A Fish-Based Tool for the Quality Assessment of Portuguese Large Rivers
Abstract
1. Introduction
2. Materials and Methods
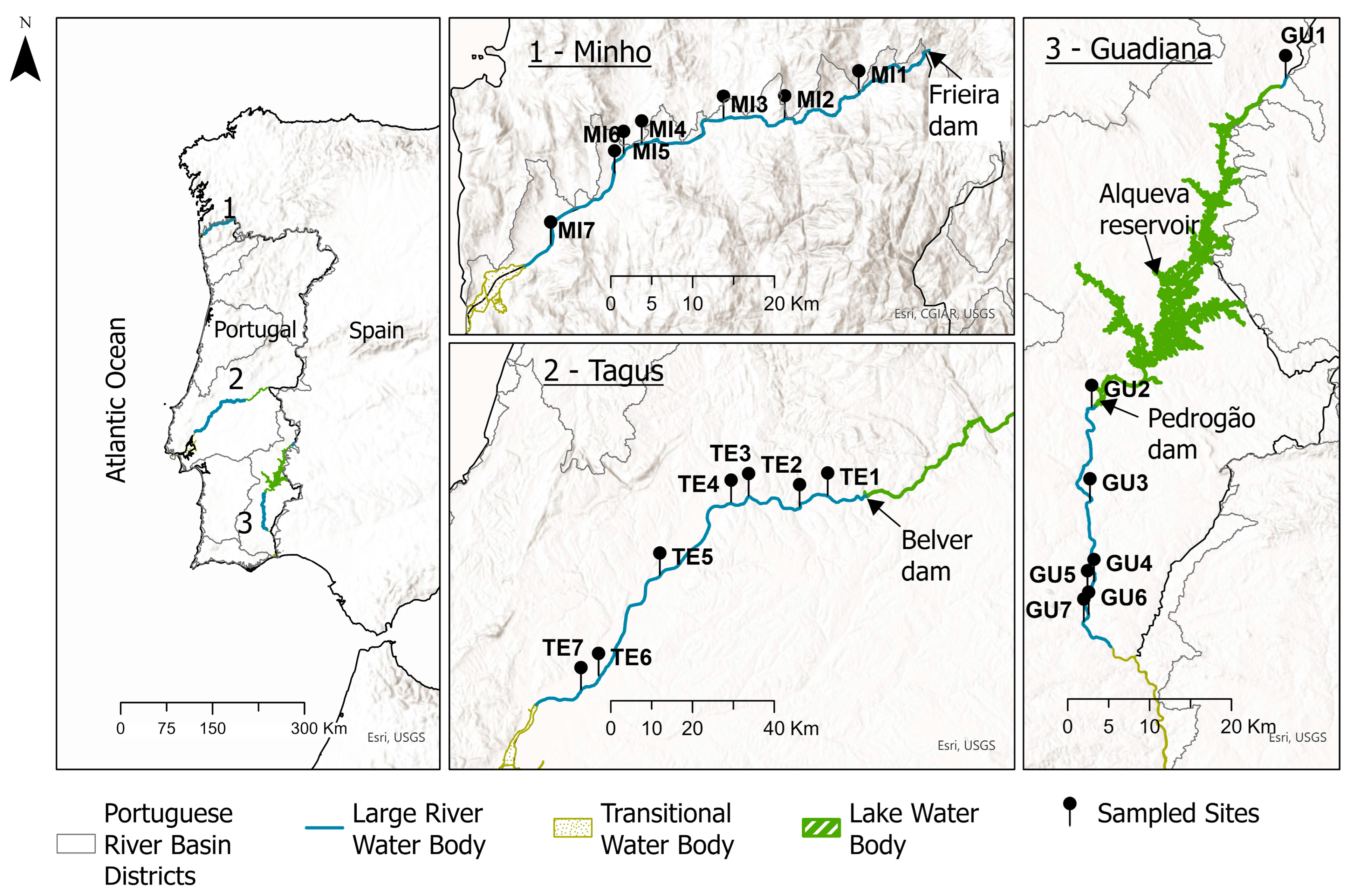
2.1. Study Area and Sampling Sites
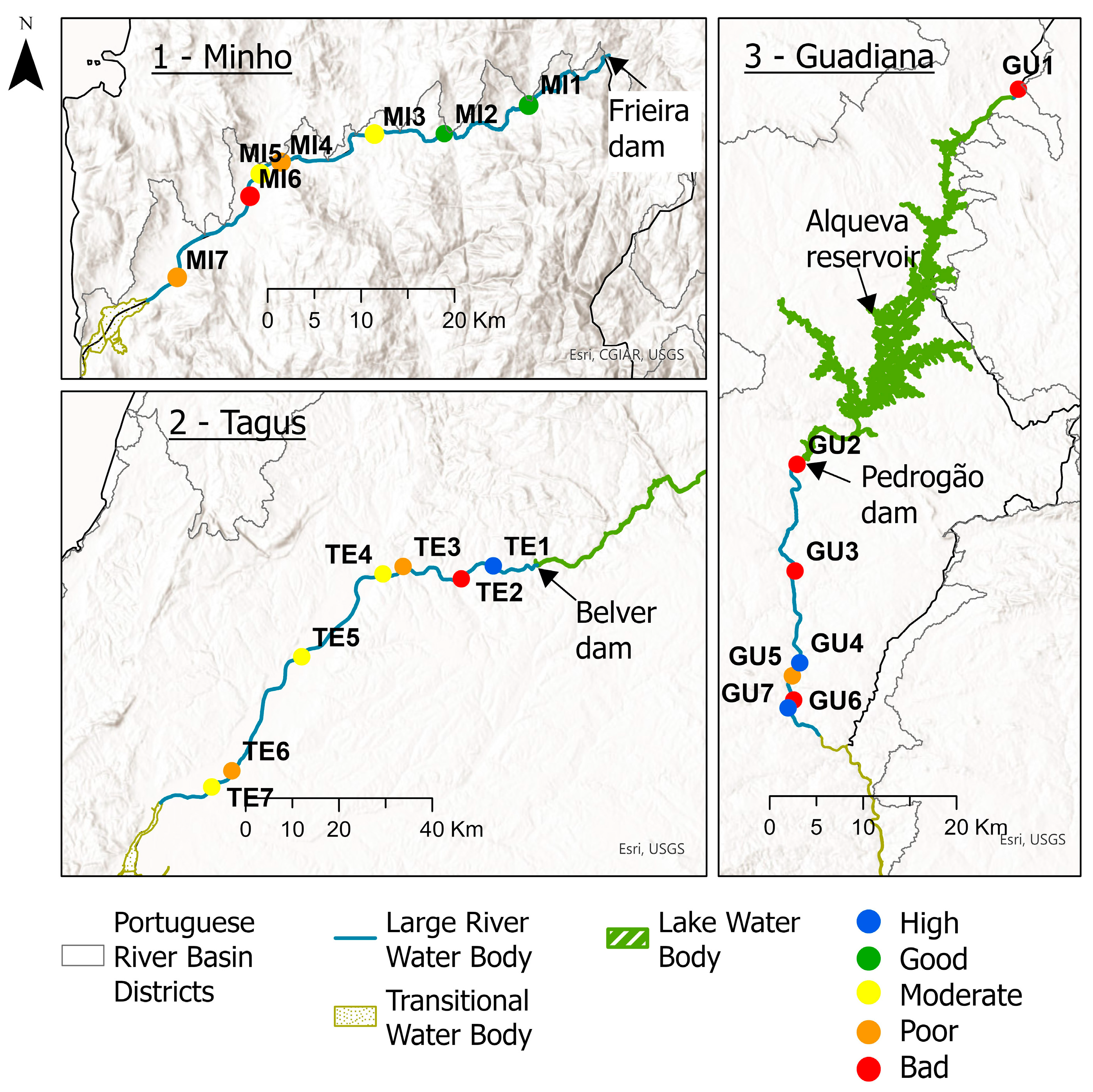
2.2. Anthropogenic Disturbance and Site Classification
2.3. Fish Sampling
2.4. Index Development
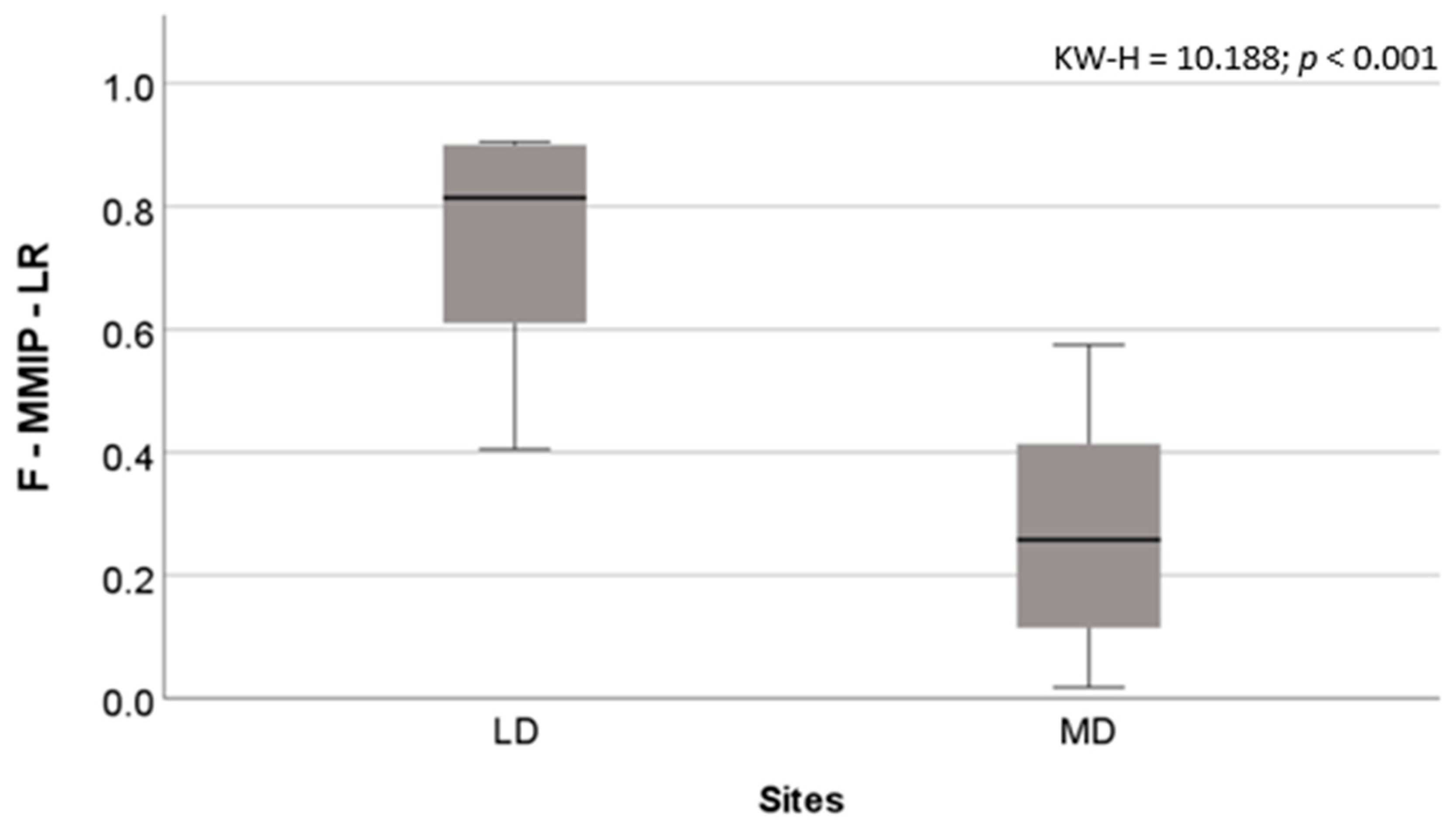
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ward, J.V.; Tockner, K.; Schiemer, F. Biodiversity of floodplain river ecosystems: Ecotones and connectivity. Regul. Rivers Res. Manag. 1999, 15, 125–139. [Google Scholar] [CrossRef]
- De Leeuw, J.J.; Buijse, A.D.; Haidvogl, G.; Lapinska, M.; Noble, R.; Repecka, R.; Virbickas, T.; Wiśniewolski, W.; Wolter, C. Challenges in developing fish-based ecological assessment methods for large floodplain rivers. Fish. Manag. Ecol. 2007, 14, 483–494. [Google Scholar] [CrossRef]
- Tockner, K.; Uehlinger, U.; Robinson, C.T. Rivers of Europe; Elsevier: Amsterdam, The Netherlands, 2009. [Google Scholar]
- Fernandes, M.R.; Aguiar, F.C.; Martins, M.J.; Rivaes, R.; Ferreira, M.T. Long-term human-generated alterations of Tagus river: Effects of hydrological regulation and land-use changes in distinct river zones. CATENA 2020, 188, 104466. [Google Scholar] [CrossRef]
- Anastácio, P.M.; Ribeiro, F.; Capinha, C.; Banha, F.; Gama, M.; Filipe, A.F.; Rebelo, R.; Sousa, R. Non-native freshwater fauna in Portugal: A review. Sci. Total Environ. 2019, 650, 1923–1934. [Google Scholar] [CrossRef]
- Collares-Pereira, M.J.; Alves, M.J.; Ribeiro, F.; Domingos, I.; Almeida, P.R.; Costa, L.; Gante, H.; Filipe, A.F.; Aboim, M.A.; Rodrigues, P.M.; et al. Guia dos Peixes de água doce e Migradores de Portugal Continental; Edições Afrontamento: Porto, Portugal, 2021. [Google Scholar]
- European Commission: Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 Establishing a Framework for the Community Action in the Field of Water Policy; Official Journal of the European Union: Strasbourg, France, 2000.
- Solheim, A.-L.; Globevnik, L.; Austnes, K.; Kristensen, P.; Moe, S.J.; Persson, J.; Phillips, G.; Poikane, S.; Van De Bund, W.; Birk, S. A new broad typology for rivers and lakes in Europe: Development and application for large-scale environmental assessments. Sci. Total Environ. 2019, 697, 134043. [Google Scholar] [CrossRef]
- Radinger, J.; Britton, J.R.; Carlson, S.M.; Magurran, A.E.; Alcaraz-Hernández, J.D.; Almodóvar, A.; Benejam, L.; Fernández-Delgado, C.; Nicola, G.G.; Oliva-Paterna, F.J.; et al. Effective monitoring of freshwater fish. Fish Fish. 2019, 20, 729–747. [Google Scholar] [CrossRef]
- Reyjol, Y.; Argillier, C.; Bonne, W.; Borja, A.; Buijse, A.D.; Cardoso, A.C.; Daufresne, M.; Kernan, M.; Ferreira, M.T.; Poikane, S.; et al. Assessing the ecological status in the context of the European Water Framework Directive: Where do we go now? Sci. Total Environ. 2014, 497–498, 332–344. [Google Scholar] [CrossRef] [PubMed]
- Ruaro, R.; Gubiani, É.A.; Hughes, R.M.; Mormul, R.P. Global trends and challenges in multimetric indices of biological condition. Ecol. Indic. 2020, 110, 105862. [Google Scholar] [CrossRef]
- Vadas, R.L.; Hughes, R.M.; Bae, Y.J.; Baek, M.J.; Gonzáles, O.C.B.; Callisto, M.; Carvalho, D.R.D.; Chen, K.; Ferreira, M.T.; Fierro, P.; et al. Assemblage-based biomonitoring of freshwater ecosystem health via multimetric indices: A critical review and suggestions for improving their applicability. Water Biol. Secur. 2022, 1, 100054. [Google Scholar] [CrossRef]
- Karr, J.R.; Fausch, K.; Angermeier, P.L.; Yant, P.R.; Schlosser, I.J. Assessing Biological Integrity in Running Waters. A Method and Its Rationale; Illinois Natural History Survey Special Publication: Champaign, IL, USA, 1986. [Google Scholar]
- Kesminas, V.; Virbickas, T. Application of an adapted index of biotic integrity to rivers of Lithuania. Assess. Ecol. Integr. Run. Water 2000, 257–270. [Google Scholar] [CrossRef]
- Breine, J.; Simoens, I.; Goethals, P.; Quataert, P.; Ercken, D.; Van Liefferinghe, C.; Belpaire, C. A fish-based index of biotic integrity for upstream brooks in Flanders (Belgium). Hydrobiologia 2004, 522, 133–148. [Google Scholar] [CrossRef]
- Pont, D.; Hugueny, B.; Rogers, C. Development of a fish-based index for the assessment of river health in Europe: The European Fish Index. Fish. Manag. Ecol. 2007, 14, 427–439. [Google Scholar] [CrossRef]
- Magalhães, M.F.; Ramalho, C.E.; Collares-Pereira, M.J. Assessing biotic integrity in a Mediterranean watershed: Development and evaluation of a fish-based index. Fish. Manag. Ecol. 2008, 15, 273–289. [Google Scholar] [CrossRef]
- Aparicio, E.; Carmona-Catot, G.; Moyle, P.B.; García-Berthou, E. Development and evaluation of a fish-based index to assess biological integrity of Mediterranean streams. Aquat. Conserv. Mar. Freshw. Ecosyst. 2011, 21, 324–337. [Google Scholar] [CrossRef]
- Adamczyk, M.; Prus, P.; Buras, P.; Wiśniewolski, W.; Ligięza, J.; Szlakowski, J.; Borzęcka, I.; Parasiewicz, P. Development of a new tool for fish-based river ecological status assessment in Poland (EFI+IBI_PL). Acta Ichthyol. Piscat. 2017, 47, 173–184. [Google Scholar] [CrossRef][Green Version]
- Ramos-Merchante, A.; Prenda, J. Macroinvertebrate taxa richness uncertainty and kick sampling in the establishment of Mediterranean rivers ecological status. Ecol. Indic. 2017, 72, 1–12. [Google Scholar] [CrossRef]
- Seegert, G. Considerations regarding development of index of biotic integrity metrics for large rivers. Environ. Sci. Pol. 2000, 3, 99–106. [Google Scholar] [CrossRef]
- Yoder, C.O.; Kulik, B.H. The development and application of multimetric indices for the assessment of impacts to fish assemblages in large rivers: A review of current science and applications. Can. Water Resour. J. 2003, 28, 301–328. [Google Scholar] [CrossRef]
- Petts, G.E.; Nestler, J.; Kennedy, R. Advancing science for water resources management. Hydrobiologia 2006, 565, 277–288. [Google Scholar] [CrossRef]
- Borgwardt, F.; Leitner, P.; Graf, W.; Birk, S. Ex uno plures–Defining different types of very large rivers in Europe to foster solid aquatic bio-assessment. Ecol. Indic. 2019, 107, 105599. [Google Scholar] [CrossRef]
- Agência Portuguesa do Ambiente: Convenção de Albufeira (Cooperação Luso-Espanhola). Available online: https://apambiente.pt/agua/convencao-de-albufeira-cooperacao-luso-espanhola (accessed on 27 March 2024).
- Feio, M.J.; Ferreira, V. Rios de Portugal: Comunidades, Processos e Alterações; Imprensa da Universidade de Coimbra: Coimbra, Portugal, 2019. [Google Scholar]
- Simon, T.P.; Evans, N.T. Environmental quality assessment using stream fishes. In Methods in Stream Ecology; Elsevier: Amsterdam, The Netherlands, 2017; Volume 39, pp. 319–334. [Google Scholar]
- Stoddard, J.L.; Larsen, D.P.; Hawkins, C.P.; Johnson, R.K.; Norris, R.H. Setting expectations for the ecological condition of streams: The concept of reference condition. Ecol. Appl. 2006, 16, 1267–1276. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, J.M.; Ferreira, M.T.; Morgado, P.; Hughes, R.M.; Teixeira, A.; Cortes, R.M.; Bochechas, J.H. A preliminary fishery quality index for Portuguese streams. N. Am. J. Fish. Manag. 2009, 29, 1466–1478. [Google Scholar] [CrossRef]
- Weigel, B.M.; Dimick, J.J. Development, validation, and application of a macroinvertebrate-based Index of biotic integrity for nonwadeable rivers of Wisconsin. J. N. Am. Benthol. Soc. 2011, 30, 665–679. [Google Scholar] [CrossRef]
- European Environmental Agency: Copernicus Land Monitoring Service. CORINE Land Cover. Available online: https://www.eea.europa.eu/en/datahub/datahubitem-view/a5144888-ee2a-4e5d-a7b0-2bbf21656348 (accessed on 15 October 2022).
- SNIRH–Sistema Nacional de Informação de Recursos Hídricos: Rede Hidrométrica do Sistema Nacional de Informação de Recursos Hídricos da APA (SNIRH). Localização Geográfica, Classificação e Caracterização. Available online: https://snirh.apambiente.pt/ (accessed on 15 October 2022).
- Smith, R.A.; Alexander, R.B.; Wolman, M.G. Water-quality trends in the nation’s rivers. Science 1987, 235, 1607–1615. [Google Scholar] [CrossRef]
- Instituto da Água I. P. Manual para a Avaliação Biológica da Qualidade da Água em Sistemas Fluviais Segundo a Directiva Quadro da Água: Protocolo de Amostragem e Análise para a Fauna Piscícola; Ministério do Ambiente, do Ordenamento do Território e do Desenvolvimento Regional; Instituto da Água: Alfragide, Portugal, 2008. [Google Scholar]
- Whittier, T.R.; Hughes, R.M.; Stoddard, J.L.; Lomnicky, G.A.; Peck, D.V.; Herlihy, A.T. A structured approach for developing indices of biotic integrity: Three examples from streams and rivers in the western USA. Trans. Am. Fish. Soc. 2007, 136, 718–735. [Google Scholar] [CrossRef]
- Krause, J.R.; Bertrand, K.N.; Kafle, A.; Troelstrup, N.H. A fish index of biotic integrity for South Dakota’s Northern glaciated plains ecoregion. Ecol. Indic. 2013, 34, 313–322. [Google Scholar] [CrossRef]
- Gonino, G.; Benedito, E.; Cionek, V.D.M.; Ferreira, M.T.; Oliveira, J.M. A fish-based index of biotic integrity for neotropical rainforest sandy soil streams—Southern Brazil. Water 2020, 12, 1215. [Google Scholar] [CrossRef]
- Noble, R.A.A.; Cowx, I.G.; Goffaux, D.; Kestemont, P. Assessing the health of European rivers using functional ecological guilds of fish communities: Standardising species classification and approaches to metric selection. Fish. Manag. Ecol. 2007, 14, 381–392. [Google Scholar] [CrossRef]
- Oliveira, J.M.; Segurado, P.; Santos, J.M.; Teixeira, A.; Ferreira, M.T.; Cortes, R.V. Modelling stream-fish functional traits in reference conditions: Regional and local environmental correlates. PLoS ONE 2012, 7, e45787. [Google Scholar] [CrossRef]
- Magalhães, M.F.; Amaral, S.D.; Sousa, M.; Alexandre, C.M.; Almeida, P.R.; Alves, M.J.; Cortes, R.; Farrobo, A.; Filipe, A.F.; Franco, A.; et al. Livro Vermelho dos Peixes Dulciaquícolas e Diádromos de Portugal Continental; FCiências.ID & ICNF, I.P.: Lisboa, Portugal, 2023. [Google Scholar]
- Pont, D.; Bady, P.; Logez, M.; Veslot, J. EFI+ Project. Improvement and Spatial Extension of the European Fish Index Deliverable 4.1: Report on the Modelling of Reference Conditions and on the Sensitivity of Candidate Metrics to Anthropogenic Pressures. Deliverable 4.2: Report on the Final Development and Validation of the New European Fish Index and Method, Including a Complete Technical Description of the New Method. 6th Framework Programme Priority FP6-2005-SSP-5-A. N° 0044096. 2009. Available online: https://hal.inrae.fr/hal-02592964/document (accessed on 15 October 2022).
- Hering, D.; Feld, C.K.; Moog, O.; Ofenböck, T. Cook book for the development of a multimetric index for biological condition of aquatic ecosystems: Experiences from the European AQEM and STAR projects and related initiatives. Hydrobiologia 2006, 566, 311–324. [Google Scholar] [CrossRef]
- Emery, E.B.; Simon, T.P.; McCormick, F.H.; Angermeier, P.L.; Deshon, J.E.; Yoder, C.O.; Sanders, R.E.; Pearson, W.D.; Hickman, G.D.; Reash, R.J.; et al. Development of a multimetric index for assessing the biological condition of the Ohio river. Trans. Am. Fish. Soc. 2003, 132, 791–808. [Google Scholar] [CrossRef]
- Lapointe, N.W.R.; Corkum, L.D.; Mandrak, N.E. A comparison of methods for sampling fish diversity in shallow offshore waters of large rivers. N. Am. J. Fish. Manag. 2006, 26, 503–513. [Google Scholar] [CrossRef]
- Hughes, R.M.; Peck, D.V. Acquiring data for large aquatic resource surveys: The art of compromise among science, logistics, and reality. J. N. Am. Benthol. Soc. 2008, 27, 837–859. [Google Scholar] [CrossRef]
- Hughes, R.M.; Kaufmann, P.R.; Herlihy, A.T.; Intelmann, S.S.; Corbett, S.C.; Arbogast, M.C.; Hjort, R.C. Electrofishing distance needed to estimate fish species richness in raftable Oregon rivers. N. Am. J. Fish. Manag. 2002, 22, 1229–1240. [Google Scholar] [CrossRef]
- Hughes, R.M.; Herlihy, A.T.; Peck, D.V. Sampling efforts for estimating fish species richness in western USA river sites. Limnologica 2021, 87, 125859. [Google Scholar] [CrossRef] [PubMed]
- Dunn, C.G.; Paukert, C.P. A flexible survey design for monitoring spatiotemporal fish richness in nonwadeable rivers: Optimizing efficiency by integrating gears. Can. J. Fish. Aquat. Sci. 2020, 77, 978–990. [Google Scholar] [CrossRef]
- Goffaux, D.G. Electrofishing versus gillnet sampling for the assessment of fish assemblages in large rivers. Arch. Hydrobiol. 2005, 162, 73–90. [Google Scholar] [CrossRef]
- Karr, J.R.; Chu, E.W. Sustaining living rivers. Hydrobiologia 2000, 422–423, 1–14. [Google Scholar] [CrossRef]
- Berkman, H.E.; Rabeni, C.F. Effect of siltation on stream fish communities. Environ. Biol. Fishes 1987, 18, 285–294. [Google Scholar] [CrossRef]
- Kemp, P.; Sear, D.; Collins, A.; Naden, P.; Jones, I. The impacts of fine sediment on riverine fish. Hydrol. Process. 2011, 25, 1800–1821. [Google Scholar] [CrossRef]
- Hughes, R.M.; Vadas, R.L. Agricultural effects on streams and rivers: A western USA focus. Water 2021, 13, 1901. [Google Scholar] [CrossRef]
- Kaufmann, P.R.; Hughes, R.M.; Paulsen, S.G.; Peck, D.V.; Seeliger, C.W.; Weber, M.H.; Mitchell, R.M. Physical habitat in conterminous US streams and rivers, Part 1: Geoclimatic controls and anthropogenic alteration. Ecol. Indic. 2022, 141, 109046. [Google Scholar] [CrossRef] [PubMed]
- Herlihy, A.T.; Sifneos, J.C.; Hughes, R.M.; Peck, D.V.; Mitchell, R.M. The relation of lotic fish and benthic macroinvertebrate condition indices to environmental factors across the conterminous USA. Ecol. Indic. 2020, 112, 105958. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Lyons, J.; Kanehl, P.; Gatti, R. Influences of watershed land use on habitat quality and biotic integrity in Wisconsin streams. Fisheries 1997, 22, 6–12. [Google Scholar] [CrossRef]
- Allan, J.D. Landscapes and riverscapes: The influence of land use on stream ecosystems. Annu. Rev. Ecol. Evol. Syst. 2004, 35, 257–284. [Google Scholar] [CrossRef]
- Hermoso, V.; Clavero, M.; Blanco-Garrido, F.; Prenda, J. Invasive species and habitat degradation in Iberian streams: An analysis of their role in freshwater fish diversity loss. Ecol. Appl. 2011, 21, 175–188. [Google Scholar] [CrossRef] [PubMed]
- Fierro, P.; Bertrán, C.; Tapia, J.; Hauenstein, E.; Peña-Cortés, F.; Vergara, C.; Cerna, C.; Vargas-Chacoff, L. Effects of local land-use on riparian vegetation, water quality, and the functional organization of macroinvertebrate assemblages. Sci. Total Environ. 2017, 609, 724–734. [Google Scholar] [CrossRef] [PubMed]
- Gregory, S.V.; Swanson, F.J.; McKee, W.A.; Cummins, K.W. An ecosystem perspective of riparian zones. BioScience 1991, 41, 540–551. [Google Scholar] [CrossRef]
- Naiman, R.J.; Decamps, H.; Pollock, M. The role of riparian corridors in maintaining regional biodiversity. Ecol. Appl. 1993, 3, 209–212. [Google Scholar] [CrossRef]
- Pusey, B.J.; Arthington, A.H. Importance of the riparian zone to the conservation and management of freshwater fish: A review. Mar. Freshw. Res. 2003, 54, 1. [Google Scholar] [CrossRef]
- Martelo, J.; Da Costa, L.; Ribeiro, D.; Gago, J.; Magalhães, M.; Gante, H.; Alves, M.; Cheoo, G.; Gkenas, C.; Banha, F.; et al. Evaluating the range expansion of recreational non-native fishes in Portuguese freshwaters using scientific and citizen science data. BioInvasions Rec. 2021, 10, 378–389. [Google Scholar] [CrossRef]
- Kennard, M.J.; Arthington, A.H.; Pusey, B.J.; Harch, B.D. Are alien fish a reliable indicator of river health? Freshw. Biol. 2005, 50, 174–193. [Google Scholar] [CrossRef]
- Ferreira, T.; Caiola, N.; Casals, F.; Oliveira, J.M.; De Sostoa, A. Assessing perturbation of river fish communities in the Iberian ecoregion. Fish. Manag. Ecol. 2007, 14, 519–530. [Google Scholar] [CrossRef]
- Ramos-Merchante, A.; Prenda, J. The ecological and conservation status of the Guadalquivir river basin (s Spain) through the application of a fish-based multimetric index. Ecol. Indic. 2018, 84, 45–59. [Google Scholar] [CrossRef]
- Hermoso, V.; Clavero, M. Revisiting ecological integrity 30 years later: Non-native species and the misdiagnosis of freshwater ecosystem health. Fish Fish. 2013, 14, 416–423. [Google Scholar] [CrossRef]
- Aparicio, E.; Alcaraz, C.; Rocaspana, R.; Pou-Rovira, Q.; García-Berthou, E. Adaptation of the European Fish Index (EFI+) to include the alien fish pressure. Fishes 2023, 9, 13. [Google Scholar] [CrossRef]
- Jungwirth, M.; Muhar, S.; Schmutz, S. Fundamentals of fish ecological integrity and their relation to the extended serial discontinuity concept. Assess. Ecol. Integr. Run. Waters 2000, 85–97. [Google Scholar] [CrossRef]
- McDowall, R.M.; Taylor, M.J. Environmental indicators of habitat quality in a migratory freshwater fish fauna. Environ. Manag. 2000, 25, 357–374. [Google Scholar] [CrossRef] [PubMed]
- Duarte, G.; Moreira, M.; Branco, P.; Da Costa, L.; Ferreira, M.T.; Segurado, P. One millennium of historical freshwater fish occurrence data for Portuguese rivers and streams. Sci. Data 2018, 5, 180163. [Google Scholar] [CrossRef]
- Duarte, G.; Branco, P.; Haidvogl, G.; Ferreira, M.T.; Pont, D.; Segurado, P. iPODfish–A new method to infer the historical occurrence of diadromous fish species along river networks. Sci. Total Environ. 2022, 812, 152437. [Google Scholar] [CrossRef]
- Lyons, J.; Piette, R.R.; Niermeyer, K.W. Development, validation, and application of a fish-based index of biotic integrity for Wisconsin’s large warmwater rivers. Trans. Am. Fish. Soc. 2001, 130, 1077–1094. [Google Scholar] [CrossRef]
- Turgeon, K.; Turpin, C.; Gregory-Eaves, I. Dams have varying impacts on fish communities across latitudes: A quantitative synthesis. Ecol. Lett. 2019, 22, 1501–1516. [Google Scholar] [CrossRef] [PubMed]
- Bernardo, J.M.; Ilhéu, M.; Matono, P.; Costa, A.M. Interannual variation of fish assemblage structure in a Mediterranean river: Implications of streamflow on the dominance of native or exotic species. River Res. Appl. 2003, 19, 521–532. [Google Scholar] [CrossRef]
- Propst, D.L.; Gido, K.B. Responses of native and nonnative fishes to natural flow regime mimicry in the San Juan river. Trans. Am. Fish. Soc. 2004, 133, 922–931. [Google Scholar] [CrossRef]
- Yoder, C.O.; Rankin, E.T. Biological response signatures and the area of degradation value: New tools for interpreting multimetric data. In Biological Assessment and Criteria: Tools for Water Resources Planning and Decision Making; Davis, W.S., Simon, T.P., Eds.; Lewis Publishers: Boca Raton, FL, USA, 1995. [Google Scholar]
- Hughes, R.M.; Kaufmann, P.R.; Herlihy, A.T.; Kincaid, T.M.; Reynolds, L.; Larsen, D.P. A process for developing and evaluating indices of fish assemblage integrity. Can. J. Fish. Aquat. Sci. 1998, 55, 1618–1631. [Google Scholar] [CrossRef]
- Sabater, S.; Elosegi, A.; Ludwig, R. Multiple Stressors in River Ecosystems; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar]


| Class | Agricultural Land Use | Artificial Land Use | TP (mg/L) | TN (mg/L) | Channel Morphology/Riparian Disturbance | Flow Regulation |
|---|---|---|---|---|---|---|
| 1 | <10% agriculture, and <3% intensive farming | <5% | <0.13 | <1.0 | No or minor impacts | Infrequent or no hydropeaking |
| 2 | 10–30% agriculture, and <10% intensive farming | 5–15% | 0.14–0.26 | 1.0–2.0 | Most of natural channel form maintained, and >70% of the streambank vegetation in natural state | Regular hydropeaking and distance > 30 km from a large hydroelectric power plant (LHPP) |
| 3 | 31–70% agriculture and <15% intensive farming | 16–25% | 0.27–0.39 | 2.1–3.0 | Channelized (some natural habitat types missing), and/or 50–70% of the streambank vegetation in natural state | Regular hydropeaking and distance < 30 km from an LHPP |
| 4 | >70% agriculture and/or >15% intensive farming | >25% | >0.39 | >3.0 | Strongly channelized (most natural habitat types missing), and/or <50% of the streambank vegetation in natural state | Regular hydropeaking and marked seasonal dewatering of the river |
| Family | Species | Basin | FO | Guilds |
|---|---|---|---|---|
| Anguillidae | Anguilla anguilla | M; T; G | 61.9% | THRE, TOLE, OMNI, BENT, DIAD |
| Atherinidae | Atherina boyeri | M; T; G | 38.1% | NOTO, INVE, PELA, GENE, DIAD |
| Centrarchidae | Lepomis gibbosus | M; T; G | 76.2% | ALIE, TOLE, GENE |
| Micropterus salmoides | M; T; G | 52.4% | ALIE, TOLE, GENE | |
| Cichlidae | Australoheros facetus | G | 28.6% | ALIE, TOLE, GENE |
| Clupeidae | Alosa alosa | M; T; G | 9.5% | THRE, NOTO, PELA, LITH, DIAD |
| Alosa fallax | M; T; G | 4.8% | THRE, NOTO, PELA, LITH, DIAD | |
| Cobitidae | Cobitis paludica | T; G | 23.8% | FNAT, TOLE, INVE, BENT, GENE |
| Cyprinidae | Carassius auratus | M; T; G | 47.6% | ALIE, TOLE, BENT |
| Carassius gibelio | T; G | 4.8% | ALIE, TOLE, BENT | |
| Cyprinus carpio | M; T; G | 42.9% | ALIE, TOLE, BENT | |
| Luciobarbus bocagei | M; T | 47.6% | FNAT, TOLE, OMNI, BENT, LITH, POTA | |
| Luciobarbus comizo | T; G | 19.0% | FNAT, TOLE, OMNI, BENT, LITH, POTA | |
| Luciobarbus microcephalus | G | 9.5% | FNAT, THRE, TOLE, OMNI, BENT, LITH, POTA | |
| Luciobarbus sclateri | G | 28.6% | FNAT, TOLE, OMNI, BENT, LITH, POTA | |
| Luciobarbus steindachneri | T; G | 9.5% | FNAT, TOLE, OMNI, BENT, LITH, POTA | |
| Gasterosteidae | Gasterosteus aculeatus | M; T | 9.5% | FNAT, THRE, NOTO, OMNI, PELA |
| Gobiidae | Pomatoschistus microps | M; T; G | 4.8% | NOTO, OMNI, BENT, GENE |
| Gobionidae | Gobio lozanoi | M; T | 38.1% | ALIE, TOLE, BENT |
| Ictaluridae | Ameirus melas | T; G | 9.5% | ALIE, TOLE, BENT |
| Ictalurus punctatus | G | 4.8% | ALIE, TOLE, BENT | |
| Leuciscidae | Achondrostoma oligolepis | M; T | 14.3% | FNAT, TOLE, OMNI, PELA, GENE |
| Alburnus alburnus | T; G | 42.9% | ALIE, TOLE, OMNI | |
| Pseudochondrostoma duriense | M | 28.6% | FNAT, NOTO, OMNI, BENT, LITH, POTA | |
| Pseudochondrostoma polylepis | T | 14.3% | FNAT, NOTO, OMNI, BENT, LITH, POTA | |
| Pseudochondrostoma willkommii | G | 4.8% | FNAT, THRE, NOTO, OMNI, BENT, LITH, POTA | |
| Squalius carolitertii | M | 14.3% | FNAT, NOTO, INVE, PELA, LITH | |
| Squalius pyrenaicus | T; G | 4.8% | FNAT, THRE, NOTO, INVE, PELA, LITH | |
| Moronidae | Dicentrarchus labrax | M; T; G | 4.8% | NOTO, INVE, PELA |
| Mugilidae | Liza ramada | M; T; G | 23.8% | TOLE, OMNI, PELA, DIAD |
| Mugil cephalus | M; T; G | 9.5% | TOLE, OMNI, PELA, DIAD | |
| Percidae | Sander lucioperca | T; G | 23.8% | ALIE, TOLE, GENE |
| Petromyzontidae | Petromyzon marinus | M; T; G | 4.8% | THRE, NOTO, BENT, LITH, DIAD |
| Pleuronectidae | Platichthys flesus | M; T; G | 9.5% | NOTO, INVE, BENT, DIAD |
| Poecilidae | Gambusia holbrooki | M; T; G | 52.4% | ALIE, TOLE |
| Salmonidae | Salmo trutta | M; T | 9.5% | FNAT, NOTO, INVE, PELA, LITH, POTA |
| Siluridae | Silurus glanis | T | 28.6% | ALIE, TOLE, BENT, GENE |
| Metrics | P5 | P95 |
|---|---|---|
| % of Lithophilic individuals | 1 | 73 |
| % of Migrant individuals | 11 | 94 |
| % of Alien individuals | 2 | 88 |
| % of Native freshwater individuals | 4 | 73 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Faro, A.T.; Ferreira, M.T.; Oliveira, J.M. A Fish-Based Tool for the Quality Assessment of Portuguese Large Rivers. Fishes 2024, 9, 149. https://doi.org/10.3390/fishes9050149
Faro AT, Ferreira MT, Oliveira JM. A Fish-Based Tool for the Quality Assessment of Portuguese Large Rivers. Fishes. 2024; 9(5):149. https://doi.org/10.3390/fishes9050149
Chicago/Turabian StyleFaro, António Tovar, Maria Teresa Ferreira, and João Manuel Oliveira. 2024. "A Fish-Based Tool for the Quality Assessment of Portuguese Large Rivers" Fishes 9, no. 5: 149. https://doi.org/10.3390/fishes9050149
APA StyleFaro, A. T., Ferreira, M. T., & Oliveira, J. M. (2024). A Fish-Based Tool for the Quality Assessment of Portuguese Large Rivers. Fishes, 9(5), 149. https://doi.org/10.3390/fishes9050149








