Morphological Distribution Patterns and Neuroimmune Communication of Ganglia in Molly Fish (Poecilia sphenops, Valenciennes 1846)
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. Histological and Histochemical Analysis
2.3. Semithin Sections and TEM
2.4. Immunohistochemical Analysis
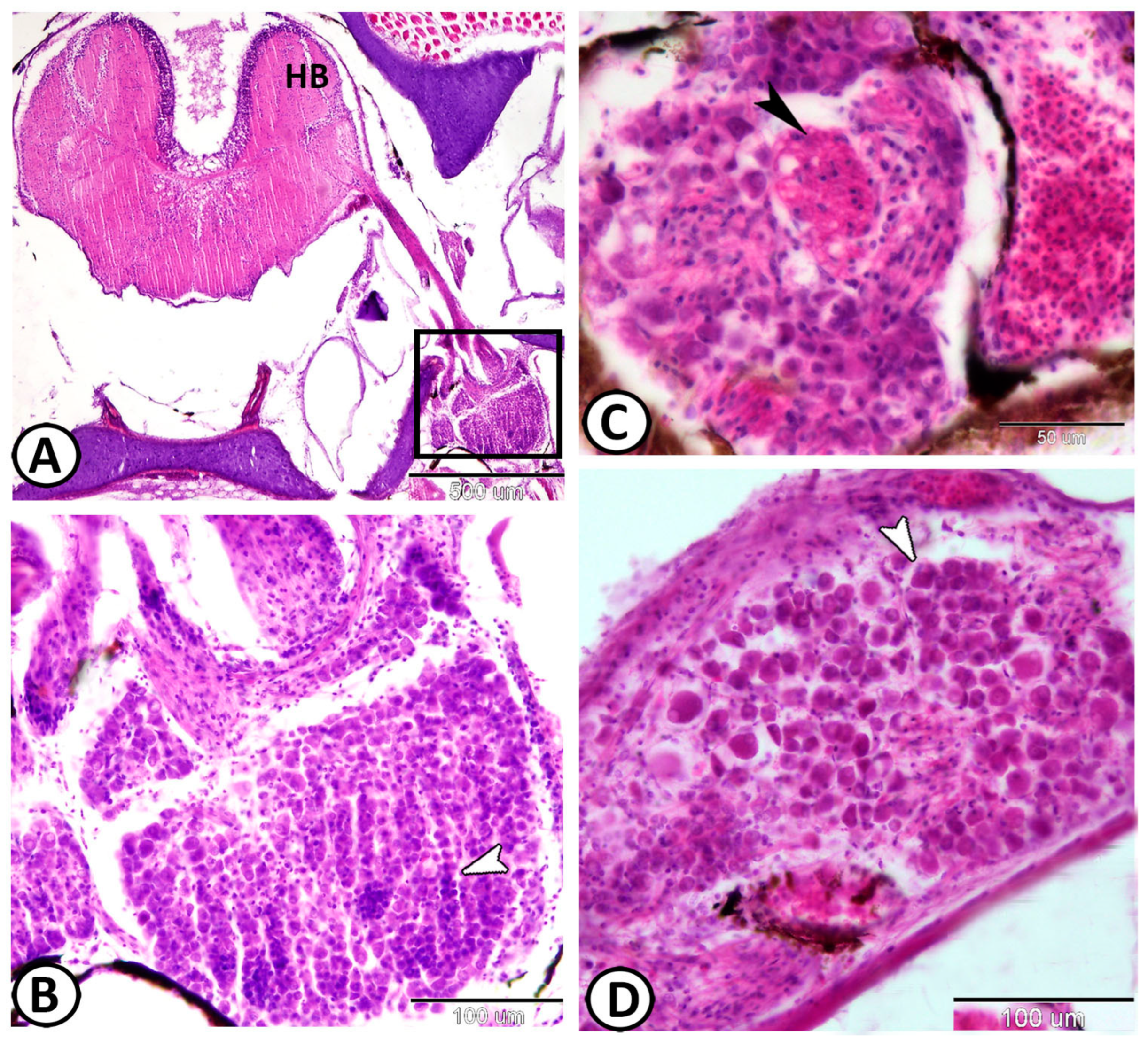
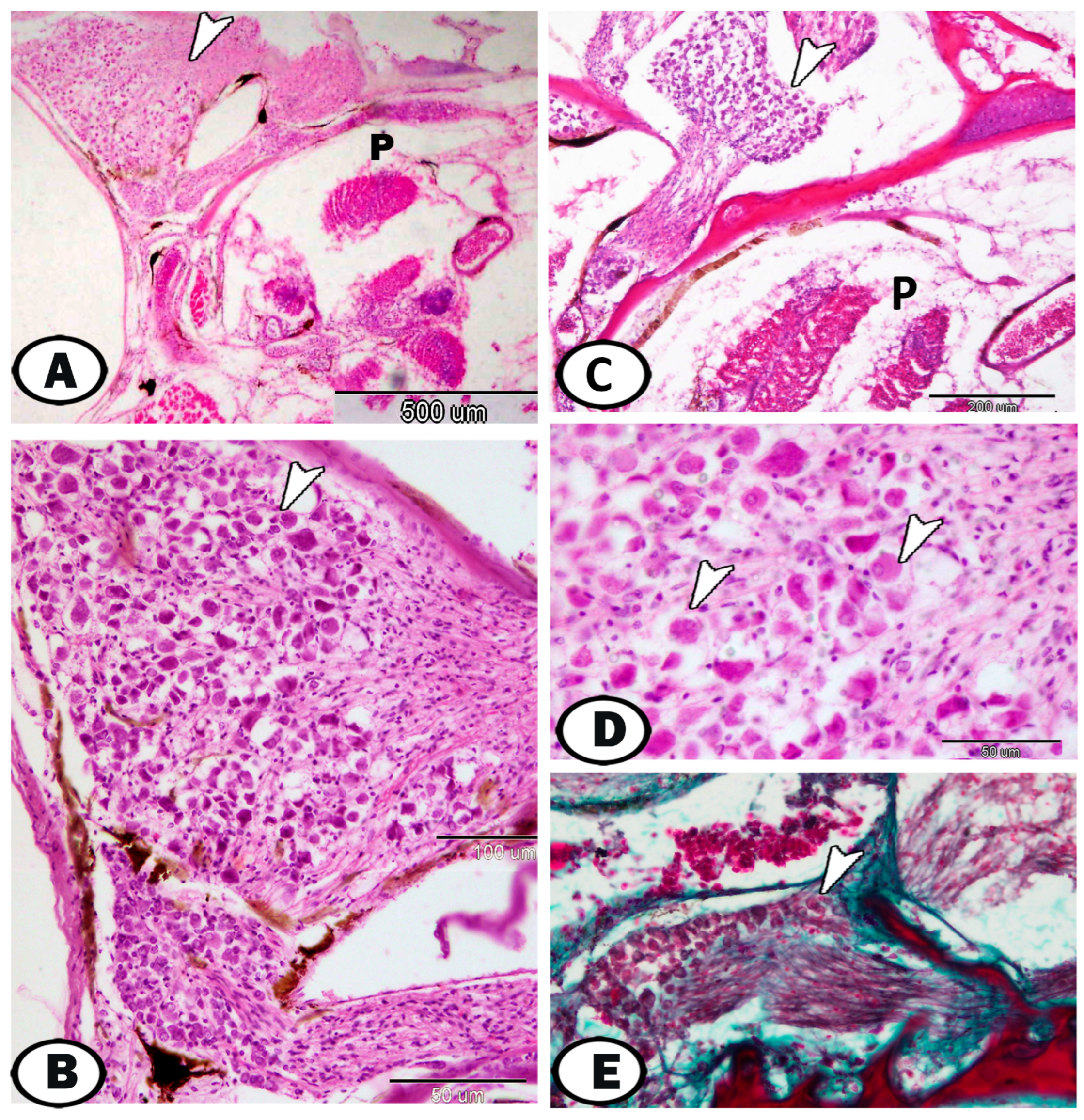
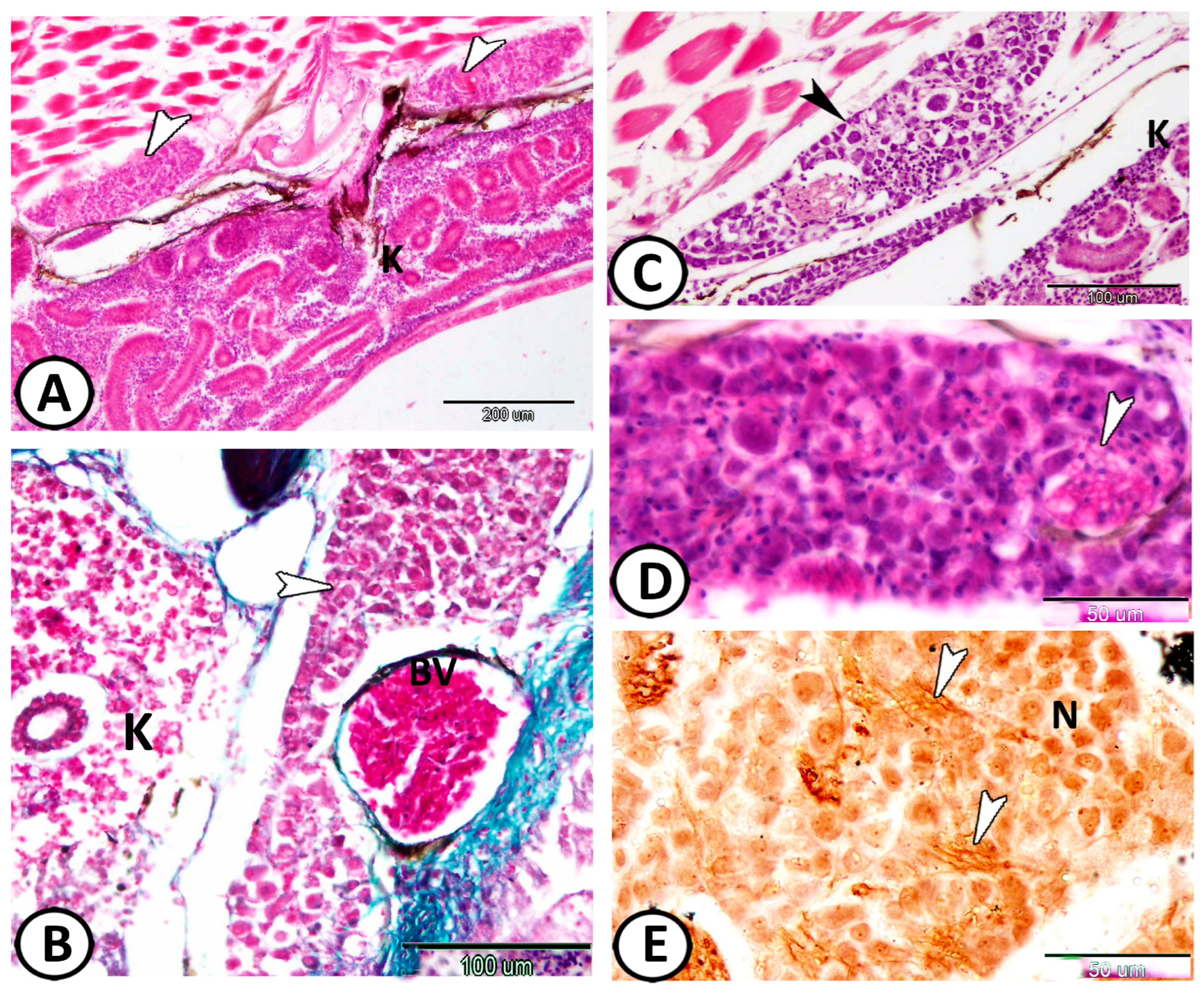
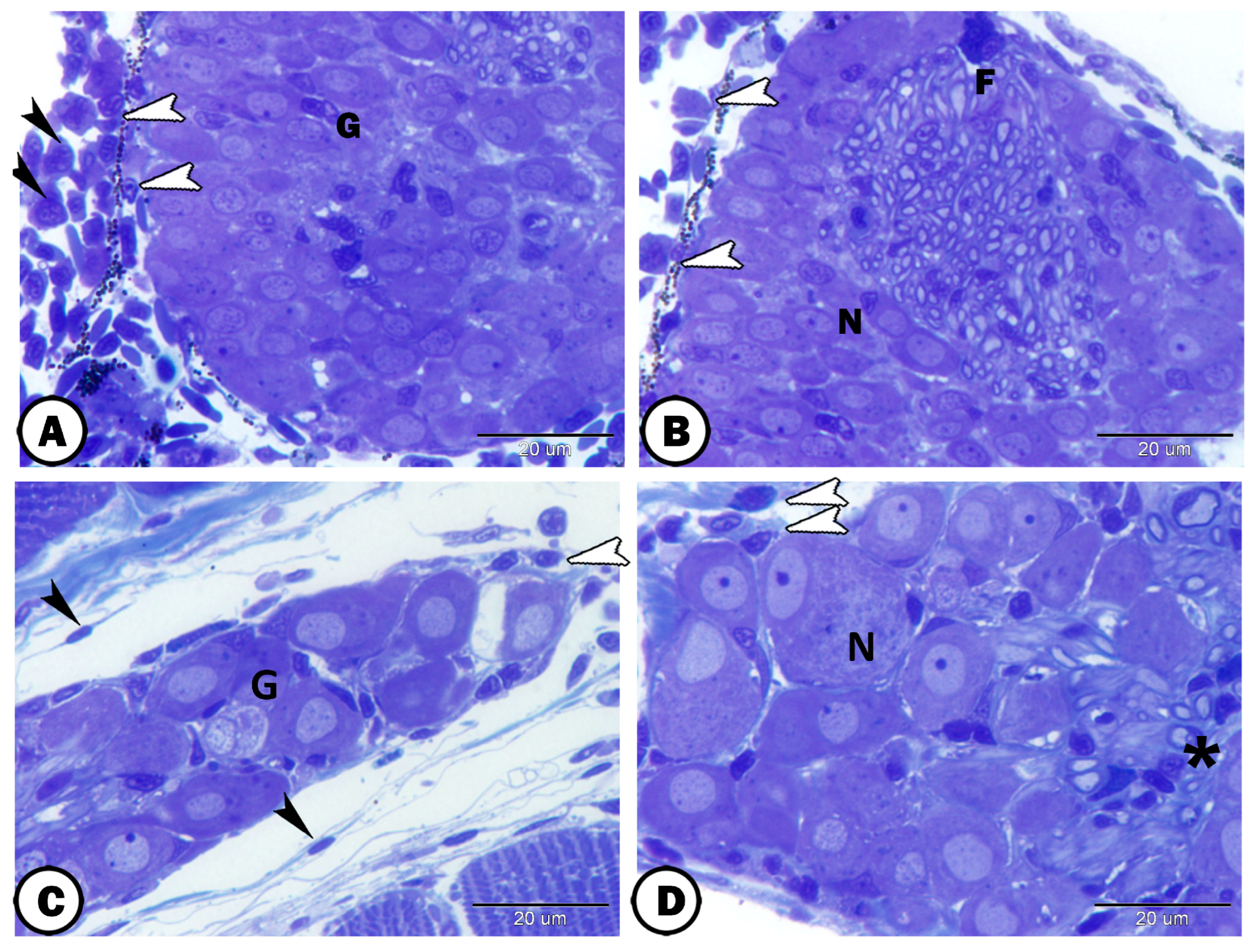
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Crane, J.F.; Trainor, P.A. Neural crest stem and progenitor cells. Annu. Rev. Cell Dev. Biol. 2006, 22, 267–286. [Google Scholar] [CrossRef] [PubMed]
- Bronner, M.E. Formation and migration of neural crest cells in the vertebrate embryo. Histochem. Cell Biol. 2012, 138, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Osório, J.; Rétaux, S. The lamprey in evolutionary studies. Dev. Genes Evol. 2008, 218, 221–235. [Google Scholar] [CrossRef] [PubMed]
- Jänig, W. Integrative Action of the Autonomic Nervous System: Neurobiology of Homeostasis; Cambridge University Press: Cambridge, UK, 2006. [Google Scholar] [CrossRef]
- Karemaker, J.M. An introduction into autonomic nervous function. Physiol. Meas. 2017, 38, R89–R118. [Google Scholar] [CrossRef]
- McCorry, L.K. Physiology of the autonomic nervous system. Am. J. Pharm. Educ. 2007, 71, 78. [Google Scholar] [CrossRef]
- Jänig, W.; Häbler, H.-J. Chapter 25—Specificity in the organization of the autonomic nervous system: A basis for precise neural regulation of homeostatic and protective body functions. In Progress in Brain Research; Mayer, E.A., Saper, C.B., Eds.; Elsevier: Amsterdam, The Netherlands, 2000; Volume 122, pp. 351–367. [Google Scholar]
- Furness, J.B.; Callaghan, B.P.; Rivera, L.R.; Cho, H.-J. The enteric nervous system and gastrointestinal innervation: Integrated local and central control. Microb. Endocrinol. Microbiota-Gut-Brain Axis Health Dis. 2014, 817, 39–71. [Google Scholar]
- Schneider, S.; Wright, C.M.; Heuckeroth, R.O. Unexpected roles for the second brain: Enteric nervous system as master regulator of bowel function. Annu. Rev. Physiol. 2019, 81, 235–259. [Google Scholar] [CrossRef]
- Aresti Sanz, J.; El Aidy, S. Microbiota and gut neuropeptides: A dual action of antimicrobial activity and neuroimmune response. Psychopharmacology 2019, 236, 1597–1609. [Google Scholar] [CrossRef]
- Bancroft, J.; Gamble, M. Theory and Practice of Histological Techniques, 5th ed; Churchill Livingstone Pub: Edinburgh, Scotland, 2002; Volume 172, pp. 593–620. [Google Scholar]
- Furness, J.B. The enteric nervous system: Normal functions and enteric neuropathies. Neurogastroenterol. Motil. 2008, 20 (Suppl. S1), 32–38. [Google Scholar] [CrossRef]
- Burns, A.J.; Pachnis, V. Development of the enteric nervous system: Bringing together cells, signals and genes. Neurogastroenterol. Motil. 2009, 21, 100–102. [Google Scholar] [CrossRef]
- Wallace, A.S.; Burns, A.J. Development of the enteric nervous system, smooth muscle and interstitial cells of Cajal in the human gastrointestinal tract. Cell Tissue Res. 2005, 319, 367–382. [Google Scholar] [CrossRef]
- Wallace, K.N.; Akhter, S.; Smith, E.M.; Lorent, K.; Pack, M. Intestinal growth and differentiation in zebrafish. Mech. Dev. 2005, 122, 157–173. [Google Scholar] [CrossRef]
- Olsson, C. Autonomic innervation of the fish gut. Acta Histochem. 2009, 111, 185–195. [Google Scholar] [CrossRef]
- Jessen, K.R.; Mirsky, R. Glial cells in the enteric nervous system contain glial fibrillary acidic protein. Nature 1980, 286, 736–737. [Google Scholar] [CrossRef]
- Mokhtar, D.M. Fish Histology: From Cells to Organs; Apple Academic Press: Oakville, ON, Canada, 2021. [Google Scholar]
- Mokhtar, D.M.; Sayed, R.K.; Zaccone, G.; Albano, M.; Hussein, M.T. Ependymal and Neural Stem Cells of Adult Molly Fish (Poecilia sphenops, Valenciennes, 1846) Brain: Histomorphometry, Immunohistochemical, and Ultrastructural Studies. Cells 2022, 11, 2659. [Google Scholar] [CrossRef]
- Rühl, A.; Nasser, Y.; Sharkey, K. Enteric glia. Neurogastroenterol. Motil. 2004, 16, 44–49. [Google Scholar] [CrossRef]
- Hagström, C.; Olsson, C. Glial cells revealed by GFAP immunoreactivity in fish gut. Cell Tissue Res. 2010, 341, 73–81. [Google Scholar] [CrossRef]
- Baker, P.A.; Meyer, M.D.; Tsang, A.; Uribe, R.A. Immunohistochemical and ultrastructural analysis of the maturing larval zebrafish enteric nervous system reveals the formation of a neuropil pattern. Sci. Rep. 2019, 9, 6941. [Google Scholar] [CrossRef]
- Holmberg, A.; Olsson, C.; Holmgren, S. The effects of endogenous and exogenous nitric oxide on gut motility in zebrafish Danio rerio embryos and larvae. J. Exp. Biol. 2006, 209, 2472–2479. [Google Scholar] [CrossRef]
- Holmberg, A.; Schwerte, T.; Pelster, B.; Holmgren, S. Ontogeny of the gut motility control system in zebrafish Danio rerio embryos and larvae. J. Exp. Biol. 2004, 207, 4085–4094. [Google Scholar] [CrossRef]
- Olden, T.; Akhtar, T.; Beckman, S.A.; Wallace, K.N. Differentiation of the zebrafish enteric nervous system and intestinal smooth muscle. Genesis 2008, 46, 484–498. [Google Scholar] [CrossRef] [PubMed]
- Uyttebroek, L.; Shepherd, I.T.; Harrisson, F.; Hubens, G.; Blust, R.; Timmermans, J.P.; Van Nassauw, L. Neurochemical coding of enteric neurons in adult and embryonic zebrafish (Danio rerio). J. Comp. Neurol. 2010, 518, 4419–4438. [Google Scholar] [CrossRef] [PubMed]
- Hanani, M.; Spray, D.C. Emerging importance of satellite glia in nervous system function and dysfunction. Nat. Rev. Neurosci. 2020, 21, 485–498. [Google Scholar] [CrossRef] [PubMed]
- Jessen, K.R.; Mirsky, R. The origin and development of glial cells in peripheral nerves. Nat. Rev. Neurosci. 2005, 6, 671–682. [Google Scholar] [CrossRef]
- Monk, K.R.; Feltri, M.L.; Taveggia, C. New insights on Schwann cell development. Glia 2015, 63, 1376–1393. [Google Scholar] [CrossRef]
- Hanani, M. Satellite glial cells in sensory ganglia: From form to function. Brain Res. Rev. 2005, 48, 457–476. [Google Scholar] [CrossRef]
- Pannese, E. The structure of the perineuronal sheath of satellite glial cells (SGCs) in sensory ganglia. Neuron Glia Biol. 2010, 6, 3–10. [Google Scholar] [CrossRef]
- Avraham, O.; Feng, R.; Ewan, E.E.; Rustenhoven, J.; Zhao, G.; Cavalli, V. Profiling sensory neuron microenvironment after peripheral and central axon injury reveals key pathways for neural repair. eLife 2021, 10, e68457. [Google Scholar] [CrossRef]
- Enes, J.; Haburčák, M.; Sona, S.; Gerard, N.; Mitchell, A.C.; Fu, W.; Birren, S.J. Satellite glial cells modulate cholinergic transmission between sympathetic neurons. PLoS ONE 2020, 15, e0218643. [Google Scholar] [CrossRef]
- van Velzen, M.; Laman, J.D.; KleinJan, A.; Poot, A.; Osterhaus, A.D.; Verjans, G.M. Neuron-interacting satellite glial cells in human trigeminal ganglia have an APC phenotype. J. Immunol. 2009, 183, 2456–2461. [Google Scholar] [CrossRef]
- Wu, H.-H.; Bellmunt, E.; Scheib, J.L.; Venegas, V.; Burkert, C.; Reichardt, L.F.; Zhou, Z.; Farinas, I.; Carter, B.D. Glial precursors clear sensory neuron corpses during development via Jedi-1, an engulfment receptor. Nat. Neurosci. 2009, 12, 1534–1541. [Google Scholar] [CrossRef]
- Germanà, A.; Marino, F.; Guerrera, M.C.; Campo, S.; De Girolamo, P.; Montalbano, G.; Germanà, G.P.; Ochoa-Erena, F.J.; Ciriaco, E.; Vega, J. Expression and distribution of S100 protein in the nervous system of the adult zebrafish (Danio rerio). Microsc. Res. Tech. 2008, 71, 248–255. [Google Scholar] [CrossRef]
- Gonzalez-Martinez, T.; Perez-Pinera, P.; Díaz-Esnal, B.; Vega, J. S-100 proteins in the human peripheral nervous system. Microsc. Res. Tech. 2003, 60, 633–638. [Google Scholar] [CrossRef]
- Meeker, N.D.; Trede, N.S. Immunology and zebrafish: Spawning new models of human disease. Dev. Comp. Immunol. 2008, 32, 745–757. [Google Scholar] [CrossRef]
- Zhang, L.; Xie, W.; Zhang, J.; Shanahan, H.; Tonello, R.; Lee, S.H.; Strong, J.A.; Berta, T.; Zhang, J.M. Key role of CCR2-expressing macrophages in a mouse model of low back pain and radiculopathy. Brain Behav. Immun. 2021, 91, 556–567. [Google Scholar] [CrossRef]
- Herbomel, P.; Thisse, B.; Thisse, C. Zebrafish early macrophages colonize cephalic mesenchyme and developing brain, retina, and epidermis through a M-CSF receptor-dependent invasive process. Dev. Biol. 2001, 238, 274–288. [Google Scholar] [CrossRef]
- Esiri, M.M.; Reading, M.C. Macrophages, lymphocytes and major histocompatibility complex (HLA) class II antigens in adult human sensory and sympathetic ganglia. J. Neuroimmunol. 1989, 23, 187–193. [Google Scholar] [CrossRef]
- Madigan, C.A.; Cambier, C.; Kelly-Scumpia, K.M.; Scumpia, P.O.; Cheng, T.-Y.; Zailaa, J.; Bloom, B.R.; Moody, D.B.; Smale, S.T.; Sagasti, A. A macrophage response to Mycobacterium leprae phenolic glycolipid initiates nerve damage in leprosy. Cell 2017, 170, 973–985.e10. [Google Scholar] [CrossRef]
- Collin, M.; Ginhoux, F. Human dendritic cells. Semin. Cell Dev. Biol. 2019, 86, 1–2. [Google Scholar] [CrossRef]
- Lucinda, N.; Figueiredo, M.M.; Pessoa, N.L.; Santos, B.S.Á.d.S.; Lima, G.K.; Freitas, A.M.; Machado, A.M.V.; Kroon, E.G.; Antonelli, L.R.d.V.; Campos, M.A. Dendritic cells, macrophages, NK and CD8+ T lymphocytes play pivotal roles in controlling HSV-1 in the trigeminal ganglia by producing IL1-beta, iNOS and granzyme B. Virol. J. 2017, 14, 37. [Google Scholar] [CrossRef]
- Robertsen, B. The interferon system of teleost fish. Fish Shellfish Immunol. 2006, 20, 172–191. [Google Scholar] [CrossRef] [PubMed]
- Ouwendijk, W.J.D.; Getu, S.; Mahalingam, R.; Gilden, D.; Osterhaus, A.D.M.E.; Verjans, G.M.G.M. Characterization of the immune response in ganglia after primary simian varicella virus infection. J. NeuroVirol. 2016, 22, 376–388. [Google Scholar] [CrossRef] [PubMed]
- Gacek, R.R. The course and central termination of first order neurons supplying vestibular end organs in the cat. Acta Otorhinolaryngol. Suppl. 1969, 254, 1–66. [Google Scholar]
- Weston, J.K. Observations on the comparative anatomy of the VIIIth nerve complex. Acta Oto-Laryngol. 1939, 27, 457–498. [Google Scholar] [CrossRef]
- Ballantyne, J.; Engström, H. Morphology of the vestibular ganglion cells. J. Laryngol. Otol. 1969, 83, 19–42. [Google Scholar] [CrossRef]
- Bas, E.; Goncalves, S.; Adams, M.; Dinh, C.T.; Bas, J.M.; Van De Water, T.R.; Eshraghi, A.A. Spiral ganglion cells and macrophages initiate neuro-inflammation and scarring following cochlear implantation. Front. Cell. Neurosci. 2015, 9, 303. [Google Scholar] [CrossRef]
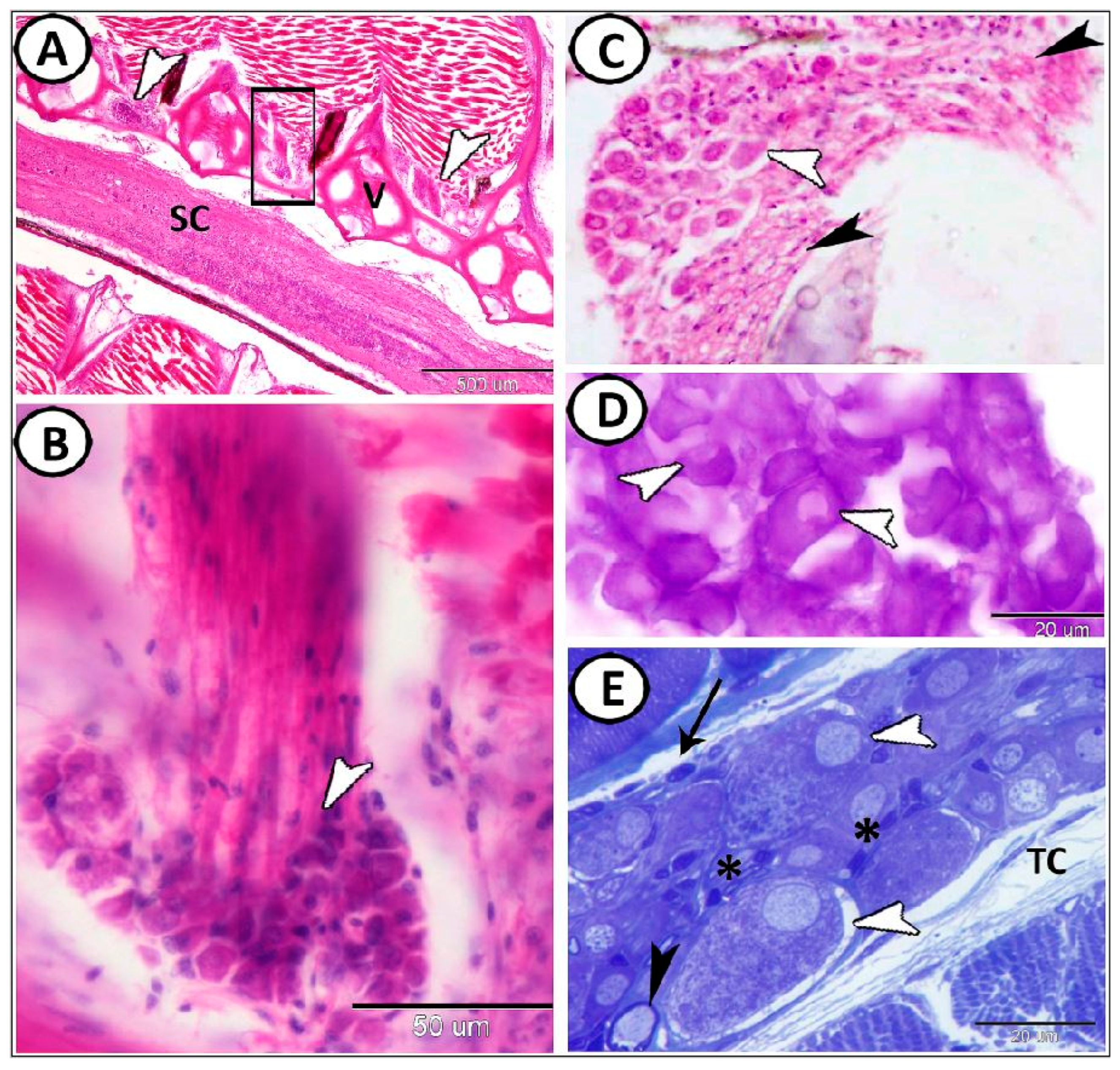
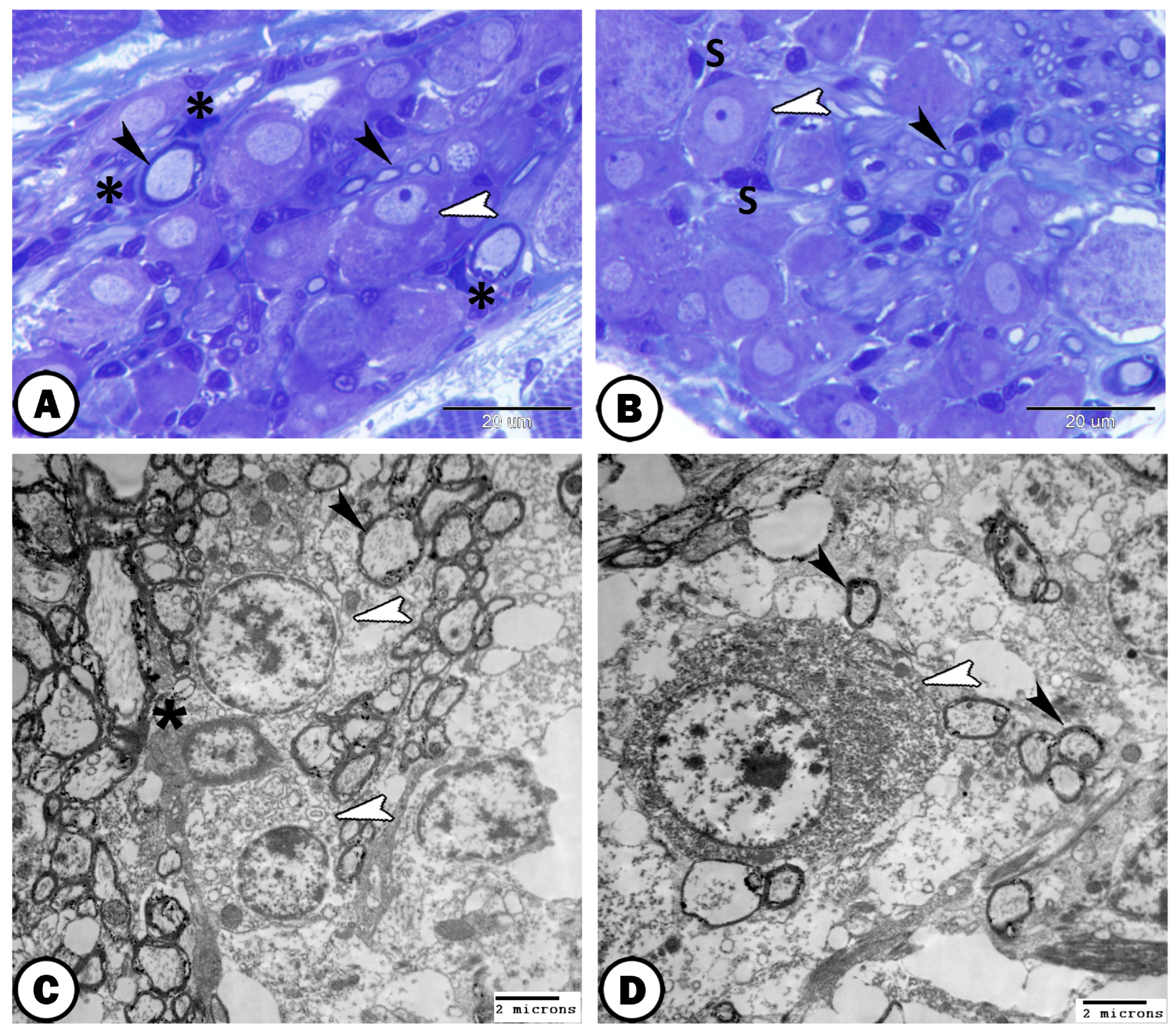

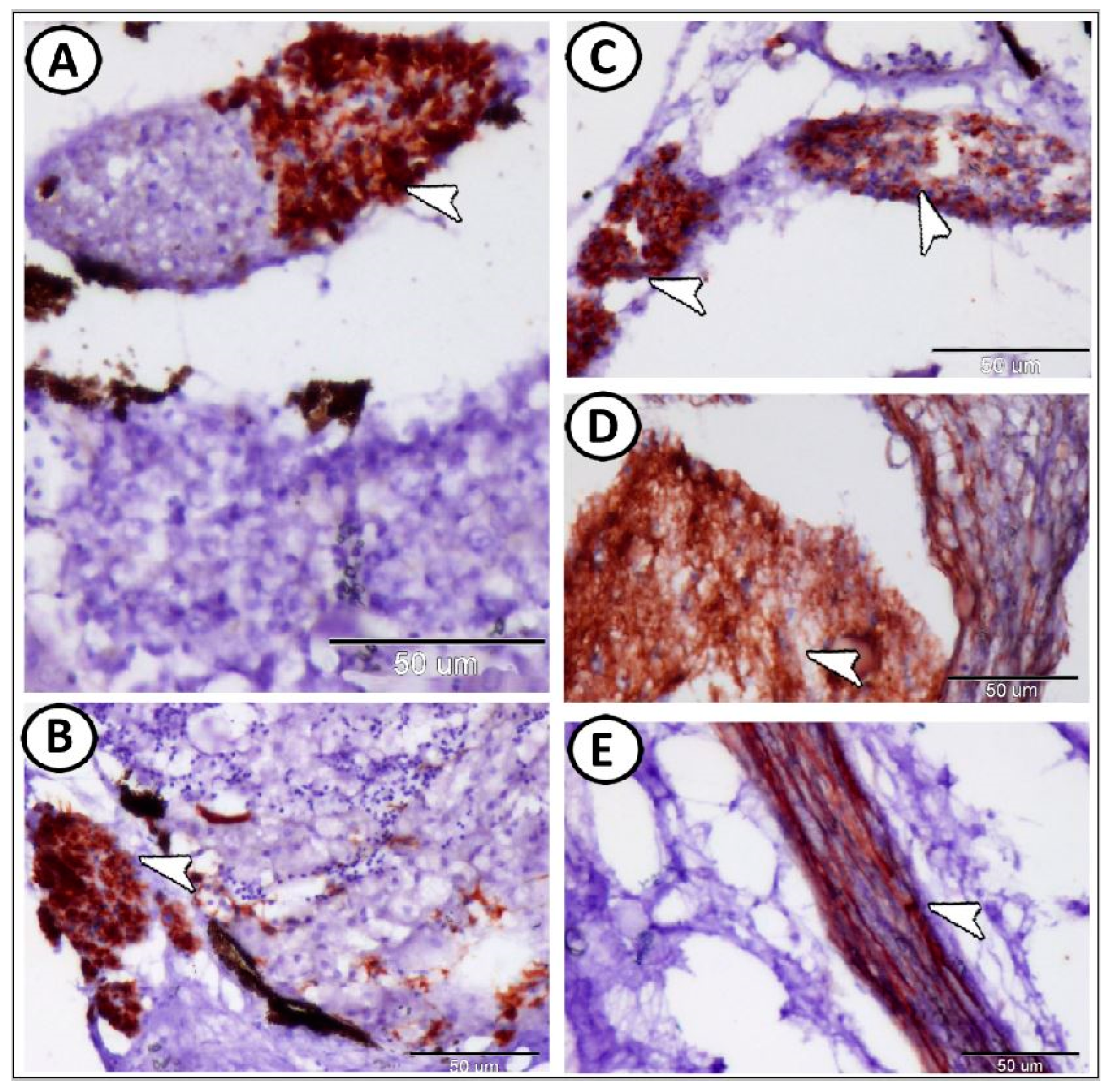
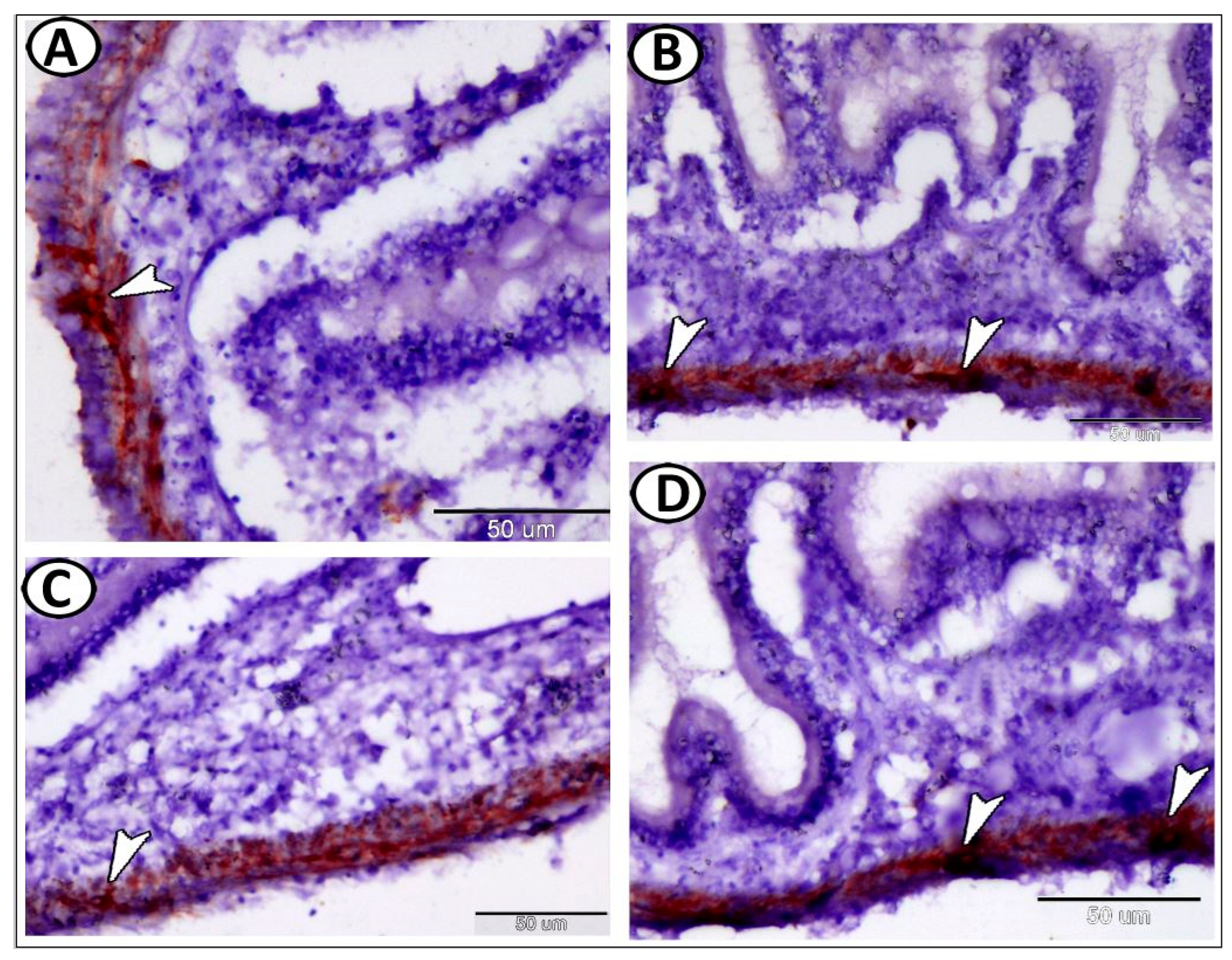



| Ganglia | Morphology | Markers | Position |
|---|---|---|---|
| Spinal G. | dorsal roots of the spinal nerves | ||
| neurons | large sizes, close together arranged in rows | some cells express GFAP | |
| round-oval cells bodies, large vesicular nucleus, distinct nucleolus, | most cells express S-100 | ||
| satellite cells | small nuclei of the flattened cells | GFAP | surround neurons |
| nerve fibers | myelinated and non-myelinated | GFAP | in the center of the ganglion |
| Schwann cells | ensheathed myelinated nerve fiber | closely related to many myelinated axons | |
| oligocyte | large vesicular round nucleus and light cytoplasm | closely related to many myelinated axons | |
| immune cells | close to the ganglion | ||
| telocytes | with distinct telopodes | close to the ganglion | |
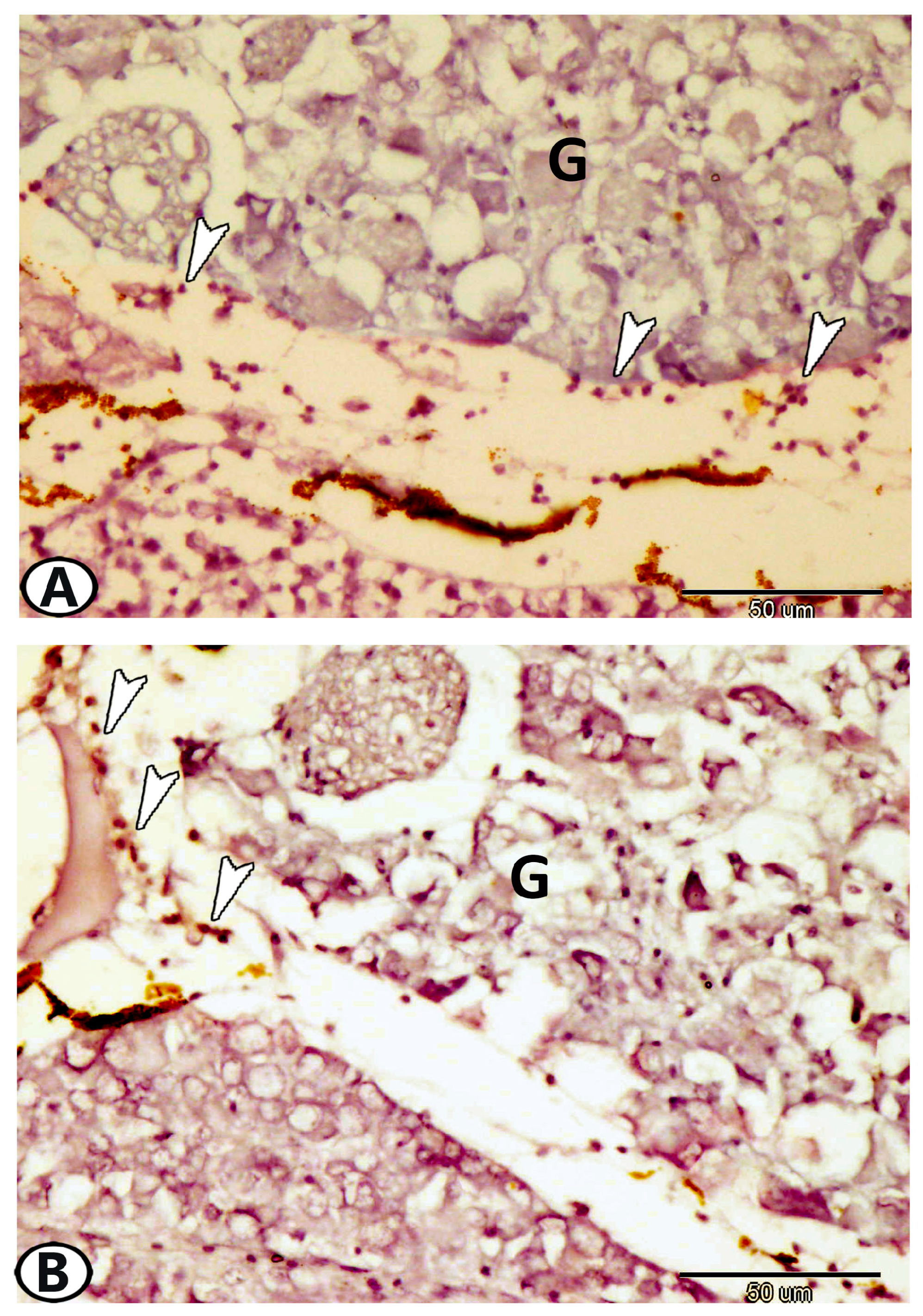
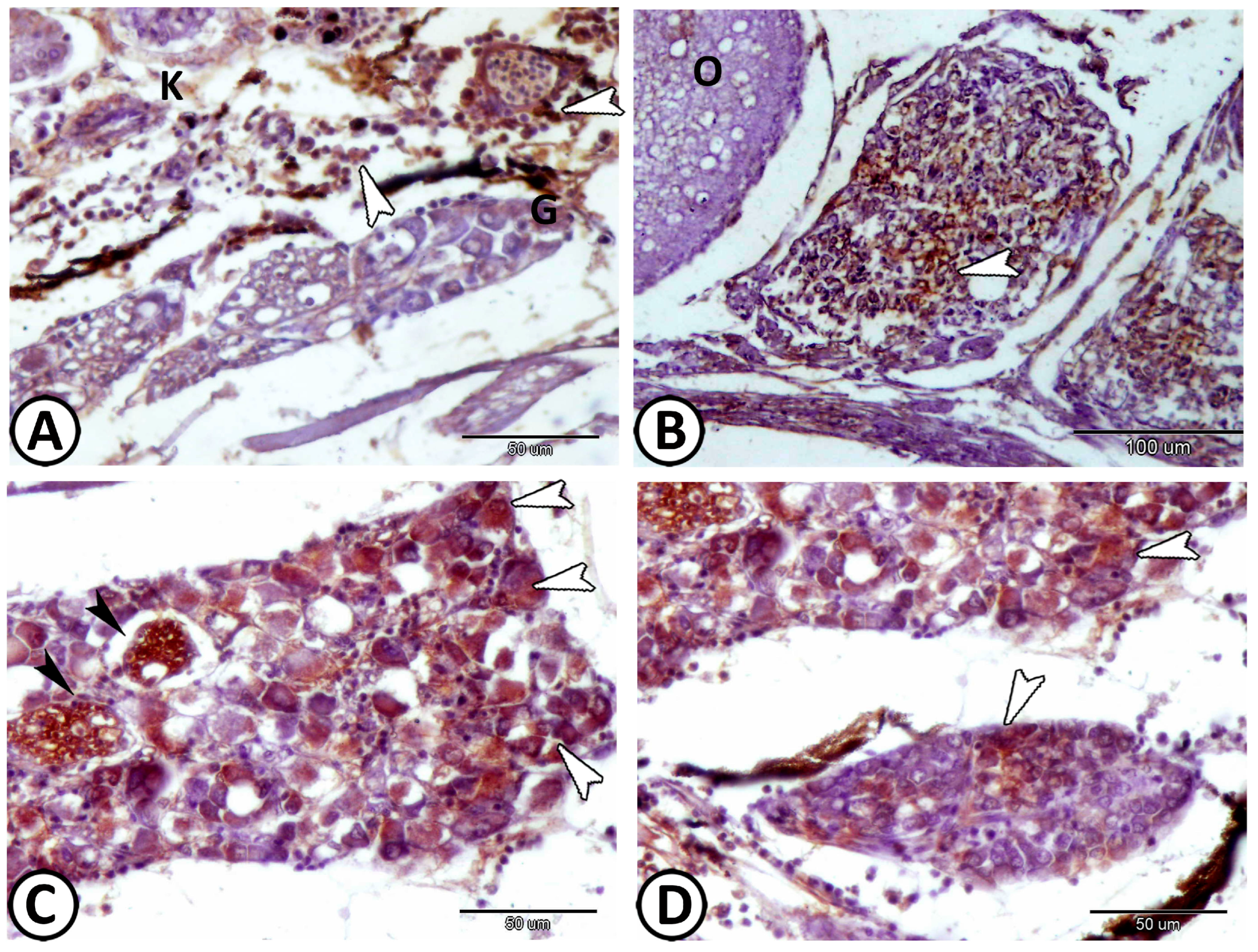
| Cervical G. | near gills, around pseudobranch, head kidney, and thymus | ||
| neurons | randomly distributed neurons | ||
| nerve fibers | peripherally situated nerve fibers | GFAP, S-100 | peripherally situated |
| immune cells | macrophages | Iba1 | around the ganglia near the head kidney |
| Glia cells | microglia | Iba1 | between neurons |
| Sympathetic G. | |||
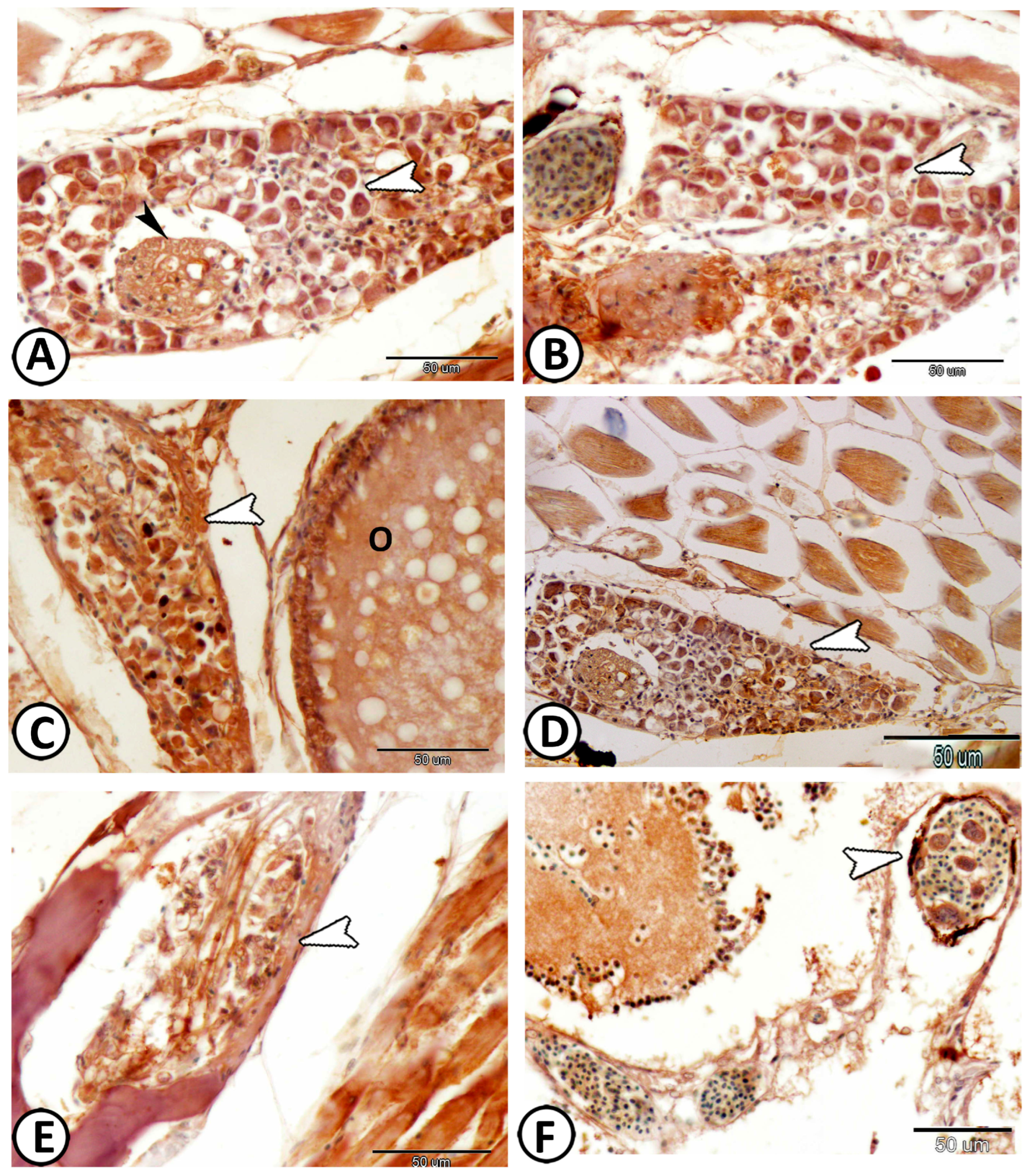
| neurons | cell bodies more widely spaced | Ach, S-100 | along the trunk kidney |
| nerve fibers | numerous axons and dendrites | S-100 | in between neurons |
| immune cells | include lymphocytes & macrophages | around the ganglia | |
| macrophages | CD68 immunoreactive cells | CD68 | near the kidney and ovary |
| lymphocytes | small sparsely distributed cells with high nucleus to cytoplasmic ratio | CD3 | along the trunk kidney |
| dendritic cells | like lymphocytes with dendrite-like processes | along the trunk kidney | |
| telocytes | with distinct telopodes | around the ganglia | |
| Enteric glia | |||
| neurons | smaller than other ganglionic cells | ACH | along the intestine muscular layer |
| glia | small cells, long cytoplasmic processes | GFAP | along the intestine muscular layer |
| macrophages | around glia cells in tunica muscularis | CD68 | the intestine |
| Vestibular G. | the largest ganglion | close to the hind brain | |
| neurons | arranged in many compact columns | S-100 | |
| ovoid or round vary in size | |||
| glial cells | a few glia cells could be identified | distributed between neurons | |
| nerve fibers | scanty within the vestibular ganglion | arranged in between the neurons |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mokhtar, D.M.; Attaai, A.; Zaccone, G.; Alesci, A.; Alonaizan, R.; Hussein, M.T. Morphological Distribution Patterns and Neuroimmune Communication of Ganglia in Molly Fish (Poecilia sphenops, Valenciennes 1846). Fishes 2023, 8, 289. https://doi.org/10.3390/fishes8060289
Mokhtar DM, Attaai A, Zaccone G, Alesci A, Alonaizan R, Hussein MT. Morphological Distribution Patterns and Neuroimmune Communication of Ganglia in Molly Fish (Poecilia sphenops, Valenciennes 1846). Fishes. 2023; 8(6):289. https://doi.org/10.3390/fishes8060289
Chicago/Turabian StyleMokhtar, Doaa M., Abdelraheim Attaai, Giacomo Zaccone, Alessio Alesci, Rasha Alonaizan, and Manal T. Hussein. 2023. "Morphological Distribution Patterns and Neuroimmune Communication of Ganglia in Molly Fish (Poecilia sphenops, Valenciennes 1846)" Fishes 8, no. 6: 289. https://doi.org/10.3390/fishes8060289
APA StyleMokhtar, D. M., Attaai, A., Zaccone, G., Alesci, A., Alonaizan, R., & Hussein, M. T. (2023). Morphological Distribution Patterns and Neuroimmune Communication of Ganglia in Molly Fish (Poecilia sphenops, Valenciennes 1846). Fishes, 8(6), 289. https://doi.org/10.3390/fishes8060289








