Productivity of Fish and Crop Growth and Characteristics of Bacterial Communities in the FLOCponics System
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Design of BFT and Coupled FP Systems
2.2. Measurements of Water Properties
2.3. DNA Extraction, PCR Amplification, and NGS Sequencing
2.4. Bioinformatic Analysis
3. Results
3.1. Growth of Fish and Crops
3.2. Physicochemical Properties of Water in BFT and FP Systems
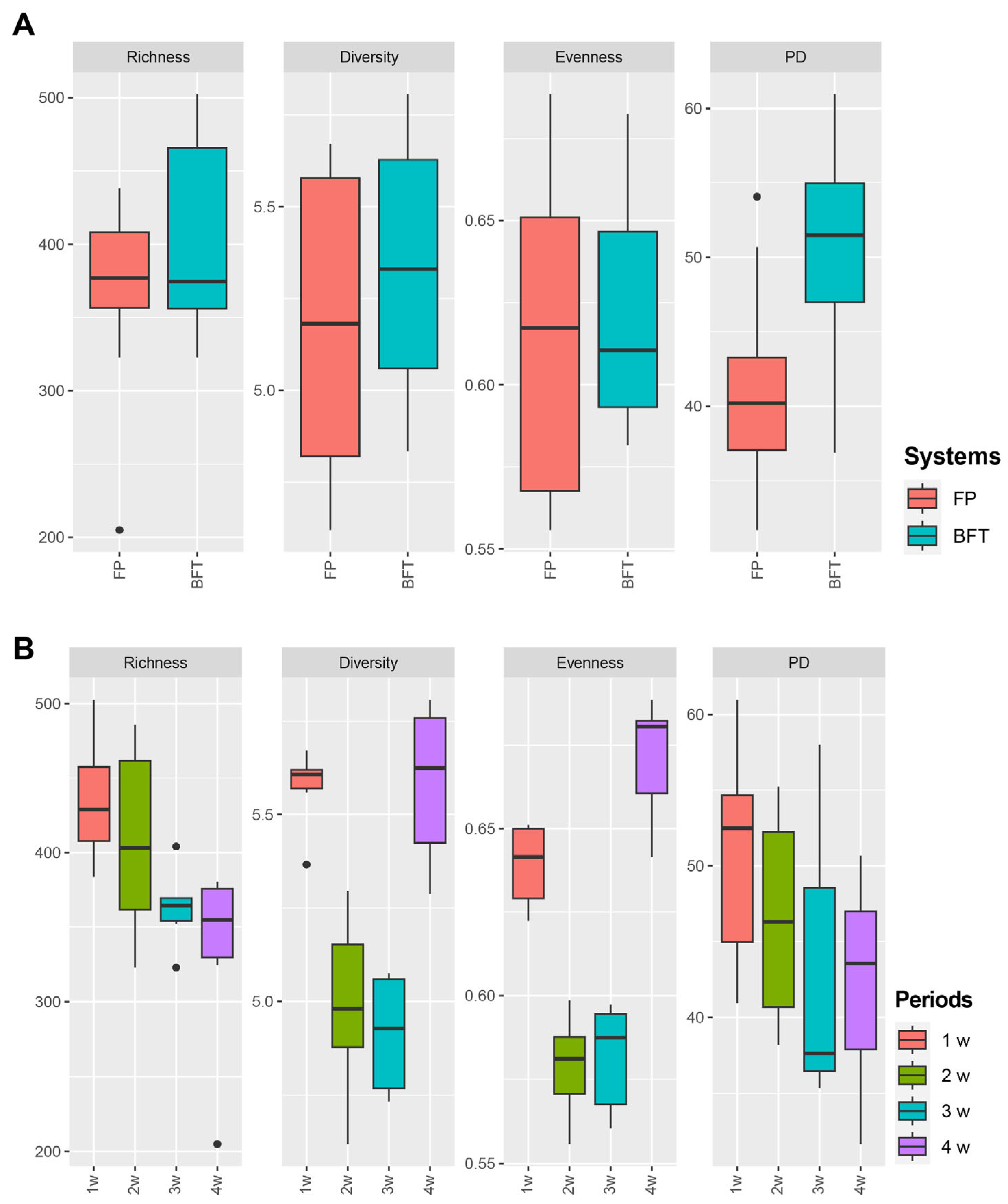
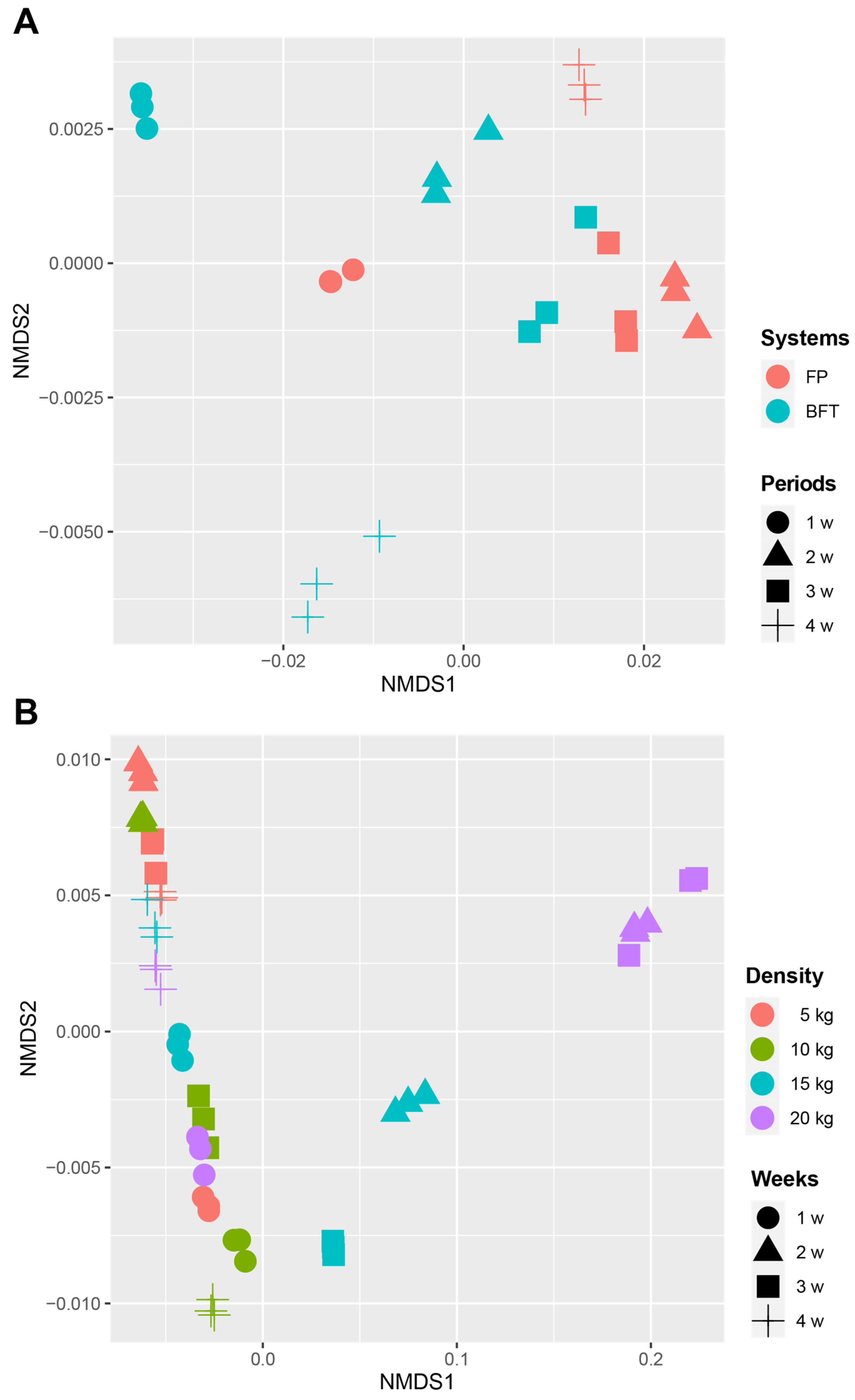
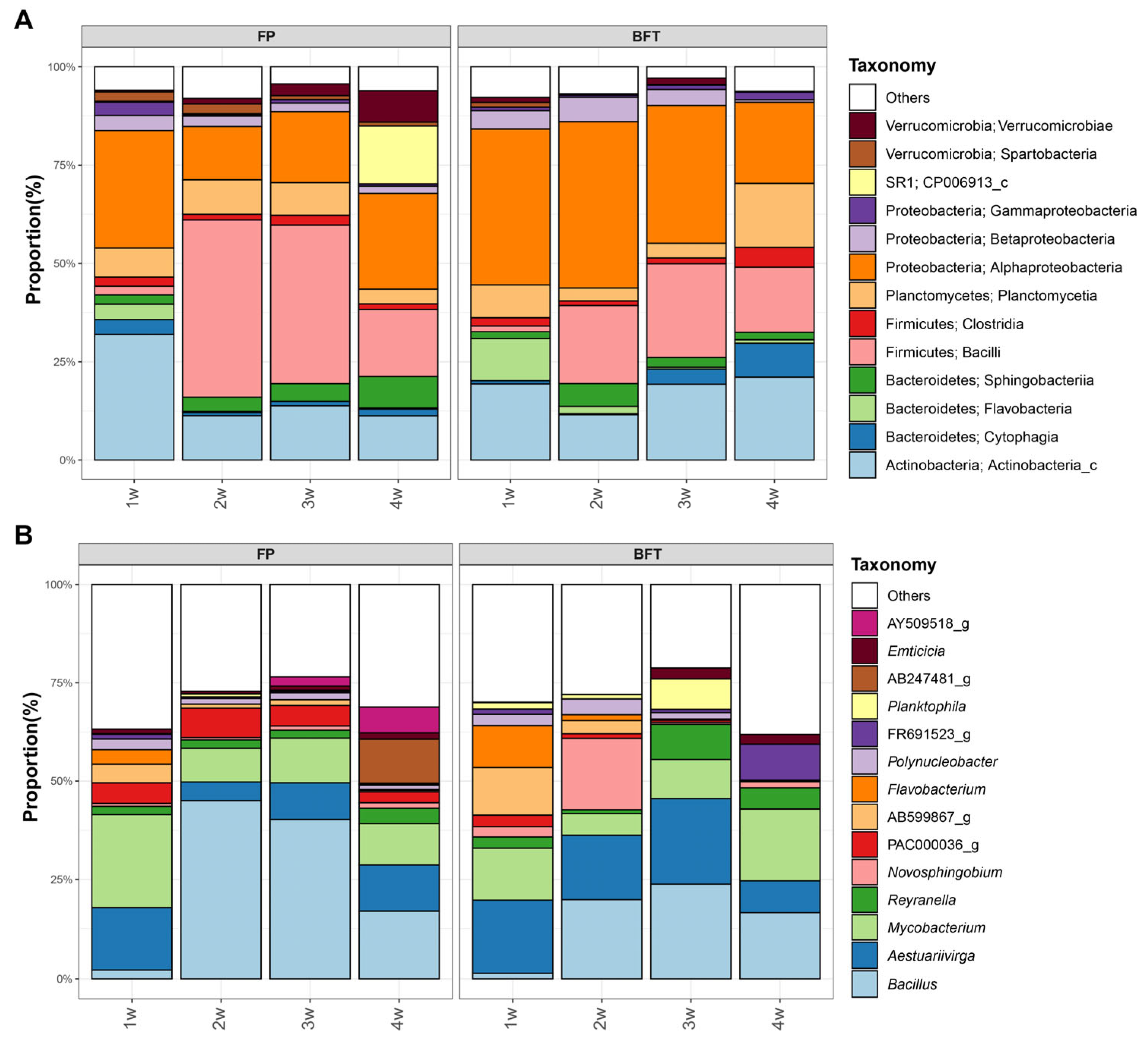
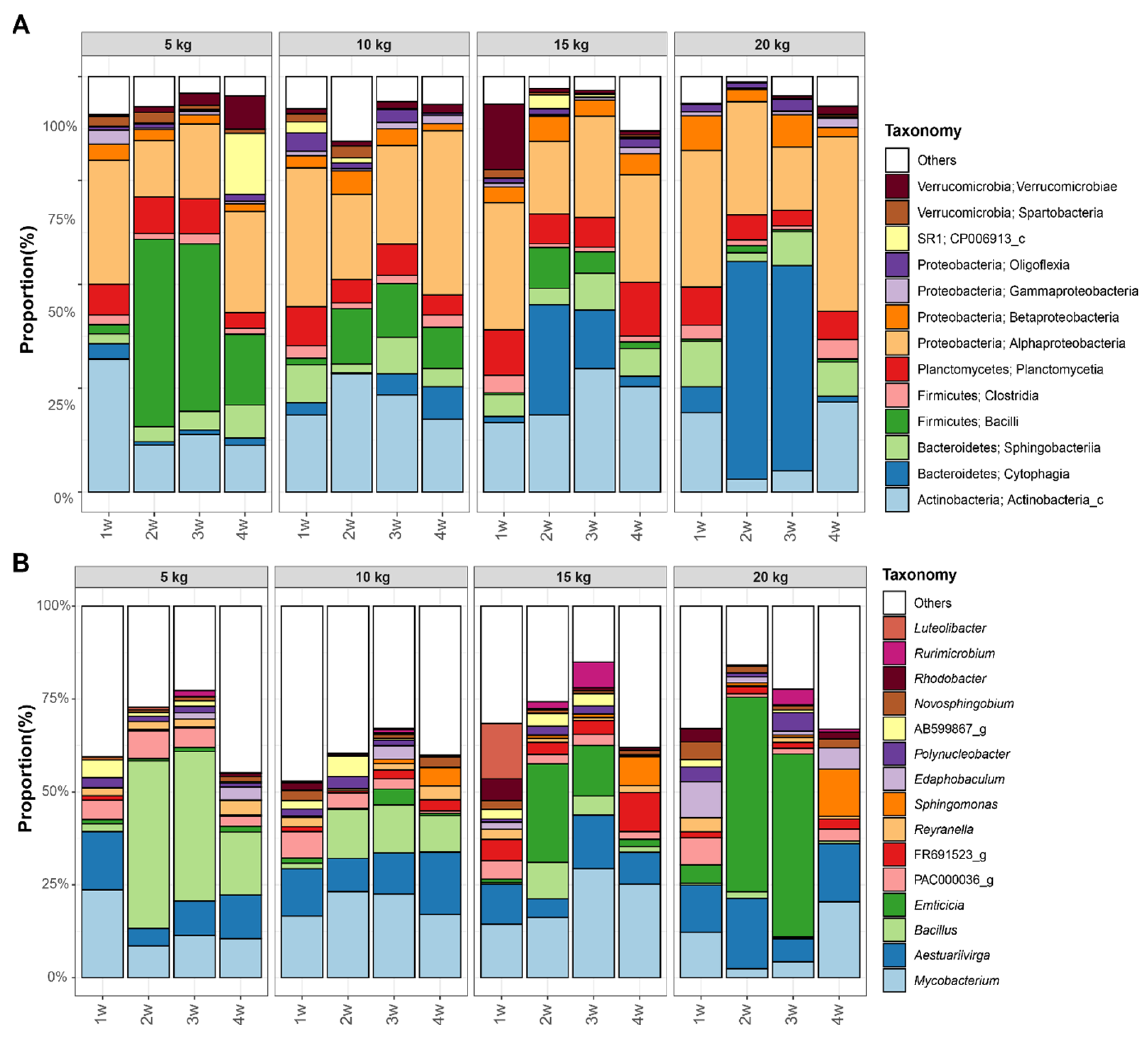
3.3. Difference of Bacterial Communities between FP and BFT Systems
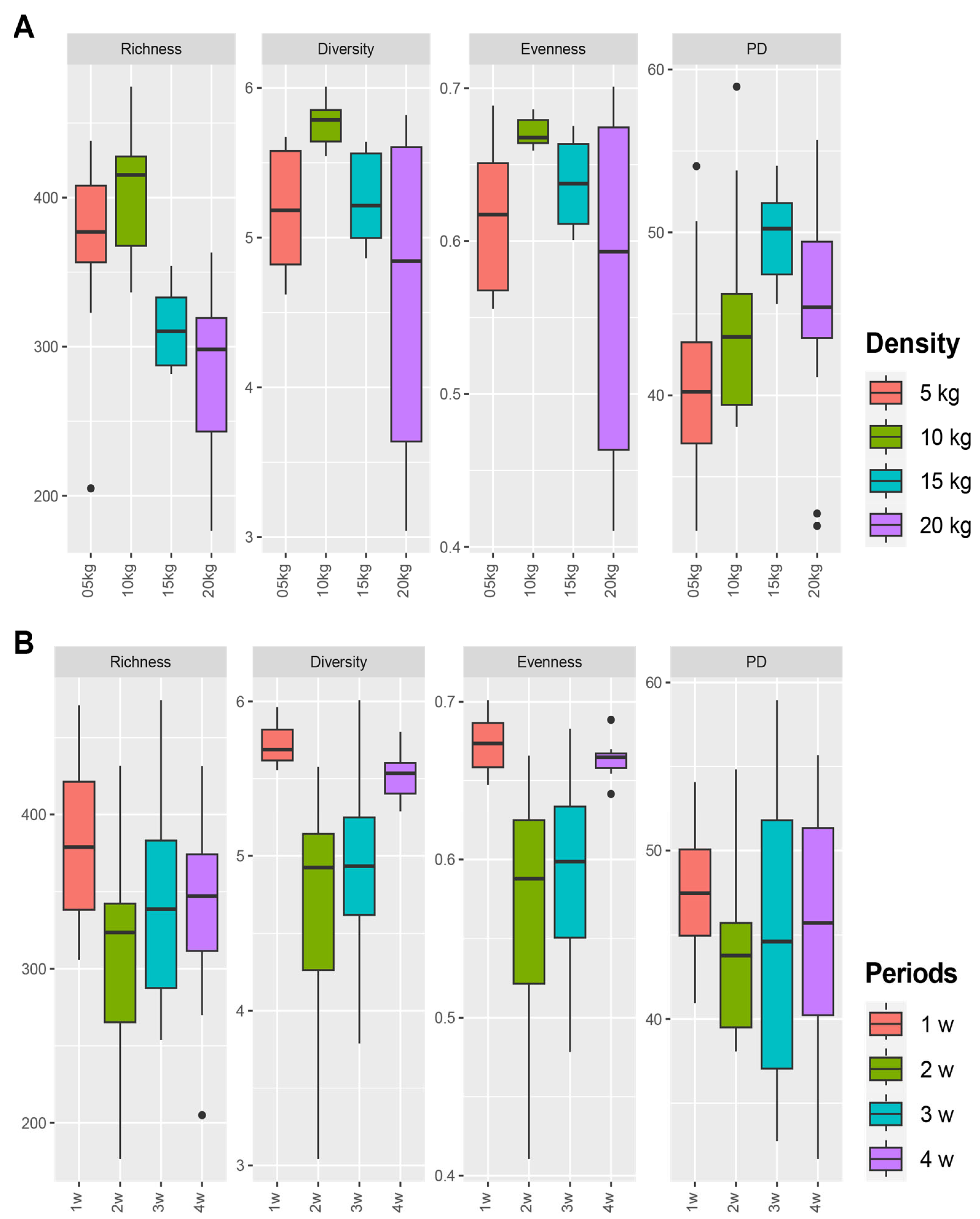
3.4. Difference of Bacterial Communities between Fish Density in FP Systems
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Boyd, C.E.; McNevin, A.A.; Clay, J.; Johnson, H.M. Certification Issues for Some Common Aquaculture Species. Rev. Fish. Sci. 2005, 13, 231–279. [Google Scholar] [CrossRef]
- Ciji, A.; Akhtar, M.S. Nitrite Implications and Its Management Strategies in Aquaculture: A Review. Rev. Aquac. 2020, 12, 878–908. [Google Scholar] [CrossRef]
- Baganz, G.F.M.; Junge, R.; Portella, M.C.; Goddek, S.; Keesman, K.J.; Baganz, D.; Staaks, G.; Shaw, C.; Lohrberg, F.; Kloas, W. The Aquaponic Principle—It Is All about Coupling. Rev. Aquac. 2022, 14, 252–264. [Google Scholar] [CrossRef]
- Pinho, S.M.; de Lima, J.P.; David, L.H.; Emerenciano, M.G.C.; Goddek, S.; Verdegem, M.C.J.; Keesman, K.J.; Portella, M.C. FLOCponics: The Integration of Biofloc Technology with Plant Production. Rev. Aquac. 2022, 14, 647–675. [Google Scholar] [CrossRef]
- Martinez-Cordova, L.R.; López-Elías, J.; Martinez-Porchas, M.; Bringas Burgos, B.; Naranjo-Paramo, J. A Preliminary Evaluation of an Integrated Aquaculture-Agriculture Systems (Tilapia and Peppers) at Mesocosm Scale. J. Aquac. Mar. Biol. 2020, 9, 19–22. [Google Scholar] [CrossRef]
- Pinheiro, I.; Arantes, R.; do Espírito Santo, C.M.; do Nascimento Vieira, F.; Lapa, K.R.; Gonzaga, L.V.; Fett, R.; Barcelos-Oliveira, J.L.; Seiffert, W.Q. Production of the Halophyte Sarcocornia Ambigua and Pacific White Shrimp in an Aquaponic System with Biofloc Technology. Ecol. Eng. 2017, 100, 261–267. [Google Scholar] [CrossRef]
- Pinho, S.M.; Molinari, D.; de Mello, G.L.; Fitzsimmons, K.M.; Coelho Emerenciano, M.G. Effluent from a Biofloc Technology (BFT) Tilapia Culture on the Aquaponics Production of Different Lettuce Varieties. Ecol. Eng. 2017, 103, 146–153. [Google Scholar] [CrossRef]
- Pinho, S.M.; David, L.H.C.; Goddek, S.; Emerenciano, M.G.C.; Portella, M.C. Integrated Production of Nile Tilapia Juveniles and Lettuce Using Biofloc Technology. Aquac. Int. 2021, 29, 37–56. [Google Scholar] [CrossRef]
- Poli, M.A.; Legarda, E.C.; de Lorenzo, M.A.; Pinheiro, I.; Martins, M.A.; Seiffert, W.Q.; do Nascimento Vieira, F. Integrated Multitrophic Aquaculture Applied to Shrimp Rearing in a Biofloc System. Aquaculture 2019, 511, 734274. [Google Scholar] [CrossRef]
- Da Rocha, A.F.; Filho, M.L.B.; Stech, M.R.; da Silva, R.P. Lettuce production in aquaponic and biofloc systems with silver catfish Rhamdia quelen. Bol. Inst. Pesca 2017, 43, 64–73. [Google Scholar] [CrossRef]
- Pinho, S.M.; de Lima, J.P.; Tarigan, N.B.; David, L.H.; Portella, M.C.; Keesman, K.J. Modelling FLOCponics Systems: Towards Improved Water and Nitrogen Use Efficiency in Biofloc-Based Fish Culture. Biosyst. Eng. 2023, 229, 96–115. [Google Scholar] [CrossRef]
- Dauda, A.B. Biofloc Technology: A Review on the Microbial Interactions, Operational Parameters and Implications to Disease and Health Management of Cultured Aquatic Animals. Rev. Aquac. 2020, 12, 1193–1210. [Google Scholar] [CrossRef]
- Faizullah, M.M.; Rajagopalsamy, C.; Ahilan, B.; Daniel, N. Application of Biofloc Technology (BFT) in the Aquaculture System. J. Entomol. Zool. Stud. 2019, 7, 204–212. [Google Scholar]
- Balcázar, J.L.; de Blas, I.; Ruiz-Zarzuela, I.; Cunningham, D.; Vendrell, D.; Múzquiz, J.L. The Role of Probiotics in Aquaculture. Vet. Microbiol. 2006, 114, 173–186. [Google Scholar] [CrossRef] [PubMed]
- El-Saadony, M.T.; Alagawany, M.; Patra, A.K.; Kar, I.; Tiwari, R.; Dawood, M.A.O.; Dhama, K.; Abdel-Latif, H.M.R. The Functionality of Probiotics in Aquaculture: An Overview. Fish Shellfish Immunol. 2021, 117, 36–52. [Google Scholar] [CrossRef]
- Eck, M.; Sare, A.R.; Massart, S.; Schmautz, Z.; Junge, R.; Smits, T.H.M.; Jijakli, M.H. Exploring Bacterial Communities in Aquaponic Systems. Water 2019, 11, 260. [Google Scholar] [CrossRef]
- Choi, J.Y.; Park, J.S.; Kim, H.; Hwang, J.; Lee, D.; Lee, J.-H. Assessment of Water Quality Parameters During a Course of Applying Biofloc Technology (BFT). J. Fishries Mar. Sci. Educ. 2020, 32, 1632–1638. [Google Scholar] [CrossRef]
- National Institute of Fisheries Science (NIFS). Standard Manual of Eel (Anguilla Japonica) Aquaculture; National Institute of Fisheries Science (NIFS): Busan, Republic of Korea, 2009. [Google Scholar]
- Klindworth, A.; Pruesse, E.; Schweer, T.; Peplies, J.; Quast, C.; Horn, M.; Glöckner, F.O. Evaluation of General 16S Ribosomal RNA Gene PCR Primers for Classical and Next-Generation Sequencing-Based Diversity Studies. Nucleic Acids Res. 2013, 41, e1. [Google Scholar] [CrossRef]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.C.; Al-Ghalith, G.A.; Alexander, H.; Alm, E.J.; Arumugam, M.; Asnicar, F.; et al. Reproducible, Interactive, Scalable and Extensible Microbiome Data Science Using QIIME 2. Nat. Biotechnol. 2019, 37, 852–857. [Google Scholar] [CrossRef]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-Resolution Sample Inference from Illumina Amplicon Data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef]
- Bokulich, N.A.; Kaehler, B.D.; Rideout, J.R.; Dillon, M.; Bolyen, E.; Knight, R.; Huttley, G.A.; Caporaso, J.G. Optimizing Taxonomic Classification of Marker-Gene Amplicon Sequences with QIIME 2’s Q2-Feature-Classifier Plugin. Microbiome 2018, 6, 90. [Google Scholar] [CrossRef]
- Yoon, S.-H.; Ha, S.-M.; Kwon, S.; Lim, J.; Kim, Y.; Seo, H.; Chun, J. Introducing EzBioCloud: A Taxonomically United Database of 16S RRNA Gene Sequences and Whole-Genome Assemblies. Int. J. Syst. Evol. Microbiol. 2017, 67, 1613–1617. [Google Scholar] [CrossRef]
- Price, M.N.; Dehal, P.S.; Arkin, A.P. FastTree: Computing Large Minimum Evolution Trees with Profiles Instead of a Distance Matrix. Mol. Biol. Evol. 2009, 26, 1641–1650. [Google Scholar] [CrossRef]
- Katoh, K.; Standley, D.M. MAFFT Multiple Sequence Alignment Software Version 7: Improvements in Performance and Usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef] [PubMed]
- McMurdie, P.J.; Holmes, S. Phyloseq: An R Package for Reproducible Interactive Analysis and Graphics of Microbiome Census Data. PLoS ONE 2013, 8, e61217. [Google Scholar] [CrossRef] [PubMed]
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; et al. Vegan: Community Ecology Package; Version 2.6-4; R Development Core Team: Vienna, Austria, 2022; Available online: https://CRAN.R-project.org/package=vegan (accessed on 20 March 2023).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021. [Google Scholar]
- Pickens, J.M.; Danaher, J.J.; Sibley, J.L.; Chappell, J.A.; Hanson, T.R. Integrating Greenhouse Cherry Tomato Production with Biofloc Tilapia Production. Horticulturae 2020, 6, 44. [Google Scholar] [CrossRef]
- Fimbres-Acedo, Y.E.; Servín-Villegas, R.; Garza-Torres, R.; Endo, M.; Fitzsimmons, K.M.; Emerenciano, M.G.C.; Magallón-Servín, P.; López-Vela, M.; Magallón-Barajas, F.J. Hydroponic Horticulture Using Residual Waters from Oreochromis Niloticus Aquaculture with Biofloc Technology in Photoautotrophic Conditions with Chlorella Microalgae. Aquac. Res. 2020, 51, 4340–4360. [Google Scholar] [CrossRef]
- Avnimelech, Y. Carbon/Nitrogen Ratio as a Control Element in Aquaculture Systems. Aquaculture 1999, 176, 227–235. [Google Scholar] [CrossRef]
- Ebeling, J.M.; Timmons, M.B.; Bisogni, J.J. Engineering Analysis of the Stoichiometry of Photoautotrophic, Autotrophic, and Heterotrophic Removal of Ammonia–Nitrogen in Aquaculture Systems. Aquaculture 2006, 257, 346–358. [Google Scholar] [CrossRef]
- Hu, Z.; Lee, J.W.; Chandran, K.; Kim, S.; Brotto, A.C.; Khanal, S.K. Effect of Plant Species on Nitrogen Recovery in Aquaponics. Bioresour. Technol. 2015, 188, 92–98. [Google Scholar] [CrossRef]
- Schmautz, Z.; Graber, A.; Jaenicke, S.; Goesmann, A.; Junge, R.; Smits, T.H.M. Microbial Diversity in Different Compartments of an Aquaponics System. Arch. Microbiol. 2017, 199, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Radhakrishnan, R.; Hashem, A.; Abd_Allah, E.F. Bacillus: A Biological Tool for Crop Improvement through Bio-Molecular Changes in Adverse Environments. Front. Physiol. 2017, 8, 667. [Google Scholar] [CrossRef] [PubMed]
- Hlordzi, V.; Kuebutornye, F.K.A.; Afriyie, G.; Abarike, E.D.; Lu, Y.; Chi, S.; Anokyewaa, M.A. The Use of Bacillus Species in Maintenance of Water Quality in Aquaculture: A Review. Aquac. Rep. 2020, 18, 100503. [Google Scholar] [CrossRef]
- Kalu, C.M.; Ogola, H.J.O.; Selvarajan, R.; Tekere, M.; Ntushelo, K. Correlations Between Root Metabolomics and Bacterial Community Structures in the Phragmites Australis Under Acid Mine Drainage-Polluted Wetland Ecosystem. Curr. Microbiol. 2021, 79, 34. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.-M.; Cai, C.-Y.; Sheu, D.-S.; Tsai, J.-M.; Sheu, S.-Y. Novosphingobium ovatum Sp. Nov., Isolated from a Freshwater Mesocosm. Int. J. Syst. Evol. Microbiol. 2020, 70, 5243–5254. [Google Scholar] [CrossRef]
- Li, X.; Salam, N.; Li, J.-L.; Chen, Y.-M.; Yang, Z.-W.; Han, M.-X.; Mou, X.; Xiao, M.; Li, W.-J. Aestuariivirga litoralis Gen. Nov., Sp. Nov., a Proteobacterium Isolated from a Water Sample, and Proposal of Aestuariivirgaceae Fam. Nov. Int. J. Syst. Evol. Microbiol. 2019, 69, 299–306. [Google Scholar] [CrossRef]
- Addison, S.L.; Foote, S.M.; Reid, N.M.; Lloyd-Jones, G. Novosphingobium nitrogenifigens Sp. Nov., a Polyhydroxyalkanoate-Accumulating Diazotroph Isolated from a New Zealand Pulp and Paper Wastewater. Int. J. Syst. Evol. Microbiol. 2007, 57, 2467–2471. [Google Scholar] [CrossRef]
- Rangjaroen, C.; Rerkasem, B.; Teaumroong, N.; Noisangiam, R.; Lumyong, S. Promoting Plant Growth in a Commercial Rice Cultivar by Endophytic Diazotrophic Bacteria Isolated from Rice Landraces. Ann. Microbiol. 2015, 65, 253–266. [Google Scholar] [CrossRef]
- Rahmatullah, R.; Das, M.; Rahmatullah, S.M. Suitable Stocking Density of Tilapia in an Aquaponic System. Bangladesh J. Fish. Res. 2010, 14, 29–35. [Google Scholar]
- Ani, J.S.; Manyala, J.O.; Masese, F.O.; Fitzsimmons, K. Effect of Stocking Density on Growth Performance of Monosex Nile Tilapia (Oreochromis niloticus) in the Aquaponic System Integrated with Lettuce (Lactuca sativa). Aquac. Fish. 2022, 7, 328–335. [Google Scholar] [CrossRef]
- Nam, G.G.; Joung, Y.; Song, J.; Lim, Y.; Cho, J.-C. Emticicia fontis Sp. Nov., Isolated from a Freshwater Pond. Int. J. Syst. Evol. Microbiol. 2016, 66, 5161–5166. [Google Scholar] [CrossRef] [PubMed]

| Group | Total Length (mm) | Body Weight (g) | F.C. 1 | Individual Growth Rate (%) 2 | Survival Rate (%) 3 |
|---|---|---|---|---|---|
| BFT | 457.9 ± 25.8 b | 140.3 ±27.0 b | 1.6 | 27.3 | 100 |
| 5 kg/m2 | 479.7 ± 31.3 a | 168.2 ± 26.8 a | 0.9 | 52.6 | 91.6 |
| 10 kg/m2 | 473.8 ± 21.2 ab | 153.6 ± 24.7 ab | 1.0 | 39.4 | 97.8 |
| 15 kg/m2 | 458.7 ± 17.5 b | 143.8 ± 16.0 b | 1.6 | 30.5 | 97.8 |
| 20 kg/m2 | 456.2 ± 15.1 b | 132.9 ± 20.0 b | 3.0 | 20.6 | 99.4 |
| Group | Total Weight (g) | Shoot Weight (g) | Shoot Length (mm) | Leaf Length (mm) | No. of Leaves |
|---|---|---|---|---|---|
| Hydroponics | 103.0 ± 17.2 ns | 78.0 ± 19.6 ns | 199.0 ± 20.1 ns | 154.0 ± 4.2 ns | 15.6 ± 2.4 b |
| 5 kg/m2 | 104.0 ± 13.4 | 85.0 ± 8.7 | 199.0 ± 25.6 | 158.0 ± 17.5 | 16.0 ± 1.4 b |
| 10 kg/m2 | 101.0 ± 4.2 | 75.0 ± 14.1 | 192.0 ± 14.8 | 160.0 ± 14.6 | 17.0 ± 3.6 ab |
| 15 kg/m2 | 100.0 ± 13.7 | 79.0 ± 14.3 | 220.0 ± 20.9 | 177.0 ± 16.4 | 22.4 ± 3.9 a |
| 20 kg/m2 | 96.0 ± 4.2 | 73.0 ± 6.7 | 206.0 ± 20.7 | 162.0 ± 24.9 | 20.6 ± 1.8 a |
| Temp 1 (°C) | DO 2 (mg/L) | pH (mV) | EC 3 (mS/cm) | TDS 4 (g/L) | |
|---|---|---|---|---|---|
| BFT | 25.7 ± 0.3 ns | 6.9 ± 0.5 a | 6.4 ± 0.9 ns | 0.5 ± 0.2 ns | 0.3 ± 0.1 ns |
| 5 kg/m2 | 25.7 ± 0.5 | 6.0 ± 0.4 ab | 6.4 ± 0.8 | 0.4 ± 0.1 | 0.3 ± 0.1 |
| 10 kg/m2 | 25.9 ± 0.5 | 6.2 ± 0.5 ab | 5.9 ± 1.2 | 0.6 ± 0.2 | 0.4 ± 0.1 |
| 15 kg/m2 | 25.8 ± 0.7 | 8.4 ± 2.2 c | 5.7 ± 1.0 | 0.7 ± 0.3 | 0.5 ± 0.2 |
| 20 kg/m2 | 26.1 ± 0.8 | 7.0 ± 1.0 a | 5.8 ± 0.9 | 0.9 ± 0.4 | 0.6 ± 0.3 |
| Nutrients (mg/L) | Experimental Groups | ||||
|---|---|---|---|---|---|
| BFT | 5 kg/m2 | 10 kg/m2 | 15 kg/m2 | 20 kg/m2 | |
| Total-N | 25.4 | 15.4 | 23.5 | 57.5 | 15.7 |
| NH4+-N | 0.1 ± 0.1 a | 0.4 ± 0.4 a | 0.9 ± 0.7 b | 0.8 ± 0.7 b | 1.7 ± 0.7 c |
| NO2−-N | 0.2 ± 0.2 ns | 0.2 ± 0.1 | 0.3 ± 0.2 | 0.5 ± 0.3 | 0.7 ± 0.5 |
| NO3−-N | 17.2 ± 7.9 ns | 20.2 ± 7.2 | 27.5 ± 10.6 | 24.9 ± 12.5 | 34.1 ± 14.5 |
| Total-P | 7.79 | 5.64 | 13.80 | 30.50 | 17.70 |
| Na | 66.15 | 60.90 | 76.50 | 81.80 | 71.70 |
| K | 14.20 | 3.10 | 3.10 | 6.70 | 4.30 |
| Ca | 38.50 | 31.40 | 49.70 | 80.50 | 56.70 |
| Mg | 8.55 | 6.40 | 8.10 | 11.60 | 9.10 |
| Fe | 0.16 | 0.16 | 0.16 | 0.18 | 0.15 |
| Zn | 0.09 | 0.06 | 0.13 | 0.22 | 0.15 |
| Cu | 0.01 | 0.01 | 0.06 | 0.09 | 0.05 |
| S | 33.25 | 40.50 | 32.10 | 65.50 | 17.20 |
| Cl | 25.35 | 24.30 | 20.00 | 36.20 | 12.10 |
| Si | 12.05 | 13.53 | 13.87 | 14.78 | 13.89 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hwang, J.-A.; Park, J.S.; Jeong, H.S.; Kim, H.; Oh, S.-Y. Productivity of Fish and Crop Growth and Characteristics of Bacterial Communities in the FLOCponics System. Fishes 2023, 8, 422. https://doi.org/10.3390/fishes8080422
Hwang J-A, Park JS, Jeong HS, Kim H, Oh S-Y. Productivity of Fish and Crop Growth and Characteristics of Bacterial Communities in the FLOCponics System. Fishes. 2023; 8(8):422. https://doi.org/10.3390/fishes8080422
Chicago/Turabian StyleHwang, Ju-Ae, Jun Seong Park, Hae Seung Jeong, Hyeongsu Kim, and Seung-Yoon Oh. 2023. "Productivity of Fish and Crop Growth and Characteristics of Bacterial Communities in the FLOCponics System" Fishes 8, no. 8: 422. https://doi.org/10.3390/fishes8080422
APA StyleHwang, J.-A., Park, J. S., Jeong, H. S., Kim, H., & Oh, S.-Y. (2023). Productivity of Fish and Crop Growth and Characteristics of Bacterial Communities in the FLOCponics System. Fishes, 8(8), 422. https://doi.org/10.3390/fishes8080422







