A Comprehensive Review of the Impacts of Climate Change on Salmon: Strengths and Weaknesses of the Literature by Life Stage
Abstract
1. Introduction
2. Methods
2.1. Study Region
2.2. Literature Collection
2.3. Classification Strategy
2.4. Synthesis
3. Results
3.1. Observed and Projected Physical Impacts of Climate Change
3.1.1. Global
- Global surface temperature in the last decade (2011–2020) was 1.09 °C higher than in 1850–1900
- Due to human influences, global average land precipitation has increased, changing near-surface salinity
- Glaciers have been retreating, Arctic sea ice in September has decreased about 40% from 1979–1988 to 2010–2019, and spring snow cover has decreased in the northern hemisphere.
- The global upper ocean (0–700 m) has warmed, the ocean surface is more acidic, oxygen levels have dropped in many upper ocean regions since the mid-20th century, and the global mean sea level has increased by 0.2 m between 1901 and 2018, and at a rate twice as fast as the long-term average from 2006 to 2018.
- Human influence is the main driver of more frequent and more intense terrestrial and marine heat waves, and concurrent events among heatwaves, droughts, wildfires and flooding.
3.1.2. Freshwater Impacts
3.1.3. Marine Impacts
3.2. Life-Stage Specific Research
3.2.1. Egg Incubation
3.2.2. Juvenile Freshwater Rearing and Migration
3.2.3. Marine Stage
3.2.4. Adult Migration and Spawning
3.2.5. Population-Level Analyses
3.3. Strengths, Weakness, Expected Responses and Management Opportunities
3.3.1. Egg Incubation
3.3.2. Juvenile Freshwater Rearing and Migration
3.3.3. Marine Stage
3.3.4. Adult Migration and Spawning
3.3.5. Population-Level Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pelletier, F.; Coltman, D.W. Will human influences on evolutionary dynamics in the wild pervade the Anthropocene? BMC Biol. 2018, 16, 7. [Google Scholar] [CrossRef]
- Reid, A.J.; Carlson, A.K.; Creed, I.F.; Eliason, E.J.; Gell, P.A.; Johnson, P.T.J.; Kidd, K.A.; MacCormack, T.J.; Olden, J.D.; Ormerod, S.J.; et al. Emerging threats and persistent conservation challenges for freshwater biodiversity. Biol. Rev. 2019, 94, 849–873. [Google Scholar] [CrossRef] [PubMed]
- Exposito-Alonso, M.; Booker, T.R.; Czech, L.; Gillespie, L.; Hateley, S.; Kyriazis, C.C.; Lang, P.L.M.; Leventhal, L.; Nogues-Bravo, D.; Pagowski, V.; et al. Genetic diversity loss in the Anthropocene. Science 2022, 377, 1431–1435. [Google Scholar] [CrossRef] [PubMed]
- Tye, S.P.; Siepielski, A.M.; Bray, A.; Rypel, A.L.; Phelps, N.B.D.; Fey, S.B. Climate warming amplifies the frequency of fish mass mortality events across north temperate lakes. Limnol. Oceanogr. Lett. 2022, 7, 510–519. [Google Scholar] [CrossRef]
- Jones, T.; Parrish, J.K.; Peterson, W.T.; Bjorkstedt, E.P.; Bond, N.A.; Ballance, L.T.; Bowes, V.; Hipfner, J.M.; Burgess, H.K.; Dolliver, J.E.; et al. Massive Mortality of a Planktivorous Seabird in Response to a Marine Heatwave. Geophys. Res. Lett. 2018, 45, 3193–3202. [Google Scholar] [CrossRef]
- Parmesan, C.; Yohe, G. A globally coherent fingerprint of climate change impacts across natural systems. Nature 2003, 421, 37–42. [Google Scholar] [CrossRef]
- Weiskopf, S.R.; Rubenstein, M.A.; Crozier, L.G.; Gaichas, S.; Griffis, R.; Halofsky, J.E.; Hyde, K.J.W.; Morelli, T.L.; Morisette, J.T.; Muñoz, R.C.; et al. Climate change effects on biodiversity, ecosystems, ecosystem services, and natural resource management in the United States. Sci. Total Environ. 2020, 733, 137782. [Google Scholar] [CrossRef]
- Wiens, J.J. Climate-related local extinctions are already widespread among plant and animal species. PLoS Biol. 2016, 14, e2001104. [Google Scholar] [CrossRef]
- Pinsky, M.L.; Rogers, L.A.; Morley, J.W.; Frölicher, T.L. Ocean planning for species on the move provides substantial benefits and requires few trade-offs. Sci. Adv. 2020, 6, eabb8428. [Google Scholar] [CrossRef]
- Morley, J.W.; Selden, R.L.; Latour, R.J.; Frölicher, T.L.; Seagraves, R.J.; Pinsky, M.L. Projecting shifts in thermal habitat for 686 species on the North American continental shelf. PLoS ONE 2018, 13, e0196127. [Google Scholar] [CrossRef]
- Cheung, W.W.L.; Brodeur, R.D.; Okey, T.A.; Pauly, D. Projecting future changes in distributions of pelagic fish species of Northeast Pacific shelf seas. Prog. Oceanogr. 2015, 130, 19–31. [Google Scholar] [CrossRef]
- Buckley, L.B.; Urban, M.C.; Angilletta, M.J.; Crozier, L.G.; Rissler, L.J.; Sears, M.W. Can mechanism inform species’ distribution models? Ecol. Lett. 2010, 13, 1041–1054. [Google Scholar] [CrossRef] [PubMed]
- Petchey, O.L.; Pontarp, M.; Massie, T.M.; Kéfi, S.; Ozgul, A.; Weilenmann, M.; Palamara, G.M.; Altermatt, F.; Matthews, B.; Levine, J.M.; et al. The ecological forecast horizon, and examples of its uses and determinants. Ecol. Lett. 2015, 18, 597–611. [Google Scholar] [CrossRef] [PubMed]
- Evans, T.G.; Diamond, S.E.; Kelly, M.W. Mechanistic species distribution modelling as a link between physiology and conservation. Conserv. Physiol. 2015, 3, cov056. [Google Scholar] [CrossRef] [PubMed]
- Urban, M.C.; Bocedi, G.; Hendry, A.P.; Mihoub, J.-B.; Pe’er, G.; Singer, A.; Bridle, J.R.; Crozier, L.G.; De Meester, L.; Godsoe, W.; et al. Improving the forecast for biodiversity under climate change. Science 2016, 353, 6304. [Google Scholar] [CrossRef] [PubMed]
- Mills, K.E.; Pershing, A.J.; Sheehan, T.F.; Mountain, D. Climate and ecosystem linkages explain widespread declines in North American Atlantic salmon populations. Glob. Chang. Biol. 2013, 19, 3046–3061. [Google Scholar] [CrossRef]
- Chaput, G.; Benoit, H.P. Evidence for bottom-up trophic effects on return rates to a second spawning for Atlantic salmon (Salmo salar) from the Miramichi River, Canada. ICES J. Mar. Sci. 2012, 69, 1656–1667. [Google Scholar] [CrossRef]
- Nicola, G.G.; Elvira, B.; Jonsson, B.; Ayllon, D.; Almodovar, A. Local and global climatic drivers of Atlantic salmon decline in southern Europe. Fish. Res. 2018, 198, 78–85. [Google Scholar] [CrossRef]
- Dadswell, M.; Spares, A.; Reader, J.; McLean, M.; McDermott, T.; Samways, K.; Lilly, J. The decline and impending collapse of the Atlantic salmon (Salmo salar) population in the North Atlantic Ocean: A review of possible causes. Rev. Fish. Sci. Aquac. 2021, 30, 215–258. [Google Scholar] [CrossRef]
- NMFS—National Marine Fisheries Service. 2015 Adult Sockeye Salmon Passage Report. 2016. Available online: https://www.columbiariverkeeper.org/sites/default/files/2017/08/8.pdf (accessed on 14 June 2023).
- Martin, B.T.; Dudley, P.N.; Kashef, N.S.; Stafford, D.M.; Reeder, W.J.; Tonina, D.; Del Rio, A.M.; Foott, J.S.; Danner, E.M. The biophysical basis of thermal tolerance in fish eggs. Proc. R. Soc. B-Biol. Sci. 2020, 287, 20201550. [Google Scholar] [CrossRef]
- Akbarzadeh, A.; Selbie, D.T.; Pon, L.B.; Miller, K.M. Endangered Cultus Lake sockeye salmon exhibit genomic evidence of hypoxic and thermal stresses while rearing in degrading freshwater lacustrine critical habitat. Conserv. Physiol. 2021, 9, coab089. [Google Scholar] [CrossRef] [PubMed]
- Ward, E.J.; Anderson, J.H.; Beechie, T.J.; Pess, G.R.; Ford, M.J. Increasing hydrologic variability threatens depleted anadromous fish populations. Glob. Chang. Biol. 2015, 21, 2500–2509. [Google Scholar] [CrossRef] [PubMed]
- Welch, D.W.; Porter, A.D.; Rechisky, E.L. A synthesis of the coast-wide decline in survival of West Coast Chinook Salmon (Oncorhynchus tshawytscha, Salmonidae). Fish Fish. 2020, 22, 194–211. [Google Scholar] [CrossRef]
- Connors, B.; Malick, M.J.; Ruggerone, G.T.; Rand, P.; Adkison, M.; Irvine, J.R.; Campbell, R.; Gorman, K. Climate and competition influence sockeye salmon population dynamics across the Northeast Pacific Ocean. Can. J. Fish. Aquat. Sci. 2020, 77, 943–949. [Google Scholar] [CrossRef]
- Crozier, L.G.; Siegel, J.E.; Wiesebron, L.E.; Trujillo, E.M.; Burke, B.J.; Sandford, B.P.; Widener, D.L. Snake River sockeye and Chinook salmon in a changing climate: Implications for upstream migration survival during recent extreme and future climates. PLoS ONE 2020, 15, e0238886. [Google Scholar] [CrossRef]
- Crozier, L.G.; Burke, B.J.; Chasco, B.E.; Widener, D.L.; Zabel, R.W. Climate change threatens Chinook salmon throughout their life cycle. Commun. Biol. 2021, 4, 222. [Google Scholar] [CrossRef]
- Bowerman, T.E.; Keefer, M.L.; Caudill, C.C. Elevated stream temperature, origin, and individual size influence Chinook salmon prespawn mortality across the Columbia River Basin. Fish. Res. 2021, 237, 105874. [Google Scholar] [CrossRef]
- FitzGerald, A.M.; John, S.N.; Apgar, T.M.; Mantua, N.J.; Martin, B.T. Quantifying thermal exposure for migratory riverine species: Phenology of Chinook salmon populations predicts thermal stress. Glob. Chang. Biol. 2021, 27, 536–549. [Google Scholar] [CrossRef]
- Martins, E.G.; Hinch, S.G.; Patterson, D.A.; Hague, M.J.; Cooke, S.J.; Miller, K.M.; Lapointe, M.F.; English, K.K.; Farrell, A.P. Effects of river temperature and climate warming on stock-specific survival of adult migrating Fraser River sockeye salmon (Oncorhynchus nerka). Glob. Chang. Biol. 2011, 17, 99–114. [Google Scholar] [CrossRef]
- Isaak, D.J.; Luce, C.H.; Horan, D.L.; Chandler, G.L.; Wollrab, S.P.; Nagel, D.E. Global warming of salmon and trout rivers in the northwestern U.S.: Road to ruin or path through purgatory? Trans. Am. Fish. Soc. 2018, 147, 566–587. [Google Scholar] [CrossRef]
- Wenger, S.J.; Isaak, D.J.; Luce, C.H.; Neville, H.M.; Fausch, K.D.; Dunham, J.B.; Dauwalter, D.C.; Young, M.K.; Elsner, M.M.; Rieman, B.E.; et al. Flow regime, temperature, and biotic interactions drive differential declines of trout species under climate change. Proc. Natl. Acad. Sci. USA 2011, 108, 14175–14180. [Google Scholar] [CrossRef] [PubMed]
- Healey, M. The cumulative impacts of climate change on Fraser River sockeye salmon (Oncorhynchus nerka) and implications for management. Can. J. Fish. Aquat. Sci. 2011, 68, 718–737. [Google Scholar] [CrossRef]
- Wainwright, T.C.; Weitkamp, L.A. Effects of climate change on Oregon coast Coho salmon: Habitat and life-cycle interactions. Northwest Sci. 2013, 87, 219–242. [Google Scholar] [CrossRef]
- McGaughran, A.; Laver, R.; Fraser, C. Evolutionary Responses to Warming. Trends Ecol. Evol. 2021, 36, 591–600. [Google Scholar] [CrossRef] [PubMed]
- Gustafson, R.G.; Waples, R.S.; Myers, J.M.; Weitkamp, L.A.; Bryant, G.J.; Johnson, O.W.; Hard, J.J. Pacific salmon extinctions: Quantifying lost and remaining diversity. Conserv. Biol. 2007, 21, 1009–1020. [Google Scholar] [CrossRef]
- NOAA Fisheries. Pacific Salmon and Steelhead. Available online: https://www.fisheries.noaa.gov/species/pacific-salmon-and-steelhead (accessed on 1 January 2021).
- Waples, R.S.; Gustafson, R.G.; Weitkamp, L.A.; Myers, J.M.; Johnson, O.W.; Busby, P.J.; Hard, J.J.; Bryant, G.J.; Waknitz, F.W.; Neely, K.; et al. Characterizing diversity in salmon from the Pacific Northwest. J. Fish Biol. 2001, 59, 1–41. [Google Scholar]
- Crozier, L. Impacts of Climate Change on Columbia River Salmon: A review of the scientific literature published in 2010. In Pages D1–D50 in Endangered Species Act Section 7(a)(2) Supplemental Biological Opinion: Consultation on Remand for Operation of the Federal Columbia River Power System; U.S. National Marine Fisheries Service: West Coast Regin, 1201 NE Lloyd Blvd, Ste 110, Portland, OR 97232, USA, 2011. [Google Scholar] [CrossRef]
- Crozier, L. Impacts of Climate Change on Columbia River Salmon: A review of the scientific literature published in 2011. In Pages D1–D50 in Endangered Species Act Section 7(a)(2) Supplemental Biological Opinion: Consultation on Remand for Operation of the Federal Columbia River Power System; U.S. National Marine Fisheries Service: West Coast Regin, 1201 NE Lloyd Blvd, Ste 110, Portland, OR 97232, USA, 2012. [Google Scholar]
- Crozier, L. Impacts of Climate Change on Columbia River Salmon: A review of the scientific literature published in 2012. In Pages D1–D50 in Endangered Species Act Section 7(a)(2) Supplemental Biological Opinion: Consultation on Remand for Operation of the Federal Columbia River Power System; U.S. National Marine Fisheries Service: West Coast Regin, 1201 NE Lloyd Blvd, Ste 110, Portland, OR 97232, USA, 2013. [Google Scholar]
- Crozier, L. Impacts of Climate Change on Columbia River Salmon: A review of the scientific literature published in 2013. In Pages D1–D50 in Endangered Species Act Section 7(a)(2) Supplemental Biological Opinion: Consultation on Remand for Operation of the Federal Columbia River Power System; U.S. National Marine Fisheries Service: West Coast Regin, 1201 NE Lloyd Blvd, Ste 110, Portland, OR 97232, USA, 2014. Available online: https://repository.library.noaa.gov/view/noaa/25641/noaa_25641_DS1.pdf (accessed on 14 June 2023).
- Crozier, L. Impacts of Climate Change on Columbia River Salmon: A review of the scientific literature published in 2014. In Pages D1–D50 in Endangered Species Act Section 7(a)(2) Supplemental Biological Opinion: Consultation on Remand for Operation of the Federal Columbia River Power System; U.S. National Marine Fisheries Service: West Coast Regin, 1201 NE Lloyd Blvd, Ste 110, Portland, OR 97232, USA, 2015. [Google Scholar]
- Crozier, L. Impacts of Climate Change on Columbia River Salmon: A review of the scientific literature published in 2015. In Pages D1–D50 in Endangered Species Act Section 7(a)(2) Supplemental Biological Opinion: Consultation on Remand for Operation of the Federal Columbia River Power System; U.S. National Marine Fisheries Service: West Coast Regin, 1201 NE Lloyd Blvd, Ste 110, Portland, OR 97232, USA, 2016. [Google Scholar]
- Crozier, L. Impacts of Climate Change on Columbia River Salmon: A review of the scientific literature published in 2016. In Pages D1–D50 in Endangered Species Act Section 7(a)(2) Supplemental Biological Opinion: Consultation on Remand for Operation of the Federal Columbia River Power System; U.S. National Marine Fisheries Service: West Coast Regin, 1201 NE Lloyd Blvd, Ste 110, Portland, OR 97232, USA, 2017. [Google Scholar]
- Crozier, L.G.; Siegel, J. Impacts of Climate Change on Columbia River Salmon: A review of the scientific literature published in 2017. In Pages D1–D50 in Endangered Species Act Section 7(a)(2) Supplemental Biological Opinion: Consultation on Remand for Operation of the Federal Columbia River Power System; U.S. National Marine Fisheries Service: West Coast Regin, 1201 NE Lloyd Blvd, Ste 110, Portland, OR 97232, USA, 2018. [Google Scholar] [CrossRef]
- Siegel, J.; Crozier, L.G. Impacts of climate change on Columbia River salmon: A review of the scientific literature published in 2018. In Pages D1–D50 in Endangered Species Act Section 7(a)(2) Supplemental Biological Opinion: Consultation on Remand for Operation of the Federal Columbia River Power System; U.S. National Marine Fisheries Service: West Coast Regin, 1201 NE Lloyd Blvd, Ste 110, Portland, OR 97232, USA, 2019. [Google Scholar] [CrossRef]
- Siegel, J.; Crozier, L.G. Impacts of Climate Change on Columbia River Salmon: A Review of The scientific Literature Published in 2019; U.S. National Marine Fisheries Service: West Coast Regin, 1201 NE Lloyd Blvd, Ste 110, Portland, OR 97232, USA, 2020. [Google Scholar] [CrossRef]
- Oesterwind, D.; Rau, A.; Zaiko, A. Drivers and pressures—Untangling the terms commonly used in marine science and policy. J. Environ. Manag. 2016, 181, 8–15. [Google Scholar] [CrossRef]
- Nielsen, J.L.; Ruggerone, G.T.; Zimmerman, C.E. Adaptive strategies and life history characteristics in a warming climate: Salmon in the Arctic? Environ. Biol. Fishes 2013, 96, 1187–1226. [Google Scholar] [CrossRef]
- IPCC. Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; Masson-Delmotte, V., Zhai, P., Pirani, A., Connors, S., Péan, C., Berger, S., Caud, N., Chen, Y., Goldfarb, L., Gomis, M., et al., Eds.; Cambridge University Press: Cambridge, UK, 2021; Available online: https://www.ipcc.ch/report/ar6/wg1/#FullReport (accessed on 1 January 2022).
- Herring, S.C.; Christidis, N.; Hoell, A.; Kossin, J.P.; Schreck II, C.J.; Stott, P.A. Explaining extreme events of 2016 from a climate perspective. Bull. Am. Meteorol. Soc. 2018, 99, S1–S157. [Google Scholar] [CrossRef]
- Mote, P.; Snover, A.K.; Capalbo, S.; Eigenbrode, S.D.; Glick, P.; Littell, J.; Raymondi, R.; Reeder, S. Northwest. In Climate Change Impacts in the United States: The Third National Climate Assessment; Melillo, J.M., Richmond, T.C., Yohe, G.W., Eds.; U.S. Global Change Research Program: Washington, DC, USA, 2014; pp. 487–513. [Google Scholar] [CrossRef]
- May, C.; Luce, C.; Casola, J.; Chang, M.; Cuhaciyan, J.; Dalton, M.; Lowe, S.; Morishima, G.; Mote, P.; Petersen, A.; et al. Northwest. In Climate Change Impacts in the United States: The Fourth National Climate Assessmentt; Reidmiller, D.R., Avery, C.W., Easterling, D.R., Kunkel, K.E., Lewis, K.L.M., Maycock, T.K., Stewart, B.C., Eds.; U.S. Global Change Research Program: Washington, DC, USA, 2018; Volume II, pp. 1036–1100. [Google Scholar] [CrossRef]
- Frankson, R.; Kunkel, K.E.; Champion, S.M.; Easterling, D.R.; Stevens, L.E.; Bumbaco, K.; Bond, N.; Casola, J.; Sweet, W. Washington State Climate Summary 2022. In NOAA Technical Report NESDIS 150-WA; NOAA/NESDIS: Silver Spring, MD, USA, 2022; p. 5. [Google Scholar]
- Runkle, J.; Kunkel, K.E.; Frankson, R.; Champion, S.M.; Stevens, L.E.; Abatzoglou, J. Idaho State Climate Summary 2022. In NOAA Technical Report NESDIS 150-ID; NOAA/NESDIS: Silver Spring, MD, USA, 2022; p. 4. [Google Scholar]
- Frankson, R.; Kunkel, K.E.; Champion, S.M.; Stevens, L.E.; Easterling, D.R.; Dello, K.; Dalton, M.; Sharp, D.; O’Neill, L. Oregon State Climate Summary 2022. In NOAA Technical Report NESDIS 150-OR; NOAA/NESDIS: Silver Spring, MD, USA, 2022; p. 5. [Google Scholar]
- Abatzoglou, J.T.; Rupp, D.E.; Mote, P.W. Seasonal climate variability and change in the Pacific Northwest of the United States. J. Clim. 2014, 27, 2125–2142. [Google Scholar] [CrossRef]
- Mutiibwa, D.; Vavrus, S.J.; McAfee, S.A.; Albright, T.P. Recent spatiotemporal patterns in temperature extremes across conterminous United States. J. Geophys. Res-Atmos. 2015, 120, 7378–7392. [Google Scholar] [CrossRef]
- Mera, R.; Massey, N.; Rupp, D.E.; Mote, P.; Allen, M.; Frumhoff, P.C. Climate change, climate justice and the application of probabilistic event attribution to summer heat extremes in the California Central Valley. Clim. Chang. 2015, 133, 427–438. [Google Scholar] [CrossRef]
- Durre, I.; Squires, M.F.; Vose, R.S.; Yin, X.G.; Arguez, A.; Applequist, S. NOAA’s 1981-2010 U.S. climate normals: Monthly precipitation, snowfall, and snow depth. J. Appl. Meteorol. Climatol. 2013, 52, 2377–2395. [Google Scholar] [CrossRef]
- Mao, Y.; Nijssen, B.; Lettenmaier, D.P. Is climate change implicated in the 2013- 2014 California drought? A hydrologic perspective. Geophys. Res. Lett. 2015, 42, 2805–2813. [Google Scholar] [CrossRef]
- Mote, P.W.; Li, S.; Lettenmaier, D.P.; Xiao, M.; Engel, R. Dramatic declines in snowpack in the western US. NPJ Clim. Atmos. Sci. 2018, 1, 2. [Google Scholar] [CrossRef]
- Lute, A.C.; Luce, C.H. Are Model Transferability And Complexity Antithetical? Insights From Validation of a Variable-Complexity Empirical Snow Model in Space and Time. Water Resour. Res. 2017, 53, 8825–8850. [Google Scholar] [CrossRef]
- Kluver, D.; Leathers, D. Regionalization of snowfall frequency and trends over the contiguous United States. Int. J. Climatol. 2015, 35, 4348–4358. [Google Scholar] [CrossRef]
- Malcomb, N.L.; Wiles, G.C. Tree-ring-based reconstructions of North American glacier mass balance through the Little Ice Age—Contemporary warming transition. Quat. Res. 2013, 79, 123–137. [Google Scholar] [CrossRef]
- Riedel, J.L.; Wilson, S.; Baccus, W.; Larrabee, M.; Fudge, T.J.; Fountain, A. Glacier status and contribution to streamflow in the Olympic Mountains, Washington, USA. J. Glaciol. 2015, 61, 8–16. [Google Scholar] [CrossRef]
- Marcinkowski, K.; Peterson, D.L. A 350-year reconstruction of the response of South Cascade Glacier to interannual and interdecadal climatic variability. Northwest Sci. 2015, 89, 14–33. [Google Scholar] [CrossRef]
- Fountain, A.G.; Gray, C.; Glenn, B.; Menounos, B.; Pflug, J.; Riedel, J.L. Glaciers of the Olympic Mountains, Washington—The Past and Future 100 Years. J. Geophys. Res. Earth Surf. 2022, 127, e2022JF006670. [Google Scholar] [CrossRef]
- Dittmer, K. Changing streamflow on Columbia Basin tribal lands-climate change and salmon. Clim. Chang. 2013, 120, 627–641. [Google Scholar] [CrossRef]
- Riche, O.; Johannessen, S.C.; Macdonald, R.W. Why timing matters in a coastal sea: Trends, variability and tipping points in the Strait of Georgia, Canada. J. Mar. Syst. 2014, 131, 36–53. [Google Scholar] [CrossRef]
- Hatcher, K.L.; Jones, J.A. Climate and streamflow trends in the Columbia River Basin: Evidence for ecological and engineering resilience to climate change. Atmos.-Ocean 2013, 51, 436–455. [Google Scholar] [CrossRef]
- Isaak, D.J.; Wollrab, S.; Horan, D.; Chandler, G. Climate change effects on stream and river temperatures across the northwest US from 1980-2009 and implications for salmonid fishes. Clim. Chang. 2012, 113, 499–524. [Google Scholar] [CrossRef]
- Diffenbaugh, N.S.; Swain, D.L.; Touma, D. Anthropogenic warming has increased drought risk in California. Proc. Natl. Acad. Sci. USA 2015, 112, 3931–3936. [Google Scholar] [CrossRef]
- Kormos, P.R.; Charles, H.L.; Seth, J.W.; Wouter, R.B. Trends and Sensitivities of Low Streamflow Extremes to Discharge Timing and Magnitude in Pacific Northwest Mountain Streams. Water Resour. Res. 2016, 52, 4990–5007. [Google Scholar] [CrossRef]
- Leppi, J.C.; Thomas, H.D.; Solomon, W.H.; Steven, W.R. Impacts of Climate Change on August Stream Discharge in the Central-Rocky Mountains. Clim. Change 2012, 112, 997–1014. [Google Scholar] [CrossRef]
- Luce, C.H. and Holden, Z.A. Declining Annual Streamflow Distributions in the Pacific Northwest United States, 1948–2006. Geophys. Res. Lett. 2009, 36, 1–6. [Google Scholar] [CrossRef]
- Reilly, M.J.; Dunn, C.J.; Meigs, G.W.; Spies, T.A.; Kennedy, R.E.; Bailey, J.D.; Briggs, K. Contemporary patterns of fire extent and severity in forests of the Pacific Northwest, USA (1985–2010). Ecosphere 2017, 8, e01695. [Google Scholar] [CrossRef]
- Johnston, J.D.; Dunn, C.J.; Vernon, M.J.; Bailey, J.D.; Morrissette, B.A.; Morici, K.E. Restoring historical forest conditions in a diverse inland Pacific Northwest landscape. Ecosphere 2018, 9, e02400. [Google Scholar] [CrossRef]
- Siegel, J.E.; Volk, C. Accurate spatiotemporal predictions of daily stream temperature from statistical models accounting for interactions between climate and landscape. PeerJ 2019, 7, e7892. [Google Scholar] [CrossRef]
- Islam, S.U.; Hay, R.W.; Dery, S.J.; Booth, B.P. Modelling the impacts of climate change on riverine thermal regimes in western Canada’s largest Pacific watershed. Sci. Rep. 2019, 9, 14. [Google Scholar] [CrossRef]
- Cheng, L.; Abraham, J.; Trenberth, K.E.; Fasullo, J.; Boyer, T.; Locarnini, R.; Zhang, B.; Yu, F.; Wan, L.; Chen, X.; et al. Upper Ocean Temperatures Hit Record High in 2020. Adv. Atmos. Sci. 2021, 38, 523–530. [Google Scholar] [CrossRef]
- Johnson, G.C.; Lumpkin, R. 2021: Global Oceans, in “State of the Climate in 2020”. Bull. Am. Meteorol. Soc. 2021, 102, S143–S198. [Google Scholar] [CrossRef]
- Johnstone, J.A.; Mantua, N.J. Atmospheric controls on northeast Pacific temperature variability and change, 1900-2012. Proc. Natl. Acad. Sci. USA 2014, 111, 14360–14365. [Google Scholar] [CrossRef]
- Cummins, P.F.; Masson, D. Climatic variability and trends in the surface waters of coastal British Columbia. Prog. Oceanogr. 2014, 120, 279–290. [Google Scholar] [CrossRef]
- Talloni-Alvarez, N.E.; Sumaila, U.R.; Le Billon, P.; Cheung, W.W.L. Climate change impact on Canada’s Pacific marine ecosystem: The current state of knowledge. Mar. Policy 2019, 104, 163–176. [Google Scholar] [CrossRef]
- Johnstone, J.A.; Mantua, N.J. Reply to Abatzoglou et al.: Atmospheric controls on northwest United States air temperatures, 1948-2012. Proc. Natl. Acad. Sci. USA 2014, 111, E5607–E5608. [Google Scholar] [CrossRef]
- Jewett, L.; Romanou, A. Ocean acidification and other ocean changes. In Climate Science Special Report: Fourth National Climate Assessment; Wuebbles, D.J., Fahey, D.W., Hibbard, K.A., Dokken, D.J., Stewart, B.C., Maycock, T.K., Eds.; U.S. Global Change Research Program: Washington, DC, USA, 2017; Volume I, pp. 364–392. [Google Scholar] [CrossRef]
- Feely, R.A.; Sabine, C.L.; Hernandez-Ayon, J.M.; Ianson, D.; Hales, B. Evidence for upwelling of corrosive "acidified" water onto the continental shelf. Science 2008, 320, 1490–1492. [Google Scholar] [CrossRef]
- Turi, G.; Lachkar, Z.; Gruber, N.; Nunnich, M. Climatic modulation of recent trends in ocean acidification in the California Current System. Environ. Res. Lett. 2016, 11, 17. [Google Scholar] [CrossRef]
- Wootton, J.T.; Pfister, C.A. Carbon system measurements and potential climatic drivers at a site of rapidly declining ocean pH. PLoS ONE 2012, 7, e53396. [Google Scholar] [CrossRef] [PubMed]
- Bond, N.A.; Cronin, M.F.; Freeland, H.; Mantua, N. Causes and impacts of the 2014 warm anomaly in the NE Pacific. Geophys. Res. Lett. 2015, 42, 3414–3420. [Google Scholar] [CrossRef]
- Amaya, D.J.; Miller, A.J.; Xie, S.-P.; Kosaka, Y. Physical drivers of the summer 2019 North Pacific marine heatwave. Nat. Commun. 2020, 11, 1903. [Google Scholar] [CrossRef]
- Jacox, M.G.; Alexander, M.A.; Mantua, N.J.; Scott, J.D.; Hervieux, G.; Webb, R.S.; Werner, F.E. Forcing of multiyear extreme ocean temperature that impacted California Current living marine resources in 2016. Bull. Am. Meteorol. Soc. 2018, 99, S27–S33. [Google Scholar] [CrossRef]
- Di Lorenzo, E.; Mantua, N. Multi-year persistence of the 2014/15 North Pacific marine heatwave. Nat. Clim. Chang. 2016, 6, 1042. [Google Scholar] [CrossRef]
- Peterson, W.T.; Fisher, J.L.; Strub, P.T.; Du, X.; Risien, C.; Peterson, J.; Shaw, C.T. The pelagic ecosystem in the Northern California Current off Oregon during the 2014-2016 warm anomalies within the context of the past 20 years. J. Geophys. Res. Ocean. 2017, 122, 7267–7290. [Google Scholar] [CrossRef] [PubMed]
- Sadowski, J.S.; Gonzalez, J.A.; Lonhart, S.I.; Jeppesen, R.; Grimes, T.M.; Grosholz, E.D. Temperature-induced range expansion of a subtropical crab along the California coast. Mar. Ecol. 2018, 39, e12528. [Google Scholar] [CrossRef]
- Brodeur, R.D.; Auth, T.D.; Phillips, A.J. Major Shifts in Pelagic Micronekton and Macrozooplankton Community Structure in an Upwelling Ecosystem Related to an Unprecedented Marine Heatwave. Front. Mar. Sci. 2019, 6, 15. [Google Scholar] [CrossRef]
- Cavole, L.M.; Demko, A.M.; Diner, R.E.; Giddings, A.; Koester, I.; Pagniello, C.; Paulsen, M.L.; Ramirez-Valdez, A.; Schwenck, S.M.; Yen, N.K.; et al. Biological Impacts of the 2013–2015 Warm-Water Anomaly in the Northeast Pacific. Oceanography 2016, 29, 273–285. [Google Scholar] [CrossRef]
- Moreira, M.; Hayes, D.S.; Boavida, I.; Schletterer, M.; Schmutz, S.; Pinheiro, A. Ecologically-based criteria for hydropeaking mitigation: A review. Sci. Total Environ. 2018, 657, 1508–1522. [Google Scholar] [CrossRef] [PubMed]
- Baker, M.R.; Matta, M.E.; Beaulieu, M.; Paris, N.; Huber, S.; Graham, O.J.; Pham, T.; Sisson, N.B.; Heller, C.P.; Witt, A.; et al. Intra-seasonal and inter-annual patterns in the demographics of sand lance and response to environmental drivers in the North Pacific. Mar. Ecol. Prog. Ser. 2019, 617, 221–244. [Google Scholar] [CrossRef]
- Brodeur, R.D.; Hunsicker, M.E.; Hann, A.; Miller, T.W. Effects of warming ocean conditions on feeding ecology of small pelagic fishes in a coastal upwelling ecosystem: A shift to gelatinous food sources. Mar. Ecol. Prog. Ser. 2019, 617, 149–163. [Google Scholar] [CrossRef]
- Auth, T.D.; Daly, E.A.; Brodeur, R.D.; Fisher, J.L. Phenological and distributional shifts in ichthyoplankton associated with recent warming in the northeast Pacific Ocean. Glob. Chang. Biol. 2018, 24, 259–272. [Google Scholar] [CrossRef]
- Swan, G.A. Chinook salmon spawning surveys in deep waters of a large, regulated river. Regul. Rivers Res. Manag. 1989, 4, 355–370. [Google Scholar] [CrossRef]
- Del Rio, A.M.; Mukai, G.N.; Martin, B.T.; Johnson, R.C.; Fangue, N.A.; Israel, J.A.; Todgham, A.E. Differential sensitivity to warming and hypoxia during development and long-term effects of developmental exposure in early life stage Chinook salmon. Conserv. Physiol. 2021, 9. [Google Scholar] [CrossRef]
- Gould, G.K.; Liu, M.L.; Barber, M.E.; Cherkauer, K.A.; Robichaud, P.R.; Adam, J.C. The effects of climate change and extreme wildfire events on runoff erosion over a mountain watershed. J. Hydrol. 2016, 536, 74–91. [Google Scholar] [CrossRef]
- Srivastava, A.; Brooks, E.S.; Dobre, M.; Elliot, W.J.; Wu, J.Q.; Flanagan, D.C.; Gravelle, J.A.; Link, T.E. Modeling forest management effects on water and sediment yield from nested, paired watersheds in the interior Pacific Northwest, USA using WEPP. Sci. Total Environ. 2020, 701, 134877. [Google Scholar] [CrossRef]
- Anttila, K.; Dhillon, R.S.; Boulding, E.G.; Farrell, A.P.; Glebe, B.D.; Elliott, J.A.K.; Wolters, W.R.; Schulte, P.M. Variation in temperature tolerance among families of Atlantic salmon (Salmo salar) is associated with hypoxia tolerance, ventricle size and myoglobin level. J. Exp. Biol. 2013, 216, 1183–1190. [Google Scholar] [CrossRef]
- Friedman, W.R.; Martin, B.T.; Wells, B.K.; Warzybok, P.; Michel, C.J.; Danner, E.M.; Lindley, S.T. Modeling composite effects of marine and freshwater processes on migratory species. Ecosphere 2019, 10, 21. [Google Scholar] [CrossRef]
- Weinheimer, J.; Anderson, J.H.; Downen, M.; Zimmerman, M.; Johnson, T. Monitoring climate impacts: Survival and migration timing of summer chum salmon in Salmon Creek, Washington. Trans. Am. Fish. Soc. 2017, 146, 983–995. [Google Scholar] [CrossRef]
- Narum, S.R.; Campbell, N.R.; Kozfkay, C.C.; Meyer, K.A. Adaptation of redband trout in desert and montane environments. Mol. Ecol. 2010, 19, 4622–4637. [Google Scholar] [CrossRef] [PubMed]
- Kavanagh, K.D.; Haugen, T.O.; Gregersen, F.; Jernvall, J.; Vollestad, L.A. Contemporary temperature-driven divergence in a Nordic freshwater fish under conditions commonly thought to hinder adaptation. BMC Evol. Biol. 2010, 10, 350. [Google Scholar] [CrossRef] [PubMed]
- Beer, W.N.; Steel, E.A. Impacts and implications of temperature variability on Chinook salmon egg development and emergence phenology. Trans. Am. Fish. Soc. 2018, 147, 3–15. [Google Scholar] [CrossRef]
- Fuhrman, A.E.; Larsen, D.A.; Steel, E.A.; Young, G.; Beckman, B.R. Chinook salmon emergence phenotypes: Describing the relationships between temperature, emergence timing and condition factor in a reaction norm framework. Ecol. Freshw. Fish 2018, 27, 350–362. [Google Scholar] [CrossRef]
- Lohmus, M.; Sundstrom, L.F.; Bjorklund, M.; Devlin, R.H. Genotype-temperature interaction in the regulation of development, growth, and morphometrics in wild-type, and growth-hormone transgenic coho salmon. PLoS ONE 2010, 5, e9980. [Google Scholar] [CrossRef]
- Whitney, C.K.; Hinch, S.G.; Patterson, D.A. Provenance matters: Thermal reaction norms for embryo survival among sockeye salmon Oncorhynchus nerka populations. J. Fish Biol. 2013, 82, 1159–1176. [Google Scholar] [CrossRef]
- Braun, D.C.; Patterson, D.A.; Reynolds, J.D. Maternal and environmental influences on egg size and juvenile life-history traits in Pacific salmon. Ecol. Evol. 2013, 3, 1727–1740. [Google Scholar] [CrossRef]
- Drinan, D.P.; Zale, A.V.; Webb, M.A.H.; Shepard, B.B.; Kalinowski, S.T. Evidence of local adaptation in westslope cutthroat trout. Trans. Am. Fish. Soc. 2012, 141, 872–880. [Google Scholar] [CrossRef]
- Goode, J.R.; Buffington, J.M.; Tonina, D.; Isaak, D.J.; Thurow, R.F.; Wenger, S.; Nagel, D.; Luce, C.; Tetzlaff, D.; Soulsby, C. Potential effects of climate change on streambed scour and risks to salmonid survival in snow-dominated mountain basins. Hydrol. Process. 2013, 27, 750–765. [Google Scholar] [CrossRef]
- MacDonald, R.J.; Boon, S.; Byrne, J.M.; Robinson, M.D.; Rasmussen, J.B. Potential future climate effects on mountain hydrology, stream temperature, and native salmonid life history. Can. J. Fish. Aquat. Sci. 2014, 71, 189–202. [Google Scholar] [CrossRef]
- Meyers, E.M.; Dobrowski, B.; Tague, C.L. Climate change impacts on flood frequency, intensity, and timing may affect trout species in Sagehen Creek, California. Trans. Am. Fish. Soc. 2010, 139, 1657–1664. [Google Scholar] [CrossRef]
- Sparks, M.M.; Falke, J.A.; Quinn, T.P.; Adkison, M.D.; Schindler, D.E.; Bartz, K.; Young, D.; Westley, P.A.H. Influences of spawning timing, water temperature, and climatic warming on early life history phenology in western Alaska sockeye salmon. Can. J. Fish. Aquat. Sci. 2019, 76, 123–135. [Google Scholar] [CrossRef]
- Beechie, T.; Imaki, H.; Greene, J.; Wade, A.; Wu, H.; Pess, G.; Roni, P.; Kimball, J.; Stanford, J.; Kiffney, P.; et al. Restoring salmon habitat for a changing climate. River Res. Appl. 2012, 29, 939–960. [Google Scholar] [CrossRef]
- Santiago, J.M.; Alonso, C.; de Jalon, D.G.; Solana-Gutierrez, J.; Munoz-Mas, R. Effects of climate change on the life stages of stream-dwelling brown trout (Salmo trutta Linnaeus, 1758) at the rear edge of their native distribution range. Ecohydrology 2020, 13, e2241. [Google Scholar] [CrossRef]
- Erkinaro, J.; Czorlich, Y.; Orell, P.; Kuusela, J.; Falkegard, M.; Lansman, M.; Pulkkinen, H.; Primmer, C.R.; Niemela, E. Life history variation across four decades in a diverse population complex of Atlantic salmon in a large subarctic river. Can. J. Fish. Aquat. Sci. 2019, 76, 42–55. [Google Scholar] [CrossRef]
- Satterthwaite, W.H.; Beakes, M.P.; Collins, E.M.; Swank, D.R.; Merz, J.E.; Titus, R.G.; Sogard, S.M.; Mangel, M. State-dependent life history models in a changing (and regulated) environment: Steelhead in the California Central Valley. Evol. Appl. 2010, 3, 221–243. [Google Scholar] [CrossRef]
- Kendall, N.W.; McMillan, J.R.; Sloat, M.R.; Buehrens, T.W.; Quinn, T.P.; Pess, G.R.; Kuzishchin, K.V.; McClure, M.M.; Zabel, R.W. Anadromy and residency in steelhead and rainbow trout (Oncorhynchus mykiss): A review of the processes and patterns. Can. J. Fish. Aquat. Sci. 2015, 72, 319–342. [Google Scholar] [CrossRef]
- Yvon-Durocher, G.; Montoya, J.M.; Trimmer, M.; Woodward, G. Warming alters the size spectrum and shifts the distribution of biomass in freshwater ecosystems. Glob. Chang. Biol. 2010, 17, 1681–1694. [Google Scholar] [CrossRef]
- Beveridge, O.S.; Petchey, O.L.; Humphries, S. Direct and indirect effects of temperature on the population dynamics and ecosystem functioning of aquatic microbial ecosystems. J. Anim. Ecol. 2010, 79, 1324–1331. [Google Scholar] [CrossRef]
- Jones, I.D.; Page, T.; Alex Elliott, J.; Thackeray, S.J.; Louise Heathwaite, A. Increases in lake phytoplankton biomass caused by future climate-driven changes to seasonal river flow. Glob. Chang. Biol. 2010, 17, 1809–1820. [Google Scholar] [CrossRef]
- Beakes, M.P.; Moore, J.W.; Hayes, S.A.; Sogard, S.M. Wildfire and the effects of shifting stream temperature on salmonids. Ecosphere 2014, 5, art63. [Google Scholar] [CrossRef]
- Howell, P.J. Changes in native bull trout and non-native brook trout distributions in the upper Powder River basin after 20 years, relationships to water temperature and implications of climate change. Ecol. Freshw. Fish 2018, 27, 710–719. [Google Scholar] [CrossRef]
- Dexter, E.; Bollens, S.M.; Rollwagen-Bollens, G.; Emerson, J.; Zimmerman, J. Persistent vs. ephemeral invasions: 8.5 years of zooplankton community dynamics in the Columbia River. Limnol. Oceanogr. 2015, 60, 527–539. [Google Scholar] [CrossRef]
- Emerson, J.E.; Bollens, S.M.; Counihan, T.D. Seasonal dynamics of zooplankton in Columbia-Snake River reservoirs, with special emphasis on the invasive copepod Pseudodiaptomus forbesi. Aquat. Invasions 2015, 10, 25–40. [Google Scholar] [CrossRef]
- Adams, J.B.; Bollens, S.M.; Bishop, J.G. Predation on the Invasive Copepod, Pseudodiaptomus forbesi, and Native Zooplankton in the Lower Columbia River: An Experimental Approach to Quantify Differences in Prey-Specific Feeding Rates. PLoS ONE 2015, 10, e0144095. [Google Scholar] [CrossRef]
- Haskell, C.A.; Beauchamp, D.A.; Bollens, S.M. Linking functional response and bioenergetics to estimate juvenile salmon growth in a reservoir food web. PLoS ONE 2017, 12, e0185933. [Google Scholar] [CrossRef]
- Hasselman, D.J.; Hinrichsen, R.A.; Shields, B.A.; Ebbesmeyer, C.C. American shad of the Pacific Coast: A harmful invasive species or benign introduction? Fisheries 2012, 37, 115–122. [Google Scholar] [CrossRef]
- Hinrichsen, R.A.; Hasselman, D.J.; Ebbesmeyer, C.C.; Shields, B.A. The Role of Impoundments, Temperature, and Discharge on Colonization of the Columbia River Basin, USA, by Nonindigenous American Shad. Trans. Am. Fish. Soc. 2013, 142, 887–900. [Google Scholar] [CrossRef]
- Hughes, R.M.; Herlihy, A.T. Patterns in catch per unit effort of native prey fish and alien piscivorous fish in 7 Pacific Northwest USA rivers. Fisheries 2012, 37, 201–211. [Google Scholar] [CrossRef]
- Christensen, D.R.; Moore, B.C. Largemouth bass consumption demand on hatchery rainbow trout in two Washington lakes. Lake Reserv. Manag. 2010, 26, 200–211. [Google Scholar] [CrossRef]
- Lawrence, D.J.; Olden, J.D.; Torgersen, C.E. Spatiotemporal patterns and habitat associations of smallmouth bass (Micropterus dolomieu) invading salmon-rearing habitat. Freshw. Biol. 2012, 57, 1929–1946. [Google Scholar] [CrossRef]
- Tabor, R.A.; Footen, B.A.; Fresh, K.L.; Celedonia, M.T.; Mejia, F.; Low, D.L.; Park, L. Smallmouth bass and largemouth bass predation on juvenile Chinook salmon and other salmonids in the Lake Washington Basin. N. Am. J. Fish. Manag. 2007, 27, 1174–1188. [Google Scholar] [CrossRef]
- Lawrence, D.J.; Beauchamp, D.A.; Olden, J.D. Life-stage-specific physiology defines invasion extent of a riverine fish. J. Anim. Ecol. 2015, 84, 879–888. [Google Scholar] [CrossRef]
- Kuehne, L.M.; Olden, J.D.; Duda, J.J. Costs of living for juvenile Chinook salmon (Oncorhynchus tshawytscha) in an increasingly warming and invaded world. Can. J. Fish. Aquat. Sci. 2012, 69, 1621–1630. [Google Scholar] [CrossRef]
- Boltana, S.; Sanhueza, N.; Aguilar, A.; Gallardo-Escarate, C.; Arriagada, G.; Valdes, J.A.; Soto, D.; Quinones, R.A. Influences of thermal environment on fish growth. Ecol. Evol. 2017, 7, 6814–6825. [Google Scholar] [CrossRef]
- Brewitt, K.S.; Danner, E.M.; Moore, J.W. Hot eats and cool creeks: Juvenile Pacific salmonids use mainstem prey while in thermal refuges. Can. J. Fish. Aquat. Sci. 2017, 74, 1588–1602. [Google Scholar] [CrossRef]
- Ohlberger, J.; Buehrens, T.W.; Brenkman, S.J.; Crain, P.; Quinn, T.P.; Hilborn, R. Effects of past and projected river discharge variability on freshwater production in an anadromous fish. Freshw. Biol. 2018, 63, 331–340. [Google Scholar] [CrossRef]
- Kock, T.J.; Liedtke, T.L.; Rondorf, D.W.; Serl, J.D.; Kohn, M.; Bumbaco, K.A. Elevated streamflows increase dam passage by juvenile coho salmon during winter: Implications of climate change in the Pacific Northwest. N. Am. J. Fish. Manag. 2012, 32, 1070–1079. [Google Scholar] [CrossRef]
- Penaluna, B.E.; Dunham, J.B.; Railsback, S.F.; Arismendi, I.; Johnson, S.L.; Bilby, R.E.; Safeeq, M.; Skaugset, A.E. Local Variability Mediates Vulnerability of Trout Populations to Land Use and Climate Change. PLoS ONE 2015, 10, e0135334. [Google Scholar] [CrossRef] [PubMed]
- Hardiman, J.M.; Mesa, M.G. The effects of increased stream temperatures on juvenile steelhead growth in the Yakima River Basin based on projected climate change scenarios. Clim. Chang. 2014, 124, 413–426. [Google Scholar] [CrossRef]
- Zhang, X.; Li, H.Y.; Deng, Z.Q.D.; Leung, L.R.; Skalski, J.R.; Cooke, S.J. On the variable effects of climate change on Pacific salmon. Ecol. Model. 2019, 397, 95–106. [Google Scholar] [CrossRef]
- Walters, A.W.; Bartz, K.K.; McClure, M.M. Interactive effects of water diversion and climate change for juvenile Chinook salmon in the Lemhi River Basin (U.S.A). Conserv. Biol. 2013, 27, 1179–1189. [Google Scholar] [CrossRef]
- Lawrence, D.J.; Stewart-Koster, B.; Olden, J.D.; Ruesch, A.S.; Torgersen, C.E.; Lawler, J.J.; Butcher, D.P.; Crown, J.K. The interactive effects of climate change, riparian management, and a nonnative predator on stream-rearing salmon. Ecol. Appl. 2014, 24, 895–912. [Google Scholar] [CrossRef] [PubMed]
- Hanson, K.C.; Peterson, D.P. Modeling the potential impacts of climate change on Pacific salmon culture programs: An example at Winthrop National Fish Hatchery. Environ. Manag. 2014, 54, 433–448. [Google Scholar] [CrossRef] [PubMed]
- Beer, W.N.; Anderson, J.J. Sensitivity of salmonid freshwater life history in western US streams to future climate conditions. Glob. Chang. Biol. 2013, 19, 2547–2556. [Google Scholar] [CrossRef] [PubMed]
- Kilduff, D.P.; Di Lorenzo, E.; Botsford, L.W.; Teo, S.L.H. Changing central Pacific El Niños reduce stability of North American salmon survival rates. Proc. Natl. Acad. Sci. USA 2015, 112, 10962–10966. [Google Scholar] [CrossRef] [PubMed]
- Mantua, N.J. Shifting patterns in Pacific climate, West Coast salmon survival rates, and increased volatility in ecosystem services. Proc. Natl. Acad. Sci. USA 2015, 112, 10823–10824. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.; Liermann, M. Using hierarchical models to estimate effects of ocean anomalies on north-west Pacific Chinook salmon Oncorhynchus tshawytscha recruitment. J. Fish Biol. 2010, 77, 1948–1963. [Google Scholar] [CrossRef]
- Sharma, R.; Quinn, T.P. Linkages between life history type and migration pathways in freshwater and marine environments for Chinook salmon, Oncorhynchus tshawytscha. Acta Oecol. -Int. J. Ecol. 2012, 41, 1–13. [Google Scholar] [CrossRef]
- Sharma, R.; Velez-Espino, L.A.; Wertheimer, A.C.; Mantua, N.; Francis, R.C. Relating spatial and temporal scales of climate and ocean variability to survival of Pacific Northwest Chinook salmon (Oncorhynchus tshawytscha). Fish. Oceanogr. 2013, 22, 14–31. [Google Scholar] [CrossRef]
- Stachura, M.M.; Mantua, N.J.; Scheuerell, M.D. Oceanographic influences on patterns in North Pacific salmon abundance. Can. J. Fish. Aquat. Sci. 2014, 71, 226–235. [Google Scholar] [CrossRef]
- Tucker, S.; Thiess, M.E.; Morris, J.F.T.; Mackas, D.; Peterson, W.T.; Candy, J.R.; Beacham, T.D.; Iwamoto, E.M.; Teel, D.J.; Peterson, M.; et al. Coastal Distribution and Consequent Factors Influencing Production of Endangered Snake River Sockeye Salmon. Trans. Am. Fish. Soc. 2015, 144, 107–123. [Google Scholar] [CrossRef]
- Malick, M.J.; Cox, S.P.; Mueter, F.J.; Peterman, R.M. Linking phytoplankton phenology to salmon productivity along a north-south gradient in the Northeast Pacific Ocean. Can. J. Fish. Aquat. Sci. 2015, 72, 697–708. [Google Scholar] [CrossRef]
- Li, S.; Wu, L.; Yang, Y.; Geng, T.; Cai, W.; Gan, B.; Chen, Z.; Jing, Z.; Wang, G.; Ma, X. The Pacific Decadal Oscillation less predictable under greenhouse warming. Nat. Clim. Chang. 2020, 10, 30–34. [Google Scholar] [CrossRef]
- Osborne, E.B.; Thunell, R.C.; Gruber, N.; Feely, R.A.; Benitez-Nelson, C.R. Decadal variability in twentieth-century ocean acidification in the California Current Ecosystem. Nat. Geosci. 2020, 13, 43–49. [Google Scholar] [CrossRef]
- IPCC. IPCC Special Report on the Ocean and Cryosphere in a Changing Climate; Poörtner, H.-O., Roberts, D.C., Masson-Delmotte, V., Zhai, P., Tignor, M., Poloczanska, E., Mintenbeck, K., Alegría, A., Nicolai, M., Okem, A., et al., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2019; p. 755. [Google Scholar] [CrossRef]
- Litzow, M.A.; Ciannelli, L.; Cunningham, C.J.; Johnson, B.; Puerta, P. Nonstationary effects of ocean temperature on Pacific salmon productivity. Can. J. Fish. Aquat. Sci. 2019, 76, 1923–1928. [Google Scholar] [CrossRef]
- Kilduff, D.P.; Botsford, L.W.; Teo, S.L.H. Spatial and temporal covariability in early ocean survival of Chinook salmon (Oncorhynchus tshawytscha) along the west coast of North America. ICES J. Mar. Sci. 2014, 71, 1671–1682. [Google Scholar] [CrossRef]
- Gosselin, J.L.; Crozier, L.G.; Burke, B.J. Shifting signals: Correlations among freshwater, marine and climatic indices often investigated in Pacific salmon studies. Ecol. Indic. 2021, 121, 107167. [Google Scholar] [CrossRef]
- Wainwright, T.C. Ephemeral relationships in salmon forecasting: A cautionary tale. Prog. Oceanogr. 2021, 193, 102522. [Google Scholar] [CrossRef]
- Alava, J.J.; Cisneros-Montemayor, A.M.; Sumaila, U.R.; Cheung, W.W.L. Projected amplification of food web bioaccumulation of MeHg and PCBs under climate change in the Northeastern Pacific. Sci. Rep. 2018, 8, 13460. [Google Scholar] [CrossRef]
- Busch, D.S.; McElhany, P. Estimates of the direct effect of seawater pH on the survival rate of species groups in the California Current Ecosystem. PLoS ONE 2016, 11, 28. [Google Scholar] [CrossRef]
- Marshall, K.N.; Kaplan, I.C.; Hodgson, E.E.; Hermann, A.; Busch, D.S.; Mcelhany, P.; Essington, T.E.; Harvey, C.J.; Fulton, E.A. Risks of ocean acidification in the California Current food web and fisheries: Ecosystem model projections. Glob. Chang. Biol. 2017, 23, 1525–1539. [Google Scholar] [CrossRef]
- Williams, C.R.; Dittman, A.H.; McElhany, P.; Busch, D.S.; Maher, M.T.; Bammler, T.K.; MacDonald, J.W.; Gallagher, E.P. Elevated CO2 impairs olfactory-mediated neural and behavioral responses and gene expression in ocean-phase coho salmon (Oncorhynchus kisutch). Glob. Chang. Biol. 2019, 25, 963–977. [Google Scholar] [CrossRef]
- McCormick, S.D.; Regish, A.M. Effects of ocean acidification on salinity tolerance and seawater growth of Atlantic salmon Salmo salar smolts. J. Fish Biol. 2018, 93, 560–566. [Google Scholar] [CrossRef] [PubMed]
- Ou, M.; Hamilton, T.J.; Eom, J.; Lyall, E.M.; Gallup, J.; Jiang, A.; Lee, J.; Close, D.A.; Yun, S.S.; Brauner, C.J. Responses of pink salmon to CO2-induced aquatic acidification. Nat. Clim. Chang. 2015, 5, 950–955. [Google Scholar] [CrossRef]
- Frommel, A.Y.; Carless, J.; Hunt, B.P.V.; Brauner, C.J. Physiological resilience of pink salmon to naturally occurring ocean acidification. Conserv. Physiol. 2020, 8, coaa059. [Google Scholar] [CrossRef]
- Keinänen, M.; Nikonen, S.; Käkelä, R.; Ritvanen, T.; Rokka, M.; Myllylä, T.; Pönni, J.; Vuorinen, P.J. High Lipid Content of Prey Fish and n−3 PUFA Peroxidation Impair the Thiamine Status of Feeding-Migrating Atlantic Salmon (Salmo salar) and Is Reflected in Hepatic Biochemical Indices. Biomolecules 2022, 12, 526. [Google Scholar] [CrossRef]
- Phillips, E.M.; Horne, J.K.; Zamon, J.E. Characterizing juvenile salmon predation risk during early marine residence. PLoS ONE 2021, 16, e0247241. [Google Scholar] [CrossRef] [PubMed]
- Moore, M.E.; Berejikian, B.A.; Greene, C.M.; Munsch, S. Environmental fluctuation and shifting predation pressure contribute to substantial variation in early marine survival of steelhead. Mar. Ecol. Prog. Ser. 2021, 662, 139–156. [Google Scholar] [CrossRef]
- Muhling, B.A.; Brodie, S.; Smith, J.A.; Tommasi, D.; Gaitan, C.F.; Hazen, E.L.; Jacox, M.G.; Auth, T.D.; Brodeur, R.D. Predictability of species distributions deteriorates under novel environmental conditions in the California Current System. Front. Mar. Sci. 2020, 7, 589. [Google Scholar] [CrossRef]
- Tolimieri, N.; Wallace, J.; Haltuch, M. Spatio-temporal patterns in juvenile habitat for 13 groundfishes in the California Current Ecosystem. PLoS ONE 2020, 15, e0237996. [Google Scholar] [CrossRef] [PubMed]
- Malick, M.J.; Hunsicker, M.E.; Haltuch, M.A.; Parker-Stetter, S.L.; Berger, A.M.; Marshall, K.N. Relationships between temperature and Pacific hake distribution vary across latitude and life-history stage. Mar. Ecol. Prog. Ser. 2020, 639, 185–197. [Google Scholar] [CrossRef]
- Brodeur, R.D.; Buchanan, J.C.; Emmett, R.L. Pelagic and demersal fish predators on juvenile and adult forage fish predators in the northern California Current: Spatial and temporal variations. Calif. Coop. Ocean. Fish. Investig. Rep. 2014, 55, 96–116. [Google Scholar]
- Litz, M.N.C.; Emmett, R.L.; Bentley, P.J.; Claiborne, A.M.; Barcelo, C. Biotic and abiotic factors influencing forage fish and pelagic nekton community in the Columbia River plume (USA) throughout the upwelling season 1999–2009. ICES J. Mar. Sci. 2014, 71, 5–18. [Google Scholar] [CrossRef]
- Kaltenberg, A.M.; Emmett, R.L.; Benoit-Bird, K.J. Timing of forage fish seasonal appearance in the Columbia River plume and link to ocean conditions. Mar. Ecol. Prog. Ser. 2010, 419, 171–184. [Google Scholar] [CrossRef]
- Ruzicka, J.J.; Brink, K.H.; Gifford, D.J.; Bahr, F. A physically coupled end-to-end model platform for coastal ecosystems: Simulating the effects of climate change and changing upwelling characteristics on the Northern California Current ecosystem. Ecol. Model. 2016, 331, 86–99. [Google Scholar] [CrossRef]
- Ruzicka, J.J.; Daly, E.A.; Brodeur, R.D. Evidence that summer jellyfish blooms impact Pacific Northwest salmon production. Ecosphere 2016, 7, 22. [Google Scholar] [CrossRef]
- Goldstein, J.; Steiner, U.K. Ecological drivers of jellyfish blooms—The complex life history of a ‘well-known’ medusa (Aurelia aurita). J. Anim. Ecol. 2020, 89, 910–920. [Google Scholar] [CrossRef]
- Siddique, A.; Purushothaman, J.; Madhusoodhanan, R.; Raghunathan, C. The rising swarms of jellyfish in Indian waters: The environmental drivers, ecological, and socio-economic impacts. J. Water Clim. Chang. 2022, 13, 3747–3759. [Google Scholar] [CrossRef]
- Heneghan, R.F.; Everett, J.D.; Blanchard, J.L.; Sykes, P.; Richardson, A.J. Climate-driven zooplankton shifts cause large-scale declines in food quality for fish. Nat. Clim. Chang. 2023, 13, 470–477. [Google Scholar] [CrossRef]
- Lee, S.H.; Tseng, L.; Yoon, Y.H.; Ramirez-Romero, E.; Hwang, J.S.; Molinero, J.C. The global spread of jellyfish hazards mirrors the pace of human imprint in the marine environment. Environ. Int. 2023, 171, 107699. [Google Scholar] [CrossRef] [PubMed]
- Fiechter, J.; Santora, J.A.; Chavez, F.; Northcott, D.; Messie, M. Krill Hotspot Formation and Phenology in the California Current Ecosystem. Geophys. Res. Lett. 2020, 47, e2020GL088039. [Google Scholar] [CrossRef] [PubMed]
- Fiechter, J.; Huff, D.D.; Martin, B.T.; Jackson, D.W.; Edwards, C.A.; Rose, K.A.; Curchitser, E.N.; Hedstrom, K.S.; Lindley, S.T.; Wells, B.K. Environmental conditions impacting juvenile Chinook salmon growth off central California: An ecosystem model analysis. Geophys. Res. Lett. 2015, 42, 2910–2917. [Google Scholar] [CrossRef]
- Fiechter, J.; Huckstadt, L.A.; Rose, K.A.; Costa, D.P. A fully coupled ecosystem model to predict the foraging ecology of apex predators in the California Current. Mar. Ecol. Prog. Ser. 2016, 556, 273–285. [Google Scholar] [CrossRef]
- Shelton, A.O.; Satterthwaite, W.H.; Ward, E.J.; Feist, B.E.; Burke, B. Using hierarchical models to estimate stock-specific and seasonal variation in ocean distribution, survivorship, and aggregate abundance of fall run Chinook salmon. Can. J. Fish. Aquat. Sci. 2019, 76, 95–108. [Google Scholar] [CrossRef]
- Weitkamp, L.A.; Teel, D.J.; Liermann, M.; Hinton, S.A.; Van Doornik, D.M.; Bentley, P.J. Stock-Specific Size and Timing at Ocean Entry of Columbia River Juvenile Chinook Salmon and Steelhead: Implications for Early Ocean Growth. Mar. Coast. Fish. 2015, 7, 370–392. [Google Scholar] [CrossRef]
- Teel, D.J.; Burke, B.J.; Kuligowski, D.R.; Morgan, C.A.; Van Doornik, D.M. Genetic Identification of Chinook Salmon: Stock-Specific Distributions of Juveniles along the Washington and Oregon Coasts. Mar. Coast. Fish. 2015, 7, 274–300. [Google Scholar] [CrossRef]
- Gosselin, J.L.; Buhle, E.R.; Van Holmes, C.; Beer, W.N.; Iltis, S.; Anderson, J.J. Role of carryover effects in conservation of wild Pacific salmon migrating regulated rivers. Ecosphere 2021, 12, e03618. [Google Scholar] [CrossRef]
- Haeseker, S.L.; McCann, J.A.; Tuomikoski, J.; Chockley, B. Assessing Freshwater and Marine Environmental Influences on Life-Stage-Specific Survival Rates of Snake River Spring-Summer Chinook Salmon and Steelhead. Trans. Am. Fish. Soc. 2012, 141, 121–138. [Google Scholar] [CrossRef]
- Russell, I.C.; Aprahamian, M.W.; Barry, J.; Davidson, I.C.; Fiske, P.; Ibbotson, A.T.; Kennedy, R.J.; Maclean, J.C.; Moore, A.; Otero, J.; et al. The influence of the freshwater environment and the biological characteristics of Atlantic salmon smolts on their subsequent marine survival. ICES J. Mar. Sci. 2012, 69, 1563–1573. [Google Scholar] [CrossRef]
- Lundin, J.I.; Spromberg, J.A.; Jorgensen, J.C.; Myers, J.M.; Chittaro, P.M.; Zabel, R.W.; Johnson, L.L.; Neely, R.M.; Scholz, N.L. Legacy habitat contamination as a limiting factor for Chinook salmon recovery in the Willamette Basin, Oregon, USA. PLoS ONE 2019, 14, e0214399. [Google Scholar] [CrossRef] [PubMed]
- Thorstad, E.B.; Uglem, I.; Finstad, B.; Kroglund, F.; Einarsdottir, I.E.; Kristensen, T.; Diserud, O.; Arechavala-Lopez, P.; Mayer, I.; Moore, A.; et al. Reduced marine survival of hatchery-reared Atlantic salmon post-smolts exposed to aluminium and moderate acidification in freshwater. Estuar. Coast. Shelf Sci. 2013, 124, 34–43. [Google Scholar] [CrossRef]
- Wells, M.L.; Trainer, V.L.; Smayda, T.J.; Karlson, B.S.; Trick, C.G.; Kudela, R.M.; Ishikawa, A.; Bernard, S.; Wulff, A.; Anderson, D.M.; et al. Harmful algal blooms and climate change: Learning from the past and present to forecast the future. Harmful Algae 2015, 49, 68–93. [Google Scholar] [CrossRef] [PubMed]
- Huisman, J.; Codd, G.A.; Paerl, H.W.; Ibelings, B.W.; Verspagen, J.M.H.; Visser, P.M. Cyanobacterial blooms. Nat. Rev. Microbiol. 2018, 16, 471–483. [Google Scholar] [CrossRef] [PubMed]
- Esenkulova, S.; Suchy, K.D.; Pawlowicz, R.; Costa, M.; Pearsall, I.A. Harmful Algae and Oceanographic Conditions in the Strait of Georgia, Canada Based on Citizen Science Monitoring. Front. Mar. Sci. 2021, 8, 725092. [Google Scholar] [CrossRef]
- Rensel, J.E.J.; Haigh, N.; Tynan, T.J. Fraser river sockeye salmon marine survival decline and harmful blooms of Heterosigrna akashiwo. Harmful Algae 2010, 10, 98–115. [Google Scholar] [CrossRef]
- Burridge, L.E.; Martin, J.L.; Lyons, M.C.; LeGresley, M.M. Lethality of microalgae to farmed Atlantic salmon (Salmo salar). Aquaculture 2010, 308, 101–105. [Google Scholar] [CrossRef]
- Lefebvre, K.A.; Frame, E.R.; Kendrick, P.S. Domoic acid and fish behavior: A review. Harmful Algae 2012, 13, 126–130. [Google Scholar] [CrossRef]
- McCabe, R.M.; Hickey, B.M.; Kudela, R.M.; Lefebvre, K.A.; Adams, N.G.; Bill, B.D.; Gulland, F.M.; Thomson, R.E.; Cochlan, W.P.; Trainer, V.L. An unprecedented coastwide toxic algal bloom linked to anomalous ocean conditions. Geophys. Res. Lett. 2016, 43, 10366–10376. [Google Scholar] [CrossRef]
- Akmajian, A.M.; Scordino, J.J.; Acevedo-Gutierrez, A. Year-round algal toxin exposure in free-ranging sea lions. Mar. Ecol. Prog. Ser. 2017, 583, 243–258. [Google Scholar] [CrossRef]
- Moore, S.K.; Johnstone, J.A.; Banas, N.S.; Salathe, E.P. Present-day and future climate pathways affecting Alexandrium blooms in Puget Sound, WA, USA. Harmful Algae 2015, 48, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Ralston, D.K.; Moore, S.K. Modeling harmful algal blooms in a changing climate. Harmful Algae 2020, 91, 101729. [Google Scholar] [CrossRef] [PubMed]
- Costa, P.R. Impact and effects of paralytic shellfish poisoning toxins derived from harmful algal blooms to marine fish. Fish Fish. 2016, 17, 226–248. [Google Scholar] [CrossRef]
- Sato, S.; Ogata, T.; Kodama, M. Trace amounts of saxitoxins in the viscera of chum salmon Oncorhynchus keta. Mar. Ecol. Prog. Ser. 1998, 175, 295–298. [Google Scholar] [CrossRef]
- Brosnan, I.G.; Welch, D.W.; Rechisky, E.L.; Porter, A.D. Evaluating the influence of environmental factors on yearling Chinook salmon survival in the Columbia River plume (USA). Mar. Ecol. Prog. Ser. 2014, 496, 181–196. [Google Scholar] [CrossRef]
- Rechisky, E.L.; Porter, A.D.; Clark, T.D.; Furey, N.B.; Gale, M.K.; Hinch, S.G.; Welch, D.W. Quantifying survival of age-2 Chilko Lake sockeye salmon during the first 50 days of migration. Can. J. Fish. Aquat. Sci. 2019, 76, 136–152. [Google Scholar] [CrossRef]
- Seitz, A.C.; Courtney, M.B.; Evans, M.D.; Manishin, K. Pop-up satellite archival tags reveal evidence of intense predation on large immature Chinook salmon (Oncorhynchus tshawytscha) in the North Pacific Ocean. Can. J. Fish. Aquat. Sci. 2019, 76, 1608–1615. [Google Scholar] [CrossRef]
- Friedman, W.R.; Santora, J.A.; Schroeder, I.D.; Huff, D.D.; Brodeur, R.D.; Field, J.C.; Wells, B.K. Environmental and geographic relationships among salmon forage assemblages along the continental shelf of the California Current. Mar. Ecol. Prog. Ser. 2018, 596, 181–198. [Google Scholar] [CrossRef]
- Henderson, M.; Fiechter, J.; Huff, D.D.; Wells, B.K. Spatial variability in ocean-mediated growth potential is linked to Chinook salmon survival. Fish. Oceanogr. 2018, 28, 334–344. [Google Scholar] [CrossRef]
- Wells, B.K.; Santora, J.A.; Schroeder, I.D.; Mantua, N.; Sydeman, W.J.; Huff, D.D.; Field, J.C. Marine ecosystem perspectives on Chinook salmon recruitment: A synthesis of empirical and modeling studies from a California upwelling system. Mar. Ecol. Prog. Ser. 2016, 552, 271–284. [Google Scholar] [CrossRef]
- Dale, K.E.; Daly, E.A.; Brodeur, R.D. Interannual variability in the feeding and condition of subyearling Chinook salmon off Oregon and Washington in relation to fluctuating ocean conditions. Fish. Oceanogr. 2017, 26, 1–16. [Google Scholar] [CrossRef]
- Daly, E.A.; Auth, T.D.; Brodeur, R.D.; Peterson, W.T. Winter ichthyoplankton biomass as a predictor of early summer prey fields and survival of juvenile salmon in the northern California Current. Mar. Ecol. Prog. Ser. 2013, 484, 203–217. [Google Scholar] [CrossRef]
- Beaugrand, G.; Reid, P.C. Relationships between North Atlantic salmon, plankton, and hydroclimatic change in the Northeast Atlantic. ICES J. Mar. Sci. 2012, 69, 1549–1562. [Google Scholar] [CrossRef]
- Thompson, S.A.; Sydeman, W.J.; Santora, J.A.; Black, B.A.; Suryan, R.M.; Calambokidis, J.; Peterson, W.T.; Bograd, S.J. Linking predators to seasonality of upwelling: Using food web indicators and path analysis to infer trophic connections. Prog. Oceanogr. 2012, 101, 106–120. [Google Scholar] [CrossRef]
- Tomaro, L.M.; Teel, D.J.; Peterson, W.T.; Miller, J.A. When is bigger better? Early marine residence of middle and upper Columbia River spring Chinook salmon. Mar. Ecol. Prog. Ser. 2012, 452, 237–252. [Google Scholar] [CrossRef]
- Trueman, C.N.; MacKenzie, K.M.; Palmer, M.R. Stable isotopes reveal linkages between ocean climate, plankton community dynamics, and survival of two populations of Atlantic salmon (Salmo salar). ICES J. Mar. Sci. 2012, 69, 784–794. [Google Scholar] [CrossRef]
- Sabal, M.C.; Huff, D.D.; Henderson, M.J.; Fiechter, J.; Harding, J.A.; Hayes, S.A. Contrasting patterns in growth and survival of Central Valley fall run Chinook salmon related to hatchery and ocean conditions. Environ. Biol. Fishes 2016, 99, 949–967. [Google Scholar] [CrossRef]
- Woodson, L.E.; Wells, B.K.; Weber, P.K.; MacFarlane, R.B.; Whitman, G.E.; Johnson, R.C. Size, growth, and origin-dependent mortality of juvenile Chinook salmon Oncorhynchus tshawytscha during early ocean residence. Mar. Ecol. Prog. Ser. 2013, 487, 163–175. [Google Scholar] [CrossRef]
- Wells, B.K.; Santora, J.A.; Henderson, M.J.; Warzybok, P.; Jahncke, J.; Bradley, R.W.; Huff, D.D.; Schroeder, I.D.; Nelson, P.; Field, J.C.; et al. Environmental conditions and prey-switching by a seabird predator impact juvenile salmon survival. J. Mar. Syst. 2017, 174, 54–63. [Google Scholar] [CrossRef]
- Daly, E.A.; Brodeur, R.D. Warming Ocean Conditions Relate to Increased Trophic Requirements of Threatened and Endangered Salmon. PLoS ONE 2015, 10, e0144066. [Google Scholar] [CrossRef] [PubMed]
- Farley, E.V.; Starovoytov, A.; Naydenko, S.; Heintz, R.; Trudel, M.; Guthrie, C.; Eisner, L.; Guyon, J.R. Implications of a warming eastern Bering Sea for Bristol Bay sockeye salmon. ICES J. Mar. Sci. 2011, 68, 1138–1146. [Google Scholar] [CrossRef]
- Turrero, P.; Horreo, J.L.; Garcia-Vazquez, E. Same old Salmo? Changes in life history and demographic trends of North Iberian salmonids since the Upper Palaeolithic as revealed by archaeological remains and beast analyses. Mol. Ecol. 2012, 21, 2318–2329. [Google Scholar] [CrossRef] [PubMed]
- Chaput, G. Overview of the status of Atlantic salmon (Salmo salar) in the North Atlantic and trends in marine mortality. ICES J. Mar. Sci. 2012, 69, 1538–1548. [Google Scholar] [CrossRef]
- Sinclair-Waters, M.; Nome, T.; Wang, J.; Lien, S.; Kent, M.P.; Saerov, H.; Floro-Larsen, B.; Bolstad, G.H.; Primmer, C.R.; Barson, N.J. Dissecting the loci underlying maturation timing in Atlantic salmon using haplotype and multi-SNP based association methods. Heredity 2022, 129, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Czorlich, Y.; Aykanat, T.; Erkinaro, J.; Orell, P.; Primmer, C.R. Rapid evolution in salmon life history induced by direct and indirect effects of fishing. Science 2022, 376, 420–423. [Google Scholar] [CrossRef]
- Czorlich, Y.; Aykanat, T.; Erkinaro, J.; Orell, P.; Primmer, C.R. Rapid sex-specific evolution of age at maturity is shaped by genetic architecture in Atlantic salmon. Nat. Ecol. Evol. 2018, 2, 1800–1807. [Google Scholar] [CrossRef]
- Ohlberger, J.; Ward, E.J.; Schindler, D.E.; Lewis, B. Demographic changes in Chinook salmon across the Northeast Pacific Ocean. Fish Fish. 2018, 19, 533–546. [Google Scholar] [CrossRef]
- Oke, K.B.; Cunningham, C.J.; Westley, P.A.H.; Baskett, M.L.; Carlson, S.M.; Clark, J.; Hendry, A.P.; Karatayev, V.A.; Kendall, N.W.; Kibele, J.; et al. Recent declines in salmon body size impact ecosystems and fisheries. Nat. Commun. 2020, 11, 4155. [Google Scholar] [CrossRef]
- Kendall, N.W.; Quinn, T.P. Quantifying and comparing size selectivity among Alaskan sockeye salmon fisheries. Ecol. Appl. 2012, 22, 804–816. [Google Scholar] [CrossRef]
- Waples, R.S.; Audzijonyte, A. Fishery-induced evolution provides insights into adaptive responses of marine species to climate change. Front. Ecol. Environ. 2016, 14, 217–224. [Google Scholar] [CrossRef]
- Otero, J.; Jensen, A.J.; L’Abee-Lund, J.H.; Stenseth, N.C.; Storvik, G.O.; Vollestad, L.A. Contemporary ocean warming and freshwater conditions are related to later sea age at maturity in Atlantic salmon spawning in Norwegian rivers. Ecol. Evol. 2012, 2, 2192–2203. [Google Scholar] [CrossRef] [PubMed]
- Chasco, B.E.; Kaplan, I.C.; Thomas, A.C.; Acevedo-Gutiérrez, A.; Noren, D.P.; Ford, M.J.; Hanson, M.B.; Scordino, J.J.; Jeffries, S.J.; Marshall, K.N.; et al. Competing tradeoffs between increasing marine mammal predation and fisheries harvest of Chinook salmon. Sci. Rep. 2017, 7, 15439. [Google Scholar] [CrossRef] [PubMed]
- Moore, M.E.; Berejikian, B.A. Population, habitat, and marine location effects on early marine survival and behavior of Puget Sound steelhead smolts. Ecosphere 2017, 8, e01834. [Google Scholar] [CrossRef]
- Moore, M.E.; Berejikian, B.A.; Goetz, F.A.; Berger, A.G.; Hodgson, S.S.; Connor, E.J.; Quinn, T.P. Multi-population analysis of Puget Sound steelhead survival and migration behavior. Mar. Ecol. Prog. Ser. 2015, 537, 217–232. [Google Scholar] [CrossRef]
- Mantyniemi, S.; Romakkaniemi, A.; Dannewitz, J.; Palm, S.; Pakarinen, T.; Pulkkinen, H.; Grdmark, A.; Karlsson, O. Both predation and feeding opportunities may explain changes in survival of Baltic salmon post-smolts. ICES J. Mar. Sci. 2012, 69, 1574–1579. [Google Scholar] [CrossRef]
- Ford, J.K.B.; Ellis, G.M. Selective foraging by fish-eating killer whales Orcinus orca in British Columbia. Mar. Ecol. Prog. Ser. 2006, 316, 185–199. [Google Scholar] [CrossRef]
- Manishin, K.A.; Cunningham, C.J.; Westley, P.A.H.; Seitz, A.C. Can late stage marine mortality explain observed shifts in age structure of Chinook salmon? PLoS ONE 2021, 16, e0247370. [Google Scholar] [CrossRef]
- Shelton, A.O.; Sullaway, G.H.; Ward, E.J.; Feist, B.E.; Somers, K.A.; Tuttle, V.J.; Watson, J.T.; Satterthwaite, W.H. Redistribution of salmon populations in the northeast Pacific ocean in response to climate. Fish Fish. 2021, 22, 503–517. [Google Scholar] [CrossRef]
- Kao, Y.C.; Madenjian, C.P.; Bunnell, D.B.; Lofgren, B.M.; Perroud, M. Temperature effects induced by climate change on the growth and consumption by salmonines in Lakes Michigan and Huron. Environ. Biol. Fishes 2015, 98, 1089–1104. [Google Scholar] [CrossRef]
- Abdul-Aziz, O.I.; Mantua, N.J.; Myers, K.W. Potential climate change impacts on thermal habitats of Pacific salmon (Oncorhynchus spp.) in the North Pacific Ocean and adjacent seas. Can. J. Fish. Aquat. Sci. 2011, 68, 1660–1680. [Google Scholar] [CrossRef]
- Cheung, W.W.L.; Dunne, J.; Sarmiento, J.L.; Pauly, D. Integrating ecophysiology and plankton dynamics into projected maximum fisheries catch potential under climate change in the Northeast Atlantic. ICES J. Mar. Sci. J. Du Cons. 2011, 68, 1008–1018. [Google Scholar] [CrossRef]
- Cheung, W.W.L.; Frolicher, T.L. Marine heatwaves exacerbate climate change impacts for fisheries in the northeast Pacific. Sci. Rep. 2020, 10, 6678. [Google Scholar] [CrossRef]
- Cheung, W.W.L.; Lam, V.W.Y.; Sarmiento, J.L.; Kearney, K.; Watson, R.E.G.; Zeller, D.; Pauly, D. Large-scale redistribution of maximum fisheries catch potential in the global ocean under climate change. Glob. Chang. Biol. 2010, 16, 24–35. [Google Scholar] [CrossRef]
- Ainsworth, C.H.; Samhouri, J.F.; Busch, D.S.; Chueng, W.W.L.; Dunne, J.; Okey, T.A. Potential impacts of climate change on Northeast Pacific marine fisheries and food webs. ICES J. Mar. Sci. 2011, 68, 1217–1229. [Google Scholar] [CrossRef]
- Blanchard, J.L.; Jennings, S.; Holmes, R.; Harle, J.; Merino, G.; Allen, J.I.; Holt, J.; Dulvy, N.K.; Barange, M. Potential consequences of climate change for primary production and fish production in large marine ecosystems. Philos. Trans. R. Soc. B Biol. Sci. 2012, 367, 2979–2989. [Google Scholar] [CrossRef]
- Heneghan, R.F.; Galbraith, E.; Blanchard, J.L.; Harrison, C.; Barrier, N.; Bulman, C.; Cheung, W.; Coll, M.; Eddy, T.D.; Erauskin-Extramiana, M.; et al. Disentangling diverse responses to climate change among global marine ecosystem models. Prog. Oceanogr. 2021, 198, 102659. [Google Scholar] [CrossRef]
- Reum, J.C.P.; Blanchard, J.L.; Holsman, K.K.; Aydin, K.; Hollowed, A.B.; Hermann, A.J.; Cheng, W.; Faig, A.; Haynie, A.C.; Punt, A.E. Ensemble Projections of Future Climate Change Impacts on the Eastern Bering Sea Food Web Using a Multispecies Size Spectrum Model. Front. Mar. Sci. 2020, 7, 124. [Google Scholar] [CrossRef]
- Hazen, E.L.; Jorgensen, S.; Rykaczewski, R.R.; Bograd, S.J.; Foley, D.G.; Jonsen, I.D.; Shaffer, S.A.; Dunne, J.P.; Costa, D.P.; Crowder, L.B.; et al. Predicted habitat shifts of Pacific top predators in a changing climate. Nat. Clim. Chang. 2012, 3, 234–238. [Google Scholar] [CrossRef]
- Wolf, S.G.; Snyder, M.A.; Sydeman, W.J.; Doak, D.F.; Croll, D.A. Predicting population consequences of ocean climate change for an ecosystem sentinel, the seabird Cassin’s auklet. Glob. Chang. Biol. 2010, 16, 1923–1935. [Google Scholar] [CrossRef]
- Bednarsek, N.; Pelletier, G.; Ahmed, A.; Feely, R.A. Chemical Exposure Due to Anthropogenic Ocean Acidification Increases Risks for Estuarine Calcifiers in the Salish Sea: Biogeochemical Model Scenarios. Front. Mar. Sci. 2020, 7, 580. [Google Scholar] [CrossRef]
- Comeau, S.; Gattuso, J.-P.; Nisumaa, A.-M.; Orr, J. Impact of aragonite saturation state changes on migratory pteropods. Proc. R. Soc. B Biol. Sci. 2012, 279, 732–738. [Google Scholar] [CrossRef] [PubMed]
- Comeau, S.; Jeffree, R.; Teyssie, J.L.; Gattuso, J.P. Response of the arctic pteropod Limacina helicina to projected future environmental conditions. PLoS ONE 2010, 5, e11362. [Google Scholar] [CrossRef]
- Flynn, K.J.; Blackford, J.C.; Baird, M.E.; Raven, J.A.; Clark, D.R.; Beardall, J.; Brownlee, C.; Fabian, H.; Wheeler, G.L. Changes in pH at the exterior surface of plankton with ocean acidification. Nat. Clim. Chang. 2012, 2, 510–513. [Google Scholar] [CrossRef]
- Magel, C.L.; Lee, E.M.J.; Strawn, A.M.; Swieca, K.; Jensen, A.D. Connecting Crabs, Currents, and Coastal Communities: Examining the Impacts of Changing Ocean Conditions on the Distribution of US West Coast Dungeness Crab Commercial Catch. Front. Mar. Sci. 2020, 7, 401. [Google Scholar] [CrossRef]
- Anttila, K.; Eliason, E.J.; Kaukinen, K.H.; Miller, K.M.; Farrell, A.P. Facing warm temperatures during migration: Cardiac mRNA responses of two adult Oncorhynchus nerka populations to warming and swimming challenges. J. Fish Biol. 2014, 84, 1439–1456. [Google Scholar] [CrossRef]
- Fellman, J.B.; Hood, E.; Nagorski, S.; Hudson, J.; Pyare, S. Interactive physical and biotic factors control dissolved oxygen in salmon spawning streams in coastal Alaska. Aquat. Sci. 2019, 81, 11. [Google Scholar] [CrossRef]
- Keefer, M.L.; Taylor, G.A.; Garletts, D.F.; Gauthier, G.A.; Pierce, T.M.; Caudil, C.C. Prespawn mortality in adult spring Chinook salmon outplanted above barrier dams. Ecol. Freshw. Fish 2010, 19, 361–372. [Google Scholar] [CrossRef]
- Sergeant, C.J.; Bellmore, J.R.; McConnell, C.; Moore, J.W. High salmon density and low discharge create periodic hypoxia in coastal rivers. Ecosphere 2017, 8, e01846. [Google Scholar] [CrossRef]
- Tillotson, M.D.; Quinn, T.P. Climate and conspecific density trigger pre-spawning mortality in sockeye salmon (Oncorhynchus nerka). Fish. Res. 2017, 188, 138–148. [Google Scholar] [CrossRef]
- Bell, D.A.; Kovach, R.P.; Vulstek, S.C.; Joyce, J.E.; Tallmon, D.A. Climate-induced trends in predator–prey synchrony differ across life-history stages of an anadromous salmonid. Can. J. Fish. Aquat. Sci. 2017, 74, 1431–1438. [Google Scholar] [CrossRef]
- Rub, A.M.W.; Sandford, B.P. Evidence of a ‘dinner bell’ effect from acoustic transmitters in adult Chinook salmon. Mar. Ecol. Prog. Ser. 2020, 641, 1–11. [Google Scholar] [CrossRef]
- Rub, A.M.W.; Som, N.A.; Henderson, M.J.; Sandford, B.P.; Van Doornik, D.M.; Teel, D.J.; Tennis, M.J.; Langness, O.P.; van der Leeuw, B.K.; Huff, D.D. Changes in adult Chinook salmon (Oncorhynchus tshawytscha) survival within the lower Columbia River amid increasing pinniped abundance. Can. J. Fish. Aquat. Sci. 2019, 76, 1862–1873. [Google Scholar] [CrossRef]
- Sergeant, C.J.; Armstrong, J.B.; Ward, E.J. Predator-prey migration phenologies remain synchronised in a warming catchment. Freshw. Biol. 2015, 60, 724–732. [Google Scholar] [CrossRef]
- Keefer, M.L.; Jepson, M.A.; Naughton, G.P.; Blubaugh, T.J.; Clabough, T.S.; Caudill, C.C. Condition-dependent en route migration mortality of adult Chinook salmon in the Willamette River main stem. N. Am. J. Fish. Manag. 2017, 37, 370–379. [Google Scholar] [CrossRef]
- Keefer, M.L.; Caudill, C.C.; Peery, C.A.; Moser, M.L. Context-dependent diel behavior of upstream-migrating anadromous fishes. Environ. Biol. Fishes 2013, 96, 691–700. [Google Scholar] [CrossRef]
- Benda, S.E.; Naughton, G.P.; Caudill, C.C.; Kent, M.L.; Schreck, C.B. Cool, Pathogen-Free Refuge Lowers Pathogen-Associated Prespawn Mortality of Willamette River Chinook Salmon. Trans. Am. Fish. Soc. 2015, 144, 1159–1172. [Google Scholar] [CrossRef]
- Keefer, M.L.; Caudill, C.C. Estimating thermal exposure of adult summer steelhead and fall Chinook salmon migrating in a warm impounded river. Ecol. Freshw. Fish 2016, 25, 599–611. [Google Scholar] [CrossRef]
- Goetz, F.A.; Quinn, T.P. Behavioral thermoregulation by adult Chinook salmon (Oncorhynchus tshawytscha) in estuary and freshwater habitats prior to spawning. Fish. Bull. 2019, 117, 258–271. [Google Scholar] [CrossRef]
- Hasler, C.T.; Cooke, S.J.; Hinch, S.G.; Guimond, E.; Donaldson, M.R.; Mossop, B.; Patterson, D.A. Thermal biology and bioenergetics of different upriver migration strategies in a stock of summer-run Chinook salmon. J. Therm. Biol. 2012, 37, 265–272. [Google Scholar] [CrossRef]
- Keefer, M.L.; Clabough, T.S.; Jepson, M.A.; Bowerman, T.; Caudill, C.C. Temperature and depth profiles of Chinook salmon and the energetic costs of their long-distance homing migrations. J. Therm. Biol. 2019, 79, 155–165. [Google Scholar] [CrossRef]
- Keefer, M.L.; Clabough, T.S.; Jepson, M.A.; Johnson, E.L.; Peery, C.A.; Caudill, C.C. Thermal exposure of adult Chinook salmon and steelhead: Diverse behavioral strategies in a large and warming river system. PLoS ONE 2018, 13, e0204274. [Google Scholar] [CrossRef] [PubMed]
- Lennox, R.J.; Eliason, E.J.; Havn, T.B.; Johansen, M.R.; Thorstad, E.B.; Cooke, S.J.; Diserud, O.H.; Whoriskey, F.G.; Farrell, A.P.; Uglem, I. Bioenergetic consequences of warming rivers to adult Atlantic salmon Salmo salar during their spawning migration. Freshw. Biol. 2018, 63, 1381–1393. [Google Scholar] [CrossRef]
- O’Sullivan, A.M.; Linnansaari, T.; Leavitt, J.; Samways, K.M.; Kurylyk, B.L.; Curry, R.A. The salmon-peloton: Hydraulic habitat shifts of adult Atlantic salmon (Salmo salar) due to behavioural thermoregulation. River Res. Appl. 2022, 38, 107–118. [Google Scholar] [CrossRef]
- Siegel, J.E.; Crozier, L.G.; Wiesebron, L.E.; Widener, D.L. Environmentally triggered shifts in steelhead migration behavior and consequences for survival in the mid-Columbia River. PLoS ONE 2021, 16, e0250831. [Google Scholar] [CrossRef] [PubMed]
- Strange, J.S. Upper thermal limits to migration in adult Chinook salmon: Evidence from the Klamath River Basin. Trans. Am. Fish. Soc. 2010, 139, 1091–1108. [Google Scholar] [CrossRef]
- Keefer, M.R.L.; Boggs, C.T.; Peery, C.; Caudill, C.C. Overwintering distribution, behavior, and survival of adult summer steelhead: Variability among Columbia river populations. N. Am. J. Fish. Manag. 2008, 28, 81–96. [Google Scholar] [CrossRef]
- Berejikian, B.A.; Tatara, C.P.; Van Doornik, D.M.; Humling, M.A.; Cooper, M.R.; Pasley, C.R.; Atkins, J.J. Duration in captivity affects competitive ability and breeding success of male but not female steelhead trout (Oncorhynchus mykiss). Can. J. Fish. Aquat. Sci. 2020, 77, 1000–1009. [Google Scholar] [CrossRef]
- Bond, M.H.; Westley, P.A.H.; Dittman, A.H.; Holecek, D.; Marsh, T.; Quinn, T.P. Combined effects of barge transportation, river environment, and rearing location on straying and migration of adult Snake River fall-run Chinook salmon. Trans. Am. Fish. Soc. 2016, 146, 60–73. [Google Scholar] [CrossRef]
- Crozier, L.; Burke, B.; Sandford, B.; Axel, G.; Sanderson, B. Passage and Survival of Adult Snake River Sockeye Salmon within and Upstream from the Federal Columbia River Power System; Fish Ecology Division, Northwest Fisheries Science Center National Marine Fisheries Service: Seattle, WA, USA, 2014. [Google Scholar]
- Pankhurst, N.W.; King, H.R. Temperature and salmonid reproduction: Implications for aquaculture. J. Fish Biol. 2010, 76, 69–85. [Google Scholar] [CrossRef]
- Burnett, N.J.; Hinch, S.G.; Bett, N.N.; Braun, D.C.; Casselman, M.T.; Cooke, S.J.; Gelchu, A.; Lingard, S.; Middleton, C.T.; Minke-Martin, V.; et al. Reducing carryover effects on the migration and spawning success of Sockeye salmon through a management experiment of dam flows. River Res. Appl. 2017, 33, 3–15. [Google Scholar] [CrossRef]
- Todd, C.D.; Friedland, K.D.; MacLean, J.C.; Whyte, B.D.; Russell, I.C.; Lonergan, M.E.; Morrissey, M.B. Phenological and phenotypic changes in Atlantic salmon populations in response to a changing climate. ICES J. Mar. Sci. 2012, 69, 1686–1698. [Google Scholar] [CrossRef]
- Jacobs, G.R.; Thurow, R.F.; Buffington, J.M.; Isaak, D.J.; Wenger, S.J. Climate, fire regime, geomorphology, and conspecifics influence the spatial distribution of Chinook salmon redds. Trans. Am. Fish. Soc. 2021, 150, 8–23. [Google Scholar] [CrossRef]
- Hague, M.J.; Ferrari, M.R.; Miller, J.R.; Patterson, D.A.; Russell, G.L.; Farrell, A.P.; Hinch, S.G. Modelling the future hydroclimatology of the lower Fraser River and its impacts on the spawning migration survival of sockeye salmon. Glob. Chang. Biol. 2011, 17, 87–98. [Google Scholar] [CrossRef]
- Boughton, D.A.; Pike, A.S. Floodplain rehabilitation as a hedge against hydroclimatic uncertainty in a migration corridor of threatened steelhead. Conserv. Biol. 2013, 27, 1158–1168. [Google Scholar] [CrossRef] [PubMed]
- Flitcroft, R.; Lewis, S.; Arismendi, I.; Davis, C.; Giannico, G.; Penaluna, B.; Santelmann, M.; Safeeq, M.; Snyder, J. Using expressed behaviour of coho salmon (Oncorhynchus kisutch) to evaluate the vulnerability of upriver migrants under future hydrological regimes: Management implications and conservation planning. Aquat. Conserv-Mar. Freshw. Ecosyst. 2019, 29, 1083–1094. [Google Scholar] [CrossRef]
- Donley, E.E.; Naiman, R.J.; Marineau, M.D. Strategic planning for instream flow restoration: A case study of potential climate change impacts in the central Columbia River basin. Glob. Chang. Biol. 2012, 18, 3071–3086. [Google Scholar] [CrossRef]
- Shanley, C.S.; Albert, D.M. Climate change sensitivity index for Pacific salmon habitat in southeast Alaska. PLoS ONE 2014, 9, e104799. [Google Scholar] [CrossRef]
- Connor, W.P.; Piston, C.E.; Garcia, A.P. Temperature during incubation as one factor affecting the distribution of Snake River Fall Chinook salmon spawning areas. Trans. Am. Fish. Soc. 2003, 132, 1236–1243. [Google Scholar] [CrossRef]
- Pitman, K.J.; Moore, J.W.; Huss, M.; Sloat, M.R.; Whited, D.C.; Beechie, T.J.; Brenner, R.; Hood, E.W.; Milner, A.M.; Pess, G.R.; et al. Glacier retreat creating new Pacific salmon habitat in western North America. Nat. Commun. 2021, 12, 6816. [Google Scholar] [CrossRef]
- Sinnatamby, R.N.; Cantin, A.; Post, J.R. Threats to at-risk salmonids of the Canadian Rocky Mountain region. Ecol. Freshw. Fish 2019, 18, 477–494. [Google Scholar] [CrossRef]
- Al-Chokhachy, R.; Lien, M.K.; Shepard, B.B.; High, B. The interactive effects of stream temperature, stream size, and non-native species on Yellowstone cutthroat trout. Can. J. Fish. Aquat. Sci. 2021, 78, 1073–1083. [Google Scholar] [CrossRef]
- Labonne, J.; Vignon, M.; Prevost, E.; Lecomte, F.; Dodson, J.J.; Kaeuffer, R.; Aymes, J.C.; Jarry, M.; Gaudin, P.; Davaine, P.; et al. Invasion dynamics of a fish-free landscape by brown trout (Salmo trutta). PLoS ONE 2013, 8, e71052. [Google Scholar] [CrossRef] [PubMed]
- Moyle, P.; Kiernan, J.; Crain, P.; Quiñones, R. Climate Change Vulnerability of Native and Alien Freshwater Fishes of California: A Systematic Assessment Approach. PLoS ONE 2013, 8, e63883. [Google Scholar] [CrossRef] [PubMed]
- Almodovar, A.; Ayllon, D.; Nicola, G.G.; Jonsson, B.; Elvira, B. Climate-driven biophysical changes in feeding and breeding environments explain the decline of southernmost European Atlantic salmon populations. Can. J. Fish. Aquat. Sci. 2019, 76, 1581–1595. [Google Scholar] [CrossRef]
- Cunningham, C.J.; Westley, P.A.H.; Adkison, M.D. Signals of large scale climate drivers, hatchery enhancement, and marine factors in Yukon River Chinook salmon survival revealed with a Bayesian life history model. Glob. Chang. Biol. 2018, 24, 4399–4416. [Google Scholar] [CrossRef] [PubMed]
- Honkanen, H.M.; Boylan, P.; Dodd, J.A.; Adams, C.E. Life stage-specific, stochastic environmental effects overlay density dependence in an Atlantic salmon population. Ecol. Freshw. Fish 2019, 28, 156–166. [Google Scholar] [CrossRef]
- Irvine, J.R.; Michielsens, C.J.G.; O’Brien, M.; White, B.A.; Folkes, M. Increasing dominance of odd-year returning pink salmon. Trans. Am. Fish. Soc. 2014, 143, 939–956. [Google Scholar] [CrossRef]
- Jeffrey, K.M.; Côté, I.M.; Irvine, J.R.; Reynolds, J.D. Changes in body size of Canadian Pacific salmon over six decades. Can. J. Fish. Aquat. Sci. 2017, 74, 191–201. [Google Scholar] [CrossRef]
- Dorner, B.; Catalano, M.J.; Peterman, R.M. Spatial and temporal patterns of covariation in productivity of Chinook salmon populations of the northeastern Pacific Ocean. Can. J. Fish. Aquat. Sci. 2018, 75, 1082–1095. [Google Scholar] [CrossRef]
- Friedland, K.D.; Shank, B.V.; Todd, C.D.; McGinnity, P.; Nye, J.A. Differential response of continental stock complexes of Atlantic salmon (Salmo salar) to the Atlantic Multidecadal Oscillation. J. Mar. Syst. 2014, 133, 77–87. [Google Scholar] [CrossRef]
- Jorgensen, J.C.; Nicol, C.; Fogel, C.; Beechie, T.J. Identifying the potential of anadromous salmonid habitat restoration with life cycle models. PLoS ONE 2021, 16, e0256792. [Google Scholar] [CrossRef]
- Nicol, C.L.; Jorgensen, J.C.; Fogel, C.B.; Timpane-Padgham, B.; Beechie, T.J. Spatially overlapping salmon species have varied population response to early life history mortality from increased peak flows. Can. J. Fish. Aquat. Sci. 2022, 79, 342–351. [Google Scholar] [CrossRef]
- Scheuerell, M.D.; Ruff, C.P.; Anderson, J.H.; Beamer, E.M. An integrated population model for estimating the relative effects of natural and anthropogenic factors on a threatened population of steelhead trout. J. Appl. Ecol. 2021, 58, 114–124. [Google Scholar] [CrossRef]
- Olmos, M.; Massiot-Granier, F.; Prevost, E.; Chaput, G.; Bradbury, I.R.; Nevoux, M.; Rivot, E. Evidence for spatial coherence in time trends of marine life history traits of Atlantic salmon in the North Atlantic. Fish Fish. 2019, 20, 322–342. [Google Scholar] [CrossRef]
- Madsen, R.P.A.; Jacobsen, M.W.; O’Malley, K.G.; Nygaard, R.; Praebel, K.; Jonsson, B.; Pujolar, J.M.; Fraser, D.J.; Bernatchez, L.; Hansen, M.M. Genetic population structure and variation at phenologyrelated loci in anadromous Arctic char (Salvelinus alpinus). Ecol. Freshw. Fish 2020, 29, 170–183. [Google Scholar] [CrossRef]
- Lehnert, S.J.; Kess, T.; Bentzen, P.; Kent, M.P.; Lien, S.; Gilbey, J.; Clement, M.; Jeffery, N.W.; Waples, R.S.; Bradbury, I.R. Genomic signatures and correlates of widespread population declines in salmon. Nat. Commun. 2019, 10, 10. [Google Scholar] [CrossRef] [PubMed]
- Kannry, S.H.; O’Rourke, S.M.; Kelson, S.J.; Miller, M.R. On the ecology and distribution of Steelhead (Oncorhynchus mykiss) in California’s Eel River. J. Hered. 2020, 111, 548–563. [Google Scholar] [CrossRef] [PubMed]
- Kelson, S.J.; Miller, M.R.; Thompson, T.Q.; O’Rourke, S.M.; Carlson, S.M. Temporal dynamics of migration-linked genetic variation are driven by streamflows and riverscape permeability. Mol. Ecol. 2020, 29, 870–885. [Google Scholar] [CrossRef]
- Schindler, D.E.; Armstrong, J.B.; Reed, T.E. The portfolio concept in ecology and evolution. Front. Ecol. Environ. 2015, 13, 257–263. [Google Scholar] [CrossRef]
- Anderson, S.C.; Moore, J.W.; McClure, M.M.; Dulvy, N.K.; Cooper, A.B. Portfolio conservation of metapopulations under climate change. Ecol. Appl. 2015, 25, 559–572. [Google Scholar] [CrossRef]
- Wade, A.A.; Beechie, T.J.; Fleishman, E.; Mantua, N.J.; Wu, H.; Kimball, J.S.; Stoms, D.M.; Stanford, J.A. Steelhead vulnerability to climate change in the Pacific Northwest. J. Appl. Ecol. 2013, 50, 1093–1104. [Google Scholar] [CrossRef]
- Wade, A.A.; Hand, B.K.; Kovach, R.P.; Luikart, G.; Whited, D.C.; Muhlfeld, C.C. Accounting for adaptive capacity and uncertainty in assessments of species’ climate-change vulnerability. Conserv. Biol. 2017, 31, 136–149. [Google Scholar] [CrossRef] [PubMed]
- Crozier, L.G.; McClure, M.M.; Beechie, T.; Bograd, S.J.; Boughton, D.A.; Carr, M.; Cooney, T.D.; Dunham, J.B.; Greene, C.M.; Haltuch, M.A.; et al. Climate vulnerability assessment for Pacific salmon and steelhead in the California Current Large Marine Ecosystem. PLoS ONE 2019, 14, e0217711. [Google Scholar] [CrossRef] [PubMed]
- Almodovar, A.; Nicola, G.G.; Ayllon, D.; Elvira, B. Global warming threatens the persistence of Mediterranean brown trout. Glob. Chang. Biol. 2012, 18, 1549–1560. [Google Scholar] [CrossRef]
- Ayllon, D.; Nicola, G.G.; Elvira, B.; Almodovar, A. Climate change will render size-selective harvest of cold-water fish species unsustainable in Mediterranean freshwaters. J. Appl. Ecol. 2021, 58, 562–575. [Google Scholar] [CrossRef]
- Neville, H.M.; Leasure, D.R.; Dauwalter, D.C.; Dunham, J.B.; Bjork, R.; Fesenmyer, K.A.; Chelgren, N.D.; Peacock, M.M.; Luce, C.H.; Isaak, D.J.; et al. Application of multiple-population viability analysis to evaluate species recovery alternatives. Conserv. Biol. 2019, 34, 482–493. [Google Scholar] [CrossRef]
- Roberts, J.J.; Fausch, K.D.; Peterson, D.P.; Hooten, M.B. Fragmentation and thermal risks from climate change interact to affect persistence of native trout in the Colorado River basin. Glob. Chang. Biol. 2013, 19, 1383–1398. [Google Scholar] [CrossRef]
- Piou, C.; Prevost, E. Contrasting effects of climate change in continental vs. oceanic environments on population persistence and microevolution of Atlantic salmon. Glob. Chang. Biol. 2013, 19, 711–723. [Google Scholar] [CrossRef]
- Piou, C.; Taylor, M.H.; Papaix, J.; Prevost, E. Modelling the interactive effects of selective fishing and environmental change on Atlantic salmon demogenetics. J. Appl. Ecol. 2015, 52, 1629–1637. [Google Scholar] [CrossRef]
- Battle, L.; Chang, H.Y.; Tzeng, C.S.; Lin, H.J. Modeling the impact of dam removal on the Formosan landlocked salmon in the context of climate change. Aquat. Sci. 2020, 82, 3. [Google Scholar] [CrossRef]
- Leppi, J.C.; Rinella, D.J.; Wilson, R.R.; Loya, W.M. Linking climate change projections for an Alaskan watershed to future coho salmon production. Glob. Chang. Biol. 2014, 20, 1808–1820. [Google Scholar] [CrossRef] [PubMed]
- McHugh, P.A.; Saunders, W.C.; Bouwes, N.; Wall, C.E.; Bangen, S.; Wheaton, J.M.; Nahorniak, M.; Ruzycki, J.R.; Tattam, I.A.; Jordan, C.E. Linking models across scales to assess the viability and restoration potential of a threatened population of steelhead (Oncorhynchus mykiss) in the Middle Fork John Day River, Oregon, USA. Ecol. Model. 2017, 355, 24–38. [Google Scholar] [CrossRef]
- Battin, J.; Wiley, M.W.; Ruckelshaus, M.H.; Palmer, R.N.; Korb, E.; Bartz, K.K.; Imaki, H. Projected impacts of climate change on salmon habitat restoration. Proc. Natl. Acad. Sci. USA 2007, 104, 6720–6725. [Google Scholar] [CrossRef]
- Honea, J.M.; McClure, M.M.; Jorgensen, J.C.; Scheuerell, M.D. Assessing freshwater life-stage vulnerability of an endangered Chinook salmon population to climate change influences on stream habitat. Clim. Res. 2016, 71, 127–137. [Google Scholar] [CrossRef]
- Pankhurst, N.W.; Munday, P.L. Effects of climate change on fish reproduction and early life history stages. Mar. Freshw. Res. 2011, 62, 1015–1026. [Google Scholar] [CrossRef]
- Tuor, K.M.F.; Shrimpton, J.M. Differences in water temperatures experienced by embryo and larval Coho Salmon (Oncorhynchus kisutch) across their geographic range in British Columbia. Environ. Biol. Fishes 2019, 102, 955–967. [Google Scholar] [CrossRef]
- Roni, P.; Johnson, C.; De Boer, T.; Pess, G.; Dittman, A.; Sear, D. Interannual variability in the effects of physical habitat and parentage on Chinook salmon egg-to-fry survival. Can. J. Fish. Aquat. Sci. 2016, 73, 1047–1059. [Google Scholar] [CrossRef]
- Campbell, E.Y.; Dunham, J.B.; Reeves, G.H.; Wondzell, S.M. Phenology of hatching, emergence, and end-of-season body size in young-of-year coho salmon in thermally contrasting streams draining the Copper River Delta, Alaska. Can. J. Fish. Aquat. Sci. 2019, 76, 185–191. [Google Scholar] [CrossRef]
- Adelfio, L.A.; Wondzell, S.M.; Mantua, N.J.; Reeves, G.H. Warm winters reduce landscape-scale variability in the duration of egg incubation for coho salmon (Oncorhynchus kisutch) on the Copper River Delta, Alaska. Can. J. Fish. Aquat. Sci. 2019, 76, 1362–1375. [Google Scholar] [CrossRef]
- Evans, M.L.; Neff, B.D.; Heath, D.D. Quantitative genetic and translocation experiments reveal genotype-by-environment effects on juvenile life-history traits in two populations of Chinook salmon (Oncorhynchus tshawytscha). J. Evol. Biol. 2010, 23, 687–698. [Google Scholar] [CrossRef] [PubMed]
- Venney, C.J.; Love, O.P.; Drown, E.J.; Heath, D.D. DNA Methylation Profiles Suggest Intergenerational Transfer of Maternal Effects. Mol. Biol. Evol. 2020, 37, 540–548. [Google Scholar] [CrossRef] [PubMed]
- Janhunen, M.; Piironen, J.; Peuhkuri, N. Parental effects on embryonic viability and growth in Arctic charr Salvelinus alpinus at two incubation temperatures. J. Fish Biol. 2010, 76, 2558–2570. [Google Scholar] [CrossRef] [PubMed]
- Le Luyer, J.; Laporte, M.; Beacham, T.D.; Kaukinen, K.H.; Withler, R.E.; Leong, J.S.; Rondeau, E.B.; Koop, B.F.; Bernatchez, L. Parallel epigenetic modifications induced by hatchery rearing in a Pacific salmon. Proc. Natl. Acad. Sci. USA 2017, 114, 12964–12969. [Google Scholar] [CrossRef]
- Gavery, M.R.; Roberts, S.B. Epigenetic considerations in aquaculture. PeerJ 2017, 5, e4147. [Google Scholar] [CrossRef]
- Eirin-Lopez, J.M.; Putnam, H.M. Marine environmental epigenetics. Annu. Rev. Mar. Sci. 2019, 11, 335–368. [Google Scholar] [CrossRef]
- Bollati, V.; Baccarelli, A. Environmental epigenetics. Heredity 2010, 105, 105–112. [Google Scholar] [CrossRef]
- Banet, A.I.; Healy, S.J.; Eliason, E.J.; Roualdes, E.A.; Patterson, D.A.; Hinch, S.G. Simulated maternal stress reduces offspring aerobic swimming performance in Pacific salmon. Conserv. Physiol. 2019, 7, coz095. [Google Scholar] [CrossRef]
- Daley, J.M.; Leadley, T.A.; Pitcher, T.E.; Drouillard, K.G. Bioamplification and the selective depletion of persistent organic pollutants in Chinook salmon larvae. Environ. Sci. Technol. 2012, 46, 2420–2426. [Google Scholar] [CrossRef]
- Burt, J.M.; Hinch, S.G.; Patterson, D.A. Parental identity influences progeny responses to incubation thermal stress in sockeye salmon Onchorhynchus nerka. J. Fish Biol. 2012, 80, 444–462. [Google Scholar] [CrossRef]
- Leblanc, C.A.; Horri, K.; Skulason, S.; Benhaim, D. Subtle temperature increase can interact with individual size and social context in shaping phenotypic traits of a coldwater fish. PLoS ONE 2019, 14, 21. [Google Scholar] [CrossRef] [PubMed]
- Pasquet, A.; Realis-Doyelle, E.; Fontaine, P.; Teletchea, F. What can we learn by studying dead fish fry? Aquac. Res. 2019, 50, 1824–1833. [Google Scholar] [CrossRef]
- Jonsson, B.; Jonsson, N. Phenotypic plasticity and epigenetics of fish: Embryo temperature affects later-developing life-history traits. Aquat. Biol. 2019, 28, 21–32. [Google Scholar] [CrossRef]
- Jonsson, B.; Jonsson, N.; Finstad, A.G. Linking embryonic temperature with adult reproductive investment in Atlantic salmon Salmo salar. Mar. Ecol. Prog. Ser. 2014, 515, 217–226. [Google Scholar] [CrossRef]
- Adams, L.E.; Lund, J.R.; Moyle, P.B.; Quiñones, R.M.; Herman, J.D.; O’Rear, T.A. Environmental hedging: A theory and method for reconciling reservoir operations for downstream ecology and water supply. Water Resour. Res. 2017, 53, 7816–7831. [Google Scholar] [CrossRef]
- Hayes, D.S.; Moreira, M.; Boavida, I.; Haslauer, M.; Unfer, G.; Zeiringer, B.; Greimel, F.; Auer, S.; Ferreira, T.; Schmutz, S. Life stage-specific hydropeaking flow rules. Sustainability 2019, 11, 17. [Google Scholar] [CrossRef]
- Langshaw, R.B.; Graf, P.J.; Pearsons, T.N. Hydropower and high productivity in the Hanford Reach: A synthesis of how flow management may benefit fall Chinook Salmon in the Columbia River, USA. Wiley Interdiscip. Rev. -Water 2018, 5, e1275. [Google Scholar] [CrossRef]
- Sapin, J.R.; Saito, L.; Dai, A.; Rajagopalan, B.; Blair Hanna, R.; Kauneckis, D. Demonstration of integrated reservoir operations and extreme hydroclimate modeling of water temperatures for fish sustainability below Shasta Lake. J. Water Resour. Plan. Manag. 2017, 143, 04017062. [Google Scholar] [CrossRef]
- Sellheim, K.; Zeug, S.; Merz, J. Informed water management alternatives for an over-allocated river: Incorporating salmon life stage effects into a decision tree process during drought. Fish. Manag. Ecol. 2020, 27, 498–516. [Google Scholar] [CrossRef]
- Dudley, P.N. Insights from an individual based model of a fish population on a large regulated river. Environ. Biol. Fishes 2019, 102, 1069–1095. [Google Scholar] [CrossRef]
- Roni, P.; Anders, P.J.; Beechie, T.J.; Kaplowe, D.J. Review of tools for identifying, planning, and implementing habitat restoration for Pacific salmon and steelhead. N. Am. J. Fish. Manag. 2018, 38, 355–376. [Google Scholar] [CrossRef]
- Isaak, D.J.; Wenger, S.J.; Peterson, E.E.; Ver Hoef, J.M.; Nagel, D.E.; Luce, C.H.; Hostetler, S.W.; Dunham, J.B.; Roper, B.B.; Wollrab, S.P.; et al. The NorWeST summer stream temperature model and scenarios for the western U.S.: A crowd-sourced database and new geospatial tools foster a user community and predict broad climate warming of rivers and streams. Water Resour. Res. 2017, 53, 9181–9205. [Google Scholar] [CrossRef]
- Fullerton, A.H.; Torgersen, C.E.; Lawler, J.J.; Steel, E.A.; Ebersole, J.L.; Lee, S.Y. Longitudinal thermal heterogeneity in rivers and refugia for coldwater species: Effects of scale and climate change. Aquat. Sci. 2018, 80, 1–15. [Google Scholar] [CrossRef]
- Lee, S.Y.; Fullerton, A.H.; Sun, N.; Torgersen, C.E. Projecting spatiotemporally explicit effects of climate change on stream temperature: A model comparison and implications for coldwater fishes. J. Hydrol. 2020, 588, 125066. [Google Scholar] [CrossRef]
- McCullough, D.A.; Bartholow, J.M.; Jager, H.I.; Beschta, R.L.; Cheslak, E.F.; Deas, M.L.; Ebersole, J.L.; Foott, J.S.; Johnson, S.L.; Marine, K.R.; et al. Research in thermal biology: Burning questions for coldwater stream fishes. Rev. Fish. Sci. 2009, 17, 90–115. [Google Scholar] [CrossRef]
- Bennett, S.; Pess, G.; Bouwes, N.; Roni, P.; Bilby, R.E.; Gallagher, S.; Ruzycki, J.; Buehrens, T.; Krueger, K.; Ehinger, W.; et al. Progress and Challenges of Testing the Effectiveness of Stream Restoration in the Pacific Northwest Using Intensively Monitored Watersheds. Fisheries 2016, 41, 92–103. [Google Scholar] [CrossRef]
- Bilby, R.; Johnson, A.; Foltz, J.R.; Puls, A.L. Management Implications from Pacific Northwest Intensively Monitored Watersheds; U.S. Geological Survey: Reston, VA, USA, 2022; 99p, Available online: https://www.pnamp.org/document/15207 (accessed on 1 June 2022).
- Erhardt, J.M.; Tiffan, K.F.; Connor, W.P. Juvenile Chinook salmon mortality in a Snake River reservoir: Smallmouth bass pedation revisited. Trans. Am. Fish. Soc. 2018, 147, 316–328. [Google Scholar] [CrossRef]
- Hawkins, B.L.; Fullerton, A.H.; Sanderson, B.L.; Steel, E.A. Individual-based simulations suggest mixed impacts of warmer temperatures and a nonnative predator on Chinook salmon. Ecosphere 2020, 11, e03218. [Google Scholar] [CrossRef]
- Tonina, D.; McKean, J.A.; Isaak, D.; Benjankar, R.M.; Tang, C.; Chen, Q. Climate Change Shrinks and Fragments Salmon Habitats in a Snow-Dependent Region. Geophys. Res. Lett. 2022, 49, e2022GL098552. [Google Scholar] [CrossRef] [PubMed]
- Pennock, C.A.; Budy, P.; Atkinson, C.L.; Barrett, N. Effects of increased temperature on arctic slimy sculpin Cottus cognatus is mediated by food availability: Implications for climate change. Freshw. Biol. 2021, 66, 549–561. [Google Scholar] [CrossRef]
- Spanjer, A.R.; Gendaszek, A.S.; Wulfkuhle, E.J.; Black, R.W.; Jaeger, K.L. Assessing climate change impacts on Pacific salmon and trout using bioenergetics and spatiotemporal explicit river temperature predictions under varying riparian conditions. PLoS ONE 2022, 17, e0266871. [Google Scholar] [CrossRef]
- Benjamin, J.R.; Dunham, J.B.; Johnson, S.L.; Ashkenas, L.; Penaluna, B.E.; Bilby, R.E.; Bateman, D.; Leer, D.; Bellmore, J.R. Pathways of productivity and influences on top consumers in forested streams. For. Ecol. Manag. 2022, 508, 120046. [Google Scholar] [CrossRef]
- Platts, P.J.; Mason, S.C.; Palmer, G.; Hill, J.K.; Oliver, T.H.; Powney, G.D.; Fox, R.; Thomas, C.D. Habitat availability explains variation in climate-driven range shifts across multiple taxonomic groups. Sci. Rep. 2019, 9, 15039. [Google Scholar] [CrossRef]
- Fuchs, H.L.; Chant, R.J.; Hunter, E.J.; Curchitser, E.N.; Gerbi, G.P.; Chen, E.Y. Wrong-way migrations of benthic species driven by ocean warming and larval transport. Nat. Clim. Chang. 2020, 10, 1052–1056. [Google Scholar] [CrossRef]
- Beer, W.N.; Anderson, J.J. Sensitivity of juvenile salmonid growth to future climate trends. River Res. Appl. 2011, 27, 663–669. [Google Scholar] [CrossRef]
- Fullerton, A.H.; Burke, B.J.; Lawler, J.J.; Torgersen, C.E.; Ebersole, J.L.; Leibowitz, S.G. Simulated juvenile salmon growth and phenology respond to altered thermal regimes and stream network shape. Ecosphere 2017, 8, e02052-23. [Google Scholar] [CrossRef]
- Huntsman, B.M.; Falke, J.A. Main stem and off-channel habitat use by juvenile Chinook salmon in a sub-Arctic riverscape. Freshw. Biol. 2019, 64, 433–446. [Google Scholar] [CrossRef]
- Armstrong, J.B.; Fullerton, A.H.; Jordan, C.E.; Ebersole, J.L.; Bellmore, J.R.; Arismendi, I.; Penaluna, B.E.; Reeves, G.H. The importance of warm habitat to the growth regime of cold-water fishes. Nat. Clim. Chang. 2021, 11, 354–361. [Google Scholar] [CrossRef] [PubMed]
- Teichert, N.; Benitez, J.P.; Dierckx, A.; Tetard, S.; de Oliveira, E.; Trancart, T.; Feunteun, E.; Ovidio, M. Development of an accurate model to predict the phenology of Atlantic salmon smolt spring migration. Aquat. Conserv. -Mar. Freshw. Ecosyst. 2020, 30, 1552–1565. [Google Scholar] [CrossRef]
- Spence, B.C.; Dick, E.J. Geographic variation in environmental factors regulating outmigration timing of coho salmon (Oncorhynchus kisutch) smolts. Can. J. Fish. Aquat. Sci. 2014, 71, 56–69. [Google Scholar] [CrossRef]
- Carlson, S.M.; Seamons, T.R. A review of quantitative genetic components of fitness in salmonids: Implications for adaptation to future change. Evol. Appl. 2008, 1, 222–238. [Google Scholar] [CrossRef] [PubMed]
- Manhard, C.V.; Joyce, J.E.; Gharrett, A.J. Evolution of phenology in a salmonid population: A potential adaptive response to climate change. Can. J. Fish. Aquat. Sci. 2017, 74, 1519–1527. [Google Scholar] [CrossRef]
- Debes, P.V.; Piavchenko, N.; Erkinaro, J.; Primmer, C.R. Genetic growth potential, rather than phenotypic size, predicts migration phenotype in Atlantic salmon. Proc. R. Soc. B. Biol. Sci. 2020, 287, 20200867. [Google Scholar] [CrossRef]
- Debes, P.V.; Solberg, M.F.; Matre, I.H.; Dyrhovden, L.; Glover, K.A. Genetic variation for upper thermal tolerance diminishes within and between populations with increasing acclimation temperature in Atlantic salmon. Heredity 2021, 127, 455–466. [Google Scholar] [CrossRef]
- Muñoz, N.J.; Farrell, A.P.; Heath, J.W.; Neff, B.D. Adaptive potential of a Pacific salmon challenged by climate change. Nat. Clim. Chang. 2015, 5, 163–166. [Google Scholar] [CrossRef]
- Timpane-Padgham, B.L.; Beechie, T.; Klinger, T. A systematic review of ecological attributes that confer resilience to climate change in environmental restoration. PLoS ONE 2017, 12, e0173812. [Google Scholar] [CrossRef]
- Klein, S.; Herron, H.; Butcher, D. EPA Region 10 Climate Change and TMDL Pilot—South Fork Nooksack River, Washington; U.S. Environmental Protection Agency: Washington, DC, USA, 2017. [Google Scholar]
- Sundt-Hansen, L.E.; Hedger, R.D.; Ugedal, O.; Diserud, O.H.; Finstad, A.G.; Sauterleute, J.F.; Tofte, L.; Alfredsen, K.; Forseth, T. Modelling climate change effects on Atlantic salmon: Implications for mitigation in regulated rivers. Sci. Total Environ. 2018, 631–632, 1005–1017. [Google Scholar] [CrossRef]
- Hallnan, R.; Saito, L.; Busby, D.; Tyler, S. Modeling Shasta Reservoir Water-Temperature Response to the 2015 Drought and Response under Future Climate Change. J. Water Resour. Plan. Manag. 2020, 146, 04020018. [Google Scholar] [CrossRef]
- Hatten, J.R.; Batt, T.R.; Connolly, P.J.; Maule, A.G. Modeling effects of climate change on Yakima River salmonid habitats. Clim. Chang. 2014, 124, 427–439. [Google Scholar] [CrossRef]
- Jager, H.I.; King, A.W.; Gangrade, S.; Haines, A.; DeRolph, C.; Naz, B.S.; Ashfaq, M. Will future climate change increase the risk of violating minimum flow and maximum temperature thresholds below dams in the Pacific Northwest? Clim. Risk Manag. 2018, 21, 69–84. [Google Scholar] [CrossRef]
- Burke, B.J.; Peterson, W.T.; Beckman, B.R.; Morgan, C.; Daly, E.A.; Litz, M. Multivariate models of adult Pacific salmon returns. PLoS ONE 2013, 8, e54134. [Google Scholar] [CrossRef] [PubMed]
- Rechisky, E.L.; Welch, D.W.; Porter, A.D.; Hess, J.E.; Narum, S.R. Testing for delayed mortality effects in the early marine life history of Columbia River Basin yearling Chinook salmon. Mar. Ecol. Prog. Ser. 2014, 496, 159–180. [Google Scholar] [CrossRef]
- Litzow, M.A.; Ciannelli, L.; Puerta, P.; Wettstein, J.J.; Rykaczewski, R.R.; Opiekun, M. Non-stationary climate-salmon relationships in the Gulf of Alaska. Proc. R. Soc. B-Biol. Sci. 2018, 285, 20181855. [Google Scholar] [CrossRef] [PubMed]
- Litzow, M.A.; Hunsicker, M.E.; Bond, N.A.; Burke, B.J.; Cunningham, C.J.; Gosselin, J.L.; Norton, E.L.; Ward, E.J.; Zador, S.G. The changing physical and ecological meanings of North Pacific Ocean climate indices. Proc. Natl. Acad. Sci. USA 2020, 117, 7665–7671. [Google Scholar] [CrossRef]
- Litzow, M.A.; Malick, M.J.; Bond, N.A.; Cunningham, C.J.; Gosselin, J.L.; Ward, E.J. Quantifying a Novel Climate Through Changes in PDO-Climate and PDO-Salmon Relationships. Geophys. Res. Lett. 2020, 47, e2020GL087972. [Google Scholar] [CrossRef]
- Burke, B.J.; Anderson, J.J.; Miller, J.A.; Tomaro, L.; Teel, D.J.; Banas, N.S.; Baptista, A.M. Estimating behavior in a black box: How coastal oceanographic dynamics influence yearling Chinook salmon marine growth and migration behaviors. Environ. Biol. Fishes 2016, 99, 671–686. [Google Scholar] [CrossRef]
- Snyder, M.N.; Schumaker, N.H.; Ebersole, J.L.; Dunham, J.B.; Comeleo, R.L.; Keefer, M.L.; Leinenbach, P.; Brookes, A.; Cope, B.; Wu, J.; et al. Individual based modeling of fish migration in a 2-D river system: Model description and case study. Landsc. Ecol. 2019, 34, 737–754. [Google Scholar] [CrossRef]
- Seeb, L.W.; Seeb, J.E.; Habicht, C.; Farley, E.V.; Utter, F.M. Single-nucleotide polymorphic genotypes reveal patterns of early juvenile migration of sockeye salmon in the eastern Bering Sea. Trans. Am. Fish. Soc. 2011, 140, 734–748. [Google Scholar] [CrossRef]
- Kuparinen, A.; Hutchings, J.A. Genetic architecture of age at maturity can generate divergent and disruptive harvest-induced evolution. Philos. Trans. R. Soc. London. Ser. B Biol. Sci. 2017, 372, 20160035. [Google Scholar] [CrossRef]
- Frölicher, T.L.; Fischer, E.M.; Gruber, N. Marine heatwaves under global warming. Nature 2018, 560, 360–364. [Google Scholar] [CrossRef]
- Keefer, M.L.; Caudill, C.C.; Peery, C.A.; Boggs, C.T. Non-direct homing behaviours by adult Chinook salmon in a large, multi-stock river system. J. Fish Biol. 2008, 72, 27–44. [Google Scholar] [CrossRef]
- Crozier, L.G.; Hutchings, J.A. Plastic and evolutionary responses to climate change in fish. Evol. Appl. 2014, 7, 68–87. [Google Scholar] [CrossRef] [PubMed]
- Reed, T.E.; Schindler, D.E.; Hague, M.J.; Patterson, D.A.; Meir, E.; Waples, R.S.; Hinch, S.G. Time to evolve? Potential evolutionary responses of Fraser River sockeye salmon to climate change and effects on persistence. PLoS ONE 2011, 6, e20380. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, N.J.; Anttila, K.; Chen, Z.; Heath, J.W.; Farrell, A.P.; Neff, B.D. Indirect genetic effects underlie oxygen-limited thermal tolerance within a coastal population of chinook salmon. Proc. R. Soc. B Biol. Sci. 2014, 281, 20141082. [Google Scholar] [CrossRef] [PubMed]
- O’Malley, K.G.; Ford, M.J.; Hard, J.J. Clock polymorphism in Pacific salmon: Evidence for variable selection along a latitudinal gradient. Proc. R. Soc. B Biol. Sci. 2010, 277, 3703–3714. [Google Scholar] [CrossRef]
- Ford, M.D.; Nichols, K.M.; Waples, R.S.; Anderson, E.C.; Kardos, M.; Koch, I.; McKinney, G.; Miller, M.R.; Myers, J.; Naish, K.; et al. Reviewing and Synthesizing the State of the Science Regarding Associations between Adult Run Timing and Specific Genotypes in Chinook Salmon and Steelhead. In Proceedings of the Report of a Workshop; National Marine Fisheries Service, Northwest Fisheries Science Center: Seattle, WA 98112, USA, 27–28 February 2020. [Google Scholar] [CrossRef]
- Collins, E.E.; Hargrove, J.S.; Delomas, T.A.; Narum, S.R. Distribution of genetic variation underlying adult migration timing in steelhead of the Columbia River basin. Ecol. Evol. 2020, 10, 9486–9502. [Google Scholar] [CrossRef]
- Donaldson, M.R.; Hinch, S.G.; Patterson, D.A.; Farrell, A.P.; Shrimpton, J.M.; Miller-Saunders, K.M.; Robichaud, D.; Hills, J.; Hruska, K.A.; Hanson, K.C.; et al. Physiological condition differentially affects the behavior and survival of two populations of sockeye salmon during their freshwater spawning migration. Physiol. Biochem. Zool. 2010, 83, 446–458. [Google Scholar] [CrossRef]
- Eliason, E.J.; Clark, T.D.; Hague, M.J.; Hanson, L.M.; Gallagher, Z.S.; Jeffries, K.M.; Gale, M.K.; Patterson, D.A.; Hinch, S.G.; Farrell, A.P. Differences in thermal tolerance among sockeye salmon populations. Science 2011, 332, 109–112. [Google Scholar] [CrossRef]
- Eliason, E.J.; Clark, T.D.; Hinch, S.G.; Farrell, A.P. Cardiorespiratory collapse at high temperature in swimming adult sockeye salmon. Conserv. Physiol. 2013, 1, cot008. [Google Scholar] [CrossRef] [PubMed]
- Eliason, E.J.; Farrell, A.P. Oxygen uptake in Pacific salmon Oncorhynchus spp.: When ecology and physiology meet. J. Fish Biol. 2016, 88, 359–388. [Google Scholar] [CrossRef] [PubMed]
- Eliason, E.J.; Wilson, S.M.; Farrell, A.P.; Cooke, S.J.; Hinch, S.G. Low cardiac and aerobic scope in a coastal population of sockeye salmon Oncorhynchus nerka with a short upriver migration. J. Fish Biol. 2013, 82, 2104–2112. [Google Scholar] [CrossRef]
- Farrell, A.P. Environment, antecedents and climate change: Lessons from the study of temperature physiology and river migration of salmonids. J. Exp. Biol. 2009, 212, 3771–3780. [Google Scholar] [CrossRef] [PubMed]
- Farrell, A.P. Aerobic scope and its optimum temperature: Clarifying their usefulness and limitations—Correspondence on. J. Exp. Biol. 216, 2771–2782. J. Exp. Biol. 2013, 216, 4493–4494. [Google Scholar] [CrossRef] [PubMed]
- Farrell, A.P.; Hinch, S.G.; Cooke, S.J.; Patterson, D.A.; Crossin, G.T.; Lapointe, M.; Mathes, M.T. Pacific salmon in hot water: Applying aerobic scope models and biotelemetry to predict the success of spawning migrations. Physiol. Biochem. Zool. 2008, 81, 697–708. [Google Scholar] [CrossRef] [PubMed]
- Connor, W.P.; Tiffan, K.F.; Chandler, J.A.; Rondorf, D.W.; Arnsberg, B.D.; Anderson, K.C. Upstream Migration and Spawning Success of Chinook Salmon in a Highly Developed, Seasonally Warm River System. Rev. Fish. Sci. Aquac. 2019, 27, 1–50. [Google Scholar] [CrossRef]
- Lusardi, R.A.; Moyle, P.B. Two-way trap and haul as a conservation strategy for anadromous salmonids. Fisheries 2017, 42, 478–487. [Google Scholar] [CrossRef]
- Colvin, M.E.; Peterson, J.T.; Sharpe, C.; Kent, M.L.; Schreck, C.B. Identifying optimal hauling densities for adult Chinook salmon trap and haul operations. River Res. Appl. 2018, 34, 1158–1167. [Google Scholar] [CrossRef]
- O’Connor, C.M.; Norris, D.R.; Crossin, G.T.; Cooke, S.J. Biological carryover effects: Linking common concepts and mechanisms in ecology and evolution. Ecosphere 2014, 5, art28. [Google Scholar] [CrossRef]
- Moyle, P.B.; Lusardi, R.; Samuel, P.; Katz, J. State of the Salmonids: Status of California’s Emblematic Fishes 2017; Center for Watershed Sciences, University of California, Davis and California Trout: San Francisco, CA, USA, 2017; p. 579. Available online: http://caltrout.org/sos/ (accessed on 1 January 2019).
- Hare, J.A.; Morrison, W.E.; Nelson, M.W.; Stachura, M.M.; Teeters, E.J.; Griffis, R.B.; Alexander, M.A.; Scott, J.D.; Alade, L.; Bell, R.J.; et al. A vulnerability assessment of fish and invertebrates to climate change on the northeast U.S. continental shelf. PLoS ONE 2016, 11, e0146756. [Google Scholar] [CrossRef]
- McClure, M.M.; Haltuch, M.A.; Willis-Norton, E.; Huff, D.D.; Hazen, E.L.; Crozier, L.G.; Jacox, M.G.; Nelson, M.W.; Andrews, K.S.; Barnett, L.A.K.; et al. Vulnerability to climate change of managed stocks in the California Current large marine ecosystem. Front. Mar. Sci. 2023, 10, 3767. [Google Scholar] [CrossRef]
- Bjornas, K.L.; Railsback, S.F.; Calles, O.; Piccolo, J.J. Modeling Atlantic salmon (Salmo salar) and brown trout (S. trutta) population responses and interactions under increased minimum flow in a regulated river. Ecol. Eng. 2021, 162, 106182. [Google Scholar] [CrossRef]
- Huang, B.; Langpap, C.; Adams, R.M. Using instream water temperature forecasts for fisheries management: An application in the Pacific Northwest. J. Am. Water Resour. Assoc. 2011, 47, 861–876. [Google Scholar] [CrossRef]
- Zeug, S.C.; Bergman, P.S.; Cavallo, B.J.; Jones, K.S. Application of a life cycle simulation model to evaluate impacts of water management and conservation actions on an endangered population of Chinook salmon. Environ. Model. Assess. 2012, 17, 455–467. [Google Scholar] [CrossRef]
- Falke, J.A.; Flitcroft, R.L.; Dunham, J.B.; McNyset, K.M.; Hessburg, P.F.; Reeves, G.H. Climate change and vulnerability of bull trout (Salvelinus confluentus) in a fire-prone landscape. Can. J. Fish. Aquat. Sci. 2015, 72, 304–318. [Google Scholar] [CrossRef]
- Almodovar, A.; Leal, S.; Nicola, G.G.; Horreo, J.L.; Garcia-Vazquez, E.; Elvira, B. Long-term stocking practices threaten the original genetic diversity of the southernmost European populations of Atlantic salmon Salmo salar. Endanger. Species Res. 2020, 41, 303–317. [Google Scholar] [CrossRef]
- Thorstad, E.B.; Bliss, D.; Breau, C.; Damon-Randall, K.; Sundt-Hansen, L.E.; Hatfield, E.M.C.; Horsburgh, G.; Hansen, H.; Maoileidigh, N.O.; Sheehan, T.; et al. Atlantic salmon in a rapidly changing environment-Facing the challenges of reduced marine survival and climate change. Aquat. Conserv-Mar. Freshw. Ecosyst. 2021, 31, 2654–2665. [Google Scholar] [CrossRef]
- Ariza, A.; Lengaigne, M.; Menkes, C.; Lebourges-Dhaussy, A.; Receveur, A.; Gorgues, T.; Habasque, J.; Gutiérrez, M.; Maury, O.; Bertrand, A. Global decline of pelagic fauna in a warmer ocean. Nat. Clim. Chang. 2022, 12, 928–934. [Google Scholar] [CrossRef]
- Hause, C.L.; Singer, G.P.; Buchanan, R.A.; Cocherell, D.E.; Fangue, N.A.; Rypel, A.L. Survival of a threatened salmon is linked to spatial variability in river conditions. Can. J. Fish. Aquat. Sci. 2022, 79, 2056–2071. [Google Scholar] [CrossRef]
- Halpern, B.S.; Kappel, C.V.; Selkoe, K.A.; Micheli, F.; Ebert, C.M.; Kontgis, C.; Crain, C.M.; Martone, R.G.; Shearer, C.; Teck, S.J. Mapping cumulative human impacts to California Current marine ecosystems. Conserv. Lett. 2009, 2, 138–148. [Google Scholar] [CrossRef]
- Andrews, K.S.; Williams, G.D.; Samhouri, J.F.; Marshall, K.N.; Gertseva, V.; Levin, P.S. The legacy of a crowded ocean: Indicators, status, and trends of anthropogenic pressures in the California Current ecosystem. Environ. Conserv. 2015, 42, 139–151. [Google Scholar] [CrossRef]
- Toft, J.D.; Munsch, S.H.; Cordell, J.R.; Siitari, K.; Hare, V.C.; Holycross, B.M.; DeBruyckere, L.A.; Greene, C.M.; Hughes, B.B. Impact of multiple stressors on juvenile fish in estuaries of the northeast Pacific. Glob. Chang. Biol. 2018, 24, 2008–2020. [Google Scholar] [CrossRef] [PubMed]
- Greene, C.M.; Blackhart, K.; Nohner, J.; Candelmo, A.; Nelson, D.M. A national assessment of stressors to estuarine fish habitats in the contiguous USA. Estuaries Coasts 2015, 38, 782–799. [Google Scholar] [CrossRef]
- Teck, S.J.; Halpern, B.S.; Kappel, C.V.; Micheli, F.; Selkoe, K.A.; Crain, C.M.; Martone, R.; Shearer, C.; Arvai, J.; Fischhoff, B.; et al. Using expert judgment to estimate marine ecosystem vulnerability in the California Current. Ecol. Appl. 2010, 20, 1402–1416. [Google Scholar] [CrossRef] [PubMed]
- Beechie, T.J.; Fogel, C.; Nicol, C.; Jorgensen, J.; Timpane-Padgham, B.; Kiffney, P. How does habitat restoration influence resilience of salmon populations to climate change? Ecosphere 2022, 14, e4402. [Google Scholar] [CrossRef]
- Justice, C.; White, S.M.; McCullough, D.A.; Graves, D.S.; Blanchard, M.R. Can stream and riparian restoration offset climate change impacts to salmon populations? J. Environ. Manag. 2017, 188, 212–227. [Google Scholar] [CrossRef] [PubMed]
- White, S.M.; Brandy, S.; Justice, C.; Morinaga, K.A.; Naylor, L.; Ruzycki, J.; Sedell, E.R.; Steele, J.; Towne, A.; Webster, J.G.; et al. Progress towards a comprehensive approach for habitat restoration in the Columbia Basin: Case study in the Grande Ronde River. Fisheries 2021, 46, 229–243. [Google Scholar] [CrossRef]
- Jordan, C.E.; Fairfax, E. Beaver: The North American freshwater climate action plan. WIREs Water 2022, 9, e1592. [Google Scholar] [CrossRef]
- Brennan, S.R.; Schindler, D.E.; Cline, T.J.; Walsworth, T.E.; Buck, G.; Fernandez, D.P. Shifting habitat mosaics and fish production across river basins. Science 2019, 364, 783–786. [Google Scholar] [CrossRef]
- Waples, R.S.; Zabel, R.W.; Scheuerell, M.D.; Sanderson, B.L. Evolutionary responses by native species to major anthropogenic changes to their ecosystems: Pacific salmon in the Columbia River hydropower system. Mol. Ecol. 2008, 17, 84–96. [Google Scholar] [CrossRef]
- Kovach, R.P.; Muhlfeld, C.C.; Wade, A.A.; Hand, B.K.; Whited, D.C.; DeHaan, P.W.; Al-Chokhachy, R.; Luikart, G. Genetic diversity is related to climatic variation and vulnerability in threatened bull trout. Glob. Chang. Biol. 2015, 21, 2510–2524. [Google Scholar] [CrossRef]
- Beechie, T.J.; Nicol, C.; Fogel, C.; Jorgensen, J.; Thompson, J.; Seixas, G.; Chamberlin, J.; Hall, J.; Timpane-Padgham, B.; Kiffney, P.; et al. Chehalis LIfe-Cycle Modeling Phase 1 Report. In NOAA Contract Report NMFS-NWFSC; CR-2021-01; United States National Marine Fisheries Service: Sacramento, CA, USA; Northwest Fisheries Science Center (U.S.): Seattle, WA, USA; NMFS (National Marine Fisheries Service): Silver Spring, MD, USA; NWFSC (Northwest Fisheries Science Center): Niceville, FL, USA, 2021. [Google Scholar]
- Bond, M.H.; Nodine, T.G.; Beechie, T.J.; Zabel, R.W. Estimating the benefits of widespread floodplain reconnection for Columbia River Chinook salmon. Can. J. Fish. Aquat. Sci. 2019, 76, 1212–1226. [Google Scholar] [CrossRef]
- FitzGerald, A.M.; Martin, B.T. Quantification of thermal impacts across freshwater life stages to improve temperature management for anadromous salmonids. Conserv. Physiol. 2022, 10, coac013. [Google Scholar] [CrossRef] [PubMed]
- Yakima River Basin Water Enhancement Project Workgroup. Yakima Basin Integrated Plan. Available online: https://yakimabasinintegratedplan.org/the-integrated-plan/ (accessed on 1 November 2022).
- McCarthy, M.D.; Rinella, D.J.; Finney, B.P. Sockeye Salmon Population Dynamics over the Past 4000 Years in Upper Russian Lake, South-Central Alaska. Journal of Paleolimnology 2018, 60, 67–75. [Google Scholar] [CrossRef]
- Chasco, B.; Burke, B.; Crozier, L.; Zabel, R. Differential impacts of freshwater and marine covariates on wild and hatchery Chinook salmon marine survival. PLoS ONE 2021, 16, e0246659. [Google Scholar] [CrossRef]
- Wells, B.K.; Huff, D.D.; Burke, B.J.; Brodeur, R.D.; Santora, J.A.; Field, J.C.; Richerson, K.; Mantua, N.J.; Fresh, K.L.; McClure, M.M.; et al. Implementing Ecosystem-Based Management Principles in the Design of a Salmon Ocean Ecology Program. Front. Mar. Sci. 2020, 7, 342. [Google Scholar] [CrossRef]
- Tillotson, M.D.; Kelly, R.P.; Duda, J.J.; Hoy, M.; Kralj, J.; Quinn, T.P. Concentrations of environmental DNA (eDNA) reflect spawning salmon abundance at fine spatial and temporal scales. Biol. Conserv. 2018, 220, 1–11. [Google Scholar] [CrossRef]
- Rubenson, E.S.; Lawrence, D.J.; Olden, J.D. Threats to rearing juvenile Chinook salmon from non-native smallmouth bass inferred from stable isotope and fatty acid biomarkers. Trans. Am. Fish. Soc. 2020, 149, 350–363. [Google Scholar] [CrossRef]
- Franklin, T.W.; Dysthe, J.C.; Rubenson, E.S.; Carim, K.J.; Olden, J.D.; McKelvey, K.S.; Young, M.K.; Schwartz, M.K. A non-invasive sampling method for detecting non-native smallmouth bass (Micropterus dolomieu). Northwest Sci. 2018, 92, 149–157. [Google Scholar] [CrossRef]
- Sepulveda, A.J.; Schmidt, C.; Amberg, J.; Hutchins, P.; Stratton, C.; Mebane, C.; Laramie, M.B.; Pilliod, D.S. Adding invasive species biosurveillance to the US Geological Survey streamgage network. Ecosphere 2019, 10, e02843. [Google Scholar] [CrossRef]
- Akbarzadeh, A.; Gunther, O.P.; Houde, A.L.; Li, S.R.; Ming, T.J.; Jeffries, K.M.; Hinch, S.G.; Miller, K.M. Developing specific molecular biomarkers for thermal stress in salmonids. BMC Genom. 2018, 19, 1–28. [Google Scholar] [CrossRef]
- Consuegra, S.; O’Rorke, R.; Rodriguez-Barreto, D.; Fernandez, S.; Jones, J.; de Leaniz, C.G. Impacts of large and small barriers on fish assemblage composition assessed using environmental DNA metabarcoding. Sci. Total Environ. 2021, 790, 148054. [Google Scholar] [CrossRef]
- Gold, Z.; Curd, E.E.; Goodwin, K.D.; Choi, E.S.; Frable, B.W.; Thompson, A.R.; Walker, H.J.; Burton, R.S.; Kacev, D.; Martz, L.D.; et al. Improving metabarcoding taxonomic assignment: A case study of fishes in a large marine ecosystem. Mol. Ecol. Resour. 2021, 21, 2546–2564. [Google Scholar] [CrossRef]
- Jeffery, N.W.; Lehnert, S.J.; Kess, T.; Layton, K.K.S.; Wringe, B.F.; Stanley, R.R.E. Application of omics tools in designing and monitoring marine protected areas for a sustainable blue economy. Front. Genet. 2022, 13, 886494. [Google Scholar] [CrossRef]
- Dauwalter, D.C.; Fesenmyer, K.A.; Bjork, R.; Leasure, D.R.; Wenger, S.J. Satellite and Airborne Remote Sensing Applications for Freshwater Fisheries. Fisheries 2017, 42, 526–537. [Google Scholar] [CrossRef]
- Naman, S.; White, S.; Bellmore, J.; McHugh, P.; Kaylor, M.; Baxter, C.; Danehy, R.; Naiman, R.; Puls, A. Food web perspectives and methods for riverine fish conservation. Wiley Interdiscip. Rev. Water 2022, 9, e1590. [Google Scholar] [CrossRef]
- Koenigstein, S.; Dahlke, F.T.; Stiasny, M.H.; Storch, D.; Clemmesen, C.; Portner, H.O. Forecasting future recruitment success for Atlantic cod in the warming and acidifying Barents Sea. Glob. Chang. Biol. 2018, 24, 526–535. [Google Scholar] [CrossRef]
- Dietze, M.C.; Fox, A.; Beck-Johnson, L.M.; Betancourt, J.L.; Hooten, M.B.; Jarnevich, C.S.; Keitt, T.H.; Kenney, M.A.; Laney, C.M.; Larsen, L.G.; et al. Iterative near-term ecological forecasting: Needs, opportunities, and challenges. Proc. Natl. Acad. Sci. USA 2018, 115, 1424–1432. [Google Scholar] [CrossRef]
- Sobocinski, K.L.; Greene, C.M.; Schmidt, M.W. Using a qualitative model to explore the impacts of ecosystem and anthropogenic drivers upon declining marine survival in Pacific salmon. Environ. Conserv. 2018, 45, 278–290. [Google Scholar] [CrossRef]
- Forget, N.L.; Duplisea, D.E.; Sardenne, F.; McKindsey, C.W. Using qualitative network models to assess the influence of mussel culture on ecosystem dynamics. Ecol. Model. 2020, 430, 109070. [Google Scholar] [CrossRef]
- Reum, J.C.P.; McDonald, P.S.; Long, W.C.; Holsman, K.K.; Divine, L.; Armstrong, D.; Armstrong, J. Rapid assessment of management options for promoting stock rebuilding in data-poor species under climate change. Conserv. Biol. 2020, 34, 611–621. [Google Scholar] [CrossRef]
- Tommasi, D.; Stock, C.A.; Hobday, A.J.; Methot, R.; Kaplan, I.C.; Eveson, J.P.; Holsman, K.; Miller, T.J.; Gaichas, S.; Gehlen, M.; et al. Managing living marine resources in a dynamic environment: The role of seasonal to decadal climate forecasts. Prog. Oceanogr. 2017, 152, 15–49. [Google Scholar] [CrossRef]
- Koenigstein, S.; Mark, F.C.; Gossling-Reisemann, S.; Reuter, H.; Poertner, H.O. Modelling climate change impacts on marine fish populations: Process-based integration of ocean warming, acidification and other environmental drivers. Fish Fish. 2016, 17, 972–1004. [Google Scholar] [CrossRef]
- Jacox, M.G.; Hazen, E.L.; Zaba, K.D.; Rudnick, D.L.; Edwards, C.A.; Moore, A.M.; Bograd, S.J. Impacts of the 2015-2016 El Nino on the California Current System: Early assessment and comparison to past events. Geophys. Res. Lett. 2016, 43, 7072–7080. [Google Scholar] [CrossRef]
- Thompson, A.R.; Bjorkstedt, E.P.; Bograd, S.J.; Fisher, J.L.; Hazen, E.L.; Leising, A.; Santora, J.A.; Satterthwaite, E.V.; Sydeman, W.J.; Alksne, M.; et al. State of the California Current Ecosystem in 2021: Winter is coming? Front. Mar. Sci. 2022, 9, 958727. [Google Scholar] [CrossRef]
- Shi, H.; García-Reyes, M.; Jacox, M.G.; Rykaczewski, R.R.; Black, B.A.; Bograd, S.J.; Sydeman, W.J. Co-occurrence of California Drought and Northeast Pacific Marine Heatwaves Under Climate Change. Geophys. Res. Lett. 2021, 48, e2021GL092765. [Google Scholar] [CrossRef]
- Smith, J.A.; Pozo Buil, M.; Fiechter, J.; Tommasi, D.; Jacox, M.G. Projected novelty in the climate envelope of the California Current at multiple spatial-temporal scales. PLoS Clim. 2022, 1, e0000022. [Google Scholar] [CrossRef]
- Brodie, S.; Abrahms, B.; Bograd, S.J.; Carroll, G.; Hazen, E.L.; Muhling, B.A.; Buil, M.P.; Smith, J.A.; Welch, H.; Jacox, M.G. Exploring timescales of predictability in species distributions. Ecography 2021, 44, 832–844. [Google Scholar] [CrossRef]
- Cheung, W.W.L.; Ota, Y.; Cisneros-Montemayor, A. Predicting Future Oceans: Sustainability of Ocean and Human Systems Amidst Global Environmental Change; Elsevier: San Diego, CA, USA, 2019; ISBN 978-0-12-817946-8. [Google Scholar]
- Holsman, K.K.; Aydin, K. Comparative methods for evaluating climate change impacts on the foraging ecology of Alaskan groundfish. Mar. Ecol. Prog. Ser. 2015, 521, 217–235. [Google Scholar] [CrossRef]
- Kaplan, I.C.; Francis, T.B.; Punt, A.E.; Koehn, L.E.; Curchitser, E.; Hurtado-Ferro, F.; Johnson, K.F.; Lluch-Cota, S.E.; Sydeman, W.J.; Essington, T.E.; et al. A multi-model approach to understanding the role of Pacific sardine in the California Current food web. Mar. Ecol. Prog. Ser. 2019, 617–618, 307–321. [Google Scholar] [CrossRef]
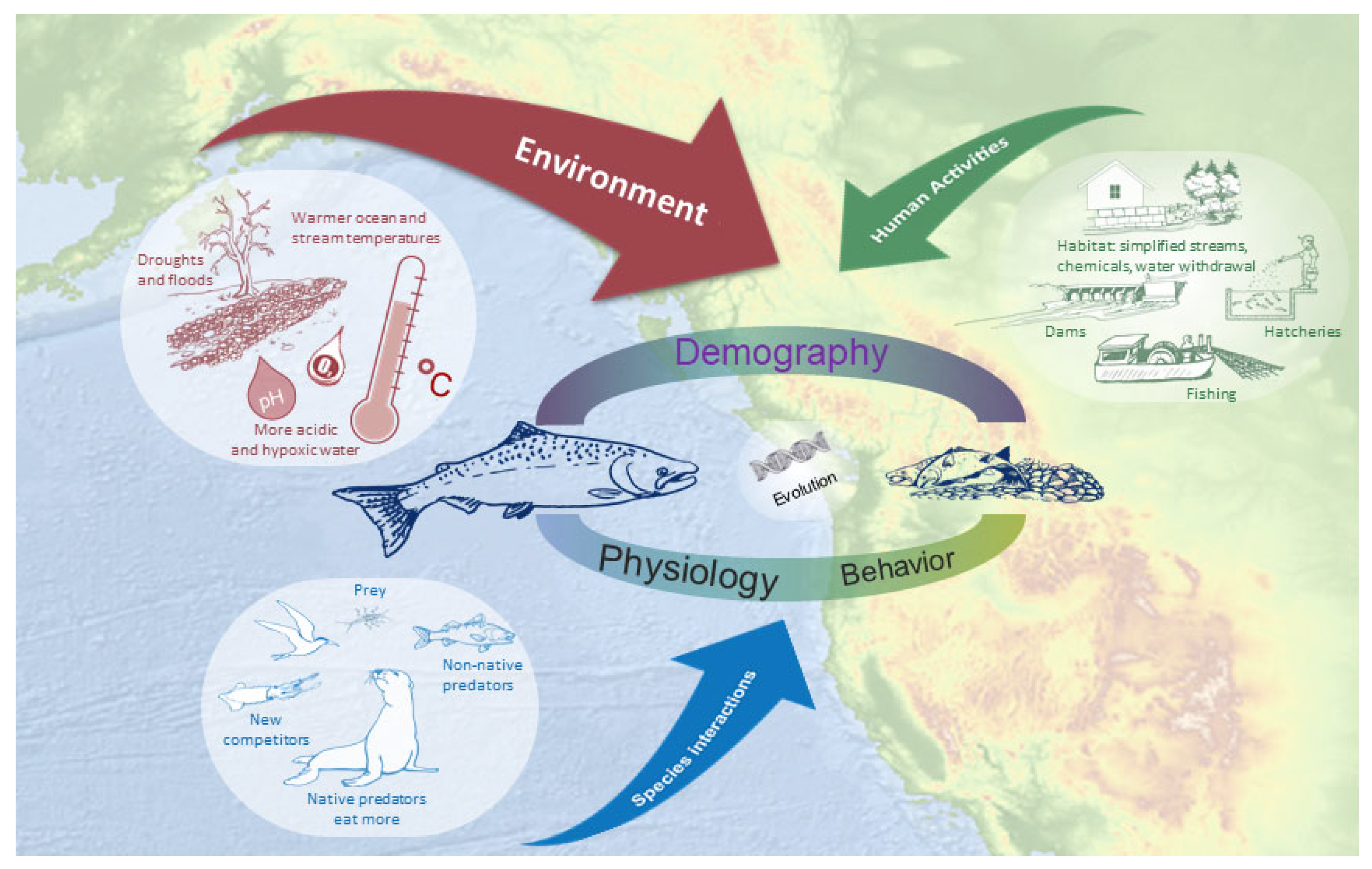
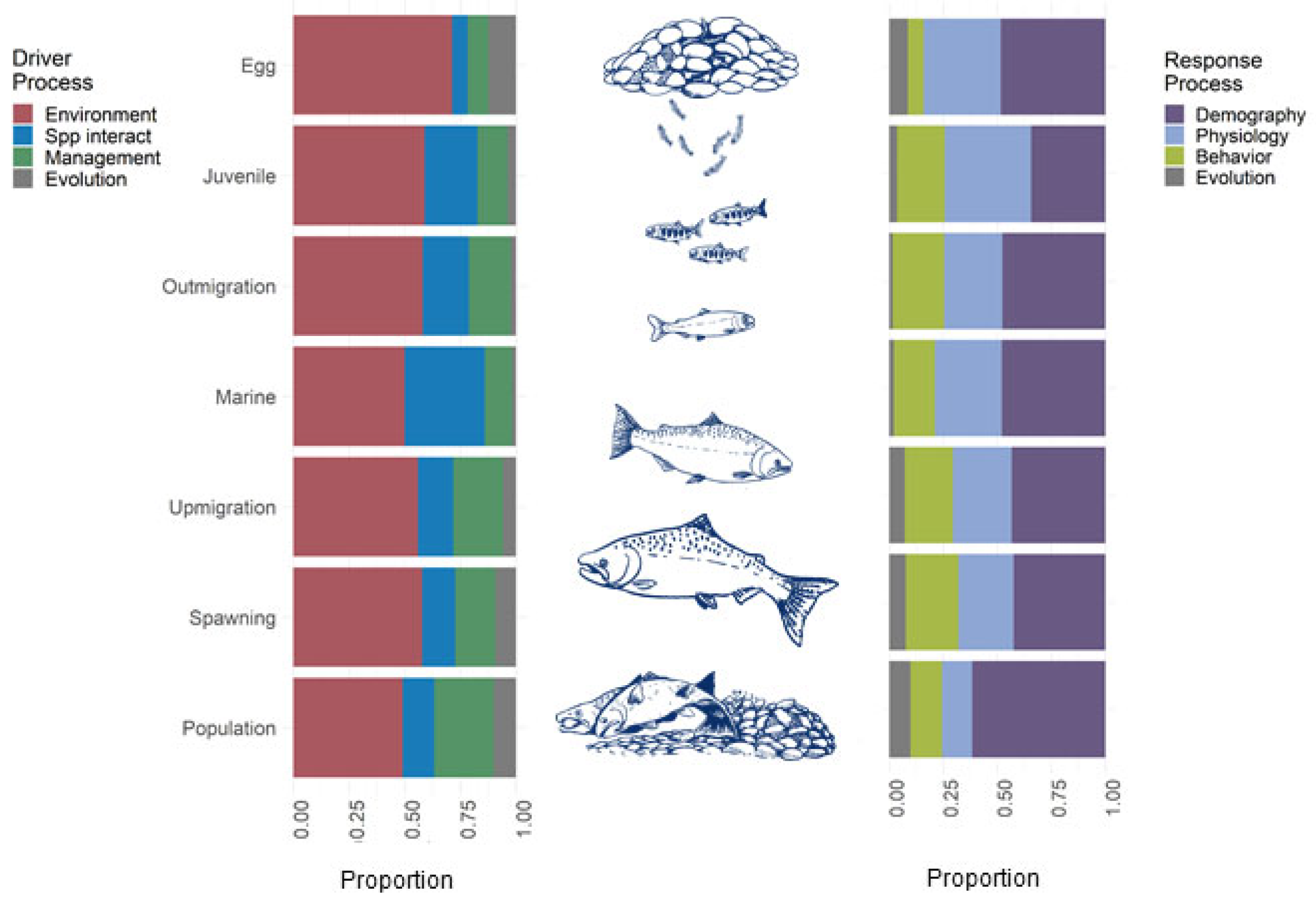

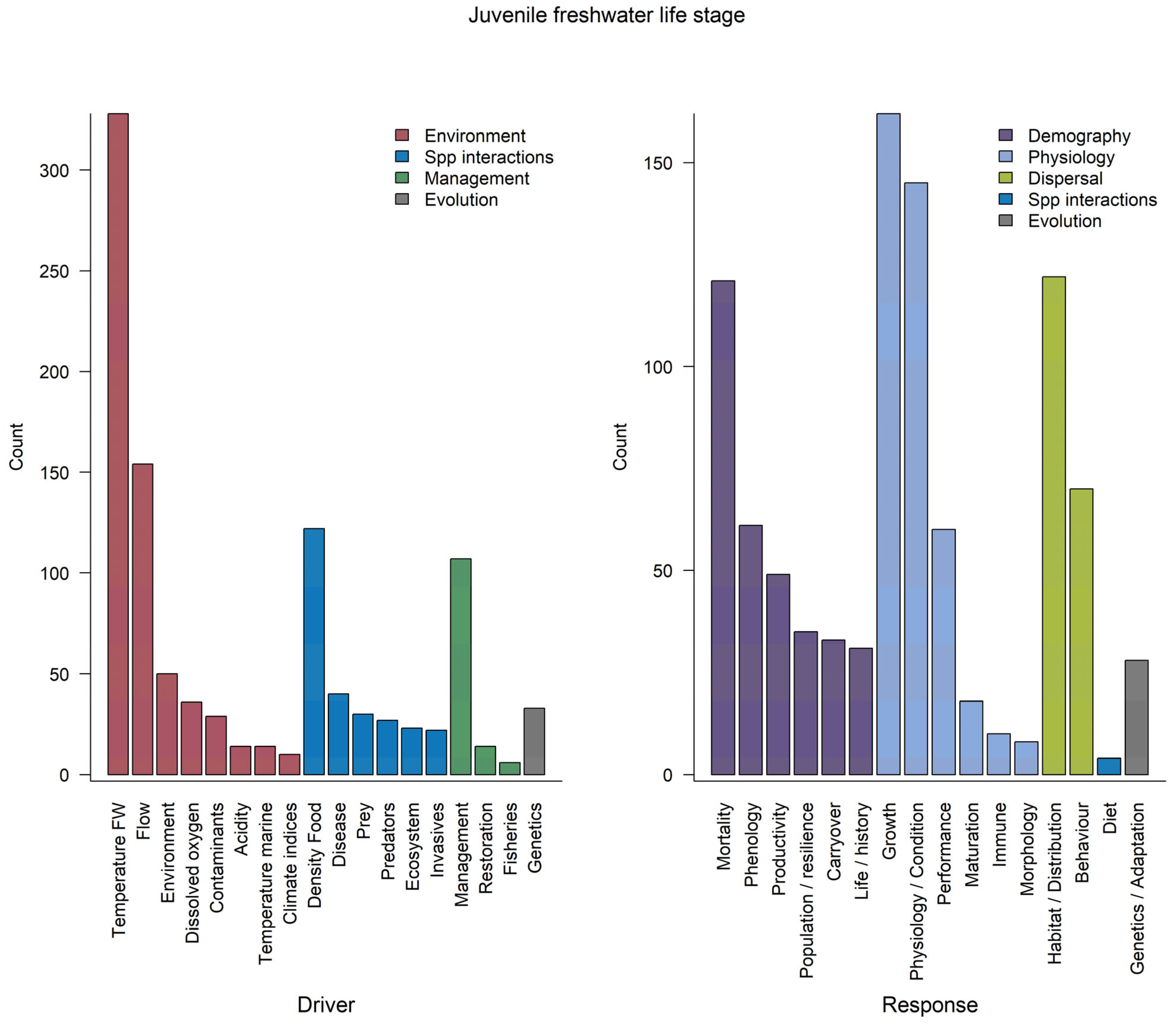
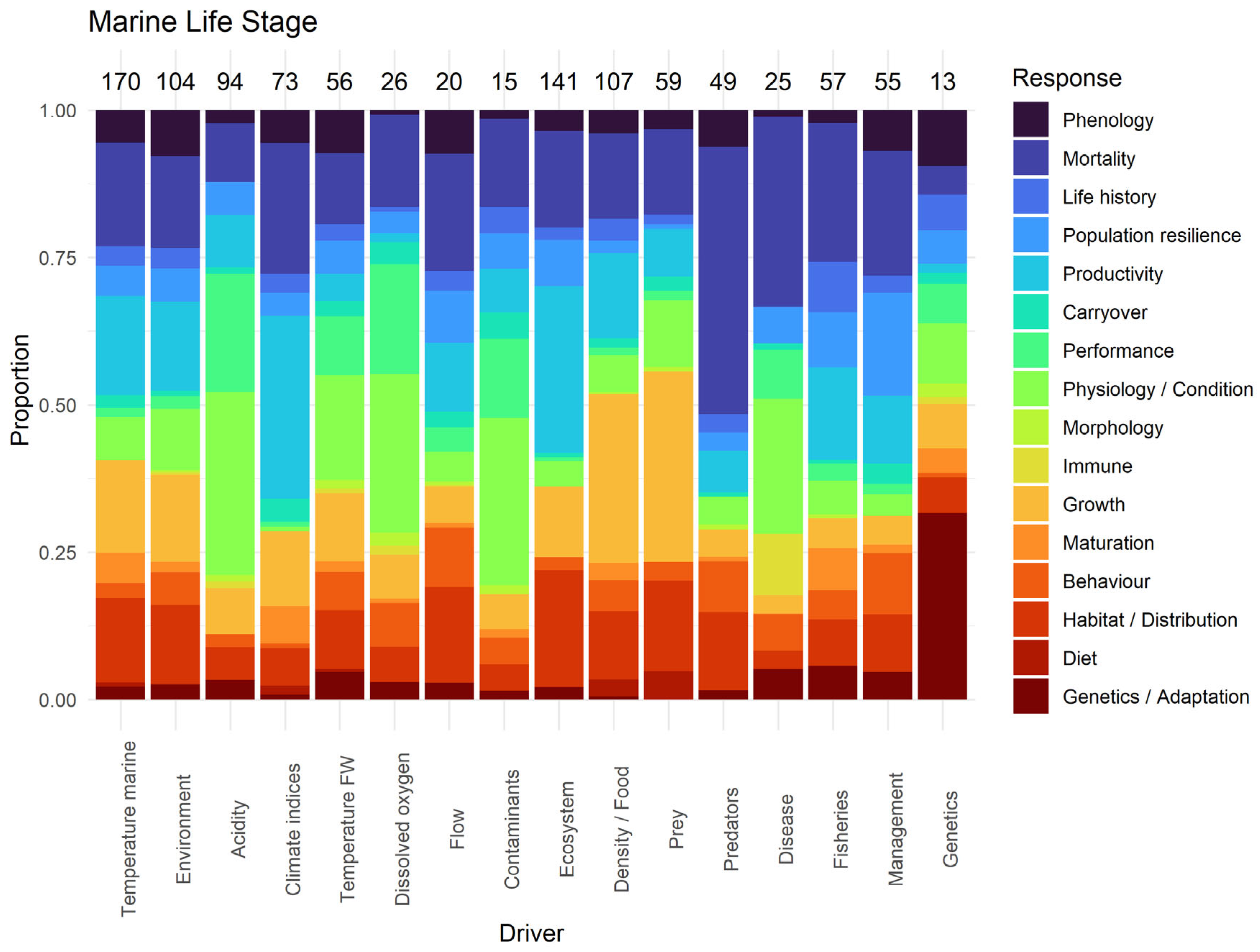
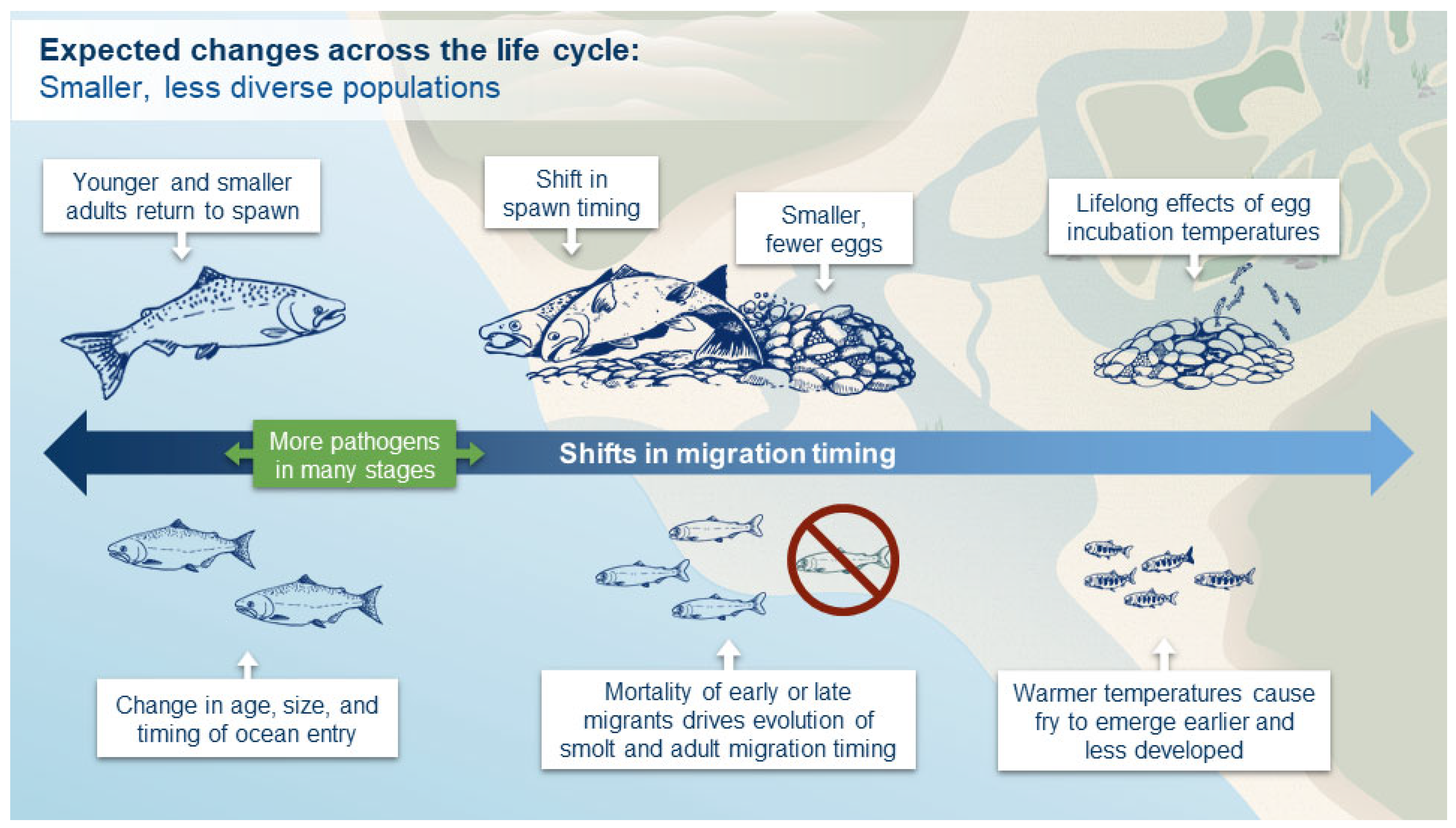
| Process | Definition | |
|---|---|---|
| Driver | ||
| Environment | Acidity | Ocean and freshwater acidification |
| Climate indices | Large-scale climate indices (PDO, ENSO) | |
| Contaminants | Contaminants such as heavy metals and PCBs | |
| Dissolved oxygen | Dissolved oxygen concentrations, hypoxic waters | |
| Environment | Other environmental drivers (e.g., salinity, upwelling) | |
| Flow | Freshwater flow levels | |
| Habitat | Physical habitat characteristics | |
| Marine temp | Marine water temperatures | |
| Freshwater temp | Freshwater water temperatures | |
| Species interactions | Density food | Density, competition, and food availability |
| Disease | Impacts of disease | |
| Ecosystem | Relevant ecosystem interactions not focused on salmon | |
| Invasives | Invasive species competing or preying on salmon | |
| Predators | Salmon predators | |
| Prey | Salmon prey | |
| Evolution | Genetics | Genetically derived traits or genetic diversity |
| Management | Fisheries | Impacts from fisheries |
| Management | Management levers (e.g., flow, hatchery, policy, framework) | |
| Restoration | Habitat restoration as a driver | |
| Methods | Methodologies for science or management | |
| Response | ||
| Physiology | Growth | Salmon growth |
| Immune | Immune system responses | |
| Maturation | Sexual maturation | |
| Morphology | Physical morphology | |
| Performance | Fish performance metrics (e.g., swim speed) | |
| Physiology/condition | Internal physiological responses/fish condition metrics | |
| Demography | Carryover | Carryover impacts from one life stage to another |
| Life history | Life history changes, often demographic | |
| Mortality | Mortality rates | |
| Phenology | Timing of life history events (e.g., spawning, migration) | |
| Population resiliency | Resilience of entire populations | |
| Productivity | Population productivity | |
| Dispersal | Behavior | Changes in behavior |
| Habitat distribution | Distribution within available habitat | |
| Species interactions | Diet | Diet composition as a response |
| Evolution | Genetic adaptation | Change in genotype or adaptive response (phenotype) |
| Management | Livelihood | Human economic or subsistence |
| Strength | Weakness | Expected Change | Management Options |
|---|---|---|---|
| Population level | |||
| Population-scale demographic processes; carryover effects in life history | Missing processes, esp. evolution and species interactions; carryover effects (other than life history); disease | Smaller, younger, fewer spawners; higher pathogen loads | Fisheries, flow management, dams, habitat restoration, forest management, stocking, invasive species management |
| Egg stage | |||
| Mechanistic understanding, evolutionary processes | Carryover effects in projection models; micro-climatic habitat projections | Egg size, emergence timing, alevin condition, epigenetic effects | Flow management, contaminants, adult condition, habitat condition |
| Juvenile stage | |||
| Widest understanding, adaptive management and hypothesis testing | Species interactions; carryover effects from eggs; data-poor watersheds (validate remote data) | Life history strategy (timing and growth rate, size and age at migration, migration timing) | Habitat; contaminants; disease; invasive species; dams; juvenile transportation; management of forests, fires, and flows |
| Marine stage | |||
| Many populations; broad spatial and temporal scales of analysis | Experiments and direct observations; behavior; species interactions; projections grounded in mechanistic understanding | Age and size at maturation; health of adult migrants | Fisheries targeting salmon, forage fish, groundfish, and predators; estuary and bottom habitat; contaminants and pathogens; hatcheries and aquaculture |
| Adult stage | |||
| Individual behavior, physiology | Less- studied systems outside main rivers; carryover effects from the ocean | Timing, condition, disease tolerance, prespawn mortality, spawn behavior | Fisheries; migration barriers; dam operations; influences on temperature; flow management; fish transportation; marine mammal predators; hatcheries; contaminant and disease reduction |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Crozier, L.G.; Siegel, J.E. A Comprehensive Review of the Impacts of Climate Change on Salmon: Strengths and Weaknesses of the Literature by Life Stage. Fishes 2023, 8, 319. https://doi.org/10.3390/fishes8060319
Crozier LG, Siegel JE. A Comprehensive Review of the Impacts of Climate Change on Salmon: Strengths and Weaknesses of the Literature by Life Stage. Fishes. 2023; 8(6):319. https://doi.org/10.3390/fishes8060319
Chicago/Turabian StyleCrozier, Lisa G., and Jared E. Siegel. 2023. "A Comprehensive Review of the Impacts of Climate Change on Salmon: Strengths and Weaknesses of the Literature by Life Stage" Fishes 8, no. 6: 319. https://doi.org/10.3390/fishes8060319
APA StyleCrozier, L. G., & Siegel, J. E. (2023). A Comprehensive Review of the Impacts of Climate Change on Salmon: Strengths and Weaknesses of the Literature by Life Stage. Fishes, 8(6), 319. https://doi.org/10.3390/fishes8060319






