Infrared Synchrotron Radiation and Its Application to the Analysis of Cultural Heritage
Abstract
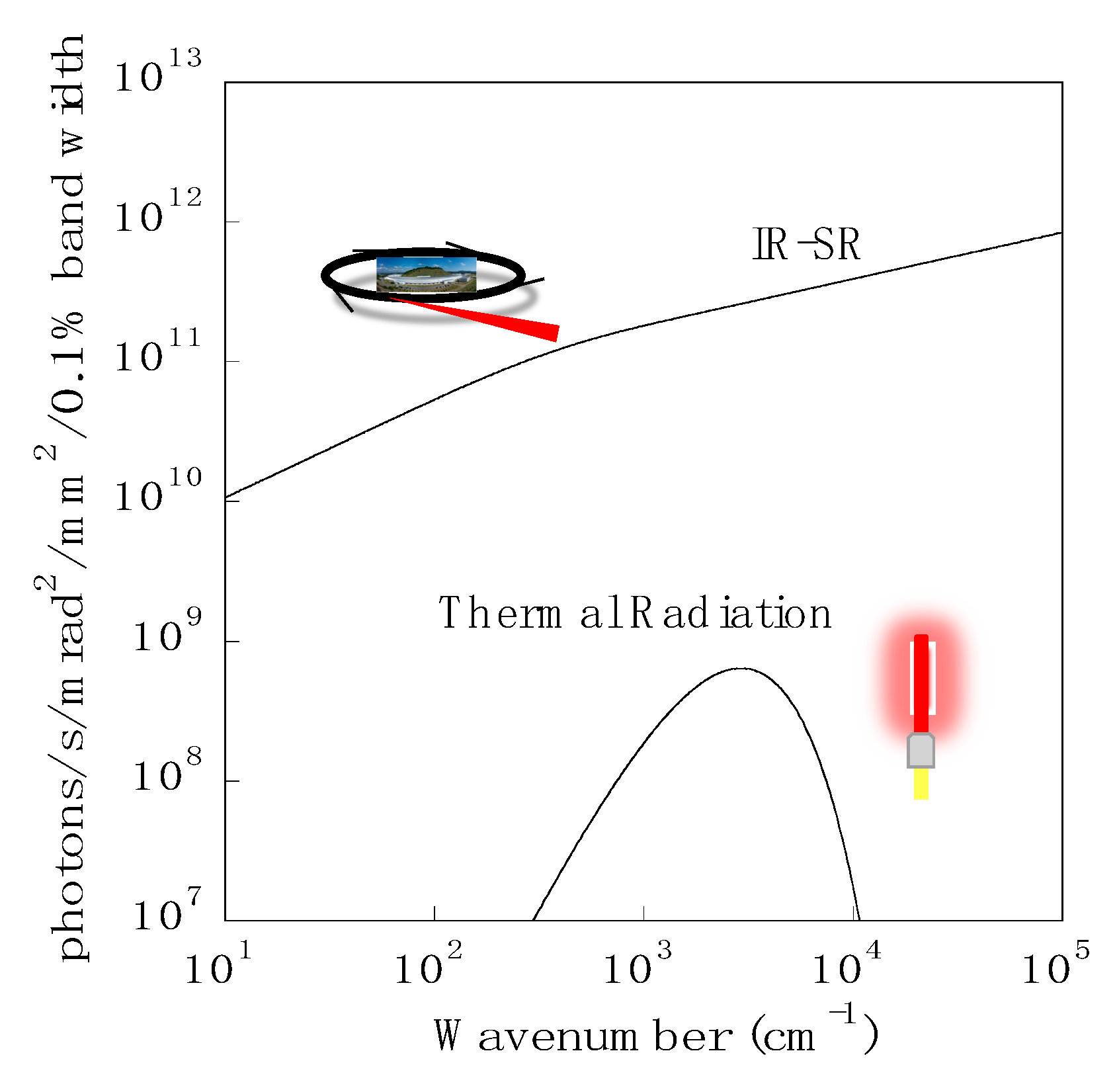
1. Introduction
2. Infrared Beamline BL43IR at SPring-8
3. Cultural Heritage Experiments
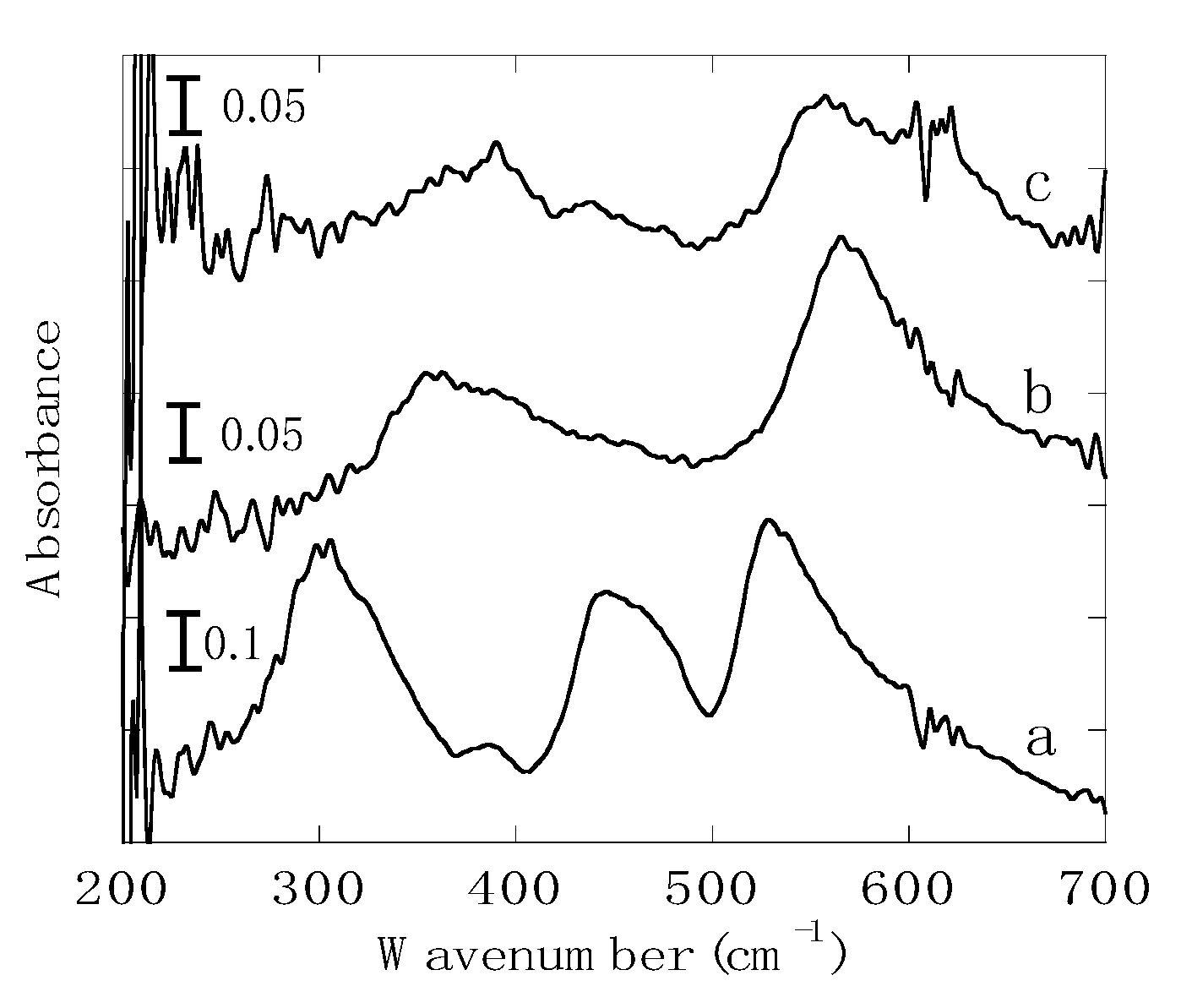
3.1. Iron Oxide in Bengala Pigment
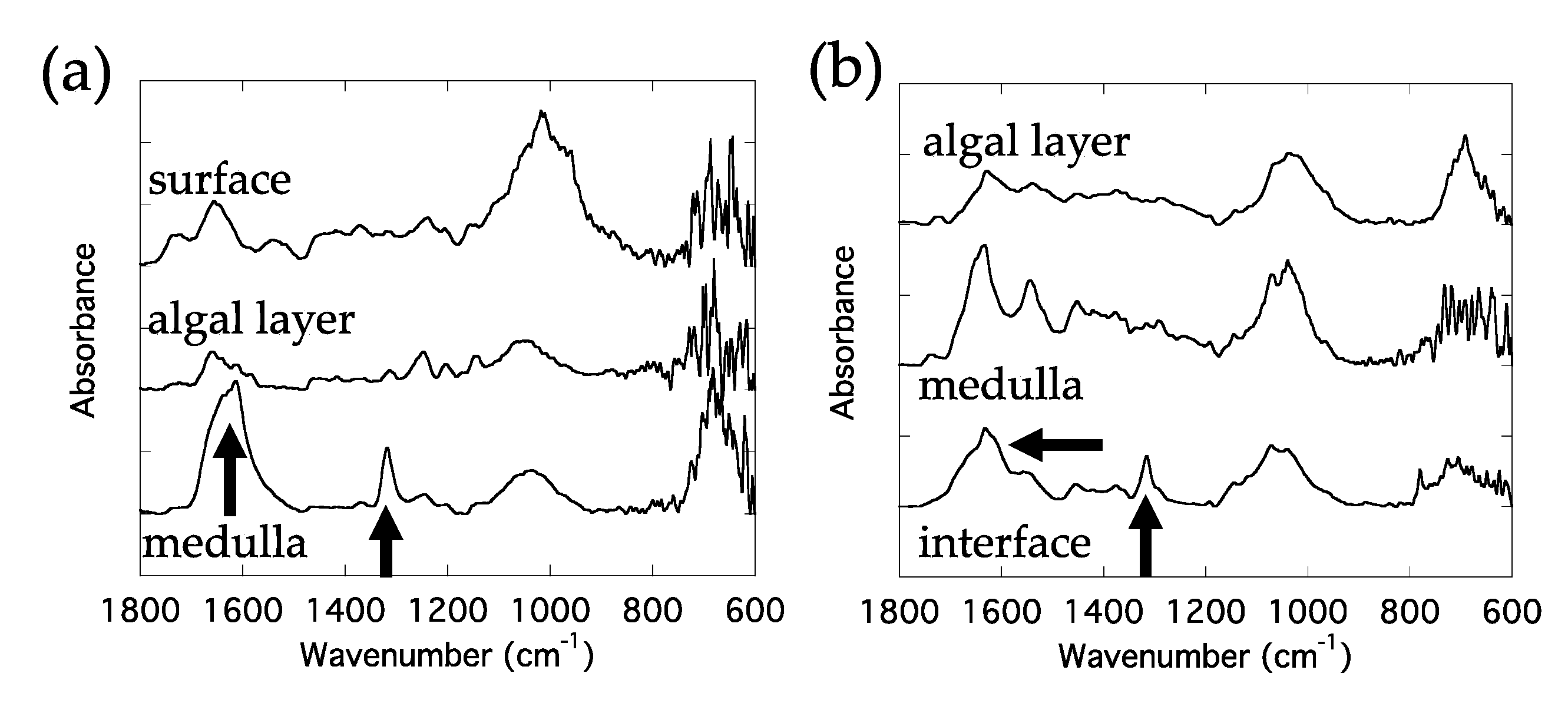
3.2. Biomineral Distribution Generated by a Lichen Growing on a Stone Cultural Heritage
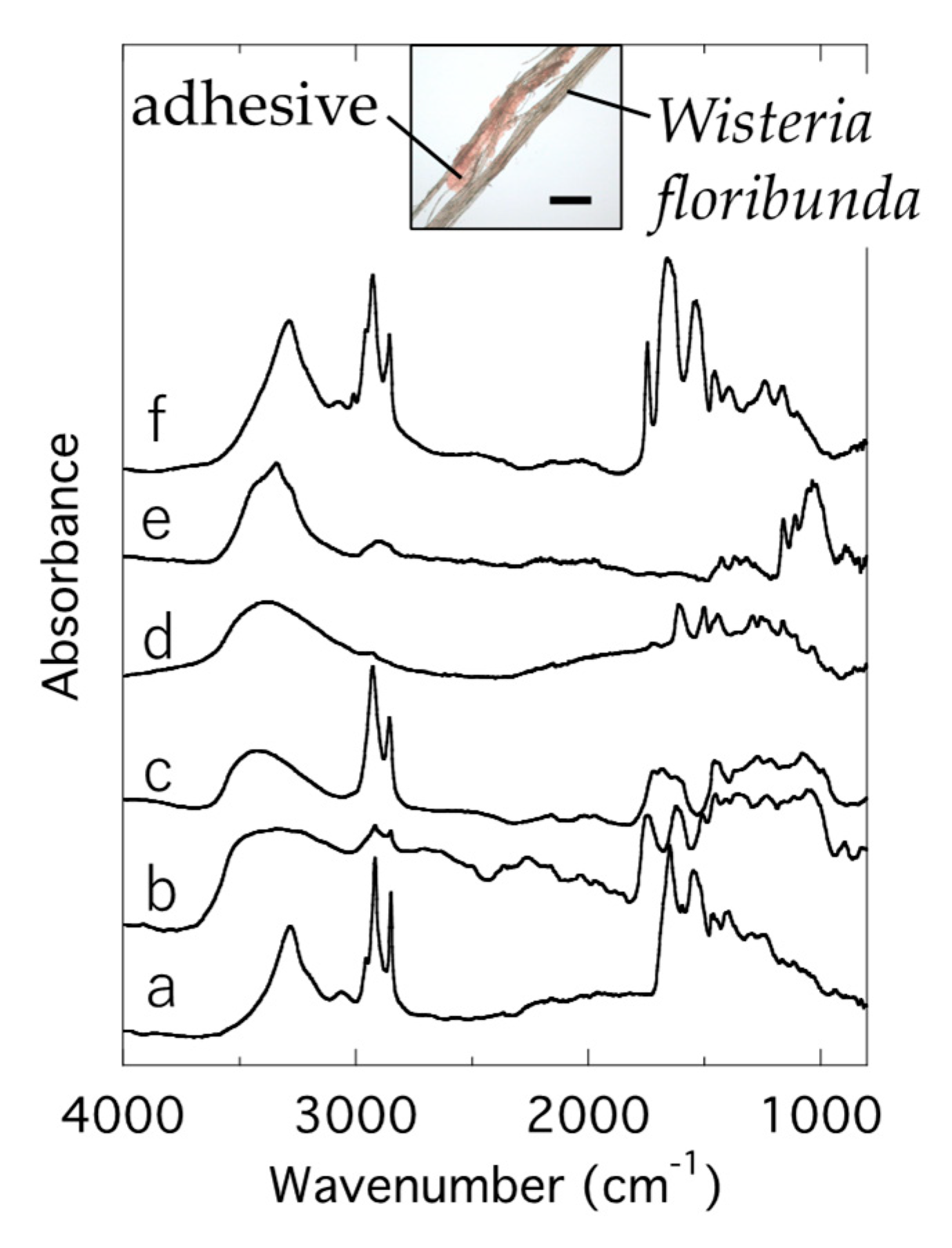
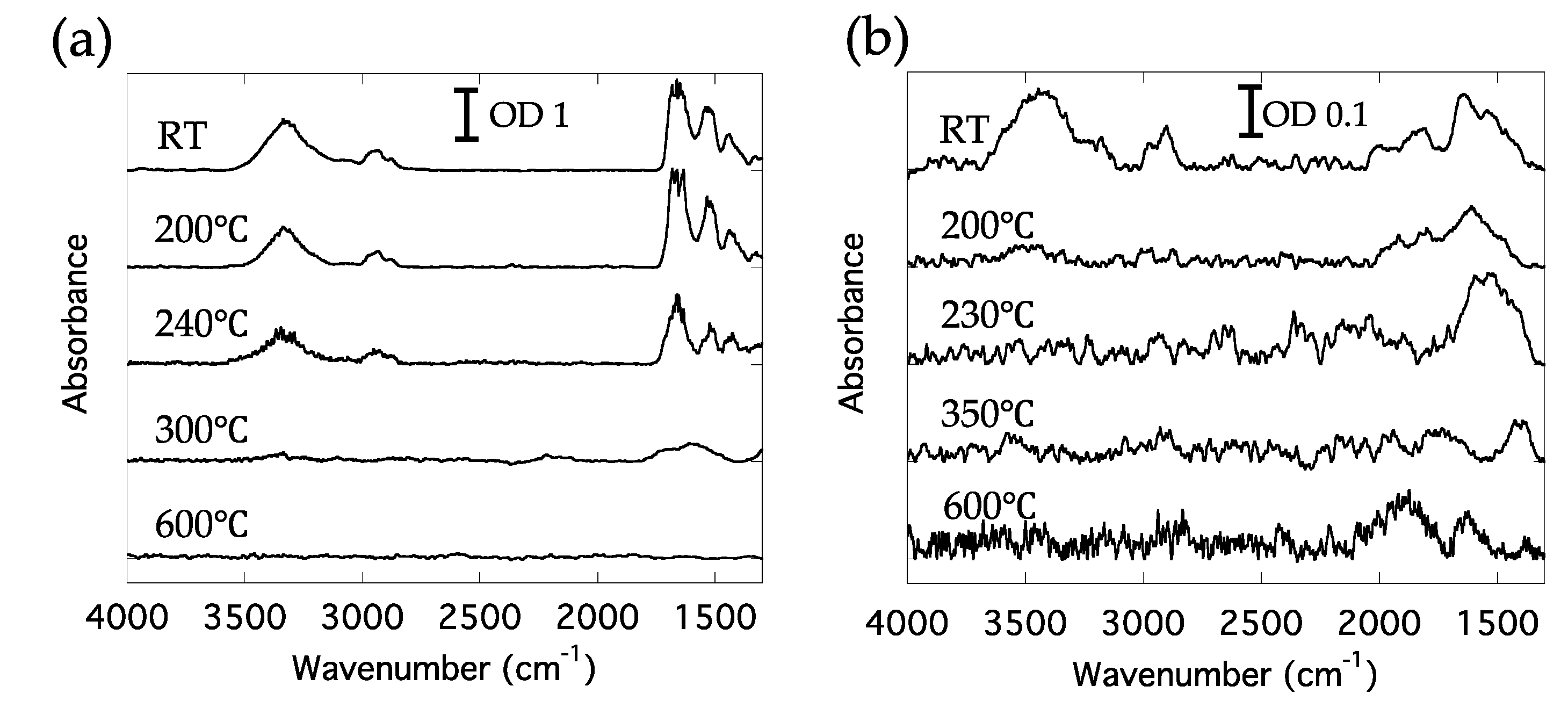
3.3. Adhesives on Fiber of Traditional Snow Festival Costume in Niino
3.4. Organic Materials in Overglaze Layers on Ceramics
4. Summary
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fukunaga, K.; Sekine, N.; Hosako, I.; Oda, N.; Yoneyama, H.; Sudou, T. Real0time terahertz imaging for art conservation science. J. Eur. Opt. Soc. Rapid 2008, 3, 08027. [Google Scholar] [CrossRef]
- Gallerano, G.P.; Doria, A.; Giovenale, E.; Messina, G.; Petralia, A.; Spassovsky, I.; Fukunaga, K.; Hosako, I. THz-ARTE: Non-Invasive Diagnostics for Art Conservation. In Proceedings of the 2008 33rd International Conference on Infrared, Millimeter and Terahertz Waves, Pasadena, CA, USA, 15–19 September 2008; pp. 1–2. [Google Scholar]
- Manceau, J.-M.; Nevin, A.; Fotakis, C.; Tzortzakis, S. Terahertz time domain spectroscopy for the analysis of cultural heritage related materials. Appl. Phys. B 2008, 90, 365–368. [Google Scholar] [CrossRef]
- Jackson, J.B.; Mourou, M.; Whitaker, J.F.; Duing, I.N.; Williamson, S.L.; Menu, M.; Mourou, G.A. Terahertz imaging for non-destructive evaluation of mural paitings. Opt. Commun. 2008, 281, 527–532. [Google Scholar] [CrossRef]
- Fukunaga, K. Terahertz spectroscopy for non-invasive analysis of cultural properties. J. Natl. Inst. Inf. Commun. Technol. 2009, 55, 67–71. [Google Scholar]
- van Loon, A.; Gambardella, A.A.; Gonalez, V.; Cotte, M.; Nolf, W.; Keune, K.; Leonhardt, E.; Groot, S.; Gaibor, A.N.P.; Vandivere, A. Out of the blue: Vermeer’s use of ultramarine in girl with a pearl earring. Herit. Sci. 2020, 8, 25. [Google Scholar] [CrossRef]
- Pronti, L.; Romani, M.; Vivian, G.; Stani, C.; Gioia, P.; Cestelli, M. Advanced methods for the analysis of Roman wall paintings: Elemental and molecular detection by means of synchrotron RT-IR and SEM icro-imaging spectroscopy. Rend. Lincei. Sci. Fis. Naurali 2020, 1–9. [Google Scholar] [CrossRef]
- Salvado, N.; Buti, S.; Tobin, M.J.; Pantos, E.; John, A.; Prag, N.W.; Pradell, T. Advantages of the use of SR-FT-IR microspectroscopy: Applications to cultural heritage. Anal. Chem. 2005, 77, 3444–3451. [Google Scholar] [CrossRef] [PubMed]
- Cinque, G.; Marcelli, A. Synchrotron radiation inraRed microspectroscopy and imaging in the characterization of archaeological materials and cultural heritage artefacts. EMU Notes Mineral. 2019, 20, 411–444. [Google Scholar]
- Ikemoto, Y.; Ishikawa, M.; Nakashima, S.; Okamura, H.; Haruyama, Y.; Matsui, S.; Moriwaki, T.; Kinoshita, T. Development of scattering near-field optical microspectroscopy apparatus using an infrared synchrotron radiation sources. Opt. Commun. 2012, 285, 2212–2217. [Google Scholar] [CrossRef]
- Torgasin, K.; Morita, K.; Zne, H.; Masuda, K.; Bakr, M.; Nagasaki, K.; Kii, T.; Ohgaki, H. Study on anormalous photoemission of LaB6 at high temperatures. Phys. Scr. 2019, 94, 7. [Google Scholar] [CrossRef]
- Bechtel, H.A.; Muller, E.A.; Olmon, R.L.; Martin, M.C.; Raschke, M.B. Ultrabroadband infrared nanospectroscopic imaging. PNAS 2014, 111, 7191–7196. [Google Scholar] [CrossRef] [PubMed]
- Kimura, H.; Moriwaki, T.; Takahashi, S.; Aoyagi, H.; Matsushita, T.; Ishizawa, Y.; Masaki, M.; Oishi, M.; Ohkuma, H.; Namba, T.; et al. Infrared beamline BL43IR at SPring-8:: design and commissioning. Nucl. Instrum. Methods Phys. Res. A 2001, 467–468, 441–444. [Google Scholar] [CrossRef]
- Moriwaki, T.; Ikemoto, Y. BL43IR at SPring-8. Infrared Phys. Technol. 2008, 51, 400–403. [Google Scholar] [CrossRef]
- Kobayashi, R.; Hashimoto, K.; Yoneyama, N.; Yoshimi, K.; Motoyama, Y.; Iguchi, S.; Ikemoto, Y.; Moriwaki, T.; Taniguchi, H.; Sasaki, T. Dimer-Mott and charge-ordered insulating states in the quasi-one-dimensional organic conducting δ′p- and δ′c-(BPDT-TTF)2ICl2. Phys. Rev. B 2017, 96, 115112. [Google Scholar] [CrossRef]
- Okamura, H.; Shoji, K.; Miyata, K.; Sugawara, H.; Moriwaki, T.; Ikemoto, Y. Pressure suppression of spin-density wave gap in the optical conductivity of SrFe2As2. J. Phys. Soc. Jpn. 2013, 82, 074720. [Google Scholar] [CrossRef]
- Kamijoh, T. Red Pigments used from Jomon Period to Kofun Period. J. Jpn. Soc. Colour Mater. 2004, 77, 86–90. [Google Scholar] [CrossRef]
- Kawasaki, E.; Matsui, T.; Yamamoto, Y.; Hara, K. The documentation method using lichens growing on stoneworks in order to protect cultural heritage stone monuments—The case of Angkor monuments of Cambodia. Lichenology 2013, 11, 39–52. [Google Scholar]
- Parsons, J.G.; Dokken, K.M.; McClure, J.; Gardea-Torresdey, J.L. FTIR, XA, and XRD study of cadmium complexes with l-crysteine. Phlyhedron 2013, 56, 237–242. [Google Scholar] [CrossRef]
- Pucetaite, M.; Tamosaityte, S.; Engdahl, A.; Ceponkus, J.; Sablinskas, V.; Uvdal, P. Microspectroscopic infrared specular reflection chemical imaging of multi-component urinary stones: MIR vs. FIR. Cent. Eur. J. Chem. 2014, 12, 44–52. [Google Scholar] [CrossRef][Green Version]
- Yanchen, H.; Shengjie, L.; Zeming, Q.; Xin, C. Application of far-infrared spectroscopy to the structural identification of protein materials. Phys. Chem. Chem. Phys. 2018, 18, 11643–11648. [Google Scholar]
- Ohnishi, S. Chapter 6 Techniques of Overglaze. In Traditional Techniques of Ceramics Art, 1st ed.; Rikogakusya Publishing Co., Ltd.: Tokyo, Japan, 2001; pp. 231–278. [Google Scholar]
- Higuchi, T.; Ninomiya, S. Attempt to Analyze the Organic Residuals in Overglaze Layers on Ceramics. In Proceedings of the 31st Annual Meeting of the Japan Society for Scientific Studies on Cultural Property, Nara, Japan, 5 July 2014; pp. 258–259. [Google Scholar]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ikemoto, Y.; Tanaka, M.; Higuchi, T.; Semba, T.; Moriwaki, T.; Kawasaki, E.; Okuyama, M. Infrared Synchrotron Radiation and Its Application to the Analysis of Cultural Heritage. Condens. Matter 2020, 5, 28. https://doi.org/10.3390/condmat5020028
Ikemoto Y, Tanaka M, Higuchi T, Semba T, Moriwaki T, Kawasaki E, Okuyama M. Infrared Synchrotron Radiation and Its Application to the Analysis of Cultural Heritage. Condensed Matter. 2020; 5(2):28. https://doi.org/10.3390/condmat5020028
Chicago/Turabian StyleIkemoto, Yuka, Manako Tanaka, Tomohiro Higuchi, Toshirou Semba, Taro Moriwaki, Emi Kawasaki, and Masayoshi Okuyama. 2020. "Infrared Synchrotron Radiation and Its Application to the Analysis of Cultural Heritage" Condensed Matter 5, no. 2: 28. https://doi.org/10.3390/condmat5020028
APA StyleIkemoto, Y., Tanaka, M., Higuchi, T., Semba, T., Moriwaki, T., Kawasaki, E., & Okuyama, M. (2020). Infrared Synchrotron Radiation and Its Application to the Analysis of Cultural Heritage. Condensed Matter, 5(2), 28. https://doi.org/10.3390/condmat5020028



