Using In-Situ Laboratory and Synchrotron-Based X-ray Diffraction for Lithium-Ion Batteries Characterization: A Review on Recent Developments
Abstract
1. Introduction
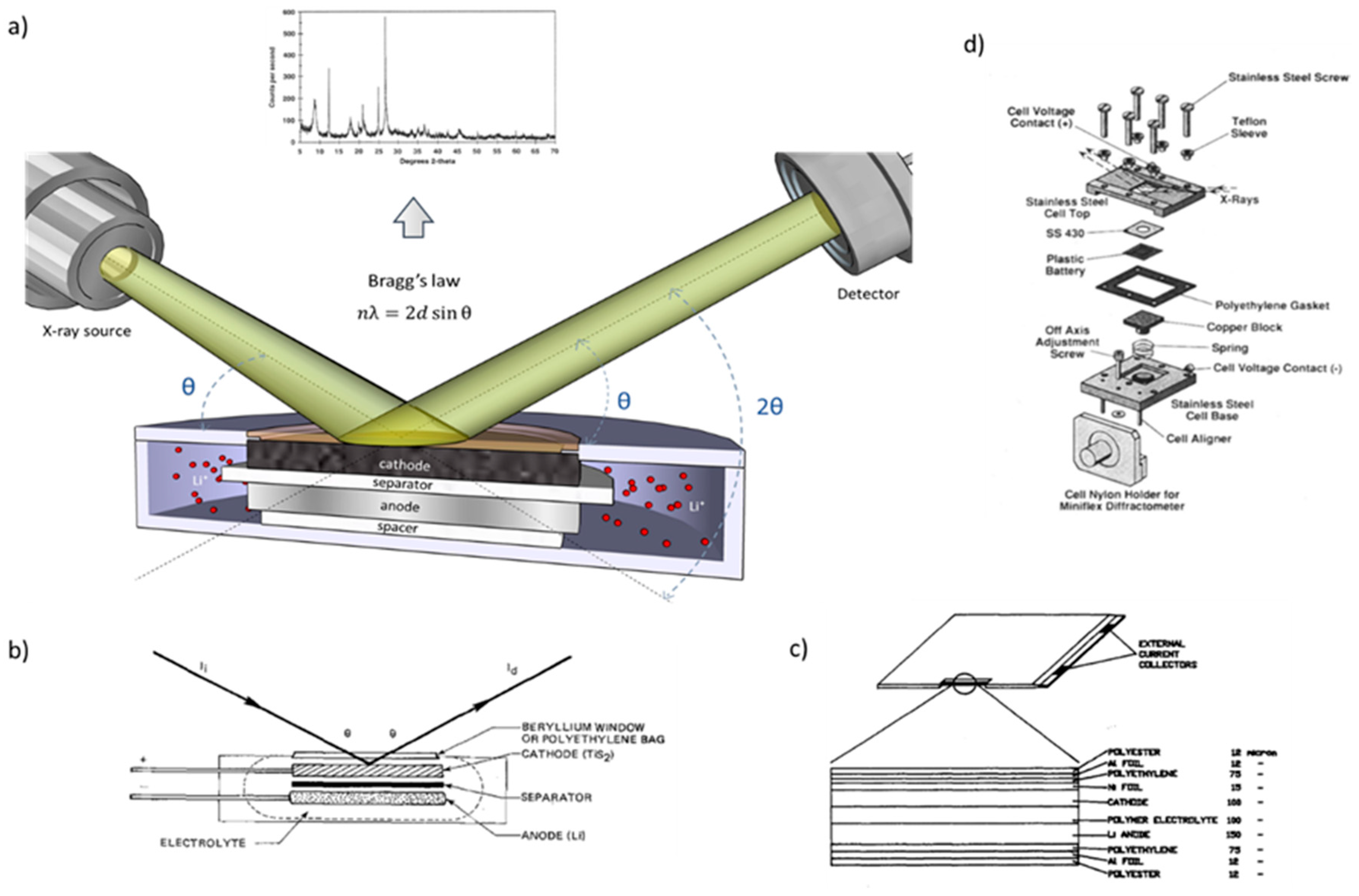
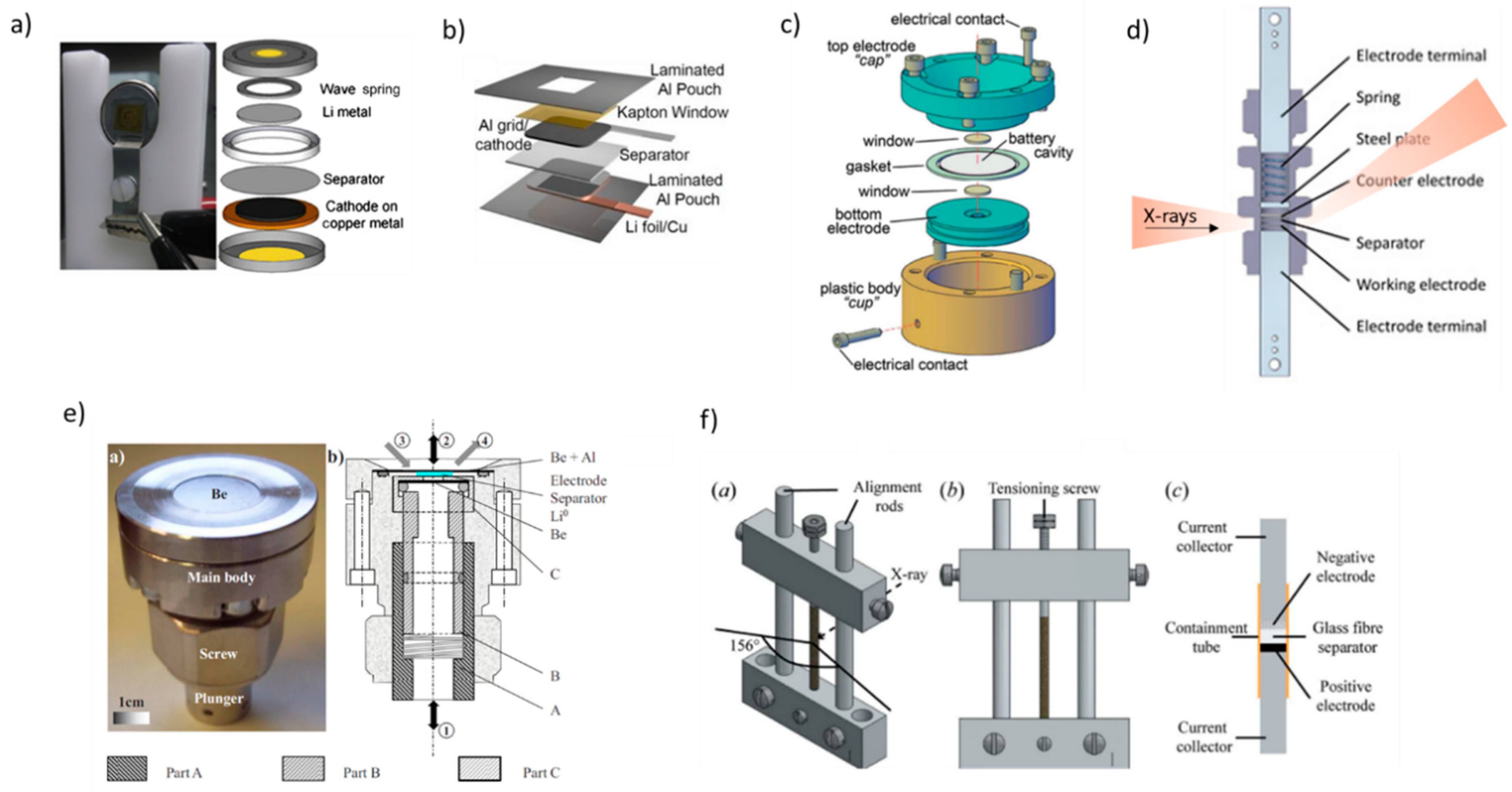
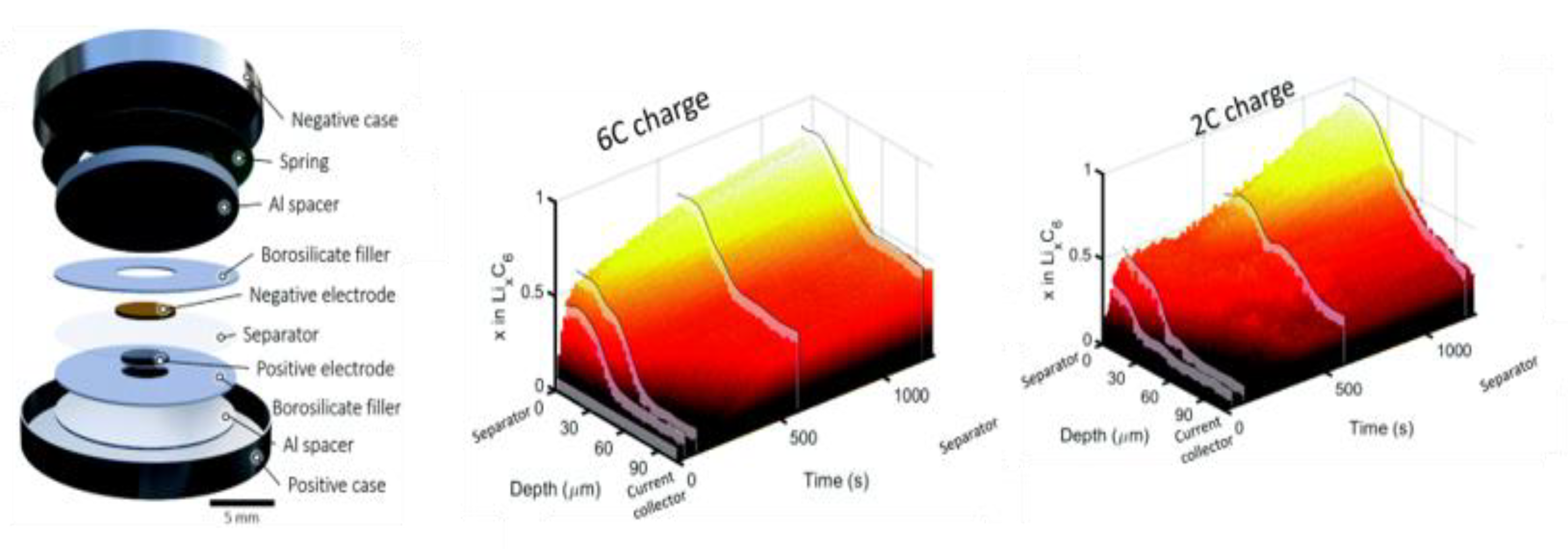
2. Design of In Situ Cells for XRD Analysis
3. Laboratory Diffractometer vs. Synchrotron XRD
4. Recent Case Studies
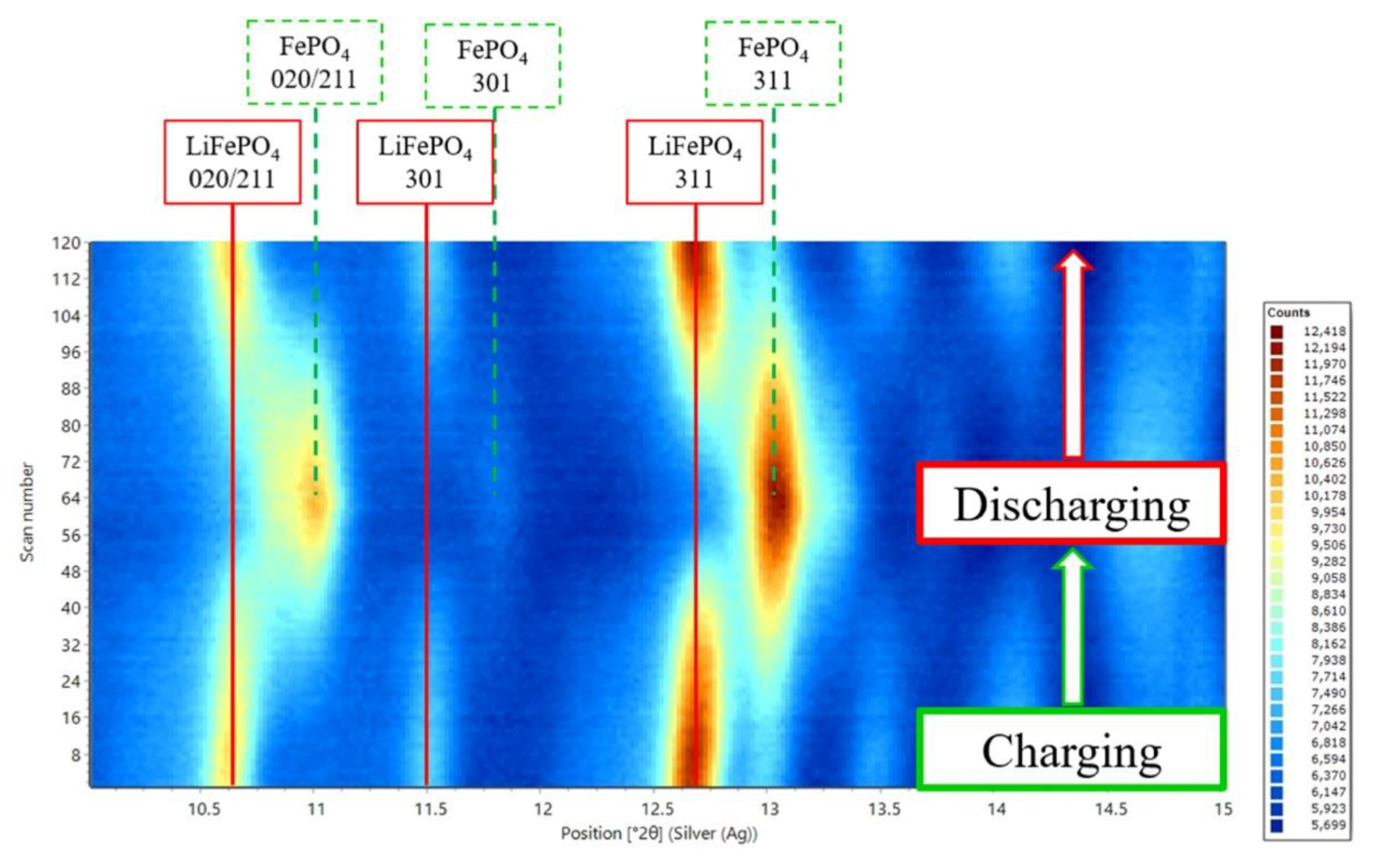
4.1. Long Duration Experiments Based at the I11 Beamline at Diamond Light Source
4.2. Diffraction Studies for Structure Determination
4.3. Bragg Coherent Diffraction Imaging
5. XRD Coupled with Other Techniques
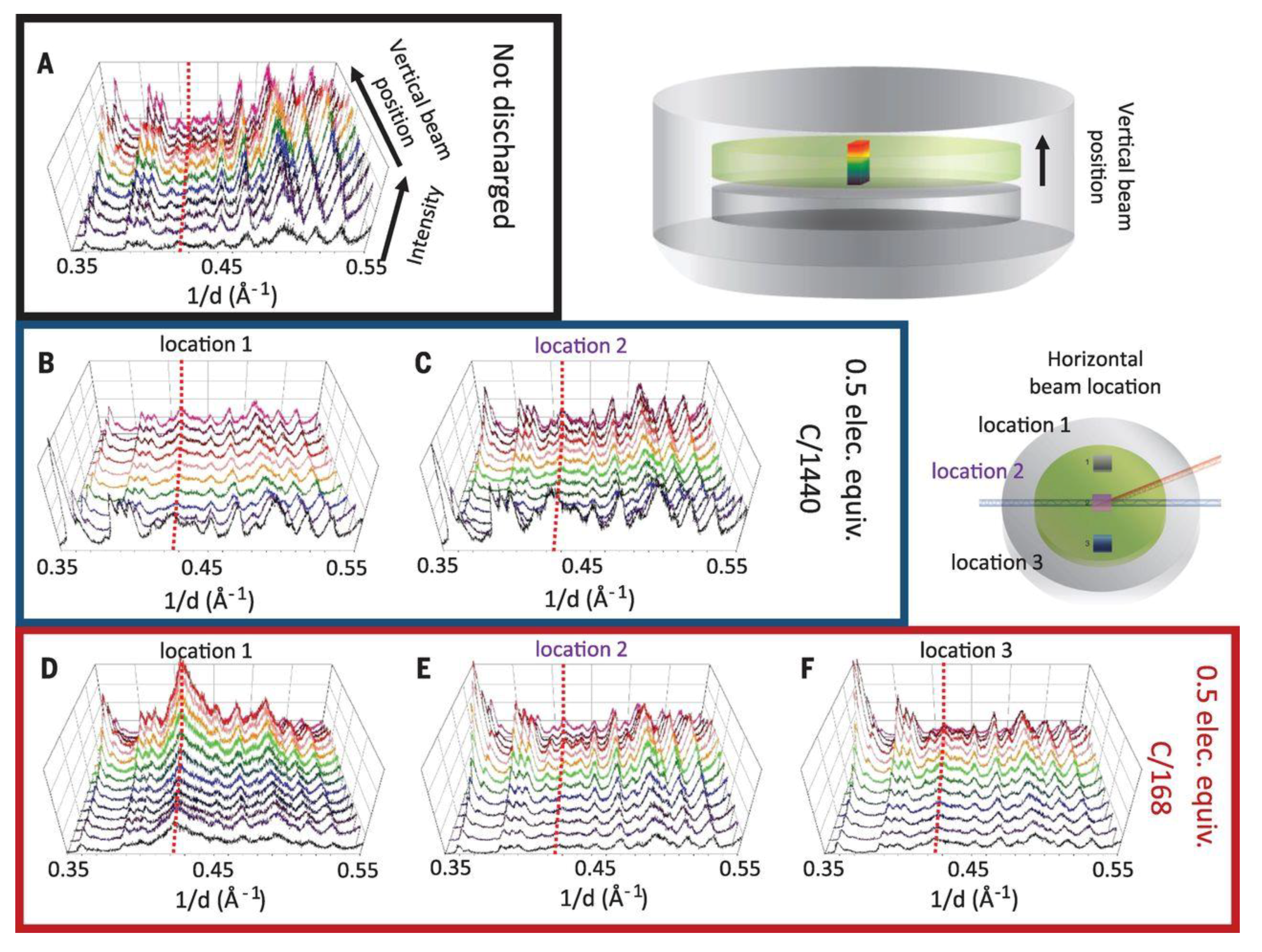
6. XRD-CT
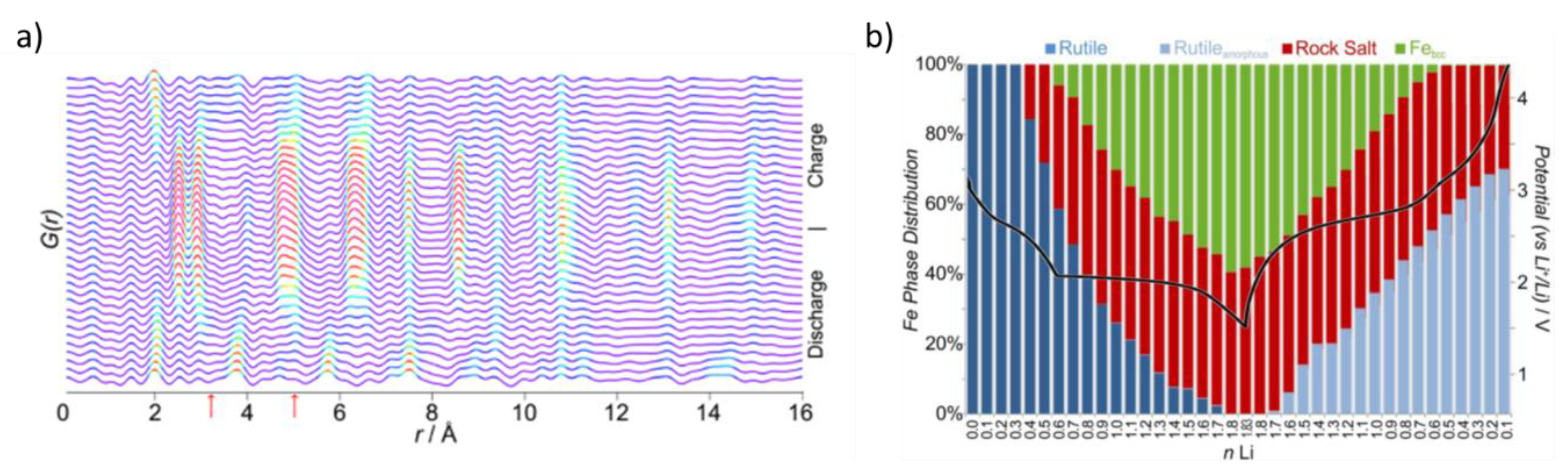
7. XRD and Pair Distribution Function
8. Conclusions and Perspective
Author Contributions
Funding
Conflicts of Interest
References
- Lithium Battery Market to Experience Rapid Growth. Available online: https://environment-analyst.com/uk/79774/lithium-battery-market-to-experience-rapid-growth (accessed on 18 June 2020).
- Armand, M.; Touzain, P. Graphite intercalation compounds as cathode materials. Mater. Sci. Eng. 1977, 31, 319–329. [Google Scholar] [CrossRef]
- Reddy, M.V.; Mauger, A.; Julien, C.M.; Paolella, A.; Zaghib, K. Brief history of early lithium-battery development. Materials 2020, 13, 1884. [Google Scholar] [CrossRef] [PubMed]
- Pender, J.P.; Jha, G.; Youn, D.H.; Ziegler, J.M.; Andoni, I.; Choi, E.J.; Heller, A.; Dunn, B.S.; Weiss, P.S.; Penner, R.M.; et al. Electrode Degradation in Lithium-Ion Batteries. ACS Nano 2020, 14, 1243–1295. [Google Scholar] [CrossRef] [PubMed]
- Müller, S.; Pietsch, P.; Brandt, B.E.; Baade, P.; De Andrade, V.; De Carlo, F.; Wood, V. Quantification and modeling of mechanical degradation in lithium-ion batteries based on nanoscale imaging. Nat. Commun. 2018, 9, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Oh, W.; Park, H.; Jin, B.S.; Thangavel, R.; Yoon, W.S. Understanding the structural phase transitions in lithium vanadium phosphate cathodes for lithium-ion batteries. J. Mater. Chem. A 2020, 8, 10331–10336. [Google Scholar] [CrossRef]
- Li, D.; Zhou, H. Two-phase transition of Li-intercalation compounds in Li-ion batteries. Mater. Today 2014, 17, 451–463. [Google Scholar] [CrossRef]
- Gauthier, M.; Carney, T.J.; Grimaud, A.; Giordano, L.; Pour, N.; Chang, H.H.; Fenning, D.P.; Lux, S.F.; Paschos, O.; Bauer, C.; et al. Electrode-Electrolyte Interface in Li-Ion Batteries: Current Understanding and New Insights. J. Phys. Chem. Lett. 2015, 6, 4653–4672. [Google Scholar] [CrossRef]
- Lukashuk, L.; Foettinger, K. In situ and operando spectroscopy: A powerful approach towards understanding catalysts. Johns. Matthey Technol. Rev. 2018, 62, 316–331. [Google Scholar] [CrossRef]
- Bañares, M.A. In situ to operando spectroscopy: From proof of concept to industrial application. Top. Catal. 2009, 52, 1301–1302. [Google Scholar] [CrossRef]
- Morcrette, M.; Chabre, Y.; Vaughan, G.; Amatucci, G.; Leriche, J.-B.; Patoux, S.; Masquelier, C.; Tarascon, J.-M. In situ X-ray diffraction techniques as a powerful tool to study battery electrode materials. Electrochim. Acta 2002, 47, 3137–3149. [Google Scholar] [CrossRef]
- Borkiewicz, O.J.; Wiaderek, K.M.; Chupas, P.J.; Chapman, K.W. Best practices for operando battery experiments: Influences of X-ray experiment design on observed electrochemical reactivity. J. Phys. Chem. Lett. 2015, 6, 2081–2085. [Google Scholar] [CrossRef] [PubMed]
- Brant, W.R.; Li, D.; Gu, Q.; Schmid, S. Comparative analysis of ex-situ and operando X-ray diffraction experiments for lithium insertion materials. J. Power Sources 2016, 302, 126–134. [Google Scholar] [CrossRef]
- McBreen, J. The application of synchrotron techniques to the study of lithium-ion batteries. J. Solid State Electrochem. 2009, 13, 1051–1061. [Google Scholar] [CrossRef]
- Lin, F.; Liu, Y.; Yu, X.; Cheng, L.; Singer, A.; Shpyrko, O.G.; Xin, H.L.; Tamura, N.; Tian, C.; Weng, T.C.; et al. Synchrotron X-ray Analytical Techniques for Studying Materials Electrochemistry in Rechargeable Batteries. Chem. Rev. 2017, 117, 13123–13186. [Google Scholar] [CrossRef] [PubMed]
- Bak, S.M.; Shadike, Z.; Lin, R.; Yu, X.; Yang, X.Q. In situ/operando synchrotron-based X-ray techniques for lithium-ion battery research. NPG Asia Mater. 2018, 10, 563–580. [Google Scholar] [CrossRef]
- Doeff, M.M.; Chen, G.; Cabana, J.; Richardson, T.J.; Mehta, A.; Shirpour, M.; Duncan, H.; Kim, C.; Kam, K.C.; Conry, T. Characterization of electrode materials for lithium ion and sodium ion batteries using synchrotron radiation techniques. J. Vis. Exp. 2013, 1–9. [Google Scholar] [CrossRef]
- Balasubramanian, M.; Sun, X.; Yang, X.Q.; Mcbreen, J. In situ X-ray diffraction and X-ray absorption studies.pdf. J. Power Sources 2001, 92, 1–8. [Google Scholar] [CrossRef]
- Pramudita, J.C.; Aughterson, R.; Dose, W.M.; Donne, S.W.; Brand, H.E.A.; Sharma, N. Using in situ synchrotron x-ray diffraction to study lithium- and sodium-ion batteries: A case study with an unconventional battery electrode (Gd2TiO5). J. Mater. Res. 2015, 30, 381–389. [Google Scholar] [CrossRef]
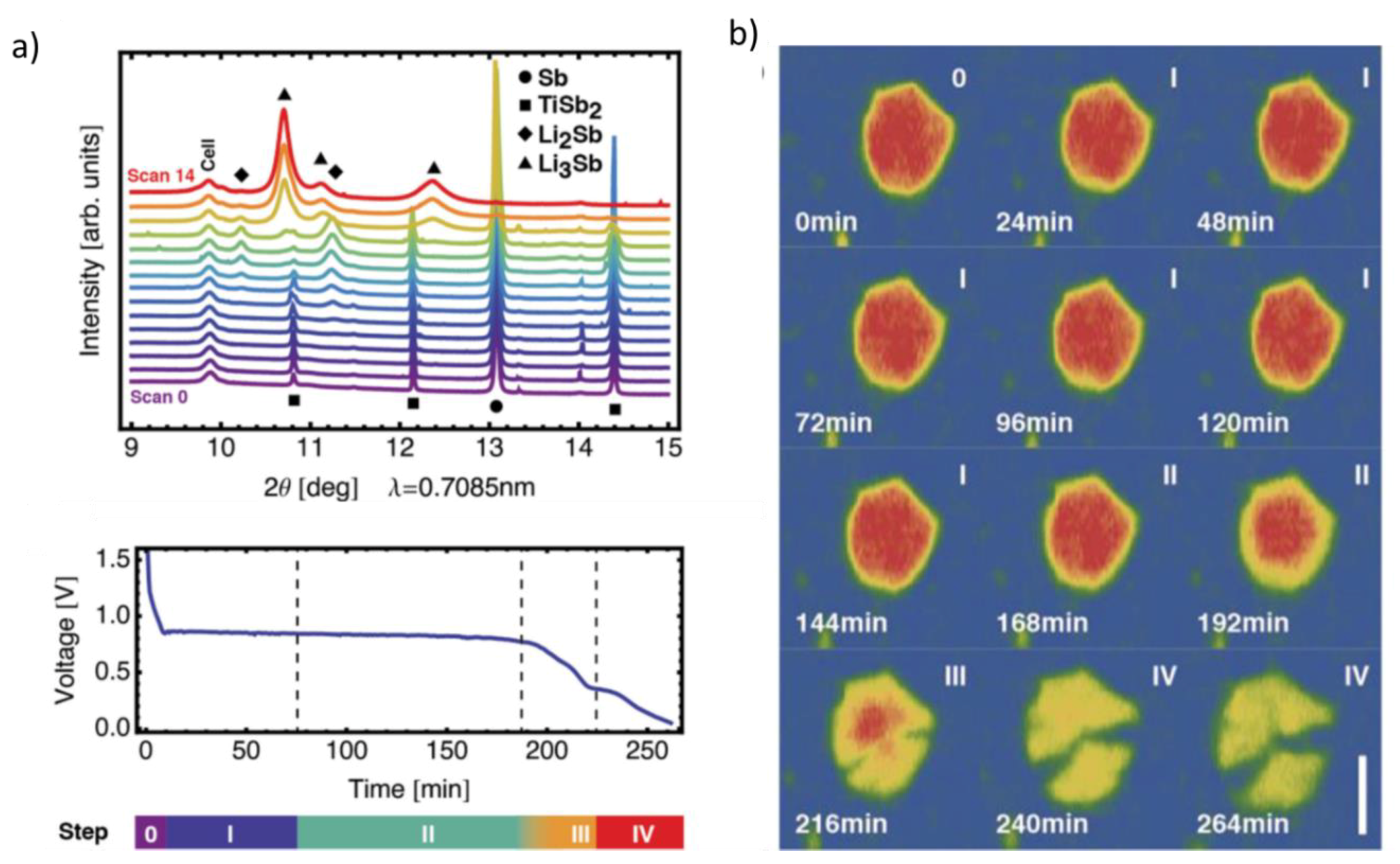
- Hu, C.W.; Chou, J.P.; Hou, S.C.; Hu, A.; Su, Y.F.; Chen, T.Y.; Liew, W.K.; Liao, Y.F.; Huang, J.L.; Chen, J.M.; et al. Cyclability evaluation on Si based Negative Electrode in Lithium ion Battery by Graphite Phase Evolution: An operando X-ray diffraction study. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef]
- Cheng, X.; Pecht, M. In situ stress measurement techniques on li-ion battery electrodes: A review. Energies 2017, 10, 591. [Google Scholar] [CrossRef]
- Koerver, R.; Zhang, W.; De Biasi, L.; Schweidler, S.; Kondrakov, A.O.; Kolling, S.; Brezesinski, T.; Hartmann, P.; Zeier, W.G.; Janek, J. Chemo-mechanical expansion of lithium electrode materials-on the route to mechanically optimized all-solid-state batteries. Energy Environ. Sci. 2018, 11, 2142–2158. [Google Scholar] [CrossRef]
- Fan, C.; Zhao, Z. Synchrotron Radiation in Materials Science: Light Sources, Techniques, and Applications, 1st ed.; John Wiley & Sons: Hoboken, NJ, USA, 2018. [Google Scholar]
- Mittemeijer, E.J.; Welzel, U. Modern Diffraction Methods; John Wiley & Sons: Hoboken, NJ, USA, 2013. [Google Scholar]
- Chianelli, R.R.; Scanlon, J.C.; Rao, B.M.L. Dynamic X—Ray Diffraction. J. Electrochem. Soc. 1978, 125, 1563. [Google Scholar] [CrossRef]
- Gustafsson, T.; Thomas, J.O.; Koksbang, R.; Farrington, G.C. The polymer battery as an environment for in situ X-ray diffraction studies of solid-state electrochemical processes. Electrochim. Acta 1992, 37, 1639–1643. [Google Scholar] [CrossRef]
- Amatucci, G.G. CoO2, The End Member of the LixCoO2 Solid Solution. J. Electrochem. Soc. 1996, 143, 1114. [Google Scholar] [CrossRef]
- Hartung, S.; Bucher, N.; Bucher, R.; Srinivasan, M. Note: Electrochemical cell for in operando X-ray diffraction measurements on a conventional X-ray diffractometer. Rev. Sci. Instrum. 2015, 86, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Reimers, J.N. Electrochemical and In Situ X-Ray Diffraction Studies of Lithium Intercalation in LixCoO2. J. Electrochem. Soc. 1992, 139, 2091. [Google Scholar] [CrossRef]
- Richard, M.N. A Cell for In Situ X-Ray Diffraction Based on Coin Cell Hardware and Bellcore Plastic Electrode Technology. J. Electrochem. Soc. 1997, 144, 554. [Google Scholar] [CrossRef]
- Roberts, G.A.; Stewart, K.D. Reflection-mode x-ray powder diffraction cell for in situ studies of electrochemical reactions. Rev. Sci. Instrum. 2004, 75, 1251–1254. [Google Scholar] [CrossRef]
- Sottmann, J.; Pralong, V.; Barrier, N.; Martin, C. An electrochemical cell for operando bench-top X-ray diffraction. J. Appl. Crystallogr. 2019, 52, 485–490. [Google Scholar] [CrossRef]
- Tripathi, A.M.; Su, W.N.; Hwang, B.J. In situ analytical techniques for battery interface analysis. Chem. Soc. Rev. 2018, 47, 736–751. [Google Scholar] [CrossRef]
- Ouvrard, G.; Zerrouki, M.; Soudan, P.; Lestriez, B.; Masquelier, C.; Morcrette, M.; Hamelet, S.; Belin, S.; Flank, A.M.; Baudelet, F. Heterogeneous behaviour of the lithium battery composite electrode LiFePO4. J. Power Sources 2013, 229, 16–21. [Google Scholar] [CrossRef]
- Tan, C.; Daemi, S.R.; Taiwo, O.O.; Heenan, T.M.M.; Brett, D.J.L.; Shearing, P.R. Evolution of electrochemical cell designs for in-situ and operando 3D characterization. Materials 2018, 11, 2157. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, M.I.; Van Campen, D.G.; Fields, J.D.; Yu, J.; Pool, V.L.; Parilla, P.A.; Ginley, D.S.; Van Hest, M.F.A.M.; Toney, M.F. Rapid thermal processing chamber for in-situ x-ray diffraction. Rev. Sci. Instrum. 2015, 86. [Google Scholar] [CrossRef]
- Jung, R.; Linsenmann, F.; Thomas, R.; Wandt, J.; Solchenbach, S.; Maglia, F.; Stinner, C.; Tromp, M.; Gasteiger, H.A. Nickel, Manganese, and Cobalt Dissolution from Ni-Rich NMC and Their Effects on NMC622-Graphite Cells. J. Electrochem. Soc. 2019, 166, A378–A389. [Google Scholar] [CrossRef]
- Yoon, W.S.; Grey, C.P.; Balasubramanian, M.; Yang, X.Q.; McBreen, J. In situ X-ray absorption spectroscopic study on LiNi0.5Mn0.5O2 cathode material during electrochemical cycling. Chem. Mater. 2003, 15, 3161–3169. [Google Scholar] [CrossRef]
- Liu, H.; Li, Z.; Grenier, A.; Kamm, G.E.; Yin, L.; Mattei, G.S.; Cosby, M.R.; Khalifah, P.G.; Chupas, P.J.; Chapman, K.W. Best practices for operando depth-resolving battery experiments. J. Appl. Crystallogr. 2020, 53, 133–139. [Google Scholar] [CrossRef]
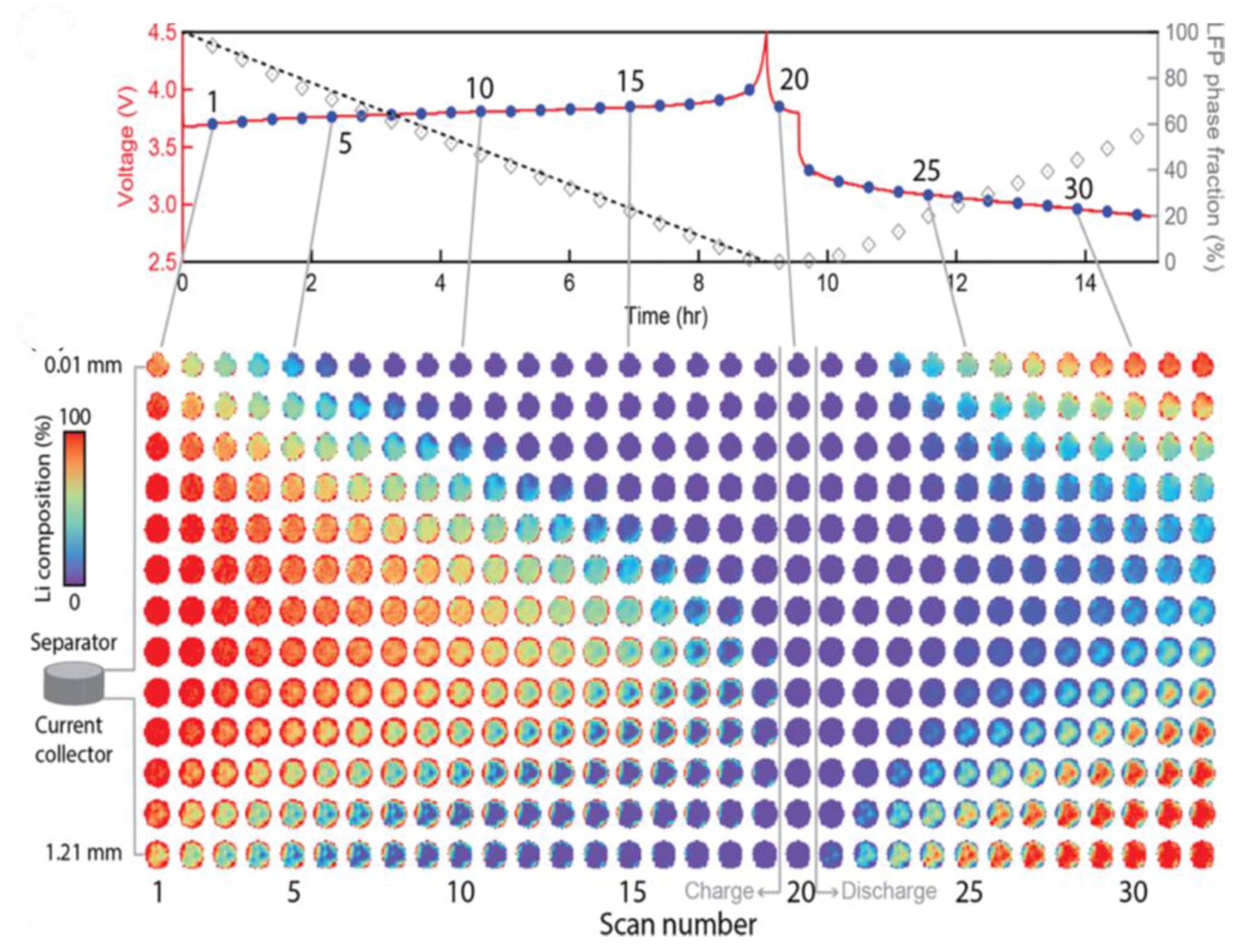
- Strobridge, F.C.; Orvananos, B.; Croft, M.; Yu, H.C.; Robert, R.; Liu, H.; Zhong, Z.; Connolley, T.; Drakopoulos, M.; Thornton, K.; et al. Mapping the inhomogeneous electrochemical reaction through porous LiFePO4-electrodes in a standard coin cell battery. Chem. Mater. 2015, 27, 2374–2386. [Google Scholar] [CrossRef]
- Ronci, F.; Scrosati, B.; Rossi Albertini, V.; Perfetti, P. In situ energy dispersive x-ray diffraction study of LiNi0.8Co0.2O2 cathode material for lithium batteries. J. Phys. Chem. B 2001, 105, 754–759. [Google Scholar] [CrossRef]
- Marschilok, A.C.; Bruck, A.M.; Abraham, A.; Stackhouse, C.A.; Takeuchi, K.J.; Takeuchi, E.S.; Croft, M.; Gallaway, J.W. Energy dispersive X-ray diffraction (EDXRD) for operando materials characterization within batteries. Phys. Chem. Chem. Phys. 2020, 22, 20972–20989. [Google Scholar] [CrossRef]
- Xu, Y.; Hu, E.; Zhang, K.; Wang, X.; Borzenets, V.; Sun, Z.; Pianetta, P.; Yu, X.; Liu, Y.; Yang, X.Q.; et al. In situ Visualization of State-of-Charge Heterogeneity within a LiCoO2 Particle that Evolves upon Cycling at Different Rates. ACS Energy Lett. 2017, 2, 1240–1245. [Google Scholar] [CrossRef]
- Holleck, G.L.; Driscoll, J.R. Transition metal sulfides as cathodes for secondary lithium batteries-II. titanium sulfides. Electrochim. Acta 1977, 22, 647–655. [Google Scholar] [CrossRef]
- Dahn, J.R.; Haering, R.R. Anomalous bragg peak widths in LixTiS2. Solid State Commun. 1981, 40, 245–248. [Google Scholar] [CrossRef]
- Ghanty, C.; Markovsky, B.; Erickson, E.M.; Talianker, M.; Haik, O.; Tal-Yossef, Y.; Mor, A.; Aurbach, D.; Lampert, J.; Volkov, A.; et al. Li+-Ion Extraction/Insertion of Ni-Rich Li1+x(NiyCozMnz)wO2 (0.005 < x < 0.03; y:z = 8:1, w ≈ 1) Electrodes: In Situ XRD and Raman Spectroscopy Study. ChemElectroChem 2015, 2, 1479–1486. [Google Scholar] [CrossRef]
- Yavuz, M.; Kiziltas-Yavuz, N.; Bhaskar, A.; Scheuermann, M.; Indris, S.; Fauth, F.; Knapp, M.; Ehrenberg, H. Influence of iron on the structural evolution of Lini0.4Fe0.2MN1.4O4 during electrochemical cycling investigated by in situ powder diffraction and spectroscopic methods. Z. Anorg. Allg. Chem. 2014, 640, 3118–3126. [Google Scholar] [CrossRef]
- Herklotz, M.; Scheiba, F.; Hinterstein, M.; Nikolowski, K.; Knapp, M.; Dippel, A.C.; Giebeler, L.; Eckert, J.; Ehrenberg, H. Advances in in situ powder diffraction of battery materials: A case study of the new beamline P02.1 at DESY, Hamburg. J. Appl. Crystallogr. 2013, 46, 1117–1127. [Google Scholar] [CrossRef]
- Schweidler, S.; De Biasi, L.; Schiele, A.; Hartmann, P.; Brezesinski, T.; Janek, J. Volume Changes of Graphite Anodes Revisited: A Combined Operando X-ray Diffraction and in Situ Pressure Analysis Study. J. Phys. Chem. C 2018, 122, 8829–8835. [Google Scholar] [CrossRef]
- De Biasi, L.; Kondrakov, A.O.; Geßwein, H.; Brezesinski, T.; Hartmann, P.; Janek, J. Between Scylla and Charybdis: Balancing among Structural Stability and Energy Density of Layered NCM Cathode Materials for Advanced Lithium-Ion Batteries. J. Phys. Chem. C 2017, 121, 26163–26171. [Google Scholar] [CrossRef]
- Borkiewicz, O.J.; Shyam, B.; Wiaderek, K.M.; Kurtz, C.; Chupas, P.J.; Chapman, K.W. The AMPIX electrochemical cell: A versatile apparatus for in situ X-ray scattering and spectroscopic measurements. J. Appl. Crystallogr. 2012, 45, 1261–1269. [Google Scholar] [CrossRef]
- Nikolowski, K.; Baehtz, G.; Bramnik, N.N.; Ehrenberg, H. A Swagelok-type in situ cell for battery investigations using synchrotron radiation. J. Appl. Crystallogr. 2005, 38, 851–853. [Google Scholar] [CrossRef]
- Leriche, J.B.; Hamelet, S.; Shu, J.; Morcrette, M.; Masquelier, C.; Ouvrard, G.; Zerrouki, M.; Soudan, P.; Belin, S.; Elkaïm, E.; et al. An Electrochemical Cell for Operando Study of Lithium Batteries Using Synchrotron Radiation. J. Electrochem. Soc. 2010, 157, A606. [Google Scholar] [CrossRef]
- Liu, H.; Allan, P.K.; Borkiewicz, O.J.; Kurtz, C.; Grey, C.P.; Chapman, K.W.; Chupas, P.J. A radially accessible tubular in situ X-ray cell for spatially resolved operando scattering and spectroscopic studies of electrochemical energy storage devices. J. Appl. Crystallogr. 2016, 49, 1665–1673. [Google Scholar] [CrossRef]
- Klein, D.; Xu, Y.; Schlögl, R.; Cap, S. Low Reversible Capacity of Nitridated Titanium Electrical Terminals. Batteries 2019, 5, 17. [Google Scholar] [CrossRef]
- Wilson, G.; Zilinskaite, S.; Unka, S.; Boston, R.; Reeves-McLaren, N. Establishing operando diffraction capability through the study of Li-ion (de) intercalation in LiFePO4. Energy Rep. 2020, 6, 174–179. [Google Scholar] [CrossRef]
- Yu, F.; Zhang, L.; Li, Y.; An, Y.; Zhu, M.; Dai, B. Mechanism studies of LiFePO4 cathode material: Lithiation/delithiation process, electrochemical modification and synthetic reaction. RSC Adv. 2014, 4, 54576–54602. [Google Scholar] [CrossRef]
- Quilty, C.D.; Bock, D.C.; Yan, S.; Takeuchi, K.J.; Takeuchi, E.S.; Marschilok, A.C. Probing Sources of Capacity Fade in LiNi0.6Mn0.2Co0.2O2 (NMC622): An Operando XRD Study of Li/NMC622 Batteries during Extended Cycling. J. Phys. Chem. C 2020, 124, 8119–8128. [Google Scholar] [CrossRef]
- Hulbert, S.L.; Williams, G.P. 1—Synchrotron Radiation Sources; Samson, J.A.R., Ederer, D.L., Eds.; Academic Press: Burlington, VT, USA, 2000; pp. 1–25. ISBN 978-0-12-617560-8. [Google Scholar]
- Beale, A.M.; Jacques, S.D.M.; Gibson, E.K.; Di Michiel, M. Progress towards five dimensional diffraction imaging of functional materials under process conditions. Coord. Chem. Rev. 2014, 277, 208–223. [Google Scholar] [CrossRef]
- Bianchini, M.; Fauth, F.; Brisset, N.; Weill, F.; Suard, E.; Masquelier, C.; Croguennec, L. Comprehensive investigation of the Na3V2(PO4)2F3-NaV2(PO4)2F3 system by operando high resolution synchrotron X-ray diffraction. Chem. Mater. 2015, 27, 3009–3020. [Google Scholar] [CrossRef]
- Bianchini, M.; Brisset, N.; Fauth, F.; Weill, F.; Elkaim, E.; Suard, E.; Masquelier, C.; Croguennec, L. Na3V2(PO4)2F3 revisited: A high-resolution diffraction study. Chem. Mater. 2014, 26, 4238–4247. [Google Scholar] [CrossRef]
- Withers, P.J. Depth capabilities of neutron and synchrotron diffraction strain measurement instruments. I. The maximum feasible path length. J. Appl. Crystallogr. 2004, 37, 596–606. [Google Scholar] [CrossRef]
- Young, B.T.; Heskett, D.R.; Woicik, J.C.; Lucht, B.L. X-ray-induced changes to passivation layers of lithium-ion battery electrodes. J. Spectrosc. 2018, 2018. [Google Scholar] [CrossRef]
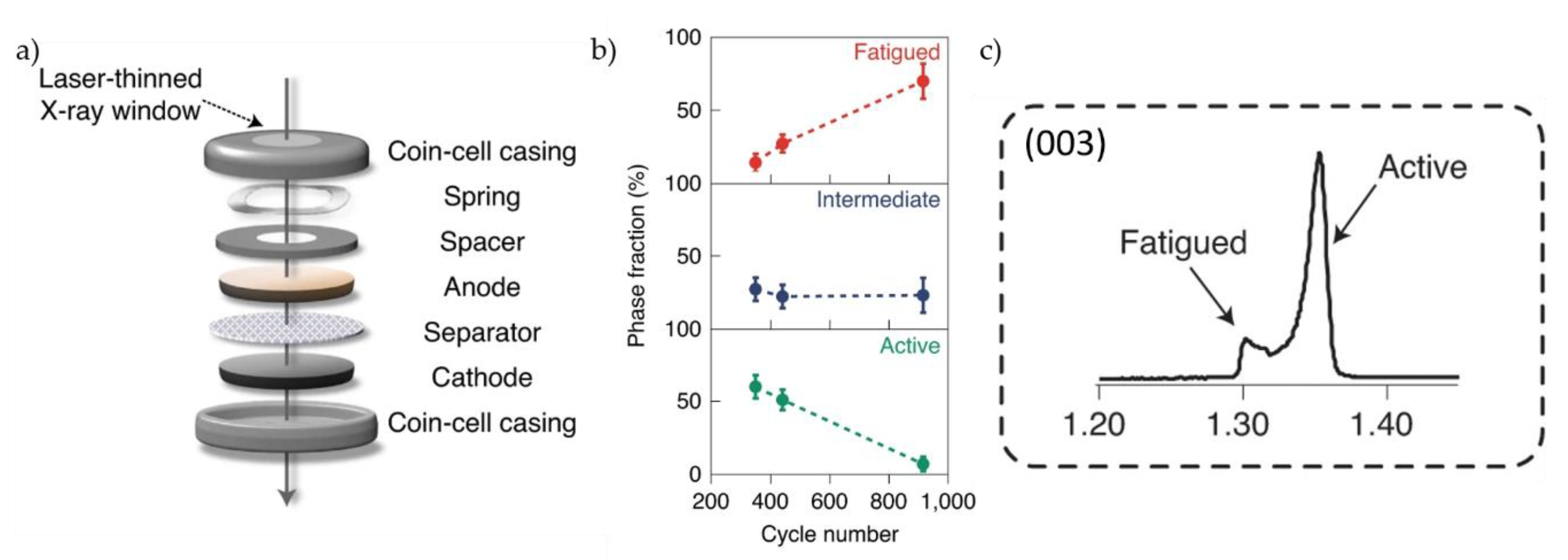
- Xu, C.; Märker, K.; Lee, J.; Mahadevegowda, A.; Reeves, P.J.; Day, S.J.; Groh, M.F.; Emge, S.P.; Ducati, C.; Layla Mehdi, B.; et al. Bulk fatigue induced by surface reconstruction in layered Ni-rich cathodes for Li-ion batteries. Nat. Mater. 2020. [Google Scholar] [CrossRef] [PubMed]
- Marker, K.; Reeves, P.J.; Xu, C.; Griffith, K.J.; Grey, C.P. Evolution of structure and lithium dynamics in LiNi0.8Mn0.1Co0.1O2 (NMC811) cathodes during electrochemical cycling. Chem. Mater. 2019, 2, 2545–2554. [Google Scholar] [CrossRef]
- Bartsch, T.; Kim, A.Y.; Strauss, F.; De Biasi, L.; Teo, J.H.; Janek, J.; Hartmann, P.; Brezesinski, T. Indirect state-of-charge determination of all-solid-state battery cells by X-ray diffraction. Chem. Commun. 2019, 55, 11223–11226. [Google Scholar] [CrossRef] [PubMed]
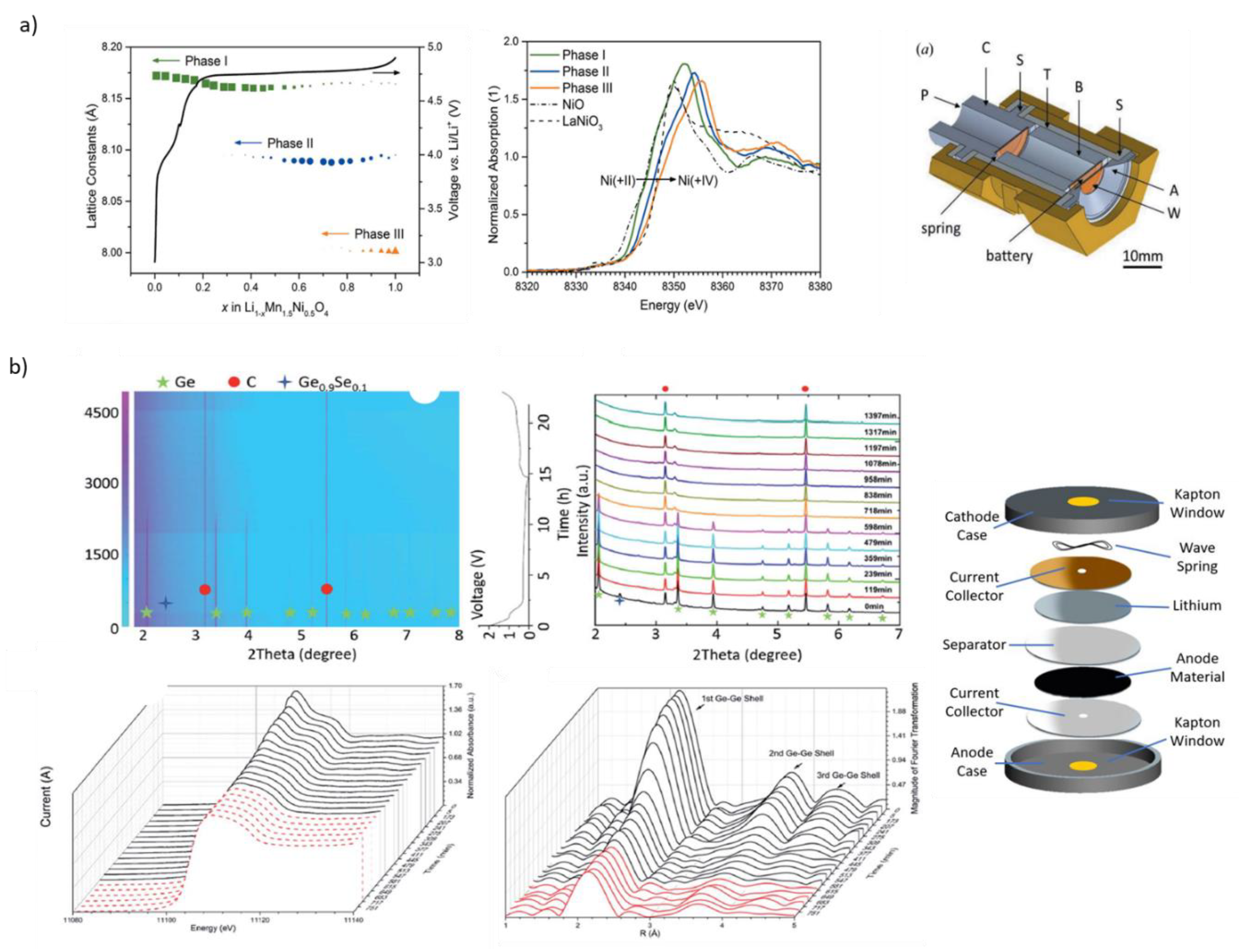
- Oh, G.; Hirayama, M.; Kwon, O.; Suzuki, K.; Kanno, R. Bulk-Type All Solid-State Batteries with 5 v Class LiNi0.5Mn1.5O4 Cathode and Li10GeP2S12 Solid Electrolyte. Chem. Mater. 2016, 28, 2634–2640. [Google Scholar] [CrossRef]
- Chattopadhyay, S.; Lipson, A.L.; Karmel, H.J.; Emery, J.D.; Fister, T.T.; Fenter, P.A.; Hersam, M.C.; Bedzyk, M.J. In situ X-ray study of the solid electrolyte interphase (SEI) formation on graphene as a model Li-ion battery anode. Chem. Mater. 2012, 24, 3038–3043. [Google Scholar] [CrossRef]
- Tippens, J.; Miers, J.C.; Afshar, A.; Lewis, J.A.; Cortes, F.J.Q.; Qiao, H.; Marchese, T.S.; Di Leo, C.V.; Saldana, C.; McDowell, M.T. Visualizing Chemomechanical Degradation of a Solid-State Battery Electrolyte. ACS Energy Lett. 2019, 4, 1475–1483. [Google Scholar] [CrossRef]
- Bianchini, M.; Fauth, F.; Suard, E.; Leriche, J.B.; Masquelier, C.; Croguennec, L. Spinel materials for Li-ion batteries: New insights obtained by operando neutron and synchrotron X-ray diffraction. Acta Crystallogr. Sect. B Struct. Sci. Cryst. Eng. Mater. 2015, 71, 688–701. [Google Scholar] [CrossRef]
- Lu, Z.; MacNeil, D.D.; Dahn, J.R. Layered cathode materials Li[NixLi(1/3−2x/3)Mn(2/3−x/3)]O2 for lithium-ion batteries. Electrochem. Solid-State Lett. 2001, 4, 3–7. [Google Scholar] [CrossRef]
- Bie, X.; Du, F.; Wang, Y.; Zhu, K.; Ehrenberg, H.; Nikolowski, K.; Wang, C.; Chen, G.; Wei, Y. Relationships between the crystal/interfacial properties and electrochemical performance of LiNi0.33Co0.33Mn0.33O2 in the voltage window of 2.5–4.6 v. Electrochim. Acta 2013, 97, 357–363. [Google Scholar] [CrossRef]
- He, P.; Yu, H.; Li, D.; Zhou, H. Layered lithium transition metal oxide cathodes towards high energy lithium-ion batteries. J. Mater. Chem. 2012, 22, 3680–3695. [Google Scholar] [CrossRef]
- Li, M.; Lu, J.; Chen, Z.; Amine, K. 30 Years of Lithium-Ion Batteries. Adv. Mater. 2018, 1800561, 1800561. [Google Scholar] [CrossRef] [PubMed]
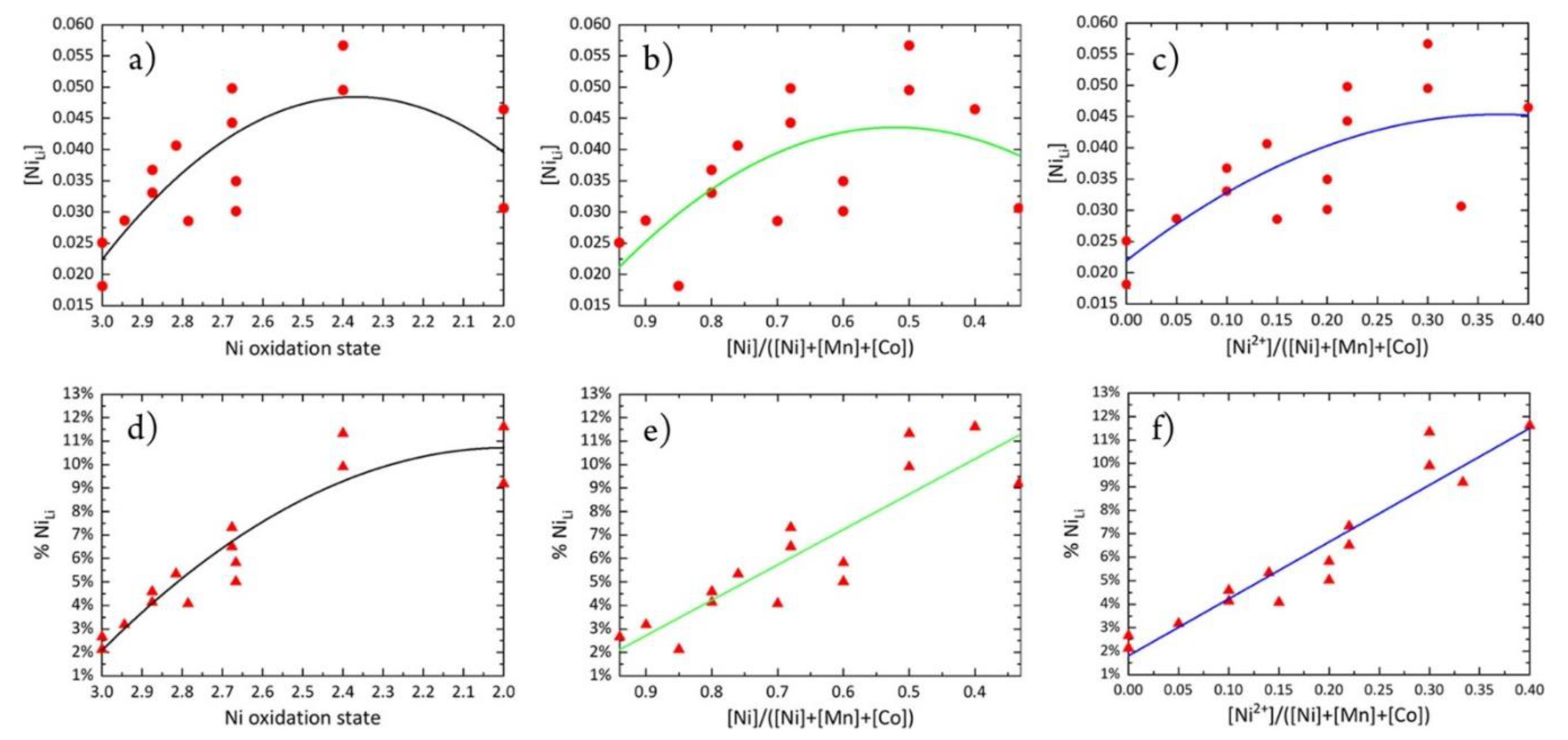
- Yin, L.; Li, Z.; Mattei, G.S.; Zheng, J.; Zhao, W.; Omenya, F.; Fang, C.; Li, W.; Li, J.; Xie, Q.; et al. Thermodynamics of Antisite Defects in Layered NMC Cathodes: Systematic Insights from High-Precision Powder Diffraction Analyses. Chem. Mater. 2020, 32, 1002–1010. [Google Scholar] [CrossRef]
- William, C. Crystal Structure Determination; Oxford University Press: New York, NY, USA, 1998. [Google Scholar]
- William, C. X-Ray Crystallography; Oxford University Press: New York, NY, USA, 2014. [Google Scholar]
- Li, L.; Xie, Y.; Maxey, E.; Harder, R. Methods for operando coherent X-ray diffraction of battery materials at the Advanced Photon Source. J. Synchrotron Radiat. 2019, 26, 220–229. [Google Scholar] [CrossRef] [PubMed]
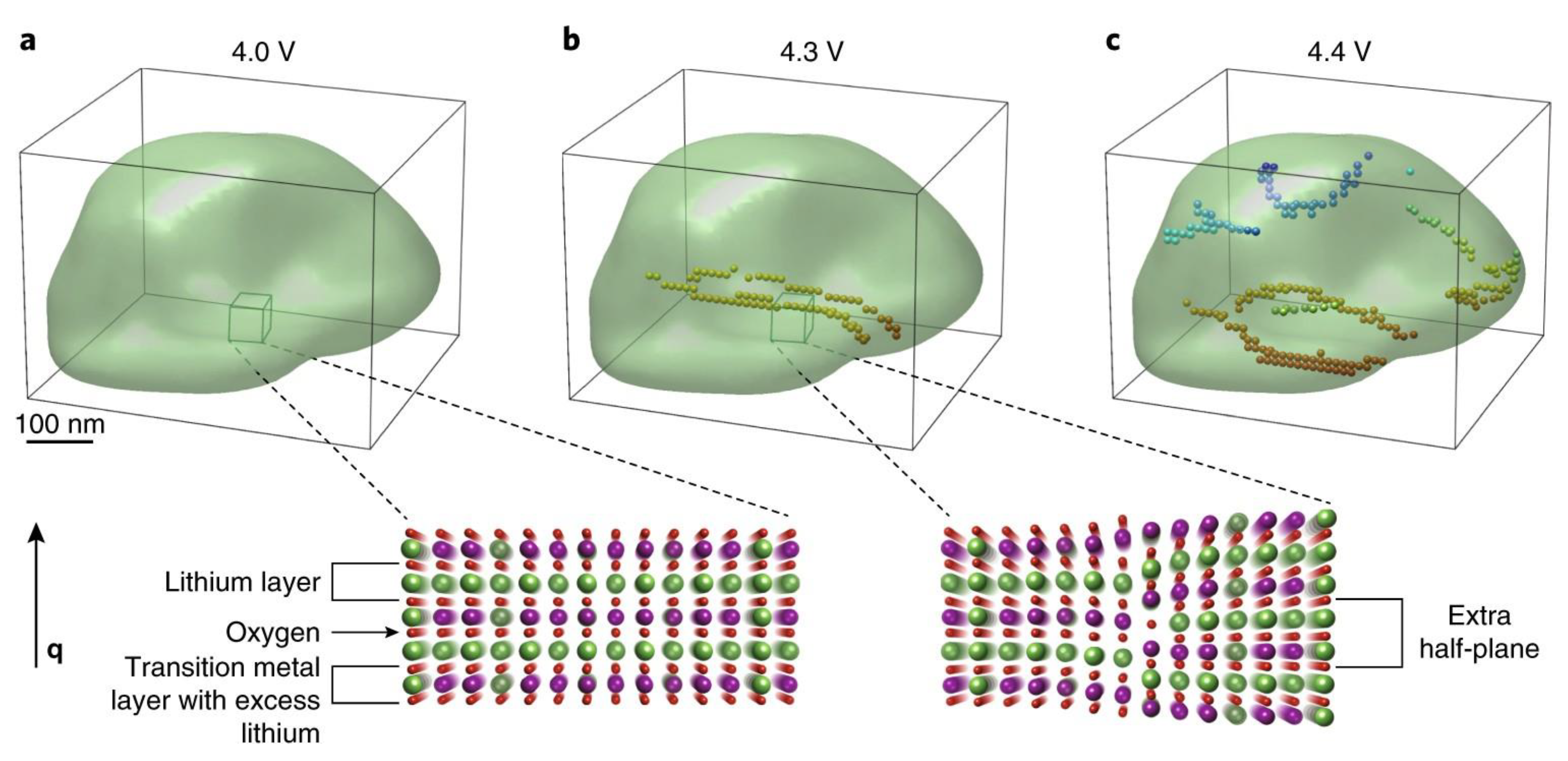
- Singer, A.; Zhang, M.; Hy, S.; Cela, D.; Fang, C.; Wynn, T.A.; Qiu, B.; Xia, Y.; Liu, Z.; Ulvestad, A.; et al. Nucleation of dislocations and their dynamics in layered oxide cathode materials during battery charging. Nat. Energy 2018, 3, 641–647. [Google Scholar] [CrossRef]
- Liu, D.; Shadike, Z.; Lin, R.; Qian, K.; Li, H.; Li, K.; Wang, S.; Yu, Q.; Liu, M.; Ganapathy, S.; et al. Review of Recent Development of In Situ/Operando Characterization Techniques for Lithium Battery Research. Adv. Mater. 2019, 31, 1–57. [Google Scholar] [CrossRef]
- Singer, A.; Ulvestad, A.; Cho, H.M.; Kim, J.W.; Maser, J.; Harder, R.; Meng, Y.S.; Shpyrko, O.G. Nonequilibrium structural dynamics of nanoparticles in LiNi1/2Mn3/2O4 cathode under operando conditions. Nano Lett. 2014, 14, 5295–5300. [Google Scholar] [CrossRef]
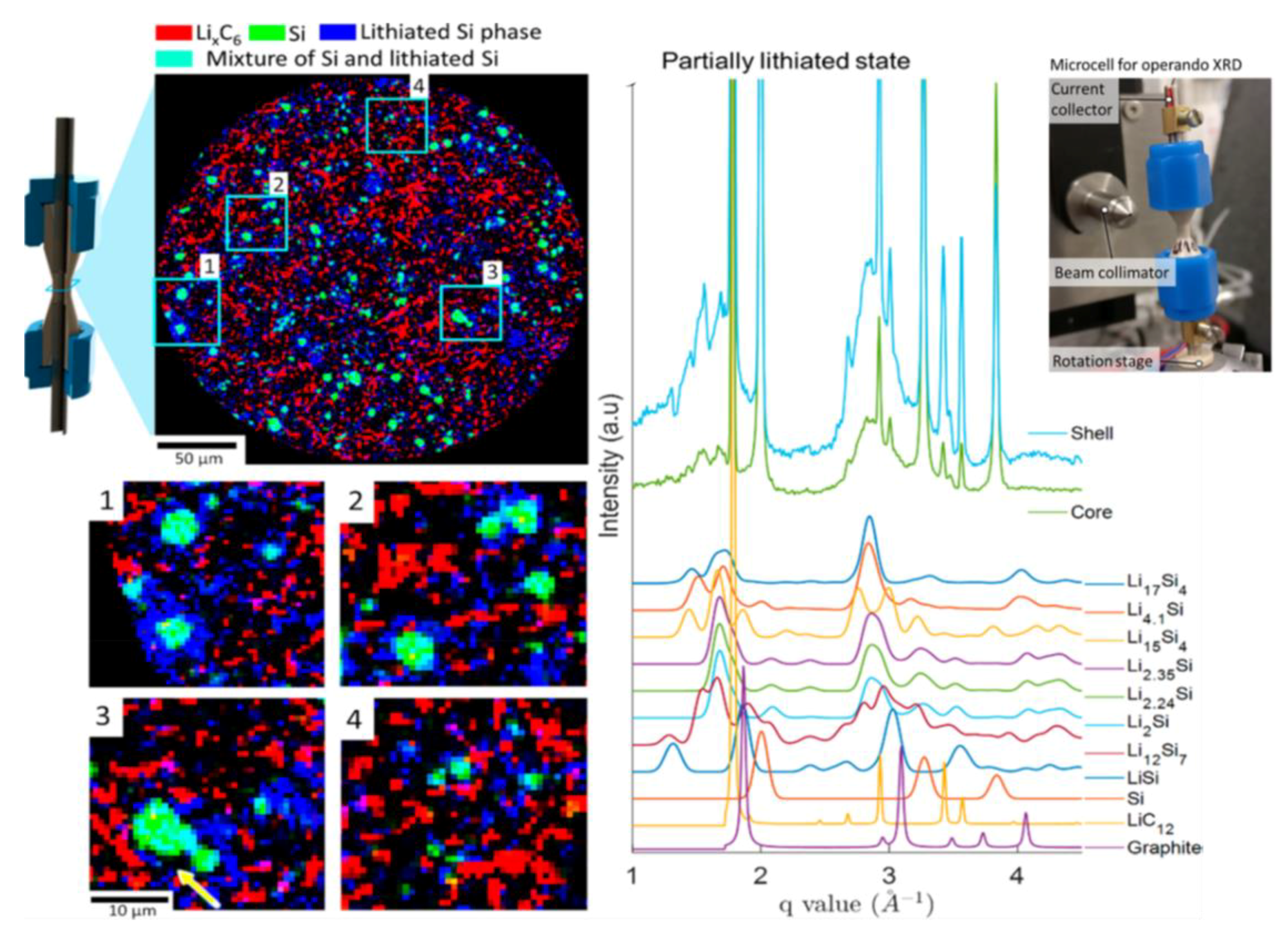
- Ehi-Eromosele, C.O.; Indris, S.; Bramnik, N.N.; Sarapulova, A.; Trouillet, V.; Pfaffman, L.; Melinte, G.; Mangold, S.; Darma, M.S.D.; Knapp, M.; et al. In Situ X-ray Diffraction and X-ray Absorption Spectroscopic Studies of a Lithium-Rich Layered Positive Electrode Material: Comparison of Composite and Core-Shell Structures. ACS Appl. Mater. Interfaces 2020, 12, 13852–13868. [Google Scholar] [CrossRef]
- Nayak, P.K.; Yang, L.; Pollok, K.; Langenhorst, F.; Aurbach, D.; Adelhelm, P. Investigation of Li1.17Ni0.20Mn0.53Co0.10O2 as an Interesting Li- and Mn-Rich Layered Oxide Cathode Material through Electrochemistry, Microscopy, and In Situ Electrochemical Dilatometry. Chem. Electro. Chem. 2019, 6, 2812–2819. [Google Scholar] [CrossRef]
- Ulvestad, A.; Singer, A.; Clark, J.N.; Cho, H.M.; Kim, J.W.; Harder, R.; Maser, J.; Meng, Y.S.; Shpyrko, O.G. Topological defect dynamics in operando battery nanoparticles. Science 2015, 348, 1344–1348. [Google Scholar] [CrossRef]
- You, H.; Liu, Y.; Ulvestad, A.; Pierce, M.S.; Komanicky, V. Studies of electrode structures and dynamics using coherent X-ray scattering and imaging. Curr. Opin. Electrochem. 2017, 4, 89–94. [Google Scholar] [CrossRef]
- Sottmann, J.; Homs-Regojo, R.; Wragg, D.S.; Fjellvåg, H.; Margadonna, S.; Emerich, H. Versatile electrochemical cell for Li/Na-ion batteries and high-throughput setup for combined operando X-ray diffraction and absorption spectroscopy. J. Appl. Crystallogr. 2016, 49, 1972–1981. [Google Scholar] [CrossRef]
- Braun, A.; Nordlund, D.; Song, S.W.; Huang, T.W.; Sokaras, D.; Liu, X.; Yang, W.; Weng, T.C.; Liu, Z. Hard X-rays in-soft X-rays out: An operando piggyback view deep into a charging lithium ion battery with X-ray Raman spectroscopy. J. Electron Spectros. Relat. Phenomena 2015, 200, 257–263. [Google Scholar] [CrossRef]
- Li, T.; Lim, C.; Cui, Y.; Zhou, X.; Kang, H.; Yan, B.; Meyerson, M.L.; Weeks, J.A.; Liu, Q.; Guo, F.; et al. In situ and operando investigation of the dynamic morphological and phase changes of a selenium-doped germanium electrode during (de)lithiation processes. J. Mater. Chem. A 2020, 8, 750–759. [Google Scholar] [CrossRef]
- Tardif, S.; Pavlenko, E.; Quazuguel, L.; Boniface, M.; Maréchal, M.; Micha, J.S.; Gonon, L.; Mareau, V.; Gebel, G.; Bayle-Guillemaud, P.; et al. Operando Raman Spectroscopy and Synchrotron X-ray Diffraction of Lithiation/Delithiation in Silicon Nanoparticle Anodes. ACS Nano 2017, 11, 11306–11316. [Google Scholar] [CrossRef] [PubMed]
- Nonaka, T.; Kawaura, H.; Makimura, Y.; Nishimura, Y.F.; Dohmae, K. In situ X-ray Raman scattering spectroscopy of a graphite electrode for lithium-ion batteries. J. Power Sources 2019, 419, 203–207. [Google Scholar] [CrossRef]
- Mukai, K.; Nonaka, T.; Uyama, T.; Nishimura, Y.F. In situ X-ray Raman spectroscopy and magnetic susceptibility study on the Li[Li0.15Mn1.85]O4 oxygen anion redox reaction. Chem. Commun. 2020, 56, 1701–1704. [Google Scholar] [CrossRef] [PubMed]
- Ketenoglu, D.; Spiekermann, G.; Harder, M.; Oz, E.; Koz, C.; Yagci, M.C.; Yilmaz, E.; Yin, Z.; Sahle, C.J.; Detlefs, B.; et al. X-ray Raman spectroscopy of lithium-ion battery electrolyte solutions in a flow cell. J. Synchrotron Radiat. 2018, 25, 537–542. [Google Scholar] [CrossRef]
- Wang, F.; Yu, H.C.; Chen, M.H.; Wu, L.; Pereira, N.; Thornton, K.; Van Der Ven, A.; Zhu, Y.; Amatucci, G.G.; Graetz, J. Tracking lithium transport and electrochemical reactions in nanoparticles. Nat. Commun. 2012, 3, 1–8. [Google Scholar] [CrossRef]
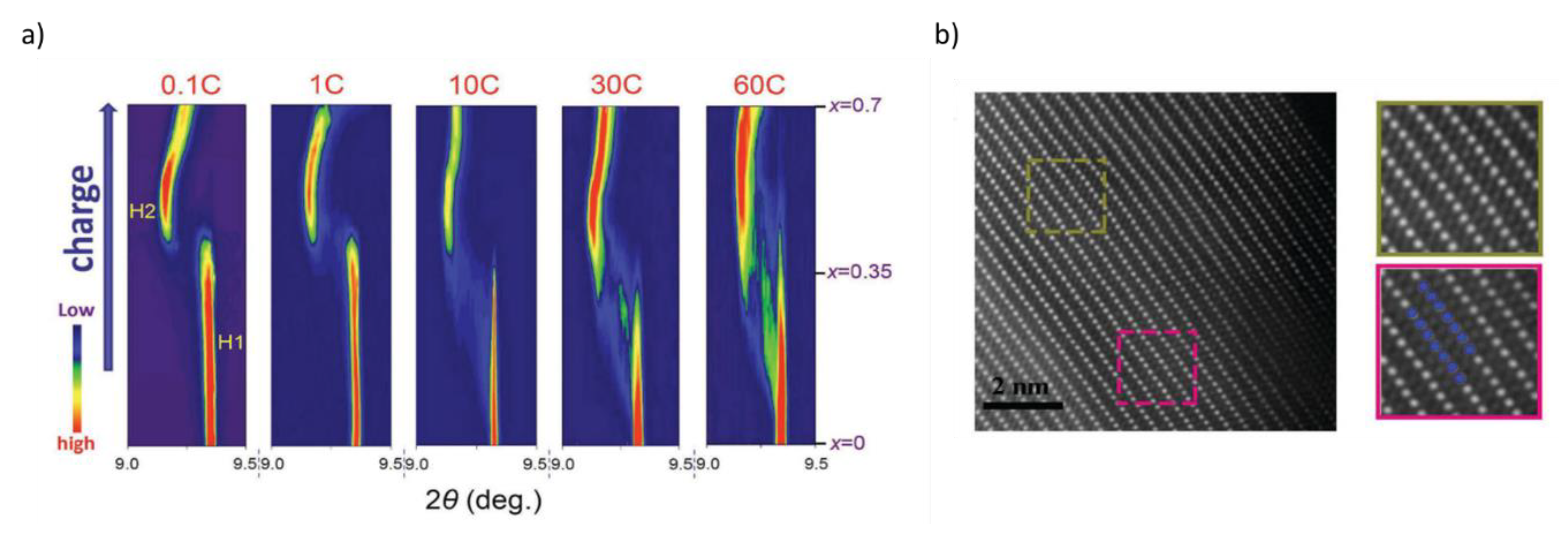
- Zhou, Y.N.; Yue, J.L.; Hu, E.; Li, H.; Gu, L.; Nam, K.W.; Bak, S.M.; Yu, X.; Liu, J.; Bai, J.; et al. High-Rate Charging Induced Intermediate Phases and Structural Changes of Layer-Structured Cathode for Lithium-Ion Batteries. Adv. Energy Mater. 2016, 6, 1–8. [Google Scholar] [CrossRef]
- Nelson, J.; Misra, S.; Yang, Y.; Jackson, A.; Liu, Y.; Wang, H.; Dai, H.; Andrews, J.C.; Cui, Y.; Toney, M.F. In operando X-ray diffraction and transmission X-ray microscopy of lithium sulfur batteries. J. Am. Chem. Soc. 2012, 134, 6337–6343. [Google Scholar] [CrossRef]
- Chueh, W.C.; El Gabaly, F.; Sugar, J.D.; Bartelt, N.C.; McDaniel, A.H.; Fenton, K.R.; Zavadil, K.R.; Tyliszczak, T.; Lai, W.; McCarty, K.F. Intercalation pathway in many-particle LiFePO4 electrode revealed by nanoscale state-of-charge mapping. Nano Lett. 2013, 13, 866–872. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; El Gabaly, F.; Ferguson, T.R.; Smith, R.B.; Bartelt, N.C.; Sugar, J.D.; Fenton, K.R.; Cogswell, D.A.; Kilcoyne, A.L.D.; Tyliszczak, T.; et al. Current-induced transition from particle-by-particle to concurrent intercalation in phase-separating battery electrodes. Nat. Mater. 2014, 13, 1149–1156. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Liu, Y.; Martha, S.K.; Wu, Z.; Andrews, J.C.; Ice, G.E.; Pianetta, P.; Nanda, J. Nanoscale morphological and chemical changes of high voltage lithium-manganese rich NMC composite cathodes with cycling. Nano Lett. 2014, 14, 4334–4341. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.H.; Huang, X.; Schwarz, K.; Huang, R.; Arias, T.A.; Brock, J.D.; Abruña, H.D. Direct visualization of sulfur cathodes: New insights into Li-S batteries via operando X-ray based methods. Energy Environ. Sci. 2018, 11, 202–210. [Google Scholar] [CrossRef]
- Xie, Y.; Wang, H.; Xu, G.; Wang, J.; Sheng, H.; Chen, Z.; Ren, Y.; Sun, C.J.; Wen, J.; Wang, J.; et al. In Operando XRD and TXM Study on the Metastable Structure Change of NaNi1/3Fe1/3Mn1/3O2 under Electrochemical Sodium-Ion Intercalation. Adv. Energy Mater. 2016, 6, 3–7. [Google Scholar] [CrossRef]
- Thomas, M.M.; Heenan, A.W.; Chun, T.; Julia, E.; Parker, D.M.; Andrew, S.; Leach, J.B.; Robinson, A.L.; Alexander, D.; Rhodri, J.; et al. Identifying the Origins of Microstructural Defects Such as Cracking within Ni-Rich NMC811 Cathode Particles for Lithium-ion Batteries. Adv. Energy Mater. 2020. [Google Scholar] [CrossRef]
- Villevieille, C.; Ebner, M.; Gómez-Cámer, J.L.; Marone, F.; Novák, P.; Wood, V. Influence of conversion material morphology on electrochemistry studied with operando X-ray tomography and diffraction. Adv. Mater. 2015, 27, 1676–1681. [Google Scholar] [CrossRef]
- Finegan, D.P.; Vamvakeros, A.; Tan, C.; Heenan, T.M.M.; Daemi, S.R.; Seitzman, N.; Di Michiel, M.; Jacques, S.; Beale, A.M.; Brett, D.J.L.; et al. Spatial quantification of dynamic inter and intra particle crystallographic heterogeneities within lithium ion electrodes. Nat. Commun. 2020, 11, 1–11. [Google Scholar] [CrossRef]
- Bleuet, P.; Susini, J.; Hodeau, J.; Eonore, E.L.; Ee, E.D.; Walter, P. Probing the structure of heterogeneous diluted materials by diffraction to mography. Nat. Mater. 2008, 7. [Google Scholar] [CrossRef]
- Daemi, S.R.; Tan, C.; Vamvakeros, A.; Heenan, T.M.M.; Finegan, D.P.; Di Michiel, M.; Beale, A.M.; Cookson, J.; Petrucco, E.; Weaving, J.S.; et al. Exploring cycling induced crystallographic change in NMC with X-ray diffraction computed tomography. Phys. Chem. Chem. Phys. 2020. [Google Scholar] [CrossRef]
- Finegan, D.P.; Vamvakeros, A.; Cao, L.; Tan, C.; Heenan, T.M.M.; Daemi, S.R.; Jacques, S.D.M.; Beale, A.M.; Di Michiel, M.; Smith, K.; et al. Spatially Resolving Lithiation in Silicon-Graphite Composite Electrodes via in Situ High-Energy X-ray Diffraction Computed Tomography. Nano Lett. 2019, 19, 3811–3820. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Kazemiabnavi, S.; Grenier, A.; Vaughan, G.; Di Michiel, M.; Polzin, B.J.; Thornton, K.; Chapman, K.W.; Chupas, P.J. Quantifying Reaction and Rate Heterogeneity in Battery Electrodes in 3D through Operando X-ray Diffraction Computed Tomography. ACS Appl. Mater. Interfaces 2019, 11, 18386–18394. [Google Scholar] [CrossRef] [PubMed]
- Vanpeene, V.; King, A.; Maire, E.; Roué, L. In situ characterization of Si-based anodes by coupling synchrotron X-ray tomography and diffraction. Nano Energy 2019, 56, 799–812. [Google Scholar] [CrossRef]
- Lemarié, Q.; Maire, E.; Idrissi, H.; Thivel, P.X.; Alloin, F.; Roué, L. Sulfur-Based Electrode Using a Polyelectrolyte Binder Studied via Coupled in Situ Synchrotron X-ray Diffraction and Tomography. ACS Appl. Energy Mater. 2020, 3, 2422–2431. [Google Scholar] [CrossRef]
- Finegan, D.P.; Quinn, A.; Wragg, D.S.; Colclasure, A.M.; Lu, X.; Tan, C.; Heenan, T.M.M.; Jervis, R.; Brett, D.J.L.; Das, S.; et al. Spatial dynamics of lithiation and lithium plating during high-rate operation of graphite electrodes. Energy Environ. Sci. 2020. [Google Scholar] [CrossRef]
- Ren, Y.; Zuo, X. Synchrotron X-Ray and Neutron Diffraction, Total Scattering, and Small-Angle Scattering Techniques for Rechargeable Battery Research. Small Methods 2018, 2, 1800064. [Google Scholar] [CrossRef]
- Permien, S.; Hansen, A.L.; Van Dinter, J.; Indris, S.; Neubüser, G.; Kienle, L.; Doyle, S.; Mangold, S.; Bensch, W. Unveiling the Reaction Mechanism during Li Uptake and Release of Nanosized “niFeMnO4”: Operando X-ray Absorption, X-ray Diffraction, and Pair Distribution Function Investigations. ACS Omega 2019, 4, 2398–2409. [Google Scholar] [CrossRef]
- Wiaderek, K.M.; Borkiewicz, O.J.; Castillo-Martínez, E.; Robert, R.; Pereira, N.; Amatucci, G.G.; Grey, C.P.; Chupas, P.J.; Chapman, K.W. Comprehensive insights into the structural and chemical changes in mixed-anion FeOF electrodes by using operando pdf and NMR spectroscopy. J. Am. Chem. Soc. 2013, 135, 4070–4078. [Google Scholar] [CrossRef]
- Benmore, C.J. A Review of High-Energy X-Ray Diffraction from Glasses and Liquids. ISRN Mater. Sci. 2012, 2012, 1–19. [Google Scholar] [CrossRef]
- Hong, X.; Ehm, L.; Zhong, Z.; Ghose, S.; Duffy, T.S.; Weidner, D.J. High-energy X-ray focusing and applications to pair distribution function investigation of Pt and Au nanoparticles at high pressures. Sci. Rep. 2016, 6, 1–8. [Google Scholar] [CrossRef]
- Poulsen, H.F.; Neuefeind, J.; Neumann, H.B.; Schneider, J.R.; Zeidler, M.D. Amorphous silica studied by high energy X-ray diffraction. J. Non. Cryst. Solids 1995, 188, 63–74. [Google Scholar] [CrossRef]
- Sánchez-Gil, V.; Noya, E.G.; Temleitner, L.; Pusztai, L. Reverse Monte Carlo modeling: The two distinct routes of calculating the experimental structure factor. J. Mol. Liq. 2015, 207, 211–215. [Google Scholar] [CrossRef][Green Version]
- Eremenko, M.; Krayzman, V.; Gagin, A.; Levin, I. Advancing reverse Monte Carlo structure refinements to the nanoscale. J. Appl. Crystallogr. 2017, 50, 1561–1570. [Google Scholar] [CrossRef]
- Soper, A.K. Tests of the empirical potential structure refinement method and a new method of application to neutron diffraction data on water. Mol. Phys. 2001, 99, 1503–1516. [Google Scholar] [CrossRef]
- Soper, A.K.; Page, K.; Llobet, A. Empirical potential structure refinement of semi-crystalline polymer systems: Polytetrafluoroethylene and polychlorotrifluoroethylene. J. Phys. Condens. Matter 2013, 25. [Google Scholar] [CrossRef]
- Pandey, A.; Biswas, P.; Drabold, D.A. Inversion of diffraction data for amorphous materials. Nat. Publ. Gr. 2016, 1–8. [Google Scholar] [CrossRef]
- Saito, Y.; Iihara, J.; Yamaguchi, K.; Haruna, T.; Onishi, M. Structure analysis technology for amorphous materials by synchrotron radiation X-ray measurements and molecular dynamics simulations. SEI Tech. Rev. 2008, 67, 27–32. [Google Scholar]
- Chao, W.; Fischer, P.; Tyliszczak, T.; Rekawa, S.; Anderson, E.; Naulleau, P. Real space soft x-ray imaging at 10 nm spatial resolution. Opt. Express 2012, 20, 9777. [Google Scholar] [CrossRef]
- Hill, J.; Campbell, S.; Carini, G.; Chen-Wiegart, Y.C.K.; Chu, Y.; Fluerasu, A.; Fukuto, M.; Idir, M.; Jakoncic, J.; Jarrige, I.; et al. Future trends in synchrotron science at NSLS-II. J. Phys. Condens. Matter 2020, 32, 374008. [Google Scholar] [CrossRef]
- Hatsui, T.; Graafsma, H. X-ray imaging detectors for synchrotron and XFEL sources. IUCrJ 2015, 2, 371–383. [Google Scholar] [CrossRef]
- Vaughan, G.B.M.; Baker, R.; Barret, R.; Bonnefoy, J.; Buslaps, T.; Checchia, S.; Duran, D.; Fihman, F.; Got, P.; Kieffer, J.; et al. ID15A at the ESRF-a beamline for high speed operando X-ray diffraction, diffraction tomography and total scattering. J. Synchrotron Radiat. 2020, 27, 515–528. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Llewellyn, A.V.; Matruglio, A.; Brett, D.J.L.; Jervis, R.; Shearing, P.R. Using In-Situ Laboratory and Synchrotron-Based X-ray Diffraction for Lithium-Ion Batteries Characterization: A Review on Recent Developments. Condens. Matter 2020, 5, 75. https://doi.org/10.3390/condmat5040075
Llewellyn AV, Matruglio A, Brett DJL, Jervis R, Shearing PR. Using In-Situ Laboratory and Synchrotron-Based X-ray Diffraction for Lithium-Ion Batteries Characterization: A Review on Recent Developments. Condensed Matter. 2020; 5(4):75. https://doi.org/10.3390/condmat5040075
Chicago/Turabian StyleLlewellyn, Alice V., Alessia Matruglio, Dan J. L. Brett, Rhodri Jervis, and Paul R. Shearing. 2020. "Using In-Situ Laboratory and Synchrotron-Based X-ray Diffraction for Lithium-Ion Batteries Characterization: A Review on Recent Developments" Condensed Matter 5, no. 4: 75. https://doi.org/10.3390/condmat5040075
APA StyleLlewellyn, A. V., Matruglio, A., Brett, D. J. L., Jervis, R., & Shearing, P. R. (2020). Using In-Situ Laboratory and Synchrotron-Based X-ray Diffraction for Lithium-Ion Batteries Characterization: A Review on Recent Developments. Condensed Matter, 5(4), 75. https://doi.org/10.3390/condmat5040075




