuzzoha Basunia, M. Nuclear Data Sheets for A = 81. Nucl. Data Sheets 2025, 199, 271–612. [Google Scholar] [CrossRef]
- Tuli, J.; Browne, E. Nuclear Data Sheets for A = 82. Nucl. Data Sheets 2019, 157, 260–494. [Google Scholar] [CrossRef]
- McCutchan, E.A. Nuclear Data Sheets for A = 83. Nucl. Data Sheets 2015, 125, 201–394. [Google Scholar] [CrossRef]
- Abriola, D.; Bostan, M.; Erturk, S.; Fadil, M.; Galan, M.; Juutinen, S.; Kibédi, T.; Kondev, F.; Luca, A.; Negret, A.; et al. Nuclear Data Sheets for A = 84. Nucl. Data Sheets 2009, 110, 2815–2944. [Google Scholar] [CrossRef]
- Singh, B.; Chen, J. Nuclear Data Sheets for A = 85. Nucl. Data Sheets 2014, 116, 1–162. [Google Scholar] [CrossRef]
- Negret, A.; Singh, B. Nuclear Data Sheets for A = 86. Nucl. Data Sheets 2015, 124, 1–156. [Google Scholar] [CrossRef]
- Johnson, T.D.; Kulp, W.D. Nuclear Data Sheets for A = 87. Nucl. Data Sheets 2015, 129, 1–190. [Google Scholar] [CrossRef]
- McCutchan, E.A.; Sonzogni, A.A. Nuclear Data Sheets for A = 88. Nucl. Data Sheets 2014, 115, 135–304. [Google Scholar] [CrossRef]
- Singh, B. Nuclear Data Sheets for A = 89. Nucl. Data Sheets 2013, 114, 1–208. [Google Scholar] [CrossRef]
- Broda, R.; Hrynkiewicz, A.; Styczeń, J.; Waluś, W. The excited states of Kr and Rb isotopes in the 78 ≦ A ≦ 85 mass region. Nucl. Phys. A 1973, 216, 493–518. [Google Scholar] [CrossRef]
- Blessing, G.; Tárkányi, F.; Qaim, S. Production of 82mRb via the 82Kr(p,n)-process on highly enriched 82Kr: A remotely controlled compact system for irradiation, safe handling and recovery of the target gas and isolation of the radioactive product. Appl. Radiat. Isot. 1997, 48, 37–43. [Google Scholar] [CrossRef]
- Kastleiner, S.; Qaim, S.; Nortier, F.; Blessing, G.; van der Walt, T.; Coenen, H. Excitation functions of 85Rb(p,xn)85m,g,83,82,81Sr reactions up to 100 MeV: Integral tests of cross section data, comparison of production routes of 83Sr and thick target yield of 82Sr. Appl. Radiat. Isot. 2002, 56, 685–695. [Google Scholar] [CrossRef]
- Horiguchi, T.; Noma, H.; Yoshizawa, Y.; Takemi, H.; Hasai, H.; Kiso, Y. Excitation functions of proton induced nuclear reactions on 85Rb. Int. J. Appl. Radiat. Isot. 1980, 31, 141–151. [Google Scholar] [CrossRef]
- Kreller, M.; Pietzsch, H.J.; Walther, M.; Tietze, H.; Kaever, P.; Knieß, T.; Füchtner, F.; Steinbach, J.; Preusche, S. Introduction of the New Center for Radiopharmaceutical Cancer Research at Helmholtz-Zentrum Dresden-Rossendorf. Instruments 2019, 3, 9. [Google Scholar] [CrossRef]
- Ido, T.; Hermanne, A.; Ditrói, F.; Szűcs, Z.; Mahunka, I.; Tárkányi, F. Excitation functions of proton induced nuclear reactions on natRb from 30 to 70 MeV. Implication for the production of 82Sr and other medically important Rb and Sr radioisotopes. Nucl. Instrum. Methods Phys. Res. B 2002, 194, 369–388. [Google Scholar] [CrossRef]
- Matlocha, T.; Štursa, J.; Běhal, R.; Zach, V.; Čihák, M. Enhanced beam extraction system at the U-120M cyclotron. Nucl. Instrum. Methods Phys. Res. A 2024, 1064, 169335. [Google Scholar] [CrossRef]
- Künzelmann, A. Zyklotron an der Leipziger Forschungsstelle. 2026. Available online: https://www.hzdr.de/db/PicUser?pOid=54397&pWidth=1920 (accessed on 15 January 2026).
- CAS, NPI Cyklotron 01. 2026. Available online: https://www.ujf.cas.cz/export/sites/ujf/.content/galerie-obrazky/fotogalerie/Pro-verejnost-a-media/Urychlovac-cyklotron/_X5A1745_LOW.jpg (accessed on 15 January 2026).
- Mansel, A.; Gruhne, S.; Franke, K.; Fischer, S. Production of 85Sr at a 18 MeV-cyclotron and purification for geochemical investigations. Appl. Radiat. Isot. 2014, 92, 22–24. [Google Scholar] [CrossRef]
- Schöngart, J.; Lindemann, M.; Klotzsche, M.; Franke, K.; Fischer, C. Quantitative tomography of contaminant phytomobilization: β+ emitters 83Sr and 86Y as tracers of fission-product analog mobility. J. Hazard. Mater. Adv. 2026, 21, 100952. [Google Scholar] [CrossRef]
- Berger, M.J.; Hubbell, J.H.; Seltzer, S.M.; Chang, J.; Coursey, J.S.; Sukumar, R.; Zucker, D.S.; Olsen, K. XCOM: Photon Cross Sections Database—NIST Standard Reference Database 8 (XGAM); National Institute of Standards and Technology: Gaithersburg, MD, USA, 2010. [CrossRef]
- Roldan, P.S.; Chereul, E.; Dietzel, O.; Magnier, L.; Pautrot, C.; Rbah, L.; Sappey-Marinier, D.; Wagner, A.; Zimmer, L.; Janier, M.; et al. Raytest ClearPET™, a new generation small animal PET scanner. Nucl. Instrum. Methods Phys. Res. A 2007, 571, 498–501. [Google Scholar] [CrossRef]
- Thielemans, K.; Tsoumpas, C.; Mustafovic, S.; Beisel, T.; Aguiar, P.; Dikaios, N.; Jacobson, M.W. STIR: Software for tomographic image reconstruction release 2. Phys. Med. Biol. 2012, 57, 867. [Google Scholar] [CrossRef]
- Ziegler, J.F.; Ziegler, M.D.; Biersack, J.P. SRIM—The Stopping and Range of Ions in Matter - SRIM2008 Code. Available online: http://www.srim.org/ (accessed on 15 January 2026).
- Horwitz, E.P.; Chiarizia, R.; Dietz, M.L. A novel strontium-selective extraction chromatoghraphic resin. Solvent Extr. Ion Exch. 1992, 10, 313–336. [Google Scholar] [CrossRef]





| Isotope | Half-Life t1/2 | Decay Modes (β+-Intensity) |
|---|---|---|
| 80Sr | 106.3 min | EC, β+ (9.2%) |
| 81Sr | 22.3 min | EC, β+ (29%) |
| 82Sr | 25.3 d | EC |
| 83Sr | 32.4 h | EC, β+ (12%) |
| 84Sr | stable | – |
| 85Sr | 64.8 d | EC |
| 86Sr | stable | – |
| 87Sr | stable | – |
| 88Sr | stable | – |
| 89Sr | 50.6 d | β− |
| 90Sr | 28.9 a | β− |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Lindemann, M.; Schöngart, J.; Štursa, J.; Franke, K. Cyclotron Production and Purification of 83Sr as a 90Sr Substitute for Positron Emission Tomography (PET). Instruments 2026, 10, 20. https://doi.org/10.3390/instruments10020020
Lindemann M, Schöngart J, Štursa J, Franke K. Cyclotron Production and Purification of 83Sr as a 90Sr Substitute for Positron Emission Tomography (PET). Instruments. 2026; 10(2):20. https://doi.org/10.3390/instruments10020020
Chicago/Turabian StyleLindemann, Marcel, Jann Schöngart, Jan Štursa, and Karsten Franke. 2026. "Cyclotron Production and Purification of 83Sr as a 90Sr Substitute for Positron Emission Tomography (PET)" Instruments 10, no. 2: 20. https://doi.org/10.3390/instruments10020020
APA StyleLindemann, M., Schöngart, J., Štursa, J., & Franke, K. (2026). Cyclotron Production and Purification of 83Sr as a 90Sr Substitute for Positron Emission Tomography (PET). Instruments, 10(2), 20. https://doi.org/10.3390/instruments10020020






