Copper-Induced Ionoregulatory Disturbance, Histopathology, and Transcriptome Responses in Freshwater Mussel (Anodonta woodiana) Gills
Abstract
1. Introduction
2. Materials and Methods
2.1. Mussels and Treatment
2.2. Copper Bioaccumulation and Ion Concentration Analyses
2.3. Histopathologic Analysis
2.4. RNA Isolation, Library Construction, and Sequencing
2.5. Transcriptome Data Analysis and Identification of Differentially Expressed Genes
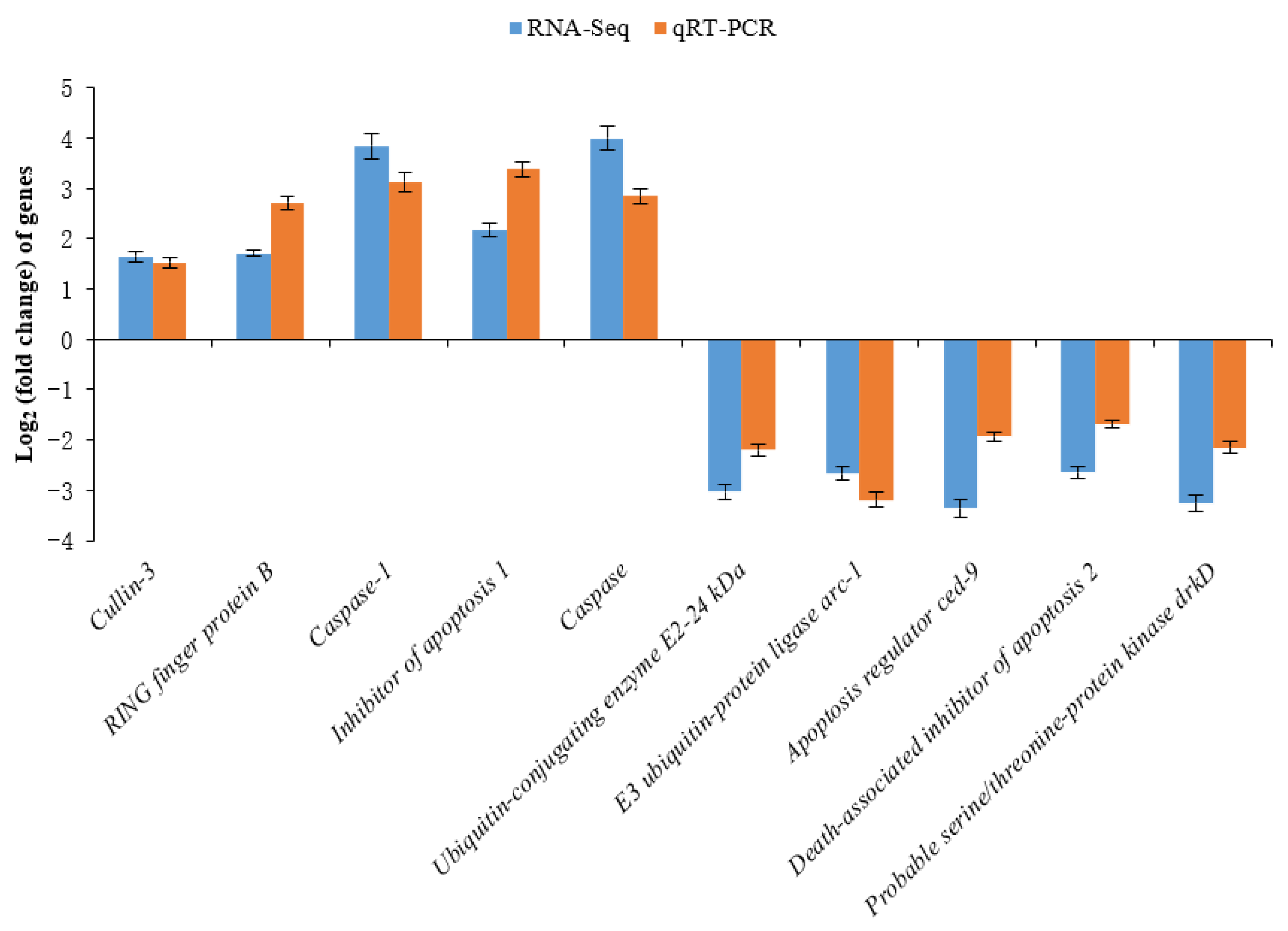
2.6. Real-Time Quantitative RT-PCR Validation
2.7. Statistical Analysis
3. Results
3.1. Cu Bioaccumulation
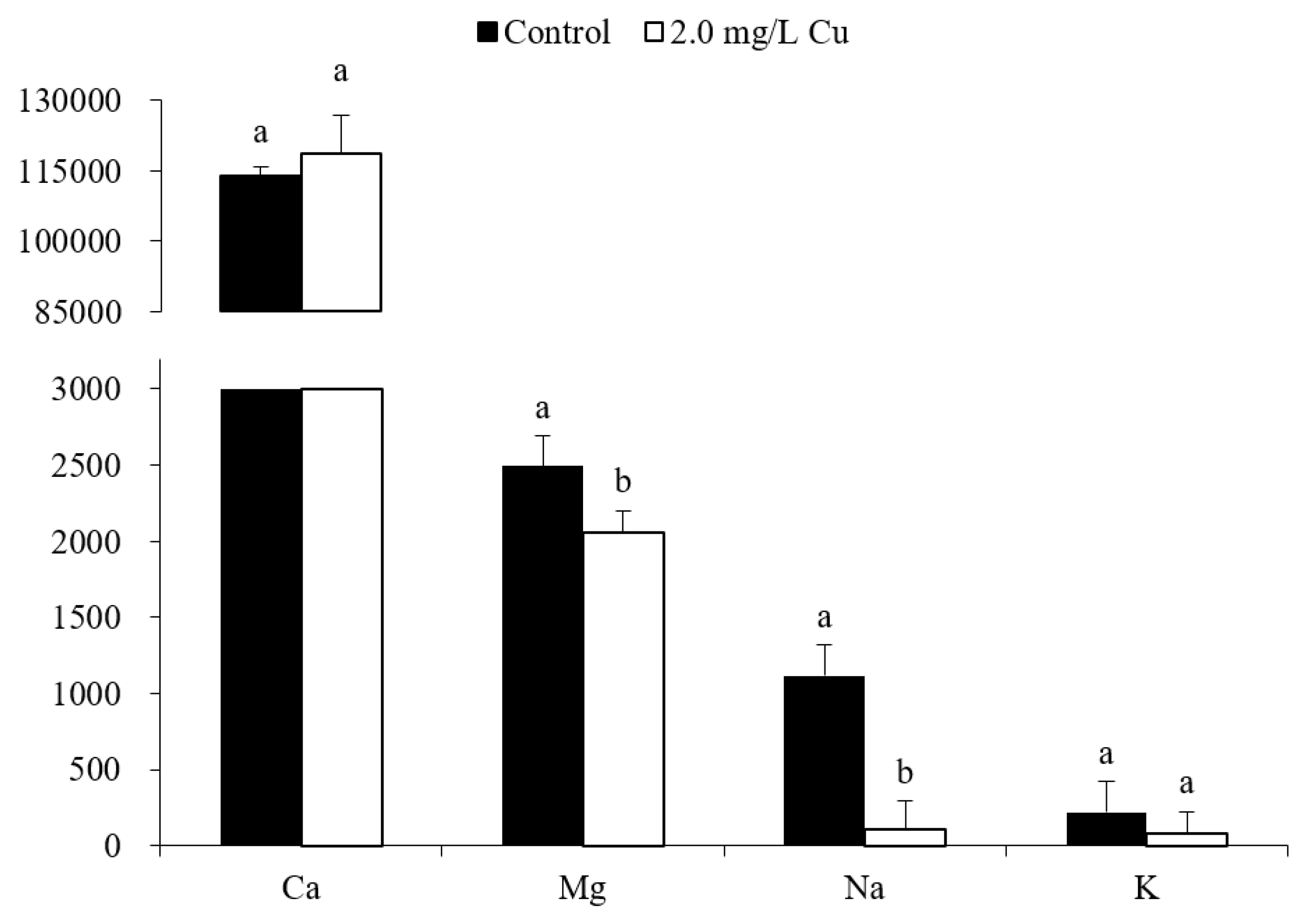
3.2. Ion Concentrations
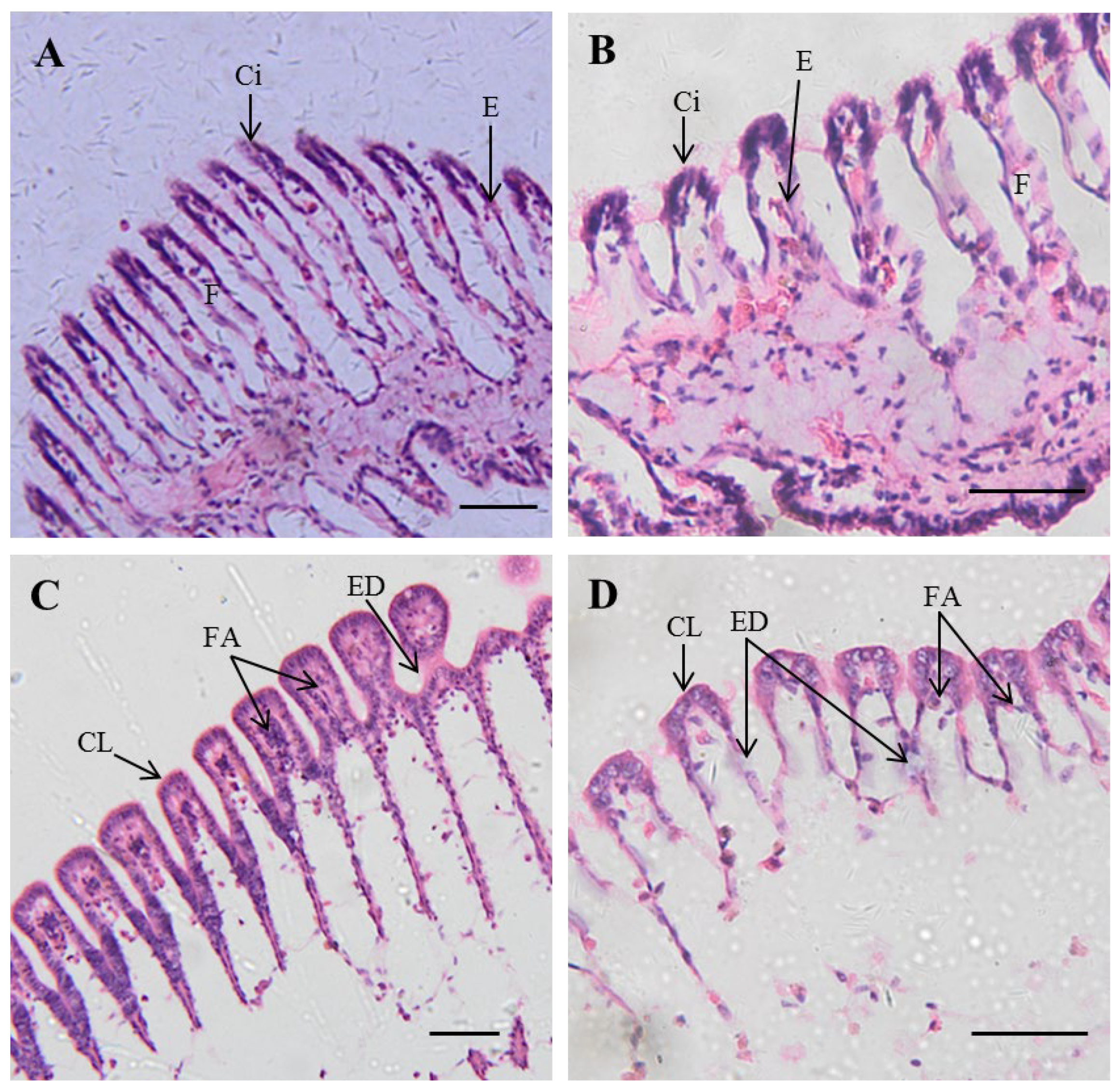
3.3. Histopathology
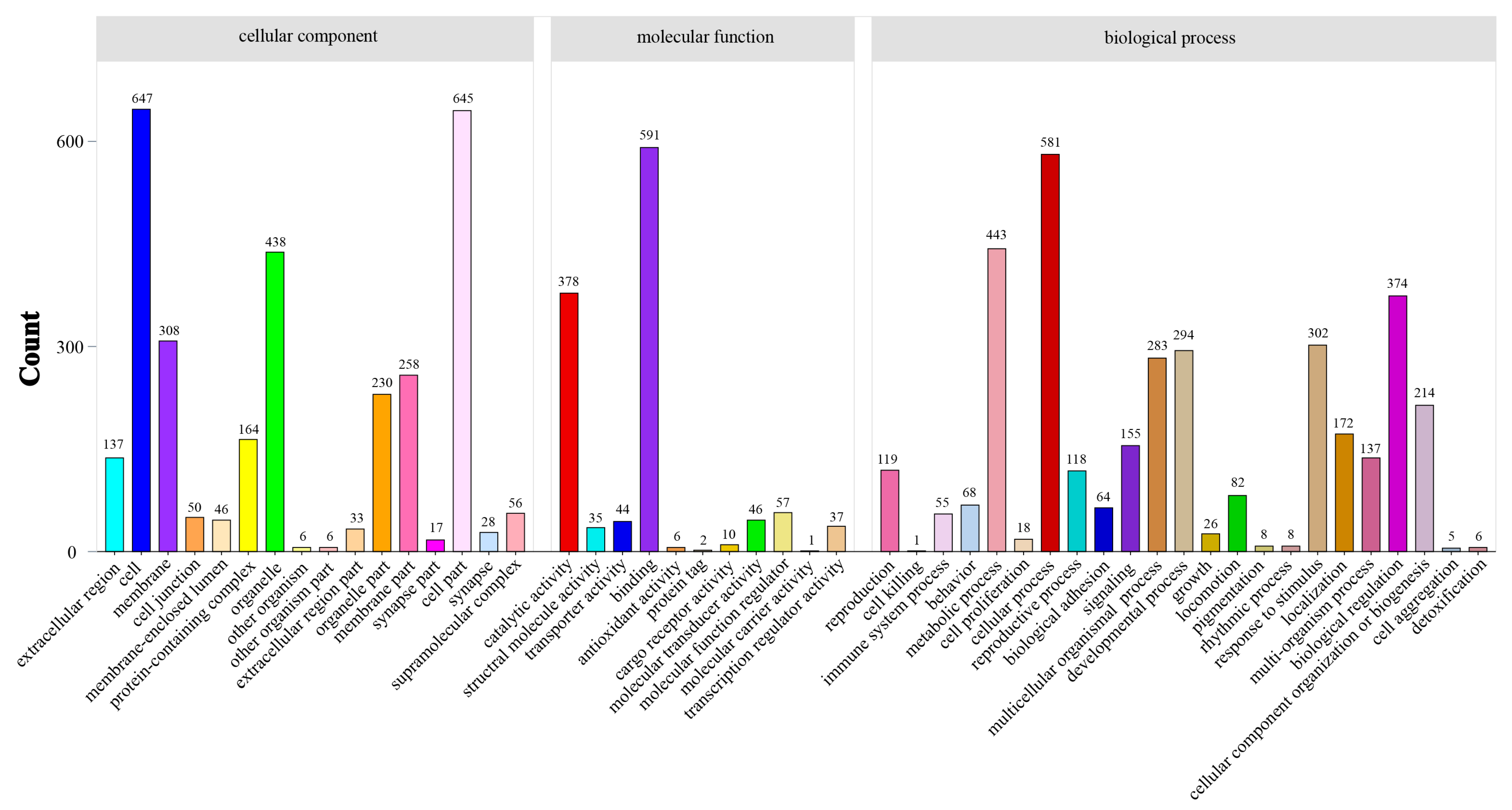
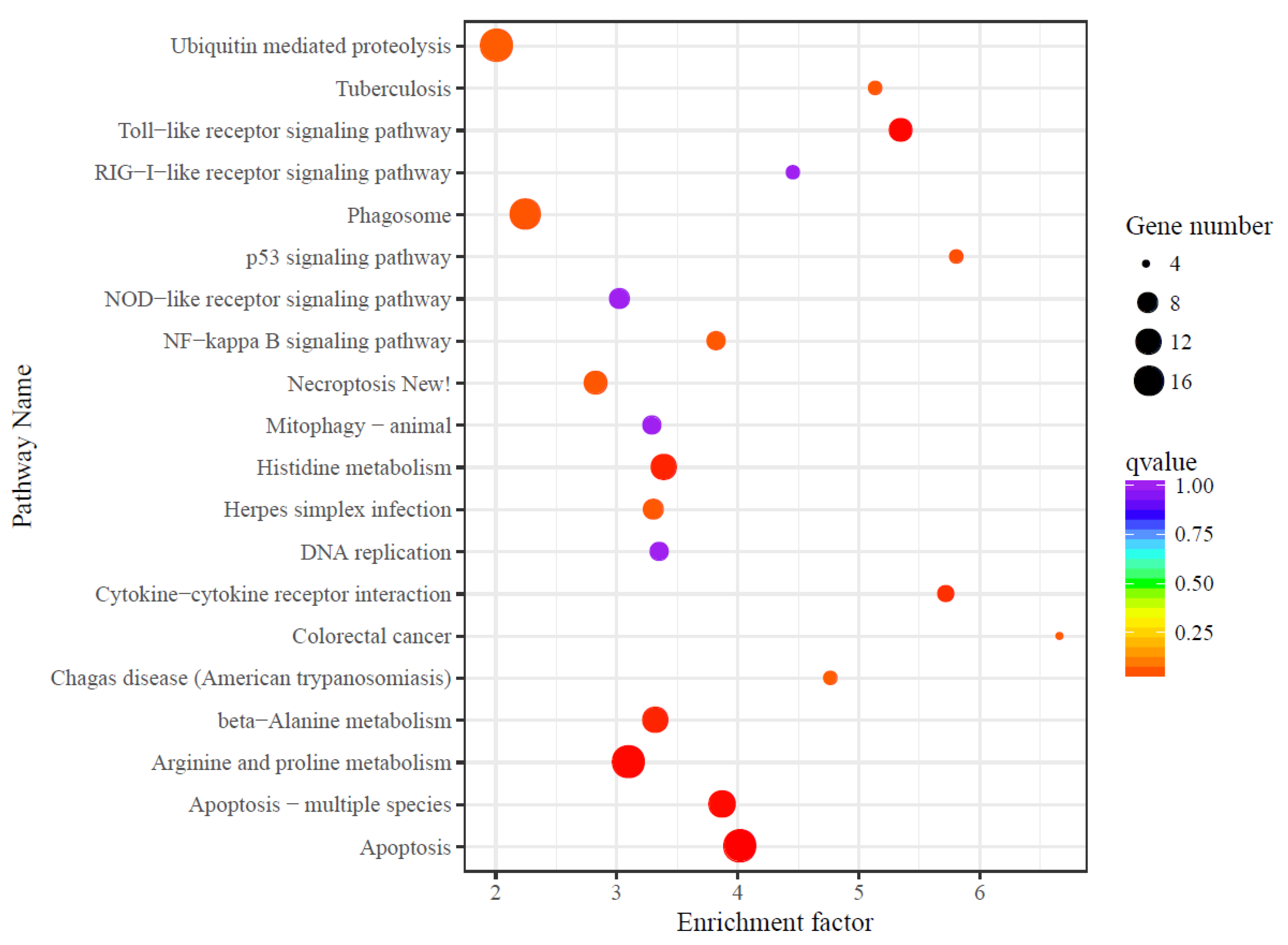
3.4. Differential Expression Analysis
4. Discussion
4.1. Cu Bioaccumulation
4.2. Ionoregulatory Disturbance
4.3. Histological Hazards
4.4. Transcriptome Responses
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Johnson, A.C.; Jin, X.; Nakada, N.; Sumpter, J.P. Learning from the past and considering the future of chemicals in the environment. Science 2020, 367, 384–387. [Google Scholar] [CrossRef] [PubMed]
- Tavares-Dias, M. Toxic, physiological, histomorphological, growth performance and antiparasitic effects of copper sulphate in fish aquaculture. Aquaculture 2021, 535, 736350. [Google Scholar] [CrossRef]
- Dorsey, A.; Ingerman, L.; Swarts, S. Toxicological Profile for Copper; Agency for Toxic Substances and Disease Registry: Atlanta, GA, USA, 2004; pp. 163–167. [Google Scholar]
- Xiao, H.Y.; Zhou, W.B.; Zeng, F.P.; Wu, D.S. Water chemistry and heavy metal distribution in an AMD highly contaminated river. Environ. Earth Sci. 2010, 59, 1023–1031. [Google Scholar] [CrossRef]
- Grande, J.A.; Aroba, J.; Andújar, J.M.; Gómez, T.; de la Torre, M.L.; Borrego, J.; Romero, S.; Barranco, C.; Santisteban, M. Tinto Versus Odiel: Two A.M.D. polluted rivers and an unresolved issue. An artificial intelligence approach. Water Resour. Manag. 2011, 25, 3575–3594. [Google Scholar] [CrossRef]
- Adewumi, A.J.; Laniyan, T.A. Ecological and human health risks associated with metals in water from Anka artisanal gold mining area, Nigeria. Hum. Ecol. Risk Assess. 2021, 27, 307–326. [Google Scholar] [CrossRef]
- Pytharopoulou, S.; Kournoutou, G.G.; Leotsinidis, M.; Georgiou, C.D.; Kalpaxis, D.L. Cadmium versus copper toxicity: Insights from an integrated dissection of protein synthesis pathway in the digestive glands of mussel Mytilus galloprovincialis. J. Hazard. Mater. 2013, 260, 263–271. [Google Scholar] [CrossRef]
- Liu, H.B.; Chen, X.B.; Su, Y.P.; Kang, I.J.; Qiu, X.C.; Shimasaki, Y.; Oshima, Y.; Yang, J. Effects of calcium and magnesium ions on acute copper toxicity to glochidia and early juveniles of the Chinese pond mussel Anodonta woodiana. Bull. Environ. Contam. Tox. 2016, 97, 504–509. [Google Scholar] [CrossRef]
- Shekh, K.; Alcaraz, A.J.; Niyogi, S.; Hecker, M. Comparative analyses of oxidative stress response and metallothionein induction in white sturgeon and rainbow trout during acute waterborne copper exposure. Comp. Biochem. Phys. C 2020, 231, 108723. [Google Scholar] [CrossRef]
- Zheng, X.; Zang, W.; Yan, Z.; Hong, Y.; Liu, Z.; Yi, X.; Wang, X.; Liu, T.; Zhou, L. Species sensitivity analysis of heavy metals to freshwater organisms. Ecotoxicology 2015, 24, 1621–1631. [Google Scholar]
- Cope, W.G.; Bringolf, R.B.; Buchwalter, D.B.; Newton, T.J.; Ingersoll, C.G.; Wang, N.; Augspurger, T.; Dwyer, F.J.; Barnhart, M.C.; Neves, R.J.; et al. Differential exposure, duration, and sensitivity of unionoidean bivalve life stages to environmental contaminants. J. N. Am. Benthol. Soc. 2008, 27, 451–462. [Google Scholar] [CrossRef]
- Bian, B.; Zhou, Y.; Fang, B. Distribution of heavy metals and benthic macroinvertebrates: Impacts from typical inflow river sediments in the Taihu Basin, China. Ecol. Indic. 2016, 69, 348–359. [Google Scholar] [CrossRef]
- Renaut, S.; Guerra, D.; Hoeh, W.R.; Stewart, D.T.; Bogan, A.E.; Ghiselli, F.; Milani, L.; Passamonti, M.; Breton, S. Genome survey of the freshwater mussel Venustaconcha ellipsiformis (bivalvia: Unionida) using a hybrid de novo assembly approach. Genome Biol. Evol. 2018, 10, 1637–1646. [Google Scholar] [CrossRef]
- Bertucci, A.; Pierron, F.; Thébault, J.; Klopp, C.; Bellec, J.; Gonzalez, P.; Baudrimont, M. Transcriptomic responses of the endangered freshwater mussel Margaritifera margaritifera to trace metal contamination in the Dronne River, France. Environ. Sci. Pollut. Res. 2017, 24, 27145–27159. [Google Scholar] [CrossRef] [PubMed]
- Donrovich, S.W.; Douda, K.; Plechingerová, V.; Rylková, K.; Horký, P.; Slavík, O.; Liu, H.Z.; Reichard, M.; Lopes-Lima, M.; Sousa, R. Invasive Chinese pond mussel Sinanodonta woodiana threatens native mussel reproduction by inducing cross-resistance of host fish. Aquat. Conserv. 2017, 27, 1325–1333. [Google Scholar] [CrossRef]
- Chen, X.B.; Yang, J.; Liu, H.B.; Jiang, T. Freshwater Mussel Watch: An innovative approach for interpretations of aquatic pollution and toxicology. J. Lake Sci. 2021, 33, 11–27, (In Chinese with English Abstract). [Google Scholar]
- Liu, J.; Gu, B.; Bian, J.; Hu, S.; Cheng, X.; Ke, Q.; Yan, H. Antitumor activities of liposome–incorporated aqueous extracts of Anodonta woodiana (Lea, 1834). Eur. Food Res. Technol. 2008, 227, 919–924. [Google Scholar] [CrossRef]
- Stangierski, J.; Andrzejewski, W.; Tomaszewska-Gras, J.; Grześ, B.; Konieczny, P.; Urbańska, M. Effect of washing on the quality of surimi-like preparation obtained from soft tissue of freshwater mussel Sinanodonta woodiana (Lea, 1834). J. Aquat. Food Prod. Technol. 2018, 27, 961–974. [Google Scholar] [CrossRef]
- Rahayu, S.Y.S.; Solihin, D.D.; Manalu, W.; Affandi, R. Nucleus pearl coating process of freshwater mussel Anodonta woodiana (Unionidae). HAYATI J. Biosci. 2013, 20, 24–30. [Google Scholar] [CrossRef]
- Yang, J.; Harino, H.; Liu, H.B.; Miyazaki, N. Monitoring the organotin contamination in the Taihu Lake of China by bivalve mussel Anodonta woodiana. Bull. Environ. Contam. Tox. 2008, 81, 164–168. [Google Scholar] [CrossRef]
- Liu, H.B.; Yang, J.; Gan, J.L. Trace element accumulation in bivalves Anodonta woodiana from the Taihu Lake, China. Arch. Environ. Contam. Tox. 2010, 59, 593–601. [Google Scholar] [CrossRef]
- Chen, X.B.; Su, Y.P.; Liu, H.B.; Yang, J. Active biomonitoring of metals with cultured Anodonta woodiana: A case study in the Taihu Lake, China. Bull. Environ. Contam. Tox. 2019, 102, 198–203. [Google Scholar] [CrossRef]
- Królak, E.; Zdanowski, B. The bioaccumulation of heavy metals by the mussels Anodonta woodiana (Lea, 1834) and Dreissena polymorpha (Pall.) in the Heated Konin Lakes. Arch. Pol. Fish. 2001, 9, 229–237. [Google Scholar]
- Kolarević, S.; Knežević-Vukčević, J.; Paunović, M.; Kračun, M.; Vasiljević, B.; Tomović, J.; Vuković-Gačić, B.; Gači, Z. Monitoring of DNA damage in haemocytes of freshwater mussel Sinanodonta woodiana sampled from the Velika Morava River in Serbia with the comet assay. Chemosphere 2013, 93, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Gecheva, G.; Yancheva, V.; Velcheva, I.; Georgieva, E.; Stoyanova, S.; Arnaudova, D.; Stefanova, V.; Georgieva, D.; Genina, V.; Todorova, B.; et al. Integrated monitoring with moss-bag and mussel transplants in reservoirs. Water 2020, 12, 1800. [Google Scholar] [CrossRef]
- Kurnia, A.I.; Purwanto, E.; Mahajoeno, E. Exposure copper heavy metal (Cu) on freshwater mussel (Anodonta woodiana) and its relation to Cu and protein content in the body shell. Nusant. Biosci. 2010, 2, 48–53. [Google Scholar] [CrossRef]
- Liu, H.B.; Chen, X.B.; Oshima, Y.; Shimasaki, Y.; Jiang, T.; Yang, J. Biochemical changes in Chinese pond mussel Anodonta woodiana (Lea, 1834) following exposure to copper. J. Fac. Agric. Kyushu Univ. 2018, 63, 311–318. [Google Scholar]
- Chen, X.B.; Liu, H.B.; Huang, H.H.; Liber, K.; Jiang, T.; Yang, J. Cadmium bioaccumulation and distribution in the freshwater bivalve Anodonta woodiana exposed to environmentally relevant Cd levels. Sci. Total Environ. 2021, 791, 148289. [Google Scholar] [CrossRef]
- Li, Y.Q.; Chen, C.M.; Liu, N.; Wang, L. Cadmium-induced ultrastructural changes and apoptosis in the gill of freshwater mussel Anodonta woodiana. Environ. Sci. Pollut. Res. 2022, 29, 23338–23351. [Google Scholar] [CrossRef]
- Meng, X.; Tian, X.; Liu, M.; Nie, G.; Jiang, K.; Wang, B.; Wang, L. The transcriptomic response to copper exposure by the gill tissue of Japanese scallops (Mizuhopecten yessoensis) using deep-sequencing technology. Fish Shellfish Immun. 2014, 38, 287–293. [Google Scholar] [CrossRef]
- Sohail, M.; Khan, M.N.; Qureshi, N.A.; Chaudhry, A.S. Monitoring DNA damage in gills of freshwater mussels (Anodonta anatina) exposed to heavy metals. Pak. J. Zool. 2017, 49, 305–311. [Google Scholar] [CrossRef]
- Dragun, Z.; Erk, M.; Ivanković, D.; Žaja, R.; Marijić, V.F.; Raspor, B. Assessment of low-level metal contamination using the Mediterranean mussel gills as the indicator tissue. Environ. Sci. Pollut. Res. 2010, 17, 977–986. [Google Scholar] [CrossRef] [PubMed]
- ASTM-E2455-06; Standard Guide for Conducting Laboratory Toxicity Tests with Freshwater Mussels. ASTM International: West Conshohocken, PA, USA, 2013.
- Zhou, C.; Huang, J.C.; Liu, F.; He, S.; Zhou, W. Removal of selenium containing algae by the bivalve Sinanodonta woodiana and the potential risk to human health. Environ. Pollut. 2018, 242, 73–81. [Google Scholar] [CrossRef]
- Goh, K.S.; Sheu, H.S.; Hua, T.E.; Kang, M.H.; Li, C.W. Uric acid spherulites in the reflector layer of firefly light organ. PLoS ONE 2013, 8, e56406. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Zhang, C.; Huang, B. Transcriptome analysis and histopathological observations of Geloina erosa gills upon Cr (VI) exposure. Comp. Biochem. Phys. C 2020, 231, 108706. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, Z.; Kholodkevich, S.; Sharov, A.; Feng, Y.; Ren, N.; Sun, K. Cadmium-induced oxidative stress, histopathology, and transcriptome changes in the hepatopancreas of freshwater crayfish (Procambarus clarkii). Sci. Total Environ. 2019, 666, 944–955. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Che, P.; Li, H.; Qiao, S.; Wang, J.; Wang, Y.; Wang, X.; Wu, B.; Liu, H.; Wang, C.; et al. Comparative transcriptome analysis reveals the differential response to cadmium stress of two Pleurotus fungi: Pleurotus cornucopiae and Pleurotus ostreatus. J. Hazard. Mater. 2021, 416, 125814. [Google Scholar] [CrossRef]
- Xia, X.; Xue, S.; Wang, X.; Zhang, Q.; Huang, C.; Guo, L.; Yao, L. Response a chronic effects of PBDE-47: Up-regulations of HSP60 and HSP70 expression in freshwater bivalve Anodonta woodiana. Fish Shellfish Immun. 2017, 65, 213–225. [Google Scholar] [CrossRef]
- Qu, F.; Li, J.; She, Q.; Zeng, X.; Li, Z.; Lin, Q.; Tang, J.; Yan, Y.; Lu, J.; Li, Y.; et al. Identification and characterization of MKK6 and AP-1 in Anodonta woodiana reveal their potential roles in the host defense response against bacterial challenge. Fish Shellfish Immun. 2022, 124, 261–272. [Google Scholar] [CrossRef]
- Qu, F.; She, Q.; Li, J.; Zeng, X.; Li, Y.; Liu, X.; Ren, L.; Liu, Z.; Gao, C.; Lu, X.; et al. Molecular characterization of MyD88 in Anodonta woodiana and its involvement in the innate immune response to bacterial infection. Front. Immunol. 2022, 13, 2893. [Google Scholar] [CrossRef]
- Bai, Z.; Lin, J.; Ma, K.; Wang, G.; Niu, D.; Li, J. Identification of housekeeping genes suitable for gene expression analysis in the pearl mussel, Hyriopsis cumingii, during biomineralization. Mol. Genet. Genom. 2014, 289, 717–725. [Google Scholar] [CrossRef]
- Dummee, V.; Tanhan, P.; Kruatrachue, M.; Damrongphol, P.; Pokethitiyook, P. Histopathological changes in snail, Pomacea canaliculata, exposed to sub-lethal copper sulfate concentrations. Ecotox. Environ. Safe. 2015, 122, 290–295. [Google Scholar] [CrossRef] [PubMed]
- Arnot, J.A.; Gobas, F.A. A review of bioconcentration factor (BCF) and bioaccumulation factor (BAF) assessments for organic chemicals in aquatic organisms. Environ. Rev. 2006, 14, 257–297. [Google Scholar] [CrossRef]
- Marigómez, I.; Soto, M.; Cajaraville, M.P.; Angulo, E.; Giamberini, L. Cellular and subcellular distribution of metals in molluscs. Microsc. Res. Tech. 2002, 56, 358–392. [Google Scholar] [CrossRef] [PubMed]
- Nugroho, A.P.; Frank, H. Uptake, distribution, and bioaccumulation of copper in the freshwater mussel Anodonta anatina. Toxicol. Environ. Chem. 2011, 93, 1838–1850. [Google Scholar] [CrossRef]
- Chen, X.B.; Yang, J.; Liu, H.B.; Su, Y.P.; Sun, L.; Oshima, Y. Element concentrations in a unionid mussel (Anodonta woodiana) at different life stages. J. Fac. Agric. Kyushu Univ. 2012, 57, 139–144. [Google Scholar] [CrossRef]
- Jorge, M.B.; Bianchini, A.; Wood, C.M.; Gillis, P.L. Copper uptake, patterns of bioaccumulation, and effects in glochidia (larvae) of the freshwater mussel (Lampsilis cardium). Environ. Toxicol. Chem. 2018, 37, 1092–1103. [Google Scholar] [CrossRef]
- Jorge, M.B.; Loro, V.L.; Bianchini, A.; Wood, C.M.; Gillis, P.L. Mortality, bioaccumulation and physiological responses in juvenile freshwater mussels (Lampsilis siliquoidea) chronically exposed to copper. Aquat. Toxicol. 2013, 126, 137–147. [Google Scholar] [CrossRef]
- Komjarova, I.; Bury, N.R. Evidence of common cadmium and copper uptake routes in zebrafish Danio rerio. Environ. Sci. Technol. 2014, 48, 12946–12951. [Google Scholar] [CrossRef]
- Le, T.T.Y.; Nachev, M.; Grabner, D.; Garcia, M.R.; Balsa-Canto, E.; Hendriks, A.J.; Peijnenburg, W.J.G.M.; Sures, B. Modelling chronic toxicokinetics and toxicodynamics of copper in mussels considering ionoregulatory homeostasis and oxidative stress. Environ. Pollut. 2021, 287, 117645. [Google Scholar] [CrossRef]
- Canli, E.G. Alterations on the activities of ion ATPases in the gill and muscle of freshwater mussel (Unio tigridis) exposed to copper. Commagene J. Biol. 2021, 5, 150–155. [Google Scholar] [CrossRef]
- Khan, M.I.; Khisroon, M.; Khan, A.; Gulfam, N.; Siraj, M.; Zaidi, F.; Ahmadullah; Abidullah; Fatima, S.H.; Noreen, S.; et al. Bioaccumulation of heavy metals in water, sediments, and tissues and their histopathological effects on Anodonta cygnea (Linea, 1876) in Kabul River, Khyber Pakhtunkhwa, Pakistan. BioMed Res. Int. 2018, 2018, 1910274. [Google Scholar] [CrossRef]
- Katsumiti, A.; Thorley, A.J.; Arostegui, I.; Reip, P.; Valsami-Jones, E.; Tetley, T.D.; Cajaraville, M.P. Cytotoxicity and cellular mechanisms of toxicity of CuO NPs in mussel cells in vitro and comparative sensitivity with human cells. Toxicol. In Vitro 2018, 48, 146–158. [Google Scholar] [CrossRef] [PubMed]
- Tsvetkov, P.; Coy, S.; Petrova, B.; Dreishpoon, M.; Verma, A.; Abdusamad, M.; Abdusamad, M.; Rossen, J.; Joesch-Cohen, L.; Humeidi, R.; et al. Copper induces cell death by targeting lipoylated TCA cycle proteins. Science 2022, 375, 1254–1261. [Google Scholar] [CrossRef] [PubMed]
- Stalin, A.; Musthafa, M.S.; Amanullah, B. Histological alterations in the gills of freshwater mussel Lamellidens marginalis exposed to sub-lethal concentration of an organophosphorus insecticide, chlorpyrifos. Asian J. Microbiol. Biotechnol. Environ. Sci. 2011, 13, 139–142. [Google Scholar]
- Wang, W.X.; Meng, J.; Weng, N. Trace metals in oysters: Molecular and cellular mechanisms and ecotoxicological impacts. Environ. Sci. Proc. Imp. 2018, 20, 892–912. [Google Scholar] [CrossRef]
- Chen, J.; Xu, Y.; Han, Q.; Yao, Y.; Xing, H.; Teng, X. Immunosuppression, oxidative stress, and glycometabolism disorder caused by cadmium in common carp (Cyprinus carpio L.): Application of transcriptome analysis in risk assessment of environmental contaminant cadmium. J. Hazard. Mater. 2019, 366, 386–394. [Google Scholar] [CrossRef]
- Abo-Al-Ela, H.G.; El-Kassas, S.; El-Naggar, K.; Abdo, S.E.; Jahejo, A.R.; Wakeel, R.A.A. Stress and immunity in poultry: Light management and nanotechnology as effective immune enhancers to fight stress. Cell Stress Chaperon. 2021, 26, 457–472. [Google Scholar] [CrossRef]
- Terahara, K.; Takahashi, K.G. Mechanisms and immunological roles of apoptosis in molluscs. Curr. Pharm. Des. 2008, 14, 131–137. [Google Scholar] [CrossRef]
- Zhang, M.; Li, L.; Liu, Y.; Gao, X. Effects of a sudden drop in salinity on immune response mechanisms of Anadara kagoshimensis. Int. J. Mol. Sci. 2019, 20, 4365. [Google Scholar] [CrossRef]
- Wei, F.; Su, T.; Wang, D.; Li, H.; You, J. Transcriptomic analysis reveals common pathways and biomarkers associated with oxidative damage caused by mitochondrial toxicants in Chironomus dilutus. Chemosphere 2020, 254, 126746. [Google Scholar] [CrossRef]
- Châtel, A.; Mouneyrac, C. Signaling pathways involved in metal-based nanomaterial toxicity towards aquatic organisms. Comp. Biochem. Phys. C 2017, 196, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Wang, X.; Chen, D.; Xu, E.G.; Luo, X.; Zeng, J.; Huan, T.; Li, L.; Wang, Y. Toxicity mechanisms of polystyrene microplastics in marine mussels revealed by high-coverage quantitative metabolomics using chemical isotope labeling liquid chromatography mass spectrometry. J. Hazard. Mater. 2021, 417, 126003. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.V.; Alfaro, A.C.; Merien, F.; Lulijwa, R.; Young, T. Copper-induced immunomodulation in mussel (Perna canaliculus) haemocytes. Metallomics 2018, 10, 965. [Google Scholar] [CrossRef] [PubMed]

| Gene Name | Primer Sequence | Amplification Efficiency (%) |
|---|---|---|
| Cullin-3 | F-AGGGCGACACACTATTTGGA | 98 |
| R-CCTGCTCCCTCAAGTAACCA | ||
| RING finger protein B | F-TGGAAAGCACACAAGTTGCA | 97 |
| R-GCTTGCCAGTTTCTTCTCGT | ||
| Caspase-1 | F-GCATGCCAGGGACAAAAGTT | 94 |
| R-AGAGCTTGAACGAACCAGGA | ||
| Inhibitor of apoptosis 1 | F-TCCACATGAGGTTCACACGA | 100 |
| R-CGCACAAAGGTTCGAATTGC | ||
| Caspase | F-CACAATACCTCTGAAGCCGC | 102 |
| R-AGGACTGGTTGTGATGAGCA | ||
| Ubiquitin-conjugating enzyme E2-24 kDa | F-CTTTGGTCCAGCACTGCAAT | 97 |
| R-CGTGGGAAGAAAAGAGCTCG | ||
| E3 ubiquitin-protein ligase arc-1 | F-CTGACATGCCCAGTTTGCTT | 102 |
| R-GATGTGCGCCTCATTCTCTG | ||
| Apoptosis regulator CED-9 | F-TCAGTGTATGGTATCGGGCC | 105 |
| R-CACTTTCACGCCCTCTGAAC | ||
| Death-associated inhibitor of apoptosis 2 | F-TCGGGATGCCTGTATTGTGT | 100 |
| R-AGCTCTTTACTCCCTACCGC | ||
| Probable serine/threonine protein kinase drkD | F-TCACTGCTGCCCGAATCATA | 103 |
| R-AGTGACACGGAAGACAGGAG | ||
| β-actin | F-GTGGCTACTCCTTCACAACC | 101 |
| R-GAAGCTAGGCTGGAACAAGG |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, X.; Liu, H.; Liber, K.; Jiang, T.; Yang, J. Copper-Induced Ionoregulatory Disturbance, Histopathology, and Transcriptome Responses in Freshwater Mussel (Anodonta woodiana) Gills. Fishes 2023, 8, 368. https://doi.org/10.3390/fishes8070368
Chen X, Liu H, Liber K, Jiang T, Yang J. Copper-Induced Ionoregulatory Disturbance, Histopathology, and Transcriptome Responses in Freshwater Mussel (Anodonta woodiana) Gills. Fishes. 2023; 8(7):368. https://doi.org/10.3390/fishes8070368
Chicago/Turabian StyleChen, Xiubao, Hongbo Liu, Karsten Liber, Tao Jiang, and Jian Yang. 2023. "Copper-Induced Ionoregulatory Disturbance, Histopathology, and Transcriptome Responses in Freshwater Mussel (Anodonta woodiana) Gills" Fishes 8, no. 7: 368. https://doi.org/10.3390/fishes8070368
APA StyleChen, X., Liu, H., Liber, K., Jiang, T., & Yang, J. (2023). Copper-Induced Ionoregulatory Disturbance, Histopathology, and Transcriptome Responses in Freshwater Mussel (Anodonta woodiana) Gills. Fishes, 8(7), 368. https://doi.org/10.3390/fishes8070368







